Abstract
Oral cryotherapy (OC) is a common preventive treatment of oral mucositis (OM) and is recommended in international guidelines. Ice and air OC have previously been shown to result in temperature reductions of 8.1–12.9 °C, and 14.5 °C, respectively, in healthy volunteers. However, no direct comparison between these two modalities has been performed. The primary aim was to investigate the tolerability and side effects of air OC using an intra-oral air-cooling (IOAC) device compared with ice OC. The secondary aim was to evaluate the temperature reduction in the mouth for the two respective methods. Cross-over study with randomization to order of treatment, in 15 healthy volunteers. We evaluated the self-reported intensity, frequency, and discomfort for 13 pre-defined side effects used in previous studies. All participants were able to complete both OC sessions, although one participant required reduced airflow in the air OC arm. The subjects reported more discomfort from being cold, having sensitive teeth, and numbness in the ice OC group, while they reported more discomfort from swallowing when subjected to air OC. No significant difference in the median temperature reduction was detected in the two modalities, except for the dorsal posterior part of the tongue where temperature reduction was larger in the ice OC group. We found that oral cooling using a new IOAC device was tolerated and seems to be safe in healthy volunteers.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Oral cryotherapy (OC) is a recommended preventive treatment of oral mucositis (OM) in international guidelines [1,2,3,4]. OC is usually achieved by letting ice chips, slushies, or popsicles melt in the mouth during chemotherapy, and is a cost-effective and safe treatment with few side effects [5].
OM is a common side effect of chemotherapy and radiotherapy in adults and children presenting as erythematous sores and ulcerations in the oral mucosa [3, 5,6,7]. OM causes pain, nutritional difficulties, and increases the risk for infections, and is considered to be one of the most debilitating side effect according to patients [9,10,11,12,13,14]. OM can be a dose-limiting factor resulting in chemotherapy dose reduction and increased healthcare costs [12, 15]. The incidence rate of OM is around 20–40% for patients receiving standard chemotherapy regimens, while for radiotherapy towards head and neck, it is close to 100%, and for patients receiving hematopoietic stem cell transplant (HSCT), it is close to 80% [6, 10, 16, 17]. Risk factors for developing OM vary with treatment regimens, genetic factors, age, and smoking, as well as previous episodes of OM [7, 16,17,18].
OC is suggested to prevent OM by local cooling causing vasoconstriction in the tissue, which leads to less chemotherapy exposure and a reduction of the metabolism of the basal epithelial cells resulting in a lower toxicity of the chemotherapeutic agent and, therefore, less tissue damage [8, 19, 20]. Treatment times of 30 to 60 min have been evaluated in randomized clinical trials [19, 21,22,23]. The reported temperature reduction of the oral mucosa was 8.1–12.9 °C using ice chips and 14.5 °C for air OC [24,25,26]. However, no direct comparison between these two modalities has been performed. To address this lack of data, we undertook a randomized cross-over study in healthy volunteers comparing the two methods.
The primary aim of this study was to investigate the tolerability and side effects of an intra-oral air-cooling (IOAC) device compared to OC with ice. The secondary aim was to evaluate the temperature reduction of the mouth for the two respective methods.
Subjects and methods
This was a cross-over study, randomized with regard to order of treatments. The study was approved by the Swedish Ethical Review Authority (Dnr 2021–01,485).
Participants
Measurements were performed on fifteen healthy volunteers, recruited by open advertisement at the Department of Anaesthesia and Intensive Care at Uppsala University Hospital, Uppsala, Sweden (n = 1) and a Facebook group (n = 14) which is dedicated mainly to medical students at Uppsala University. The study recruited more women (n = 9), and the mean age was 25.9 years (SD 4.9, range 20 to 40) (Table 1).
Methods
Tolerability was defined as the participant being able to maintain the OC protocol for > 90% of the total intended treatment time. Short interruptions were allowed in both study arms.
A questionnaire based on a modified numeric rating scale (NRS) rating the different side effects was created. Each side effect was quantified from intensity, frequency, and discomfort perspectives, respectively. Thus, zero represented no intensity at all, never experiencing the side effect or no discomfort in the first, second, and third categories, respectively. In analogy, the number 10 represented maximum intensity, continuous experience of the side effect, or maximum discomfort in the three respective categories. The choice of parameters was based on previous publications [25,26,27]. In order to make the grading more comprehensible, it was decided to categorize the grade of side effects as low (median score 0–4), moderate (median score > 4–7), and severe (median score > 7–10).
Temperature measurements were obtained using a FLIR® C digital thermal imaging camera. All participants were placed seated in a position 25 cm from the camera. The specifications of the thermal camera used for this study were as follows: minimal focus distance of 0.15 m, accuracy 0.01 °C, thermal sensitivity < 0.10 °C, and penetration of 4.5–6.5 mm (spectral range 7.5–14 µm) of the outer mucosal surface [28, 29]. The temperature differences on the output JPG images are visualized as color gradients based on a choosable palette. Using a rainbow color palette, the colors on the image ranges from white/yellow, representing the highest temperature, to black, representing the lowest temperature. Images were downloaded to a computer using the FLIR tools + software (Version 6.4.18031.1003) for data analysis (Fig. 1a).
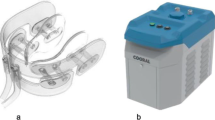
a Example of digital thermal images of the oral mucosa before and after the intervention. The left image with white/yellow color range areas represents the highest temperatures. The right image with black/purple color range represents the sites of lowest temperatures. b Schematic drawing of the IOAC device. RT room temperature
The air-cooling device
We used a novel IOAC that was described for the first time in a previous study [24]. It consists of a cooling unit, a cooling delivery system, and a mouthpiece (Fig. 1b). The average temperature of the cooled air was 5.1 °C, measured at the mouthpiece.
Experimental procedure
Before participating in the study, each participant had received written information about the study and was asked to schedule two separate appointments that would have to be at least one day apart. Each participant was asked not to consume tobacco, alcohol, caffeinated drinks, medications, or food 60 min prior to the study protocol. Informed consent was obtained at the first appointment after additional verbal information about the study had been provided. Basic demographic and health data were obtained from the participants to ensure that they fulfilled the inclusion criteria (age 18–65 years, speak, and understand Swedish or English fluently, and being healthy) and had none of the exclusion criteria (diseases requiring medical care or dental problems requiring care from a dentist). Afterwards, the volunteers were placed in a room for acclimatization with an average temperature of 23.7 °C for 30 min before initiating the experiment. At this point, the participants were randomized to initiate the experiment with either ice or air OC. Randomization was based on a simple coin toss.
Baseline images of the oral mucosa were obtained from the right and left buccal mucosa, upper and lower lips, anterior, posterior, and ventral aspects of the tongue and the hard palate. During the experiment, output data regarding temperature and flow were continuously collected from the IOAC device.
If the participant was randomized to air OC, the experiment was initiated with a five-minute test phase to test the IOAC, and in case, any adjustments were needed. Thereafter, the experiment continued through 60 min and a new set of images was collected of the above-mentioned areas.
If the participant was randomized to ice OC, the same procedure with baseline images was performed, and thereafter, a bucket of crushed ice was made available. Each participant was encouraged to keep the ice in the mouth for as long as they could (maximum 60 min). The participants were free to swallow or spit out the ice water. After this, a new set of images was collected of the above-mentioned areas. The average amount of ice consumed was 256 g, which was calculated by measuring the quantity of ice before initiating and after completing the experiment. After completing the experiment, the participants were asked to answer a questionnaire.
During the participant’s second session, they would repeat the same procedure but with the other type of OC, and the same questionnaire was given after they completed the experiment. After approximately one month, the participants were asked if they would consider participating in the experiment again, which OC treatment type they preferred, and in a hypothetical scenario of OC treatment for four hours (assuming equal effectiveness), which type they would prefer.
Statistics
No formal power analysis was performed since it was not an effect study, but rather a proof-of-concept study. The IBM SPSS Statistics (version 28.0.1.0 (142)) was used for statistical analysis. Shapiro–Wilk’s test (p > 0.05), visual inspection of histograms, and normal Q-Q plots indicated that most temperature reduction variables were approximately normally distributed, but some were not. Due to the small study population and mixed normality test results, it was decided to treat all data as non-normally distributed and a non-parametric Wilcoxon-Signed Ranks Test was used. Significance level was set to p < 0.01 due to multiple testing and relatively few observations.
Results
All participants reported being healthy and had no dental conditions requiring dentist intervention. Nine self-reported not having sensitive teeth, 5 rarely had sensitive teeth, and 1 had sensitive teeth sometimes (Table 1).
Sample characteristics
All participants (n = 15) were able to complete both OC sessions, although one participant required reduced airflow in the air OC arm. The most common side effects for ice OC were feeling cold (n = 15), numbness (n = 14), or pain in the mouth (n = 11). The most common side effects for air OC were increased saliva production (n = 14), swallowing difficulties (n = 13), mouth dryness (n = 12), and feeling cold in the mouth (n = 12) (Table 2).
We evaluated self-reported scores for intensity, frequency, and discomfort (median [IQR]) for the various side effects. Each of the side effects was thereafter ranked according to grade.
We found higher intensity scores for ice OC compared to air OC: feeling cold in the mouth (6.0 [2.0] vs. 3.3 [3.3]), numbness (5.0 [3.5] vs. 2.0 [5.5]), and sensitive teeth (3.9 [2.0] vs. 1.0 [0.0]). In contrast, we found higher scores for hypersalivation (6.7 [1] vs. 6.7 [1.0]), dry mouth (6 [3.5] vs. 0.4 [2.1]), and difficulty swallowing (4 [4] vs. 4.0 [4.0]) for air OC (Fig. 2a & Table 3).
Regarding frequency, the higher score in the ice OC group was numbness (4.5 [2] vs. 4.0 [6.0]). In contrast, difficulty swallowing (9 [5] vs. 9.0 [5.0]), mouth dryness (7.5 [2.8] vs. 6.8 [3.3]), and increased saliva production (7 [2.7] vs. 3.1 [1.8]) were more frequently experienced in the air OC group (Fig. 2b & Table 3).
Finally, we found higher discomfort scores from being cold (4 [2] vs 1.5 [2.5]), sensitive teeth (3.9 [3] vs. 1 [0]), and numbness (3 [3] vs 1 [1]) in the ice OC compared to air OC. In contrast, the participants reported higher discomfort scores from difficulty in swallowing (3 [2] vs 2.5 [2.7]) when using air OC (Fig. 2c and Table 3).
One participant was not able to utilize air OC per protocol and needed reduced air flow levels to tolerate the treatment. For this reason, this participant was removed from subsequent analyses. After this, the median temperature reduction was 14.1 ± 4.3 °C and 12.0 ± 3.5 °C in the oral mucosa for ice and air OC, respectively. This difference was not significant; the only location in the mouth where we found a significant difference in temperature reduction was the dorsal posterior part of the tongue for ice OC 24.3 ± 7.1 °C compared to air OC 16.4 ± 4.2 °C (Table 4).
When participants were asked if they would consider participating in the experiment again, 13 responded that they would and 2 were indecisive. Six participants said they preferred air OC, four preferred ice OC, and one had equal preferences for both. Four individuals could not give a preference regarding preferred type of OC. In the hypothetical scenario of four hours of OC, six said that they would prefer air OC, four would prefer ice OC, one indicated equal preferences for both, and four were indecisive.
Discussion
All participants completed the study protocol; both ice and air OC met the pre-defined tolerability criterion. Tolerability was similar or better than what has been previously reported for ice OC [24,25,26]. The most common side effects for ice OC were feeling cold, numbness, or pain in the mouth, probably related to the cooling effect of ice. Similar results were reported by Walladbegi et al. [26]. Swallowing difficulties and poor fit seem to be common side effects of the IOAC device and could be related to device design [26, 27]. In addition, increased salivation and dry mouth seem to affect participants more often with the IOAC device than with ice OC. This is probably related to the air flow, which we have previously reported [24].
To the best of our knowledge, this is the first study to measure and compare the side effects of two different OC modalities in healthy individuals. The participants’ evaluation of the intensity of side effects for ice OC was of moderate to low grade (cold, numbness and sensitive teeth), and similarly, that for air OC was of moderate to low grade (hypersalivation, dry mouth and difficult swallowing), indicating an acceptable intensity level of the experienced side effect. In contrast, the frequency for some of these side effects was of low grade for ice OC (numbness), while severe to moderate grade for air OC (difficulty swallowing, dry mouth and hypersalivation). These differences in the frequency can be explained by the IOAC device being continuously in the month, thereby making for a more frequent perception of these side effects, while side effects of ice OC will remain only for as long as the ice slowly melts in the mouth. Reassuringly, when the participants were asked to evaluate the total discomfort of the various side effects, the result was low grade in both the ice OC (cold, sensitive teeth, and numbness) and air OC arms (difficulties swallowing).
With regard to the temperature reduction in the oral mucosa, the only significant result was observed on the dorsal part of the tongue, where ice OC resulted in a larger temperature reduction. The temperature reduction in the ice OC group in our study was larger than in previously reported studies, and our air OC temperature reduction was also similar or larger than the ice temperature reduction previously reported [25,26,27]. This indicates that air OC has the capacity to reduce the temperature in the oral mucosa to a degree that is clinically relevant. Larger temperature reductions accomplished by air OC have been demonstrated previously by our group [24]. This suggests that if the treatment is tolerable at the intended air flows, temperature reduction in the oral mucosa achieved by air OC is similar to that of ice OC.
The strengths of this study lie in the experimental nature of the cross-over trial design making each participant their own control in combination with the randomization of the order of treatments to eliminate order effect. We focused on collecting only a few data points, which were continuously monitored, and the study was performed in a controlled environment. The same person collected the raw data and evaluated the images for all subjects. This is on one hand a strength because it provides for a more consistent analysis, but on the other hand presents a limitation due to the risk of observer bias. We allowed for a minimum of one day wash-out to restore oral mucosa temperature and sensibility and to enable the participants to answer the questionnaires with some time passing in between. Furthermore, the follow-up questions regarding willingness to participate in a similar study again took place a month later, in order to allow the participants plenty of time for reflection, and to monitor the development of any late side effects.
A limitation of the study was that the power was low due to few participants and a large number of measurements; however, we considered the sample size to be adequate for a proof-of-concept study. Likewise, some of the participants indicated that they would take part in a similar study and more of them preferred air OC compared to ice OC in a hypothetical scenario of 4 h of treatment, but due to the small sample size, no firm conclusion can be made from this data. Another limitation is the use of healthy volunteers who are not at risk of developing OM, which might increase or decrease motivation to fulfill the intended treatment, resulting in worse or better tolerability than the target population would have demonstrated. However, for ethical reasons, it was necessary to start with a study in volunteers. Finally, it was for obvious reasons not feasible to blind the participants to the treatment modality.
Conclusion
We found that oral cooling using a new IOAC device was tolerated and seems to be safe in healthy volunteers. Further studies are needed to explore its efficacy for preventing oral mucositis in patients receiving chemotherapy.
References
Sung L, Robinson P, Treister N, Baggott T, Gibson P, Tissing W, et al. Guideline for the prevention of oral and oropharyngeal mucositis in children receiving treatment for cancer or undergoing haematopoietic stem cell transplantation. BMJ Support Palliat Care. 2017;7:7–16.
Patel P, Robinson P, Baggott C, Gibson P, Ljungman G, Massey N, Ottaviani G, Phillips R, Revon-Riviere G, Treister N, White M, Cabral S, Dupuis LL, Sung L. Guideline for the prevention of oral and oro-pharyngeal mucositis in pediatric cancer or hematopoietic stem cell transplant patients: 2020 update. Eur J Cancer. 2021;154:92–101.
Elad S, Cheng KKF, Lalla RV, Yarom N, Hong C, Logan RM, et al. MASCC/ISOO clinical practice guidelines for the management of mucositis secondary to cancer therapy. Cancer. 2020;126:4423–31.
Correa MEP, Cheng KKF, Chiang K, Kandwal A, Loprinzi CL, Mori T, et al. Systematic review of oral cryotherapy for the management of oral mucositis in cancer patients and clinical practice guidelines. Support Care Cancer. 2020;28:2449–56.
Riley P, Glenny A-M, Worthington HV, Littlewood A, Clarkson JE, McCabe MG. Interventions for preventing oral mucositis in patients with cancer receiving treatment: oral cryotherapy. Cochrane Oral Health Group, editor. Cochrane Database Syst Rev [Internet]. 2015 [cited 2020 Nov 9]; Available from: http://doi.wiley.com/https://doi.org/10.1002/14651858.CD011552.pub2
Sonis ST, Elting LS, Keefe D, Peterson DE, Schubert M, Hauer-Jensen M, et al. Perspectives on cancer therapy-induced mucosal injury: pathogenesis, measurement, epidemiology, and consequences for patients. Cancer. 2004;100:1995–2025.
Cheng KKF, Lee V, Li CH, Goggins W, Thompson DR, Yuen HL, et al. Incidence and risk factors of oral mucositis in paediatric and adolescent patients undergoing chemotherapy. Oral Oncol. 2011;47:153–62.
Treister N, Sonis S. Mucositis: biology and management. Curr Opin Otolaryngol Head Neck Surg. 2007;15:123–9.
Ljungman G, Gordh T, Sörensen S, Kreuger A. Pain variations during cancer treatment in children: a descriptive survey. Pediatr Hematol Oncol. 2000;17:211–21.
Vagliano L, Feraut C, Gobetto G, Trunfio A, Errico A, Campani V, et al. Incidence and severity of oral mucositis in patients undergoing haematopoietic SCT—results of a multicentre study. Bone Marrow Transplant. 2011;46:727–32.
Elting LS, Cooksley C, Chambers M, Cantor SB, Manzullo E, Rubenstein EB. The burdens of cancer therapy. Clinical and economic outcomes of chemotherapy-induced mucositis. Cancer. 2003;98:1531–9.
Sonis ST, Oster G, Fuchs H, Bellm L, Bradford WZ, Edelsberg J, et al. Oral mucositis and the clinical and economic outcomes of hematopoietic stem-cell transplantation. J Clin Oncol Off J Am Soc Clin Oncol. 2001;19:2201–5.
Borbasi S, Cameron K, Quested B, Olver I, To B, Evans D. More than a sore mouth: patients’ experience of oral mucositis. Oncol Nurs Forum. 2002;29:1051–7.
Kamsvåg-Magnusson T, Thorsell-Cederberg J, Svanberg A, von Essen L, Arvidson J, Mellgren K, et al. Parents and children’s perceptions of distress related to oral mucositis during haematopoietic stem cell transplantation. Acta Paediatr Oslo Nor. 1992;2014(103):630–6.
Berger K, Schopohl D, Bollig A, Strobach D, Rieger C, Rublee D, et al. Burden of oral mucositis: a systematic review and implications for future research. Oncol Res Treat. 2018;41:399–405.
Maria OM, Eliopoulos N, Muanza T. Radiation-Induced Oral Mucositis. Front. Oncol. 2017;7:89.
Jones JA, Avritscher EBC, Cooksley CD, Michelet M, Bekele BN, Elting LS. Epidemiology of treatment-associated mucosal injury after treatment with newer regimens for lymphoma, breast, lung, or colorectal cancer. Support Care Cancer. 2006;14:505–15.
Barasch A, Peterson DE. Risk factors for ulcerative oral mucositis in cancer patients: unanswered questions. Oral Oncol. 2003;39:91–100.
Mahood DJ, Dose AM, Loprinzi CL, Veeder MH, Athmann LM, Therneau TM, et al. Inhibition of fluorouracil-induced stomatitis by oral cryotherapy. J Clin Oncol Off J Am Soc Clin Oncol. 1991;9:449–52.
Lundgren-Eriksson L, Carlsson A, Eksborg S, Ryd W, Vesanen R, Hultborn R. Pharmacokinetics of doxorubicin and epirubicin in mice during chlorpromazine-induced hypothermia. Cancer Chemother Pharmacol. 1997;40:419–24.
Salvador P, Azusano C, Wang L, Howell D. A pilot randomized controlled trial of an oral care intervention to reduce mucositis severity in stem cell transplant patients. J Pain Symptom Manag. 2012;44:64–73.
Svanberg A, Birgegård G, Öhrn K. Oral cryotherapy reduces mucositis and opioid use after myeloablative therapy—a randomized controlled trial. Support Care Cancer. 2007;15:1155–61.
Lilleby K, Garcia P, Gooley T, McDonnnell P, Taber R, Holmberg L, et al. A prospective, randomized study of cryotherapy during administration of high-dose melphalan to decrease the severity and duration of oral mucositis in patients with multiple myeloma undergoing autologous peripheral blood stem cell transplantation. Bone Marrow Transplant. 2006;37:1031–5.
Blacker C, Kamsvåg T, Bejhed RS, Ljungman G. Primary evaluation of an air-cooling device to reduce oral mucositis: a pilot study in healthy volunteers. Med Oncol. 2020;37:110.
Svanberg A, Öhrn K, Broström H, Birgegård G. The effect of cryotherapy on oral mucosa: a study in healthy volunteers. Med Oncol. 2012;29:3587–91.
Walladbegi J, Gellerstedt M, Svanberg A, Jontell M. Innovative intraoral cooling device better tolerated and equally effective as ice cooling. Cancer Chemother Pharmacol. 2017;80:965–72.
Mahdi A, Stübner J, Bergling M, Jontell M, Walladbegi J. Can cryoprevention of oral mucositis be obtained at a higher temperature? Clin Oral Investig. 2021;25:4519–26.
FLIR C2 - Powerful, Compact Thermal Imaging System. 2014. https://flir.netx.net/file/asset/7595/original. Accessed 19 Sep 2022
Bashkatov AN, Genina EA, Kochubey VI, Tuchin VV. Optical properties of human skin, subcutaneous and mucous tissues in the wavelength range from 400 to 2000\hspace0.167emnm. J Phys D. 2005;38:2543–55.
Funding
Open access funding provided by Uppsala University. This study was funded by the Swedish Childhood Cancer Fund (MT2021-0004 and TJ2018-0046) and the Sjöberg Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Blacker, C., Bejhed, R.S., Frykholm, P. et al. Randomized cross-over study investigating the tolerability and side effects of an intra-oral air-cooling device compared to ice in healthy volunteers. Med Oncol 40, 58 (2023). https://doi.org/10.1007/s12032-022-01932-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-022-01932-4