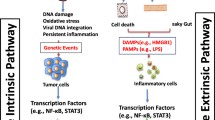
Abstract
Hepatocellular carcinoma is one of the major causes of cancer-related deaths worldwide and is associated with several inflammatory mediators, since 90% of HCCs occur based on chronic hepatitis B or C, alcoholism or increasingly metabolic syndrome-associated inflammation. EMT is a physiological process, with coordinated changes in epithelial gene signatures and is regulated by multiple factors, including cytokines and growth factors such as TGFβ, EGF, and FGF. Recent reports propose a strong association between EMT and inflammation, which is also correlated with tumor aggressiveness and poor outcomes. Cellular heterogeneity results collectively as an outcome of EMT, inflammation, and the tumor microenvironment, and it plays a fundamental role in the progression, complexity of cancer, and chemoresistance. In this review, we highlight recent developments concerning the association of EMT and inflammation in the context of HCC progression. Identifying potential EMT-related biomarkers and understanding EMT regulatory molecules will likely contribute to promising developments in clinical practice and will be a valuable tool for predicting metastasis in general and specifically in HCC.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Epithelial to mesenchymal transition or EMT is the switch from an epithelial, polarized state to the mesenchymal and unpolarized phenotype. It is a fundamental physiological process for healthy embryonic development; however, it is hijacked in certain pathological conditions such as cancer and inflammation [1,2,3]. There are 3 classes of EMT. Developmental (type I) EMT is required for mesoderm formation, neural crest delamination, the establishment of the heart valve, palatogenesis, and myogenesis [4,5,6]. Following tissue damage, EMT becomes activated during fibrosis and wound healing (type II) [7, 8]. However, it is also aberrantly activated during tumorigenesis when cancer cells start to disseminate, invade, and form metastasis (type III) [1, 2]. Mesenchymal cells can also acquire the epithelial state by undergoing a mesenchymal to epithelial transition (MET).
EMT-MET transitions cause cellular plasticity that is a feature of tumor initiation, progression, and metastatic colonization [9, 10]. The roles of EMT and MET in metastasis vary depending on the context. Tumor cells, which are phenotypically very heterogeneous, can benefit from EMT to gain some properties such as increased invasiveness and metastatic capacity, acquisition of stem-like features allowing them to adapt to the rapid shifts of tumor microenvironment resulting in more malignant tumors [11,12,13].
Plasticity and heterogeneity during EMT play pivotal roles in hepatocellular carcinoma (HCC) [14, 15]. HCC, the predominant form of primary liver cancer, is one of the major causes of cancer-related mortality worldwide and is associated with inflammatory mediators as more than 90% of HCCs occur due to inflammation [16,17,18].
Inflammation of the liver can result from chronic viral infections (Hepatitis B or C-HBV/HCV), which are among the major causes of HCC, as well as chronic alcohol consumption, aflatoxins, and drug derivatives [19, 20]. Due to the poor understanding of the inflammatory mechanisms underlying heterogeneous HCC development, the treatment options are very limited. Growing data show that inflammatory cytokines produced predominantly by macrophages and T lymphocytes lead to the activation of HCC carcinogenesis [21, 22]. For example, recent studies suggest that osteopontin (OPN), a matricellular cytokine that mediates various biological functions such as cell adhesion, migration, and survival is linked to HCC tumor progression and metastasis [23]. It can induce EMT via regulating vimentin expression in HCC cells [24]. In addition, in HCV-infected cells, cellular kinases such as p38 MAPK and JNK, which are involved in EMT regulation, are activated during OPN induction [25, 26].
EMT is an epigenetic reprogramming event accompanied by changes in the epithelial gene signature. This involves alterations in the expression of E-cadherin, which is the hallmark of EMT and is significantly associated with poor prognosis of most cancers, including HCC [1, 27, 28]. EMT is mediated by a group of transcription factors known as the EMT inducers, such as SNAI1/Slug, TWIST1/2, and ZEB1/2 [29, 30]. For instance, it has been shown that EMT inducers such as SNAI1 can regulate the expression and activation of several interleukins in different cancer types such as HCC, thus triggering an inflammatory response, which in some cases may result in the development of EMT-like features [12, 13, 31].
Inflammation is known as an adaptive response to infection, and some infections result in non-resolving inflammation due to host defense mechanisms, which are thought to contribute to the progression of various cancer types by promoting proliferative and survival signaling pathways, becoming a key component of the tumor microenvironment [18, 32]. EMT and inflammation are complex and highly controlled processes, and the functional connection between malignancy and inflammation has long been accepted. However, available data about the molecular mechanisms of these complex processes remain limited. Several recent studies addressed the relationship between EMT and inflammation in cancer progression. High levels of ZEB1 expression were recently shown to promote the malignant transformation of HCC due to activation of EMT, accompanied by reduced patient survival [33]. The ZEB1-attributed induction of EMT is usually associated with increased chemoresistance and eventually poor patient survival [34]. This is intriguing not only for the potential usage of ZEB1 inhibitors as potential treatment strategies, but also as a way for scoring HCC stages based on the predictive power of ZEB1 expression. ZEB1 has been shown to influence the inflammatory pathways in other cancer types [35,36,37], and together with its ability to induce pronounced EMT, leading to metastasis, puts it in a unique position as a prognostic factor and as a scoring criterion for HCC.
The long-established link between the metastatic capability of tumor cells, stemness, and inflammatory responses strengthens the connection between EMT and inflammatory mediators [10, 38]. Cancer cells that have undergone EMT have been shown to produce proinflammatory factors such as cytokines that contribute to tumor cell phenotypic changes and aggressiveness. Over the past decade, it became evident that a wide variety of inflammatory factors such as transforming growth factor beta (TGFβ), Wnt, several cytokines, and some long non-coding RNAs (lncRNAs) can stimulate EMT activation in cancer cells [12, 39, 40]. Moreover, metastatic progression and poor tumor prognosis are tightly correlated with EMT [12, 15, 41].
Signaling Pathways Involved in EMT and Carcinogenesis.
TGFβ, Wnt, Notch, NF-kβ, hedgehog, and receptor tyrosine kinases are essential signaling pathways inducing EMT during carcinogenesis and metastasis. These pathways are guided by critical mediators including Snai1/Slug, Smads, and Twist activated by signals from the tumor microenvironment [42,43,44].
TGFβ
The cytokine TGFβ is a potent inducer of EMT and associates with several critical signaling pathways, such as Wnt. The role of TGFβ in the stimulation of EMT and metastasis is intensively studied. Additionally, it is one of the crucial factors of cancer-related inflammation [13, 45]. TGFβ works as a tumor suppressor via regulating cell proliferation as well as a tumor promoter [46, 47]. However, there is limited knowledge available on which mechanisms are involved in mediating the process. TGFβ is well established as a potent inducer of EMT in HCC [48, 49].
TGFβ and hypoxia are dual factors that may induce EMT by increasing the stability of the EMT inducers in the inflammatory environment [20, 40, 50]. Moreover, it has been demonstrated that hypoxia affects the aggressiveness of cancer cells, illustrated by the enrichment of several gene sets correlated with poor prognosis and low survival [20, 51, 52]. For instance, it has been recently shown that HCC metastasis is promoted by the overexpression of HIF-1α, which triggers EMT-like features [12, 50, 53, 54]. Moreover, some ubiquitin ligases responsible for HIF-1α protein stabilization also have key functions in the progression of HCC. One of them is USP14, which is highly expressed in HCC, has a major function in the prognosis of the disease, and emerges as a potential therapeutic target [55]. Although hypoxia is known to be involved in the progression of HCC, because limited oxygenation microenvironment is associated with cancer progression, more research is needed to establish the link between the prognostic role of hypoxia and EMT in cancers.
In response to TGFβ, primary mouse hepatocytes lose their epithelial features due to a rapid decrease in Cdh1 expression [15]. The expression of Smad7, which is known to antagonize the TGFβ/Smad signaling pathway, confirms the reduction of the epithelial traits. Additionally, cirrhotic hepatocytes exposed to TGFβ differ from healthy mouse livers in morphology and gene signature pattern. The underlying mechanisms of hepatocarcinogenesis seem to be related to inflammation due to in part to increased TGFβ levels [15]. Furthermore, a recent study showed that hepatic TGFβ expression was increased after chronic alcohol consumption resulting in the activation of fibrosis and EMT [56].
The stabilization of the SMAD3/SMAD4 complex, which plays an integral role in TGFβ signaling, was recently reported to be mediated by the dynamic scaffolding protein β2-spectrin (β2SP). Overall, nuclear translocation of β2SP, which regulates the nuclear localization of β-catenin and induction of stemness genes, together with the inflammatory cytokine IL6, are related to HCC progression [57,58,59]. Additionally, sorafenib was shown to upregulate IL6 via the upregulation of STAT3 phosphorylation in hepatocytes [60, 61].
Notch
Notch is a highly conserved developmental signaling pathway required in many fundamental physiological processes, including proliferation, differentiation, apoptosis, homeostasis, and the regulation of the cell fate in several tissues [62,63,64]. The Notch pathway mediates harmonious equilibrium between cell proliferation and cell death; thus, it is fundamental for tumor progression. Notch can act as a tumor suppressor and oncogene, a dual feature defined by cellular context and signal dose or strength [65,66,67]. A dysfunctional Notch pathway is implicated in many cancers, including HCC [63]. Activation of the pathway leads to liver fibrosis and is mostly associated with poor prognosis in HCC [68]. The Notch coactivator MAML1 was recently found to contribute to the aggressiveness of most cancers, including HCC [69,70,71]. Besides, GPR50 endorses HCC progression through Notch pathway by regulating ADAM17 [72]. Notch target genes, such as Myc, cooperate with many signaling pathways to induce EMT during carcinogenesis [73, 74]. TGFβ also induces Notch activity via Smad pathway and suppresses E-cadherin indirectly through Slug [75]. Notch is also known to induce EMT and metastasis through Snai1 expression [76].
NF-kβ
The nuclear factor kappa β (NF-kβ) is a major signaling pathway connected to EMT and inflammation. NF-kβ is a prototypical proinflammatory signaling pathway activated by various inflammatory stimuli such as TNF-α and interleukin-1 (IL-1) and has been associated with the metastatic capacity of tumor cells [40, 77]. The activation of EMT inducers such as Snail1/Slug, Twist, and Zeb1/2 is highly demanded to promote tumors’ metastatic potential in several types of cancer [40, 77]. It was revealed that stabilization of Snai1 through the NF-kβ signaling pathway is needed for inflammation-induced metastasis [78]. It is also known that some target genes of NF-kβ and STAT3 pathways are linked and result in more aggressive tumors, which could be explained by the stabilization of cancer stem cells in connection to the activation of STAT3 [18, 79]. CRIF1 plays a paramount role in HCC progression via induction of ROS/NF-kβ signaling cascade resulting in tumor aggressiveness [80].
Cytokines
Cytokines are soluble mediators which are highly associated with cancer-related inflammation, and are mainly divided into 4 types: chemokines, interferons, interleukins, and tumor necrosis factors [12, 81].
Chemokines
Chemokines are small, secreted proteins that bind to G protein-coupled receptors [82, 83]. There are two types of chemokines; inflammatory chemokines, which are activated by inflammation, and homeostatic chemokines, which are constitutively active. Chemokines play critical roles in several biological processes such as embryonic development, angiogenesis, wound healing, migration, inflammation, and immune system homeostasis [83, 84]. They are also tightly associated with carcinogenesis; hence, they can induce proliferation, tumorigenesis, metastasis, stemness, and EMT [85, 86]. CXCL8, CCL5, and CXCL1 are critical chemokines related to EMT. CXCL8 is a well-studied chemokine involved in the TGFβ stimulated EMT through binding to the chemokine receptor CXCR1 during EMT [87, 88]. Beyond EMT, TGFβ itself is a widely accepted immunosuppressive cytokine associated with critical functions, including angiogenesis, metastasis, and cell motility [89,90,91]. A recent study indicated that TGFβ has an immunosuppression effect on CD8+ T cells via CXCR3 blockage [90]. In liver cancer, chemokines are present in the HCC microenvironment, and some HCC cells express chemokine receptors such as CCR5 to regulate migration, invasion, and growth of the tumor [84, 92, 93].
Interferons
Interferons (IFNs) are a family of anti-inflammatory cytokines involved in many cellular and physiological functions, such as inducing intrinsic pathways in response to viral infection. There are 3 types of IFNs described so far: types I, II, and III, classified based on functional and structural differences [94, 95]. IFN-α has been recently described to be involved in inflammation via induction of IFN-stimulated genes. IFN-α signaling pathways are known to crosstalk with many cancer-associated pathways such as JAK-STAT [96, 97], Wnt/GSK-3 [98], and Ras/Raf/MEK/ERK [99]. IFN-α also contributes to metastasis through the upregulation of HIF genes. IFN-α induces the expression of HIF-1α, which then activates JAK/PI3K/PTEN/mTOR/AKT and Ras/p38/ MEK/ERK signaling pathways. It has been documented that HIF-1α then stimulates the expression of EMT genes which also contribute to the tumorigenic progression in various cancer cell lines [100]. IFN-α has recently been recognized as a promising therapeutic target in HCC [101]. Besides conflicting results of IFN treatment in HCC, IFN-α and sorafenib seem to have a dual effect on HCC progression and metastasis through tumor-associated macrophage (TAM) polarization [102].
Interleukins
Interleukin family members IL-1α, IL-1β, IL-6, and IL-8, are cytokines known to be involved in cancer-associated inflammation [102, 103]. IL-6 and TNF-α secreted by adipocytes are the keystone proinflammatory cytokines in the tumor microenvironment due to their contribution to many biological processes, including inflammation, EMT, and cancer [104,105,106]. IL-6 typically induces EMT via STAT3-induced SNAIL expression [107, 108]. IL-6 production in cancer patients elevates platelet counts (para-neoplastic thrombocytosis), which is an indicator of poor prognosis in several types of cancer, such as HCC and ovarian cancer [109, 110]. IL-1α plays a critical role in activating vascular endothelium, which causes infiltration of inflammatory cells in a tumor-promoting environment [111].
TNF-α
TNF-α is crucial both for hepatocyte apoptosis and liver proliferation and is a well-known factor promoting tumor cell proliferation and angiogenesis [112, 113]. Recent reports highlight its essential role as an inflammatory mediator that can induce EMT in synergetic collaboration with TGFβ [12, 103]. TNF-α induces Snai1 promoter activity in some cancer cells, such as colorectal cancer, and has a role in stabilizing Snai1 protein [40, 114]. During inflammation, TNF-α also induces apoptosis by stimulating inflammatory signaling pathways [115, 116].
The Inflammatory Microenvironment in HCC
The tumor microenvironment plays a significant role in cancer progression [20, 117]. It contains inflammatory cells such as macrophages, neutrophils, and dendritic cells, which secrete numerous types of cytokines, ECM remodeling factors, and growth factors [12, 118]. HCC is one of the leading inflammation-related cancer types. Tumor-associated macrophages, which have a central role in tumor progression show EMT-like features in several cancer types such as HCC. TAMs produce multiple factors such as HGF, EGF, IL-1β, IL-6, and TNF-α, which are capable of inducing EMT in a tumorigenic environment [119, 120]. TAMs are also crucial for various biological processes such as angiogenesis, extracellular matrix (ECM) remodeling, and metastasis [40, 121]. There is increasing evidence that the tumor microenvironment has both pro- or anti-inflammatory responses in a context-dependent manner and is highly affected by cell heterogeneity and plasticity [122, 123]. Cell plasticity allows cancer cells to escape from the tumor microenvironment, which also leads to tumor heterogeneity. Tumor heterogeneity is acknowledged as a primary cause behind the failure of current cancer therapies. However, reducing inflammation seems to be a promising solution for better therapeutic strategies.
Other Factors
Despite the recent developments regarding the inflammatory factors associated with EMT, several unknown factors link inflammation to EMT in cancer. miRNAs and long non-coding RNAs (lncRNAs) are among them [124]. miRNAs are single-stranded non-coding RNAs, which play crucial functions in the epigenetic regulation of gene expression at the post-transcriptional level. miRNAs have been indicated to have essential roles during inflammation and carcinogenesis via their tight association with transcription factors. For instance, miR-122 is a tissue-specific miRNA that has an anti-inflammatory role in the liver. It is well described that transcription factors such as hepatocyte nuclear factor (HNF)-1α, HNF-4α, and CCAAT/enhancer binding protein α (C/EBPα) can induce the expression of miR-122 [125,126,127].
LncRNAs are essential for the advancement and progress in various cancer models such as HCC. One example is lncRNA HULC, which is aberrantly upregulated in liver cancer [128]. It is significant since it promotes EMT via miR-200a-3p/ZEB1 signaling pathway, which is also correlated with the clinical stage in HCC [129].
Cellular heterogeneity of HCC is partially explained by the EMT program [15, 31, 130]. A recent study reported an inflammatory mediator called polymeric immunoglobulin receptor (pIgR) as a prognostic marker of HCC. It induces EMT via Smad2/3 and is critical for the stem cell properties and the TGFβ linked EMT, closely related to HCC progression and tumor heterogeneity [130,131,132]. Aberrant expression of pIgR was also correlated with poor survival from HCC [133]. More supporting evidence would eventually corroborate the involvement of inflammatory pathways in HCC heterogeneity.
Activation of hepatic stellate cells (HSCs) is an essential event for the progression of hepatocarcinogenesis. The proteoglycan Agrin utilizes (PDGF)-induced HSCs by providing a tumorigenic microenvironment. Agrin secreted by platelet-derived growth factor (PDGF)-induced HSCs contributes to proliferation, invasion, and the metastatic ability of HCC and HSCs can be inhibited by the multi-kinase inhibitor sorafenib; a drug that has been approved for HCC therapy [134]. Overall, Agrin seems to be a key target for the treatment of HCC because of its contribution to HCC development in liver cirrhosis patients [135, 136].
Recent advances in tumor metastasis research revealed that the absent in melanoma 2 (AIM2) protein is found in the center of the inflammation-carcinogenesis axis [137]. AIM2 promotes HCC metastasis since its silencing activates EMT via targeting FN1 (fibronectin 1), which, in turn, contributes to the regulation of Snai1, N-cadherin, and vimentin [138]. It is demonstrated that HBx (hepatitis B virus X protein) induced loss of AIM2 is correlated with EMT. Recently, several studies have revealed a strong association between EMT and inflammation, which is also correlated with the aggressiveness of the tumor and poor overall duration of survival [103].
Conclusions
Despite significant efforts in the past decade, there remains a gap to be filled to understand the underlying mechanisms controlling EMT in HCC. HCC progression is influenced by complex pathways of EMT and inflammation. Thus, the inflammatory tumor microenvironment and EMT are tightly linked in relation to several aspects of tumor development and tumor metastasis. Since multiple mechanisms underlying both EMT and inflammation have been identified, as listed in this review, these processes provide suitable targets for future tumor intervention strategies. Furthermore, several immune checkpoint inhibitors such as atezolizumab have also been approved, that target the balance between inflammation and immunity. Therefore, it is highly likely that identifying potential EMT-related biomarkers and understanding EMT regulatory molecules will contribute to clinical practice and be a valuable tool for predicting HCC aggression and metastasis. It is now becoming clear that inflammation can both affect cancer progression and treatment responses. However, further efforts are still needed to understand the connection between EMT and cancer stemness in HCC progression.
Data Availability
Not applicable.
References
Brabletz T. To differentiate or not–routes towards metastasis. Nat Rev Cancer. 2012;12(6):425–36. https://doi.org/10.1038/nrc3265.
Kalluri R, Weinberg RA. The basics of epithelial-mesenchymal transition. J Clin Invest. 2009;119(6):1420–8. https://doi.org/10.1172/JCI39104.
Kim DH, Xing T, Yang Z, Dudek R, Lu Q, et al. Epithelial mesenchymal transition in embryonic development, tissue repair and cancer: a comprehensive overview. J Clin Med. 2017;7(1). https://doi.org/10.3390/jcm7010001.
Theveneau E, Mayor R. Neural crest delamination and migration: from epithelium-to-mesenchyme transition to collective cell migration. Dev Biol. 2012;366(1):34–54. https://doi.org/10.1016/j.ydbio.2011.12.041.
Thiery JP, Acloque H, Huang RY, Nieto MA. Epithelial-mesenchymal transitions in development and disease. Cell. 2009;139(5):871–90. https://doi.org/10.1016/j.cell.2009.11.007.
Yokoyama S, Asahara H. The myogenic transcriptional network. Cell Mol Life Sci. 2011;68(11):1843–9. https://doi.org/10.1007/s00018-011-0629-2.
Haensel D, Dai X. Epithelial-to-mesenchymal transition in cutaneous wound healing: where we are and where we are heading. Dev Dyn. 2018;247(3):473–80. https://doi.org/10.1002/dvdy.24561.
Stone RC, Pastar I, Ojeh N, Chen V, Liu S, et al. Epithelial-mesenchymal transition in tissue repair and fibrosis. Cell Tissue Res. 2016;365(3):495–506. https://doi.org/10.1007/s00441-016-2464-0.
Bakir B, Chiarella AM, Pitarresi JR, Rustgi AK. EMT, MET, Plasticity, and Tumor Metastasis. Trends Cell Biol. 2020;30(10):764–76. https://doi.org/10.1016/j.tcb.2020.07.003.
Lu W, Kang Y. Epithelial-mesenchymal plasticity in cancer progression and metastasis. Dev Cell. 2019;49(3):361–74. https://doi.org/10.1016/j.devcel.2019.04.010.
Ribatti D, Tamma R, Annese T. Epithelial-mesenchymal transition in cancer: a historical overview. Transl Oncol. 2020;13(6): 100773. https://doi.org/10.1016/j.tranon.2020.100773.
Suarez-Carmona M, Lesage J, Cataldo D, Gilles C. EMT and inflammation: inseparable actors of cancer progression. Mol Oncol. 2017;11(7):805–23. https://doi.org/10.1002/1878-0261.12095.
Zhou C, Liu J, Tang Y, Liang X. Inflammation linking EMT and cancer stem cells. Oral Oncol. 2012;48(11):1068–75. https://doi.org/10.1016/j.oraloncology.2012.06.005.
Chan LK, Tsui YM, Ho DW, Ng IO. Cellular heterogeneity and plasticity in liver cancer. Semin Cancer Biol. 2021. https://doi.org/10.1016/j.semcancer.2021.02.015.
van Zijl F, Zulehner G, Petz M, Schneller D, Kornauth C, et al. Epithelial-mesenchymal transition in hepatocellular carcinoma. Future Oncol. 2009;5(8):1169–79. https://doi.org/10.2217/fon.09.91.
Bishayee A. The role of inflammation and liver cancer. Adv Exp Med Biol. 2014;816:401–35. https://doi.org/10.1007/978-3-0348-0837-8_16.
Refolo MG, Messa C, Guerra V, Carr BI, D'Alessandro R. Inflammatory mechanisms of HCC development. Cancers (Basel). 2020;12(3). https://doi.org/10.3390/cancers12030641.
Yu LX, Ling Y, Wang HY. Role of nonresolving inflammation in hepatocellular carcinoma development and progression. NPJ Precis Oncol. 2018;2(1):6. https://doi.org/10.1038/s41698-018-0048-z.
Kew MC. Aflatoxins as a cause of hepatocellular carcinoma. J Gastrointestin Liver Dis. 2013;22(3):305–10.
Severi T, van Malenstein H, Verslype C, van Pelt JF. Tumor initiation and progression in hepatocellular carcinoma: risk factors, classification, and therapeutic targets. Acta Pharmacol Sin. 2010;31(11):1409–20. https://doi.org/10.1038/aps.2010.142.
Budhu A, Wang XW. The role of cytokines in hepatocellular carcinoma. J Leukoc Biol. 2006;80(6):1197–213. https://doi.org/10.1189/jlb.0506297.
Kany S, Vollrath JT, Relja B. Cytokines in inflammatory disease. Int J Mol Sci. 2019;20(23). https://doi.org/10.3390/ijms20236008.
Ding K, Fan L, Chen S, Wang Y, Yu H, et al. Overexpression of osteopontin promotes resistance to cisplatin treatment in HCC. Oncol Rep. 2015;34(6):3297–303. https://doi.org/10.3892/or.2015.4306.
Dong Q, Zhu X, Dai C, Zhang X, Gao X, et al. Osteopontin promotes epithelial-mesenchymal transition of hepatocellular carcinoma through regulating vimentin. Oncotarget. 2016;7(11):12997–3012. https://doi.org/10.18632/oncotarget.7016.
Cabiati M, Gaggini M, Cesare MM, Caselli C, De Simone P, et al. Osteopontin in hepatocellular carcinoma: a possible biomarker for diagnosis and follow-up. Cytokine. 2017;99:59–65. https://doi.org/10.1016/j.cyto.2017.07.004.
Shang S, Plymoth A, Ge S, Feng Z, Rosen HR, et al. Identification of osteopontin as a novel marker for early hepatocellular carcinoma. Hepatology. 2012;55(2):483–90. https://doi.org/10.1002/hep.24703.
Kim H, Park YN. Hepatocellular carcinomas expressing ‘stemness’-related markers: clinicopathological characteristics. Dig Dis. 2014;32(6):778–85. https://doi.org/10.1159/000368021.
Kong FF, Qu ZQ, Yuan HH, Wang JY, Zhao M, et al. Overexpression of FOXM1 is associated with EMT and is a predictor of poor prognosis in non-small cell lung cancer. Oncol Rep. 2014;31(6):2660–8. https://doi.org/10.3892/or.2014.3129.
Bolos V, Peinado H, Perez-Moreno MA, Fraga MF, Esteller M, et al. The transcription factor Slug represses E-cadherin expression and induces epithelial to mesenchymal transitions: a comparison with Snail and E47 repressors. J Cell Sci. 2003;116(Pt 3):499–511.
Lamouille S, Xu J, Derynck R. Molecular mechanisms of epithelial-mesenchymal transition. Nat Rev Mol Cell Biol. 2014;15(3):178–96. https://doi.org/10.1038/nrm3758.
Giannelli G, Koudelkova P, Dituri F, Mikulits W. Role of epithelial to mesenchymal transition in hepatocellular carcinoma. J Hepatol. 2016;65(4):798–808. https://doi.org/10.1016/j.jhep.2016.05.007.
Coussens LM, Werb Z. Inflammation and cancer. Nature. 2002;420(6917):860–7. https://doi.org/10.1038/nature01322.
Zhou YM, Cao L, Li B, Zhang RX, Sui CJ, et al. Clinicopathological significance of ZEB1 protein in patients with hepatocellular carcinoma. Ann Surg Oncol. 2012;19(5):1700–6. https://doi.org/10.1245/s10434-011-1772-6.
Sreekumar R, Emaduddin M, Al-Saihati H, Moutasim K, Chan J, et al. Protein kinase C inhibitors override ZEB1-induced chemoresistance in HCC. Cell Death Dis. 2019;10(10):703. https://doi.org/10.1038/s41419-019-1885-6.
Al-Ismaeel Q, Neal CP, Al-Mahmoodi H, Almutairi Z, Al-Shamarti I, et al. ZEB1 and IL-6/11-STAT3 signalling cooperate to define invasive potential of pancreatic cancer cells via differential regulation of the expression of S100 proteins. Br J Cancer. 2019;121(1):65–75. https://doi.org/10.1038/s41416-019-0483-9.
de Barrios O, Sanchez-Moral L, Cortes M, Ninfali C, Profitos-Peleja N, et al. ZEB1 promotes inflammation and progression towards inflammation-driven carcinoma through repression of the DNA repair glycosylase MPG in epithelial cells. Gut. 2019;68(12):2129–41. https://doi.org/10.1136/gutjnl-2018-317294.
Katsura A, Tamura Y, Hokari S, Harada M, Morikawa M, et al. ZEB1-regulated inflammatory phenotype in breast cancer cells. Mol Oncol. 2017;11(9):1241–62. https://doi.org/10.1002/1878-0261.12098.
Ricciardi M, Zanotto M, Malpeli G, Bassi G, Perbellini O, et al. Epithelial-to-mesenchymal transition (EMT) induced by inflammatory priming elicits mesenchymal stromal cell-like immune-modulatory properties in cancer cells. Br J Cancer. 2015;112(6):1067–75. https://doi.org/10.1038/bjc.2015.29.
Gugnoni M, Ciarrocchi A. Long noncoding RNA and epithelial mesenchymal transition in cancer. Int J Mol Sci. 2019;20(8). https://doi.org/10.3390/ijms20081924.
Lopez-Novoa JM, Nieto MA. Inflammation and EMT: an alliance towards organ fibrosis and cancer progression. EMBO Mol Med. 2009;1(6–7):303–14. https://doi.org/10.1002/emmm.200900043.
Aruga N, Kijima H, Masuda R, Onozawa H, Yoshizawa T, et al. Epithelial-mesenchymal transition (EMT) is correlated with patient’s prognosis of lung squamous cell carcinoma. Tokai J Exp Clin Med. 2018;43(1):5–13.
Deshmukh AP, Vasaikar SV, Tomczak K, Tripathi S, den Hollander P, et al. Identification of EMT signaling cross-talk and gene regulatory networks by single-cell RNA sequencing. Proc Natl Acad Sci USA. 2021;118(19). https://doi.org/10.1073/pnas.2102050118.
Gonzalez DM, Medici D. Signaling mechanisms of the epithelial-mesenchymal transition. Sci Signal. 2014;7(344):re8. https://doi.org/10.1126/scisignal.2005189.
Moustakas A, Heldin CH. Signaling networks guiding epithelial-mesenchymal transitions during embryogenesis and cancer progression. Cancer Sci. 2007;98(10):1512–20. https://doi.org/10.1111/j.1349-7006.2007.00550.x.
Fuxe J, Karlsson MC. TGF-beta-induced epithelial-mesenchymal transition: a link between cancer and inflammation. Semin Cancer Biol. 2012;22(5–6):455–61. https://doi.org/10.1016/j.semcancer.2012.05.004.
Kubiczkova L, Sedlarikova L, Hajek R, Sevcikova S. TGF-beta - an excellent servant but a bad master. J Transl Med. 2012;10:183. https://doi.org/10.1186/1479-5876-10-183.
Yang L, Moses HL. Transforming growth factor beta: tumor suppressor or promoter? Are host immune cells the answer? Cancer Res. 2008;68(22):9107–11. https://doi.org/10.1158/0008-5472.CAN-08-2556.
Dituri F, Mancarella S, Cigliano A, Chieti A, Giannelli G. TGF-beta as multifaceted orchestrator in HCC progression: signaling, EMT, immune microenvironment, and novel therapeutic perspectives. Semin Liver Dis. 2019;39(1):53–69. https://doi.org/10.1055/s-0038-1676121.
Su Q, Fan M, Wang J, Ullah A, Ghauri MA, et al. Sanguinarine inhibits epithelial-mesenchymal transition via targeting HIF-1alpha/TGF-beta feed-forward loop in hepatocellular carcinoma. Cell Death Dis. 2019;10(12):939. https://doi.org/10.1038/s41419-019-2173-1.
Zhang J, Zhang Q, Lou Y, Fu Q, Chen Q, et al. Hypoxia-inducible factor-1alpha/interleukin-1beta signaling enhances hepatoma epithelial-mesenchymal transition through macrophages in a hypoxic-inflammatory microenvironment. Hepatology. 2018;67(5):1872–89. https://doi.org/10.1002/hep.29681.
Tam SY, Wu VWC, Law HKW. Hypoxia-induced epithelial-mesenchymal transition in cancers: HIF-1alpha and beyond. Front Oncol. 2020;10:486. https://doi.org/10.3389/fonc.2020.00486.
Yeo CD, Kang N, Choi SY, Kim BN, Park CK, et al. The role of hypoxia on the acquisition of epithelial-mesenchymal transition and cancer stemness: a possible link to epigenetic regulation. Korean J Intern Med. 2017;32(4):589–99. https://doi.org/10.3904/kjim.2016.302.
Kung-Chun Chiu D, Pui-Wah Tse A, Law CT, Ming-Jing Xu I, Lee D, et al. Hypoxia regulates the mitochondrial activity of hepatocellular carcinoma cells through HIF/HEY1/PINK1 pathway. Cell Death Dis. 2019;10(12):934. https://doi.org/10.1038/s41419-019-2155-3.
Liu Z, Tu K, Wang Y, Yao B, Li Q, et al. Hypoxia accelerates aggressiveness of hepatocellular carcinoma cells involving oxidative stress, epithelial-mesenchymal transition and non-canonical hedgehog signaling. Cell Physiol Biochem. 2017;44(5):1856–68. https://doi.org/10.1159/000485821.
Lv C, Wang S, Lin L, Wang C, Zeng K, et al. USP14 maintains HIF1-alpha stabilization via its deubiquitination activity in hepatocellular carcinoma. Cell Death Dis. 2021;12(9):803. https://doi.org/10.1038/s41419-021-04089-6.
Yan G, Wang X, Sun C, Zheng X, Wei H, et al. Chronic alcohol consumption promotes diethylnitrosamine-induced hepatocarcinogenesis via immune disturbances. Sci Rep. 2017;7(1):2567. https://doi.org/10.1038/s41598-017-02887-7.
Mitra A, Yan J, Xia X, Zhou S, Chen J, et al. IL6-mediated inflammatory loop reprograms normal to epithelial-mesenchymal transition(+) metastatic cancer stem cells in preneoplastic liver of transforming growth factor beta-deficient beta2-spectrin(+/-) mice. Hepatology. 2017;65(4):1222–36. https://doi.org/10.1002/hep.28951.
Bergmann J, Muller M, Baumann N, Reichert M, Heneweer C, et al. IL-6 trans-signaling is essential for the development of hepatocellular carcinoma in mice. Hepatology. 2017;65(1):89–103. https://doi.org/10.1002/hep.28874.
Chen Y, Meng L, Shang H, Dou Q, Lu Z, et al. Beta2 spectrin-mediated differentiation repressed the properties of liver cancer stem cells through beta-catenin. Cell Death Dis. 2018;9(4):424. https://doi.org/10.1038/s41419-018-0456-6.
Deng YR, Ma HD, Tsuneyama K, Yang W, Wang YH, et al. STAT3-mediated attenuation of CCl4-induced mouse liver fibrosis by the protein kinase inhibitor sorafenib. J Autoimmun. 2013;46:25–34. https://doi.org/10.1016/j.jaut.2013.07.008.
Li Y, Chen G, Han Z, Cheng H, Qiao L, et al. IL-6/STAT3 Signaling contributes to sorafenib resistance in hepatocellular carcinoma through targeting cancer stem cells. Onco Targets Ther. 2020;13:9721–30. https://doi.org/10.2147/OTT.S262089.
Wael H, Yoshida R, Kudoh S, Hasegawa K, Niimori-Kita K, et al. Notch1 signaling controls cell proliferation, apoptosis and differentiation in lung carcinoma. Lung Cancer. 2014;85(2):131–40. https://doi.org/10.1016/j.lungcan.2014.05.001.
Bray SJ. Notch signalling: a simple pathway becomes complex. Nat Rev Mol Cell Biol. 2006;7(9):678–89. https://doi.org/10.1038/nrm2009.
Bray SJ. Notch signalling in context. Nat Rev Mol Cell Biol. 2016;17(11):722–35. https://doi.org/10.1038/nrm.2016.94.
Allenspach EJ, Maillard I, Aster JC, Pear WS. Notch signaling in cancer. Cancer Biol Ther. 2002;1(5):466–76. https://doi.org/10.4161/cbt.1.5.159.
Aster JC, Pear WS, Blacklow SC. The varied roles of notch in cancer. Annu Rev Pathol. 2017;12:245–75. https://doi.org/10.1146/annurev-pathol-052016-100127.
Nowell CS, Radtke F. Notch as a tumour suppressor. Nat Rev Cancer. 2017;17(3):145–59. https://doi.org/10.1038/nrc.2016.145.
Zhu C, Ho YJ, Salomao MA, Dapito DH, Bartolome A, et al. Notch activity characterizes a common hepatocellular carcinoma subtype with unique molecular and clinicopathologic features. J Hepatol. 2021;74(3):613–26. https://doi.org/10.1016/j.jhep.2020.09.032.
Shen H, McElhinny AS, Cao Y, Gao P, Liu J, et al. The Notch coactivator, MAML1, functions as a novel coactivator for MEF2C-mediated transcription and is required for normal myogenesis. Genes Dev. 2006;20(6):675–88. https://doi.org/10.1101/gad.1383706.
Wang J, Zhang SM, Wu JM, Lu ZC, Yang JR, et al. Mastermind-like transcriptional coactivator 1 overexpression predicts poor prognosis in human with hepatocellular carcinoma. Ann Clin Lab Sci. 2016;46(5):502–7.
Zema S, Pelullo M, Nardozza F, Felli MP, Screpanti I, et al. A dynamic role of mastermind-like 1: a journey through the main (path)ways between development and cancer. Front Cell Dev Biol. 2020;8: 613557. https://doi.org/10.3389/fcell.2020.613557.
Saha SK, Choi HY, Yang GM, Biswas PK, Kim K, et al. GPR50 promotes hepatocellular carcinoma progression via the notch signaling pathway through direct interaction with ADAM17. Mol Ther Oncolytics. 2020;17:332–49. https://doi.org/10.1016/j.omto.2020.04.002.
Garg M. Epithelial-mesenchymal transition - activating transcription factors - multifunctional regulators in cancer. World J Stem Cells. 2013;5(4):188–95. https://doi.org/10.4252/wjsc.v5.i4.188.
Wang Z, Li Y, Kong D, Sarkar FH. The role of Notch signaling pathway in epithelial-mesenchymal transition (EMT) during development and tumor aggressiveness. Curr Drug Targets. 2010;11(6):745–51. https://doi.org/10.2174/138945010791170860.
Zavadil J, Cermak L, Soto-Nieves N, Bottinger EP. Integration of TGF-beta/Smad and Jagged1/Notch signalling in epithelial-to-mesenchymal transition. EMBO J. 2004;23(5):1155–65. https://doi.org/10.1038/sj.emboj.7600069.
Timmerman LA, Grego-Bessa J, Raya A, Bertran E, Perez-Pomares JM, et al. Notch promotes epithelial-mesenchymal transition during cardiac development and oncogenic transformation. Genes Dev. 2004;18(1):99–115. https://doi.org/10.1101/gad.276304.
Pires BR, Mencalha AL, Ferreira GM, de Souza WF, Morgado-Diaz JA, et al. NF-kappaB is involved in the regulation of EMT genes in breast cancer cells. PLoS ONE. 2017;12(1): e0169622. https://doi.org/10.1371/journal.pone.0169622.
Wu Y, Deng J, Rychahou PG, Qiu S, Evers BM, et al. Stabilization of snail by NF-kappaB is required for inflammation-induced cell migration and invasion. Cancer Cell. 2009;15(5):416–28. https://doi.org/10.1016/j.ccr.2009.03.016.
Abbott DW, Laszczak M, Lewis JD, Su H, Moore SC, et al. Structural characterization of macroH2A containing chromatin. Biochemistry. 2004;43(5):1352–9. https://doi.org/10.1021/bi035859i.
Chang H, Li J, Qu K, Wan Y, Liu S, et al. CRIF1 overexpression facilitates tumor growth and metastasis through inducing ROS/NFkappaB pathway in hepatocellular carcinoma. Cell Death Dis. 2020;11(5):332. https://doi.org/10.1038/s41419-020-2528-7.
Jing Y, Han Z, Liu Y, Sun K, Zhang S, et al. Mesenchymal stem cells in inflammation microenvironment accelerates hepatocellular carcinoma metastasis by inducing epithelial-mesenchymal transition. PLoS ONE. 2012;7(8): e43272. https://doi.org/10.1371/journal.pone.0043272.
Turner MD, Nedjai B, Hurst T, Pennington DJ. Cytokines and chemokines: at the crossroads of cell signalling and inflammatory disease. Biochim Biophys Acta. 2014;1843(11):2563–82. https://doi.org/10.1016/j.bbamcr.2014.05.014.
Hughes CE, Nibbs RJB. A guide to chemokines and their receptors. FEBS J. 2018;285(16):2944–71. https://doi.org/10.1111/febs.14466.
Huang F, Geng XP. Chemokines and hepatocellular carcinoma. World J Gastroenterol. 2010;16(15):1832–6. https://doi.org/10.3748/wjg.v16.i15.1832.
Sarvaiya PJ, Guo D, Ulasov I, Gabikian P, Lesniak MS. Chemokines in tumor progression and metastasis. Oncotarget. 2013;4(12):2171–85. https://doi.org/10.18632/oncotarget.1426.
Nagarsheth N, Wicha MS, Zou W. Chemokines in the cancer microenvironment and their relevance in cancer immunotherapy. Nat Rev Immunol. 2017;17(9):559–72. https://doi.org/10.1038/nri.2017.49.
Liang CM, Chen L, Hu H, Ma HY, Gao LL, et al. Chemokines and their receptors play important roles in the development of hepatocellular carcinoma. World J Hepatol. 2015;7(10):1390–402. https://doi.org/10.4254/wjh.v7.i10.1390.
Liu Q, Li A, Tian Y, Wu JD, Liu Y, et al. The CXCL8-CXCR1/2 pathways in cancer. Cytokine Growth Factor Rev. 2016;31:61–71. https://doi.org/10.1016/j.cytogfr.2016.08.002.
Batlle R, Andres E, Gonzalez L, Llonch E, Igea A, et al. Regulation of tumor angiogenesis and mesenchymal-endothelial transition by p38alpha through TGF-beta and JNK signaling. Nat Commun. 2019;10(1):3071. https://doi.org/10.1038/s41467-019-10946-y.
Gunderson AJ, Yamazaki T, McCarty K, Fox N, Phillips M, et al. TGFbeta suppresses CD8(+) T cell expression of CXCR3 and tumor trafficking. Nat Commun. 2020;11(1):1749. https://doi.org/10.1038/s41467-020-15404-8.
Yang L. TGFbeta and cancer metastasis: an inflammation link. Cancer Metastasis Rev. 2010;29(2):263–71. https://doi.org/10.1007/s10555-010-9226-3.
Barashi N, Weiss ID, Wald O, Wald H, Beider K, et al. Inflammation-induced hepatocellular carcinoma is dependent on CCR5 in mice. Hepatology. 2013;58(3):1021–30. https://doi.org/10.1002/hep.26403.
Singh SK, Mishra MK, Rivers BM, Gordetsky JB, Bae S, et al. Biological and clinical significance of the CCR5/CCL5 axis in hepatocellular carcinoma. Cancers (Basel). 2020;12(4). https://doi.org/10.3390/cancers12040883.
Kopitar-Jerala N. The role of interferons in inflammation and inflammasome activation. Front Immunol. 2017;8:873. https://doi.org/10.3389/fimmu.2017.00873.
Walter MR. The role of structure in the biology of interferon signaling. Front Immunol. 2020;11: 606489. https://doi.org/10.3389/fimmu.2020.606489.
Rauch I, Muller M, Decker T. The regulation of inflammation by interferons and their STATs. JAKSTAT. 2013;2(1): e23820. https://doi.org/10.4161/jkst.23820.
Platanias LC. Mechanisms of type-I- and type-II-interferon-mediated signalling. Nat Rev Immunol. 2005;5(5):375–86. https://doi.org/10.1038/nri1604.
Wang H, Brown J, Garcia CA, Tang Y, Benakanakere MR, et al. The role of glycogen synthase kinase 3 in regulating IFN-beta-mediated IL-10 production. J Immunol. 2011;186(2):675–84. https://doi.org/10.4049/jimmunol.1001473.
Romerio F, Riva A, Zella D. Interferon-alpha2b reduces phosphorylation and activity of MEK and ERK through a Ras/Raf-independent mechanism. Br J Cancer. 2000;83(4):532–8. https://doi.org/10.1054/bjoc.2000.1263.
Yeh YH, Hsiao HF, Yeh YC, Chen TW, Li TK. Inflammatory interferon activates HIF-1alpha-mediated epithelial-to-mesenchymal transition via PI3K/AKT/mTOR pathway. J Exp Clin Cancer Res. 2018;37(1):70. https://doi.org/10.1186/s13046-018-0730-6.
Herzer K, Hofmann TG, Teufel A, Schimanski CC, Moehler M, et al. IFN-alpha-induced apoptosis in hepatocellular carcinoma involves promyelocytic leukemia protein and TRAIL independently of p53. Cancer Res. 2009;69(3):855–62. https://doi.org/10.1158/0008-5472.CAN-08-2831.
Zhang Z, Zhu Y, Xu D, Li TE, Li JH, et al. IFN-alpha facilitates the effect of sorafenib via shifting the M2-like polarization of TAM in hepatocellular carcinoma. Am J Transl Res. 2021;13(1):301–13.
Wang H, Liu J, Hu X, Liu S, He B. Prognostic and therapeutic values of tumor necrosis factor-alpha in hepatocellular carcinoma. Med Sci Monit. 2016;22:3694–704. https://doi.org/10.12659/msm.899773.
Xing Z, Gauldie J, Cox G, Baumann H, Jordana M, et al. IL-6 is an antiinflammatory cytokine required for controlling local or systemic acute inflammatory responses. J Clin Invest. 1998;101(2):311–20. https://doi.org/10.1172/JCI1368.
Popko K, Gorska E, Stelmaszczyk-Emmel A, Plywaczewski R, Stoklosa A, et al. Proinflammatory cytokines Il-6 and TNF-alpha and the development of inflammation in obese subjects. Eur J Med Res. 2010;15(Suppl 2):120–2. https://doi.org/10.1186/2047-783x-15-s2-120.
Sullivan NJ, Sasser AK, Axel AE, Vesuna F, Raman V, et al. Interleukin-6 induces an epithelial-mesenchymal transition phenotype in human breast cancer cells. Oncogene. 2009;28(33):2940–7. https://doi.org/10.1038/onc.2009.180.
Wendt MK, Balanis N, Carlin CR, Schiemann WP. STAT3 and epithelial-mesenchymal transitions in carcinomas. JAKSTAT. 2014;3(1): e28975. https://doi.org/10.4161/jkst.28975.
Gyamfi J, Lee YH, Eom M, Choi J. Interleukin-6/STAT3 signalling regulates adipocyte induced epithelial-mesenchymal transition in breast cancer cells. Sci Rep. 2018;8(1):8859. https://doi.org/10.1038/s41598-018-27184-9.
Lin RJ, Afshar-Kharghan V, Schafer AI. Paraneoplastic thrombocytosis: the secrets of tumor self-promotion. Blood. 2014;124(2):184–7. https://doi.org/10.1182/blood-2014-03-562538.
Porta C, De Amici M, Quaglini S, Paglino C, Tagliani F, et al. Circulating interleukin-6 as a tumor marker for hepatocellular carcinoma. Ann Oncol. 2008;19(2):353–8. https://doi.org/10.1093/annonc/mdm448.
Bevilacqua MP, Pober JS, Wheeler ME, Cotran RS, Gimbrone MA, Jr. Interleukin-1 activation of vascular endothelium. Effects on procoagulant activity and leukocyte adhesion. Am J Pathol. 1985;121(3):394–403.
Faletti L, Peintner L, Neumann S, Sandler S, Grabinger T, et al. TNFalpha sensitizes hepatocytes to FasL-induced apoptosis by NFkappaB-mediated Fas upregulation. Cell Death Dis. 2018;9(9):909. https://doi.org/10.1038/s41419-018-0935-9.
Wang Y, Xu J, Zhang X, Wang C, Huang Y, et al. TNF-alpha-induced LRG1 promotes angiogenesis and mesenchymal stem cell migration in the subchondral bone during osteoarthritis. Cell Death Dis. 2017;8(3): e2715. https://doi.org/10.1038/cddis.2017.129.
Wang H, Wang HS, Zhou BH, Li CL, Zhang F, et al. Epithelial-mesenchymal transition (EMT) induced by TNF-alpha requires AKT/GSK-3beta-mediated stabilization of snail in colorectal cancer. PLoS ONE. 2013;8(2): e56664. https://doi.org/10.1371/journal.pone.0056664.
Jang MK, Kim HS, Chung YH. Clinical aspects of tumor necrosis factor-alpha signaling in hepatocellular carcinoma. Curr Pharm Des. 2014;20(17):2799–808. https://doi.org/10.2174/13816128113199990587.
Page MJ, Bester J, Pretorius E. The inflammatory effects of TNF-alpha and complement component 3 on coagulation. Sci Rep. 2018;8(1):1812. https://doi.org/10.1038/s41598-018-20220-8.
Neophytou, C.M., M. Panagi, T. Stylianopoulos, and P. Papageorgis, The role of tumor microenvironment in cancer metastasis: molecular mechanisms and therapeutic opportunities. Cancers (Basel), 2021. 13(9). https://doi.org/10.3390/cancers13092053.
Kim J, Bae JS. Tumor-Associated macrophages and neutrophils in tumor microenvironment. Mediators Inflamm. 2016;2016:6058147. https://doi.org/10.1155/2016/6058147.
Shirabe K, Mano Y, Muto J, Matono R, Motomura T, et al. Role of tumor-associated macrophages in the progression of hepatocellular carcinoma. Surg Today. 2012;42(1):1–7. https://doi.org/10.1007/s00595-011-0058-8.
Li Z, Wu T, Zheng B, Chen L. Individualized precision treatment: targeting TAM in HCC. Cancer Lett. 2019;458:86–91. https://doi.org/10.1016/j.canlet.2019.05.019.
Capece D, Fischietti M, Verzella D, Gaggiano A, Cicciarelli G, et al. The inflammatory microenvironment in hepatocellular carcinoma: a pivotal role for tumor-associated macrophages. Biomed Res Int. 2013;2013: 187204. https://doi.org/10.1155/2013/187204.
Faurobert E, Bouin AP, Albiges-Rizo C. Microenvironment, tumor cell plasticity, and cancer. Curr Opin Oncol. 2015;27(1):64–70. https://doi.org/10.1097/CCO.0000000000000154.
Runa F, Hamalian S, Meade K, Shisgal P, Gray PC, et al. Tumor microenvironment heterogeneity: challenges and opportunities. Curr Mol Biol Rep. 2017;3(4):218–29. https://doi.org/10.1007/s40610-017-0073-7.
Cao MX, Jiang YP, Tang YL, Liang XH. The crosstalk between lncRNA and microRNA in cancer metastasis: orchestrating the epithelial-mesenchymal plasticity. Oncotarget. 2017;8(7):12472–83. https://doi.org/10.18632/oncotarget.13957.
Jin K, Li T, Sanchez-Duffhues G, Zhou F, Zhang L. Involvement of inflammation and its related microRNAs in hepatocellular carcinoma. Oncotarget. 2017;8(13):22145–65. https://doi.org/10.18632/oncotarget.13530.
Coulouarn C, Factor VM, Andersen JB, Durkin ME, Thorgeirsson SS. Loss of miR-122 expression in liver cancer correlates with suppression of the hepatic phenotype and gain of metastatic properties. Oncogene. 2009;28(40):3526–36. https://doi.org/10.1038/onc.2009.211.
Ha SY, Yu JI, Choi C, Kang SY, Joh JW, et al. Prognostic significance of miR-122 expression after curative resection in patients with hepatocellular carcinoma. Sci Rep. 2019;9(1):14738. https://doi.org/10.1038/s41598-019-50594-2.
Yu X, Zheng H, Chan MT, Wu WK. HULC: an oncogenic long non-coding RNA in human cancer. J Cell Mol Med. 2017;21(2):410–7. https://doi.org/10.1111/jcmm.12956.
Li SP, Xu HX, Yu Y, He JD, Wang Z, et al. LncRNA HULC enhances epithelial-mesenchymal transition to promote tumorigenesis and metastasis of hepatocellular carcinoma via the miR-200a-3p/ZEB1 signaling pathway. Oncotarget. 2016;7(27):42431–46. https://doi.org/10.18632/oncotarget.9883.
Sphyris N, Mani SA. pIgR: frenemy of inflammation, EMT, and HCC progression. J Natl Cancer Inst. 2011;103(22):1644–5. https://doi.org/10.1093/jnci/djr421.
Ai J, Tang Q, Wu Y, Xu Y, Feng T, et al. The role of polymeric immunoglobulin receptor in inflammation-induced tumor metastasis of human hepatocellular carcinoma. J Natl Cancer Inst. 2011;103(22):1696–712. https://doi.org/10.1093/jnci/djr360.
Critelli R, Milosa F, Faillaci F, Condello R, Turola E, et al. Microenvironment inflammatory infiltrate drives growth speed and outcome of hepatocellular carcinoma: a prospective clinical study. Cell Death Dis. 2017;8(8): e3017. https://doi.org/10.1038/cddis.2017.395.
Zhang Y, Zhang J, Chen X, Yang Z. Polymeric immunoglobulin receptor (PIGR) exerts oncogenic functions via activating ribosome pathway in hepatocellular carcinoma. Int J Med Sci. 2021;18(2):364–71. https://doi.org/10.7150/ijms.49790.
Ben Mousa A. Sorafenib in the treatment of advanced hepatocellular carcinoma. Saudi J Gastroenterol. 2008;14(1):40–2. https://doi.org/10.4103/1319-3767.37808.
Lv X, Fang C, Yin R, Qiao B, Shang R, et al. Agrin para-secreted by PDGF-activated human hepatic stellate cells promotes hepatocarcinogenesis in vitro and in vivo. Oncotarget. 2017;8(62):105340–55. https://doi.org/10.18632/oncotarget.22186.
Zhang QJ, Wan L, Xu HF. High expression of agrin is associated with tumor progression and poor prognosis in hepatocellular carcinoma. Math Biosci Eng. 2019;16(6):7375–83. https://doi.org/10.3934/mbe.2019368.
Lozano-Ruiz B, Gonzalez-Navajas JM. The emerging relevance of AIM2 in liver disease. Int J Mol Sci. 2020;21(18). https://doi.org/10.3390/ijms21186535.
Chen SL, Liu LL, Lu SX, Luo RZ, Wang CH, et al. HBx-mediated decrease of AIM2 contributes to hepatocellular carcinoma metastasis. Mol Oncol. 2017;11(9):1225–40. https://doi.org/10.1002/1878-0261.12090.
Funding
This work was supported in part by NIH grant number CA 82723 (to BIC) and TÜBİTAK grant numbers 114Z245, 117Z223, 120N556, and 219Z034 (to H.A).
Author information
Authors and Affiliations
Contributions
Conceptualization, H. A. and B. I. C.; writing—original draft preparation, B. S.; writing—review and editing, B. I. C and H. A.; funding acquisition, B. I. C. and H. A. All the authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Ethics Approval
Not applicable.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sengez, B., Carr, B.I. & Alotaibi, H. EMT and Inflammation: Crossroads in HCC. J Gastrointest Canc 54, 204–212 (2023). https://doi.org/10.1007/s12029-021-00801-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12029-021-00801-z