Abstract
The purpose of this prospective study was to determine the incremental diagnostic value of single photon emission computed tomography/computed tomography with iodine-131 over planar whole body scan in the staging of patients with differentiated thyroid carcinoma. A total of 365 patients (270 female, 95 male) with differentiated thyroid carcinoma were treated with radioiodine therapy for thyroid remnant ablation with radical intent after thyroidectomy between January 2013 and November 2014. In addition to planar whole body scan, single photon emission computed tomography/computed tomography of neck and chest were performed. Each radioactive focus at whole body scan was classified as positive or equivocal with respect of specific territories: thyroid bed, cervical lymph nodes and distant metastases.Whole-body scan detected focal uptake in 353 patients and no uptake in 12. The location was considered equivocal in 100. Single photon emission computed tomography/computed tomography detected focal uptake in 356 patients and no uptake in nine. In three patients with negative wholebody scan, single photon emission computed tomography/computed tomography provided information about residual activity in the thyroid bed. By single photon emission computed tomography/computed tomography the location was equivocal in 18 patients only. Single photon emission computed tomography/computed tomography was helpful in 82 out of 100 patients with equivocal findings by whole body scan allowing a correct identification of the uptake sites. In a great number of equivocal whole body scan, due to high remnant activity, single photon emission computed tomography/computed tomography was able to differentiate between thyroid remnant and lymph nodes uptake. In 22 out of 100 patients with doubtful whole body scan, single photon emission computed tomography/computed tomography correctly identified nodal or distant metastases, and in 2/100 patients, focal uptake classified as metastatic by whole body scan was reclassified as para-physiological by single photon emission computed tomography/computed tomography. The TNM classification changed in 13 out of 22 patients. Single photon emission computed tomography/computed tomography improves detection and localization of the iodine-131 uptake after thyroidectomy in patients with differentiated thyroid carcinoma and it is more accurate than whole body scan to evaluate lymph nodes and to identify and characterize distant metastases. Single photon emission computed tomography/computed tomography aids assessment of lower/upper stage in a significant number of patients with differentiated thyroid carcinoma and it can affect therapy decision-making and patient management.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Differentiated thyroid carcinoma (DTC) is the most common endocrine cancer accounting for 1 % of all cancers diagnosed each year [1]. Based on histology, thyroid cancer is classified as papillary (80.2 %), follicular (11.4 %), Hürthle cell (3.1 %), medullary (3.5 %), and anaplastic (1.7 %) [2]. Papillary and follicular carcinomas account for the majority of DTCs and these histological variants are characterized by radioiodine (131I) uptake. The 5-year survival rates for well-differentiated thyroid cancer, are 99.8 % for localized tumors, 97.0 % for tumors with regional metastases, and 57.3 % for tumors with distant metastases [3]. Dinneen et al. reported that the 10-year overall and cause-specific survival rates decreased to 24 and 27 %, respectively, in patients with distant metastasis [4].
After initial thyroidectomy, ablation of thyroid remnants by 131I is performed in the vast majority of patients. Treatment of regional and/or distant metastases by 131I improves the prognosis of patients with metastatic disease [5–8].
Accurate diagnosis of neck lymph node (LN) and distant metastases is mandatory for treatment planning and follow-up. The extent of localized neck nodal disease or distant metastases has a major prognostic impact [9]. Planar 131I WBS, in association with serum thyroglobulin measurement, is considered the routine diagnostic procedure in patients with DTC for diagnostic work-up after thyroidectomy [10]. 131I scintigraphy integrates morphologic imaging modalities, such as ultrasonography, computed tomography, and magnetic resonance imaging, for staging of patients with DTC. In restaging, treatment of metastases and long-term follow-up WBS can be useful, in addition to neck ultrasound and serum thyroglobulin, in selected patients (e.g., metastatic patients and patients with antithyroglobulin antibodies).
131I scintigraphy is conventionally performed as a whole body planar scan in anterior and posterior projections. The limitations of this technology are related to the absence of reliable anatomical references, to the overimposition of different anatomical structures and to physiologic other than pathologic uptake. A sensitivity of 45–75 % has been reported in the literature for diagnostic planar 131I WBS in detecting recurrences or metastases from DTC [11, 12].
Hybrid Single photon emission computed tomography/computed tomography (SPECT/CT) with 131I provides metabolic and morphological information, allowing accurate alignment of anatomical and functional findings. The incremental diagnostic value of SPECT/CT in the management of DTC patients has been investigated in several studies [13–16]. The cost-effectiveness of SPECT/CT was evaluated as well [17].
Purpose of this prospective study was to evaluate the incremental diagnostic value of 131I SPECT/CT over conventional planar WBS in clarifying equivocal findings in a large cohort of consecutive patients. Moreover, the efficacy of SPECT/CT in the prognostic stratification and, as a consequence, in the assessment of therapeutic strategy and planning in DTC patients was investigated.
Materials and methods
From January 2013 until November 2014 we enrolled, prospectively, 365 consecutive patients (270 female and 95 male) after total or nearly total thyroidectomy for DTC. They were admitted to the Nuclear Medicine Department of our Institution for the ablation of thyroid remnant and for 131I WBS.
The patients age ranged from 16 to 83 years with a mean age of 51 ± 14 years. All patients had histopathologically diagnosed DTC: 150 papillary carcinoma, 118 follicular variant of papillary carcinoma, 53 aggressive papillary variants (tall cell, columnar cell or diffuse sclerosing variants), 37 follicular, 5 papillary plus follicular variant of papillary and one papillary plus follicular type. Two hundred and forty-four patients underwent L-thyroxine withdrawal for 40 days, replaced by L-triiodothyronine in the first 20 days.
In 121 patients recombinant human thyrotropin (rhTSH—Thyrogen, Genzyme Corporation) was administered intramuscularly at a dose of 0.9 mg on 2 consecutive days during treatment with levothyroxine, and radioiodine was administered on the day after the second injection. All patients followed a low-iodine diet for 2 weeks in preparation for radioiodine administration. In all patients the serum thyrotropin concentration was higher than 30 m UI/L before 131I administration.
Serum thyrotropin, fT3, fT4, thyroglobulin, antithyroglobulin antibody levels, and ioduria were measured before 131I therapy.
The administered dose of 131I ranged from 1.1 to 4.2 GBq (average 2.6 GBq), that was defined according to the risk class based on the TNM staging of the American Joint Committee on Cancer/International Union against Cancer, currently in the seventh edition [18].
Tipically, 1.1 GBq [30 mCi]) were administered to low risk patients, 1.85 GBq [50 mCi]) to intermediate risk patients, and a high dose (3.7 GBq [100 mCi]) to high risk patients.
All patients underwent WBS followed SPECT/CT scanning on the same day.
The imaging was acquired 3–4 days after 131I administration, by hybrid dual-detector SPECT/CT (Infinia Hawkeye II, GE Healthcare, Haifa Israel), equipped with 1 inch StarBriteTM Crystal and a high energy collimator. The StarBriteTM Crystal has a sensitivity more than two folds higher than that provided by a 3/8 inch crystal without a significant loss in resolution, therefore contributing to improve the reliability of SPECT with 131I.
The WBS was perfomed in countinous mode with high-energy general purpose (HEGP) parallel holes collimator, 364-keV photopeak with ±10 % energy windows setting and scatter correction. The infrared-based real-time automatic body contouring system was activated for simultaneous dual view (anterior/posterior) scans with a matrix of 256 × 1024. The scan speed was 11 cm/min for injected activities ranging from 1100 to 1850 MBq and 15 cm/min for 3700 Mbq.
Hybrid SPECT/CT scans from skull base to the lung bases were routinely obtained in all patients and additional SPECT/CT scans of other areas were performed depending on whole body scintigraphy findings. SPECT images were acquired with HEGP collimator, matrix size of 128 × 128, 364-keV photopeak with ±10 % energy and scatter windows, dual-detector 180° acquisition, angular step of 3°, 15′′ time per step/view. The CT parameters were 140 kV, 2.5 mA, 30′′ rotation speed, 10 mm slice thickness, 256 × 256 matrix. CT acquisition was performed with a 2-slices elicoidal acquisition. An ordered subset expectation maximization iterative reconstruction with CT-based attenuation correction and scatter correction was performed.
The studies were performed for clinical purposes, according to the institutional ethical committee rules. All patients provided written, informed consent.
Data analysis
WBS were considered positive when one or more areas of 131I uptake higher than background activity were identified. A hot nose was considered normal, as well as symmetrical uptake of the salivary glands of the gastrointestinal tract and urinary bladder. Focal uptake localized on the medial portion of the neck was categorized as positive for thyroid bed uptake; foci laterally located in the neck or apart from the medial portion or in the mediastinal region were categorized as positive for LN metastases; foci adjacent to the medial portion but not clearly localized, or streak artifacts, commonly observed with high activity of radioiodine, were interpreted as equivocal for thyroid bed uptake or LN. Foci whose location could not be differentiated between the mediastinum and lung were categorized as equivocal for LN metastases or distant metastases. Uptake in the lung fields, or in the skeleton or in other areas, when they were not clear physiologic accumulation, was categorized as positive for distant metastases.
Planar images were compared with SPECT/CT: Changes in the interpretations from WBS to SPECT/CT were analyzed in the thyroid bed, LN, and distant metastases.
Moreover, we evaluated the change in TNM stage [18].
Statistical analysis
Data were analyzed using cross-tabulation for categorical data in order to examine relationship between variables.
Data analysis was performed using SPSS (Version 20.0 for Mac OS X).
Results
In planar imaging neck focal uptake was observed in 353/365 patients, whereas no uptake was found in 12 (with no correlation with tumor histology). Three hundred and fifty-six SPECT/CT studies were positive and nine were negative, thus documenting thyroid remnant not seen by WBS in three patients.
One hundred planar studies showed equivocal radioactive foci, which could not be interpreted as thyroid remnant, LN, distant metastases, or physiologic uptake.
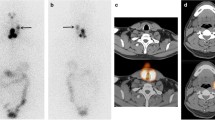
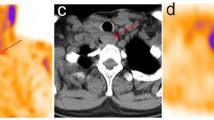
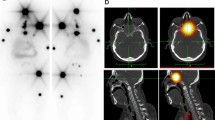
SPECT/CT studies were conclusive in 82/100 patients with equivocal radioactive foci at WBS (22.5 % of total patients). In detail, in 60 patients (16.4 %) SPECT/CT confirmed thyroid remnant uptake, excluding extrathyroidal uptake; in 22 (6.0 % of total patient group) SPECT/CT documented or excluded nodal (Fig. 1) or distant lesions. Specifically, in 20/22 subjects SPECT/CT demonstrated nodal or distant metastases and 2/22 demonstrated para-physiological uptake in a hiatal hernia (Fig. 2) and in a bowel diverticulum. In 18 patients SPECT/CT also was inconclusive (uncertain anatomical attribution). Table 1 summarizes the detailed data for 22 patients for whom additional conclusive information was provided by 131I SPECT/CT fusion imaging after planar imaging.
In 13 subjects out of 22 with equivocal planar WBS, SPECT/CT findings modified the TNM stage and consequently the treatment planning were changed in 13/365 patients, 3.6 % of the patients studied. The results of the study are summarized in Fig. 3.
Discussion
Thyroid cancer is the most frequently occurring endocrine cancer [19], with an incidence of 1 % of all cancers diagnosed each year [1]. The primary treatment of DTC is total or nearly total thyroidectomy with or without LN dissection [20]. Papillary and follicular carcinomas account for the majority of DTC and these histological variants show iodine uptake in the majority of patients due to a presence of the sodium-iodine symporter in the cancer cells [21].
Planar 131I WBS is the standard method for identifying thyroid remnants or metastastic disease [22]. It boasts good sensitivity and high specificity, but anatomical localization of the lesions and the differentiation of neoplastic from normal iodine-avid tissue is frequently critical [23]. The high activity contained in the thyroid residue may hamper the detection of radioiodine-positive cervical LN because of streak artifacts.
Compared with planar imaging, SPECT/CT more clearly localizes and better characterizes the nature of neck and distant radioactivity foci and allows clarification of equivocal or inconclusive results on planar imaging, with subsequent impact on management [24].
In fact hybrid, SPECT/CT systems allow combination of scintigraphic functional imaging (SPECT) data with anatomical (CT) information in a single examination, improving the accuracy of the technique.
Several studies have demonstrated that 131I SPECT/CT has incremental diagnostic value in the diagnosis, treatment, and management of DTC [13, 16, 17, 25–29].
Particularly, 131I SPECT/CT was found to allow a gain in information on nodal staging in 35–36.4 % of patients and resulted in new risk stratification in 6.4–25 % [30, 31].
Disadvantages of SPECT/CT include additional imaging time and possible patient discomfort, claustrophobia from lying in a fixed position for 20 min in the tightly enclosed space of the SPECT/CT gantry and the additional radiation exposure from the CT component of the study (1 mSv for each acquisition).
Barwick and colleagues demonstrated that the use of 131I SPECT/CT changes clinical management in significant numbers of patients with DTC, both when used routinely on all consecutive patients and when used on selected patients with inconclusive planar images [32].
The most commonly used staging system in DTC is the TNM staging of the American Joint Committee on Cancer/International Union against Cancer, currently in the seventh edition [18]. Because this staging was developed to predict risk for death—not for recurrence—and does not take into account several independent prognostic variables, the ATA has developed a 3-level risk stratification for patients with DTC7.
Avram concluded that risk stratification and staging of patients should not be based solely on clinical and histopathological criteria, but should include specific imaging, in particular 131I SPECT/CT imaging, to evaluate for the presence of regional and distant metastases [33].
Kohlfuerst et al. showed that SPECT/CT had a diagnostic effect in 21 (64 %) of 33 patients, and therapeutic planning was changed in eight (24 %) of 33 patients [25]; Schmidt et al. reported that node status was changed with SPECT/CT in 20 (35 %) of 57 patients at the first radioablation [26].
In comparison to previously published studies, specific features of our study are:
-
a.
prospective evaluation of the largest cohort of patients affected by DTC prospectively enrolled for comparison between WBS and SPECT/CT [24];
-
b.
a hybrid SPECT/CT system was adopted in all patients, whereas in other studies fusion imaging was used;
-
c.
to our knowledge the first large cohort of patients evaluated after administration of therapeutic 131I activities (1.1–4.2 GBq) by SPECT/CT equipped with high sensitivity 1′′ StarbriteTM detector allowing higher sensitivity and better image contrast in comparison to 3/8 or 5/8 inch crystal.
The characteristics of this equipment may explain, at least partially, the very low equivocal studies found after SPECT/CT, 18/365 (4.9 %).
Depending on the location of the iodine uptake site, the findings of the 131I SPECT/CT may change a treatment planning, the surveillance, and the need for additional imaging, laboratory tests or invasive procedures like biopsies [33].
Beyond the impact on staging, the utility of 131I SPECT/CT is defined by its impact on clinical management, which may be changed in 23.5–25 % of patients, as reported by Xue et al. in their recent systematic review on the incremental value of 131I SPECT/CT [30].
Our study results demonstrated that the interpretation of equivocal radioactive focal uptake at WBS was clarified by SPECT/CT in 82 (22.5 %) out of 365 patients.
In addition, clinical staging according to the TNM classification and therapeutic planning by addition of SPECT/CT findings were modified in 13 (3.6 %) out of 365 patients. A controversial issue is the selection criteria of patients for SPECT/CT imaging after 131I therapy: all patients or only subjects with positive or equivocal studies. We think that our findings (upstaging in six patients and downstaging in seven, respectively, are sufficient to justify the need of SPECT/CT in the global population, especially in the era of defensive medicine. It is, however, our opinion that at least all patients with equivocal and/or positive findings for nodal or distant metastatic lesions should compulsorily undergo SPECT/CT after 131I treatment.
Study limitations
The main limitation of this study is the use of a low-dose and low-resolution CT; it is, therefore not clear, if the use of a high resolution CT scan might have limited the number of studies (18 patients) still equivocal after SPECT/CT. It should be, however underlined, that a combination of StarbriteTM crystals and high resolution CT is not currently offered by different vendors.
The results of the present study were obtained by a SPECT/CT with StarbriteTM Crystals and consequently they could not be reproduced by centers adopting SPECT/CT equipped with 3/8′′ or 5/8′′ crystals. PET/CT with 124I could represent a competitive alternative, but its application is limited by the difficult supplying of this radiopharmaceutical.
Conclusion
In this study, SPECT/CT showed incremental value over WBS in increasing diagnostic accuracy, reducing pitfalls, and modifying the TNM classification and clinical management defined by planar imaging. SPECT/CT is a powerful diagnostic tool that allows accurate anatomical localization and characterization of radioiodine foci, and both patients and physicians will benefit from this technique in terms of diagnosis, proper staging, restaging, and treatment planning.
Abbreviations
- SPECT/CT:
-
single photon emission computed tomography/computed tomography
- WBS:
-
planar whole body scan
- DTC:
-
differentiated thyroid carcinoma
- LN:
-
neck lymph node
- rhTSH:
-
recombinant human thyrotropin
- HEGP:
-
high-energy general purpose
- ABC:
-
automatic body contouring
References
L. Davies, H.G. Welch, Increasing incidence of thyroid cancer in the United States, 1973–2002. JAMA 295, 2164–2167 (2006)
S.A. Hundahl, I.D. Fleming, A.M. Fremgen, H.R. Menck, A National Cancer Data Base report on 53,856 cases of thyroid carcinoma treated in the U.S., 1985–1995. Cancer 83, 2638–2648 (1998)
E.P. Simard, E.M. Ward, R. Siegel, A. Jemal, Cancers with increasing incidence trends in the United States: 1999 through 2008. CA Cancer J. Clin. 62(2), 118–128 (2012)
S.F. Dinneen, M.J. Valimaki, E.J. Bergstralh, J.R. Goellner, C.A. Gorman, I.D. Hay, Distant metastases in papillary thyroid carcinoma:100 cases observed at one institution during 5 decades. J. Clin. Endocrinol. Metab. 80(7), 2041–2045 (1995)
F. Pacini et al., Post-surgical use of radioiodine (131I) in patients with papillary and follicular thyroid cancer and the issue of remnant ablation: a consensus report. Eur. J. Endocrinol. 153(5), 651–659 (2005)
M. Luster, S.E. Clarke, M. Dietlein et al., Guidelines for radioiodine therapy of differentiated thyroid cancer. Eur. J. Nucl. Med. Mol. Imaging 35(10), 1941–1959 (2008)
B.R. Haugen, E.K. Alexander, K.C. Bible, G.M. Doherty, S.J. Mandel, Y.E. Nikiforov, F. Pacini, G.W. Randolph, A.M. Sawka, M. Schlumberger, K.G. Schuff, S.I. Sherman, J.A. Sosa, D.L. Steward, R.M. Tuttle, L. Wartofsky, 2015 American Thyroid Association Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer: The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid 26(1), 1–133 (2016)
M. Schlumberger, C. Challeton, F. De Vathaire et al., Radioactive iodine treatment and external radiotherapy for lung and bone metastases from thyroid carcinoma. J. Nucl. Med. 37(4), 598–605 (1996)
E.L. Mazzaferri, S.M. Jhiang, Long-term impact of initial surgical and medical therapy on papillary and follicular thyroid cancer. Am. J. Med. 97, 418–428 (1994)
S.I. Sherman, Thyroid carcinoma. Lancet 361, 501–511 (2003)
W.G. Spies, C.H. Wojtowicz, S.M. Spies, A.Y. Shah, A.M. Zimmer, Value of post-therapy whole-body I-131 imaging in the evaluation of patients with thyroid carcinoma having undergone high-dose I- 131 therapy. Clin. Nucl. Med 14, 793–800 (1989)
M. Filesi, A. Signore, G. Ventroni, F.F. Melacrinis, G. Ronga, Role of initial iodine-131 whole-body scan and serum thyroglobulin in differentiated thyroid carcinoma metastases. J. Nucl. Med. 39, 1542–1546 (1998)
K. Tharp, O. Israel, J. Hausmann et al., Impact of 131I-SPECT/CT images obtained with an integrated system in the follow-up of patients with thyroid carcinoma. Eur. J. Nucl. Med. Mol. Imaging 31(10), 1435–1442 (2004)
J. Ruf, L. Lehmkuhl, H. Bertram et al., Impact of SPECT and integrated low-dose CT after radioiodine therapy on the management of patients with thyroid carcinoma. Nucl. Med. Commun. 25(12), 1177–1182 (2004)
L. Chen, Q. Luo, Y. Shen et al., Incremental value of 131I SPECT/CT in the management of patients with differentiated thyroid carcinoma. J. Nucl. Med. 49(12), 1952–1957 (2008)
K.K. Wong, N. Zarzhevsky, J.M. Cahill, K.A. Frey, A.M. Avram, Incremental value of diagnostic 131I SPECT/CT fusion imaging in the evaluation of differentiated thyroid carcinoma. Am. J. Roentgenol. 191(6), 1785–1794 (2008)
Y. Yamamoto, Y. Nishiyama, T. Monden, Y. Matsumura, K. Satoh, M. Ohkawa, Clinical usefulness of fusion of 131I SPECT and CT images in patients with differentiated thyroid carcinoma. J. Nucl. Med. 44, 1905–1910 (2003)
SB Edge, DR Byrd, CC Compton, AG Fritz, FL Greene, A Trotti, (eds.) AJCC Cancer Staging Manual. 7th edn. (New York, NY, Springer, 2010)
National Cancer Institute. Thyroid cancer, http://www.cancer.gov/cancertopics/types/thyroid (2014). Accessed 27 Nov 2015
R.H. Nishiyama, Overview of surgical pathology of the thyroid gland. World J. Surg. 24, 898–906 (2000)
M. Bongiovanni, G. Paone, L. Ceriani, M. Pusztaszer, Cellular and molecular basis for thyroid cancer imaging in nuclear medicine. Clin. Transl. Imaging 1, 149–161 (2013)
B.R. Haugen, E.C. Lin, Isotope imaging for metastatic thyroid cancer. Endocrinol. Metab. Clin. North Am. 30(2), 469–492 (2001)
D.I. Glazer, R.K. Brown, K.K. Wong, H. Savas, M.D. Gross, A.M. Avram, SPECT/CT evaluation of unusual physiologic radioiodine biodistributions: pearls and pitfalls in imaging interpretation. Radiographics 33, 397–418 (2013)
K.K. Wong, D.J. Wale, L.M. Fig, M.D. Gross, SPET–CT in thyroid cancer: a systematic review. Clin. Transl. Imaging 2, 459–475 (2014)
S. Kohlfuerst, I. Igerc, M. Lobnig, H.J. Gallowitsch, I. Gomez-Segovia, S. Matschnig, Z. Mayr, P. Mikosch, M. Beheshti, P. Lind, Posttherapeutic 131I SPECT-CT offers high diagnostic accuracy when the findings on conventional planar imaging are inconclusive and allows a tailored patient treatment regimen. Eur. J. Nucl. Med. Mol. Imaging 36, 886–893 (2009)
D. Schmidt, A. Szikszai, R. Linke, W. Bautz, T. Kuwert, Impact of 131I SPECT/spiral CT on nodal staging of differentiated thyroid carcinoma at the first radioablation. J. Nucl. Med. 50, 18–23 (2009)
N. Aide, N. Heutte, J.P. Rame, E. Rousseau, C. Loiseau, M.H. Amar, S. Bardet, Clinical relevance of single-photon emission computed tomography/computed tomography of the neck and thorax in postablation 131I scintigraphy for thyroid cancer. J. Clin. Endocrinol. Metab. 94, 2075–2084 (2009)
A. Spanu, M.E. Solinas, F. Chessa, D. Sanna, S. Nuvoli, G. Madeddu, 131I SPECT/CT in the follow-up of differentiated thyroid carcinoma: incremental value versus planar imaging. J. Nucl. Med. 50, 184–190 (2009)
H. Wakabayashi, K. Nakajima, M. Fukuoka, A. Inaki, A. Nakamura, D. Kayano, S. Kinuya, Double-phase 131I whole body scan and 131I SPECT-CT images in patients with differentiated thyroid cancer: their effectiveness for accurate identification. Ann. Nucl. Med. 25, 609–615 (2011)
Y.L. Xue, Z.L. Qiu, H.J. Song, Q.Y. Luo, Value of 131I SPECT/CT for the evaluation of differentiated thyroid cancer: a systematic review of the literature. Eur. J. Nucl. Med. Mol. Imaging 40, 768–778 (2013)
Y. Maruoka, K. Abe, S. Baba, T. Isoda et al., Incremental diagnostic value of SPECT/CT with 131I scintigraphy after radioiodine therapy in patients with well-differentiated thyroid carcinoma. Radiology 265, 902–909 (2012)
T. Barwick, I. Murray, H. Megadmi, W.M. Drake, P.N. Plowman, S.A. Akker et al., Single photon emission computed tomography (SPECT)/computed tomography using iodine-123 in patients with differentiated thyroid cancer: additional value over whole body planar imaging and SPECT. Eur. J. Endocrinol. 162, 1131–1139 (2010)
A.M. Avram, Radioiodine scintigraphy with SPECT/CT: an important diagnostic tool for thyroid cancer staging and risk stratification. J. Nucl. Med. 53, 754–764 (2012)
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Zilioli, V., Peli, A., Panarotto, M.B. et al. Differentiated thyroid carcinoma: Incremental diagnostic value of 131I SPECT/CT over planar whole body scan after radioiodine therapy. Endocrine 56, 551–559 (2017). https://doi.org/10.1007/s12020-016-1086-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-016-1086-3