Abstract
The U.S. Institute of Medicine considers that a serum 25-hydroxyvitamin D (25OHD) concentration >20 ng/mL corresponds to optimal vitamin D status in the general population. Old studies of vitamin D status in the French general population have demonstrated high prevalence of insufficiency. We measured serum 25OHD, 1,25(OH)2D, PTH, calcium, phosphorus, and creatinine levels in 892 French Caucasian healthy subjects (463 men, 429 women) aged from 18 to 89 years. The 25OHD concentration was similar in men (24.1 ± 8.2 ng/mL) and women (23.4 ± 8.0 ng/mL). 25OHD concentrations of <10, <12, <20, and <30 ng/mL were found in respectively 6.3, 9.9, 34.6, and 80.3 % of subjects. Residence in northern France (odds ratio [OR] 1.91), blood sampling between January and March (OR 7.74), BMI ≥24 kg/m2 (OR 1.81), and age 60 years or more (OR 1.99) were significant determinants of hypovitaminosis D (25OHD <20 ng/mL). The serum 25OHD level correlated positively with 1,25(OH)2D and negatively with PTH. 25OHD values below 20 ng/mL were associated with lower 1,25(OH)2D levels, and 25OHD values below 27 ng/mL were associated with higher PTH levels. Many French healthy adults have a 25OHD concentration <20 ng/mL, especially during winter months. Actions to improve the vitamin D status of the French general population are urgently needed.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The serum 25-hydroxyvitamin D (25OHD) concentration is the generally accepted marker of vitamin D status [1, 2], but the range of 25OHD levels associated with optimal vitamin D status is controversial. There is good agreement that a 25OHD concentration <10–12 ng/mL reflects severe vitamin D deficiency associated with a risk of rickets/osteomalacia and/or diffuse pain. The U.S. Institute of Medicine considers that a 25OHD concentration above 20 ng/mL represents optimal vitamin D status for the general population [2], while the Endocrine Society considers that a concentration above 30 ng/mL is more appropriate for patients with osseous, renal, digestive, or phosphocalcic disorders [1].
Only two epidemiological studies have examined the vitamin D status of the healthy general population in France. Among 1569 subjects who participated in the SUVIMAX study in 1994, the 25OHD level was 24.4 ng/mL on average and 12 ng/mL or lower in 14 % of cases [3]. In another group of 1824 subjects included in the same study, the 25OHD concentration was 20.0 ng/mL on average, <20 ng/mL in 57.7 % of cases, and <10 ng/mL in 14.8 % of cases [4]. In the ENNS study (Etude Nationale Nutrition Santé, 2006–2007), the 25OHD concentration was 23.0 ng/mL on average (n = 1587), <10 ng/mL in 4.8 % of participants, <12 ng/mL in 9.0 %, <20 ng/mL in 42.5 %, and <30 ng/mL in 80.1 % [5]. These studies showed that vitamin D insufficiency, and severe vitamin D deficiency, were frequent in the French general population, and the authors called for urgent measures to improve the situation. These studies also identified several risk factors for vitamin D deficiency, such as old age, ample clothing, and obesity/overweight. Consumption of vitamin D-fortified foods or vitamin D supplements, and frequent outdoor activity, were associated with better vitamin D status.
As these studies were conducted many years ago, and vitamin D supplementation was intensively promoted in France, we re-examined the current vitamin D status of a large group of French healthy adults enrolled in the VARIETE study. We also sought risk factors for low vitamin D levels (25OHD <20 ng/mL).
Subjects and methods
Subjects
We studied healthy volunteers who participated in the VARIETE study, a population-based cross-sectional study designed to recruit a reference population in order to harmonize normal adult serum IGF-I values (ClinicalTrials.gov identifier: NCT01831648). The subjects were recruited, with their written informed consent, by the clinical research units of ten university hospitals distributed throughout France (Fig. 1), between January 2011 and February 2012. To be included in the study, subjects had to have a normal physical work-up (weight, height, blood pressure, nutritional status, and gonadal/sexual status), normal laboratory values determined after an overnight fast (plasma sodium, potassium, calcium, phosphate, creatinine, glycemia, total cholesterol, liver enzymes, TSH, blood cell counts, albuminemia, prothrombin time, and HIV and HCV serology), age 18–89 years, and BMI between 19 and 28 kg/m2. The exclusion criteria were a medical history of thyroid, renal, hepatic, cardiovascular, pulmonary, intestinal or psychiatric disorders, cancer, epilepsy, onset of illness during the week preceding inclusion, ongoing consumption of tobacco or other toxics, and any treatment potentially modifying IGF-I or calcium/phosphorus metabolism (antiandrogens or antiestrogens, loop diuretics, hydrochlorothiazide, CYP-inducing drugs, bisphosphonates, and other anti-resorptive drugs). In addition to the blood samples necessary for the screening biological evaluation, 50 mL of whole blood and 30 mL of EDTA blood was obtained from each subject. Blood was promptly centrifuged (3000 rpm at 4 °C), and serum or plasma was aliquoted in polypropylene tubes that were immediately stored at −80°C. This study was funded by Programme Hospitalier de Recherche Clinique, French Ministry of Health, No. P081216/IDRCB 2009-A00892-55, and was approved by the Paris-Sud Ethics Committee in November 2009.
Laboratory methods
The biological parameters of the healthy volunteer screening evaluation were determined locally by the laboratories attached to the clinical research units, using standard chemistry. The CKDepi formula was used to estimate the glomerular filtration rate (eGFR). PTH, 25OHD, and 1,25(OH)2D measurements were centralized and done in batches by means of immunochemiluminescent assays on the LIAISON XL automated platform (DiaSorin, Stillwater, Mn, USA), using serum samples that had never been thawed. Table 1 shows the analytical performance of the 3rd-generation PTH and 25OHD assays, as evaluated by one of us (CM), and the analytical performance of the 1,25(OH2)D assay as reported in [6].
Statistical analysis
Quantitative variables are reported as mean ± SD. Associations between serum 25OHD concentrations and other quantitative variables were assessed by simple regression analysis. The LOWESS representation was used to smooth the relationships between 25OHD and PTH and between 25OHD and 1,25(OH)2D. Variables significantly associated with 25OHD were included in a model for multiple regression analysis. The odds ratios of 25OHD <20 ng/mL were calculated for potential risk factors using the Woolf's method. Unpaired data were compared by ANOVA. p-values <0.05 were considered significant.
Results
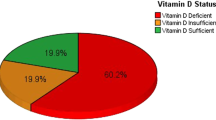
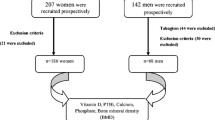
Nine hundred and seventy-two subjects were initially recruited. Eighty were excluded for several reasons (see Fig. 2). The study population thus consisted of 892 subjects (463 men and 429 women) whose main characteristics are summarized in Table 2. All had normal calcemia, phosphatemia, albuminemia, and eGFR values. The 25OHD serum concentration did not differ between men (24.1 ± 8.2 ng/mL) and women (23.4 ± 8.0 ng/mL). The 25OHD concentration was <10 ng/mL in 56 (6.3 %), <12 ng/mL in 88 (9.9 %), <20 g/mL in 309 (34.6 %), and <30 ng/mL in 716 subjects (80.3 %). Residence in Northern France, blood sampling between January and March, BMI ≥24 kg/m2, and age ≥60 years were significant risk factors for suboptimal vitamin D status (Tables 3, 4).
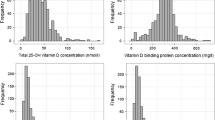
In simple regression analysis the serum 25OHD concentration correlated positively with serum 1,25(OH)2D (r = 0.210; p < 0.001) and negatively with serum PTH (r = −0.272; p < 0.001), age (r = −0.159; p < 0.001), and BMI (r = −0.158; p < 0.001), but did not correlate with eGFR, serum calcium, phosphate, or albumin. These correlations remained significant in multiple regression analysis. The Lowess curve of the relationship between serum 25OHD and 1,25(OH)2D showed an inflexion point at a 25OHD concentration of 20 ng/mL, below which the slope was steeper (Fig. 3a). Similarly, the Lowess curve of the relationship between serum 25OHD and PTH showed an inflexion point at a 25OHD concentration of 27 ng/mL, below which the slope was again steeper (Fig. 3b).
Relationship between serum 1,25(OH)2D and 25OHD levels (Fig. 1a) and between PTH and 25OHD levels (Fig. 1b) in 892 French Caucasian healthy subjects. The solid curves are the Lowess representations of the relationships. An inflexion point in the curve between 1,25(OH)2D and 25OHD, below which the slope is steeper, is found at a 25OHD concentration of approximately 20 ng/mL (vertical arrow Fig. 2a). An inflexion point in the curve between PTH and 25OHD, below which the slope is steeper, is found at a 25OHD concentration of approximately 27 ng/mL (vertical arrow Fig. 2b)
Discussion
Data on the vitamin D status of the general populations in the USA, Canada, and Scandinavian countries are plentiful [7–10], but only two old studies were previously available for France [3–5]. Here, in a large group of French Caucasian healthy adults, we found that the 25OHD concentration was below 20 ng/mL in more than one-third of cases. Although large, this fraction is smaller than that found in the French studies SUVIMAX, conducted in 1994 (57.7 % [3, 4]) and ENNS conducted in 2005 (42.5 % [5]). This apparent decrease in the prevalence of low vitamin D levels might be related to greater public awareness of the potential beneficial effects of vitamin D and of the high frequency of vitamin D deficiency in the general population. However, it could also be due to a recruitment bias. Indeed, we applied numerous inclusion/exclusion criteria and, despite being apparently healthy, 60 (6.7 %) of the subjects who were initially recruited to the study were subsequently excluded because of abnormal results in the screening biological evaluation. In addition, overweight, a well-known risk factor for vitamin D deficiency, was not an exclusion criteria in the SUVIMAX and ENNS studies, while most of our subjects had a normal BMI (average 23 kg/m2). Another point that may partly explain the above-mentioned discrepancies between the results of the present study and those of the SUVIMAX and ENNS study relates to the fact that different 25OHD assays were used in these three studies (Liaison XL in the present study, IDS ELISA in the ENNS study [5], and Roche Cobas [4] or IncStar RIA [3] in the SUVIMAX study). Indeed, inter-method variability in the measurement of 25OHD concentration has been widely reported [11–13]. It is thus probable that some differences in the proportion of vitamin-deficient/insufficient subjects would have been found in these three studies if other 25OHD assays were used.
Interestingly, despite a lower prevalence of vitamin D insufficiency in our study (25OHD <20 ng/mL), the prevalence of severe deficiency was only slightly lower than in the SUVIMAX study and very similar to that found in the ENNS study: almost 10 % of our subjects had 25OHD values below 12 ng/mL. Similarly, the proportion of subjects with a 25OHD concentration <30 ng/mL (i.e. the cut-off value proposed by the Endocrine Society to define optimal vitamin D status [1]) was also similar to what was found in the ENNS and the SUVIMAX study.
It is now widely recognized that vitamin D deficiency in adults is a risk factor for poor bone and muscle health [1], at least in the elderly, and that vitamin D supplementation, usually combined with calcium, significantly reduces the risk of non-vertebral fractures [14] and falls [15]. Many other potential beneficial effects of vitamin D have also been identified [16]. Even if the results of intervention studies focusing on these so-called “non classical” effects are conflicting, and although there is no evidence-based 25OHD threshold value above which these potential benefits occur, it is clearly preferable to avoid or correct vitamin D insufficiency. Our finding that more than one-third of our very healthy volunteers had a serum 25OHD concentration below 20 ng/mL and that more than 80 % had a 25OHD <30 ng/mL is therefore unacceptable, especially as this situation would be easy to correct. One way to improve the vitamin D status of the general population is mandatory fortification of some staple foods. Indeed, trials of food fortification [17–19] show that adding about 8–10 µg of vitamin D to the usual daily diet would virtually eradicate severe vitamin D deficiency (25(OH)D ≤12 ng/mL) in the general population, even if some individuals would still have a serum 25OHD concentration below 20 ng/mL and many still have a 25OHD <30 ng/mL. In France, vitamin D fortification by food manufacturers is voluntary. Pending mandatory fortification, pharmacological supplementation is the best option. However, the cost of serum 25OHD assay is only covered by the French healthcare system in a few specific clinical situations [20], meaning that vitamin D supplementation of the healthy general population would need to take place without prior 25OHD measurement. This makes it even more important to identify subjects at the risk of vitamin D deficiency. We identified several risk factors for low 25OHD serum concentrations, including residence in Northern France and age over 60 years. Also, although most of our subjects had a normal BMI, we found a negative correlation between serum 25OHD and BMI, subjects with BMI ≥24 kg/m2 being twice as likely as leaner subjects to have vitamin D insufficiency (25OHD <20 ng/mL). Studies conducted in France [5] and elsewhere [21] have shown that dark skin, ample clothing, and little outdoor activity are associated with low 25OHD levels. As we recruited Caucasian subjects wearing western clothes, and as we did not record the level of outdoor activity in our subjects, we were unable to confirm these findings. As in the French ENNS study [5], we found that blood sampling for 25OHD assay between January and March was the factor most strongly associated with low 25OHD levels (almost eight times more frequent than during the rest of the year), suggesting that the Health Authorities should consider recommending routine supplementation of the general population during the winter months.
We found that the serum 25OHD concentration correlated positively with 1,25(OH)2D and negatively with PTH. We identified a 25OHD threshold of 20 ng/mL below which the 1,25(OH)2D concentration decreased, suggesting that 1,25(OH)2D production is somewhat impaired below this value. Likewise, we identified a 25OHD threshold of 27 ng/mL below which the PTH concentration increased, suggesting that this value is needed at least to control secondary hyperparathyroidism. It must be noted that 1,25(OH)2D levels continued to increase, albeit less strongly, when 25OHD increased above 20 ng/mL, and that PTH levels continued to fall when 25OHD increased above 27 ng/mL. It was not possible to determine whether the relationship between 25OHD and 1,25(OH)2D or between 25OHD and PTH starts to plateau above a higher 25OHD concentration, as very few subjects had high 25OHD concentrations (>40 ng/mL in 2.9 % and >50 ng/mL in 0.5 % of subjects). Furthermore, the 25OHD threshold value below which PTH increases is reported to be dependent on calcium intake, as subjects with the highest calcium intake need a lower 25OHD concentration to control PTH secretion [22, 23]. Unfortunately, no data on dietary calcium intake were available in our study.
The main strengths of this study are the large number of healthy subjects, the population-based recruitment with strict inclusion criteria, and the centralization of 25OHD, 1,25(OH)2D, and PTH assays in a single laboratory. Several limitations must also be underlined. First, although also representing a strength of this study, the strict inclusion/exclusion criteria probably resulted in the selection of a population with better-than-average health and nutritional status. Thus, the prevalence of vitamin D deficiency might be higher in a less strictly selected population. Second, we did not evaluate dietary/supplemental vitamin D and calcium intake and could not therefore examine their impact on vitamin D status. Third, although we employed the most widely used automated 25OHD immunoassay, and although our laboratory performs very well in the DEQAS international quality control programme, it is possible that another assay method would have given different values and thus different proportions of vitamin D-deficient/insufficient subjects. Indeed, although harmonization of 25OHD assay results has improved since the availability of a reference method [24], and implementation of the Vitamin D Standardization Program (VDSP) [25], some between-method variability persists as indicated above.
In conclusion, approximately 80, 35, and 10 % of a large group of French Caucasian healthy adults had a serum 25OHD concentration below 30, 20, and 12 ng/mL, respectively. The percentage of subjects with low vitamin D status was especially high among those sampled during winter months. As vitamin D deficiency may lead to impaired musculoskeletal health and, possibly, poorer overall health, and can be easily corrected, our results call for more widespread vitamin D supplementation and/or food fortification, especially during the winter months. We confirm the existence of several risk factors for vitamin D deficiency that could be used by family physicians to identify patients most likely to benefit from vitamin D supplementation.
References
M.F. Holick, N.C. Binkley, H.A. Bischoff-Ferrari, C.M. Gordon, D.A. Hanley, R.P. Heaney, M.H. Murad, C.M. Weaver, S. Endocrine, Evaluation, treatment, and prevention of vitamin D deficiency: an Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 96(7), 1911–1930 (2011)
A.C. Ross, J.E. Manson, S.A. Abrams, J.F. Aloia, P.M. Brannon, S.K. Clinton, R.A. Durazo-Arvizu, J.C. Gallagher, R.L. Gallo, G. Jones, C.S. Kovacs, S.T. Mayne, C.J. Rosen, S.A. Shapses, The 2011 report on dietary reference intakes for calcium and vitamin D from the Institute of Medicine: what clinicians need to know. J. Clin. Endocrinol. Metab. 96(1), 53–58 (2011)
M.C. Chapuy, P. Preziosi, M. Maamer, S. Arnaud, P. Galan, S. Hercberg, P.J. Meunier, Prevalence of vitamin D insufficiency in an adult normal population. Osteoporos. Int. 7(5), 439–443 (1997)
M. Touvier, M. Deschasaux, M. Montourcy, A. Sutton, N. Charnaux, E. Kesse-Guyot, L.K. Fezeu, P. Latino-Martel, N. Druesne-Pecollo, D. Malvy, P. Galan, S. Hercberg, K. Ezzedine, J.C. Souberbielle, Interpretation of plasma PTH concentrations according to 25OHD status, gender, age, weight status, and calcium intake: importance of the reference values. J. Clin. Endocrinol. Metab. 99(4), 1196–1203 (2014)
M. Vernay, M. Sponga, B. Salanave, A. Oléko, V. Deschamps, A. Malon, K. Castetbon, Statut en vitamine D de la population adulte en France: l’Étude nationale nutrition santé (ENNS, 2006–2007). Bull. Epidemiol. Hebd. 16–17, 189–194 (2012)
J. van Helden, R. Weiskirchen, Experience with the first fully automated chemiluminescence immunoassay for the quantification of 1alpha, 25-dihydroxy-vitamin D. Clin. Chem. Lab. Med. 53(5), 761–770 (2015)
J. Hilger, A. Friedel, R. Herr, T. Rausch, F. Roos, D.A. Wahl, D.D. Pierroz, P. Weber, K. Hoffmann, A systematic review of vitamin D status in populations worldwide. Br. J. Nutr. 111(1), 23–45 (2014)
T. Skaaby, L.L. Husemoen, B.H. Thesen, C. Pisinger, A. Hannemann, T. Jorgensen, A. Linneberg, Longitudinal associations between lifestyle and vitamin D: a general population study with repeated vitamin D measurements. Endocrine 51, 342–350 (2016)
E. Klingberg, G. Oleröd, J. Konar, M. Petzold, O. Hammarsten, Seasonal variations in serum 25-hydroxyvitamin D levels in a swedish cohort. Endocrine 49, 800–808 (2015)
K.D. Cashman, M. Kiely, Tackling inadequate vitamin D intakes within the population: fortification of dairy products with vitamin D may not be enough. Endocrine 51, 38–46 (2016)
N. Binkley, D.C. Krueger, S. Morgan, D. Wiebe, Current status of clinical 25-hydroxyvitamin D measurement: an assessment of between-laboratory agreement. Clin. Chim. Acta 411, 1976–1982 (2010)
G. Carter, J.L. Berry, E. Gunter, G. Jones, J.C. Jones, H.L. Makin, S. Sufi, M.J. Wheeler, Proficiency testing of 25-hydroxyvitamin D (25-OHD) assays. J. Steroid Biochem. Mol. Biol. 121, 176–179 (2010)
G. Carter, J.C. Jones, J. Shannon, E.L. Williams, G. Jones, M. Kaufmann, C. Sempos, 25-Hydroxyvitamin D assays: potential interference from other circulating vitamin D metabolites. J. Steroid Biochem. Mol. Biol. (2015). doi:10.1016/jsbmb.2015.12.018
H.A. Bischoff-Ferrari, W.C. Willett, E.J. Orav, P. Lips, P.J. Meunier, R.A. Lyons, L. Flicker, J. Wark, R.D. Jackson, J.A. Cauley, H.E. Meyer, M. Pfeifer, K.M. Sanders, H.B. Stahelin, R. Theiler, B. Dawson-Hughes, A pooled analysis of vitamin D dose requirements for fracture prevention. N. Engl. J. Med. 367, 40–49 (2012)
H.A. Bischoff-Ferrari, B. Dawson-Hughes, H.B. Staehelin, J.E. Orav, A.E. Stuck, R. Theiler, J.B. Wong, A. Egli, D.P. Kiel, J. Henschkowski, Fall prevention with supplemental and active forms of vitamin D: a meta-analysis of randomised controlled trials. BMJ 339, b3692 (2009)
S. Christakos, M. Hewison, D.G. Gardner, C.L. Wagner, I.N. Sergeev, E. Rutten, A.G. Pittas, R. Boland, L. Ferrucci, D.D. Bikle, Vitamin D: beyond bone. Ann. N. Y. Acad. Sci. 1287, 45–58 (2013)
L.J. Black, K.M. Seamans, K.D. Cashman, M. Kiely, An updated systematic review and meta-analysis of the efficacy of vitamin D food fortification. J. Nutr. 142(6), 1102–1108 (2012)
K.H. Madsen, L.B. Rasmussen, R. Andersen, C. Molgaard, J. Jakobsen, P.J. Bjerrum, E.W. Andersen, H. Mejborn, I. Tetens, Randomized controlled trial of the effects of vitamin D-fortified milk and bread on serum 25-hydroxyvitamin D concentrations in families in Denmark during winter: the VitmaD study. Am. J. Clin. Nutr. 98(2), 374–382 (2013)
S.J. Whiting, J.P. Bonjour, F.D. Payen, B. Rousseau, Moderate amounts of vitamin D3 in supplements are effective in raising serum 25-hydroxyvitamin D from low baseline levels in adults: a systematic review. Nutrients 7(4), 2311–2323 (2015)
J.C. Souberbielle, C.L. Benhamou, B. Cortet, M. Rousiere, C. Roux, V. Abitbol, M. Audran, J. Bachetta, O. Beauchet, H. Blain, V. Breuil, K. Briot, P. Brunet, P. Chanson, C. Cormier, M. Courbebaisse, P. Fardellone, D. Fouque, G. Friedlander, J.B. Gauvain, L. Groussin, P. Houillier, W. Jacot, G. Jean, P. Kamenicky, M.H. Lafage-Proust, E. Legrand, F. Levy-Weil, A. Linglart, E. Mallet, C. Marcelli, G. Maruani, F. Montagnon, V. Personne, D. Prie, A. Raynaud-Simon, Y. Rolland, B. Salle, C. Sault, A.M. Schott, E. Thervet, P. Urena-Torres, J.P. Viard, G. Weryha, C. Pierrot-Deseilligny, J. Young, T. Thomas, HAS report on vitamin D measurement: don’t go from an extreme situation to another as extreme situation. Presse Med. 43(1), 5–8 (2014)
N.M. van Schoor, P. Lips, Worldwide vitamin D status. Best Pract Res Clin Endocrinol Metab 25(4), 671–680 (2011)
J.F. Aloia, S.A. Talwar, S. Pollack, M. Feuerman, J.K. Yeh, Optimal vitamin D status and serum parathyroid hormone concentrations in African American women. Am. J. Clin. Nutr. 84(3), 602–609 (2006)
P. Patel, M.Z. Mughal, P. Patel, B. Yagnik, N. Kajale, R. Mandlik, V. Khadilkar, S.A. Chiplonkar, S. Phanse, V. Patwardhan, A. Patel, A. Khadilkar, Dietary calcium intake influences the relationship between serum 25-hydroxyvitamin D3 (25OHD) concentration and parathyroid hormone (PTH) concentration. Arch. Dis. Child. 101(4), 316–319 (2015)
H.C. Stepman, A. Vanderroost, K. Van Uytfanghe, L.M. Thienpont, Candidate reference measurement procedures for serum 25-hydroxyvitamin D3 and 25-hydroxyvitamin D2 by using isotope-dilution liquid chromatography-tandem mass spectrometry. Clin. Chem. 57(3), 441–448 (2011)
C.T. Sempos, H.W. Vesper, K.W. Phinney, L.M. Thienpont, P.M. Coates, D.S.P. Vitamin, Vitamin D status as an international issue: national surveys and the problem of standardization. Scand. J. Clin. Lab. Invest. Suppl. 243, 32–40 (2012)
Acknowledgments
We thank DiaSorin for its kind donation of the PTH, 1,25(OH)2D, and 25OHD assay kits. We thank the physicians and technicians of the clinical research units that recruited and examined the healthy subjects, and/or collected data.
Funding
This research was supported by a Grant from Programme Hospitalier de Recherche Clinique, French Ministry of Health, No. P081216/IDRCB 2009-A00892-55.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
JCS reports lecture fees and/or travel/hotel expenses from DiaSorin, Roche Diagnostics, Abbott, Amgen, Shire, MSD, Lilly, and Rottapharm; CM reports lecture fees and travel/hotel expenses from DiaSorin; EC is a consultant for IDS and DiaSorin and has received lecture fees from IDS, DiaSorin, Roche, Abbott, and Amgen; SBT and PC have nothing to disclose.
Rights and permissions
About this article
Cite this article
Souberbielle, JC., Massart, C., Brailly-Tabard, S. et al. Prevalence and determinants of vitamin D deficiency in healthy French adults: the VARIETE study. Endocrine 53, 543–550 (2016). https://doi.org/10.1007/s12020-016-0960-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-016-0960-3