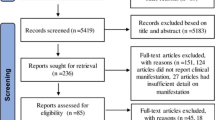
Abstract
Selective immunoglobulin A deficiency (SIgAD) is the most common primary immunodeficiency, defined as an isolated deficiency of IgA (less than 0.07 g/L). Although the majority of people born with IgA deficiency lead normal lives without significant pathology, there is nonetheless a significant association of IgA deficiency with mucosal infection, increased risks of atopic disease, and a higher prevalence of autoimmune disease. To explain these phenomena, we have performed an extensive literature review to define the geoepidemiology of IgA deficiency and particularly the relative risks for developing systemic lupus erythematosus, hyperthyroidism, hypothyroidism, type 1 diabetes mellitus, Crohn’s disease, ulcerative colitis, rheumatoid arthritis, juvenile idiopathic arthritis, ankylosing spondylitis, and vitiligo; these diseases have strong data to support an association. We also note weaker associations with scleroderma, celiac disease, autoimmune hepatitis, immune thrombocytopenic purpura, and autoimmune hemolytic anemia. Minimal if any associations are noted with myasthenia gravis, lichen planus, and multiple sclerosis. Finally, more recent data provide clues on the possible immunologic mechanisms that lead to the association of IgA deficiency and autoimmunity; these lessons are important for understanding the etiology of autoimmune disease.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Selective immunoglobulin A deficiency (SIgAD) is a relatively common primary immunodeficiency which is associated with a number of clinically significant complications. Interestingly, autoimmune conditions are more common among SIgAD populations. This review touches on proposed mechanisms for the development of SIgAD and its complications, and then summarizes the current data regarding autoimmune conditions in individuals with this disease.
Diagnostic Criteria
SIgAD is defined as immunoglobulin A (IgA) levels less than 0.07 g/L in a patient 4 years of age or older with normal immunoglobulin M (IgM) and immunoglobulin G (IgG), and no other identified causes of immunodeficiency [1, 2]. This standardized definition is useful for clinical and research applications, but assumes that normal IgA levels are similar across all demographics—an inaccurate assumption. Data regarding IgA levels in healthy populations are limited, but have shown that average IgA levels increase by 20 mg/dL per decade in women, and are generally higher in male populations than female [3]. IgA levels vary to such an extent with ethnicity and seasons that the prevalence of SIgAD falls during winter months [3]. This raises questions about the validity of current SIgAD diagnostic criteria, because the same patient may have SIgAD when measured in summer, but not in winter [4, 5]. Seasonal variation may be caused by increased exposure to pathogens in the winter, resulting in stimulation of the immune system. It should be noted that this seasonal variation was not seen in a large study of Nigerians which did find IgA variability with age and sex [6]. IgA levels are also known to be associated with metabolic syndrome, heavy drinking, and liver fibrosis [7, 8].
It is also important to note that the definition of SIgAD has changed over time. Indeed, the scientific community did not reach consensus on a single definition of SIgAD until 1999 [9]. Prior studies have identified individuals with SIgAD using variable IgA cutoff values, meaning that some older studies may have included individuals without the disease or excluded patients with the disease under the current definition.
Epidemiology
SIgAD is the most prevalent primary immunodeficiency, found at the highest frequency of 1 in 142 in Caucasians and a low of 1 in 18,550 among Japanese, with a prevalence among other ethnicities ranging between these values [1, 3, 10,11,12,13]. Genetic factors likely play a role in the development of SIgAD, as 33.3% of patients are from consanguineous unions [1]. The disease does not follow simple Mendelian inheritance patterns, but does exhibit familial clustering. For example, the general SIgAD prevalence among Turkish individuals is 1 in 188, compared to 1 in 34 among first-degree relatives of Turkish individuals with SIgAD [14]. Additionally, 20–25% of SIgAD patients have a family history of SIgAD or CVID [2]. Although IgA levels are typically higher in males, men are more likely to have SIgAD (0.28% in men, compared to 0.04% in women) [3].
Function and Structure of IgA
Two thirds of all immunoglobulin produced by the body is IgA, which has an important role in both humoral and mucosal immunity [15]. In humans, IgA exists in two forms—IgA1 and IgA2, with the former more common in blood and the latter primarily in secretions [16]. Both types may exist in a membrane-bound form, free monomeric form, or free dimeric form (Fig. 1). The free, monomeric form is dominant in the serum, whereas the dimeric form is most common in secretions. The dimeric form is wrapped by the secretory component—a cleaved portion of the polymeric immunoglobulin receptor that prevents breakdown of secreted IgA by proteases and gastric acid in the gastrointestinal system [16]. In its various forms, IgA binds antigens on bacteria, viruses, and various toxins to prevent their action at the mucosal barrier, exerts an anti-inflammatory effect, and has been noted in mouse models to influence the intestinal microbiota by binding non-beneficial bacteria and inhibiting their growth, while allowing beneficial bacteria such as Lactobacillus casei to proliferate [15, 17].
The structure of IgA in its monomeric form (left) and dimeric form (right). While both forms are found in secretions and serum, the monomeric form predominates in serum, while the dimeric form predominates in secretions. The monomeric form is composed of an Fc region made of two heavy chains attached via disulfide bonds in the hinge region to the antigen-binding fragment (Fab), which is made up of two arms composed of one heavy chain and one light chain each. The Fc region has a constant structure across all IgA molecules and allows IgA to interact with immune cells via the IgA receptor. The tip of each Fab arm contains a hypervariable region which is specifically designed to bind a particular antigen. The dimeric IgA form consists of two monomeric IgA molecules linked end to end by a J chain at the Fc regions, and wrapped with the secretory component, which is a protein that inhibits digestion of the IgA by proteases
Complications
An increased risk of infections has been widely reported in IgA deficiency, most commonly in the form of mucosal infections. One study found pneumonia in 57.8% of children with SIgAD (compared to an annual incidence of community-acquired pneumonia in the USA of 24.8 per 10,000 persons), recurrent infections in 24.5%, and bronchiectasis in 14.0% (compared to an estimated prevalence of 37–56.6 per 10,000 persons) [1, 18, 19]. This study relied upon caregivers to report diagnoses in a questionnaire. A separate study found 17.5% of children with SIgAD had recurrent URIs, 11.8% had recurrent otitis, 17.8% had pneumonia, and 6.0% had recurrent pneumonia [20]. However, this study did not provide a definition of recurrent URIs, recurrent otitis, or recurrent pneumonia, making it difficult to compare the reported prevalence with the population-level prevalence, particularly when as many as 46% of children in the general population have three or more episodes of otitis media by 3 years of age [21]. In another study, the prevalence of recurrent URI (defined as 3 episodes per year for 3 successive years) and recurrent UTI (defined as 3 episodes over a 2-year period) was 42% and 9%, respectively, among patients with SIgAD, compared to 29% and 3% among individuals with normal IgA levels [22]. In addition to the increased frequency of minor infections observed among patients with SIgAD, the authors also noted a significantly increased incidence of serious infections and complications, including pulmonary tuberculosis and septicemia [22]. Fungal hair and nail infections are seen in 56.3% of individuals with SIgAD, which is significantly more common than the 20.6% in the general population [23].
IgA Deficiency, Allergy, and Atopy
SIgAD is associated with increased prevalence of allergies in patients, as well as their first-degree relatives, although this pattern does not bear out in all studies [24]. According to one pediatric study, 18.78% of SIgAD patients exhibited allergic sequelae (12.1% asthma, 8.8% rhinitis, 3.6% atopic dermatitis, and 4.2% food allergy), with 11.46% undergoing adenoidectomy compared to 1.14% of the general population over the same period [20]. In comparison, population studies have shown that 5–7% of children in the general population have asthma, and 5–12% have rhinitis [25]. Atopic dermatitis has such a variable range of diagnostic criteria among studies that prevalence estimates in the general population vary so much as to be meaningless [26]. Another study showed allergies in 56% of individuals with SIgAD, and asthma in 29.8% [1]. A study of only 39 cases calculated an odds ratio 3.57 (23%) for having asthma among known SIgAD patients [27]. Among Turkish pediatric patients, 45.7% had an allergic disorder (34.6% asthma, 27.2% allergic rhinitis, 11.1% atopic dermatitis, 22.2% positive skin prick), and among their first-degree relatives, 10.9% had asthma, 9.1% had allergic rhinitis, and 7.7% had eczema [14]. One study of SIgAD patients found that 57.84% had atopic dermatitis, although only 10.17% of those had elevated IgE [28]. These studies are generally small, yielding significant variability between resulting prevalence estimates. Although the literature would benefit from larger population studies, the existing data suggest that allergic symptoms are more common both in patients with SIgAD and their first-degree relatives.
IgA Deficiency and Anaphylactic Transfusion Reactions
For decades, the most feared ramification of SIgAD has been the potential for anaphylactic transfusion reactions. Anti-IgA IgE antibodies have been reported in individuals with SIgAD, suggesting the possibility of anaphylactic reactions to blood or IVIG transfusions to sensitized patients. The incidence of such reactions was originally reported as 1 in 20,000–47,000 transfusions [13, 29]. At great cost, many hospitals created vigilance schemes to provide IgA-deficient patients with only blood products that were donated by IgA-deficient donors, or that were washed of IgA. IgA was effectively removed from blood products via washing with buffered saline solutions [30]. Recent findings have cast doubt on the existence of such transfusion reactions, however. Only 17.0% of reported cases were found to have anti-IgA antibodies; many reported cases have subsequently been transfused IgA containing products without experiencing a repeat reaction; and IgA deficiency is not more common among patients who have had anaphylactic transfusion reactions [31]. This evidence suggests that if SIgAD-related anaphylactic transfusion reactions occur, they are less common than previously believed. Additional research is needed to determine conclusively whether SIgAD is truly a cause of anaphylactic transfusion reactions. While it may be safe to give IgA-containing blood to IgA-deficient individuals, until more information is available, this should be done carefully and with close monitoring. Patients with a suspected history of SIgAD-related transfusion reactions should not be given IgA-containing blood products.
IgA Deficiency and CVID
In some cases, SIgAD has also been observed to progress to common variable immunodeficiency (CVID) [32]. Interestingly, one study showed that 23.5% of SIgAD patients with autoimmunity progressed to CVID, while none of the SIgAD patients without autoimmunity did [1]. However, diagnostic uncertainty makes it impossible to determine whether these patients had true SIgAD progressing to CVID, or simply subclinical CVID that progressed over time.
IgA Deficiency and Gastrointestinal Disease
SIgAD has also been associated with lactase deficiency. Twenty-one percent of individuals with SIgAD had confirmed or presumed lactase deficiency, versus 8% of individuals with normal levels of IgA in one study [22]. SIgAD does not appear to be associated with Helicobacter pylori-associated dyspepsia, but among patients with symptomatic H. pylori dyspepsia, those with SIgAD have a significantly higher incidence of gastritis, duodenal ulcers, and nodular lymphoid hyperplasia [33].
Nodular lymphoid hyperplasia (NLH) has been noted in individuals with SIgAD, but it is difficult to determine whether this is a true association because NLH is asymptomatic in most cases and therefore may go undiagnosed in the absence of other GI pathology. It is also known to be associated with Giardia lamblia infection and celiac disease, two conditions that are more common among individuals with SIgAD, further clouding the picture [34, 35]. Many case reports have suggested a possible association, but no studies of prevalence have been published to date [36].
Both recurrent and chronic diarrhea are significantly more common among individuals with SIgAD, but many of these cases may be associated with celiac disease or inflammatory bowel disease, which will be covered more thoroughly below [20].
IgA Deficiency and Malignancy
Although other primary immunodeficiencies have been associated with higher rates of malignancy, the data regarding SIgAD is limited. One study of Spaniards with SIgAD revealed a prevalence of 1.5% for a subset of malignancies including Hodgkin lymphoma, ALL, Wilm’s, Burkitt’s, and ganglioneuroma [20]. A second study of 386 patients with SIgAD found a small increase in incidence of cancer compared to a healthy cohort, but the increase was nonsignificant [37].
IgA Deficiency and Quality of Life
One small study found individuals with SIgAD to have lower average health-related quality of life [38]. Risk factors for lower quality of life scores within the SIgAD cohort included higher number of antibiotic treatments in the past year, higher number of daily medications, allergic rhinoconjunctivitis, chronic MSK symptoms at least every other week, and anxiety or insomnia.
Despite its known association with autoimmune diseases and its putative link with malignancies, SIgAD does not appear to significantly affect mortality, and it has a better prognosis than other primary antibody deficiencies [22, 39].
IgA Deficiency and Autoimmunity
Perhaps the most interesting feature of SIgAD is its association with various autoimmune conditions. Between 25.5 and 31.7% of individuals with SIgAD are afflicted with an autoimmune condition [1, 40, 41]. A study of Turkish children with SIgAD found autoimmune manifestations in only 17.3%, possibly because autoimmunity has less time to manifest in these young individuals [14].
Individuals with SIgAD are also more likely to have family members with autoimmune disorders than healthy controls. In the aforementioned Turkish study, 14.6% of the first-degree relatives of children with SIgAD had autoimmune disorders, compared to 5% of the general population [14]. A second study found that 17.5% of SIgAD individuals had first- or second-degree relatives with autoimmune disorders [1]. Notably, the authors found that a family history of autoimmunity or primary immunodeficiency did not predict whether a given individual with SIgAD would develop autoimmune manifestations. However, not all studies have borne out the same pattern. One study of Spaniards with SIgAD found that only 2.16% of them had a family history of autoimmunity [20].
Interestingly, some studies have suggested a substantive difference between SIgAD individuals who develop autoimmunity and those who do not. One study of 57 patients with symptomatic SIgAD found that mean serum level of IgM significantly higher, regulatory T cell count was lower, and switched memory B cell count was lower in patients with autoimmunity, compared to patients without autoimmunity [1]. The same study noted that 76.4% of those individuals with autoimmunity exhibited a class-switching defect, versus 5% of SIgAD cases without autoimmunity. Unfortunately, the mechanism of autoimmunity in SIgAD remains unknown (Table 1).
Although there is a clear correlation between SIgAD and autoimmunity, not all autoimmune diseases are associated with SIgAD. While a number of studies have examined the prevalence of specific autoimmune diseases among individuals with SIgAD, small study populations lead to significant variability of results. The following is a summary of the available data.
Mechanisms of Autoimmunity in IgA Deficiency
Previous papers have suggested six possible mechanisms for the increased incidence of autoimmunity in individuals with SIgAD, although the mechanisms are not mutually exclusive, and multiple mechanisms are likely at play simultaneously (Table 1) [56].
The first hypothesis suggests that certain human leukocyte antigen (HLA) genes on the MHC locus of chromosome 6 favor the development of both SIgAD and autoimmune diseases. The pattern of familial clustering observed in SIgAD is a strong argument in favor of this hypothesis [46]. An inordinate proportion of patients with SIgAD express haplotype 8.1 (HLA-A1, B8, DR3, DQ2) (45% of SIgAD patients, compared to 16% of general population). Others have noted that HLA-DR7, DQ2 and HLA-DR1, DQ5 are also more prevalent among SIgAD patients, compared to the general population [13, 42,43,44,45, 59]. Many of the HLA types that appear frequently in SIgAD patients overlap with those commonly observed in autoimmune diseases, including celiac disease, type 1 diabetes mellitus, systemic lupus erythematosus, and Grave’s disease [47]. However, a large Swedish study demonstrated no appreciable difference between HLA frequencies in SIgAD versus healthy individuals [60].
A second proposed mechanism suggests that both SIgAD and autoimmunity may arise secondary to the same B cell, T cell, or cytokine abnormalities. Regulatory T cell deficiency is very common in SIgAD (prevalence of 64%) and is also associated with autoimmunity [40]. Abolhassani et al. observed that SIgAD complicated by autoimmunity had fewer regulatory T cells, as well as CD27+IgD− memory B cells, compared to SIgAD without autoimmunity [1]. On the other hand, Nechvatalova and colleagues found that patients with SIgAD had greater numbers of regulatory T cells and differentiated memory T cells, compared to healthy individuals, although the findings may be due to the higher rate of CMV infection among SIgAD patients in this study [61]. In another study, the same group found lower numbers of CD4+ lymphocytes and switched memory B cells, similar to the patterns commonly observed in CVID patients [48]. Another study observed that SIgAD patients with lower numbers of switched memory B cells were more prone to infection and autoimmunity [49]. Indeed, over one quarter of individuals with SIgAD also have low levels of IgG3 or IgG4, and low IgG2 has also been observed in this population suggesting possible subclinical B cell abnormalities [50, 62]. Borte, Lio, and colleagues demonstrated an increase in IgA production in SIgAD patients after stimulation with certain cytokines, arguing for the existence of a class-switching defect [51, 52]. Indeed, one study found that 76.4% SIgAD cases complicated by autoimmunity had a class-switching defect, compared to 5% of SIgAD cases without autoimmunity [1]. However, other studies have noted the existence of immature IgA-positive B cells in SIgAD patients, suggesting that in some cases, the defect is in maturation, rather than class switching of B cells [53]. T and B cells have a higher rate of spontaneous and induced apoptosis in patients with CVID, and Yazdani and colleagues have recently suggested that the same mechanism may be present in those with SIgAD, although this mechanism has yet to be confirmed in individuals with SIgAD [63]. Additionally, SIgAD has been transferred between patients via bone marrow grafting, which further suggests a defect in immune cells [44].
The monogenic hypothesis suggests that certain as-yet undiscovered monogenic mutations predispose both to the development of SIgAD and autoimmune conditions. Researchers have observed that certain primary immunodeficiency syndromes are secondary to monogenic mutations that predispose the same patients to autoimmune manifestations [10]. Bronson and colleagues noted several monogenic associations between SIgAD and autoimmune disease in a genome-wide study [54]. Another paper found a similar variant of CTLA4-ICOS common in celiac disease, SIgAD, and CVID [55]. Mutations in the TACI gene have also been implicated in certain cases of SIgAD [53]. While this mechanism may be at play for certain associations, no single mutation has yet been found to explain the correlation of SIgAD with any specific autoimmune disease, or autoimmunity in general. If a single-gene mutation associated with SIgAD predisposes to autoimmunity, it appears that additional mechanisms must also be at play for any individual to develop autoimmune manifestations.
The fourth hypothesis is more specific to the functions of IgA, which protects mucosal barriers from entry of foreign pathogens and antigens. Studies have clearly established that rates of infection and atopy are higher among patients with SIgAD, suggesting greater mucosal permeability to pathogens and antigens, likely due to the lack of IgA to bind them [1, 14, 20, 22,23,24, 27, 28]. Because a greater number of foreign antigens cross the mucosal barrier in the setting of SIgAD, B and T cells will become sensitized to a greater number of antigenic combinations, which may place patients at increased risk for the development of autoimmune disease. Exposure to infectious agents is known to induce autoimmune disease in some circumstances—important examples being the molecular mimicry of cardiac myosin by group A streptococci causing rheumatic fever, HLA-B27 by Klebsiella pneumoniae causing spondyloarthropathies, and peripheral nerve antigens by Campylobacter jejuni causing Guillain-Barré, among others [64, 65]. Indeed, T cells may become autoreactive and initiate autoimmune disease via molecular mimicry, when the same TCR is capable of recognizing both a foreign antigen and a self-antigen, expression of two distinct T cell receptors (TCRs) by a single T cell such that the same T cell may recognize both a foreign and self-antigen through different TCRs, or expression of a chimeric TCR that may recognize either foreign or self-antigens depending on its configuration [64].
IgA also helps to resolve inflammation in the body, partly via clearance of pathogens and immune complexes [1]. As discussed above, individuals with SIgAD have increased proclivity to infections, which are likely to foster a state of chronic inflammation. The lack of IgA may propagate a state of lingering local and systemic inflammation. Lingering inflammation has a number of negative consequences for health, partly via the release of interferon (IFN)-γ, type I IFN, interleukin (IL)-1β, IL-12, IL-17, and tumor necrosis factor-α, and may predispose to the sensitization of immune cells to auto-antigens [47, 56, 64].
Finally, IgA interacts with cell receptors such as FcαRI to downregulate immune pathways that in turn propagate inflammation [57]. This manifests in such downstream effects as downregulation of neutrophil chemotaxis [58]. In SIgAD patients, IgA is not available to carry out this function, and resulting lingering inflammation may predispose to autoimmunity, as we have discussed above.
Systemic Autoimmune Diseases
Systemic Lupus Erythematosus
Systemic lupus erythematosus (SLE) is a complex autoimmune disease process whose relationship to SIgAD has been studied more thoroughly than many of the disease entities in this review. It does appear to be associated with SIgAD, but it also has other known associations that may cause confounding. For instance, one systematic review found an average SLE prevalence of 3.8% in patients with SIgAD, with prevalence varying based on ethnicity [66]. Prevalence was 1.5% among Caucasians, versus 0.8% among Chinese with SIgAD. However, SLE is very rarely seen in Chinese individuals, so 0.8% is a significant elevation in prevalence, compared to the general population. The same review noted an association between SLE and HLA-B8, DR3, and DQ2 in Caucasian populations. One large study examined the prevalence of SLE among SIgAD individuals (0.57%) and among normal controls (0.06%) in Sweden and found the difference to be statistically significant, with a prevalence ratio of 8.9 [67]. Across all studies in this review, prevalence of SLE among patients with SIgAD was 1.25%, compared to 0.24%, the highest accepted prevalence of SLE in the general population [66, 68,69,70,71,72,73,74,75,76,77,78,79,80] (Table 2). As is the case with many of the autoimmune conditions considered here, SLE has been increasing in prevalence in many populations, so older studies may underestimate the modern prevalence of SLE in this population [68].
Among patients with known SLE, the prevalence of SIgAD is approximately 2.02%, which is much higher than the prevalence of SIgAD observed in any healthy population [57, 67, 81,82,83,84]. One study found that these patients were significantly more likely to have anti-Sm antibodies, anti-La antibodies, and a speckled ANA pattern than patients with SLE but without SIgAD [77]. These individuals did not differ from their counterparts with SLE in symptoms, anti-Ro antibody status, or HLA.
The existing evidence suggests an association between SIgAD and SLE, although future studies will elucidate the role of confounding variables, such as ethnicity and HLA types in this association.
Scleroderma/Systemic Sclerosis
Scleroderma has not been studied exhaustively in SIgAD populations and has only rarely been observed. We found only four studies which mentioned scleroderma or systemic sclerosis. In these studies, the prevalence of scleroderma in SIgAD patients ranged from 0.3 to 6.7%, with a weighted average of 0.78%, compared to a prevalence of 0.004–0.04% in populations with normal serum immunoglobulins [20, 72, 82, 83, 85] (Table 3). It is important to note that the largest of these studies found the lowest prevalence. This may reflect an observation bias, in which only studies which observed systemic sclerosis mentioned it, thus artificially inflating the average prevalence. But even the lowest observed prevalence in SIgAD populations is tenfold higher than in populations without SIgAD. Thus, the available data suggests a possible association between SIgAD and scleroderma which would benefit from confirmation by additional studies.
Sjogren’s Syndrome
Three studies examined the prevalence of Sjogren’s syndrome in SIgAD patients. Two small studies found no patients with Sjogren’s syndrome [40, 84] (Table 4). A third, larger study found a single individual with both SIgAD and Sjogren’s syndrome, but the difference in prevalence compared to a sample of the general population was not significant [22]. A larger study is necessary to determine whether any correlation exists between these diseases.
Sarcoidosis
According to two studies evaluating a total of 247 patients, the prevalence of sarcoidosis among individuals with SIgAD is 3.5% [22, 84] (Table 5). The larger of the two studies found the highest prevalence, but did not find it to be statistically different from the prevalence in the general population. Further studies are required to determine whether any association exists.
Endocrine Disorders
Thyroiditis
Rather than distinguishing between hyperthyroidism and hypothyroidism, many studies simply noted “thyroiditis,” which may evolve into either entity or may resolve completely. The prevalence of thyroiditis among patients with SIgAD ranged from 0.6 to 10.8%, with an overall weighted average prevalence of 3.3% [1, 14, 20, 40, 57, 81, 82, 86, 87] (Table 6).
Hyperthyroidism
Hyperthyroidism appears to have a moderate association with SIgAD. The prevalence of SIgAD among patients with hyperthyroidism ranged from 0 to 2.7%, with a weighted average of 1.6% among five studies [88,89,90,91,92]. Five studies examined the prevalence of hyperthyroidism among individuals with SIgAD, with estimates ranging from 0 to 11.8%, and a weighted average of 2% [14, 22, 67, 84, 92] (Table 7). An early study of 204 patients failed to find a statistically significant difference in prevalence between SIgAD individuals and the general population [22]. However, a 2011 paper found a prevalence of Graves’ disease among Swedish SIgAD individuals that was tenfold higher than the prevalence in the general Swedish population [92]. The largest study to date examined 2100 Swedish SIgAD individuals, finding a prevalence of hyperthyroidism of 1.7%, compared to 0.43% in the general Swedish population [67]. The difference was statistically significant with a PR of 3.9. This suggests an association between SIgAD and hyperthyroidism.
Hypothyroidism
Hypothyroidism appears to have a weak association with SIgAD deficiency. A total of six studies have examined the prevalence of hypothyroidism among SIgAD individuals, finding values between 0.8 and 4.9%, with a weighted average of 1.1% [14, 22, 67, 84, 93, 94] (Table 8). Two studies used a cohort of matched controls. The first found a prevalence of 1.89% and 0% in SIgAD and control populations, respectively, and found that the difference was statistically significant [22]. The second study found a prevalence of 0.76% and 0.16% in SIgAD and control populations, respectively [67]. The difference was significant, with a PR of 3.9. These data support an association between SIgAD and hypothyroidism.
Type 1 Diabetes Mellitus
Type 1 diabetes mellitus (T1DM) is the destruction of insulin-producing pancreatic beta cells, typically due to autoimmunity. The incidence of T1DM varies by ethnicity, but it has been most extensively studied in Caucasians, in whom it is associated with HLA B8, DR3, and DQ2 [66]. Among the general population, T1DM has an estimated prevalence of 0.26 to 0.34% in the USA, versus 0.17% in Italy and 0.014% in Japan [95,96,97]. T1DM appears to be associated with SIgAD, although individuals with T1DM have a similar prevalence of allergy and atopy when compared with the general population [98]. Compared to 0.34%—the highest estimated prevalence of T1DM among the general population of Caucasians, 11 studies have found a prevalence ranging from 0 to 6.35% among SIgAD individuals, with a weighted average of 5.12% [1, 14, 20, 40, 66, 67, 82, 84, 87, 93, 94] (Table 9). Two studies made use of a control population. The first was a study of 330 Spanish individuals with SIgAD and found a prevalence of 3.03% among SIgAD individuals, compared to 0.08% in the general population [20]. The second was a study of 2100 Swedes with SIgAD, which found a statistically significant difference between 5.9% in the SIgAD population and 0.57% in the general population of Sweden, with a prevalence ratio of 10 [67].
Fifteen studies have examined the prevalence of SIgAD among individuals with T1DM, finding a prevalence ranging between 0 and 3.7%, with a weighted average of 1.06% [66, 99,100,101,102,103,104,105,106,107,108,109,110,111]. This prevalence is higher than 1 in 142 (0.7%), the most generous estimate of SIgAD prevalence in the general population. In a study of 200 Greek children, 3% of those with T1DM also had SIgAD, whereas the prevalence of SIgAD among healthy children was 0.2% [111].
These data support an association between T1DM and SIgAD, although individuals with both conditions may have fewer allergic symptoms than those with SIgAD alone.
Gastrointestinal Disorders
Pernicious Anemia
Pernicious anemia is an autoimmune disease process consisting of antibodies against intrinsic factor, its binding site in the terminal ileum, or destruction of the parietal cells which produce it, thereby preventing effective absorption of cobalamin [112]. Five small studies have examined the prevalence of this disease in the population of SIgAD individuals, finding a value between 0 and 2.36%, with a weighted average of 1.26% [22, 40, 82, 84, 94] (Table 10). None of the five studies was sufficiently powered to demonstrate a correlation between pernicious anemia and SIgAD, and none examined the prevalence of SIgAD among individuals with pernicious anemia. Although the prevalence among SIgAD individuals is higher than the estimated prevalence of 0.1% in the general population, it is similar to the estimated 1.9% prevalence among individuals over 60 years of age [113]. Most of these studies did not elaborate on the surveillance method used for pernicious anemia. The observed prevalence would naturally be higher if all individuals were tested, as opposed to symptomatic individuals only. Larger studies are necessary to determine whether a correlation exists between these two diseases. Pernicious anemia has occasionally been noted to coexist as a trifecta with SIgAD and celiac disease [114]. Interestingly, individuals with this trifecta typically lack antibodies against intrinsic factor or parietal cells, raising suggestions that the pernicious anemia in these individuals may represent a distinct disease process that is unrelated to the classically understood pathogenesis of pernicious anemia [115].
The existing data are insufficient to determine whether an association exists between pernicious anemia and SIgAD.
Autoimmune Gastritis
Autoimmune gastritis, also known as autoimmune metaplastic atrophic gastritis, is a broad diagnosis which is based on pathology and exclusion of infectious etiology, and includes the subtype of pernicious anemia involving antibodies against parietal cells [116]. It has an estimated prevalence of 2 to 5% in the general population, but these numbers are based on low-quality studies that are likely affected by sampling bias [117]. Only two studies have estimated the prevalence of this disease among SIgAD individuals, with a weighted average of 0.51% [20, 94] (Table 11). These studies both face the same limitations as those dealing with pernicious anemia, above. More studies are necessary to draw any conclusion about autoimmune gastritis in individuals with SIgAD.
Celiac Disease
Celiac disease is an autoimmune gastrointestinal disease that results in gluten intolerance. It has been associated with multiple genetic loci and certain dietary practices and has an estimated prevalence of 1% in the Western world, where it has been studied most thoroughly [118].
Eleven studies have examined the prevalence of SIgAD among patients with celiac disease, finding a wide variation in prevalence from 0.55 to 16.67%, with a weighted average of 0.57% [66, 119,120,121,122,123,124,125,126,127] (Table 12). A recent study of 317 Americans with celiac disease found a 1.9% prevalence of SIgAD and compared it to a control group, which had a 0.4% prevalence [125]. This difference in prevalence was not deemed statistically significant; however, the study used a more stringent 0.03 g/L cutoff, rather than the generally accepted 0.07 g/L cutoff for diagnosing SIgAD, and may not have been sufficiently powered to detect a true difference in prevalence.
Twelve studies evaluated the prevalence of celiac disease among individuals with SIgAD, finding values between 0.63 and 9.9%, with a weighted average of 6.05%, which is higher than the estimated 1% prevalence in the general population [1, 14, 20, 22, 40, 57, 66, 67, 86, 93, 94, 128]. Three of these studies compared the prevalence among SIgAD individuals to that found in a control population. The smallest of these examined 204 SIgAD individuals with a disease prevalence of 0.63%, compared to 0 cases in the control population, and did not find the difference to be statistically significant [22]. A slightly larger study of 330 SIgAD individuals found a prevalence of 6.67%, compared to 0.5–1.0% in control populations, and did not mention whether the difference was significant [20]. Finally, a study of 2100 Swedes with SIgAD found a disease prevalence of 6.7%, compared with 0.19% in the control population [67]. The authors determined this difference to be statistically significant, with a prevalence ratio of 35.
Importantly, studies of celiac disease in the context of SIgAD face significant limitations. The first test for celiac disease is typically IgA antibodies against TTG, but these antibodies are rarely present in IgA-deficient individuals [120]. Indeed, according to one study, only 9.3% of SIgAD individuals with biopsy-confirmed celiac disease had IgG antibodies against TTG [66]. Therefore, celiac disease in SIgAD individuals cannot be reliably diagnosed with the assays most widely used in the general population [129]. A small study suggested that individuals with SIgAD and celiac disease are not only more likely to have an additional autoimmune disease than individuals with celiac disease and not SIgAD (67% vs 23.5%) but they may also have a lower frequency of GI symptoms (17.7% vs 66.7%) [125]. This has potential clinical significance, since asymptomatic individuals are less likely to be tested for celiac disease, but it is unknown whether untreated celiac disease carries the same risk of lymphomas in these individuals. Studies suggest that individuals with SIgAD and celiac disease have statistically greater TNF-alpha and IL-10 than CD without SIgAD [130]. If the pathogenesis of malignancy in celiac disease is associated with inflammatory pathways, these individuals would continue to be at risk, despite their relative lack of symptoms.
While the existing data suggests a likely association between SIgAD and celiac disease, more carefully designed studies are necessary to confirm this association. It should be noted that celiac disease cannot be reliably diagnosed in SIgAD individuals using IgA and IgG assays, so clinicians should rely on biopsies in this population. Individuals with SIgAD and celiac disease are also less likely to have GI symptoms, so a higher suspicion should be maintained by clinicians caring for these individuals.
Inflammatory Bowel Disease
Inflammatory bowel disease (IBD) is a category of autoimmune bowel disease including both ulcerative colitis and Crohn’s disease. Most studies which remark on IBD treat Crohn’s disease and ulcerative colitis separately, but one study did not distinguish between the two disease entities, and another treated them both individually and also as IBD. These two studies found an average IBD prevalence of 3.88% among SIgAD individuals [40, 67] (Table 13). Ludvigsson et al. compared a prevalence of 3.9% among 2100 people with SIgAD to 0.81% among controls, and found that the difference was statistically significant with a prevalence ratio of 5 [67].
Crohn’s Disease
Crohn’s disease is a type of IBD that affects small and large bowel, often in a patchy, non-contiguous pattern. A total of six studies have estimated the prevalence of Crohn’s disease among SIgAD individuals, finding values between 1.21 and 15.8%, with a weighted average of 2.49% [1, 20, 67, 83, 86, 94] (Table 14). One study of 330 Spaniards compared a prevalence of 1.21% among individuals with SIgAD with a prevalence of 0.13% in a control population, although the authors did not determine whether the difference was statistically significant [20]. A study of 2100 Swedes with SIgAD found the prevalence of 2.4% to be significantly higher than 0.42% among controls, with a prevalence ratio of 5.7 [67]. These results suggest that Crohn’s disease is more prevalent among individuals with SIgAD.
Ulcerative Colitis
Unlike Crohn’s disease, ulcerative colitis (UC) affects only the large bowel and typically presents in a contiguous pattern. Five studies have examined the relationship of UC and SIgAD, finding a prevalence from 0.63 to 7.9%, with a weighted average of 1.73% [1, 22, 67, 83, 87] (Table 15). Koskinen et al. found a prevalence of 0.63% among 204 individuals with SIgAD, which was not significantly different from the prevalence of 0% in the control group [22]. However, a larger study of 2100 individuals with SIgAD found a significant difference between the 1.7% prevalence in the SIgAD population, compared to the 0.46% prevalence in the control population, with a prevalence ratio of 3.9 [67]. These data suggest an association between SIgAD and UC.
Autoimmune Hepatitis
Autoimmune hepatitis (AIH) is an inflammatory condition with unclear pathogenesis involving T cells targeting the liver. Although precise antigens have yet to be identified for many forms of AIH, confirmed subtypes include anti-LKM1, which targets CYP2D6, anti-soluble liver antigen/liver pancreas, which targets SEPSECS, and anti-LC1, which targets formiminotransferase cyclodeaminase [131]. Other variants of the disease are associated with the nonspecific antinuclear antibody (ANA) or anti-smooth muscle antibody (SMA), and still others have not yet been associated with any known autoantibody [131]. The prevalence of this disease varies based on gender, ethnicity, and age, but it is estimated at 0.005% in South Korea, 0.024% in Denmark, and 0.023% in Japan, and appears in a female predominant pattern [132,133,134].
Three studies have examined the prevalence of SIgAD among patients with AIH, finding a weighted average of 4.75% [135,136,137] (Table 16). However, one study of anti-LKM1-positive AIH involved only 5 patients and reported a 40% prevalence [135]. Removing this study from the weighted average yields a 2.87% prevalence rate. The significance of this result is uncertain due to the small sample size of the study; however, a larger study of pediatric patients found that the prevalence of partial IgA deficiency (defined as 1.20 g/L or less) was 45% in anti-LKM1-positive AIJ, versus 9% in ANA/SMA-positive AIH [136]. An additional study of 65 French adults with anti-LKM1-positive AIH reported that 29% has low (defined as 1.2 g/L or less) and 4.6% had undetectable (did not report a specified cutoff) serum IgA [137]. More studies are warranted to determine whether any connection exists between SIgAD and the specific subtypes of AIH.
Five studies reported the prevalence of AIH among patients with AIH, with a range of 0.79 to 5%, and a weighted average of 1.57% [20, 24, 40, 81, 82]. None of these five studies used a control group, so the significance of these results cannot be determined.
The existing data suggests a likely association with AIH, specifically the anti-LKM1-positive subtype. However, larger studies are required to confirm this association.
Primary Sclerosing Cholangitis
Primary sclerosing cholangitis (PSC) is an idiopathic condition involving inflammation and destruction of the bile ducts, with a prevalence in North America and Europe of 0.006 to 0.016% [138]. Only a single study reported prevalence of PSC among individuals with SIgAD, finding a value of 0.79% among 126 Brazilians [57]. This value is higher than the estimated prevalence in the general population of North America, but these numbers represent different populations and cannot be directly compared. No general prevalence data was found for Brazil, so further studies are needed to determine whether an association exists.
Musculoskeletal Disorders
Rheumatoid Arthritis
Rheumatoid arthritis (RA) is an autoimmune disease characterized primarily by synovial inflammation and joint destruction, and classically characterized by rheumatoid factor and anti-cyclic citrullinated peptide antibodies. It has an estimated prevalence of 0.41 to 0.54% among insured adults in the USA [139].
Five studies have examined the prevalence of SIgAD among individuals with RA, finding a range of 0.25 to 4%, with a weighted average of 0.5% [71, 75, 140,141,142]. Seven studies reported the prevalence of RA among individuals with SIgAD, finding a range of 2.2 to 5.7%, and a weighted average of 2.4% [22, 24, 67, 81,82,83,84] (Table 17). Two of these studies used control groups. Koskinen et al. found no individuals with RA in the control group and did not determine the significance of the prevalence discrepancy [22]. Ludvigsson et al. found a prevalence of 2.2% for RA among those with SIgAD versus 0.5% in the control group, which is consistent with other prevalence estimates for RA in the general population [67]. The authors determined that the difference in prevalence was statistically significant, with a prevalence ratio of 4.5. Individuals with both RA and SIgAD did not differ in terms of long-term disease activity or mortality compared to those with RA alone [140].
The available data support an association between RA and SIgAD.
Juvenile Idiopathic Arthritis
Juvenile idiopathic arthritis (JIA) is a chronic arthritis affecting children, with a general prevalence between 0.0038 and 0.4% [143]. Nine studies have examined the prevalence of SIgAD among children with JIA, finding a range from 1 to 4.35%, with a weighted average of 2.7% [79, 144,145,146,147,148,149,150,151] (Table 18). A total of nine studies reported the prevalence of JIA among individuals with SIgAD, finding 0 to 15.67%, with a weighted average of 1.97% [1, 20, 39, 67, 72, 81, 82, 86, 93]. Two of these studies utilized control groups. One study found that 3.33% of patients with SIgAD and only 0.11% of the general population had JIA, but the significance of this difference was not reported [20]. The other study showed that 0.76% of patients with known SIgAD had JIA, compared to 0.09% of controls, for a prevalence ratio of 8.9, and the authors determined this to be a statistically significant difference [67]. These data support an association between JIA and SIgAD.
Ankylosing Spondylitis
The prevalence of ankylosing spondylitis (AS) has been estimated as ranging from a high of 0.238% in Europe to a low of 0.074 in Africa [152]. Only four studies have reported the prevalence of AS among individuals with SIgAD, finding a range of 0 to 2.52%, with a weighted average of 1.73% [14, 22, 81, 84] (Table 19). The largest of these studies involved only 204 individuals with SIgAD; however, it compared the 2.52% prevalence in this group to a control group, in which the prevalence was 0.42% [22]. This difference was not found to be statistically significant, possibly because the study was not sufficiently powered.
This disease appears to manifest differently, depending on the HLA type of the affected individual. Serum levels of IgA are negatively associated with disease activity in HLA-B27-negative individuals, but not in B27-positive patients [153]. It has additionally been suggested that SIgAD is a poor prognostic marker for AS [154].
Limited evidence suggests that an association may exist between SIgAD and AS—particularly in HLA-B27-negative individuals—but more studies are necessary to elucidate this connection.
Myositis
Myositis refers to a broad spectrum of disease processes, whose breadth makes it difficult to determine the overall prevalence in the general population. However, most studies of SIgAD populations do not distinguish between individual etiologies of myositis when reporting prevalence. Five studies report the prevalence of myositis in populations with SIgAD, ranging from 0 to 7.32%, with a weighted average of 3.58% [1, 40, 72, 81, 155] (Table 20). No studies reported the prevalence of SIgAD among patients with myositis, although one study observed that IgA levels in people with polymyositis and dermatomyositis were similar to those in controls [155]. The existing data is inadequate to determine whether any relationship exists between SIgAD and myositis.
Dermatologic Disorders
Vitiligo
Vitiligo is an autoimmune disorder resulting in the destruction of melanocytes in a patchy distribution and is estimated to have a global prevalence of approximately 1% [156]. Only a single study reported the prevalence of SIgAD among individuals with vitiligo, finding a prevalence of 50% in the SIgAD population, which was significantly higher than a prevalence of 36.7% among controls [157]. It should be noted that the cutoff used for SIgAD in this study was very high at 4 g/L, so the study likely overestimates the prevalence of SIgAD in both groups. A larger study found no significant difference in IgA levels between individuals with and without vitiligo, but it was not designed to detect SIgAD [158].
Nine studies investigated the prevalence of vitiligo among patients with SIgAD, with results varying between 0.79 and 15.38%, with a weighted average of 2.89% [1, 20, 22, 28, 40, 57, 86, 87, 94] (Table 21). Only one of these studies used a control group, finding a prevalence of 4.4% and 0.42% among patients with SIgAD and controls, respectively, and found the difference to be significant [22]. These results suggest a relationship between vitiligo and SIgAD.
Psoriasis
Psoriasis is an inflammatory skin disease that is associated with arthritis in some individuals. It has a prevalence between 0.73 and 3.2%, with prevalence increasing with greater distance from the equator, and in adults, compared to children [159,160,161]. Psoriasis has been associated with multiple sclerosis, metabolic syndrome, inflammatory bowel disease, and psychiatric disease, although no studies have reported on the prevalence of SIgAD among individuals with psoriasis [162,163,164,165]. Seven studies have reported the prevalence of psoriasis among individuals with SIgAD, with results varying between 0 and 6.3%, with a weighted average of 2.72% [22, 23, 28, 40, 84, 93, 94] (Table 22). Each of these studies was relatively small, and although two used control populations, neither was sufficiently powered to detect a difference between the populations [22, 23]. Studies of psoriasis patients have shown high and low levels of IgA, and it has been postulated that IgA may be involved in the pathogenesis of persistent stationary psoriasis, while acting as a protective factor against guttate-type psoriasis [166]. It has been suggested that IgA impairs neutrophil chemotaxis, which would support this observation [58]. Unfortunately, none of the studies of psoriasis in individuals with SIgAD reliably distinguish between subtypes of psoriasis. Larger studies are necessary to elucidate any such relationship with specific subtypes of psoriasis.
Alopecia Areata
Alopecia areata is an autoimmune condition resulting in destruction of hair follicles, resulting in nonscarring hair loss. It has a reported prevalence in the USA of 0.1 to 0.2% [167]. No studies were found to report the prevalence of SIgAD among individuals with alopecia areata, but four studies have reported the prevalence of alopecia areata within SIgAD populations, varying from 0.9 to 3.51%, with a weighted average of 2.3% [1, 28, 40, 86] (Table 23). All of these studies were small, with the largest only involving 102 participants. No study used a control group. While all four studies found a prevalence higher than would be expected in the general population, it is impossible to determine whether any true association exists, based on the current data.
Recurrent Aphthous Stomatitis
Recurrent aphthous stomatitis (RAS) is an inflammatory condition resulting in painful ulcerations on the oropharyngeal mucosa, and is thought to affect approximately 1.6% of the general population [168]. Three studies reported prevalence of RAS among individuals with SIgAD, finding values from 1.2 to 4.9%, with a weighted average of 2.4% [20, 28, 128] (Table 24). However, RAS is associated with other autoimmune disorders including celiac disease and inflammatory bowel disease, and may be a manifestation of these diseases in some cases [168]. Without controlling for these disease entities, it is impossible to determine whether RAS has a separate correlation with SIgAD.
Lichen Planus
Lichen planus is a disease affecting the stratified squamous epithelia with uncertain etiology and with a prevalence ranging 0.22 to 5% [169]. The prevalence of lichen planus among individuals with SIgAD has only been recorded by two studies, with a weighted average of 0.97% [22, 40] (Table 25). Koskinen et al. used a control group, and although the prevalence was higher among the SIgAD group, the difference was not significant. The limited available data does not suggest that the prevalence of lichen planus in SIgAD individuals differs from that of the general population.
Renal
Glomerulonephritis
Glomerulonephritis denotes inflammation of the glomerulus, which may occur secondary to multiple primary and secondary etiologies. Observed prevalence varies between 0.12 and 1.23%, depending on the population [170].
A single study noted the prevalence of SIgAD among individuals with glomerulonephritis as 0.7%, compared to 0% of a healthy control group, and this difference was not statistically significant [80].
Two studies reported the prevalence of glomerulonephritis in SIgAD populations, finding a weighted average of 1.49% [22, 24] (Table 26). The larger of the two studies compared the results to a control populations and did not find a significant difference in prevalence [22]. The existing data is very limited, but it does not support a relationship between SIgAD and glomerulonephritis.
Neuromuscular Disorders
Myasthenia Gravis
Myasthenia gravis (MG) is a neuromuscular disorder in which antibodies form against the post-synaptic acetylcholine receptor, inhibiting signaling at the neuromuscular junction. It has an estimated prevalence of 0.08% in the general population, with greater prevalence among older populations [171].
Seven studies have reported the prevalence of SIgAD among individuals with MG, ranging from 0 to 1.85%, with a weighted average of 0.44% [66, 155, 172,173,174,175,176] (Table 27). The most recent of these papers used a control group and found no significant difference in prevalence [176].
Five studies examined the prevalence of MG among SIgAD populations, finding a rate of 0 to 2.7%, with a weighted average of 0.18% [1, 40, 67, 86, 155]. The largest of these studies used a control group, finding no significant difference in prevalence [67]. The existing evidence does not support a correlation between SIgAD and MG.
Multiple Sclerosis
There have been two case reports of multiple sclerosis (MS) in patients with SIgAD, raising the possibility of a correlation [178, 179]. However, two small studies have failed to find any cases of MS among patients with SIgAD [40, 84]. Moreover, any association between MS and SIgAD is unlikely to be missed, because the diagnostic workup for MS typically includes CSF immunoglobulins, which would likely reveal any previously undiagnosed SIgAD [180]. There is therefore no evidence to support a correlation between MS and SIgAD.
Hematologic Disorders
Immune Thrombocytopenic Purpura
Immune thrombocytopenic purpura (ITP) is an autoimmune disorder resulting in the destruction of platelets. Good quality studies of prevalence are lacking, and it has been estimated at 0.024% in the general population in the USA [181]. It is widely assumed to be one of the most prevalent autoimmune manifestations of SIgAD. Multiple reviews report a prevalence of 1:200, citing the same article, which does not supply data about ITP [182]. Seven studies have reported on the prevalence of ITP among individuals with SIgAD, varying from 0 to 5.71%, with a weighted average of 1.8% [20, 24, 39, 40, 82, 84, 93] (Table 28). None of these studies utilized control groups, so although the reported prevalence is greater among SIgAD populations, it is impossible to determine whether this difference is statistically significant.
Autoimmune Hemolytic Anemia
Autoimmune hemolytic anemia (AIHA) is the activation of the immune system against red blood cells, causing their lysis. Because it encompasses multiple etiologies, there is little good quality data on prevalence in the general population. However, prevalence has been estimated at 0.017% [183].
Only a single study examined the prevalence of SIgAD among individuals with AIHA, finding a prevalence of 17% [184]. Rather than using the accepted definition of SIgAD, this study defined it as a value 2 standard deviations below the mean, which limits the applicability of the results.
Eight studies reported the prevalence of AIHA among individuals with SIgAD, reporting values between 0 and 15.38%, yielding a weighted average of 2.18% [1, 20, 39, 40, 57, 82, 86, 87] (Table 29). The small size of the studies, the wide variability of the results, and the lack of control groups make it difficult to draw conclusions from these findings. However, it is interesting to note that on average, French children with isolated AIHA (not associated with Evans syndrome) had lower-than-average IgA levels [184].
Although the existing data suggest a possible relationship between SIgAD and AIHA, it cannot be confirmed without additional studies.
Insufficient Data
Multiple additional conditions have been noted in SIgAD individuals only in single studies, with insufficient data to compare prevalence to that of the general population. These included autoimmune lymphoproliferative syndrome [1], uveitis [20], rheumatic fever [22], Guillain-Barré [40], Still’s disease [71], pemphigus vulgaris [84], Stevens-Johnson [84], interstitial cystitis [84], polymyalgia rheumatica [84], reactive arthritis [84], cryoglobulinemia [82], vasculitis [82], Henoch-Schonlein purpura [22], Kawasaki disease [40], anti-sperm antibodies [82], granuloma annulare [93], familial Mediterranean fever [14], and pulmonary hemosiderosis [14]. The available data can be found in the appendices.
No prevalence data could be found regarding mixed connective tissue disease, hidradenitis suppurativa, primary biliary cirrhosis, or bullous pemphigoid.
Discussion
The data presented in this review makes it clear that in addition to other clinically important manifestations of SIgAD, this disease is also associated with a number of autoimmune diseases. Between one quarter and one third of individuals with SIgAD are afflicted with autoimmune disease, but SIgAD does not guarantee the development of such a condition [1, 40, 41]. Clinicians should keep this association in mind, maintaining a high level of suspicion for autoimmune disease in patients with known SIgAD, and in some cases screening for systemic lupus erythematosus, hyperthyroidism, hypothyroidism, type 1 diabetes mellitus, Crohn’s disease, ulcerative colitis, rheumatoid arthritis, juvenile idiopathic arthritis, ankylosing spondylitis, and vitiligo—the autoimmune entities with the strongest association with SIgAD. Clinicians should also be aware that the current 0.07 g/L cutoff for diagnosing SIgAD is somewhat arbitrary. It remains unclear whether patients with IgA levels near, but not below this level carry a similarly increased risk for developing autoimmune disease. There is some evidence that SIgAD may be stratified into variants that do or do not carry an increased risk for autoimmune disease, based, for instance on the existence of a class-switching defect, which may favor autoimmune manifestations [1]. Nevertheless, further studies are necessary to elucidate the clinical utility of assessing complement profiles, IgG subtypes, class-switching defects, or monogenic variants in patients with SIgAD. We hope that further research in this area will reveal interventions for preventing the development of autoimmunity in people with selective IgA deficiency.
References
Abolhassani H, Gharib B, Shahinpour S, Masoom SN, Havaei A, Mirminachi B, Arandi N, Torabi-Sagvand B, Khazaei HA, Mohammadi J, Rezaei N, Aghamohammadi A (2015) Autoimmunity in patients with selective IgA deficiency. J Investig Allergol Clin Immunol 25(2):112–119
Bonilla FA, Khan DA, Ballas ZK, Chinen J, Frank MM, Hsu JT, Keller M, Kobrynski LJ, Komarow HD, Mazer B, Nelson RP Jr, Orange JS, Routes JM, Shearer WT, Sorensen RU, Verbsky JW, Bernstein DI, Blessing-Moore J, Lang D, Nicklas RA, Oppenheimer J, Portnoy JM, Randolph CR, Schuller D, Spector SL, Tilles S, Wallace D, Joint Task Force on Practice Parameters rtAAoAA, Immunology, the American College of Allergy A, Immunology, the Joint Council of Allergy A, Immunology (2015) Practice parameter for the diagnosis and management of primary immunodeficiency. J Allergy Clin Immunol 136(5):1186–1205 e1181–1178. https://doi.org/10.1016/j.jaci.2015.04.049
Weber-Mzell D, Kotanko P, Hauer AC, Goriup U, Haas J, Lanner N, Erwa W, Ahmaida IA, Haitchi-Petnehazy S, Stenzel M, Lanzer G, Deutsch J (2004) Gender, age and seasonal effects on IgA deficiency: a study of 7293 Caucasians. Eur J Clin Investig 34(3):224–228. https://doi.org/10.1111/j.1365-2362.2004.01311.x
Buckley RH, Dees SC (1967) Serum immunoglobulins. 3. Abnormalities associated with chronic urticaria in children. J Allergy Ther 40(5):294–303
Stiehm ER, Fudenberg HH (1966) Serum levels of immune globulins in health and disease: a survey. Pediatrics 37(5):715–727
Oyeyinka GO, Salimonu LS, Williams AI, Johnson AO, Ladipo OA, Osunkoya BO (1984) Range of normal serum immunoglobulin (IgG, IgA and IgM) values in Nigerians. Afr J Med Med Sci 13(3–4):169–176
Gonzalez-Quintela A, Alende R, Gude F, Campos J, Rey J, Meijide LM, Fernandez-Merino C, Vidal C (2008) Serum levels of immunoglobulins (IgG, IgA, IgM) in a general adult population and their relationship with alcohol consumption, smoking and common metabolic abnormalities. Clin Exp Immunol 151(1):42–50. https://doi.org/10.1111/j.1365-2249.2007.03545.x
Maleki I, Aminafshari MR, Taghvaei T, Hosseini V, Rafiei A, Torabizadeh Z, Barzin M, Orang E (2014) Serum immunoglobulin A concentration is a reliable biomarker for liver fibrosis in non-alcoholic fatty liver disease. World J Gastroenterol 20(35):12566–12,573. https://doi.org/10.3748/wjg.v20.i35.12566
Conley ME, Notarangelo LD, Etzioni A (1999) Diagnostic criteria for primary immunodeficiencies. Representing PAGID (Pan-American Group for Immunodeficiency) and ESID (European Society for Immunodeficiencies). Clin Immunol 93(3):190–197. https://doi.org/10.1006/clim.1999.4799
Abolhassani H, Aghamohammadi A, Hammarstrom L (2016) Monogenic mutations associated with IgA deficiency. Expert Rev Clin Immunol 12(12):1321–1335. https://doi.org/10.1080/1744666x.2016.1198696
Yazdani R, Azizi G, Abolhassani H, Aghamohammadi A (2017) Selective IgA deficiency: epidemiology, pathogenesis, clinical phenotype, diagnosis, prognosis and management. Scand J Immunol 85(1):3–12. https://doi.org/10.1111/sji.12499
Modell V, Quinn J, Orange J, Notarangelo LD, Modell F (2016) Primary immunodeficiencies worldwide: an updated overview from the Jeffrey Modell Centers Global Network. Immunol Res 64(3. United States):736–753. https://doi.org/10.1007/s12026-016-8784-z
Singh K, Chang C, Gershwin ME (2014) IgA deficiency and autoimmunity. Autoimmun Rev 13(2):163–177. https://doi.org/10.1016/j.autrev.2013.10.005
Erkocoglu M, Metin A, Kaya A, Ozcan C, Akan A, Civelek E, Capanoglu M, Ginis T, Kocabas CN (2017) Allergic and autoimmune disorders in families with selective IgA deficiency. Turk J Med Sci 47(2):592–598. https://doi.org/10.3906/sag-1605-50
Pabst O (2012) New concepts in the generation and functions of IgA. Nat Rev Immunol 12(12):821–832. https://doi.org/10.1038/nri3322
Woof JM, Kerr MA (2004) IgA function—variations on a theme. Immunology 113(2):175–177. https://doi.org/10.1111/j.1365-2567.2004.01958.x
Okai S, Usui F, Ohta M, Mori H, Kurokawa K, Matsumoto S, Kato T, Miyauchi E, Ohno H, Shinkura R (2017) Intestinal IgA as a modulator of the gut microbiota. Gut Microbes 8(5):486–492. https://doi.org/10.1080/19490976.2017.1310357
Jain S, Self WH, Wunderink RG, Fakhran S, Balk R, Bramley AM, Reed C, Grijalva CG, Anderson EJ, Courtney DM, Chappell JD, Qi C, Hart EM, Carroll F, Trabue C, Donnelly HK, Williams DJ, Zhu Y, Arnold SR, Ampofo K, Waterer GW, Levine M, Lindstrom S, Winchell JM, Katz JM, Erdman D, Schneider E, Hicks LA, McCullers JA, Pavia AT, Edwards KM, Finelli L, Team CES (2015) Community-acquired pneumonia requiring hospitalization among U.S. adults. N Engl J Med 373(5):415–427. https://doi.org/10.1056/NEJMoa1500245
Chalmers JD, Sethi S (2017) Raising awareness of bronchiectasis in primary care: overview of diagnosis and management strategies in adults. NPJ Prim Care Respir Med 27(1):18. https://doi.org/10.1038/s41533-017-0019-9
Dominguez O, Giner MT, Alsina L, Martin MA, Lozano J, Plaza AM (2012) [Clinical phenotypes associated with selective IgA deficiency: a review of 330 cases and a proposed follow-up protocol]. An Pediatr (Barc) 76(5):261–267. https://doi.org/10.1016/j.anpedi.2011.11.006
Cheong KH, Hussain SS (2012) Management of recurrent acute otitis media in children: systematic review of the effect of different interventions on otitis media recurrence, recurrence frequency and total recurrence time. J Laryngol Otol 126(9):874–885. https://doi.org/10.1017/S0022215112001338
Koskinen S (1996) Long-term follow-up of health in blood donors with primary selective IgA deficiency. J Clin Immunol 16(3):165–170
Jorgensen GH, Gardulf A, Sigurdsson MI, Sigurdardottir ST, Thorsteinsdottir I, Gudmundsson S, Hammarstrom L, Ludviksson BR (2013) Clinical symptoms in adults with selective IgA deficiency: a case-control study. J Clin Immunol 33(4):742–747. https://doi.org/10.1007/s10875-012-9858-x
Burgio GR, Duse M, Monafo V, Ascione A, Nespoli L (1980) Selective IgA deficiency: clinical and immunological evaluation of 50 pediatric patients. Eur J Pediatr 133(2):101–106
Weeke ER (1992) Epidemiology of allergic diseases in children. Rhinol Suppl 13:5–12
Dizon MP, Yu AM, Singh RK, Wan J, Chren MM, Flohr C, Silverberg JI, Margolis DJ, Langan SM, Abuabara K (2018) Systematic review of atopic dermatitis disease definition in studies using routinely collected health data. Br J Dermatol 178(6):1280–1287. https://doi.org/10.1111/bjd.16340
Urm SH, Yun HD, Fenta YA, Yoo KH, Abraham RS, Hagan J, Juhn YJ (2013) Asthma and risk of selective IgA deficiency or common variable immunodeficiency: a population-based case-control study. Mayo Clin Proc 88(8):813–821. https://doi.org/10.1016/j.mayocp.2013.05.021
Gualdi G, Lougaris V, Baronio M, Vitali M, Tampella G, Moratto D, Tanghetti P, Monari P, Calzavara-Pinton P, Plebani A (2015) Burden of skin disease in selective IgA deficiency and common variable immunodeficiency. J Investig Allergol Clin Immunol 25(5):369–371
Sandler SG, Mallory D, Malamut D, Eckrich R (1995) IgA anaphylactic transfusion reactions. Transfus Med Rev 9(1):1–8
Davenport RD, Burnie KL, Barr RM (1992) Transfusion management of patients with IgA deficiency and anti-IgA during liver transplantation. Vox Sang 63(4):247–250
Sandler SG, Eder AF, Goldman M, Winters JL (2015) The entity of immunoglobulin A-related anaphylactic transfusion reactions is not evidence based. Transfusion 55(1):199–204. https://doi.org/10.1111/trf.12796
Aghamohammadi A, Mohammadi J, Parvaneh N, Rezaei N, Moin M, Espanol T, Hammarstrom L (2008) Progression of selective IgA deficiency to common variable immunodeficiency. Int Arch Allergy Immunol 147(2):87–92. https://doi.org/10.1159/000135694
Magen E, Waitman DA, Goldstein N, Schlesinger M, Dickstein Y, Kahan NR (2016) Helicobacter pylori infection in patients with selective immunoglobulin a deficiency. Clin Exp Immunol 184(3):332–337. https://doi.org/10.1111/cei.12765
Albuquerque A (2014) Nodular lymphoid hyperplasia in the gastrointestinal tract in adult patients: a review. World J Gastrointest Endosc 6(11):534–540. https://doi.org/10.4253/wjge.v6.i11.534
Agarwal S, Mayer L (2010) Gastrointestinal manifestations in primary immune disorders. Inflamm Bowel Dis 16(4):703–711. https://doi.org/10.1002/ibd.21040
Ajdukiewicz AB, Youngs GR, Bouchier IA (1972) Nodular lymphoid hyperplasia with hypogammaglobulinaemia. Gut 13(8):589–595
Mellemkjaer L, Hammarstrom L, Andersen V, Yuen J, Heilmann C, Barington T, Bjorkander J, Olsen JH (2002) Cancer risk among patients with IgA deficiency or common variable immunodeficiency and their relatives: a combined Danish and Swedish study. Clin Exp Immunol 130(3):495–500
Jorgensen GH, Gardulf A, Sigurdsson MI, Arnlaugsson S, Hammarstrom L, Ludviksson BR (2014) Health-related quality of life (HRQL) in immunodeficient adults with selective IgA deficiency compared with age- and gender-matched controls and identification of risk factors for poor HRQL. Qual Life Res 23(2):645–658. https://doi.org/10.1007/s11136-013-0491-9
Mohammadinejad P, Pourhamdi S, Abolhassani H, Mirminachi B, Havaei A, Masoom SN, Sadeghi B, Ghajar A, Afarideh M, Parvaneh N, Mirsaeed-Ghazi B, Movahedi M, Gharagozlou M, Chavoushzadeh Z, Mahdaviani A, Zandieh F, Sherkat R, Sadeghi-Shabestari M, Faridhosseini R, Jabbari-Azad F, Ahanchian H, Zandkarimi M, Cherghi T, Fayezi A, Mohammadzadeh I, Amin R, Aleyasin S, Moghtaderi M, Ghaffari J, Bemanian M, Shafiei A, Kalantari N, Ahmadiafshar A, Khazaei HA, Mohammadi J, Nabavi M, Rezaei N, Aghamohammadi A (2015) Primary antibody deficiency in a tertiary referral hospital: a 30-year experiment. J Investig Allergol Clin Immunol 25(6):416–425
Azizi G, Tavakol M, Rafiemanesh H, Kiaee F, Yazdani R, Heydari A, Abouhamzeh K, Anvari P, Mohammadikhajehdehi S, Sharifia L, Bagheri Y, Mohammadi H, Abolhassani H, Aghamohammadi A (2017) Autoimmunity in a cohort of 471 patients with primary antibody deficiencies. Expert Rev Clin Immunol 13(11):1099–1106. https://doi.org/10.1080/1744666X.2017.1384312
Sarmiento E, Mora R, Rodriguez-Mahou M, Rodriguez-Molina J, Fernandez-Cruz E, Carbone J (2005) [Autoimmune disease in primary antibody deficiencies]. Allergol Immunopathol 33(2):69–73
Ferreira RC, Pan-Hammarstrom Q, Graham RR, Fontan G, Lee AT, Ortmann W, Wang N, Urcelay E, Fernandez-Arquero M, Nunez C, Jorgensen G, Ludviksson BR, Koskinen S, Haimila K, Padyukov L, Gregersen PK, Hammarstrom L, Behrens TW (2012) High-density SNP mapping of the HLA region identifies multiple independent susceptibility loci associated with selective IgA deficiency. PLoS Genet 8(1):e1002476. https://doi.org/10.1371/journal.pgen.1002476
Mohammadi J, Ramanujam R, Jarefors S, Rezaei N, Aghamohammadi A, Gregersen PK, Hammarstrom L (2010) IgA deficiency and the MHC: assessment of relative risk and microheterogeneity within the HLA A1 B8, DR3 (8.1) haplotype. J Clin Immunol 30(1):138–143. https://doi.org/10.1007/s10875-009-9336-2
Hammarstrom L, Lonnqvist B, Ringden O, Smith CI, Wiebe T (1985) Transfer of IgA deficiency to a bone-marrow-grafted patient with aplastic anaemia. Lancet 1(8432):778–781
Hammarstrom L, Vorechovsky I, Webster D (2000) Selective IgA deficiency (SIgAD) and common variable immunodeficiency (CVID). Clin Exp Immunol 120(2):225–231
Vorechovsky I, Webster AD, Plebani A, Hammarstrom L (1999) Genetic linkage of IgA deficiency to the major histocompatibility complex: evidence for allele segregation distortion, parent-of-origin penetrance differences, and the role of anti-IgA antibodies in disease predisposition. Am J Hum Genet 64(4):1096–1109
Wang N, Hammarstrom L (2012) IgA deficiency: what is new? Curr Opin Allergy Clin Immunol 12(6):602–608. https://doi.org/10.1097/ACI.0b013e3283594219
Nechvatalova J, Pikulova Z, Stikarovska D, Pesak S, Vlkova M, Litzman J (2012) B lymphocyte subpopulations in patients with selective IgA deficiency. J Clin Immunol 32(3):441–448. https://doi.org/10.1007/s10875-012-9655-6
Aghamohammadi A, Abolhassani H, Biglari M, Abolmaali S, Moazzami K, Tabatabaeiyan M, Asgarian-Omran H, Parvaneh N, Mirahmadian M, Rezaei N (2011) Analysis of switched memory B cells in patients with IgA deficiency. Int Arch Allergy Immunol 156(4):462–468. https://doi.org/10.1159/000323903
Bonilla FA, Geha RS (2006) 2. Update on primary immunodeficiency diseases. J Allergy Clin Immunol 117(2 Suppl Mini-Primer):S435–S441. https://doi.org/10.1016/j.jaci.2005.09.051
Borte S, Pan-Hammarstrom Q, Liu C, Sack U, Borte M, Wagner U, Graf D, Hammarstrom L (2009) Interleukin-21 restores immunoglobulin production ex vivo in patients with common variable immunodeficiency and selective IgA deficiency. Blood 114(19):4089–4098. https://doi.org/10.1182/blood-2009-02-207,423
Lio D, D’Anna C, Leone F, Curro MF, Candore G, Caruso C (1998) Hypothesis: interleukin-5 production impairment can be a key point in the pathogenesis of the MHC-linked selective IgA deficiency. Autoimmunity 27(3):185–188. https://doi.org/10.3109/08916939809003866
Vo Ngoc DT, Krist L, van Overveld FJ, Rijkers GT (2017) The long and winding road to IgA deficiency: causes and consequences. Expert Rev Clin Immunol 13(4):371–382. https://doi.org/10.1080/1744666x.2017.1248410
Bronson PG, Chang D, Bhangale T, Seldin MF, Ortmann W, Ferreira RC, Urcelay E, Pereira LF, Martin J, Plebani A, Lougaris V, Friman V, Freiberger T, Litzman J, Thon V, Pan-Hammarstrom Q, Hammarstrom L, Graham RR, Behrens TW (2016) Common variants at PVT1, ATG13-AMBRA1, AHI1 and CLEC16A are associated with selective IgA deficiency. Nat Genet 48(11):1425–1429. https://doi.org/10.1038/ng.3675
Haimila K, Einarsdottir E, de Kauwe A, Koskinen LL, Pan-Hammarstrom Q, Kaartinen T, Kurppa K, Ziberna F, Not T, Vatta S, Ventura A, Korponay-Szabo IR, Adany R, Pocsai Z, Szeles G, Dukes E, Kaukinen K, Maki M, Koskinen S, Partanen J, Hammarstrom L, Saavalainen P (2009) The shared CTLA4-ICOS risk locus in celiac disease, IgA deficiency and common variable immunodeficiency. Genes Immun 10(2. England):151–161. https://doi.org/10.1038/gene.2008.89
Todoric K, Koontz JB, Mattox D, Tarrant TK (2013) Autoimmunity in immunodeficiency. Curr Allergy Asthma Rep 13(4):361–370. https://doi.org/10.1007/s11882-013-0350-3
Jacob CM, Pastorino AC, Fahl K, Carneiro-Sampaio M, Monteiro RC (2008) Autoimmunity in IgA deficiency: revisiting the role of IgA as a silent housekeeper. J Clin Immunol 28(Suppl 1):S56–S61. https://doi.org/10.1007/s10875-007-9163-2
Schroder JM, Szperalski B, Koh CJ, Christophers E (1981) IgA-associated inhibition of polymorphonuclear leukocyte chemotaxis in neutrophilic dermatoses. J Invest Dermatol 77(6):464–468
Cuccia-Belvedere M, Monafo V, Martinetti M, Plebani A, De Paoli F, Burgio GR (1989) Recurrent extended HLA haplotypes in children with selective IgA deficiency. Tissue Antigens 34(2):127–132
Frankowiack M, Kovanen RM, Repasky GA, Lim CK, Song C, Pedersen NL, Hammarstrom L (2015) The higher frequency of IgA deficiency among Swedish twins is not explained by HLA haplotypes. Genes Immun 16(3. England):199–205. https://doi.org/10.1038/gene.2014.78
Nechvatalova J, Pavlik T, Litzman J, Vlkova M (2017) Terminally differentiated memory T cells are increased in patients with common variable immunodeficiency and selective IgA deficiency. Cent Eur J Immunol 42(3):244–251. https://doi.org/10.5114/ceji.2017.70966
Tezcan I, Ersoy F, Sanal O (1992) IgG subclasses in symptomatic IgA deficiency. Turk J Pediatr 34(1):1–4
Yazdani R, Fatholahi M, Ganjalikhani-Hakemi M, Abolhassani H, Azizi G, Hamid KM, Rezaei N, Aghamohammadi A (2016) Role of apoptosis in common variable immunodeficiency and selective immunoglobulin A deficiency. Mol Immunol 71:1–9. https://doi.org/10.1016/j.molimm.2015.12.016
Cusick MF, Libbey JE, Fujinami RS (2012) Molecular mimicry as a mechanism of autoimmune disease. Clin Rev Allergy Immunol 42(1):102–111. https://doi.org/10.1007/s12016-011-8293-8
Sfriso P, Ghirardello A, Botsios C, Tonon M, Zen M, Bassi N, Bassetto F, Doria A (2010) Infections and autoimmunity: the multifaceted relationship. J Leukoc Biol 87(3):385–395. https://doi.org/10.1189/jlb.0709517
Wang N, Shen N, Vyse TJ, Anand V, Gunnarson I, Sturfelt G, Rantapää-Dahlqvist S, Elvin K, Truedsson L, Andersson BA, Dahle C, Örtqvist E, Gregersen PK, Behrens TW, Hammarström L (2011) Selective IgA deficiency in autoimmune diseases. Mol Med 17(11–12):1383–1396. https://doi.org/10.2119/molmed.2011.00195
Ludvigsson JF, Neovius M, Hammarstrom L (2014) Association between IgA deficiency & other autoimmune conditions: a population-based matched cohort study. J Clin Immunol 34(4):444–451. https://doi.org/10.1007/s10875-014-0009-4
Rees F, Doherty M, Grainge MJ, Lanyon P, Zhang W (2017) The worldwide incidence and prevalence of systemic lupus erythematosus: a systematic review of epidemiological studies. Rheumatology (Oxford) 56(11):1945–1961. https://doi.org/10.1093/rheumatology/kex260
Alarcon-Segovia D, Fishbein E (1972) Serum immunoglobulins in systemic lupus erythematosus. Clin Sci 43(1):121–131
Calabozo RM, Gamir GM, Medina LJ, Diaz-Miguel PC, Alonso RA (1990) Selective deficiency of IgA in autoimmunity diseases [in Spanish]. Rev Clin Esp 186(4):163–165
Cassidy JT, Burt A, Petty R, Sullivan D (1969) Selective IgA deficiency in connective tissue diseases. N Engl J Med 280(5):275
Cassidy JT, Kitson RK, Selby CL (2007) Selective IgA deficiency in children and adults with systemic lupus erythematosus. Lupus 16(8):647–650. https://doi.org/10.1177/0961203307077543
Gershwin ME, Blaese RM, Steinberg AD, Wistar R Jr, Strober W (1976) Antibodies to nucleic acids in congenital immune deficiency states. J Pediatr 89(3):377–381
Kaufman LD, Heinicke MH, Hamburger M, Gorevic PD (1991) Male lupus: prevalence of IgA deficiency, 7S IgM and abnormalities of reticuloendothelial system Fc-receptor function. Clin Exp Rheumatol 9(3):265–269
Kustimur S, Gulmezoglu E (1985) [Selective IgA deficiency in patients with systemic lupus erythematosus and rheumatoid arthritis]. Mikrobiyol Bul 19(4):190–199
Mantovani AP, Monclaro MP, Skare TL (2010) Prevalence of IgA deficiency in adult systemic lupus erythematosus and the study of the association with its clinical and autoantibody profiles. Rev Bras Reumatol 50(3):273–282
Rankin EC, Isenberg DA (1997) IgA deficiency and SLE: prevalence in a clinic population and a review of the literature. Lupus 6(4):390–394. https://doi.org/10.1177/096120339700600408
Rifle G, Bielefeld P, Chalopin JM, Besancenot JF, Guiguet M, Mousson C, Tanter Y, Poirier D, Cortet P (1988) Selective IgA deficiency and systemic lupus erythematosus. Ann Med Interne (Paris) 139(2):134–137
Sparchez M, Lupan I, Delean D, Bizo A, Damian L, Muntean L, Tamas MM, Bolba C, Simionescu B, Slavescu C, Felea I, Lazar C, Sparchez Z, Rednic S (2015) Primary complement and antibody deficiencies in autoimmune rheumatologic diseases with juvenile onset: a prospective study at two centers. Pediatr Rheumatol Online J 13:51. https://doi.org/10.1186/s12969-015-0050-8
Yewdall V, Cameron JS, Nathan AW, Neild G, Ogg CS, Williams DG (1983) Systemic lupus erythematosus and IgA deficiency. J Clin Lab Immunol 10(1):13–18
Petty RE, Palmer NR, Cassidy JT, Tubergen DG, Sullivan DB (1979) The association of autoimmune diseases and anti-IgA antibodies in patients with selective IgA deficiency. Clin Exp Immunol 37(1):83–88
Edwards E, Razvi S, Cunningham-Rundles C (2004) IgA deficiency: clinical correlates and responses to pneumococcal vaccine. Clin Immunol 111(1):93–97. https://doi.org/10.1016/j.clim.2003.12.005
Santaella ML, Cox PR, Colon M, Ramos C, Disdier OM (2005) Rheumatologic manifestations in patients with selected primary immunodeficiencies evaluated at the university hospital. P R Health Sci J 24(3):191–195
Jorgensen GH, Thorsteinsdottir I, Gudmundsson S, Hammarstrom L, Ludviksson BR (2009) Familial aggregation of IgAD and autoimmunity. Clin Immunol 131(2):233–239. https://doi.org/10.1016/j.clim.2008.11.013
Ingegnoli F, Ughi N, Mihai C (2018) Update on the epidemiology, risk factors, and disease outcomes of systemic sclerosis. Best Pract Res Clin Rheumatol 32(2):223–240. https://doi.org/10.1016/j.berh.2018.08.005
Aghamohammadi A, Cheraghi T, Gharagozlou M, Movahedi M, Rezaei N, Yeganeh M, Parvaneh N, Abolhassani H, Pourpak Z, Moin M (2009) IgA deficiency: correlation between clinical and immunological phenotypes. J Clin Immunol 29(1):130–136. https://doi.org/10.1007/s10875-008-9229-9
Soheili H, Abolhassani H, Arandi N, Khazaei HA, Shahinpour S, Hirbod-Mobarakeh A, Rezaei N, Aghamohammadi A (2013) Evaluation of natural regulatory T cells in subjects with selective IgA deficiency: from senior idea to novel opportunities. Int Arch Allergy Immunol 160(2):208–214. https://doi.org/10.1159/000339867
Carroccio A, Custro N, Montalto G, Giannitrapani L, Soresi M, Notarbartolo A (1999) Evidence of transient IgA anti-endomysial antibody positivity in a patient with Graves’ disease. Digestion 60(1):86–88. https://doi.org/10.1159/000007595
Ch’ng CL, Biswas M, Benton A, Jones MK, Kingham JG (2005) Prospective screening for coeliac disease in patients with Graves’ hyperthyroidism using anti-gliadin and tissue transglutaminase antibodies. Clin Endocrinol 62(3):303–306. https://doi.org/10.1111/j.1365-2265.2005.02214.x
Collin P, Salmi J, Hallstrom O, Reunala T, Pasternack A (1994) Autoimmune thyroid disorders and coeliac disease. Eur J Endocrinol 130(2):137–140
Cuoco L, Certo M, Jorizzo RA, De Vitis I, Tursi A, Papa A, De Marinis L, Fedeli P, Fedeli G, Gasbarrini G (1999) Prevalence and early diagnosis of coeliac disease in autoimmune thyroid disorders. Ital J Gastroenterol Hepatol 31(4):283–287
Jorgensen GH, Ornolfsson AE, Johannesson A, Gudmundsson S, Janzi M, Wang N, Hammarstrom L, Ludviksson BR (2011) Association of immunoglobulin A deficiency and elevated thyrotropin-receptor autoantibodies in two Nordic countries. Hum Immunol 72(2):166–172. https://doi.org/10.1016/j.humimm.2010.10.014
Aytekin C, Tuygun N, Gokce S, Dogu F, Ikinciogullari A (2012) Selective IgA deficiency: clinical and laboratory features of 118 children in Turkey. J Clin Immunol 32(5):961–966. https://doi.org/10.1007/s10875-012-9702-3
Shkalim V, Monselize Y, Segal N, Zan-Bar I, Hoffer V, Garty BZ (2010) Selective IgA deficiency in children in Israel. J Clin Immunol 30(5):761–765. https://doi.org/10.1007/s10875-010-9438-x
Menke A, Orchard TJ, Imperatore G, Bullard KM, Mayer-Davis E, Cowie CC (2013) The prevalence of type 1 diabetes in the United States. Epidemiology 24(5):773–774. https://doi.org/10.1097/EDE.0b013e31829ef01a
Onda Y, Sugihara S, Ogata T, Yokoya S, Yokoyama T, Tajima N, Type 1 Diabetes Study G (2017) Incidence and prevalence of childhood-onset type 1 diabetes in Japan: the T1D study. Diabet Med 34(7):909–915. https://doi.org/10.1111/dme.13295
Bruno G, Pagano E, Rossi E, Cataudella S, De Rosa M, Marchesini G, Miccoli R, Vaccaro O, Bonora E (2016) Incidence, prevalence, costs and quality of care of type 1 diabetes in Italy, age 0-29 years: the population-based CINECA-SID ARNO Observatory, 2002-2012. Nutr Metab Cardiovasc Dis 26(12):1104–1111. https://doi.org/10.1016/j.numecd.2016.09.002
Jasser-Nitsche H, Varga EM, Borkenstein HM, Hontzsch J, Suppan E, Weinhandl G, Pieringer L, Avian A, Frohlich-Reiterer E (2017) Type 1 diabetes in children and adolescents is not associated with a reduced prevalence of atopy and allergic diseases. Pediatr Diabetes 18(8):890–894. https://doi.org/10.1111/pedi.12504
Smith WI Jr, Rabin BS, Huellmantel A, Van Thiel DH, Drash A (1978) Immunopathology of juvenile-onset diabetes mellitus. I. IgA deficiency and juvenile diabetes. Diabetes 27(11):1092–1097
Hoddinott S, Dornan J, Bear JC, Farid NR (1982) Immunoglobulin levels, immunodeficiency and HLA in type 1 (insulin-dependent) diabetes mellitus. Diabetologia 23(4):326–329
Bertrams J, Schoeps L, Baur MP, Luboldt W, van Loghem E (1983) IgG and IgA heavy chain allotypes in type 1 diabetes. J Immunogenet 10(4):305–310
Cerutti F, Urbino A, Sacchetti C, Palomba E, Zoppo M, Tovo PA (1988) [Selective IgA deficiency in juvenile-onset insulin-dependent diabetes mellitus]. Pediatr Med Chir 10(2):197–201
Liblau RS, Caillat-Zucman S, Fischer AM, Bach JF, Boitard C (1992) The prevalence of selective IgA deficiency in type 1 diabetes mellitus. APMIS 100(8):709–712
Page SR, Lloyd CA, Hill PG, Peacock I, Holmes GK (1994) The prevalence of coeliac disease in adult diabetes mellitus. QJM 87(10):631–637
Acerini CL, Ahmed ML, Ross KM, Sullivan PB, Bird G, Dunger DB (1998) Coeliac disease in children and adolescents with IDDM: clinical characteristics and response to gluten-free diet. Diabet Med 15(1):38–44. https://doi.org/10.1002/(SICI)1096-9136(199801)15:1<38::AID-DIA520>3.0.CO;2-L
Fraser-Reynolds KA, Butzner JD, Stephure DK, Trussell RA, Scott RB (1998) Use of immunoglobulin A-antiendomysial antibody to screen for celiac disease in North American children with type 1 diabetes. Diabetes Care 21(11):1985–1989
Schober E, Bittmann B, Granditsch G, Huber WD, Huppe A, Jager A, Oberhuber G, Rami B, Reichel G (2000) Screening by anti-endomysium antibody for celiac disease in diabetic children and adolescents in Austria. J Pediatr Gastroenterol Nutr 30(4):391–396
Picarelli A, Sabbatella L, Di Tola M, Vetrano S, Casale C, Anania MC, Porowska B, Vergari M, Schiaffini R, Gargiulo P (2005) Anti-endomysial antibody of IgG1 isotype detection strongly increases the prevalence of coeliac disease in patients affected by type I diabetes mellitus. Clin Exp Immunol 142(1):111–115. https://doi.org/10.1111/j.1365-2249.2005.02866.x
Laadhar L, Zitouni M, Kallel-Sellami M, Masmoudi S, Bouguerra R, Chaabouni H, Ben Slama C, Makni S (2006) [Prevalence of celiac disease serological markers in Tunisian type 1 diabetic adults]. Ann Biol Clin (Paris) 64(5):439–444
Sayarifard F, Aghamohammadi A, Haghi-Ashtiani MT, Rajab A, Irani H, Ahmadian JH, Zaridoost A, Parvaneh N, Rezaei N, Rabbani A (2012) Evaluation of serum IgA levels in Iranian patients with type 1 diabetes mellitus. Acta Diabetol 49(2):131–135. https://doi.org/10.1007/s00592-010-0183-7
Giza S, Kotanidou E, Papadopoulou-Alataki E, Antoniou MC, Maggana I, Kyrgios I, Galli-Tsinopoulou A (2016) Prevalence of selective immunoglobulin A deficiency in Greek children and adolescents with type 1 diabetes. World J Pediatr 12(4):470–476. https://doi.org/10.1007/s12519-016-0039-5
Bizzaro N, Antico A (2014) Diagnosis and classification of pernicious anemia. Autoimmun Rev 13(4–5):565–568. https://doi.org/10.1016/j.autrev.2014.01.042
Andres E, Serraj K (2012) Optimal management of pernicious anemia. J Blood Med 3:97–103. https://doi.org/10.2147/JBM.S25620
Quigley EM, Carmichael HA, Watkinson G (1986) Adult celiac disease (celiac sprue), pernicious anemia and IgA deficiency. Case report and review of the relationships between vitamin B12 deficiency, small intestinal mucosal disease and immunoglobulin deficiency. J Clin Gastroenterol 8(3 Pt 1):277–281
Ginsberg A, Mullinax F (1970) Pernicious anemia and monoclonal gammopathy in a patient with IgA deficiency. Am J Med 48(6):787–791
Minalyan A, Benhammou JN, Artashesyan A, Lewis MS, Pisegna JR (2017) Autoimmune atrophic gastritis: current perspectives. Clin Exp Gastroenterol 10:19–27. https://doi.org/10.2147/CEG.S109123
Coati I, Fassan M, Farinati F, Graham DY, Genta RM, Rugge M (2015) Autoimmune gastritis: pathologist’s viewpoint. World J Gastroenterol 21(42):12179–12,189. https://doi.org/10.3748/wjg.v21.i42.12179
Gujral N, Freeman HJ, Thomson AB (2012) Celiac disease: prevalence, diagnosis, pathogenesis and treatment. World J Gastroenterol 18(42):6036–6059. https://doi.org/10.3748/wjg.v18.i42.6036
Cataldo F, Marino V, Bottaro G, Greco P, Ventura A (1997) Celiac disease and selective immunoglobulin A deficiency. J Pediatr 131(2):306–308
Cataldo F, Marino V, Ventura A, Bottaro G, Corazza GR (1998) Prevalence and clinical features of selective immunoglobulin A deficiency in coeliac disease: an Italian multicentre study. Italian Society of Paediatric Gastroenterology and Hepatology (SIGEP) and “Club del Tenue” Working Groups on Coeliac Disease. Gut 42(3):362–365
Demir H, Yuce A, Kocak N, Ozen H, Gurakan F (2000) Celiac disease in Turkish children: presentation of 104 cases. Pediatr Int 42(5):483–487
Heneghan MA, Stevens FM, Cryan EM, Warner RH, McCarthy CF (1997) Celiac sprue and immunodeficiency states: a 25-year review. J Clin Gastroenterol 25(2):421–425
Lavo B, Knutson F, Knutson L, Sjoberg O, Hallgren R (1992) Jejunal secretion of secretory immunoglobulins and gliadin antibodies in celiac disease. Dig Dis Sci 37(1):53–59
McGowan KE, Lyon ME, Butzner JD (2008) Celiac disease and IgA deficiency: complications of serological testing approaches encountered in the clinic. Clin Chem 54(7):1203–1209. https://doi.org/10.1373/clinchem.2008.103606
Pallav K, Xu H, Leffler DA, Kabbani T, Kelly CP (2016) Immunoglobulin A deficiency in celiac disease in the United States. J Gastroenterol Hepatol 31(1):133–137. https://doi.org/10.1111/jgh.13176
Sardi J, Casellas F, de Torres I, Malagelada JR (2000) [Clinical relevance of immunoglobulin A deficiency in celiac disease]. Med Clin (Barc) 115(18):687–689
Sinclair D, Saas M, Turk A, Goble M, Kerr D (2006) Do we need to measure total serum IgA to exclude IgA deficiency in coeliac disease? J Clin Pathol 59(7):736–739. https://doi.org/10.1136/jcp.2005.031864
Meini A, Pillan NM, Villanacci V, Monafo V, Ugazio AG, Plebani A (1996) Prevalence and diagnosis of celiac disease in IgA-deficient children. Ann Allergy Asthma Immunol 77(4):333–336. https://doi.org/10.1016/S1081-1206(10)63329-7
Kumar V, Jarzabek-Chorzelska M, Sulej J, Karnewska K, Farrell T, Jablonska S (2002) Celiac disease and immunoglobulin a deficiency: how effective are the serological methods of diagnosis? Clin Diagn Lab Immunol 9(6):1295–1300
Cataldo F, Lio D, Marino V, Scola L, Crivello A, Corazza GR, Working Group of the S, Working Group of ‘Club del T (2003) Plasma cytokine profiles in patients with celiac disease and selective IgA deficiency. Pediatr Allergy Immunol 14(4):320–324
Sebode M, Hartl J, Vergani D, Lohse AW, International Autoimmune Hepatitis G (2018) Autoimmune hepatitis: from current knowledge and clinical practice to future research agenda. Liver Int 38(1):15–22. https://doi.org/10.1111/liv.13458
Gronbaek L, Vilstrup H, Jepsen P (2014) Autoimmune hepatitis in Denmark: incidence, prevalence, prognosis, and causes of death. A nationwide registry-based cohort study. J Hepatol 60(3):612–617. https://doi.org/10.1016/j.jhep.2013.10.020
Yoshizawa K, Joshita S, Matsumoto A, Umemura T, Tanaka E, Morita S, Maejima T, Ota M (2016) Incidence and prevalence of autoimmune hepatitis in the Ueda area, Japan. Hepatol Res 46(9):878–883. https://doi.org/10.1111/hepr.12639
Kim BH, Choi HY, Ki M, Kim KA, Jang ES, Jeong SH (2017) Population-based prevalence, incidence, and disease burden of autoimmune hepatitis in South Korea. PLoS One 12(8):e0182391. https://doi.org/10.1371/journal.pone.0182391
Duchini A, McHutchison JG, Pockros PJ (2000) LKM-positive autoimmune hepatitis in the western United States: a case series. Am J Gastroenterol 95(11):3238–3241. https://doi.org/10.1111/j.1572-0241.2000.03207.x
Gregorio GV, Portmann B, Reid F, Donaldson PT, Doherty DG, McCartney M, Mowat AP, Vergani D, Mieli-Vergani G (1997) Autoimmune hepatitis in childhood: a 20-year experience. Hepatology 25(3):541–547. https://doi.org/10.1002/hep.510250308
Homberg JC, Abuaf N, Bernard O, Islam S, Alvarez F, Khalil SH, Poupon R, Darnis F, Levy VG, Grippon P et al (1987) Chronic active hepatitis associated with antiliver/kidney microsome antibody type 1: a second type of “autoimmune” hepatitis. Hepatology 7(6):1333–1339
Gossard AA, Gores GJ (2017) Primary sclerosing cholangitis: what the gastroenterologist and hepatologist needs to know. Clin Liver Dis 21(4):725–737. https://doi.org/10.1016/j.cld.2017.06.004
Hunter TM, Boytsov NN, Zhang X, Schroeder K, Michaud K, Araujo AB (2017) Prevalence of rheumatoid arthritis in the United States adult population in healthcare claims databases, 2004-2014. Rheumatol Int 37(9):1551–1557. https://doi.org/10.1007/s00296-017-3726-1
Badcock LJ, Clarke S, Jones PW, Dawes PT, Mattey DL (2003) Abnormal IgA levels in patients with rheumatoid arthritis. Ann Rheum Dis 62(1):83–84
Natvig JB, Harboe M, Fausa O, Tveit A (1971) Family studies in individuals with selective absence of gamma-A-globulin. Clin Exp Immunol 8(2):229–236
Thakar YS, Chande C, Dhanvijay AG, Pande S, Saoji AM (1997) Analysis of immunoglobulin deficiency cases: a five year study. Indian J Pathol Microbiol 40(3):309–313
Thierry S, Fautrel B, Lemelle I, Guillemin F (2014) Prevalence and incidence of juvenile idiopathic arthritis: a systematic review. Joint Bone Spine 81(2):112–117. https://doi.org/10.1016/j.jbspin.2013.09.003
Barkley DO, Hohermuth HJ, Howard A, Webster DB, Ansell BM (1979) IgA deficiency in juvenile chronic polyarthritis. J Rheumatol 6(2):219–224
Bluestone R, Goldberg LS, Katz RM, Marchesano JM, Calabro JJ (1970) Juvenile rheumatoid arthritis—a serologic survey of 200 consecutive patients. J Pediatr 77(1):98–102
Cassidy JT, Petty RE, Sullivan DB (1973) Abnormalities in the distribution of serum immunoglobulin concentrations in juvenile rheumatoid arthritis. J Clin Invest 52(8):1931–1936. https://doi.org/10.1172/JCI107377
Cassidy JTPR, Sullivan DB (1977) Occurrence of selective IgA deficiency in children with juvenile rheumatoid arthritis. Arthritis Rheum 20:181–183
Moradinejad MH, Rafati AH, Ardalan M, Rabiei M, Farghadan M, Ashtiani MT, Pourpak Z, Moin M (2011) Prevalence of IgA deficiency in children with juvenile rheumatoid arthritis. Iran J Allergy Asthma Immunol 10(1):35–40. https://doi.org/010.01/ijaai.3540
Panush RS, Bianco NE, Schur PH, Rocklin RE, David JR, Stillman JS (1972) Juvenile rheumatoid arthritis. Cellular hypersensitivity and selective IgA deficiency. Clin Exp Immunol 10(1):103–115
Pelkonen P, Savilahti E, Makela AL (1983) Persistent and transient IgA deficiency in juvenile rheumatoid arthritis. Scand J Rheumatol 12(3):273–279
Salmi TT, Schmidt E, Laaksonen AL, Anttila R, Kouvalainen K (1973) Levels of serum immunoglobulins in juvenile rheumatoid arthritis. Ann Clin Res 5(6):395–397
Dean LE, Jones GT, MacDonald AG, Downham C, Sturrock RD, Macfarlane GJ (2014) Global prevalence of ankylosing spondylitis. Rheumatology (Oxford) 53(4):650–657. https://doi.org/10.1093/rheumatology/ket387
Reynolds TL, Khan MA, van der Linden S, Cleveland RP (1991) Differences in HLA-B27 positive and negative patients with ankylosing spondylitis: study of clinical disease activity and concentrations of serum IgA, C reactive protein, and haptoglobin. Ann Rheum Dis 50(3):154–157
Herrero-Beaumont G, Armas JB, Elswood J, Will RK, Calin A (1990) Selective IgA deficiency and spondyloarthropathy: a distinct disease? Ann Rheum Dis 49(8):636–637
Lisak RP, Zweiman B (1976) Serum immunogloblin levels in myasthenia gravis, polymyositis, and dermatomyositis. J Neurol Neurosurg Psychiatry 39(1):34–37
Ezzedine K, Eleftheriadou V, Whitton M, van Geel N (2015) Vitiligo. Lancet 386(9988):74–84. https://doi.org/10.1016/S0140-6736(14)60763-7
Ali R, Ahsan MS, Azad MA, Ullah MA, Bari W, Islam SN, Yeasmin S, Hasnat A (2010) Immunoglobulin levels of vitiligo patients. Pak J Pharm Sci 23(1):97–102
Li Y, Yang M, Zhang R, Liu W, Zhang K, Wen W, Yi L, Wang Q, Hao M, Yang H, Chang J, Li J (2016) Evaluation of serum immunoglobulins concentrations and distributions in vitiligo patients. Immunol Res 64(5–6):1150–1156. https://doi.org/10.1007/s12026-016-8809-7
Parisi R, Symmons DP, Griffiths CE, Ashcroft DM, Identification, Management of P, Associated ComorbidiTy project t (2013) Global epidemiology of psoriasis: a systematic review of incidence and prevalence. J Invest Dermatol 133(2):377–385. https://doi.org/10.1038/jid.2012.339
Egeberg A, Skov L, Gislason GH, Thyssen JP, Mallbris L (2017) Incidence and prevalence of psoriasis in Denmark. Acta Derm Venereol 97(7):808–812. https://doi.org/10.2340/00015555-2672
Rachakonda TD, Schupp CW, Armstrong AW (2014) Psoriasis prevalence among adults in the United States. J Am Acad Dermatol 70(3):512–516. https://doi.org/10.1016/j.jaad.2013.11.013
Marrie RA, Patten SB, Tremlett H, Wolfson C, Leung S, Fisk JD (2017) Increased incidence and prevalence of psoriasis in multiple sclerosis. Mult Scler Relat Disord 13:81–86. https://doi.org/10.1016/j.msard.2017.02.012
Milcic D, Jankovic S, Vesic S, Milinkovic M, Marinkovic J, Cirkovic A, Jankovic J (2017) Prevalence of metabolic syndrome in patients with psoriasis: a hospital-based cross-sectional study. An Bras Dermatol 92(1):46–51. https://doi.org/10.1590/abd1806-4841.20175178
Eppinga H, Poortinga S, Thio HB, Nijsten TEC, Nuij V, van der Woude CJ, Vodegel RM, Fuhler GM, Peppelenbosch MP (2017) Prevalence and phenotype of concurrent psoriasis and inflammatory bowel disease. Inflamm Bowel Dis 23(10):1783–1789. https://doi.org/10.1097/MIB.0000000000001169
Ferreira BR, Pio-Abreu JL, Reis JP, Figueiredo A (2017) Analysis of the prevalence of mental disorders in psoriasis: the relevance of psychiatric assessment in dermatology. Psychiatr Danub 29(4):401–406. https://doi.org/10.24869/psyd.2017.401
van de Kerkhof PC, Steijlen PM (1995) IgA deficiency and psoriasis: relevance of IgA in the pathogenesis of psoriasis. Dermatology 191(1):46–48. https://doi.org/10.1159/000246487
Strazzulla LC, Wang EHC, Avila L, Lo Sicco K, Brinster N, Christiano AM, Shapiro J (2018) Alopecia areata: disease characteristics, clinical evaluation, and new perspectives on pathogenesis. J Am Acad Dermatol 78(1):1–12. https://doi.org/10.1016/j.jaad.2017.04.1141
Queiroz S, Silva M, Medeiros AMC, Oliveira PT, Gurgel BCV, Silveira E (2018) Recurrent aphthous ulceration: an epidemiological study of etiological factors, treatment and differential diagnosis. An Bras Dermatol 93(3):341–346. https://doi.org/10.1590/abd1806-4841.20186228
Gorouhi F, Davari P, Fazel N (2014) Cutaneous and mucosal lichen planus: a comprehensive review of clinical subtypes, risk factors, diagnosis, and prognosis. ScientificWorldJournal 2014:742826. https://doi.org/10.1155/2014/742826
Wetmore JB, Guo H, Liu J, Collins AJ, Gilbertson DT (2016) The incidence, prevalence, and outcomes of glomerulonephritis derived from a large retrospective analysis. Kidney Int 90(4):853–860. https://doi.org/10.1016/j.kint.2016.04.026
Binks S, Vincent A, Palace J (2016) Myasthenia gravis: a clinical-immunological update. J Neurol 263(4):826–834. https://doi.org/10.1007/s00415-015-7963-5
Behan PO, Simpson JA, Behan WM (1976) Letter: Decreased serum-IgA in myasthenia gravis. Lancet 1(7959):593–594
Bramis J, Sloane C, Papatestas AE, Genkins G, Aufses AH Jr (1976) Letter: Serum-IgA in myasthenia gravis. Lancet 1(7971):1243–1244
Bundey S, Doniach D, Soothill JF (1972) Immunological studies in patients with juvenile-onset myasthenia gravis and in their relatives. Clin Exp Immunol 11(3):321–332
Liblau R, Fischer AM, Shapiro DE, Morel E, Bach JF (1992) The frequency of selective IgA deficiency in myasthenia gravis. Neurology 42(3 Pt 1):516–518
Ramanujam R, Piehl F, Pirskanen R, Gregersen PK, Hammarstrom L (2011) Concomitant autoimmunity in myasthenia gravis—lack of association with IgA deficiency. J Neuroimmunol 236(1–2):118–122. https://doi.org/10.1016/j.jneuroim.2011.05.008
Liblau RS, Bach JF (1992) Selective IgA deficiency and autoimmunity. Int Arch Allergy Immunol 99(1):16–27. https://doi.org/10.1159/000236330
Iwasaki Y, Kinoshita M, Uchida A (1987) [A case of multiple sclerosis demonstrating ossification of anterior, posterior longitudinal ligament, thickening of the skull, and selective IgA deficiency]. Rinsho Shinkeigaku 27(9):1122–1126
Remolina Lopez AJ, Uribe Rueda C, Patrucco L, Rojas JI, Cristiano E (2011) Selective IgA deficiency and multiple sclerosis. Neurologia 26(6):375–377. https://doi.org/10.1016/j.nrl.2010.12.012
Pereira LF, Gomez M, Garcia Trujillo JA, Romero Chala S, Camara Hijon C (2012) [Selective immunoglobulin A deficiency is exceptionally associated with multiple sclerosis]. Neurologia 27(5):316. https://doi.org/10.1016/j.nrl.2011.08.004
Feudjo-Tepie MA, Robinson NJ, Bennett D (2008) Prevalence of diagnosed chronic immune thrombocytopenic purpura in the US: analysis of a large US claim database: a rebuttal. J Thromb Haemost 6(4):711–712; author reply 713. https://doi.org/10.1111/j.1538-7836.2008.02911.x
Cunningham-Rundles C (2002) Hematologic complications of primary immune deficiencies. Blood Rev 16(1):61–64. https://doi.org/10.1054/blre.2001.0185
Zanella A, Barcellini W (2014) Treatment of autoimmune hemolytic anemias. Haematologica 99(10):1547–1554. https://doi.org/10.3324/haematol.2014.114561
Aladjidi N, Leverger G, Leblanc T, Picat MQ, Michel G, Bertrand Y, Bader-Meunier B, Robert A, Nelken B, Gandemer V, Savel H, Stephan JL, Fouyssac F, Jeanpetit J, Thomas C, Rohrlich P, Baruchel A, Fischer A, Chene G, Perel Y, Centre de Reference National des Cytopenies Auto-immunes de lE (2011) New insights into childhood autoimmune hemolytic anemia: a French national observational study of 265 children. Haematologica 96(5):655–663. https://doi.org/10.3324/haematol.2010.036053
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflicts of interest.
Ethical Approval
This review was performed according to applicable ethical guidelines, using previously published and publicly available data.
Informed Consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
ESM 1
(XLSX 26 kb)
Rights and permissions
About this article
Cite this article
Odineal, D.D., Gershwin, M.E. The Epidemiology and Clinical Manifestations of Autoimmunity in Selective IgA Deficiency. Clinic Rev Allerg Immunol 58, 107–133 (2020). https://doi.org/10.1007/s12016-019-08756-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12016-019-08756-7