Abstract
Injury to the peripheral nerve causes potential loss of sensory and motor functions, and peripheral nerve repair (PNR) remains a challenging endeavor. The current clinical methods of nerve repair, such as direct suture, autografts, and acellular nerve grafts (ANGs), exhibit their respective disadvantages like nerve tension, donor site morbidity, size mismatch, and immunogenicity. Even though commercially available nerve guidance conduits (NGCs) have demonstrated some clinical successes, the overall clinical outcome is still suboptimal, especially for nerve injuries with a large gap (≥ 3 cm) due to the lack of biologics. In the last two decades, the combination of advanced tissue engineering technologies, stem cell biology, and biomaterial science has significantly advanced the generation of a new generation of NGCs incorporated with biological factors or supportive cells, including mesenchymal stem cells (MSCs), which hold great promise to enhance peripheral nerve repair/regeneration (PNR). Orofacial MSCs are emerging as a unique source of MSCs for PNR due to their neural crest-origin and easy accessibility. In this narrative review, we have provided an update on the pathophysiology of peripheral nerve injury and the properties and biological functions of orofacial MSCs. Then we have highlighted the application of orofacial MSCs in tissue engineering nerve guidance for PNR in various preclinical models and the potential challenges and future directions in this field.
Graphical Abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Peripheral nerve injury (PNI) quickly becomes permanent if immediate action for repair is not taken, occurring in around 2.8% of trauma patients in which the natural peripheral nerve repair (PNR) process exhibits poor results [9, 111]. According to the statistics from the whole year of 2014, the average cost of nerve injury repair for patients is about $5779 US dollars, with digital nerve damage being the most expensive at an average of $8931 US dollars [105]. Therefore, due to the long-term disability, subsequent sick-leave and permanent disability pension, PNI has a significant impact on health care costs.
The type and severity of PNI usually determine the treatment modalities, either non-surgical or surgical treatment. A mild injury, such as neurapraxia, may be able to heal naturally or completely be treated by non-operative means, while neurotmesis and the resulting nerve gap usually requires surgical techniques to repair via direct end-to-end coaptation or bridging the gap defect with nerve grafts [42]. Currently, nerve autograft remains to be the “gold-standard care” for severe PNI treatment because it provides native ECM, autologous Schwann cells and growth factors to promote and sustain nerve regeneration [85]. However, several disadvantages exist for nerve autografts, including limited availability, mismatch of the donor nerve, donor site morbidity and possible complications like neuroma. Therefore, there is an increasing clinical demand of generation and commercialization of acellularized allografts and tissue-engineered nerve guidance conduits (NGCs) [76, 85, 110].
In recent years, the progress in stem cell biology, biomaterial science, and tissue engineering technologies have shed light on the development of cell-based NGCs as potential alternatives for nerve autografts. Due to their critical roles in nerve regeneration, primary Schwann cells harvested from nerve tissues are considered the optimal source of supporting cells for generation of engineered NGCs. However, harvesting, isolating, and expanding Schwann cells face similar challenges and shortcomings as harvesting nerve autografts [8, 35, 99], Mesenchymal stem/stromal cells (MSCs), a unique subpopulation of adult stem cells, have been identified and isolated from almost all tissues of the body, e.g. bone marrow, adipose and umbilical cord tissues, etc., and are emerging as a good source of stem cells for nerve regeneration due to their self-renewal, multipotent differentiation, and immunomodulatory capabilities [6, 7, 12, 35]. MSC have also been identified and isolated from various dental and orofacial tissues, such as dental pulp (DPSC), periodontal ligament (PDLSC), gingiva/oral mucosa (GMSC), and dental follicle (DFSC). These orofacial/dental MSCs possess similar phenotypical and functional properties, e. g. self-renewal, multipotency and immunomodulatory effects, to other sources of MSCs, but have a unique neural crest origin and propensity to differentiate into neural cells, particularly, Schwann-like cells, making them a superior source of MSCs for nerve regeneration [64, 112, 113]. To date, the incorporation of MSCs in various types of bio-engineered tissue and scaffolds has shown great promise for the development of the new generation of functionalized NGCs with potential application in clinic [16, 22, 36]. In this review article, we focus on the recent progress in orofacial MSC-based tissue engineering for PNR.
Anatomy and Physiology
The examination of the anatomy and physiology of the peripheral nerve will show the parallel between its structure and the layers within an ideal conduit design and demonstrate the necessity for biomimetics (Fig. 1).
Axon
The axon conducts signals from the cell body to the axon terminal, the signal traveling to the next neuron’s dendrites or the target organ’s effector cells via neurotransmitters. Axons are unmyelinated in the CNS and myelinated in the PNS, each myelin sheath separated by a node of Ranvier for conducting saltatory conduction, allowing nerve impulses to jump from one node of Ranvier to the next [17].
Endoneurium
Surrounding the axons is the endoneurium which contains micro-capillaries and vessel networks to provide nutrients to the cells inside, and this layer is fragile and susceptible to trauma damage [59]. The endoneurium supports the Schwann cells (SC) that produces the myelin sheath, degrades the degenerated axons, cells, and debris during PNR, and create the “track” called the Bands of Büngner for the regenerating axon to grow along [40, 67].
Perineurium and the Fascicles
Made of layers of perineurial cells and collagen, the perineurium maintains endoneurial fluid homeostasis, the fluid surrounding the myelinated and unmyelinated nerve fibers inside [100]. The fluid is also a blood-nerve barrier and gives the nerve a degree of elasticity with the endoneurium [78]. The perineurium surrounds the fascicles which are bundles composed of axons, Schwann cells, endoneurium, and related cells and structures [78]. These bundles form the fascicular plexus that rapidly changes in the cross-sectional topography traveling along the nerve due to division and union of fascicles [51]. Because of the frequently changing topography, fascicular structure becomes critical in peripheral nerve biomimetics. Techniques such as micro-CT and micro-MRI create 3D images of the damaged nerve to provide a guide for accurate nerve conduit designs, streamlining the method for creating NGCs with high-resolution and accurate fascicular matching [118, 119, 133].
Epineurium
The epineurium is the most outer layer of the peripheral nerve, made of two layers: the inner epifascicular epineurium, the loose connective tissue layer that directly surrounds the fascicles, and the outer epineurial epineurium, the outside sheath [100]. The epineurium has nutrient blood vessels on the surface and serves as a crucial shock absorber for the nerve to dissipate the forces of impact [102].
Pathophysiology of Peripheral Nerve Injury
The Sunderland Classification ranks injuries from minor to major as 1st (neuropraxia) to 5th degree (complete neurotmesis) [4]. A detailed display is shown in Fig. 2 (Fig. 2).
A schematic representation of increasing levels in severity of peripheral nerve injury (PNI). The Sunderland Classification (in red) increases from level I to V, level I as a minor, recoverable injury to V as a complete sever of the peripheral nerve that requires surgical intervention. The Seddon Classification (bottom right) classifies PNI into three classes: neurapraxia, axonotmesis, and neurotmesis
Injuries limited to the axon alone are usually self-repaired due to the presence of intact endoneurium, perineurium, and epineurium serving as the architecture for PNR guidance [78]. Compression injury results in neurapraxia, the 1st degree of injury characterized by focal demyelination or ischemia resulting from mild compression and nerve traction [10]. Thus, the recovery is expected to be via spontaneous myelination [5].
Axonotmesis, the 2nd degree of injury, results in damages to the axon itself (example: severing of the axon), requiring axonal regeneration to replace the damaged sections [102]. Invasive surgery may not be required for 1st and 2nd degree due to the preservation of the architecture for natural nerve regeneration guidance [78].
A 3rd degree of injury represents a more severe type with the endoneurium damaged, whereby direct repair may be difficult. If there is a gap present in the injury site, direct repairing can cause unwanted tension that can disrupt capillaries to reduce axonal function. The possibility of tension-derived devascularization makes nerve graft a sound alternative [31].
Type 4 injury involves damage to the perineurium along with the fascicle, endoneurium, and axons that is more difficult to treat, requiring invasive surgery to remove scar tissue in the injury area [112, 113]. The difficulty lies in the diagnosis as well, involving a “wait-and-see period” over 3 months when signs of natural injury repair may show in Type 2 and 3 injuries. If the natural repair does not show, then the injury is likely type 4 or above, but the long “wait-and-see period” may have decreased the chances of rescuing the damaged nerves [78]. Invasive exploration could damage the nerve even further.
Epineurium damage indicates the most severe type of nerve injury, classified as a 5th degree of injury called neurotmesis. The epineurial suture physically connects the severed peripheral nerve by aligning the blood vessels that are visible on the surface. Although this procedure is minimally invasive to the nerve itself, it may not connect the severed inner fascicles, which are important for the guidance of successful axon repair [4]. Tension of the sutured epineurial nerve in large gaps is a big drawback due to the resulting scarring that can block the regeneration occurring at the injury site. To repair these larger gaps, nerve grafts or synthesized/engineered nerve guidance conduits (NGCs) are appropriate options [9].
A 6th degree nerve injury involves a combination of different degrees of nerve injury together on a continuous nerve, which may require microsurgery to repair [4].
Natural Nerve Repair: What Happens?
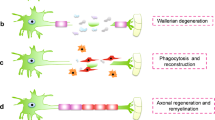
The natural nerve repair process includes three overlapped phases: Wallerian degeneration (the clearance of the myelin sheath debris), axonal regeneration, and end-organ reinnervation.
After axons are heavily injured, the axonal cytoskeleton and the neurofilaments all degrade. This massive amount of degeneration sends out signals in response to the damage, to alter the glial cell behavior and recruit macrophages to the injury site [30]. Debris from myelin and axonal breakdown can serve as axon regrowth blockers and axonal growth inhibitory signals [13]. Upon injury, Schwann cells at the site undergo demyelination/dedifferentiation and acquire repair phenotypes characterized by increased proliferation, migration, and secretion of various factors to recruit macrophages that help with the clearance of debris through phagocytosis [134]. The resident macrophages also engage in the early phagocytosis of the debris, and then both types of macrophages polarize into M1 proinflammatory phenotype contributing to acute inflammatory response at the injury site [18, 134]. Later, macrophages transform to the M2 phenotype to release soluble factors contributing to axonal regeneration and remyelination process. The modulation of the macrophages throughout the entire repair process involves the regulation of their inflammatory and anti-inflammatory responses [134].
After successful debris clearance, the Schwann cells migrate to form the Bands of Bunger, the injured axon creates a growth cone to begin PNR. Depending on the amount of time has passed since the injury, the microenvironment that supports the regeneration, and the distance the axon has to traverse, axons may successfully reconnect with the other axonal stump across the injury site, completing the regeneration process Without the continual environmental support across the gap and the correct formation of the bands of Bunger, the process of regeneration is slow, and the chance of failure remains high [29, 89]. This depends on the type of injury as well: types 1 and 2 both have endoneurium, perineurium and epineurium intact, which can provide that structure for regeneration. Types 3 or above results in damages to any of the three layers, which lowers the probability of successful PNR [112, 113].
Orofacial Mesenchymal Stem Cells
Mesenchymal Stem Cells
In the last two decades, much progress has been made in the development of various types of biomimetic NGCs by incorporating various types of growth factors and/or supporting cells for the repair/regeneration of severe PNIs with a large gap [106]. Due to their potent self-renewal, multipotent differentiation, and immunomodulatory capabilities, MSCs, particularly, orofacial tissue-derived MSCs, represent a good candidate source of supporting cells for generating the new generation of functionalized NGCs [50, 109].
The first type of MSCs to be found was bone marrow derived MSCs (BMSCs), which was initially described by Friedenstein et al. as the stromal cells characterized by self-renewal or fibroblast-like colony-forming units (CFU-F) [26]. Since then, a similar subpopulation of MSCs has been identified and isolated from various parts of the body, e. g. adipose and skin etc. [7, 12]. Other sources of MSCs include peripheral blood, muscles, and perinatal and fetal sources like amniotic fluid, umbilical cord, and placental MSCs [6]. The minimum criteria to define MSCs set by the International Society for Cellular Therapy (ISCT) included: 1) the expression of characteristic surface markers like cluster of differentiation (CD)73, CD90, CD105 should be in more than 95% of the population, while less than 2% of the population is positive for the hematopoietic markers, CD14, CD34, CD45, CD79α, CD19, and HLA class II [23, 74]; 2) Plastic adherence and forming fibroblast-like colony unit (CFU-F); 3) multi-lineage differentiation potential into mesodermal cell lineages, e. g. osteocytes, chondrocytes, and adipocytes [20, 23]. In addition, MSCs should also be very flexible in converting from quiescence to proliferation and reversing differentiation [12]. MSCs of different origins may exhibit some different characteristics. For example, BMSCs can not only differentiate into mesodermal cell lineages (adipogenic, osteogenic, chondrogenic) but also other types of cells, e. g. vascular smooth muscle cells, tenocytes, and even cells of neuro-ectodermal origins [12]. In addition to these properties, it is commonly observed that MSCs from different tissues are immunotolerant and have potent pleiotropic, immunomodulatory, and anti-inflammatory functions through their paracrine secretion of a myriad of bioactive factors, making them good candidates in the application in tissue engineering/regenerative medicine (TE/RM) [47].
Orofacial MSC: A Unique Cell Population with a Neural Crest-Origin
Origin and Characteristics
Up to date, several distinct types of MSCs have been identified and isolated from human orofacial tissues, such as dental pulp (DPSCs) [32], exfoliated deciduous teeth (SHED) [70], gingiva (GMSCs) [129], apical papilla (SCAPs) [98], periodontal ligament (PDLSCs) [91], dental follicle (DFSCs) [72], and tooth germ (TGSCs) [114]. Similar to other sources of MSCs, orofacial MSCs also possess multipotent differentiation and immunomodulatory capabilities and express a panel of MSC-related cells surface markers, e. g. CD90, CD146, CD73, and STRO-1 [91], but lack the expression of hematopoietic cell markers, CD45, CD14, and CD34 [23, 32]. Embryonically, neural crest (NC) composed of a unique population of ectomesenchymal progenitor cells gives rise to various types of body tissues, including most of orofacial and cranial tissues [64]. In recent years, a unique population of MSCs has been isolated and characterized from different NC-derived tissues, called NC-derived MSCs [95]. It has been well recognized that orofacial MSCs share a common embryonic neural crest-origin and maintain certain properties of neural cells, making orofacial tissues a reservoir of NC-derived MSCs [112, 113] (Fig. 3).
A schematic of the origins of orofacial mesenchymal stem cells (orofacial MSCs) and their neurogenic capabilities. a The developing nervous system of the embryo. b The formation of the neural tube releases migrating neural crest cells. c neural crest cells proceed to various differentiation lineages, including neural differentiation, which develops into peripheral glial cells and neurons, and odontogenic differentiation, which develops into dentition. d The stages of tooth development. e The various types of orofacial MSCs found in the teeth at different stages of development. f Orofacial MSC characterization and markers of neurogenic capabilities
Neural Differentiation of Orofacial MSCs
Compared to mesoderm-derived MSCs, orofacial MSCs constitutively express a relatively high level of neural progenitor markers and neurotrophic factors, such as Nestin, S100-beta, GFAP, BDNF, and GDNF, even under standard culture conditions [32, 38, 70], and possess intrinsic ability to differentiate towards neural types of cells due to their NC-origins [1, 52, 112, 113, 132]. For instance, DPSCs can directly differentiate into neural cells evidenced by the expression of neurogenic cell markers and NGF [108]. GMSCs have been shown to express Nestin in addition to other neural crest stem cell (NCSC)-related genes like Twist1, Pax3, Sox9, and FoxD3 [25]. PDLSCs and TGSCs are shown to express Nestin and β3-tubulin, suggesting that both of them have characteristics of neuro-progenitor cells and immature neurons [24, 43]. Under 3D-neural sphere culture conditions, most of the orofacial MSCs, including DPSC, SHED, GMSCs. SCAPs, PDLSCs, and DFSCs, can aggregate into 3D spheroids with increased expression of neural crest cell markers, e. g. nestin, GFAP, CD271 and SOX-10, and a strong differentiation propensity towards neurons and glial cells [46, 71, 79, 94, 126,127,128]. The neural differentiation capacity of TGSCs has been shown to be unwavering in differentiation quality after cryopreservation, able to retain its level of pluripotency associated gene expression (i.e. Nanog, Oct4, Sox2) [114]. Taken together, these studies support the notion that orofacial MSCs of a NC-origin represent a superior source of stem cells for cell-based regenerative therapy of PNI due to their intrinsic neurogenic potentials [80, 112, 113].
Immunomodulatory Function of Orofacial MSCs
In addition to their multipotent differentiation capabilities, MSCs derived from different tissues, including orofacial MSCs, possess potent anti-inflammatory and immunomodulatory effects on both innate and adaptive immune cells through direct cell–cell contact or their secretion of various soluble bioactive factors [107]. Upon the activation of Toll-like receptors (TLR), DPSCs exert the immunosuppressive effects on lymphocytes by upregulating the production of anti-inflammatory cytokines such as IL-10 and TGF-β1 [107]. They also recruit monocytes and skew their differentiation into anti-inflammatory M2 macrophages and cause apoptosis in activated T-cells via cell-to-cell interactions [63]. It has been shown that SHEDs can directly interact and inhibit CD4+ T helper (Th) cells, including T helper 17 (Th17) cells that produces inflammatory cytokine IL-17, but promote the generation and function of anti-inflammatory regulatory T cells (Tregs) [115]. SHED can directly promote the M2 macrophage polarization or the shift of M1 to M2 macrophages, and/or directly suppress M1 macrophage activation [27, 54]. It has been reported that DFSCs promote M2 macrophage polarization through the secretion of thrombospondin-1 (TSP-1) and TGF-B3 [14]. Similarly, GMSCs, SCAPS, and PDLSCs have been shown to suppress lymphocyte activation or converting pro-inflammatory T cells to anti-inflammatory Tregs through INF-γ-induced upregulation of indoleamine 2,3-dioxygenase (IDO) and TGF-β [21, 58, 83, 115, 129]. Zhang et al. reported that GMSCs potently suppress M1 macrophage activation and promote M2 polarization partially through the secretion of soluble factors, IL-6 and granulocyte macrophage colony-stimulating factor (GM-CSF) [130]. Taken together, these studies have demonstrated that orofacial MSCs possess potent anti-inflammatory and immunomodulatory functions on both innate and adaptive immune cells, which may add another layer of beneficial effects on repair/regeneration of PNIs [92].
Harnessing Extracellular Vesicles (EVs) of Orofacial MSCs for Nerve Regeneration
It has been well recognized that MSCs, including orofacial MSCs, exert their therapeutic effects mainly through their secretomes composed of a spectrum of soluble bioactive factors such as cytokines, growth factors, and extracellular vesicles (EVs) or exosomes, which exert immunomodulatory/anti-inflammatory, anti-apoptotic, anti-oxidant, anti-fibrotic, proangiogenic, and pro-regenerative functions. Due to the relative stability, safety, and equivalent biological effects, the use of MSC secretome can circumvent the drawbacks of the administration of viable cells, thus considered as a new paradigm for the development of cell-free therapeutics in TE/RM [48], including in regenerative therapy of PNI. The conditioned media from SHED and DPSC contained a variety of neurogenic factors, such as NGF, BDNF, CNTF, and VEGF, showing the possibility to use the secretomes of orofacial MSCs instead of cells themselves for regenerative therapy of PNI [101, 112, 113]. Interestingly, a comparative study showed that SCAP-derived secretomes had stronger neuroregenerative capabilities, while BMSC-derived secretomes possessed stronger proangiogenic capabilities, suggesting that SCAPs do have a stronger pro-regenerative effect on PNI than that of BMSCs in a comparable environment [121].
EVs or exosomes are nano-sized vesicles enclosed by a lipid bilayer membrane and loaded with cargos containing various types of bioactive factors such as lipids, DNA, miRNA, non-coding RNA, cytokines, and growth factors, etc., which play critical roles in cell–cell communications or interactions through the horizontal transfer of these bioactive cargo contents [81]. Accumulating evidence supports that EVs contribute to orofacial MSC-mediated therapeutic effects in different preclinical disease models, including PNIs [54]. For instance, it has been reported that GMSC-derived EVs can promote proliferation and migration and the expression of repair phenotype-related genes in cultured Schwann cells and facilitate functional recovery of crush-injured sciatic nerves of mice [62]. Rao F et al. also reported that GMSC-EVs could significantly promote Schwann cell proliferation and the outgrowth of ex vivo cultured DRGs [84]. In vivo, they demonstrated that GMSC-EVs implanted with chitin conduit significantly promoted axonal regeneration, remyelination, and functional recovery in a rat sciatic nerve defect model [84]. Meanwhile, Shi Q et al. reported that application of a chitosan/silk hydrogel sponge loaded with GMSC-derived EVs promotes skin wound healing in a diabetic rat model through multiple mechanisms, including enhancing neuronal outgrowth in skin [93]. In addition, a recent study showed that DPSC-derived exosomes can inhibit activation of M1 macrophages and exert therapeutic effects on spinal cord injury (SCI) [56, 57]. Taken together, these findings have shed light on the application of orofacial MSC-derived secretomes, particularly EVs/exosomes, as cell-free platforms for neuroprotective treatment or regenerative therapy of PNIs, including the development of bioengineered nerve guidance conduits (NGCs) laden with orofacial MSCs or their cell-free EVs [33] (Fig. 4).
A diagram of the use of orofacial MSCs for peripheral nerve repair (PNR). Orofacial MSCs can differentiate into neural-like cells (neuron-like, neural progenitor-like, or Schwann-like cells), produce conditioned medium (CM) containing extracellular vesicles and bioactive factors with immunomodulatory and/or pro-nerve regenerative potentials, or be applied directly into the injury site alone or in combination with a scaffold. The bottom represents the two types of orofacial MSC-facilitated PNR by stem cells or their secretomes
Bioengineering Orofacial MSC-laden Nerve Guidance Conduits (NGC)
Due to their potent neurogenic and immunomodulatory capabilities through the production of secretomes composed of extracellular vesicles/exosomes and various soluble bioactive factors, orofacial MSCs represent excellent sources of MSCs for the development of the next generation of cell-based NGCs as potential alternatives of nerve autografts to facilitate for PNR.
Progress in the Construction of NGCs
As a common surgical treatment method, direct suturing is a relatively simple procedure for nerve injuries with a small gap (< 1.0 cm) where two ends of the injured nerve are connected to allow for natural regeneration. However, for nerve injuries with a large gap (≥ 2 cm), the tension presented at the direct suturing site often leads to poor clinical outcomes [110]. Nerve autografts and allografts have been shown to yield good clinical outcomes for PNR of large gap nerve injuries because of the existing native scaffold and biological elements. However, nerve autografts, the current “gold standard” for PNR, can cause donor site morbidity due to excision of a nerve from another part of the body, risk of infection due to multiple invasive surgeries required, and size mismatching when donor nerves of a different size is transplanted [49]. Nerve allografts have similar disadvantages, including size mismatching and immunogenicity, although the latter can be mitigated via decellularized nerve grafts [110, 116]. The scarcity of implantable allografts for PNR presents another dimension of disadvantages that can make procedures and clinical care less efficient than optimal [116].
To circumvent the disadvantages of nerve grafts, about twenty different types of NGCs have been developed and commercially available [77]. NGCs are any types of structures made of natural or synthetic materials or a combination of both and capable of providing mechanical and biochemical support to facilitate PNR [44, 110]. NGCs are applied to connect the proximal and distal ends of the injured nerves, thus providing a “bridge” or “channel” to guide the outgrowing axons and prevent the adverse influence from outside the injury area, such as the surrounding tissues or cells which can invade the space [44]. Generally, an ideal NGCs should meet the following criteria: 1) Biocompatible with minimal host immune responses; 2) Biodegradable but with an optimal degrading rate to provide sufficient time for axon growth guidance; 3) Optimal mechanical properties (e.g., permeability, tensile strength, elasticity, compression resistance, inner wall stability, biomimetic architecture) [44, 86, 110, 116]. Therefore, NGCs have demonstrated the benefits of low immunogenicity thus lower risk of complications during and after PNR, often attributed to the materials that allow for biocompatibility, biodegradability, and structural integrity [44, 86, 110, 116] (Fig. 5).
A diagram of peripheral nerve repair methods. a Direct repair is a simple method but requires tensionless repair. b Biological grafts, including autograft and acellular nerve graft (ANG), demonstrate disadvantages of donor site morbidity, size mismatch, and complication risks. ANGs, however, can be repopulated with orofacial MSCs to become functionalized. c Bio-engineered nerve grafts incorporate orofacial MSCs or orofacial MSCs secretomes. Cells can be filled into the graft during transplant or incorporated into tissue-engineered nerve graft (TENG) before transplant. d A comparison of the process of using autograft or TENG for PNR
A representative of the first-generation of NGCs is a hollow nerve tube made of silicone in 1982 [60], which possess a favorable structural strength and can provide the space and area for the growing axons. However, obvious disadvantages exist for such a type of NGCs, e. g. chronic nerve compression, neuroma formation, nerve pain, and the requirement of a “follow-up” surgery to remove the tube because of their non-biodegradable property [36, 65]. In the last two decades, much progress has been made in the construction of NGCs with various types of biocompatible and biodegradable synthetic materials, such as poly (lactic acid) (PLA), poly(lactic-co-glycolic acid) (PLGA), poly(ε-caprolactone) (PCL), and poly(glycolide-co-caprolactone) (PGC), allowing for the avoidance of a “follow-up” surgery after PNR [36, 110]. In addition, NGCs made of these synthetic materials can maintain the needed structural strength while being biodegradable over time, suitable for controlled release of compounds, cell products, and factors for promoting PNR [36]. Natural polymers or scaffolds, such as collagen, chitosan, gelatin, cellulose, silk, hyaluronic acid, alginate, and laminin, have a common advantage of high biocompatibility, thus compatible for the use with cells and tissues. However, natural biomaterials usually lack the structural stability, so they are frequently combined with synthetic materials to balance the biocompatibility, structural stability, and biodegradability for the construction of the second-generation of NGCs [77].
In the last decade, the advancement in tissue engineering technology and stem cell biology has significantly advanced the fabrication of the third generation of functionalized NGCs loaded with biological factors and/or supportive cells, particularly, MSCs [77]. Even though there are still no FDA-approved supplemental biological factors nor cells loaded with NGCs for PNR, accumulating preclinical studies have observed significant beneficial effects of supplemental bioactive factors or MSCs in facilitating PNR when they are combined with NGC implantation. Generally, supportive cells can be combined with NGCs through various approaches, including but not limited to random or organized encapsulation in different carriers (e. g. hydrogels) as the filler of NGCs, even coating on the inner NGC surface, or incorporation into the wall matrix of NGCs [77]. The incorporated supportive MSCs improve the functional ability of NGCs through multiple mechanisms, such as their differentiation capability into Schwann-like cells and their immunomodulatory and neurogenic effects via their paracrine secretion of a variety of bioactive factors, thus supporting and maintaining a regenerative microenvironment to facilitate axon growth, protect neurons from further damage, promote neovascularization, suppress inflammation and fibrosis, all of which can contribute to improving clinical outcomes [11, 120].
Application of Orofacial MSCs Combined with NGCs
In the last decades, MSCs of different origins, including orofacial MSCs, particularly DPSCs and GMSCs, have been widely explored to be used in combination with NGCs through different approaches with significantly improved beneficial effects on PNR. Herein, we focus on how orofacial MSCs are combined with NGCs to facilitate PNR (Table 1).
Orofacial MSCs Filled within NGCs
Various studies have demonstrated that orofacial MSCs are compatible and functional when they were premixed in either culture medium or different types of hydrogels and then directly filled into the tube of NGCs. Beigi MH et al. have reported the fabrication of nanofibrous NGCs made of electrospun poly(ε-caprolactone)/gelatin, which were subsequently filled with 3 µL culture medium containing 1 × 105 SHED cells. In a sciatic nerve defect (10-mm) model in rats, implantation of nanofibrous NGCs seeded with SHED significantly support axonal regeneration [3]. Recently, Luo L et al. have fabricated a cellulose/soy protein isolate composite membrane (CSM) conduit filled with bioactive GelMA hydrogels encapsulated with recombinant human basic fibroblast growth factor and DPSCs (CSM-GFD conduits) [61]. In a long sciatic nerve defect model (15-mm) in rats, they have demonstrated that implantation of CSM-GFD conduits significantly promoted axonal regeneration, remyelination, and functional recovery, which are comparable to the therapeutic effect achieved by implantation of nerve autografts. Interestingly, they have shown that almost all newly formed nerve tissue at defect site was originated from the direct differentiation of exogeneous DPSCs in CSM-GFD [61].
Using a popular facial nerve defect model in rats, whereby a 7-mm gap is created in the buccal branch of the facial nerve, Sasaki R et al. have demonstrated that implantation of a degradable poly-DL-lactide-co-glycolide (PLGA) conduit filled with 10 µl type I collagen solution mixed with 5 × 105 DPSCs promoted axonal regeneration and remyelination, while the PLGA conduits were readily resorbed 2 months after the implantation [88]. In a facial nerve defect (7-mm) model in rabbits, implantation of chitosan tubes filled with 40 µL of DPSC suspension (3 × 106/each) supplemented with stem cell factor (SCF) significantly promote axonal regeneration, remyelination, functional recovery, and angiogenesis compared with implantation with chitosan tubes alone [73].
In addition to non-differentiated orofacial MSCs, neural types of cells pre-differentiated from orofacial MSCs have also been explored as supportive cells in combination with NGCs for PNR. A study by Takaoka S et al display owed that implantation of NGCs (composite seamless tube; Atree, Tokyo, Japan) filled 1 × 105 neural lineage cells (NLCs) differentiated from DPSCs mixed in Matrigel to bridge a 10-mm sciatic nerve defect in immunodeficient rats displays enhanced beneficial effects on axon growth, remyelination, electrophysiological activities, and muscle atrophy improvement compared with NGCs filled with Matrigel only. Interestingly, they found that numerous transplanted DPSC-derived NLCs differentiated into platelet-derived growth factor receptor alpha (PDGFRα +) oligodendrocyte progenitor cells (OPCs) and a few PDGFRα + /p75 + Schwann cell-like cells at 2 weeks post-transplantation [103]. A research group has differentiated hDPSCs into neuronal or cholinergic neuronal cells (DF-chN), mixed them with 0.1 mL of fibrin glue (1 × 106), and then filled the mixture into the biodegradable bovine collagen dura mater (Lyoplant®). At 8 weeks following implantation of the NGCs filled with DF-chNs into the transected sciatic nerve defect of rats, enhanced motor nerve regeneration as evidenced by a notable increase in behavioral activities detected with an open-field test [39]. Recently, we showed that GMSCs could be readily converted into neural crest stem-like cells (NCSC) under defined cultured conditions without introduction of exogeneous gene expressions. Implantation of Axoguard nerve conduits filled with GMSC-derived NCSCs mixed in Matrigel to a 7-mm facial nerve defect in rats significantly promoted axonal regeneration, remyelination, and the functional recovery [127, 128].
Orofacial MSCs-Repopulated Acellular Nerve Grafts (ANGs)
Acellular nerve grafts (ANGs) can provide native nerve scaffolds and certain types of neurotrophic factors, thus they serve as “alternative natural NGCs” for PNR. Studies have shown that ANGs repopulated with MSCs can improve their functions to promote PNR. Recently, a study by Qiao W et al. fabricated xenogenic ANGs harvested from the sciatic nerves of SD rats and then repopulated the ANGs by microinjection of a total volume of 30 μl of allogenic rat DPSC cell suspension (6 × 105 cells per graft). ANGs loaded with DPSCs (ANG-DPSCs) were further cultured in complete culture medium for three days before they were implanted to bridge a 10-mm sciatic nerve gap defect in New Zealand White Rabbits. They found that ANGs loaded with DPSCs significantly facilitated the nerve regeneration and functional recovery compared with an ANG without DPSCs and allografts in rabbits as determined by electrophysiological and histological analysis [82].
NGCs Loaded with Orofacial MSCs-Derived Secretomes
To date, it has been well accepted that the therapeutic and regenerative actions of MSCs, including orofacial MSCs, are attributed, at least in part, to their secretomes containing all the bioactive molecules released in the conditioned medium (CM) or in extracellular vesicles (EVs)/exosomes [33]. Several studies have explored the combined use of NGCs and orofacial MSC-derived conditioned medium (CM) or EVs in PNR. For instance, Sugimura-Wakayama Y et al. have shown that implantation of silicone conduits loaded with SHED-CM significantly enhanced axonal regeneration, remyelination, and functional recovery in a 10-mm sciatic nerve gap model in rats compared with implantation of the empty conduits [101]. Recently, Rao F et al. have explored the combined use of the biodegradable chitin conduit and GMSC-derived exosomes to repair rat sciatic nerve defect. They found that GMSC-exosomes significantly promote Schwann cells proliferation and DRG axon growth in vitro. In a 10-mm sciatic nerve gap model in rats, implantation of the hollow chitin conduits loaded with 10 µg of GMSC-exosomes significantly increased the number and diameter of nerve fibers and myelin formation along with a significant improvement in muscle function, nerve conduction function, and motor function compared with animals receiving implantation of empty conduits [84]. These studies have shed light on the application of orofacial MSC-derived secretomes as cell-free products in combination with NGCs for novel therapeutic intervention in PNR.
Fabrication of Orofacial MSC-Laden Tissue Engineered Nerve Grafts (TENGS) or Engineered Neural Tissues (EngNT) for PNR
In recent years, several studies have demonstrated the feasibility to fabricate MSC-laden tissue engineered nerve grafts (TENGS) or engineered neural tissues (EngNT) to facilitate PNR [37]. For instance, different types of allogeneic supportive cells, including Schwann cells (SCs), adipose-derived adult stem cells (ASCs), dental pulp stem cells (DPSCs), or their combinations (1 × 106 cells/graft), were filled into PLA nerve conduits and dynamically cultured at 37 °C on a roller for three days, allowing the uniform attachment of cells on the inner surface of the NGCs [15]. Overall, cell-laden PLA NGCs were superior to the cell-free NGCs in term of facilitating nerve regeneration after they were implanted to bridge a 15-mm long sciatic nerve gap defect of rats, whereby the group of animals implanted with NGCs seeded with a combination of SCs and ASCs showed the greatest axonal regeneration and functional recovery while rats receiving NGCs seeded with SCs and DPSCs had better values in term of NCV and neovascularization than those seeded with DPSCs only [15].
A previous study has demonstrated the generation of EngNT laden with differentiated ADSCs (EngNT-dADSCs), which promoted nerve regeneration when implanted as the filler within NeuraWrap conduits [28]. Using the similar approach, Sanen K et al. fabricated a tethered type-1 collagen gel mixed with Schwann cells differentiated from hDPSCs in which the cells underwent self-alignment, followed by stabilization to generate 40-μm-thick sheets of EngNT d-hDPSCs [87]. The EngNT-d-hDPSC sheets were rolled up to form rods (~ 200 μm in diameter × 15 mm in length), two of which were used as the filler of NeuraWrap™ conduits and transplanted to repair a 15-mm rat sciatic nerve defects. Eight weeks following transplantation, newly regenerated nerves from rats receiving NeuraWrap™ filled with EngNT-d-hDPSCs showed ingrowing neurites, myelinated nerve fibers, and blood vessels along the construct as evaluated by immunohistochemistry and ultrastructural analysis [87]. In addition, human dental follicle stem cells (hDFSCs) have also been combined with aligned electrospun PCL/PLGA material (AEM) to generate DFSC-seeded AEM constructs, wherein DFSCs could stretch along the oriented fibers and maintain their proliferative ability and implantation of DFSC-seeded AEM constructs contributed to the restoration of spinal cord injury in rats [53].
Most recently, we have shown that GMSCs undergo spontaneous conversion into Schwann cell precursor (SCP)-like cells when they were encapsulated in soft methylated 3D collagen hydrogel [125, 131]. Interestingly, GMSCs encapsulated in methylated 3D collagen hydrogel are capable of migrating and incorporating into the wall matrix of Axoguard nerve connectors or protectors made of porcine small intestine submucosa matrix (SIS). In vivo, implantation of functionalized NGCs laden with GMSC-derived SCPs remarkably improved the functional recovery and axonal regeneration in the segmental facial nerve defects and the crush-injured sciatic nerves in rats [124, 125, 131]. Taken together, these studies have demonstrated the feasibility to generate cell-based functionalized NGCs by incorporating orofacial MSCs into the scaffolds of NGCs with enhanced pro-regenerative potentials in preclinical models of transected nerve defects.
3D-Bioprinting of Orofacial MSC-Laden Nerve Grafts for PNR
Traditionally 3D-printed nonbiological NGCs could support PNR through providing the anatomic and geometric guidance for injured nerves, but their outcomes of functional restoration are limited, thus prompting the fabrication of biomimetic nerve grafts by incorporating cellular and biological components via 3D bioprinting technologies [90]. One of the key components for 3D-bioprinting is the selection and formulation of appropriate “bio-inks” composed of cells and biologics such as signaling molecules and growth factors [96, 122]. Various methods can produce 3D structures via bio-printing, including but not limited to extrusion-based, inkjet, stereolithography, bioplotting, and the Kenzan method which involves the use of a needle array to arrange cell spheroids to allow self-aggregation to form the 3D construct [122]. Currently, there exist two approaches, scaffold-based and scaffold-free, to 3D-bioprint biomimetic nerve grafts as described below.
Scaffold-Based Biofabricated Nerve Grafts
Bio-inks used for 3D-bioprinting of scaffold-based nerve grafts are usually composed of various types of hydrogels/scaffolds or ECMs encapsulated with different types of cells [90]. For instance, neural stem cells (NSCs) encapsulated with different types of hydrogels/scaffolds, such as conductive composite hydrogel (CCH) scaffolds [97], GelMA/HAMA hydrogels [117], sodium alginate/gelatin scaffolds [56, 57], collagen/silk fibroin scaffold [41], have been fabricated into nerve-like grafts by 3D bioprinting, which show positive effects on repairing of spinal cord injury (SCI).
Scaffold-Free Biofabricated Nerve Grafts
In 2013, Owens CM et al. was the first to report a novel approach to biofabricate a fully cellular nerve graft through 3D bioprinting (NovoGen MMX Bioprinter™; Organovo) with 90% of BMSCs and 10% of Schwann cells (SC) as the cellular bio-ink [75]. In a rat sciatic nerve gap injury (10-mm) model, the biofabricated nerve grafts exhibited similar functional motor and sensory recoveries to nerve autografts and might be superior to the hollow collagen tube (Neuragen) [75]. Later on, Yurie H et al. utilized a Kenzan method-based bioprinter system (Regenova®; Cyfuse, Tokyo, Japan) and biofabricated a scaffold-free Bio 3D nerve conduit with 3D-spheroids of human dermal fibroblasts as the cellular “bio-ink” [123]. In a rat sciatic nerve gap injury (5-mm) model, implantation of the scaffold-free Bio 3D nerve conduit significantly promoted both motor and sensory functional recovery and remyelination compared with the silicone NGC control group [123]. To extend their studies, the same group of researchers used the same approach to biofabricate scaffold-free Bio 3D nerve conduits from normal human dermal fibroblasts (NHDF) and tested their pro-nerve regenerative potentials in a larger 10-mm sciatic nerve defect model in immune deficient F344 rats [104]. In the distal regions, the number of myelinated axons, the myelinated axon diameter, and the myelin thickness (MT) of the regenerated axons in the Bio 3D group were significantly higher/larger than those of the control silicone conduit group [104]. In a 5-mm canine ulnar nerve defect model in Beagle dogs, transplantation of biofabricated scaffold-free Bio 3D conduits developed from autologous dermal fibroblasts also showed significant supportive effects on axonal regrowth, remyelination, and Schwann cell migration [68]. In addition to dermal fibroblasts, human induced pluripotent stem cell-derived mesenchymal stem cells (iMSCs) have also been utilized for biofabrication of scaffold-free Bio 3D conduits, which displayed pro-angiogenic and neurotrophic effects after transplantation in vivo. In a 5-mm sciatic nerve defect model in immunodeficient rats, functional, histological, and morphometric assessments indicated that the regenerated nerve in the Bio 3D group was significantly superior to that in the silicone group [69]. Similarly, scaffold-free 3D nerve constructs have been biofabricated with human GMSC spheroids as the cellular “bio-ink”. In a 5-mm facial nerve defect model in rats, transplantation of GMSC-based scaffold-free 3D nerve construct resulted in functional (CMAP) recovery of facial nerve to a degree similar to the autograft group and better axonal regeneration and remyelination than the silicon control group [127, 128]. Taken together, these studies have shed light on the application of 3D bioprinting as a promising approach to nerve graft fabrication and nerve regeneration.
Future and Challenges
Due to their neural crest-origin and easily accessibility, orofacial MSCs as a unique source of MSCs hold great promise for the development of cell-based TE/RM products for the treatment of a variety of pathological conditions, particularly, peripheral injuries [112, 113]. However, several major challenges have been identified in the application of orofacial MSCs in peripheral nerve repair/regeneration. 1) Similar to other sources of MSCs, orofacial MSCs are heterogeneous in their properties and biological functions, and there are either no standard protocols for their isolation and ex vivo expansion or no consistent markers for their characterization [2, 125, 131]. Future studies should optimize the isolation and culture conditions, and consequently, the establishment of standardized procedures for ex vivo expansion of orofacial MSCs that can be translated into large scale production of orofacial MSCs with good manufacturing practice (GMP) quality [66]. 2) Even though many studies have implicated that the secretomes or paracrine factors contribute to MSC-based therapeutic effects on various diseases [34], to date, the mechanism of action of orofacial MSC-based regenerative therapies of PNR, remains largely unknown. For instance, it is less known about the immunomodulatory effects of orofacial MSCs in the treatment of PNI, wherein the interaction of immune cells, particularly macrophages, and Schwann cells, play a critical role in Wallerian degeneration and axonal regeneration. Moreover, the various pathways involved in the mediation of MSC-derived benefits remain to be explored. 3) In most studies, the fate of MSCs following transplantation in combination with various types of scaffolds or NGCs in injured nerves remain largely unknown. In the future, it is critical to develop novel approaches for the long-term track of transplanted orofacial MSCs in injured nerves, thus enabling reliable evaluation of the therapeutic outcome conferred by the transplanted cells. 4) Even though the therapeutic efficacy of orofacial MSCs combined with various types of NGCs has been demonstrated in various rodent models of peripheral nerve injury with a small gap (usually 5-15 mm), it is worthy to further evaluate the pro-nerve regenerative potentials of orofacial MSCs in larger animal models with critical gap nerve injuries [55] in the hope to obtain substantial evidence supporting the application of orofacial MSCs in translational clinical studies. 5) 3D-bioprinting is emerging as a novel platform for fabricating cell-based functionalized NGCs. However, little has been explored for the use of orofacial MSCs in 3D bioprinting nerve grafts. Further studies are necessary to extend the application of orofacial MSCs as unique “bio-inks” for biofabrication of nerve grafts and test their regenerative potentials in different pre-clinical models of PNI. In summary, the optimization and standardization of the procedures for isolation and ex vivo expansion of orofacial MSCs, the combination of biomaterial science and advanced tissue engineering technology for generation of new generation of orofacial-MSC based functionalized NGCs, and the employment of larger animal models of PNIs, will pave the way for translational application of orofacial MSCs in PNR.
Future Implementation
Although there are numerous orofacial technologies in development for treatment of peripheral nerve injuries, successful clinical implementation will require a paradigm-shift in surgical workflow. The proposed technologies in this article will likely be regulated as a combined therapeutic (i.e. medical devices and biologic). In the United States, FDA-regulated medical products must comply with current Good Manufacturing Practice (cGMP) for advanced clinical trials and eventual commercialization. The FDA cGMP guidelines are designed to ensure safety and reliability of the final clinical product.
Implementing orofacial MSCs for PNR requires MSC isolation, expansion, and modification usually in vitro, then incorporation into constructs or scaffolds for implantation. This is unlike the process for obtaining minimally modified cells such as BMSCs and ADSCs in bone marrow aspirate concentrate (BMAC) and adipose-derived stromal vascular fraction (SVF), respectively. BMSCs and ADSCs are extracted via needle aspiration and liposuction, respectively, then centrifugated (instead of expanded and modified) to obtain the MSCs to be transplanted back into the target area in the patient [19]. Minimally modified cells circumvent the strict regulations regarding cell expansion. Thus, conditions remain challenging. Notably, autologous cGMP-grade Schwann cells have previously been transplanted into patients with spinal cord or peripheral nerve injury [45]. Schwann cells were isolated and expanded from autologous donor nerves that were enzymatically dissociated over the course of 21 days. Similar protocols will be necessary to generate cGMP-compliant orofacial stem cells at scale for clinical implementation.
Our interdisciplinary team of oral surgeons, dentists, scientists, and bioengineers have been pioneering several regenerative therapies using orofacial stem cell products. We envision a future workflow where a patient with a nerve injury is referred by surgeon to their local dentist for a relatively minor tissue extraction (e.g. gingiva biopsy, tooth extraction, etc.). Over the next few weeks, autologous orofacial stem cells will be isolated at cGMP facility and subsequently engineered into replacement nerve tissue. The final engineered tissue will be delivered to the clinician who will be responsible for ensuring quality prior to transplantation into the patient. Additionally, adequate non-destructive quality control metrics (e.g. biomarkers) and tissue preservation strategies will be necessary. The time between peripheral nerve injury and nerve graft implantation should be minimal to maximize therapeutic outcome. Thus, a future solution could be a tissue bank of pre-made TENGs, with a variety for different injury types, severity, and nerve sizes ready to be transplanted in the case of an emergency [55]. Overall, success of orofacial MSCs for PNR will need the expertise and disciplines of a variety of fields to improve the PNR clinical outcomes (Fig. 6).
A potential future process for producing and implementing orofacial-MSC-ladened tissue-engineered nerve grafts. a Neurosurgeon refers the patient with peripheral nerve injury to a dentist for orofacial tissue extraction. b Patient undergoes minor tissue extraction. c Tissues are transported to a GMP facility for further processing. d Orofacial MSCs are obtained from the tissues. e The cells are isolated and expanded in the GMP facility over a time period. f Cells are incorporated into pre-made nerve conduits or grafts, allowed for maturation for a time period. g The tissue-engineered nerve graft (TENG) is shipped to the operating room. h The TENG is transplanted into the patient’s injured nerve
Conclusion
A more effective method of peripheral nerve injury repair is an urgent clinical need, one that surpasses the performance of current biological nerve grafts. The use of a functionalized tissue-engineered nerve graft (TENG) incorporated with orofacial MSCs presents a sound solution because of their superior qualities for nerve regeneration, due to their neural crest origins, demonstrated by their neurogenic markers and neural differentiation capability. In addition, they possess immunomodulatory functions for regulating immune cells during PNR and produce secretome that shows similar neurogenic and immunomodulatory qualities, presenting an option for acellular repair method. Practically, orofacial MSC extraction presents a non-invasive option, rivaling the invasiveness of extracting BMSCs and ADSCs from the body. In recent years, various types of orofacial MSCs or their product-ladened nerve grafts have demonstrated promising results for PNR in vitro and in vivo. In conclusion, their strong neurogenic and immunomodulatory properties, as well as the ease of harvesting and incorporation, make orofacial MSCs a promising cellular component for future application in peripheral nerve injury repair.
Data Availability
Not applicable.
Abbreviations
- MSC:
-
mesenchymal stem cells
- BMSC:
-
bone marrow-derived mesenchymal stem cells
- ADSC:
-
adipose derived stem ells
- NGC:
-
nerve guidance conduit
- PNI:
-
peripheral nerve injury
- PNR:
-
peripheral nerve repair/regeneration
- DPSC:
-
dental pulp stem cell
- SHED:
-
exfoliated deciduous teeth derived stem cell
- SCAP:
-
apical papilla derived stem cells
- TGSC:
-
tooth germ derived stem cell
- PDLSCs:
-
periodontal ligament stem cells
- GMSCs:
-
gingiva-derived mesenchymal stem cells
- DFSCs:
-
dental follicle stem cells
- CNS:
-
central nervous system
- PNS:
-
peripheral nervous system
- SC:
-
Schwann cell
- NC:
-
neural crest
- NCSC:
-
neural crest derived stem cell
- CFU-F:
-
fibroblast-like colony-forming units
- TE/RM:
-
tissue engineering/regenerative medicine
- TENGs:
-
tissue engineered nerve grafts
- EngNT:
-
engineered neural tissues
References
Ahmadi, N., Razavi, S., Kazemi, M., & Oryan, S. (2012). Stability of neural differentiation in human adipose derived stem cells by two induction protocols. Tissue and Cell, 44(2), 87–94. https://doi.org/10.1016/j.tice.2011.11.006
Bar, J. K., Lis-Nawara, A., & Grelewski, P. G. (2021). Dental pulp stem cell-derived secretome and its regenerative potential. International Journal of Molecular Sciences, 22(21). https://doi.org/10.3390/ijms222112018
Beigi, M. H., Ghasemi-Mobarakeh, L., Prabhakaran, M. P., Karbalaie, K., Azadeh, H., Ramakrishna, S., Baharvand, H., & Nasr-Esfahani, M. H. (2014). In vivo integration of poly(epsilon-caprolactone)/gelatin nanofibrous nerve guide seeded with teeth derived stem cells for peripheral nerve regeneration. Journal of Biomedical Materials Research. Part A, 102(12), 4554–4567. https://doi.org/10.1002/jbm.a.35119
Beris, A., Gkiatas, I., Gelalis, I., Papadopoulos, D., & Kostas-Agnantis, I. (2019). Current concepts in peripheral nerve surgery. European Journal of Orthopaedic Surgery & Traumatology, 29(2), 263–269. https://doi.org/10.1007/s00590-018-2344-2
Biso, G., & Munakomi, S. (2023). Neuroanatomy, Neurapraxia. In StatPearls [Internet]. StatPearls Publishing. https://www.ncbi.nlm.nih.gov/pubmed/32491678. Accessed 24 Oct 2022
Brown, C., McKee, C., Bakshi, S., Walker, K., Hakman, E., Halassy, S., Svinarich, D., Dodds, R., Govind, C. K., & Chaudhry, G. R. (2019). Mesenchymal stem cells: Cell therapy and regeneration potential. Journal of Tissue Engineering and Regenerative Medicine, 13(9), 1738–1755. https://doi.org/10.1002/term.2914
Bunnell, B. A. (2021). Adipose tissue-derived mesenchymal stem cells. Cells, 10(12). https://doi.org/10.3390/cells10123433
Burks, S. S., Diaz, A., Haggerty, A. E., Oliva, N., Midha, R., & Levi, A. D. (2021). Schwann cell delivery via a novel 3D collagen matrix conduit improves outcomes in critical length nerve gap repairs. Journal of Neurosurgery, 135(4), 1241–1251. https://doi.org/10.3171/2020.8.JNS202349
Campbell, W. W. (2008). Evaluation and management of peripheral nerve injury. Clinical Neurophysiology, 119(9), 1951–1965. https://doi.org/10.1016/j.clinph.2008.03.018
Carballo Cuello, C. M., & De Jesus, O. (2023). Neurapraxia. In StatPearls [Internet]. StatPearls Publishing. https://www.ncbi.nlm.nih.gov/pubmed/32809336. Accessed 25 July 2023
Carvalho, C. R., Oliveira, J. M., & Reis, R. L. (2019). Modern trends for peripheral nerve repair and regeneration: Beyond the hollow nerve guidance conduit. Frontiers in Bioengineering and Biotechnology, 7, 337. https://doi.org/10.3389/fbioe.2019.00337
Charbord, P. (2010). Bone marrow mesenchymal stem cells: Historical overview and concepts. Human Gene Therapy, 21(9), 1045–1056. https://doi.org/10.1089/hum.2010.115
Chen, P., Piao, X., & Bonaldo, P. (2015). Role of macrophages in Wallerian degeneration and axonal regeneration after peripheral nerve injury. Acta Neuropathologica, 130(5), 605–618. https://doi.org/10.1007/s00401-015-1482-4
Chen, X., Yang, B., Tian, J., Hong, H., Du, Y., Li, K., Li, X., Wang, N., Yu, X., & Wei, X. (2018). Dental follicle stem cells ameliorate lipopolysaccharide-induced inflammation by secreting TGF-β3 and TSP-1 to elicit macrophage M2 polarization. Cellular Physiology and Biochemistry, 51(5), 2290–2308. https://doi.org/10.1159/000495873
Dai, L. G., Huang, G. S., & Hsu, S. H. (2013). Sciatic nerve regeneration by cocultured Schwann cells and stem cells on microporous nerve conduits. Cell Transplantation, 22(11), 2029–2039. https://doi.org/10.3727/096368912x658953
de Ruiter, G. C., Malessy, M. J., Yaszemski, M. J., Windebank, A. J., & Spinner, R. J. (2009). Designing ideal conduits for peripheral nerve repair. Neurosurgical Focus, 26(2), E5. https://doi.org/10.3171/FOC.2009.26.2.E5
Debanne, D., Campanac, E., Bialowas, A., Carlier, E., & Alcaraz, G. (2011). Axon physiology. Physiological Reviews, 91(2), 555–602. https://doi.org/10.1152/physrev.00048.2009
DeFrancesco-Lisowitz, A., Lindborg, J. A., Niemi, J. P., & Zigmond, R. E. (2015). The neuroimmunology of degeneration and regeneration in the peripheral nervous system. Neuroscience, 302, 174–203. https://doi.org/10.1016/j.neuroscience.2014.09.027
Di Matteo, B., Vandenbulcke, F., Vitale, N. D., Iacono, F., Ashmore, K., Marcacci, M., & Kon, E. (2019). Minimally manipulated mesenchymal stem cells for the treatment of knee osteoarthritis: A systematic review of clinical evidence. Stem Cells International, 2019, 1735242. https://doi.org/10.1155/2019/1735242
Ding, D. C., Shyu, W. C., & Lin, S. Z. (2011). Mesenchymal stem cells. Cell Transplantation, 20(1), 5–14. https://doi.org/10.3727/096368910X
Ding, G., Liu, Y., An, Y., Zhang, C., Shi, S., Wang, W., & Wang, S. (2010). Suppression of T cell proliferation by root apical papilla stem cells in vitro. Cells, Tissues, Organs, 191(5), 357–364. https://doi.org/10.1159/000276589
Dixon, A. R., Jariwala, S. H., Bilis, Z., Loverde, J. R., Pasquina, P. F., & Alvarez, L. M. (2018). Bridging the gap in peripheral nerve repair with 3D printed and bioprinted conduits. Biomaterials, 186, 44–63. https://doi.org/10.1016/j.biomaterials.2018.09.010
Dominici, M., Le Blanc, K., Mueller, I., Slaper-Cortenbach, I., Marini, F., Krause, D., Deans, R., Keating, A., Prockop, D., & Horwitz, E. (2006). Minimal criteria for defining multipotent mesenchymal stromal cells. The international society for cellular therapy position statement. Cytotherapy, 8(4), 315–317. https://doi.org/10.1080/14653240600855905
Fortino, V. R., Chen, R. S., Pelaez, D., & Cheung, H. S. (2014). Neurogenesis of neural crest-derived periodontal ligament stem cells by EGF and bFGF. Journal of Cellular Physiology, 229(4), 479–488. https://doi.org/10.1002/jcp.24468
Fournier, B. P., Loison-Robert, L. S., Ferre, F. C., Owen, G. R., Larjava, H., & Hakkinen, L. (2016). Characterisation of human gingival neural crest-derived stem cells in monolayer and neurosphere cultures. European Cells and Materials, 31, 40–58. https://doi.org/10.22203/ecm.v031a04
Friedenstein, A. J., Chailakhjan, R. K., & Lalykina, K. S. (1970). The development of fibroblast colonies in monolayer cultures of guinea-pig bone marrow and spleen cells. Cell and Tissue Kinetics, 3(4), 393–403. https://doi.org/10.1111/j.1365-2184.1970.tb00347.x
Gao, X., Shen, Z., Guan, M., Huang, Q., Chen, L., Qin, W., Ge, X., Chen, H., Xiao, Y., & Lin, Z. (2018). Immunomodulatory role of stem cells from human exfoliated deciduous teeth on periodontal regeneration. Tissue Engineering Part A, 24(17–18), 1341–1353. https://doi.org/10.1089/ten.TEA.2018.0016
Georgiou, M., Golding, J. P., Loughlin, A. J., Kingham, P. J., & Phillips, J. B. (2015). Engineered neural tissue with aligned, differentiated adipose-derived stem cells promotes peripheral nerve regeneration across a critical sized defect in rat sciatic nerve. Biomaterials, 37, 242–251. https://doi.org/10.1016/j.biomaterials.2014.10.009
Gordon, T. (2020). Peripheral nerve regeneration and muscle reinnervation. International Journal of Molecular Sciences, 21(22). https://doi.org/10.3390/ijms21228652
Griffin, J. W., George, E. B., Hsieh, S.-T., & Glass, J. D. (1995). 375Axonal degeneration and disorders of the axonal cytoskeleton. In S. G. Waxman, J. D. Kocsis, & P. K. Stys (Eds.), The Axon: Structure, function and pathophysiology (p. 0). Oxford University Press. https://doi.org/10.1093/acprof:oso/9780195082937.003.0020
Grinsell, D., & Keating, C. P. (2014). Peripheral nerve reconstruction after injury: A review of clinical and experimental therapies. Biomed Research International, 2014, 698256. https://doi.org/10.1155/2014/698256
Gronthos, S., Brahim, J., Li, W., Fisher, L. W., Cherman, N., Boyde, A., DenBesten, P., Robey, P. G., & Shi, S. (2002). Stem cell properties of human dental pulp stem cells. Journal of Dental Research, 81(8), 531–535. https://doi.org/10.1177/154405910208100806
Gugliandolo, A., & Mazzon, E. (2021). Dental mesenchymal stem cell secretome: An intriguing approach for neuroprotection and neuroregeneration. International Journal of Molecular Sciences, 23(1). https://doi.org/10.3390/ijms23010456
Han, Y., Yang, J., Fang, J., Zhou, Y., Candi, E., Wang, J., Hua, D., Shao, C., & Shi, Y. (2022). The secretion profile of mesenchymal stem cells and potential applications in treating human diseases. Signal Transduction and Targeted Therapy, 7(1), 92. https://doi.org/10.1038/s41392-022-00932-0
Hopf, A., Schaefer, D. J., Kalbermatten, D. F., Guzman, R., & Madduri, S. (2020). Schwann cell-like cells: Origin and usability for repair and regeneration of the peripheral and central nervous system. Cells, 9(9). https://doi.org/10.3390/cells9091990
Houshyar, S., Bhattacharyya, A., & Shanks, R. (2019). Peripheral nerve conduit: Materials and structures. ACS Chemical Neuroscience, 10(8), 3349–3365. https://doi.org/10.1021/acschemneuro.9b00203
Hu, N., Wu, H., Xue, C., Gong, Y., Wu, J., Xiao, Z., Yang, Y., Ding, F., & Gu, X. (2013). Long-term outcome of the repair of 50 mm long median nerve defects in rhesus monkeys with marrow mesenchymal stem cells-containing, chitosan-based tissue engineered nerve grafts. Biomaterials, 34(1), 100–111. https://doi.org/10.1016/j.biomaterials.2012.09.020
Janebodin, K., Horst, O. V., Ieronimakis, N., Balasundaram, G., Reesukumal, K., Pratumvinit, B., & Reyes, M. (2011). Isolation and characterization of neural crest-derived stem cells from dental pulp of neonatal mice. PLoS One, 6(11), e27526. https://doi.org/10.1371/journal.pone.0027526
Jang, S., Kang, Y. H., Ullah, I., Shivakumar, S. B., Rho, G. J., Cho, Y. C., Sung, I. Y., & Park, B. W. (2018). Cholinergic nerve differentiation of mesenchymal stem cells derived from long-term cryopreserved human dental pulp in vitro and analysis of their motor nerve regeneration potential in vivo. International Journal of Molecular Sciences, 19(8). https://doi.org/10.3390/ijms19082434
Jessen, K. R., & Mirsky, R. (2016). The repair Schwann cell and its function in regenerating nerves. Journal of Physiology, 594(13), 3521–3531. https://doi.org/10.1113/JP270874
Jiang, J. P., Liu, X. Y., Zhao, F., Zhu, X., Li, X. Y., Niu, X. G., Yao, Z. T., Dai, C., Xu, H. Y., Ma, K., Chen, X. Y., & Zhang, S. (2020). Three-dimensional bioprinting collagen/silk fibroin scaffold combined with neural stem cells promotes nerve regeneration after spinal cord injury. Neural Regeneration Research, 15(5), 959–968. https://doi.org/10.4103/1673-5374.268974
Kamble, N., Shukla, D., & Bhat, D. (2019). Peripheral nerve injuries: Electrophysiology for the neurosurgeon. Neurology India, 67(6), 1419–1422. https://doi.org/10.4103/0028-3886.273626
Kang, J., Fan, W., Deng, Q., He, H., & Huang, F. (2019). Stem cells from the apical papilla: A promising source for stem cell-based therapy. BioMed Research International, 2019, 6104738. https://doi.org/10.1155/2019/6104738
Kang, N. U., Lee, S. J., & Gwak, S. J. (2022). Fabrication Techniques of nerve guidance conduits for nerve regeneration. Yonsei Medical Journal, 63(2), 114–123. https://doi.org/10.3349/ymj.2022.63.2.114
Khan, A., Diaz, A., Brooks, A. E., Burks, S. S., Athauda, G., Wood, P., Lee, Y. S., Silvera, R., Donaldson, M., Pressman, Y., Anderson, K. D., Bunge, M. B., Pearse, D. D., Dietrich, W. D., Guest, J. D., & Levi, A. D. (2022). Scalable culture techniques to generate large numbers of purified human Schwann cells for clinical trials in human spinal cord and peripheral nerve injuries. Journal of Neurosurgery. Spine, 36(1), 135–144. https://doi.org/10.3171/2020.11.Spine201433
Kim, B. C., Bae, H., Kwon, I. K., Lee, E. J., Park, J. H., Khademhosseini, A., & Hwang, Y. S. (2012). Osteoblastic/cementoblastic and neural differentiation of dental stem cells and their applications to tissue engineering and regenerative medicine. Tissue Engineering. Part B, Reviews, 18(3), 235–244. https://doi.org/10.1089/ten.TEB.2011.0642
Kou, M., Huang, L., Yang, J., Chiang, Z., Chen, S., Liu, J., Guo, L., Zhang, X., Zhou, X., Xu, X., Yan, X., Wang, Y., Zhang, J., Xu, A., Tse, H. F., & Lian, Q. (2022). Mesenchymal stem cell-derived extracellular vesicles for immunomodulation and regeneration: A next generation therapeutic tool? Cell Death & Disease, 13(7), 580. https://doi.org/10.1038/s41419-022-05034-x
Praveen Kumar, L., Kandoi, S., Misra, R., Vijayalakshmi, S., Rajagopal, K., & Verma, R. S. (2019). The mesenchymal stem cell secretome: A new paradigm towards cell-free therapeutic mode in regenerative medicine. Cytokine & Growth Factor Reviews, 46, 1–9. https://doi.org/10.1016/j.cytogfr.2019.04.002
Lackington, W. A., Ryan, A. J., & O’Brien, F. J. (2017). Advances in nerve guidance conduit-based therapeutics for peripheral nerve repair. ACS Biomaterials Science & Engineering, 3(7), 1221–1235. https://doi.org/10.1021/acsbiomaterials.6b00500
Lavorato, A., Raimondo, S., Boido, M., Muratori, L., Durante, G., Cofano, F., Vincitorio, F., Petrone, S., Titolo, P., Tartara, F., Vercelli, A., & Garbossa, D. (2021). Mesenchymal stem cell treatment perspectives in peripheral nerve regeneration: Systematic review. International Journal of Molecular Sciences, 22(2). https://doi.org/10.3390/ijms22020572
Lee, S. K., & Wolfe, S. W. (2000). Peripheral nerve injury and repair. Journal of American Academy of Orthopaedic Surgeons, 8(4), 243–252. https://doi.org/10.5435/00124635-200007000-00005
Li, J. F., Yin, H. L., Shuboy, A., Duan, H. F., Lou, J. Y., Li, J., Wang, H. W., & Wang, Y. L. (2013). Differentiation of hUC-MSC into dopaminergic-like cells after transduction with hepatocyte growth factor. Molecular and Cellular Biochemistry, 381(1–2), 183–190. https://doi.org/10.1007/s11010-013-1701-z
Li, X., Yang, C., Li, L., Xiong, J., Xie, L., Yang, B., Yu, M., Feng, L., Jiang, Z., Guo, W., & Tian, W. (2015). A therapeutic strategy for spinal cord defect: human dental follicle cells combined with aligned PCL/PLGA electrospun material. Biomed Research International, 2015, 197183. https://doi.org/10.1155/2015/197183
Li, Y., Duan, X., Chen, Y., Liu, B., & Chen, G. (2022). Dental stem cell-derived extracellular vesicles as promising therapeutic agents in the treatment of diseases. International Journal of Oral Science, 14(1), 2. https://doi.org/10.1038/s41368-021-00152-2
Lischer, M., di Summa, P. G., Petrou, I. G., Schaefer, D. J., Guzman, R., Kalbermatten, D. F., & Madduri, S. (2023). Mesenchymal stem cells in nerve tissue engineering: Bridging nerve gap injuries in large animals. International Journal of Molecular Sciences, 24(9). https://doi.org/10.3390/ijms24097800
Liu, C., Hu, F., Jiao, G., Guo, Y., Zhou, P., Zhang, Y., Zhang, Z., Yi, J., You, Y., Li, Z., Wang, H., & Zhang, X. (2022). Dental pulp stem cell-derived exosomes suppress M1 macrophage polarization through the ROS-MAPK-NFkappaB P65 signaling pathway after spinal cord injury. Journal of Nanobiotechnology, 20(1), 65. https://doi.org/10.1186/s12951-022-01273-4
Liu, S., Yang, H., Chen, D., Xie, Y., Tai, C., Wang, L., Wang, P., & Wang, B. (2022). Three-dimensional bioprinting sodium alginate/gelatin scaffold combined with neural stem cells and oligodendrocytes markedly promoting nerve regeneration after spinal cord injury. Regenerative Biomaterials, 9, rbac038. https://doi.org/10.1093/rb/rbac038
Liu, X. M., Liu, Y., Yu, S., Jiang, L. M., Song, B., & Chen, X. (2019). Potential immunomodulatory effects of stem cells from the apical papilla on Treg conversion in tissue regeneration for regenerative endodontic treatment. International Endodontic Journal, 52(12), 1758–1767. https://doi.org/10.1111/iej.13197
Lopes, B., Sousa, P., Alvites, R., Branquinho, M., Sousa, A. C., Mendonca, C., Atayde, L. M., Luis, A. L., Varejao, A. S. P., & Mauricio, A. C. (2022). Peripheral nerve injury treatments and advances: One health perspective. International Journal of Molecular Sciences, 23(2). https://doi.org/10.3390/ijms23020918
Lundborg, G., Gelberman, R. H., Longo, F. M., Powell, H. C., & Varon, S. (1982). In vivo regeneration of cut nerves encased in silicone tubes: Growth across a six-millimeter gap. Journal of Neuropathology and Experimental Neurology, 41(4), 412–422. https://doi.org/10.1097/00005072-198207000-00004
Luo, L., He, Y., Jin, L., Zhang, Y., Guastaldi, F. P., Albashari, A. A., Hu, F., Wang, X., Wang, L., Xiao, J., Li, L., Wang, J., Higuchi, A., & Ye, Q. (2021). Application of bioactive hydrogels combined with dental pulp stem cells for the repair of large gap peripheral nerve injuries. Bioactive Materials, 6(3), 638–654. https://doi.org/10.1016/j.bioactmat.2020.08.028
Mao, Q., Nguyen, P. D., Shanti, R. M., Shi, S., Shakoori, P., Zhang, Q., & Le, A. D. (2019). Gingiva-derived mesenchymal stem cell-extracellular vesicles activate Schwann cell repair phenotype and promote nerve regeneration. Tissue Engineering Part A, 25(11–12), 887–900. https://doi.org/10.1089/ten.TEA.2018.0176
Martinez, V. G., Ontoria-Oviedo, I., Ricardo, C. P., Harding, S. E., Sacedon, R., Varas, A., Zapata, A., Sepulveda, P., & Vicente, A. (2017). Overexpression of hypoxia-inducible factor 1 alpha improves immunomodulation by dental mesenchymal stem cells. Stem Cell Research & Therapy, 8(1), 208. https://doi.org/10.1186/s13287-017-0659-2
Mayo, V., Sawatari, Y., Huang, C. Y., & Garcia-Godoy, F. (2014). Neural crest-derived dental stem cells–where we are and where we are going. Journal of Dentistry, 42(9), 1043–1051. https://doi.org/10.1016/j.jdent.2014.04.007
Merle, M., Dellon, A. L., Campbell, J. N., & Chang, P. S. (1989). Complications from silicon-polymer intubulation of nerves. Microsurgery, 10(2), 130–133. https://doi.org/10.1002/micr.1920100213
Mezey, É. (2022). Human Mesenchymal stem/stromal cells in immune regulation and therapy. Stem Cells Translational Medicine, 11(2), 114–134. https://doi.org/10.1093/stcltm/szab020
Min, Q., Parkinson, D. B., & Dun, X. P. (2021). Migrating Schwann cells direct axon regeneration within the peripheral nerve bridge. Glia, 69(2), 235–254. https://doi.org/10.1002/glia.23892
Mitsuzawa, S., Ikeguchi, R., Aoyama, T., Takeuchi, H., Yurie, H., Oda, H., Ohta, S., Ushimaru, M., Ito, T., Tanaka, M., Kunitomi, Y., Tsuji, M., Akieda, S., Nakayama, K., & Matsuda, S. (2019). The efficacy of a Scaffold-free Bio 3D conduit developed from autologous dermal fibroblasts on peripheral nerve regeneration in a canine ulnar nerve injury model: A preclinical proof-of-concept study. Cell Transplantation, 28(9–10), 1231–1241. https://doi.org/10.1177/0963689719855346
Mitsuzawa, S., Zhao, C., Ikeguchi, R., Aoyama, T., Kamiya, D., Ando, M., Takeuchi, H., Akieda, S., Nakayama, K., Matsuda, S., & Ikeya, M. (2020). Pro-angiogenic scaffold-free Bio three-dimensional conduit developed from human induced pluripotent stem cell-derived mesenchymal stem cells promotes peripheral nerve regeneration. Science and Reports, 10(1), 12034. https://doi.org/10.1038/s41598-020-68745-1
Miura, M., Gronthos, S., Zhao, M., Lu, B., Fisher, L. W., Robey, P. G., & Shi, S. (2003). SHED: Stem cells from human exfoliated deciduous teeth. Proceedings of the National Academy of Sciences of the United States of America, 100(10), 5807–5812. https://doi.org/10.1073/pnas.0937635100
Mohebichamkhorami, F., Niknam, Z., Khoramjouy, M., Heidarli, E., Ghasemi, R., Hosseinzadeh, S., Mohseni, S. S., Hajikarim-Hamedani, A., Heidari, A., Ghane, Y., Mahmoudifard, M., Zali, H., & Faizi, M. (2022). Brain homogenate of a rat model of Alzheimer’s disease modifies the secretome of 3D cultured periodontal ligament stem cells: A potential neuroregenerative therapy. Iranian Journal of Pharmaceutical Research, 21(1), e133668. https://doi.org/10.5812/ijpr-133668
Morsczeck, C., Gotz, W., Schierholz, J., Zeilhofer, F., Kuhn, U., Mohl, C., Sippel, C., & Hoffmann, K. H. (2005). Isolation of precursor cells (PCs) from human dental follicle of wisdom teeth. Matrix Biology, 24(2), 155–165. https://doi.org/10.1016/j.matbio.2004.12.004
Mu, X., Liu, H., Yang, S., Li, Y., Xiang, L., Hu, M., & Wang, X. (2022). Chitosan tubes inoculated with dental pulp stem cells and stem cell factor enhance facial nerve-vascularized regeneration in rabbits. ACS Omega, 7(22), 18509–18520. https://doi.org/10.1021/acsomega.2c01176
Mushahary, D., Spittler, A., Kasper, C., Weber, V., & Charwat, V. (2018). Isolation, cultivation, and characterization of human mesenchymal stem cells. Cytometry. Part A, 93(1), 19–31. https://doi.org/10.1002/cyto.a.23242
Owens, C. M., Marga, F., Forgacs, G., & Heesch, C. M. (2013). Biofabrication and testing of a fully cellular nerve graft. Biofabrication, 5(4), 045007. https://doi.org/10.1088/1758-5082/5/4/045007
Panagopoulos, G. N., Megaloikonomos, P. D., & Mavrogenis, A. F. (2017). The present and future for peripheral nerve regeneration. Orthopedics, 40(1), e141–e156. https://doi.org/10.3928/01477447-20161019-01
Parker, B. J., Rhodes, D. I., O’Brien, C. M., Rodda, A. E., & Cameron, N. R. (2021). Nerve guidance conduit development for primary treatment of peripheral nerve transection injuries: A commercial perspective. Acta Biomaterialia, 135, 64–86. https://doi.org/10.1016/j.actbio.2021.08.052
Pfister, B. J., Gordon, T., Loverde, J. R., Kochar, A. S., Mackinnon, S. E., & Cullen, D. K. (2011). Biomedical engineering strategies for peripheral nerve repair: Surgical applications, state of the art, and future challenges. Critical Reviews in Biomedical Engineering, 39(2), 81–124. https://doi.org/10.1615/critrevbiomedeng.v39.i2.20
Pisciotta, A., Bertoni, L., Riccio, M., Mapelli, J., Bigiani, A., La Noce, M., Orciani, M., de Pol, A., & Carnevale, G. (2018). Use of a 3D floating sphere culture system to maintain the neural crest-related properties of human dental pulp stem cells. Frontiers in Physiology, 9, 547. https://doi.org/10.3389/fphys.2018.00547
Pisciotta, A., Bertoni, L., Vallarola, A., Bertani, G., Mecugni, D., & Carnevale, G. (2020). Neural crest derived stem cells from dental pulp and tooth-associated stem cells for peripheral nerve regeneration. Neural Regeneration Research, 15(3), 373–381. https://doi.org/10.4103/1673-5374.266043
Poongodi, R., Chen, Y. L., Yang, T. H., Huang, Y. H., Yang, K. D., Lin, H. C., & Cheng, J. K. (2021). Bio-scaffolds as cell or exosome carriers for nerve injury repair. International Journal of Molecular Sciences, 22(24). https://doi.org/10.3390/ijms222413347
Qiao, W., Lu, L., Wu, G., An, X., Li, D., & Guo, J. (2019). DPSCs seeded in acellular nerve grafts processed by Myroilysin improve nerve regeneration. Journal of Biomaterials Applications, 33(6), 819–833. https://doi.org/10.1177/0885328218812136
Rajan, T. S., Giacoppo, S., Diomede, F., Ballerini, P., Paolantonio, M., Marchisio, M., Piattelli, A., Bramanti, P., Mazzon, E., & Trubiani, O. (2016). The secretome of periodontal ligament stem cells from MS patients protects against EAE. Science and Reports, 6, 38743. https://doi.org/10.1038/srep38743
Rao, F., Zhang, D., Fang, T., Lu, C., Wang, B., Ding, X., Wei, S., Zhang, Y., Pi, W., Xu, H., Wang, Y., Jiang, B., & Zhang, P. (2019). Exosomes from human gingiva-derived mesenchymal stem cells combined with biodegradable chitin conduits promote rat sciatic nerve regeneration. Stem Cells International, 2019, 2546367. https://doi.org/10.1155/2019/2546367
Ray, W. Z., & Mackinnon, S. E. (2010). Management of nerve gaps: Autografts, allografts, nerve transfers, and end-to-side neurorrhaphy. Experimental Neurology, 223(1), 77–85. https://doi.org/10.1016/j.expneurol.2009.03.031
Sanchez Rezza, A., Kulahci, Y., Gorantla, V. S., Zor, F., & Drzeniek, N. M. (2022). Implantable biomaterials for peripheral nerve regeneration-technology trends and translational tribulations. Frontiers in Bioengineering and Biotechnology, 10, 863969. https://doi.org/10.3389/fbioe.2022.863969
Sanen, K., Martens, W., Georgiou, M., Ameloot, M., Lambrichts, I., & Phillips, J. (2017). Engineered neural tissue with Schwann cell differentiated human dental pulp stem cells: Potential for peripheral nerve repair? Journal of Tissue Engineering and Regenerative Medicine, 11(12), 3362–3372. https://doi.org/10.1002/term.2249
Sasaki, R., Aoki, S., Yamato, M., Uchiyama, H., Wada, K., Ogiuchi, H., Okano, T., & Ando, T. (2011). PLGA artificial nerve conduits with dental pulp cells promote facial nerve regeneration. Journal of Tissue Engineering and Regenerative Medicine, 5(10), 823–830. https://doi.org/10.1002/term.387
Scheib, J., & Hoke, A. (2013). Advances in peripheral nerve regeneration. Nature Reviews. Neurology, 9(12), 668–676. https://doi.org/10.1038/nrneurol.2013.227
Selim, O. A., Lakhani, S., Midha, S., Mosahebi, A., & Kalaskar, D. M. (2022). Three-dimensional engineered peripheral nerve: Toward a new era of patient-specific nerve repair solutions. Tissue Engineering. Part B, Reviews, 28(2), 295–335. https://doi.org/10.1089/ten.TEB.2020.0355
Seo, B. M., Miura, M., Gronthos, S., Bartold, P. M., Batouli, S., Brahim, J., Young, M., Robey, P. G., Wang, C. Y., & Shi, S. (2004). Investigation of multipotent postnatal stem cells from human periodontal ligament. The Lancet, 364(9429), 149–155. https://doi.org/10.1016/S0140-6736(04)16627-0
Shang, L., Shao, J., & Ge, S. (2021). Immunomodulatory functions of oral mesenchymal stem cells: Novel force for tissue regeneration and disease therapy. Journal of Leukocyte Biology, 110(3), 539–552. https://doi.org/10.1002/jlb.3mr0321-766r
Shi, Q., Qian, Z., Liu, D., Sun, J., Wang, X., Liu, H., Xu, J., & Guo, X. (2017). GMSC-derived exosomes combined with a chitosan/silk hydrogel sponge accelerates wound healing in a diabetic rat skin defect model. Frontiers in Physiology, 8, 904. https://doi.org/10.3389/fphys.2017.00904
Simonovic, J., Toljic, B., Nikolic, N., Peric, M., Vujin, J., Panajotovic, R., Gajic, R., Bekyarova, E., Cataldi, A., Parpura, V., & Milasin, J. (2018). Differentiation of stem cells from apical papilla into neural lineage using graphene dispersion and single walled carbon nanotubes. Journal of Biomedical Materials Research. Part A, 106(10), 2653–2661. https://doi.org/10.1002/jbm.a.36461
Solis-Castro, O. O., Rivolta, M. N., & Boissonade, F. M. (2022). Neural Crest-Derived Stem Cells (NCSCs) obtained from Dental-Related Stem Cells (DRSCs): A literature review on current knowledge and directions toward translational applications. International Journal of Molecular Sciences, 23(5). https://doi.org/10.3390/ijms23052714
Soman, S. S., & Vijayavenkataraman, S. (2020). Perspectives on 3D bioprinting of peripheral nerve conduits. International Journal of Molecular Sciences, 21(16). https://doi.org/10.3390/ijms21165792
Song, S., Li, Y., Huang, J., Cheng, S., & Zhang, Z. (2023). Inhibited astrocytic differentiation in neural stem cell-laden 3D bioprinted conductive composite hydrogel scaffolds for repair of spinal cord injury. Biomaterials Advances, 148, 213385. https://doi.org/10.1016/j.bioadv.2023.213385
Sonoyama, W., Liu, Y., Fang, D., Yamaza, T., Seo, B. M., Zhang, C., Liu, H., Gronthos, S., Wang, C. Y., Wang, S., & Shi, S. (2006). Mesenchymal stem cell-mediated functional tooth regeneration in swine. PLoS One, 1(1), e79. https://doi.org/10.1371/journal.pone.0000079
Stang, F., Fansa, H., Wolf, G., Reppin, M., & Keilhoff, G. (2005). Structural parameters of collagen nerve grafts influence peripheral nerve regeneration. Biomaterials, 26(16), 3083–3091. https://doi.org/10.1016/j.biomaterials.2004.07.060
Stewart, J. D. (2003). Peripheral nerve fascicles: Anatomy and clinical relevance. Muscle and Nerve, 28(5), 525–541. https://doi.org/10.1002/mus.10454
Sugimura-Wakayama, Y., Katagiri, W., Osugi, M., Kawai, T., Ogata, K., Sakaguchi, K., & Hibi, H. (2015). Peripheral nerve regeneration by secretomes of stem cells from human exfoliated deciduous teeth. Stem Cells Dev, 24(22), 2687–2699. https://doi.org/10.1089/scd.2015.0104
Sunderland, S. (1990). The anatomy and physiology of nerve injury. Muscle and Nerve, 13(9), 771–784. https://doi.org/10.1002/mus.880130903
Takaoka, S., Uchida, F., Ishikawa, H., Toyomura, J., Ohyama, A., Watanabe, M., Matsumura, H., Marushima, A., Iizumi, S., Fukuzawa, S., Ishibashi-Kanno, N., Yamagata, K., Yanagawa, T., Matsumaru, Y., & Bukawa, H. (2022). Transplanted neural lineage cells derived from dental pulp stem cells promote peripheral nerve regeneration. Human Cell, 35(2), 462–471. https://doi.org/10.1007/s13577-021-00634-9
Takeuchi, H., Ikeguchi, R., Aoyama, T., Oda, H., Yurie, H., Mitsuzawa, S., Tanaka, M., Ohta, S., Akieda, S., Miyazaki, Y., Nakayama, K., & Matsuda, S. (2020). A scaffold-free Bio 3D nerve conduit for repair of a 10-mm peripheral nerve defect in the rats. Microsurgery, 40(2), 207–216. https://doi.org/10.1002/micr.30533
Tapp, M., Wenzinger, E., Tarabishy, S., Ricci, J., & Herrera, F. A. (2019). The epidemiology of upper extremity nerve injuries and associated cost in the US emergency departments. Annals of Plastic Surgery, 83(6), 676–680. https://doi.org/10.1097/SAP.0000000000002083
Terenghi, G. (1999). Peripheral nerve regeneration and neurotrophic factors. Journal of Anatomy, 194((Pt 1)(Pt 1)), 1–14. https://doi.org/10.1046/j.1469-7580.1999.19410001.x
Tomic, S., Djokic, J., Vasilijic, S., Vucevic, D., Todorovic, V., Supic, G., & Colic, M. (2011). Immunomodulatory properties of mesenchymal stem cells derived from dental pulp and dental follicle are susceptible to activation by toll-like receptor agonists. Stem Cells and Development, 20(4), 695–708. https://doi.org/10.1089/scd.2010.0145
Tsutsui, T. W. (2020). Dental pulp stem cells: Advances to applications. Stem Cells Cloning, 13, 33–42. https://doi.org/10.2147/SCCAA.S166759
Väänänen, H. K. (2005). Mesenchymal stem cells. Annals of Medicine, 37(7), 469–479. https://doi.org/10.1080/07853890500371957
Vijayavenkataraman, S. (2020). Nerve guide conduits for peripheral nerve injury repair: A review on design, materials and fabrication methods. Acta Biomaterialia, 106, 54–69. https://doi.org/10.1016/j.actbio.2020.02.003
Walsh, S., & Midha, R. (2009). Practical considerations concerning the use of stem cells for peripheral nerve repair. Neurosurgical Focus, 26(2), E2. https://doi.org/10.3171/FOC.2009.26.2.E2
Wang, D., Wang, Y., Tian, W., & Pan, J. (2019). Advances of tooth-derived stem cells in neural diseases treatments and nerve tissue regeneration. Cell Proliferation, 52(3), e12572. https://doi.org/10.1111/cpr.12572
Wang, M. L., Rivlin, M., Graham, J. G., & Beredjiklian, P. K. (2019). Peripheral nerve injury, scarring, and recovery. Connective Tissue Research, 60(1), 3–9. https://doi.org/10.1080/03008207.2018.1489381
Yalvaç, M. E., Ramazanoglu, M., Tekguc, M., Bayrak, O. F., Shafigullina, A. K., Salafutdinov, I. I., Blatt, N. L., Kiyasov, A. P., Sahin, F., Palotás, A., & Rizvanov, A. A. (2010). Human tooth germ stem cells preserve neuro-protective effects after long-term cryo-preservation. Current Neurovascular Research, 7(1), 49–58. https://doi.org/10.2174/156720210790820181
Yamaza, T., Kentaro, A., Chen, C., Liu, Y., Shi, Y., Gronthos, S., Wang, S., & Shi, S. (2010). Immunomodulatory properties of stem cells from human exfoliated deciduous teeth. Stem Cell Research & Therapy, 1(1), 5. https://doi.org/10.1186/scrt5
Yan, Y., Yao, R., Zhao, J., Chen, K., Duan, L., Wang, T., Zhang, S., Guan, J., Zheng, Z., Wang, X., Liu, Z., Li, Y., & Li, G. (2022). Implantable nerve guidance conduits: Material combinations, multi-functional strategies and advanced engineering innovations. Bioactive Materials, 11, 57–76. https://doi.org/10.1016/j.bioactmat.2021.09.030
Yang, J., Yang, K., Man, W., Zheng, J., Cao, Z., Yang, C. Y., Kim, K., Yang, S., Hou, Z., Wang, G., & Wang, X. (2023). 3D bio-printed living nerve-like fibers refine the ecological niche for long-distance spinal cord injury regeneration. Bioactive Materials, 25, 160–175. https://doi.org/10.1016/j.bioactmat.2023.01.023
Yao, Z., Yan, L. W., Qiu, S., He, F. L., Gu, F. B., Liu, X. L., Qi, J., & Zhu, Q. T. (2019). Customized scaffold design based on natural peripheral nerve fascicle characteristics for biofabrication in tissue regeneration. BioMed Research International, 2019, 3845780. https://doi.org/10.1155/2019/3845780
Yao, Z., Yan, L. W., Wang, T., Qiu, S., Lin, T., He, F. L., Yuan, R. H., Liu, X. L., Qi, J., & Zhu, Q. T. (2018). A rapid micro-magnetic resonance imaging scanning for three-dimensional reconstruction of peripheral nerve fascicles. Neural Regeneration Research, 13(11), 1953–1960. https://doi.org/10.4103/1673-5374.238718
Yousefi, F., Lavi Arab, F., Nikkhah, K., Amiri, H., & Mahmoudi, M. (2019). Novel approaches using mesenchymal stem cells for curing peripheral nerve injuries. Life Sciences, 221, 99–108. https://doi.org/10.1016/j.lfs.2019.01.052
Yu, S., Zhao, Y., Ma, Y., & Ge, L. (2016). Profiling the secretome of human stem cells from dental apical papilla. Stem Cells and Development, 25(6), 499–508. https://doi.org/10.1089/scd.2015.0298
Yu, X., Zhang, T., & Li, Y. (2020). 3D printing and bioprinting nerve conduits for neural tissue engineering. Polymers (Basel), 12(8). https://doi.org/10.3390/polym12081637
Yurie, H., Ikeguchi, R., Aoyama, T., Kaizawa, Y., Tajino, J., Ito, A., Ohta, S., Oda, H., Takeuchi, H., Akieda, S., Tsuji, M., Nakayama, K., & Matsuda, S. (2017). The efficacy of a scaffold-free Bio 3D conduit developed from human fibroblasts on peripheral nerve regeneration in a rat sciatic nerve model. PLoS One, 12(2), e0171448. https://doi.org/10.1371/journal.pone.0171448
Zhang, Q., Burrell, J. C., Zeng, J., Motiwala, F. I., Shi, S., Cullen, D. K., & Le, A. D. (2022). Implantation of a nerve protector embedded with human GMSC-derived Schwann-like cells accelerates regeneration of crush-injured rat sciatic nerves. Stem Cell Research & Therapy, 13(1), 263. https://doi.org/10.1186/s13287-022-02947-4
Zhang, Q., Nguyen, P., Burrell, J. C., Zeng, J., Shi, S., Shanti, R. M., Kulischak, G., Cullen, D. K., & Le, A. D. (2021). Harnessing 3D collagen hydrogel-directed conversion of human GMSCs into SCP-like cells to generate functionalized nerve conduits. NPJ Regenerative Medicine, 6(1), 59. https://doi.org/10.1038/s41536-021-00170-y
Zhang, Q., Nguyen, P., Xu, Q., Park, W., Lee, S., Furuhashi, A., & Le, A. D. (2017). Neural progenitor-like cells induced from human gingiva-derived mesenchymal stem cells regulate myelination of Schwann cells in rat sciatic nerve regeneration. Stem Cells Translational Medicine, 6(2), 458–470. https://doi.org/10.5966/sctm.2016-0177
Zhang, Q., Nguyen, P. D., Shi, S., Burrell, J. C., Cullen, D. K., & Le, A. D. (2018). 3D bio-printed scaffold-free nerve constructs with human gingiva-derived mesenchymal stem cells promote rat facial nerve regeneration. Science and Reports, 8(1), 6634. https://doi.org/10.1038/s41598-018-24888-w
Zhang, Q., Nguyen, P. D., Shi, S., Burrell, J. C., Xu, Q., Cullen, K. D., & Le, A. D. (2018). Neural crest stem-like cells non-genetically induced from human gingiva-derived mesenchymal stem cells promote facial nerve regeneration in rats. Molecular Neurobiology, 55(8), 6965–6983. https://doi.org/10.1007/s12035-018-0913-3
Zhang, Q., Shi, S., Liu, Y., Uyanne, J., Shi, Y., Shi, S., & Le, A. D. (2009). Mesenchymal stem cells derived from human gingiva are capable of immunomodulatory functions and ameliorate inflammation-related tissue destruction in experimental colitis. The Journal of Immunology, 183(12), 7787–7798. https://doi.org/10.4049/jimmunol.0902318
Zhang, Q. Z., Su, W. R., Shi, S. H., Wilder-Smith, P., Xiang, A. P., Wong, A., Nguyen, A. L., Kwon, C. W., & Le, A. D. (2010). Human gingiva-derived mesenchymal stem cells elicit polarization of m2 macrophages and enhance cutaneous wound healing. Stem Cells, 28(10), 1856–1868. https://doi.org/10.1002/stem.503
Zhang, R. C., Du, W. Q., Zhang, J. Y., Yu, S. X., Lu, F. Z., Ding, H. M., Cheng, Y. B., Ren, C., & Geng, D. Q. (2021). Mesenchymal stem cell treatment for peripheral nerve injury: A narrative review. Neural Regeneration Research, 16(11), 2170–2176. https://doi.org/10.4103/1673-5374.310941
Zheng, Y. H., Xiong, W., Su, K., Kuang, S. J., & Zhang, Z. G. (2013). Multilineage differentiation of human bone marrow mesenchymal stem cells in vitro and in vivo. Experimental and Therapeutic Medicine, 5(6), 1576–1580. https://doi.org/10.3892/etm.2013.1042
Zhu, S., Zhu, Q., Liu, X., Yang, W., Jian, Y., Zhou, X., He, B., Gu, L., Yan, L., Lin, T., Xiang, J., & Qi, J. (2016). Three-dimensional reconstruction of the microstructure of human acellular nerve allograft. Science and Reports, 6, 30694. https://doi.org/10.1038/srep30694
Zigmond, R. E., & Echevarria, F. D. (2019). Macrophage biology in the peripheral nervous system after injury. Progress in Neurobiology, 173, 102–121. https://doi.org/10.1016/j.pneurobio.2018.12.001
Funding
This work was supported by NIH/NIDCR R21DE029926-01 (AL) and the Schoenleber funding support (to AL and QZ).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Q. Y. M.: Writing the original draft; J. B. and Q. Z. Z.: revising & editing; A. L. and Q. Z.: Supervision. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethical Approval
Not applicable.
Consent to Participate
Not applicable.
Consent to Publish
All authors agree to the submission and publication of the manuscript.
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Meng, Q., Burrell, J.C., Zhang, Q. et al. Potential Application of Orofacial MSCs in Tissue Engineering Nerve Guidance for Peripheral Nerve Injury Repair. Stem Cell Rev and Rep 19, 2612–2631 (2023). https://doi.org/10.1007/s12015-023-10609-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12015-023-10609-y