Abstract
The association between the serum essential metal elements (magnesium, iron, copper, zinc, and calcium) and thyroid nodules is still inconsistent. The current study aims to investigate the relationship of metal elements with thyroid nodules and their malignant tendency. A total of 6480 Chinese euthyroid adults were included in our study. We collect basic information through questionnaires and medical checkups. We diagnose thyroid nodules by ultrasound and detect serum trace metal concentrations by using an automatic biochemical analyzer. Binary and multinomial logistic regressions were used to investigate the associations. As a result, we found that serum copper concentrations were positively associated with thyroid nodules in the second, third, and fourth quartiles, compared to the first quartile (P = 0.024, P = 0.016, P = 0.032) in women and P for trend is 0.038. There is a significant sex-specific association between copper concentrations and thyroid nodules (P for interaction = 0.009). The results of the multinomial logistic regression analyses indicate high serum calcium and magnesium concentrations emerged as consistent risk factors for thyroid nodules in both genders, whereas low zinc was a sex-specific factor. We also observed significant sex interactions in the relationships of magnesium (P for interaction = 0.043) with thyroid nodules with malignant tendency among participants with thyroid nodules. In conclusion, our study suggests that gender is an important factor when studying the association between serum metals and thyroid nodules. The imbalance of selected metal elements (calcium, copper, zinc, and magnesium) may relate to thyroid nodules and their malignant tendency, and future prospective studies are needed to further confirm the associations.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Thyroid nodules are local thyroid space-occupying lesions caused by various reasons, which are common diseases of the endocrine system with a prevalence in the general population as high as 67% [1]. Although only 5% of nodules are malignant, they have attracted public attention due to the psychological burden and anxiety they cause to patients [2, 3]. Pieces of evidences showed that imbalances of metal elements in the blood may contribute to the development of thyroid malignancies [4]. It is thought that nutritional deficiencies, exposure to various environmental substances, and changes in dietary habits may also be factors contributing to the increased incidence of thyroid nodules [5]. The use of dietary supplements, especially those containing vitamin/mineral supplements (VMS), is becoming increasingly prevalent which may lead to simultaneous supplementation with essential metal elements [6]. Supplements are widely used in Europe and the USA [7, 8]. Currently, many types of dietary supplements, especially mineral supplements, are also marketed and available in China now [9, 10]. There is no doubt that serum metal element concentrations are influenced by dietary supplements, especially mineral supplements.
Serum metal element concentrations including calcium, copper, iron, magnesium, and zinc have been applied in daily clinical work. These metals are mainly obtained through the daily diets or mineral supplements. They have an important effect on endocrine homeostasis and play an essential role in cell proliferation and differentiation, intercellular signal transduction, metabolic regulation, oxidation, and reduction reactions [11,12,13]. Metal elements are essential to the physiological processes of the thyroid gland, and there were higher levels of many metal elements in the thyroid gland [14]. Several small-sample studies have focused on the relationship between serum metal element levels and thyroid nodules, but the relationship is still inconsistent. In a study with 56 participants, it was shown that low serum concentration of calcium, magnesium, zinc, copper, and selenium was associated with an increased risk of developing nodular goiter [15]. In another study, copper and manganese levels were significantly higher in patients with multinodular goiter, whereas the differences in zinc and iron concentrations between the two groups were not found [16].
In this current study, we conducted a cross-sectional study with a large sample size to explore the sex-specific association of metal elements with thyroid nodules and their malignant tendency by using logistic regression. The outcomes of this research may offer some suggestions for micronutrient supplementation and further prevention of thyroid nodules.
Materials and Methods
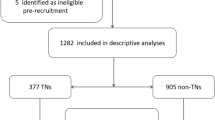
Study Population
This current study has been registered at www.chictr.org.cn (ChiCTR2100054130). A total of 13535 adults (aged ≥ 18 years) from Shunde District, Guangdong Province, China, were selected for this study, in which women who are pregnant or have recently given birth were not included. Participants who had taken mineral supplements in the past 3 months and had missing data on serum metal concentrations (n = 4350); those with missing ultrasound data (n = 263); those who had received iodinated contrast agent injections; taken thyroid-affecting medications such as antithyroid drugs, thyroid hormone supplement medications, and lithium carbonate; had a history of thyroid surgery; or had abnormal thyroid function (n = 2442) were all excluded from this study (Fig. 1). The purpose and procedures of the survey were explained to the participants, and they provided informed consent before enrollment. The ethics committee of Shunde Hospital of Southern Medical University approved the research protocol (20211103).
Data Collection
We surveyed all participants in the form of self-made questionnaire, including general demographic characteristics (sex, age, education) and behavior of lifestyle (alcohol consumption status, smoking status, iodine intake status, thyroid surgery history, medication history including anti-glycemic drugs and antihypertensive drugs). For anthropometric parameters, we measured the blood pressure, height, and weight of the participants according to standard methods. We also collected their biochemical data at the same time.
Laboratory Assays
Participants’ fasting blood was collected by intravenous sampling between 7 and 8 am. The blood was cooled to 4 °C and transported to a center laboratory (certified by the College of American Pathologists) within 2–4 h. Total cholesterol (TC), triglycerides (TG), low-density lipoprotein (LDL), high-density lipoprotein (HDL), and serum essential metal elements (magnesium, iron, copper, zinc, and calcium) were measured by the automatic biochemical analyzer (Beckman Coulter AU580). Thyroid peroxidase antibodies (TPOAb), thyroid-stimulating hormone (TSH), free triiodothyronine (FT3), and free thyroxine (FT4) levels were detected by the fully automated microparticle chemiluminescence instrument (Beckman Coulter UniCel DxI 800). Glycated hemoglobin (HbA1c) was assessed by the automatic glycated hemoglobin analyzer (TOSOH, HLC-723 G8, Tokyo, Japan). The reference range below is for normal thyroid function: FT3 (3.53–7.37 pmol/L), FT4 (7.98–16.02 pmol/L), and TSH (0.56–5.91 mIU/mL). A value of TPOab > 4.9 IU/mL was defined as positive. The following are the laboratory reference ranges for serum metal elements: magnesium (0.75–1.02 mmol/L); copper (10.0–24.0 μmol/L); zinc (10.7–17.7 μmol/L); iron (7–30 μmol/L); calcium (2.15–2.57 mmol/L).
Definition
Definition of Thyroid Nodules and Their Malignant Tendency
Ultrasound was performed using B-mode US imaging (MX7, Mindray Shenzhen, P.R. China) with a 13-MHz linear array probe and we defined thyroid nodules as present when the size of the nodule was found to be ≥ 2 mm on ultrasound by some highly trained technicians. Ultrasound was also used to observe whether the subjects had different types of thyroid nodules including nodules larger than 1 cm in diameter, multiple nodules, calcified nodules, and vertically growing nodules. Although fine needle puncture is the gold standard for the diagnosis of malignant nodules for people with thyroid nodules, due to its invasiveness, it is difficult to be popularized in large cohort studies. We therefore made a definition of nodules with a malignant tendency according to ultrasound parameters and guidelines. In a study assessing the predictive accuracy of thyroid nodule benignity and malignancy using ultrasound, Brito et al. found that both calcified nodules and vertically oriented nodules were somewhat predictive of malignant thyroid nodules. Additionally, the Chinese guidelines for ultrasound-based malignancy risk stratification of thyroid nodules indicate that calcified nodules and vertically growing nodules exhibit higher malignant tendency. Thus, vertical growth of nodules or calcification of nodules, or both were defined as thyroid nodules with a malignant tendency [17, 18].
Definition of Covariates
We categorized participants into three groups based on age: young people (18–40), middle-aged people (41–60), and elderly people (> 60) [19, 20]. Smoking status was divided into three groups: never smoke, former smoke (more than 6 months before quitting), and current smoke (used to smoke at least 100 cigarettes and smoking currently) [21]. Alcoholics were defined as men who consumed ≥ 210 g of alcohol per week or women who drank ≥ 140 g of alcohol per week [22]. We classify BMI into four categories, which are underweight (< 18.5 kg/m2), normal (18.5–24.9 kg/m2), overweight (25–29.9 kg/m2), and obese (≥ 30 kg/m2) groups [23]. Participants who met a systolic blood pressure (SBP) ≥ 140 mmHg or diastolic blood pressure (DBP) ≥ 90 mmHg or had a history of taking antihypertensive medication were considered hypertensive. Subjects with a previous diagnosis of diabetes mellitus or HbA1c ≥ 6.5% were considered diabetic [22]. Dyslipidemia was defined as having either or a combination of TG, LDL-C, HDL-C, and TC representing ≥ 2.26, ≥ 4.14, < 1.04, and ≥ 6.22 mmol/L, respectively, or a current use of lipid-modifying medications [24]. The iodine intake status of the subjects was divided into three groups according to the consumption of iodized salt, non-iodized salt, and both.
Statistical Analyses
We present continuous variables with mean (standard deviation). As for the categorical variables, we present them in the form of percentages. We used one-way analyses of variance (ANOVA) to compare normally distributed continuous variables between the three groups. Pearson χ2 test was used to analyze the difference between categorical variables.
We use logistic regression to analyze the association between serum metal concentration and thyroid nodules and their malignant tendency. The results of the logistic regression are presented in ORs and 95% confidential intervals (95% CIs). We divided the concentration of each metal into quartiles and the first quartile was used as reference. The other quartiles at individual metal levels were compared with the first quartile to discover the relationship between each serum metal element and thyroid nodules. P values for trend were calculated across serum metal element quartiles using the Wald test, based on a score derived from the median value of each baseline quartile [25]. In addition, restricted cubic spline (RCS) analyses were conducted using a three-knot restricted cubic spline function (with knots at the 10th, 50th, and 90th percentiles) to further assess the prediction-response relationship between metals and thyroid nodule risk. Furthermore, multinomial logistic regression analyses were conducted with gender stratification. Participants without thyroid nodules were designated as the reference group, allowing assessment of the relationships between various thyroid nodule types and serum metal.
Analyses including logistic regression and RCS applied in this study were adjusted for age categories, sex, smoking status, excessive alcohol consumption, iodine intake status, BMI categories, diabetes, hypertension, dyslipidemia, TSH, and TPOab. In the gender-stratified analyses, the model adjusted for all potential confounders except sex.
We analyzed the data using IBM SPSS Statistics (version 27) and R (version 4.2.1). A two-tailed P < 0.05 was considered statistically significant.
Results
The Characteristics of the Research Subjects
A total of 6480 subjects were enrolled, and their general demographic and clinical features are summarized in Table 1. A total of 2251 participants were found to have thyroid nodules. Among participants with thyroid nodules, the incidence of thyroid nodules with a malignant tendency was 13.06%. The mean age of the survey population was 46.62 years and the mean BMI was 23.77 kg/m2. Compared to participants without thyroid nodules (NCs), the proportion of women and older individuals was significantly higher among those with benign thyroid nodules (BTNs) and malignant thyroid nodules (MTNs). BTNs and MTNs groups also exhibited a greater prevalence of comorbidities including diabetesand hypertension. Notable differences were observed in thyroid function tests between groups, with higher TPO antibody levels and lower TSH levels in the BTNs and MTNs. FT3 and FT4 levels conversely trended higher in these same groups. Additional disparities were revealed in serum biomarkers, as the BTNs and MTNs groups displayed elevated total cholesterol, LDL, copper, calcium, and magnesium, and decreased zinc relative to NCs. Then we further separated the men and women groups for analyses and found serum copper and calcium levels were higher in women with thyroid nodules versus women without thyroid nodules. Men with thyroid nodules have lower calcium levels (Table 2).
Associations Between Serum Essential Metal Elements and the Appearance of Thyroid Nodules by Logistic Regression
We first studied the association between the presence of thyroid nodules and the concentration of each metal element by logistic regression. The metal levels were processed as categorical variables and classified into four quartiles, and the lowest concentration (first quartile) group was used as a reference. Forest plots (Fig. 2) were created to vividly present the outcome’s odds ratios (OR), 95% confidence interval (CI), and P values to better reflect the relationship between individual metals and thyroid nodules. As a result, in the total population, the OR for thyroid nodules obtained from the second quartile of serum copper levels compared to the lowest quartile was 1.197 (P = 0.020, 95% CI: 1.054, 1.360). We observed a significant sex interaction in the relationship between copper and the appearance of thyroid nodules (P for interaction = 0.009). After stratifying the analyses by gender, we found that serum copper concentrations were particularly statistically significant in the second, third, and fourth quartiles, compared to the first quartile (P = 0.024, P = 0.016, P = 0.032) in women and P for trend is 0.038. And the risk of thyroid nodules in women in the highest quartile of copper levels is 1.241 folds (P = 0.032, 95% CI: 1.052, 1.464). We further utilized restricted cubic spline modeling to flexibly visualize and assess the prediction-response relationship between serum metals and thyroid nodule risk. In women, the association between copper and thyroid nodules was largely consistent with the logistic regression results (P for all = 0.016, P for nonlinear = 0.024) (Supplementary Fig. 1). Interestingly, in men, serum calcium concentrations showed a positive correlation with the occurrence of thyroid nodules in the second quartile when compared with the lowest quartile (P = 0.050, OR = 1.280, 95% CI: 1.036, 1.583).
Relationships between thyroid nodules and serum metals by logistic analyses in the total population (a), female (b), and male (c). The model was adjusted for age categories, sex, smoking status, excessive alcohol consumption, iodine intake status, BMI categories, diabetes, hypertension, dyslipidemia, TSH, and TPOab. In the gender-stratified analyses, the model adjusted for all potential confounders except sex. Abbreviations: Cu, copper; Zn, zinc; Ca, calcium; Fe, iron; Mg, magnesium; TNs ( +), participants with thyroid nodules; TNs ( −), participants without thyroid nodules
Associations Between Serum Metal Elements and Two Types of Thyroid Nodules by Multinomial Logistic Analyses
Next, we set participants without thyroid nodules as the reference group and performed multinomial logistic regression analyses to assess the relationships between two types of thyroid nodules (benign thyroid nodules and thyroid nodules with malignant tendency) and serum metal elements stratified by gender (Table 3). For women, elevated serum calcium levels were positively associated with both benign thyroid nodules (OR = 5.310, P < 0.001) and thyroid nodules with malignant tendency (OR = 6.600, P = 0.009) after adjusting for confounders. In addition, higher serum magnesium (OR = 45.859, P < 0.001) and lower serum zinc (OR = 0.935, P = 0.002) were also associated with increased benign thyroid nodule risk in women. In men, elevated serum calcium levels (OR = 2.880, P = 0.030) and magnesium levels (OR = 11.72, P = 0.002) were positively associated with the risk of benign thyroid nodules after accounting for confounding variables. In summary, higher serum calcium and magnesium concentrations emerged as consistent risk factors for thyroid nodules in both genders, whereas lower zinc was a factor associated with increased risk only in women.
Associations Between Serum Metal Elements and Malignant Thyroid Nodules Among Participants with Thyroid Nodules by Logistic Analyses
Last, we also studied the association between serum concentrations of different metals and thyroid nodules with malignant tendency among participants with thyroid nodules (Fig. 3). In the overall population, serum magnesium concentration showed a negative association with the appearance of thyroid nodules with malignant tendency. The ORs of quartiles 2, 3, and 4 were lowered to 0.649 (P = 0.015, 95% CI: 0.484, 0.869), 0.598 (P = 0.004, 95% CI: 0.447, 0.800), and 0.452 (P < 0.001, 95% CI: 0.332, 0.612) respectively. After further gender stratification analyses, we found that in women, the negative association of serum magnesium on thyroid nodules with malignant tendency still existed. The OR for the highest quartile versus the lowest magnesium quartile was 0.400 (95% CI: 0.274–0.579, P < 0.001) and the trend was significant (P for trend < 0.001). We observed significant sex interactions in the relationships of magnesium (P for interaction = 0.043) with thyroid nodules with malignant tendency.
Relationships between malignant thyroid nodules and serum metals among participants with thyroid nodules by logistic analyses in the total population (a), female (b), and male (c). The model was adjusted for age categories, sex, smoking status, excessive alcohol consumption, iodine intake status, BMI categories, diabetes, hypertension, dyslipidemia, TSH, and TPOab. In the gender-stratified analyses, the model was adjusted for all potential confounders except sex. Abbreviations: BTN, benign thyroid nodules; MTN, thyroid nodules with malignant tendency; Cu, copper; Zn, zinc; Ca, calcium; Fe, iron; Mg, magnesium
Discussion
In this current study, we studied the correlation of metal elements with thyroid nodules and their malignant tendency. Gopinathan et al. concluded that microcalcifications have the highest accuracy (76%), specificity (44–95%), and positive predictive value (77.9%) for detecting malignancy in a thyroid nodule and vertically growing nodules have a higher likelihood of malignancy with accuracy, specificity, and sensitivity in the range of 67%, 92%, and 40% respectively. Despite the inability to pathologically confirm the proportion of thyroid cancer in participants with nodules displaying malignant tendencies, the utilization of ultrasound to identify such nodules enabled physicians to identify patients requiring more vigilant monitoring or potential biopsy [26]. Consequently, to some extent, this may hold promise in facilitating early prevention of thyroid cancer.
Overall, we found a positive association between serum copper concentrations and the prevalence of thyroid nodules as well as a negative association between serum zinc concentrations and benign thyroid nodules in women by logistic regression analyses. These significant associations were observed in women but not in man. That is, we found a sex-specific association between these serum essential metal elements and thyroid nodules. Differences in hormonal, genetic, metabolic, anatomical, and epigenetic factors in different gender populations may contribute to their different susceptibility and response to metals [27]. As it has been shown in a study, a positive association between increased levels of metal mixtures and cancer mortality was only observed in women [23]. And our previous study showed there is a sex-specific association between urinary metal mixtures and the prevalence of NAFLD in US adults [28]. Therefore, we should emphasize the effect of sex differences on the results when studying metals and human health.
The association between the essential metal elements and thyroid nodules is still unclear. Our current study shows that excessive copper is a risk factor for thyroid nodules, which is consistent with our previous findings [29]. Since the OR for copper was not the highest in the fourth quartile in the logistic regression analyses, a supplementary assessment of the prediction-response relationship was warranted [30]. We further utilized restricted cubic spline modeling to assess the prediction-response relationship between serum metals and thyroid nodule risk. The results were largely consistent with the logistic regression. Copper acts as an oxidation–reduction active element [31]. With regard to thyroid health, imbalances in Cu levels have been linked the development of goiter and other thyroid diseases [32]. Copper levels were significantly higher in patients with multinodular goiter in another study [16]. It has been suggested that serum copper has a negative effect on thyroid function [33]. Copper can promote cell proliferation by promoting Mek1-Erk physical interactions before Mek1 phosphorylation of Erk. This may be a potential mechanism by which high levels of blood copper promote the development of thyroid nodules [29].
Zinc is an important trace element for the synthesis and metabolism of thyroid hormones. Zinc deficiency or excess may affect thyroid function and lead to the development of thyroid nodules [34]. A case–control study found that urinary zinc levels were lower in patients with nodular goiter than in healthy controls, and that urinary zinc levels were negatively associated with the risk of developing nodular goiter [35]. In addition, a recent study has shown that microscopic calcifications of benign nodules contain essentially no zinc [36]. One cross-sectional study showed that patients with thyroid disease had relatively low blood zinc concentrations compared to healthy adult subjects [37]. In our study, we found that zinc concentration was negatively associated with benign nodules in women.
Magnesium is considered to play a role in preventing cancer including thyroid cancer. The results of Huaijin et al. demonstrated that patients with papillary thyroid carcinoma had lower serum magnesium levels than patients with benign nodules [38]. Our present findings suggest a positive correlation between serum magnesium levels and benign thyroid nodules [30]. Interestingly, participants with malignant thyroid nodules had lower serum magnesium levels than those with benign nodules which is agreed with the findings of Huaijin et al. Furthermore, we highlight the important role played by gender factors in the influence of serum magnesium on nodules with malignant tendency.
As for calcium, in cross-sectional studies, there was no statistically significant difference in serum calcium levels between benign thyroid nodules and controls with normal thyroid function [39]. However, Christopher et al. show that among short-term (< 5 years) users of calcium supplements, an increased risk of thyroid cancer was observed. The positive correlation between serum calcium and the presence of thyroid nodules without malignant tendency was also verified in our research. Last, studies on the association between iron and thyroid nodules are scarce. Sana et al. found that iron concentrations were significantly higher in children with goiter than in those without goiter [40]. However, no significant correlation was found between iron and thyroid nodules in our study.
The safety, efficacy, and effectiveness of dietary supplementation have been hotly debated. The positive effect of serum copper on the incidence of thyroid nodules as well as the negative effect of zinc on thyroid nodules with malignant tendency observed in our study will provide some suggestions for the application of dietary supplements in specific populations. Avoiding excessive intake of copper may have a preventive effect on the development of thyroid nodules, while supplementing with moderate amounts of zinc and avoiding excessive calcium intake may help prevent the development of thyroid nodules especially for women. In addition, our results may be helpful for clinical management. People with higher serum copper levels, especially women, should check themselves for thyroid nodules while adults with or without thyroid nodules who have low serum zinc concentration or high blood calcium and copper concentration should be followed up to screen for thyroid nodules at an early stage.
Although there are some advantages in our study, such as a large-sample community-based population and homogeneous detection, there are still some limitations. Firstly, biopsy remains the most reliable method for detecting malignant thyroid nodules. We could not perform fine-needle aspiration biopsy for all participants due to the large size of the study population. Thus, the proportion of pathological diagnosis of thyroid cancer in participants with malignant thyroid nodules is unclear. Secondly, as a cross-sectional study, this study is not able to confirm the causal relationship between serum metal elements and thyroid nodules. Future studies may focus on longitudinal observations and mechanistic explanations. Finally, the participants in this study were mainly from coastal areas, so the results may be biased by ethnicity and geographical location.
In conclusion, our study suggests that gender is an important factor when studying the association between serum metals and thyroid nodules. The imbalance of selected metal elements (calcium, copper, zinc, and magnesium) may relate to thyroid nodules and their malignant tendency and future prospective studies are needed to further confirm the associations.
Data Availability
The data supporting the study findings are available from the corresponding authors upon reasonable request.
References
Daniels K et al (2020) Machine learning by ultrasonography for genetic risk stratification of thyroid nodules. JAMA Otolaryngol Head Neck Surg 146(1):36–41. https://doi.org/10.1001/jamaoto.2019.3073
Kobaly K, Kim CS, Mandel SJ (2022) Contemporary management of thyroid nodules. Annu Rev Med 73:517–528. https://doi.org/10.1146/annurev-med-042220-015032
Chen P et al (2022) Exploring the research landscape of the past, present, and future of thyroid nodules. Front Med (Lausanne) 9:831346. https://doi.org/10.3389/fmed.2022.831346
Bibi K, Shah MH (2020) Appraisal of metal imbalances in the blood of thyroid cancer patients in comparison with healthy subjects. Biol Trace Elem Res 198(2):410–422. https://doi.org/10.1007/s12011-020-02088-w
Barrea L et al (2021) Nutritional status and follicular-derived thyroid cancer: an update. Crit Rev Food Sci Nutr 61(1):25–59. https://doi.org/10.1080/10408398.2020.1714542
Sicińska E et al (2019) Different socio-demographic and lifestyle factors can determine the dietary supplement use in children and adolescents in central-eastern Poland. Nutrients 11(3) https://doi.org/10.3390/nu11030658
Bailey RL et al (2011) Dietary supplement use in the United States, 2003–2006. J Nutr 141(2):261–266. https://doi.org/10.3945/jn.110.133025
Skeie G et al (2009) Use of dietary supplements in the European Prospective Investigation into Cancer and Nutrition calibration study. Eur J Clin Nutr 63(Suppl 4):S226–S238. https://doi.org/10.1038/ejcn.2009.83
Gu JH et al (2021) Association between pre-diagnostic dietary supplements intake and ovarian cancer survival: findings from a prospective cohort study in Chinese women. Front Nutr 8:758178. https://doi.org/10.3389/fnut.2021.758178
Tang L et al (2017) Consumption of dietary supplements by Chinese women during pregnancy and postpartum: a prospective cohort study. Matern Child Nutr 13(4) https://doi.org/10.1111/mcn.12435
Yang W et al (2022) Genome-wide association and Mendelian randomization study of blood copper levels and 213 deep phenotypes in humans. Commun Biol 5(1):405. https://doi.org/10.1038/s42003-022-03351-7
Cooper DS (2004) Subclinical thyroid disease: consensus or conundrum? Clin Endocrinol (Oxf) 60(4):410–412. https://doi.org/10.1111/j.1365-2265.2004.02031.x
Zhang H et al (2022) Disordered serum essential element levels are associated with increased risk of kidney tumors. Environ Sci Pollut Res Int 29(21):31675–31685. https://doi.org/10.1007/s11356-021-18201-y
Zaichick V, Tsyb AF, Vtyurin BM (1995) Trace elements and thyroid cancer. Analyst 120(3):817–821. https://doi.org/10.1039/an9952000817
Kravchenko VI et al (2020) Association between thyroid hormone status and trace elements in serum of patients with nodular goiter. Biol Trace Elem Res 196(2):393–399. https://doi.org/10.1007/s12011-019-01943-9
Giray B et al (2010) Trace elements status in multinodular goiter. J Trace Elem Med Biol 24(2):106–110. https://doi.org/10.1016/j.jtemb.2009.11.003
Brito JP et al (2014) The accuracy of thyroid nodule ultrasound to predict thyroid cancer: systematic review and meta-analysis. J Clin Endocrinol Metab 99(4):1253–1263. https://doi.org/10.1210/jc.2013-2928
Zhou J et al (2020) 2020 Chinese guidelines for ultrasound malignancy risk stratification of thyroid nodules: the C-TIRADS. Endocrine 70(2):256–279. https://doi.org/10.1007/s12020-020-02441-y
Glassock RJ, Rule AD (2012) The implications of anatomical and functional changes of the aging kidney: with an emphasis on the glomeruli. Kidney Int 82(3):270–277. https://doi.org/10.1038/ki.2012.65
Bennett JP et al (2022) Assessment of clinical measures of total and regional body composition from a commercial 3-dimensional optical body scanner. Clin Nutr 41(1):211–218. https://doi.org/10.1016/j.clnu.2021.11.031
Wan H et al (2020) Associations between abdominal obesity indices and diabetic complications: Chinese visceral adiposity index and neck circumference. Cardiovasc Diabetol 19(1):118. https://doi.org/10.1186/s12933-020-01095-4
Wan H et al (2022) Chronic lead exposure induces fatty liver disease associated with the variations of gut microbiota. Ecotoxicol Environ Saf 232:113257. https://doi.org/10.1016/j.ecoenv.2022.113257
Duan H et al (2022) Associations of uric acid with liver steatosis and fibrosis applying vibration controlled transient elastography in the United States: a nationwide cross-section study. Front Endocrinol (Lausanne) 13:930224. https://doi.org/10.3389/fendo.2022.930224
(2018) 2016 Chinese guidelines for the management of dyslipidemia in adults. J GeriatrCardiol 15(1):1–29. https://doi.org/10.11909/j.issn.1671-5411.2018.01.011
Veronese N et al (2022) Dietary acrylamide and incident osteoporotic fractures: an 8-year prospective cohort study. Aging Clin Exp Res 34(10):2441–2448. https://doi.org/10.1007/s40520-022-02214-9
Popova NM et al (2021b) Impact of the hypoechogenicity criteria on thyroid nodule malignancy risk stratification performance by different TIRADS systems. Cancers (Basel) 13(21). https://doi.org/10.3390/cancers13215581
Gade M, Comfort N, Re DB (2021) Sex-specific neurotoxic effects of heavy metal pollutants: epidemiological, experimental evidence and candidate mechanisms. Environ Res 201:111558. https://doi.org/10.1016/j.envres.2021.111558
Wan H et al (2022) Sex-specific associations of the urinary fourteen-metal mixture with NAFLD and liver fibrosis among US adults: a nationally representative study. Ecotoxicol Environ Saf 248:114306. https://doi.org/10.1016/j.ecoenv.2022.114306
Zeng H et al (2022) The association of thyroid nodules with blood trace elements identified in a cross-section study. Front Nutr 9:870873. https://doi.org/10.3389/fnut.2022.870873
Feng X et al (2023) Relationship of multiple metals mixture and osteoporosis in older Chinese women: an aging and longevity study. Environ Pollut 317:120699. https://doi.org/10.1016/j.envpol.2022.120699
Olin KL, Walter RM, Keen CL (1994) Copper deficiency affects selenoglutathione peroxidase and selenodeiodinase activities and antioxidant defense in weanling rats. Am J Clin Nutr 59(3):654–658. https://doi.org/10.1093/ajcn/59.3.654
Theophanides T, Anastassopoulou J (2002) Copper and carcinogenesis. Crit Rev Oncol Hematol 42(1):57–64. https://doi.org/10.1016/S1040-8428(02)00007-0
Kazi TG et al (2010) Interaction of copper with iron, iodine, and thyroid hormone status in goitrous patients. Biol Trace Elem Res 134(3):265–279. https://doi.org/10.1007/s12011-009-8478-7
Zhou Q et al (2022) Trace elements and the thyroid. Front Endocrinol (Lausanne) 13:904889. https://doi.org/10.3389/fendo.2022.904889
YildirimSimsir I, Cetinkalp S, Kabalak T (2020) Review of factors contributing to nodular goiter and thyroid carcinoma. Med Princ Pract 29(1):1–5. https://doi.org/10.1159/000503575
Gotnayer L et al (2023) Zinc in microscopic calcifications isolated from thyroid fine needle aspiration may serve as a biomarker of thyroid nodule malignancy: a promising proof-of-concept. Acta Biomater 161:275–284. https://doi.org/10.1016/j.actbio.2023.03.010
Mehl S et al (2020) Cross-sectional analysis of trace element status in thyroid disease. J Trace Elem Med Biol 58:126430. https://doi.org/10.1016/j.jtemb.2019.126430
Xu H et al (2023) The inverse association of serum magnesium with papillary thyroid cancer in thyroid nodules: a cross-sectional survey based on thyroidectomy population. Biol Trace Elem Res 201(7):3279–3289. https://doi.org/10.1007/s12011-022-03448-4
Bolat H, Erdoğan A (2022) Benign nodules of the thyroid gland and 25-hydroxy-vitamin D levels in euthyroid patients. Ann Saudi Med 42(2):83–88. https://doi.org/10.5144/0256-4947.2022.83
El-Fadeli S et al (2016) Effects of imbalance in trace element on thyroid gland from Moroccan children. Biol Trace Elem Res 170(2):288–293. https://doi.org/10.1007/s12011-015-0485-2
Acknowledgements
The authors thank all team members and participants in the cross-sectional study.
Funding
This work was supported by the National Natural Science Foundation of China (82200960, 82170800) and the Guangdong Basic and Applied Basic Research Foundation (2021A1515110682).
Author information
Authors and Affiliations
Contributions
Study concept and design: QM, JS, and HW. Literature searches: SL, YJ, HD, YH, XC, NY, XL, and DW. Statistical analysis: QM, YL, and GY. Drafting of the manuscript: QM, YL, and GY. Data collection: all authors. Critical revision: JS and HW. Study supervision: JS and HW. All authors contributed to the manuscript for important intellectual content and approved the submission.
Corresponding authors
Ethics declarations
Ethics Approval and Consent to Participate
The purpose and procedures of the survey were explained to the participants, and they provided informed consent before enrollment. The ethics committee of Shunde hospital of Southern Medical University approved the research protocol (20211103).
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Qintao Ma, Ying Li, and Genfeng Yu contributed equally to this work.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ma, Q., Li, Y., Yu, G. et al. Sex-Specific Associations of Five Serum Essential Metal Elements with Thyroid Nodules in Euthyroid Adults: a Cross‑sectional Study. Biol Trace Elem Res 202, 4357–4366 (2024). https://doi.org/10.1007/s12011-023-04024-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-023-04024-0