Abstract
The main aim of this study was to determine the levels of Al, Cd, Cr, Cu, Fe, Hg, Mn, Ni, Pb, and Zn in commercial canned and pouched cat foods (salmon, tuna, liver, fish, and other aquatic products) and assess the potential health risks to kitten (≤ 1 years old) and adult cats (≥ 1 years old) associated with the recommended average consumption rate of labels. The study was also aimed to adapt the health risk assessment method to animal health and to support clinical prevention and diagnosis. The detected levels of the metals were below the data from other studies, except the mean Fe in all and Pb levels in salmon and kitten foods. Target hazard quotient (THQ) and total target hazard quotient (TTHQ) values did not exceed 1. That means the studied metals do not pose a health risk for adult cats and kittens. Dietary Hg and Cd should also be considered in the differential diagnosis of cases with clinical or postmortem findings, especially regarding neurological, kidney, and liver tissues. In conclusion, although canned/pouched consumption does not pose a health risk with regard to metals, further studies of health risk assessment for other pollutants by this first adaptation method will be necessary.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Anthropogenic or naturally originated chemical pollutants like metals (such as aluminum [Al], cadmium [Cd], chromium [Cr], copper [Cu], iron [Fe], lead [Pb], manganese [Mn], mercury [Hg], nickel [Ni], and zinc [Zn]), metalloids (such as arsenic [As]), pesticides (such as insecticides, herbicides, fungicides, and molluscicides), endocrine disruptors (such as bisphenol A, phthalates, and tributyltin), persistent organic pollutants (such as polyaromatic hydrocarbons and halogenated compounds), Maillard reaction products (such as acrylamide and furans), human/veterinary pharmaceuticals, personal care products, nano-pollutants, and food/feed additives enter food/feed webs and threaten not only human but also animal health [1, 2]. These potentially toxic pollutants are mainly carcinogens, teratogens, mutagens, and immune, neural, cardiovascular, hepatic, renal, reproductive, and endocrine disruptors [3, 4]. Heavy metals are a broad class of pollutants and toxicologically distinguished from others by intensive uses, non-biodegradability, accumulation, and magnification in the food chain. Some of the most affected/accumulated food chain members are aquatic animals and health risk assessment based on metal levels in the tissues of various fish species is a hot research topic [5,6,7]. Owners prefer the canned/pouched or homemade diets with fish and liver the most because they are more appetizing to cats. Yet, as demonstrated in previous studies, heavy metals accumulate in higher levels in aquatic organisms [5, 8]. Also, heavy metals accumulate in higher levels in target organs like the liver (metabolism), kidneys (extraction), and bones. As demonstrated in previous studies, some metals accumulate in highest levels in the liver because it is the main organ for the metabolism of substances [9,10,11]. Therefore, diets containing seafood and liver could pose more health risks than others.
Like all living animals, cats require a balanced diet to grow normally and maintain health once they are mature [12]. Commercial feeds formulated in line with the nutritional needs of cats are an effective factor in prolonging the lifespan by supporting health [13]. Energy is necessary for the body’s metabolic work, which means cats’ daily food intake must provide adequate metabolic energy [12]. Therefore, the daily feed requirement can alter according to the lifestyle (indoor, outdoor, etc.), health status (healthy, sick, obese, or cachectic), or physiological state (sex, pregnant, lactating, geriatric, pediatric, kitten, adult, etc.) of the cat. An ideal diet for a cat is programmed by veterinarians according to its lifestyle, species, breed, and physiological needs. Consequently, the daily consumption rate and quantity should be calculated considering the nutritional and energy requirements of the cat and the nutritional content of the food [14]. On the contrary, pet owners prefer market foods and feeding based on their own free choice primarily and the manufacturer’s recommendation secondarily [15]. Cat feeds are classified as dry, wet (canned/pouched), and semi-moist; commercially natural and organic foods; raw feed diets; vegetarian products; and homemade or veterinary (therapeutic) diets according to specific needs [12]. However, animal health specialists suggest consumption of canned feed according to individual nutrient, calorie, or metabolic energy requirements [14, 16], and wet foods form half of the diets or feeding preferences for a large percentage of cats in some countries [13, 17]. The commercially canned adult and kitten cat feed weights range between 70 and 415 g, and the range for pouches is between 80 and 100 g. The average daily consumption recommendations are 300–400 g for a normal active 4–5 kg adult cat and 200–300 g for a normal active 4–10-month-old kitten in two or three portions according to the labels.
Even if health risk assessment is not suitable in farm animals (such as cattle, sheep, goats, pigs, fishes, rabbits, poultry, turkey, geese, and ducks) because of their short lifespan, a health risk assessment would need to be considered (a hazard characterization especially including genotoxic effects, developmental neurotoxicology, and behavioral alterations) in companion animals (such as dogs, cats, or horses) which can reach the geriatric stage of life [1].
Health risk assessment is a scientific tool used for the characterization of health risk potential on the target population in various exposure scenarios [18]. Several methods are used in human health risk assessment [5, 19]. Among these, target hazard quotient (THQ) is one of the most commonly used methods to evaluate chronic non-carcinogenic effects on consumers and the most suitable for adaptation in animal health risk assessment.
There are some parameters that should be considered in the adaptation of human health risk assessment to animal health. The average body weight, which is an important parameter for assessment, varies widely in animals compared to humans, relative to breed and gender (male: 2.5–12 kg, female: 2–8 kg). Like body weight, the average life span of cats, 14 years (12–20 years), can vary considerably among breeds [14, 15, 20, 21]. Therefore, calculations should be based on the average body weight or life span by breed and gender. On the other hand, there are some common parameters that facilitate the adaptation of assessment. The oral reference doses (RfD) between these are one of the most important parameters of the risk assessment and calculated according to toxicity studies that have been conducted on laboratory animals for extrapolation to human toxicity [18]. Therefore, considering the variables and parameters used in the method, the risk assessment can be adapted to animal health.
The objectives of this study are (i) to determine heavy metal (Al, Cd, Cr, Cu, Fe, Hg, Mn, Ni, Pb, and Zn) contamination levels most associated with environmental pollution in canned and pouched cat foods, (ii) to quantify the potential health risks to cats (male/female and adult/kitten) by recommended average consumption rate of products, and (iii) to adapt the health risk assessment method to animal health science.
Materials and Methods
Sample Collection
A total of 50 commercial (15 different trademarks) canned and pouched cat foods were collected from local markets in Istanbul during 2019 and 2020. The foods were labeled as with salmon (n = 10), tuna (n = 10), and liver (n = 10) for adults, with aquatic products (fish, shrimp, etc.) (n = 20) for kittens.
Chemical Analyses
Microwave acid digestion (Cem, Matthews, NC, USA) oven was used to remove the organic parts in the samples. Firstly, all of the canned/pouched cat foods were homogenized with Ultra Turrax homogenizer (IKA T25, Germany), and 0.5 g (± 0.1 mg) was weighed into Teflon vessels. Samples were digested by 8 mL of nitric acid (HNO3; 65%, Merck, Germany) and 2 mL of hydrogen peroxide (H2O2; 30%, Merck, Germany) in a microwave system (CEM MARS X press, closed Teflon vessel microwave oven) using the 4-step program in accordance with the procedure for 15 min at 85% power and 200 °C. After completing digestion process, the residues were cooled and filtered through a 0.45-µm filter paper (Whatman, USA). The filtrates were transferred to a volumetric flasks and filled up to 25 mL with deionized water. Finally, the digested solutions were kept at + 4 °C until analysis. The Al, Cd, Cr, Cu, Fe, Hg, Mn, Ni, Pb, and Zn contamination levels in canned and pouched cat foods were analyzed by inductively coupled plasma optical emission spectrophotometer (ICP-OES-Perkin-Elmer-Norwalk, USA). The following wavelength lines of the ICP-OES analysis were used: Al 396.2 nm, Cd 228.8 nm, Cr 267.7 nm, Cu 324.8 nm, Fe 259.9 nm, Hg 184.95 nm, Mn 257.6 nm, Ni 231.6 nm, Pb 220.4 nm, and Zn 206.2 nm. Prior to metal analysis, standard solutions were prepared from stock solution (ICP multi-element standard solution; Merck, Germany) and used to calibrate the ICP-OES device. The method quality was validated by using certified reference material (DORM-4; National Research Council, Canada), correlation coefficients (r2) (> 0.99), relative standard deviation (RSD) (< 10), recovery (94.2–103.8%), limit of detection (LOD), and quantification (LOQ) (mg L−1; Cd, Fe, Hg, Mn, and Zn: 0.0003/0.001; Cr and Cu: 0.0006/0.002; Ni: 0.0008/0.003; Pb: 0.002/0.007; Al: 0.003/0.010). LOD was calculated as 3 SD/m and LOQ was calculated as 10 SD/m (SD: standard deviation, m: the slope of calibration curves). Each sample was analyzed in triplicate and the metal concentrations were expressed as mg kg−1 wet weight (ww).
Animal Health Risk Assessment Adaptation
The potential health risk was assessed regarding metal contamination levels according to recommended consumption rates of commercial canned and pouched (salmon, tuna, liver, and aquatic products) cat foods. The THQ method [19] was adapted to assess the health risks of heavy metal contamination in samples according to the formula given below:
where EF is exposure frequency (365 days year−1), ED is exposure corresponding to average lifespan (14 years for adult and 1 year for kitten) [12, 21], FIR is the consumption rate (recommended average consumption rate of products is 400 g day−1 for adults and 300 g day−1 for kittens), C is the metal concentration in feed (mg kg−1 ww), RfD is the oral reference doses (mg kg−1/day) as given in Table 1 [22,23,24], WAB is the average body weight (bw) (5.0 kg for male and 4.2 for female adults and 2.4 for male and 1.4 for female 24–26 week old kittens) [15, 25], and TA is the average non-carcinogenic exposure duration (EFxED) [5].
The values less than 1 of THQ indicate that the consumption of the selected canned/pouched feeds in terms of heavy metals does not pose a risk to the specified individuals according to the consumption scenario.
The total THQs (TTHQs) were calculated by summing up the THQ values of each metal for each wet cat food as formulated below:
Statistical Analysis
After normality and homogeneity tests, the one-way analysis of variance (ANOVA) with post hoc Tukey’s HSD was performed to evaluate results statistically. The differences at level p < 0.05 were considered significant.
Results
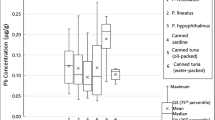
The sampled international trademarked cat food product countries were Austria, Czech Republic, England, France, Germany, Italy, Malaysia, the USA, and Turkey. The determined mean metal concentrations of samples are shown in Table 2. The highest mean Cd and Pb concentrations were determined in canned/pouched salmon; the highest mean Cu, Fe, Hg, and Ni concentrations were determined in canned/pouched tuna; the highest mean Al, Mn, and Zn concentrations were determined in canned/pouched liver for adults; and the highest mean Cr and Hg concentrations were determined in canned/pouched aquatic products for kittens.
The differences in Al levels between liver and others, in Cd levels between salmon and others, and in Mn levels between salmon and liver were statistically significant (p < 0.05).
Authorities have not set a maximum limit for essential metals (Cu, Fe, Mn, Se, and Zn), but some sectoral federations recommended maximum (Cu: 28, Fe: 1420, Mn: 170, Se: 568, and Zn: 284) and minimum (for kitten, Cu: 10, Fe: 80, Mn: 10, Se: 300, Zn: 75, and for adults, Cu: 5, Fe: 80, Mn: 5, Se: 300, and Zn: 75) limits for essential metals in cat foods [26]. Authorities have set maximum limits (mg kg−1) for some nonessential toxic metals such as As, Cd, Hg, and Pb. The limits in the EC were Cd: 2 for “feed materials of animal origin,” Hg: 0.5 for “feedingstuffs produced by the processing of fish or other marine animals,” 0.4 for “complete feedingstuffs for dogs and cats,” 0.2 for “complementary feedingstuffs for dogs and cats,” and Pb: 5 for “complete feedingstuffs” [27]. The US Food and Drug Administration set maximum limits as Cd: 10, Cr: 10, Hg: 0.267, Ni: 50, and Pb: 10, while the Association of American Feed Control Officials has only set a maximum limit for Se in dog and cat food [28, 29].
Although none of the contamination levels in the study exceeded the legal limits of metals and commercial animal feed/foods labeled these metals between recommended essential and toxic levels as contents, it is not possible to reveal toxic potential without a species-specific risk assessment of a dietary toxicants intake and eating habits.
The target hazard quotient values calculated according to mean metal concentrations, shown in Table 3, were all below 1 for both male/female adult cats and kittens. The highest TTHQs for female and male adults were 2.9E-01 and 3.5E-01, respectively, by a tuna consumption scenario, and for female and male kittens were 7.2E-01 and 4.2E-01 by an aquatic products consumption scenario in producer-recommended consumption rates. The TTHQ values were ordered as female kitten > male kitten > female adults > male adults from highest to lowest. Total target hazard quotient values did not exceed 1 in recommended average consumption rates of products, which means that the studied metals do not pose a health risk for adult cats and kittens. However, consumption of canned/pouched fish over 418 g per day may pose a risk for 24–26 week old female kittens with a body weight of 1.4 kg or less.
There are critical points in the THQ calculation method for non-adult animals that may lead to incorrect results. It should be noted that the ED, bw, and FIR values can alter for kitten and in different age-staged animals.
Discussion
Although potential health risk assessment of contaminants in foods through consumption rates is frequently conducted for human, they have yet to be adapted to animal health. It is also important that these foods do not pose a health risk for animals, especially for those feeds in the same food group used daily and for long periods, as with cats [30]. Contaminants accumulate in fish tissues based on the trophic level, life span, and feeding habits as well as the chemical characteristics of the contaminant. This accumulation biomagnifies up through the food chain and reflects a risk for the consumer’s (both human and animal) health [2, 5, 31]. Similar to human health risk assessments, in the study, the scenario of if the wet foods (randomly purchased and feed with the recommended consumption amounts in their labels) pose a health risk to adult and kitten cats, in terms of food safety-related toxic heavy metals was created. The reason for the selection of foods with fish and liver is that they are the most preferred ones by cats. Metals are potentially toxic above tolerable limits whether they are essential (Cu, Fe, Mn, Zn, etc.) or not (Cd, Hg, Pb, etc.) for the biological structure and functions of organisms [5, 32,33,34,35]. Although the determination of metal levels in biological samples (tissue, blood, urine, feces, and hair) of animal species and feed or foods can play a key role in diagnosis, there are no reference values (for biological samples) or maximum tolerable limits (MTL) for cats. For example, it has been reported that the concentrations of > 6 mg kg−1 in blood and 4 mg kg−1 in the feed are points to the diagnosis of Hg poisoning in most animal species [34].
Comparison of the selected metal concentrations with other study results analyzed by ICP-OES and set or recommended limits by international authorities or federation is shown in Table 4 [26, 27, 29, 44,45,46,47,48,49,50,51,52,53,54]. The contamination levels of the metals included in the study were well below the legal or recommended limits. Among these metals, the closest level to the limit was determined as Pb. There is no legal limit or recommended maximum level for Al. Average Al levels were in the ranges determined by other studies on seafood (Table 4).
Al is neurotoxic and leads to behavioral changes such as memory loss, anemia, bone abnormalities, osteoarthritis, and myocardial infarction [3]. The average Al concentration of tuna measured in the study is higher than measured by Ababneh and Al-Momani [51]. The average Al concentrations of all aquatic products in the study are lower than measured in fishes by Ahmad Al-Subeihi [44], and within the ranges measured in fishes by Sunjog et al. and Subotić et al. [48, 50]. There is no legal or recommended maximum contamination level for Al in the feed or food of animals. Although no MTL value has been specified for cats, it has been reported as 200–1000 mg kg−1 for other species [35]. In this respect, according to the THQ result of the study, Al contamination does not pose a risk in any product.
Cd is nephrotoxic, mutagen, carcinogen, teratogen, endocrine disruptor and regulators have set legal maximum contamination limits for pet foods [3, 4, 27, 29, 37]. The average Cd concentration of Salmon measured in the study is higher than measured in fishes by Nikolić et al., Subotić et al., Ababneh and Al-Momani, and Sobhanardakani [46, 50, 51, 53], and measured in the liver by Naseri et al. [54]. The average Cd concentrations of all aquatic products measured in the study are within the ranges measured in fishes by Milenkovic et al., Rašković et al., and Ural et al. [45, 47, 52]. The average Cd concentrations of all aquatic products measured in the study are lower than both the legal (2.5-fold lower) and recommended (12-fold lower) maximum level. Although no MTL value has been specified for cats, it has been reported as 10 mg kg−1 for other species [35]. Also according to the THQ result of the study, Cd contamination does not pose a risk in any product.
Hypoactivity, mydriasis, lacrimation, and diarrhea are the signs of acute Cr toxicosis and regulators have set a legal maximum contamination limit for pet foods [29, 35]. The average Cr concentrations of all aquatic products measured in the study are higher than measured in fishes by Nikolić et al. and Heshmati et al. [46, 49], lower than measured in fishes by Rašković et al. [47], and within the ranges measured in fishes by Sunjog et al. [48]. The average Cr concentration of liver measured in the study is lower than measured by Naseri et al. [54]. The average Cr concentrations of all products measured in the study are 50-fold lower than the legal maximum level. Although no MTL value has been specified for cats, it has been reported as 100–500 mg kg−1 for other species [35]. Also according to the THQ result of the study, Cr contamination does not pose a risk in any product.
The excessive dietary intake of Cu can cause liver damage primarily in animals and a maximum contamination limit has been recommended for pet foods [26, 35, 36]. The average Cu concentrations of all aquatic products measured in the study are higher than measured in fishes by Ahmad Al-Subeihi and Heshmati et al. [44, 49], and within the ranges measured in fishes by Rašković et al., Sunjog et al., and Subotić et al. [47, 48, 50]. The average Cu concentration of liver measured in the study is lower than measured by Naseri et al. [54]. The average Cu concentrations of all products measured in the study are more than tenfold lower than the recommended maximum level. Although no MTL value has been specified for cats, it has been reported as 15–500 mg kg−1 for other species [35]. Also according to the THQ result of the study, Cu contamination does not pose a risk in any product.
Fe damage the cardiovascular and digestive systems, liver, and brain, and a maximum contamination limit has been recommended for pet foods [26, 35, 36]. The average Fe concentrations of all aquatic products measured in the study are lower than measured in fishes by Ababneh and Al-Momani [51], and within the ranges measured in fishes by Rašković et al., Sunjog et al., Subotić et al., and Ural et al. [47, 48, 50, 52]. The average Fe concentrations of all aquatic products measured in the study are more than tenfold lower than the recommended maximum level. Although no MTL value has been specified for cats, it has been reported as 200–1000 mg kg−1 for other species [35]. Also according to the THQ result of the study, Fe contamination does not pose a risk in any product.
Mercury causes neurotoxic effects like ataxia, imbalance, and seizures and can lead to death, and regulators have set legal maximum contamination limits for pet foods [3, 27, 29, 30, 38]. The average Hg concentrations of tuna and other aquatic products measured in the study are higher than measured in fishes by Nikolić et al., Heshmati et al., Ababneh and Al-Momani, and Ural et al. [46, 49, 51, 52], lower than measured in fishes by Ahmad Al-Subeihi and Subotić et al. [44, 50], and within the ranges measured in fishes by Milenkovic et al. and Sunjog et al. [45, 48]. The average Hg concentrations of all aquatic products measured in the study are fivefold lower than legal maximum levels for animal food and feeds. Although no MTL value has been specified for cats, it has been reported as 500–3000 mg kg−1 for other species [35]. Also according to the THQ result of the study, Hg contamination does not pose a risk in any product.
Decrease in Fe level and hematological changes are the signs of chronic Mn toxicosis and a maximum contamination limit has been recommended for pet foods [26, 35]. The average Mn concentrations of fishes measured in the study are lower than measured in fishes by Ababneh and Al-Momani and Ural et al. [51, 52], and within the ranges measured in fishes by Rašković et al., Sunjog et al., and Subotić et al. [47, 48, 50]. The average Mn concentrations of all aquatic products measured in the study are 56-fold lower than recommended maximum levels for animal food and feeds. Although no MTL value has been specified for cats, it has been reported as 200–1000 mg kg−1 for other species [35]. Also according to the THQ result of the study, Mn contamination does not pose a risk in any product.
Decrease in feed intake, growth, and reproductive performance, kidney damage, and hematological changes are the common signs of chronic Mn toxicosis [35]. Regulators have set a legal maximum contamination limit for pet foods [29]. The average Ni concentrations of fishes measured in the study are lower than measured in fishes by Heshmati et al. and Ababneh and Al-Momani [49, 51], and within the ranges measured in all products by Rašković et al. and Naseri et al. [47, 54]. The average Ni concentrations of all products measured in the study are 500-fold lower than legal maximum levels for animal food and feeds. Although no MTL value has been specified for cats, it has been reported as 400–2000 mg kg−1 for other species [35]. Also according to the THQ result of the study, Ni contamination does not pose a risk in any product.
Clinical symptoms of Pb toxicosis in cats include neurologic (behavior changes and intermittent seizures) and gastrointestinal (abdominal discomfort, anorexia, vomiting, diarrhea, lethargy, and weight loss) signs [36]. Regulators have set legal maximum contamination limits for pet foods [27, 29]. The average Pb concentration of fishes and liver measured in the study is higher than measured in fishes by Nikolić et al., Sunjog et al., Ababneh and Al-Momani, and Sobhanardakani [46, 48, 51, 53], and within the ranges measured in aquatic products by Rašković et al. and Milenkovic et al. [45, 47]. The average Pb concentrations of all products measured in the study are 1.6–eightfold lower than legal maximum levels for animal food and feeds. Although no MTL value has been specified for cats, it has been reported as 10–100 mg kg−1 for other species [35]. Also according to the THQ result of the study, Pb contamination does not pose a risk in any product.
Zn toxicity limits the absorption of Fe, Cu, and calcium and causes clinical symptoms in cats and a maximum contamination limit has been recommended for pet foods [26, 35, 36]. The average Zn concentrations of all aquatic products measured in the study are higher than measured by Ahmad Al-Subeihi, Nikolić et al., Sunjog et al., Heshmati et al., and Sobhanardakani [44, 46, 48, 49, 53], lower than measured by Ural et al. [52], and within the ranges measured in all products by Rašković et al., Sunjog et al., and Subotić et al. [47, 48, 50]. The average Zn concentrations of all products measured in the study are 13.5-fold lower than recommended maximum levels for animal food and feeds. Although no MTL value has been specified for cats, it has been reported as 250–1000 mg kg−1 for other species [35]. Also according to the THQ result of the study, Zn contamination does not pose a risk in any product.
Compared to the mean metal concentrations (analyzed with atomic absorption spectrometer) from a study of canned fish cat foods in Turkey, Cd, Cr, Cu, Mn, and Pb in the present study were low, while the Fe and Ni means were high [39]. Since that study did not specify if the sampled food was for adult cats or kittens and since no risk assessment was evaluated, it is thought that the results suggested cat foods pose a higher health risk than that indicated in our study, especially for Cu. Compared to another study conducted in Egypt, the levels of Cd and Pb (in tuna for adults and in aquatic products for kittens and analyzed with atomic absorption spectrometer) were high, while Cr and Pb levels (in salmon) were low in the present study [40]. In a study conducted in Brazil (analyzed with ICP-MS), estimated daily intakes (EDI) of cats were calculated over the metal levels of six canned adult cat foods which contained 83.5% fish and 16.5% beef and/or liver [41]. According to the results, the studied metal levels (Cr, Cu, Fe, Mn, and Zn) were 7–30 times higher than those determined in the present study. The concern about the presence of Hg and Cd, which exceeded the MTLs in canned foods, was especially emphasized in the study [41]. In a study conducted in chicken liver, while Cd, Cu, and Pb levels were higher than our results, the Zn level was found to be lower than ours [42]. Compared to a study conducted in Pakistan, in chicken, lamb, and beef livers, Cd was not detected as in our study, Ni level was higher while Cu, Fe, Pb, and Zn levels were low than ours [43]. According to the THQ and TTHQ results of the study, contamination of each metal does not pose a risk in any product alone or together.
Conclusions
This study illuminated that THQ and TTHQ values of selected metals did not exceed 1 and may mean that consumption of canned or pouched food at the recommended rate does not pose a health risk for adult cats and kittens. Even though the levels of the selected metals are below the legal or recommended limits, in the ranges indicated in other studies, possible intake of other dietary contaminants (unevaluated metals and other pollutants) and consumption over the considered rate in the study may increase the risk. Whereas heavy metal concentration levels in human diets and health risk assessment are a popular and important field of study, it is an omitted subject in animal health science. Species-specific animal risk assessment adaptation studies should be conducted based on dietary habit surveys with high participation rates and should include consideration of other contaminants. The consumption rates of canned foods, especially in breeds predisposed to neurological, liver, and kidney diseases and animals in risk groups (with chronic disease, pediatric, geriatric, etc.) must be well evaluated. Preventing metal contamination in the production stages of canned foods and keeping levels below the legal limits are important for animal food safety. However, it will be a more accurate approach to create consumption suggestions as a result of the risk assessment to be made over the consumption scenarios and potential consumer groups. Extensive studies are needed to identify potential risks and clinical impacts associated with heavy metal exposure due to the canned/pouched food consumption of animals of different breeds, life stages, health status, feeding habits, and physical and physiological conditions.
Data Availability
Not applicable.
Code Availability
Not applicable.
References
Dorne JLCM, Fink-Gremmels J (2013) Human and animal health risk assessments of chemicals in the food chain: comparative aspects and future perspectives. Toxicol Appl Pharmacol 270(3):187–195. https://doi.org/10.1016/j.taap.2012.03.013
Yarsan E, Yipel M (2013) The important terms of marine pollution “biomarkers and biomonitoring, bioaccumulation, bioconcentration, biomagnification.” J Mol Biomark Diagn S1:1–2. https://doi.org/10.4172/2155-9929.S1-003
Gupta RC (2012) Veterinary toxicology: Basic and clinical principles, 2nd edn. Academic press, USA
Bampidis VA, Nistor E, Nitas D (2013) Arsenic, cadmium, lead and mercury as undesirable substances in animal feeds. J Anim Sci Biotechnol 46(1):17–22
Yipel M, Yarsan E (2014) A risk assessment of heavy metal concentrations in fish and an invertebrate from the Gulf of Antalya. B Envıron Contam Tox 93(5):542–548. https://doi.org/10.1007/s00128-014-1376-5
Jia Y, Kong Q, Yang Z, Wang L (2016) Accumulation behavior and risk assessment of heavy metals and arsenic in tissues of white bream (Parabramis pekinensis) from the Xiang River, southern China. Environ Sci Pollut 23(24):25056–25064. https://doi.org/10.1007/s11356-016-7734-6
Jia Y, Wang L, Qu Z, Wang C, Yang Z (2017) Effects on heavy metal accumulation in freshwater fishes: species, tissues, and sizes. Environ Sci Pollut 24(10):9379–9386. https://doi.org/10.1007/s11356-017-8606-4
Bosch AC, O’Neill B, Sigge GO, Kerwath SE, Hoffman LC (2016) Heavy metals in marine fish meat and consumer health: a review. J Sci Food Agric 96(1):32–48. https://doi.org/10.1002/jsfa.7360
Abou-Arab AAK (2001) Heavy metal contents in Egyptian meat and the role of detergent washing on their levels. Food Chem Toxicol 39(6):593–599. https://doi.org/10.1016/s0278-6915(00)00176-9
Sedki A, Lekouch N, Gamon S, Pineau A (2003) Toxic and essential trace metals in muscle, liver and kidney of bovines from a polluted area of Morocco. Sci Total Environ 317(1–3):201–205. https://doi.org/10.1016/S0048-9697(03)00050-0
Caggiano R, Sabia S, D’Emilio M, Macchiato M, Anastasio A, Ragosta M, Paino S (2005) Metal levels in fodder milk dairy products and tissues sampled in ovine farms of Southern Italy. Environ Res 99(1):48–57. https://doi.org/10.1016/j.envres.2004.11.002
Case LP, Daristotle L, Hayek MG, Raasch MF (2010) Canine and feline nutrition-E-book: a resource for companion animal professionals. Elsevier Health Sciences.
Laflamme DP, Abood SK, Fascetti AJ, Fleeman LM, Freeman LM, Michel KE, Willoughby KN (2008) Pet feeding practices of dog and cat owners in the United States and Australia. J Am Vet Med Assoc 232(5):687–694. https://doi.org/10.2460/javma.232.5.687
Hand MS, Thatcher CD, Remillard RL, Roudebush P (2000) Small animal clinical nutrition, 4th edn. Mark Morris Institute, USA
Colliard L, Paragon BM, Lemuet B, Bénet JJ, Blanchard G (2009) Prevalence and risk factors of obesity in an urban population of healthy cats. J Feline Med Surg 11:135–140. https://doi.org/10.1016/j.jfms.2008.07.002
NRC (2006) National Research Council. Nutrient requirements of dogs and cats. National Academies Press, Washington, USA
Davies M, Alborough R, Jones L, Davis C, Williams C, Gardner DS (2017) Mineral analysis of complete dog and cat foods in the UK and compliance with European guidelines. Sci Rep 7(1):17107. https://doi.org/10.1038/s41598-017-17159-7
Asante-Duah DK (2002) Public health risk assessment for human exposure to chemicals, vol 6. Kluwer Academic, London
US EPA (2000) US environmental protection agency. Guidance for Assessing Chemical Contaminant Data for Use in Fish Advisory, vol. II: Risk Assessment and Fish Consumption Limits. https://www.epa.gov/sites/production/files/2015-06/documents/volume2.pdf Accessed 1 October 2020
Chase MH, Morales FR, Boxer PA, Fung SJ (1985) Aging of motoneurons and synaptic processes in the cat. Exp Neurol 90(2):471–478. https://doi.org/10.1016/0014-4886(85)90035-4
Taylor EJ, Adams C, Neville R (1995) Some nutritional aspects of ageing in dogs and cats. Proc Nutr Soc 54(3):645–656. https://doi.org/10.1079/PNS19950064
MDEQ (2019) The Michigan Department of Environmental Quality. https://www.michigan.gov/documents. Accessed 1 October 2020
US EPA (2005) US environmental protection agency. IRIS Guidance Documents. http:// www.epa.gov/IRIS. Accessed 1 October 2020
US ORNL (2019) US Department of Energy science and energy laboratory. Risk Assessment Information System. https://rais.ornl.gov/tools/tox_profiles.html. Accessed 1 Oct 2020
Yu S, Howard KA, Wedekind K J, Morris J G, Rogers Q R (2002) A low‐selenium diet increases thyroxine and decreases 3. 5. 3′ triiodothyronine in the plasma of kittens. J Anim Physiol Anim Nutr 86(1‐2):36–41. https://doi.org/10.1046/j.1439-0396.2002.00338.x
FEDIAF (European Pet Food Industry) (2013) Nutritional guidelines for complete and complementary pet food for cats and dogs. http://www.fediaf.org/self-regulation/nutrition.html Accessed 1 October 2020
EC (2002) Commission directive 2002/32/EC of the European Parliment and of the council of 7 May 2002 on undesirable substances in animal feed (2002) Official Journal of the European Communities Brussels, Belgium. https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX%3A02002L0032-20131227 Accessed 1 October 2020
AAFCO (2014) Association of American Feed Control Officials Publications. Dog and Cat Food Nutrient Profiles. https://www.aafco.org/Regulatory/Committees/Pet-Food. Accessed 1 October 2020
FDA (2011). Target animal safety review memorandum: trace metal analysis of commercial pet food for toxic metals. https://www.fda.gov/media/81895/download
Dunham-Cheatham SM, Klingler K, Peacock M, Teglas MB, Gustin MS (2019) What is in commercial cat and dog food? The case for mercury and ingredient testing. Science of The Total Environ 684:276–280. https://doi.org/10.1016/j.scitotenv.2019.05.337
Adel M, Conti GO, Dadar M, Mahjoub M, Copat C, Ferrante M (2016) Heavy metal concentrations in edible muscle of whitecheek shark, Carcharhinus dussumieri (elasmobranchii, chondrichthyes) from the Persian Gulf: a food safety issue. Food Chem Toxicol 97:135–140. https://doi.org/10.1016/j.fct.2016.09.002
Pirsaheb M, Fattahi N, Sharafi K, Khamotian R, Atafar Z (2016) Essential and toxic heavy metals in cereals and agricultural products marketed in Kermanshah, Iran, and human health risk assessment. Food Addit Contam: Part B 9(1):15–20
Okpala COR, Sardo G, Vitale S, Bono G, Arukwe A (2018) Hazardous properties and toxicological update of mercury: from fish food to human health safety perspective. Crit Rev Food Sci Nutr 58(12):1986–2001. https://doi.org/10.1080/10408398.2017.1291491
Blakley B R (2013) Overview of mercury poisoning. merck manual veterinary manual. https://www.merckvetmanual.com/toxicology/mercury-poisoning/overview-of-mercury-poisoning
National Research Council (2005) Mineral tolerance of animals, 2nd edn. The National Academies Press, USA
Moeller RB, Albretsen JC, Coppock R, Dziwenka MM,Ensley S, Gwaltney-Brant S(2004) Metals and Minerals. In: Plumbee K (ed) Clinical veterinary toxicology. Mosby, Philadelphia, USA
López-Alonso M (2012) Animal feed contamination by toxic metals. In: Fink-Gremmels J (ed) Animal Feed Contamination. Woodhead Publishing, Philadelphia, 19102–3406, USA. https://doi.org/10.1533/9780857093615.2.183
Luippold A, Gustin MS (2016) Mercury concentrations inwet and dry cat and dog food. Anim Feed Sci Tech 222:190–193. https://doi.org/10.1016/j.anifeedsci.2016.10.021
Duran A, Tuzen M, Soylak M (2010) Trace element concentrations of some pet foods commercially available in Turkey. Food Chem Toxicol 48(10):2833–2837. https://doi.org/10.1016/j.fct.2010.07.014
Abd-Elhakim YM, El Sharkawy NI, Moustafa GG (2016) An investigation of selected chemical contaminants in commercial pet foods in Egypt. J Vet Diagn Invest 28(1):70–75. https://doi.org/10.1177/1040638715624733
Paulelli ACC, Martins AC, de Paula ES, Souza JMO, Carneiro MFH, Júnior FB, Batista BL (2018) Risk assessment of 22 chemical elements in dry and canned pet foods. J Consum Prot Food S 13(4):359–365. https://doi.org/10.1007/s00003-018-1178-5
Albashr TKM, Ahmed AS, Khidir ZK (2021) Determination of the level of trace elements (copper, lead, cadmium, zinc) and the level of cholesterol and triglycerides in some frozen meat imported in Tikrit markets. In IOP Conference Series. Earth Environ Sci 735(1):012004
Arif A, Khan B, Shahid N, Ahmed R (2021) Detection and validation studies of trace metals, protein and steroid in different organs of local and brand meat (poultry, cattle and fish). S Asian J Life Sci 9(1):1–9. https://doi.org/10.17582/journal.sajls/2021/9.1.1.9
Ahmad Al-Subeihi, AA (2021) Risk assessment of trace elements in selected imported frozen fish fillet in Jordanian market. Int J Environ Anal Chem 1-10https://doi.org/10.1080/03067319.2021.1897797
Milenkovic B, Stajic JM, Stojic N, Pucarevic M, Strbac S (2019) Evaluation of heavy metals and radionuclides in fish and seafood products. Chemosphere 229:324–331. https://doi.org/10.1016/j.chemosphere.2019.04.189
Nikolić D, Skorić S, Lenhardt M, Hegediš A, Krpo-Ćetković J (2020) Risk assessment of using fish from different types of reservoirs as human food–A study on European perch (Perca fluviatilis). Environ Pollut 257:113586. https://doi.org/10.1016/j.envpol.2019.113586
Rašković B, Poleksić V, Skorić S, Jovičić K, Spasić S, Hegediš A, Lenhardt M (2018) Effects of mine tailing and mixed contamination on metals, trace elements accumulation and histopathology of the chub (Squalius cephalus) tissues: Evidence from three differently contaminated sites in Serbia. Ecotoxicol Environ Saf 153:238–247. https://doi.org/10.1016/j.ecoenv.2018.01.058
Sunjog K, Kolarević S, Kračun-Kolarević M, Višnjić-Jeftić Ž, Skorić S, Gačić Z, Vuković-Gačić B (2016) Assessment of status of three water bodies in Serbia based on tissue metal and metalloid concentration (ICP-OES) and genotoxicity (comet assay). Environ Pollut 213:600–607. https://doi.org/10.1016/j.envpol.2016.03.008
Heshmati A, Sadati R, Ghavami M, Khaneghah AM (2019) The concentration of potentially toxic elements (PTEs) in muscle tissue of farmed Iranian rainbow trout (Oncorhynchus mykiss), feed, and water samples collected from the west of Iran: a risk assessment study. Environ Sci Pollut Res 26(33):34584–34593. https://doi.org/10.1007/s11356-019-06593-x
Subotić S, Spasić S, Višnjić-Jeftić Ž, Hegediš A, Krpo-Ćetković J, Mićković B, Lenhardt M (2013) Heavy metal and trace element bioaccumulation in target tissues of four edible fish species from the Danube River (Serbia). Ecotoxicol Environ Saf 98:196–202. https://doi.org/10.1016/j.ecoenv.2013.08.020
Ababneh FA, Al-Momani IF (2013) Levels of mercury, cadmium, lead and other selected elements in canned tuna fish commercialised in Jordan. Intern J Environ Anal Chem 93(7):755–766. https://doi.org/10.1080/03067319.2012.672981
Ural M, Arca S, Örnekçi GN, Demirol F, Yüce S, Uysal K, Koçer MAT (2012) Metal accumulation in sediment, water, and freshwater fish in a Dam Lake. Toxicol Environ Chem 94(1):49–55. https://doi.org/10.1080/02772248.2011.633912
Sobhanardakani S (2017) Tuna fish and common kilka: health risk assessment of metal pollution through consumption of canned fish in Iran. J Consum Prot Food Saf 12(2):157–163. https://doi.org/10.1007/s00003-017-1107-z
Naseri K, Salmani F, Zeinali M, Zeinali T (2021) Health risk assessment of Cd, Cr, Cu, Ni and Pb in the muscle, liver and gizzard of hen’s marketed in East of Iran. Toxicol Rep 8:53–59. https://doi.org/10.1016/j.toxrep.2020.12.012
Author information
Authors and Affiliations
Contributions
Not applicable.
Corresponding author
Ethics declarations
Ethics Approval
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Conflict of İnterest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Altınok-Yipel, F., Yipel, M. & Tekeli, İ.O. Health Risk Assessment of Essential and Toxic Metals in Canned/Pouched Food on Kitten and Adult Cats: an Animal Health Risk Assessment Adaptation Assay. Biol Trace Elem Res 200, 1937–1948 (2022). https://doi.org/10.1007/s12011-021-02792-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-021-02792-1