Abstract
In recent years, because of its significant biological roles, the usage of boron has been started in animal feeding. In this research, it was aimed to investigate the ulexite’s action mechanism on the zebrafish brain with an evaluation of the oxidative parameters. The adult zebrafish were exposed to four ulexite doses (5, 10, 20, and 40 mg/l) in a static test apparatus for 96 h. For assessing the oxidative responses, multiple biochemical analyses were performed in brain tissues. The results indicated the supporting potential of low ulexite doses on the antioxidant system (< 40 mg/l) and that low-dose ulexite does not lead to oxidative stress in the zebrafish brain. Again, our results showed that low ulexite concentrations did not cause DNA damage or apoptosis. As a final result, in aquatic environments, ulexite (a boron compound) can be used in a safe manner, but it would be useful at higher concentrations to consider the damages of the cells that are probable to develop because of the oxidative stress
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Boron element is found at the rate of 10 ppm in the earth’s crust and 3–5 ppm in sea water. Boron minerals are named according to the metal found in their structures, their water content, and crystal structures. Boron element is not found alone in nature. Generally, boron is found by forming minerals such as colemanite (2CaO • 3B2O3 • 5H2O) and ulexite (Na2O • 2CaO • 5B2O3 • 16H2O) containing some oxidized alkaline ions [1,2,3]. Ulexite (the chemical formula NaCaB5O9.8H2O) is an important boron mineral containing calcium and sodium [4] and is commonly used worldwide [5]. Ulexite ore, which is in sodium-calcium-hydroborate composition, is localized in rare places in the earth’s crust [6]. Uleksit is the most important boron mineral used in the production of boron compounds [7]. Boron minerals are effective not only in many enzymes’ activity, but also in hormone and lipid metabolism [8]. The action mechanism may not be purely understandable; however, it has been demonstrated that borates are not metabolized and they control the damage of the organism by changing the oxidative stress parameters [9,10,11,12,13].
The formation of reactive oxygen species (ROS) occurs during normal metabolism in a healthy organism; however, in some problematic situations (inflammation and drug, exogenous source, and radiation exposure), the production of this compound (ROS) is increasing. The main known ROS in the cells are superoxide anion, singlet oxygen, hydroxyl radical, and hydrogen peroxide; the levels of these in the cell are in constant balance under normal conditions [14]. The excess of free radicals can initiate chemical chain reactions that damage cells and even their DNA. Under normal conditions, there is a protected balance between oxidants and antioxidants in cells. However, an imbalance caused by excessive production of oxidants or a decrease in antioxidants leads to abnormal oxidant production, so “oxidative stress” occurs. In oxidative stress, in vivo–produced ROS cause oxidative damage of nucleic acids, proteins, and lipids. Among all purine and pyrimidine bases in DNA, guanine is more prone to oxidation. Therefore, the main target of ROS is DNA modification. The most common ROS in the organism is the lipid radical formed by the removal of a hydrogen from the allyl group of unsaturated fatty acids. This structure reacts with oxygen first and forms lipid peroxy radical and then lipid hydroperoxides by chain reaction with lipids. The iron (Fe+ 2) and copper (Cu+1) ions in the environment both accelerate the formation of lipid peroxide and turn this radical into cytotoxic products. Reactive oxygen species react with lipids, proteins, carbohydrates, and DNA for taking electrons. When these oxidants are in excess, they lead to peroxidation of lipids in the membrane, leading to impaired permeability, thereby causing intracellular ion imbalance. They also play an important role in the formation of many diseases by disrupting the structure of proteins and molecules such as DNA and RNA. In oxidation, an OH radical is added to the 8th position of the guanine molecule and 8-OHdG which is one of the free radical lesions of oxidized DNA. This shows the true toxic effects of unsaturated fatty acids in the ROS cell membrane, causing peroxidation. Lipid peroxide radicals, on the one hand, affect other unsaturated fatty acids in the cell membrane, leading to the formation of new lipid radicals, on the other hand, taking the hydrogen atoms released into lipid peroxides. So, the event continues by self-catalyzing. Thiobarbituric acid reagents such as malondialdehyde (MDA) are the most-known aldehyde form of lipid peroxides, resulting from the oxygenation of arachidonic acid or non-enzymatic oxidative degradation of polyunsaturated fatty acids. MDA changes the ionization, enzymatic activity, aggregation states of cell surface determinants, and intrinsic membrane properties by causing polymerization and cross-linking of components in the cell membrane. Defense systems that function in the body to prevent damage caused by ROS are called antioxidant defense systems. There are many different substances that can act as antioxidants. Antioxidants inhibit lipid peroxidation by stopping the peroxidation chain reaction or by collecting reactive oxygen species. The antioxidants have a protective effect against cancer and abnormal increase in cell division by reducing the effect of oxidative damage on DNA [15,16,17,18,19]. Due to their ability to take electrons from target molecules, oxidants are known to alter the structure and functions of this target molecule, causing cell damage by affecting the cell membrane, genetic material such as DNA, RNA, and various enzymatic events. These oxidants are monitored at the level of malondialdehyde (MDA) with enzymatic systems such as cytoplasmic, mitochondrial, and extracellular forms superoxide dismutase (SOD), glutathione peroxidase (GPx), and catalase (CAT) in living organisms [20]. Environmental factors that cause DNA damage are physical and chemical agents. Although there are about 23 different types of oxidative base damage on DNA, the most common base modification is 8-hydroxy-2-deoxyguanosine (8-OHdG) [21]. For this reason, the most commonly measured base damage is 8-OHdG and is considered as a marker of oxidative DNA damage [20]. In the formation of DNA damage, poly (ADP-ribose) polymerase enzyme (DNA Polymerase enzymes) is inhibited and DNA breaks occur [22]. In the case of DNA damage, the cell is eliminated by directing to apoptosis. The central component of the apoptotic program is caspases. Caspase activation is cell-specific, and caspase inhibitors (IAP) have been shown to inhibit effector caspases and inhibit apoptosis [23].
No study with ulexite content in aquatic livings was found in literature searches. For this reason, ulexite (a boron derivative) which has a protective effect in fish was used in this study, and its protective mechanism was investigated for the first time considering the use of the substance in question and the oxidative stress response.
Material and Methods
Fish Material
Adult zebrafish were obtained from Atatürk University Fisheries Faculty. The ulexite treatments were implemented in Atatürk University, Fisheries Faculty, Zebrafish Research and Toxicology Trial Unit. The fish were acclimated in the same conditions before the trial for 14 days. This study was conducted in agreement with the precepts of Atatürk University Animal Experimental and Ethical Committee number with 75296309-050.01.04-E.2000066104. The study was approved by the committee in Erzurum, Turkey.
Ulexite Source
Ulexite (Cas No. 1319-33-1; NaCa[B5O6(OH)6] 5H2O) was obtained from a commercial company (Eti Mine Works, Ankara, Turkey), and the test concentrations were determined according to the previous study by Rowe et al. [24].
Trial Plan and Application of Ulexite
According to the static test rules, the trial plan was composed in four replications (control 0 mg/l, and 5, 10, 20, 40 mg/l ulexite added to the water environment). For this purpose, the study was planned with 4 replications, 56 fish were used (randomly selected) in each group in aquarium environments, and the trial was conducted with a total of 1120 fish. The adult larvae were treated with determined applications for 96 h. The activities of enzymes (SOD, CAT, GPx, PON, and AR) and myeloperoxidase (MPO), MDA, 8-OHdG, and caspase-3 levels were analyzed in zebrafish brain tissues which were obtained from the living materials.
Determination of Enzyme Activities and Lipid Peroxidation (MDA) Level
Homogenate Preparation
All groups of fish were anesthetized in ice-cold water and subsequently sacrificed. All biochemical analyses were determined by combining the brain tissues of 8 fish (4 females and 4 males) for each group at the end of the trial period. To study oxidative stress parameters, firstly, tissues were homogenized. For this, the brain tissues taken from each application group were homogenized in phosphate buffer and centrifuged at 1300 rpm for 30 min [25, 26]. Bradford method (via bovine serum albumin as standard) was used for the determination of protein concentration (at 595 nm) [27].
Measurement of Antioxidant Enzyme Activity Enzyme Activity
Superoxide dismutase (SOD) accelerates the dismutation of toxic superoxide radicals (O2−) formed during oxidative energy production to hydrogen peroxide and molecular oxygen. This method is based on reading the optical density (OD) of the blue-colored formazan paint (at 560-nm wavelength) created by the superoxide radicals created using xanthine and xanthine oxidase (XOD) [28]. Catalase (CAT) activity was measured according to the Aebi [29] method. Catalase catalyzes the destruction of H2O2 to water and molecular oxygen. By using H2O2 to absorb light, the destruction rate of the enzyme at 240-nm wavelength was measured spectrophotometrically. Glutathione peroxidase (GPx) activity was measured in the presence of reaction with GR. The oxidized GSSG formed by the reduction of hydroperoxide with GPx is returned to its reduced state with GR and NADPH. Oxidation of NADPH to NADP+ results in a decrease in absorbance and was measured by a decrease in 340-nm absorbance [30]. Myeloperoxidase (MPO) activity was measured spectrophotometrically using o-dianicidine and hydrogen peroxide. In the presence of H2O2 as the oxidizing agent, MPO catalyzes the oxidation of o-dianicidine and gives a brown product to oxidized o-dianicidine. The absorbency of the samples was measured by spectrophotometry at a wavelength of 460 nm [31]. Paraoxonase (PON) and arylesterase (AR) activities were measured using commercially available kits. Paraoxane hydrolysis rate (diethyl p-nitrophenyl phosphate) was measured at 37 degrees with an increase in absorption at 412 nm [32].
Measurement of Lipid Peroxidation (MDA)
The principle of this analysis is based on the spectrophotometric (wavelength of 532 nm) measurement of the pigment, which is the result of the reaction of TBA (thiobarbituric acid) with the acidic pH and hot environment of the pink-colored pigment [33].
Determination of Apoptosis (Caspase-3) Level
Caspase-3 enzyme activity in the control and experimental group samples was determined using the “Caspase-3 Analysis Kit” (Fish (CASP3) ELISA Kit (Catalog No: 201-00-0031) (SunRed)). The main point of this analysis is the determination of the product formed by the reaction of the substrate with the caspase-3 enzyme. The readings were done in 10 min ELISA (plate reader) devices at 450-nm absorbance [34].
Determination DNA Damage (8-OHdG) Level
8-OHdG analysis in brain tissue homogenates was done with a ready-to-use commercial kit (Fish (8-OHdG) using the competitive binding of the 8-OHdG monoclonal antigen (anti-8-OHdG) to the sample, standard, or 8-OHdGs that have not been previously bound to the wells of the plate: 201-00-0041/SunRed). Samples taken from − 80 °C were dissolved and mixed with chitin solutions and standards. They had been incubated for 10 min in the plate shaker at 37 °C in the dark. Then, the Stop Solution (50 μl) was added to the plate wells and the measurement was performed in ELISA (plate reader) device with an absorbance of 450 nm within 10 min [34].
Statistical Analyses
Statistical analysis of the data obtained from the biochemical analysis was done using the SPSS (Standard Version Copyright © SPSS Inc. 20) prepackage program. The data of the study groups were shown as mean ± standard deviation (mean ± S d), and one-way ANOVA test was used for statistical comparison between groups. Significance was determined by Duncan’s test. Whether the differences were statistically significant and their significance levels were determined (p < 0.05).
Results
As a result of 96 h of application, the potential regulatory effect of ulexite, as well as the reference range for zebrafish, was investigated by multiple biochemical analyzes in brain tissue.
Enzyme Activities and Lipid Peroxidation Level
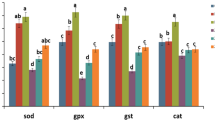
Ninety-six-hour ulexite application (5, 10, 20, and 40 mg/l) caused alterations in enzyme activities of zebrafish brain tissue (p < 0.05). The low-dose ulexite treatments (5, 10, and 20 mg/l) induced SOD, CAT, GPx, PON, and AR enzyme activities as compared with the control group. Forty milligrams per liter of ulexite led to the inhibition of these enzymes and an increase in MDA and MPO levels (Table 1).
DNA Damage Grade and Apoptosis Level
After 96 h, ulexite administration caused significant changes in 8-OHdG levels in zebrafish brain tissue (p < 0.05). The lower concentrations of ulexite (5, 10, and 20 mg/l) decreased the 8-OHdG levels comparing with the control group, whereas the 40-mg/l ulexite application caused an increase in the 8-OHdG level (Fig. 1). A situation similar to that of 8-OHdG was observed in caspase-3 activity, and the difference between the groups was found significant (p < 0.05) (Fig. 2).
Discussion
Enzyme Activities and Lipid Peroxidation Level
A general definition of oxidative stress is when the proxy-to-antioxidant balance shifts to the proxoid direction, causing potential cellular damage [8]. However, the measurement of oxidative stress status can be difficult due to the complex endogenous defense systems that perform correction and repair. Oxidative stress may be the result of increased free radical production and decreased antioxidant defense. Therefore, the investigation of antioxidant consumption as a biomarker of oxidative stress may be by evaluating the decrease in antioxidant amounts or the increase in their metabolites [20].
The supporting potential of low-dose ulexite on the antioxidant system was in the present research. In addition, comparing with the control group, low ulexite concentrations yielded lower results in brain MDA levels. Also, the determined enzyme activities for these groups (5, 10, and 20 mg/l) supported the other findings in our research that oxidative stress did not occur. As supporting the present research’s findings, Türkez et al. [35] reported that boron compounds (boric acid, borax, ulexite, and colemanite) assisted the antioxidant enzyme activities at doses which are not causing any genetic damage. In this line, the results of Geyikoğlu and Türkez [36] were parallel with our findings which obtained that boron compounds increase the activities of antioxidant enzymes at low concentrations (< 20 mg/l) and reduce at high concentrations (80–500 mg/l). Similarly, Nielsen [37] reported that boron increases SOD levels. In this study, SOD activity increasing (another prediction is about increasing antioxidant capacity) was effective in inhibiting boron free radical formation [38]. In addition, the GPx activity increasing in brain tissues is thought to be an adaptive answer to the effects of boron [33, 34, 39]. The antioxidant mechanism of boron and its derivatives has not been fully understood in conducted studies until now. However, boron supplementation (100 mg/kg/day) had been led to an increase in lipid peroxidation by increasing antioxidant activity [40]. It shows that the increase in CAT activity as a result of the use of boron compound plays a protective role against oxygen radicals. In this sense, it can be said that boron-derived ulexite has protective effects by both preventing LPO and increasing the antioxidant defense system activity.
Due to its antioxidant properties, PON and AR have been updated in recent years. PON and AR are enzymes in the esterase group that are encoded by the same gene and whose active centers are similar [41]. It is due to its success in preventing lipid hydroperoxide hydrolysis and oxidative damage in the atheroprotective effects of paraoxonase enzymes [42]. In this study, which constituted the first data on the ulexite application in aquaculture, it was concluded that ulexite also supports antioxidant enzyme activities by determining that these increases are statistically significant compared with control in enzyme activities. It is known that boron compounds have negative effects on GSH-Px at lower levels than those associated with other enzymes. In this sense, a dose-response relationship can be established between boron derivatives and GPx activity. Selenium should be taken into consideration especially in its effect on this enzyme [35, 43]. However, we cannot say that the effects of boron compounds on antioxidant enzymes are caused solely by selenium. Enzyme inhibitions seen in high concentrations of ulexite (40 mg/l) are a result of lipid peroxidation. Because lipid peroxidation increases fluidity by disrupting the integrity of cell membranes, this inactivates membrane-bound receptors and enzymes [44]. PON1 and AR enzymes are included in the antioxidant defense system because they prevent oxidation of lipid peroxides. It has been stated in previous studies that decreased specific activity may occur as a result of the inhibition of one of the oxidation products released into the circulation when glycolysis or oxidative stress increases, or as a result of decreased serum concentration [41]. As known, PON is inactive under oxidative stress and can be inhibited by oxidant [45].
In addition to the decrease in MDA level, ulexite can be interpreted to strengthen the antioxidant system, in the change of PON activation [46,47,48]. Similar mechanisms may be considered to be effective since this is consistent with AR enzyme activity results [34].
DNA Damage Grade and Apoptosis Level
DNA damage occurs spontaneously or under the influence of environmental factors during normal DNA metabolism. The most important cause of DNA damage during normal DNA metabolism is spontaneous changes in the chemical structure of the bases. The keto-enol tautomerism or deamination of the purine and pyrimidine bases alters the pairing specificity of the bases, leading to a mismatch. Also, depending on the thermal resistance of the bases, hydrolytic base loss occurs and as a result apurinic/apyrimidinic regions occur. Loss of base affects both replications, and chain fractures easily occur in these areas. Oxidative DNA damage can be created endogenously by oxygen radicals formed in some reaction steps during normal metabolism, as well as exogenous factors such as ionizing radiation and various chemicals. The oxygen required for life leads to the formation of reactive products that cause damage to cellular components in the organism [21].
High levels of MDA and MPO obtained at high doses of ulexite (40 mg/l) indicate the presence of oxidative stress. As a result of increased oxidative stress, 8-OHdG levels in this group of brain tissues were determined as high [10]. In parallel with the increase in 8-OHdG level, MDA level also increased. This is a verification feature. As a known case, nitrogen bases of DNA react with MDA, which can diffuse [49,50,51]. Boron/borax has a modulating effect on 8-OHdG levels as determined in low-dose ulexite applications [10].
When DNA damage occurs in the cell, it remains in the G1 phase and stops the transition to the S phase. It repairs the DNA damage that occurs in the cell by providing the synthesis of DNA repair enzymes; however, if this damage is irreparable, it triggers the synthesis of the base protein on the mitochondria membrane and enables the cell to undergo apoptosis [52].
The caspases activate other procaspases in a series of events. Caspases contribute to the production of cytokines (caspase-1, 4, 5, 13); they are classified as the initiators of proteolysis (caspase-3, 6, 7). The death signaling starter caspases connect to the adapter and lead to death, but they cannot perform the execution, only activate those who will do it. The practitioner caspases who carry out death activate the flow of the starter caspases [23]. Considering the increased caspase-3 activity, the important issue is to explain as to what extent caspase-3 activates ulexite application. The caspase-3 increase in brain tissues in high dose (40 mg/l) was also supported by a decrease in enzyme activity in the same group, high MDA levels, 8-OHgD level, and MPO activity. It can be said that this increase occurs in apoptotic cells due to the disruption of the body enzyme balance and the change of the apoptosis pathway.
Reactive oxygen metabolites occur as a natural result of oxygen use. ROS occurs in phagocytic activation; various synthesis and degradation reactions, especially mitochondrial electron transport; and oxidative stress, which develops as a result of shifting of the proxy/antioxidant balance in favor of proxydanes and damages biomolecules with various mechanisms. As a result of the increase in ROS formation and decrease in antioxidant enzyme levels, the cell’s defense line against oxidative stress is broken and dragged to death. Among the radical reactions characterized by biological damage, the most prominent is lipid peroxidation. Oxygen radical exposition to membranes of cell stimulates lipid peroxidation reactions. Lipid peroxidation ends with the conversion of lipid hydroperoxides to aldehyde MDA and other carbonyl compounds. Decreases in intracellular glutathione drag the cell to apoptosis via oxidative stress. In various animal model studies with low intracellular glutathione levels, it has been observed that they induce glucocorticoid-induced platelet apoptosis and increase anti-Fas/APO-1-induced lymphocyte apoptosis [53]. High levels of boron derivatives (ulexite, colemanite, borax, etc.) can induce apoptosis, as in this study. It does this by activating caspase-3 and suppressing some mechanisms [54]. Again, in the field of aquaculture, the anti-apoptotic effect of borax and/or its derivatives on toxicity models has been reported only in a few studies [10,11,12,13, 34]. Boron supplementation was found to reduce tissue antioxidant defense and reduce genotoxic effects [55]. In this study, ulexite, used at low doses, has no negative effect on some biochemical analysis results (DNA damage and apoptosis). We can evaluate this situation as the strengthening of the antioxidant defense for the studied tissue. Similarly, Khaliq [56] reported that at low doses, boron regulates cellular apoptosis, and it does this by overcoming oxidative stress.
Conclusion
This research showed clearly that ulexite, one of the commercially important boron compounds, promotes antioxidant capacity via increasing enzyme activities at low doses. However, this proposed mechanism should be supported by different biochemical investigations concerning the dose for each boron compound. Again, our results show that low concentrations of ulexite do not cause apoptosis or DNA damage in the brain. In summary, although high dose (40 mg/l) caused oxidative stress (with decreased enzyme activities and increased MDA and MPO levels), low-dose ulexite administration did not cause oxidative stress, DNA damage, and apoptosis. However, when we look at the effectiveness of the different concentrations tested, low-dose (5, 10, and 20 mg/l ulexite) applications were effective in preventing oxidative stress compared with high dose (40 mg/l).
The obtained data showed that it would be safe to use ulexite considering the cellular damages likely to develop due to oxidative stress. As a result, it may be important to study the applications and possible side effects in explaining the effectiveness of boron derivatives.
References
Gungoren SGOC (2012) Mikrodalga enerjisinin kolemanit ve üleksitin sudaki çözünürlüğüne etkisinin arastırılması. Istanbul J Earth Sci 22(1):85–93
Tekin G (2016) Kalsine uleksitin amonyum klorür çözeltileri içinde çözünürleştirilmesi kinetiği. J Balikesir Univ Inst Sci Technol 6(1):100–114
Kaykıoglu G (2016) Removal of methylene blue with colemanite and ulexite core waste: evaluation of kinetic and ısotherm. Celal Bayar Univ J Sci 12(3):499–509
Eskıbalcı MF, Isıktas OV (2017) Examination of the efficiencies of furnaces with different heating mechanisms in drying ulexite. Erzincan Univ J Sci Technol 10(1):11–22
Helvacı C (2003) Türkiye borat yatakları jeolojik konumu, ekonomik önemi ve bor politikası. J Balikesir Univ Inst Sci Technol 5(1):4–41
Gulensoy H, Kocakerim MM (1978) Uleksit mineralinin karbondioksitli sulardaki çözünürlüğü. MTA Derg (89):34-45
Demirkıran N, Kunkul A (2007) Dissolution kinetics of ulexite in perchloric acid solutions. Int J Miner Process 83:76–80
Comba B, Oto G, Mis L, Özdemir H, Comba A (2016) Effects of borax on inflammation, haematological parameters and total oxidant-antioxidant status in rats applied 3–methylcholanthrene. Kafkas Univ Vet Fak Derg 22(4):539–544
Pawa S, Shakir A (2006) Boron ameliorates fulminant hepatic failure by counteracting the changes associated with the oxidative stress. Chem Biol Interact 160:89–98
Alak G, Parlak V, Aslan ME, Ucar A, Atamanalp M, Türkez H (2018) Borax supplementation alleviates hematotoxicity and DNA damage in rainbow trout (Oncorhynchus mykiss) exposed to copper. Biol Trace Elem Res 187(2):536–542
Alak G, Parlak V, Yeltekin AÇ, Ucar A, Çomaklı S, Topal A, Atamanalp M, Özkaraca M, Türkez H (2019) The protective effect exerted by dietary borax on toxicity metabolism in rainbow trout (Oncorhynchus mykiss) tissues. Comp Biochem Physiol C Toxicol Pharmacol 216:82–92
Alak G, Yeltekin AÇ, Ucar A, Parlak V, Türkez H, Atamanalp M (2019) Borax alleviates copper-induced renal injury via inhibiting the DNA damage and apoptosis in rainbow trout. Biol Trace Elem Res 191(2):495–501
Alak G, Parlak V, Aslan ME, Ucar A, Atamanalp M, Turkez H (2019) Borax supplementation alleviates hematotoxicity and DNA damage in rainbow trout (Oncorhynchus mykiss) exposed to copper. Biol Trace Elem Res 187(2):536–542
Halliwell B, Gutteridge JMC (1998) Free radicals in biology and medicine, 2nd edn. Clarendon Press, Oxford, pp 188–196
Çiftçi N (2017) The role of oxidative stress in cancer: could antioxidants fuel the progression of cancer? Ahi Evran Tıp Dergisi 1(1):8–13
Borek C, Ong A, Mason H, Donahue L, Biaglow JE (1986) Selenium and vitamin E inhibit radiogenic and chemically induced transformation in vitro via different mechanisms. Proc Natl Acad Sci 83:1490–1494
Schuyer M, Berns EM (1999) Is TP53 dysfunction required for BRCA1-associated carcinogenesis? Mol Cell Endocrinol 155:143–152
Uysal M (1998) Serbest radikaller, lipit peroksitleri ve organizmada prooksidan-antioksidan dengeyi etkileyen koşullar. Klinik Gelişim 2:336–341
Sezer K, Keskin M (2014) Role of the free oxygen radicals on the pathogenesis of the diseases. FÜ Sağ Bil Vet Dergisi 28(1):49–56
Eken A (2017) Rat kan ve doku örneklerinde oksidatif stres parametreleri. J Clin Anal Med
Dincer Y, Kankaya S (2010) DNA hasarının belirlenmesinde comet assay. Turkiye Klinikleri J Med Sci 30(4):1365–1373
Baydas G, Reiter RJ, Akbulut M, Tuzcu M, Tamer S (2005) Melatonin inhibits neural apoptosis induced by homocysteine in hippocampus of rats via inhibition of cytochrome C translocation and caspase-3 activation and by regulating pro-and anti-apoptotic protein levels. Neuroscience 135(3):879–886
Tomatır AG (2003) Apoptosıs: programmed cell death. T Klin J Med Sci 23(6):499–508
Rowe RI, Bouzan C, Nabili S, Eckhert CD (1998) The response of trout and zebrafish embryos to low and high boron concentrations is U-shaped. Biol Trace Elem Res 66(1-3):261–270
Alak G, Yeltekin AÇ, Tas IH, Ucar A, Parlak V, Topal A, Kocaman EM, Atamanalp M (2017) Investigation of 8-OHdG, CYP1A, HSP70 and transcriptional analyses of antioxidant defense system in liver tissues of rainbow trout exposed to eprinomectin. Fish Shellfish Immunol 65:136–144
Alak G, Ucar A, Parlak V, Yeltekin AÇ, Tas IH, Ölmez D, Kocaman EM, Yılgın M, Atamanalp M, Yanık T (2017) Assessment of 8-hydroxy-2-deoxyguanosine activity, gene expression and antioxidant enzyme activity on rainbow trout (Oncorhynchus mykiss) tissue exposed to biopesticide. Comparative Biochemistry and Physiology Part C: Toxicology & Pharmacology 203:51–58
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein dye binding. Anal Biochem 72:248–254
Sun Y, Oberley LW, Li Y (1988) A simple method for clinical assay of superoxide dismutase. Clin Chem 34(3):497–500
Aebi H (1974) Catalase. In: Methods of enzymatic analysis. Academic Press, New York, Bergmeyer, HU, USA, p 673–678.
Beutler E (1984) Red cell metabolism: a manual of biochemical methods, Second edn. Grune and Starton, New York
Bradley PP, Priebat DA, Christensen RD, Rothstein G (1982) Measurement of cutaneous inflammation: estimation of neutrophilcontent with an enzyme marker. J Invest Dermatol 78:206–209
Gülcü F, Gürsü MF (2003) The standardization of paraoxonase and arylesterase activity measurements. Turk J Biochem 28(2):45–49
Alak G, Ucar A, Çilingir Yeltekin A, Parlak V, Nardemir G, Kızılkaya M, Taş IH, Yılgın M, Atamanalp M, Topal A, Kocaman ME, Yanık T (2019) Neurophysiological responses in the brain tissues of rainbow trout (Oncorhynchus mykiss) treated with bio-pesticide. Drug Chem Toxicol 42(2):203–209
Alak G, Parlak V, Uçar A, Yeltekin AÇ, Ozgeris FB, Cağlar O, Atamanalp M, Türkez H (2020) Oxidative and DNA damage potential of colemanite on zebrafish: brain, liver and blood. Turk J Fish Aquat Sci 20(8):593–602
Türkez H, Geyikoǧlu F, Tatar A, Keleş S, Özkan A (2007) Effects of some boron compounds on peripheral human blood. Z Naturforsch C 62(11-12):889–896
Geyikoğlu F, Türkez H (2008) Boron compounds reduce vanadium tetraoxide genotoxicity in human lymphocytes. Environ Toxicol Pharmacol 26:342–347
Nielsen FH (1994) Biochemical and physiologic consequences of boron deprivation in humans. Environ Health Perspect 102:59–63
Hunt DC (1998) Regulation of enzymatic activity, one possible role of dietary boron in higher animals and humans. Biol Trace Elem Res 66:205–225
Mohora M, Boghianu L, Muscurel C, Dute C, Dumitrache C (2002) Effect of boric acid redox status in the rat livers. Rom J Biophys 12(3–4):77–82
Sogut I, Oglakci A, Kartkaya K, Ol KK, Sogut MS, Kanbak G, Inal ME (2015) Effect of boric acid on oxidative stress in rats with fetal alcohol syndrome. Exp Ther Med 9(3):1023–1027
Caner C, Vural Özeç A, Aydın H, Topalkara A, Arıcı MK, Erdoğan H, Toker Mİ (2012) Comparison of total oxidative stress, total antioxidant capacity, and paraoxonase, arylesterase, and lipid peroxidase levels in aqueous humor and serum of diabetic and non-diabetic patients with cataract. TJO 42:1
Draganov DI, LaDu NB (2004) Pharmacogenetics of paraoxonases: a brief review. Naunyn Schmiedeberg's Arch Pharmacol 369(1):78–88
Al-Saleh IA, Al-Doush I (1997) Selenium levels in wheat grains grown in Saudi Arabia. Bull Environ Contam Toxicol 59:590–594
Kang DH (2002) Oxidative stress, DNA damage, and breast cancer. AACN Clin Issues 13:540–549
Abdel Salam OME, El-Shamarka ME, Omara EA (2018) Brain oxidative stress and neurodegeneration in the ketamine model of schizophrenia during antipsychotic treatment: effects of N-Acetyl cysteine treatment. Reactive Oxygen Species J 6(16):253–266
Ozdulger A, Cinel I, Koksel O, Cinel L, Avlan D, Unlu A, Okcu H, Dikmengil M, Oral U (2003) The protective effect of N-acetylcysteine on apoptotic lung injury in cecal ligation and puncture-induced sepsis model. Shock. 19(4):366–372
Prıce TA, Uras F, Banks WA, Ercal N (2006) A novel antioxidant N-acetylcysteine amide prevents gp120- and Tat-induced oxidative stress in brain endothelial cells. Exp Neurol 201:193–202
Jasna JM, Anandbabu K, Bharathi SR, Angayarkanni N (2014) Paraoxonase enzyme protects retinal pigment epithelium from chlorpyrifos insult. PLoS One 9(6):e101380
Frank L, Massaro D (1980) Oxygen toxicity. Am J Med 69(1):117–126
Freeman BA, Crapo JD (1982) Biology of disease: free radicals and tissue injury. Lab Investig 47:412–426
Niki E, Yoshida Y, Saito Y, Noguchi N (2005) Lipid peroxidation: mechanisms, inhibition, and biological effects. Biochem Biophys Res Commun 338:668–676
Deckbar D, Jeggo PA, Löbrich M (2011) Understanding the limitations of radiation-induced cell cycle checkpoints. Crit Rev Biochem Mol Biol 46(4):271–283
Strasser A, Harris AW, Huang DC, Krammer PH, Cory S (1995) Bcl-2 and Fas/APO-1 regulate distinct pathways to lymphocyte apoptosis. EMBO J 14(24):6136–6147
Hazman Ö, Bozkurt MF, Fidan AF, Uysal FE, Çelik S (2018) The effect of boric acid and borax on oxidative stress, inflammation, ER stress and apoptosis in cisplatin Toxication and nephrotoxicity developing as a result of toxication. Inflammation 41(3):1032–1048
Yazıcı S, Aksit H, Korkut O, Sunay B, Çelik T (2014) Effects of boric acid and 2-aminoethoxydiphenyl borate on necrotizing enterocolitis. Gastroenterology 58(1):61–67
Khaliq H, Jing W, Ke X, Ke-Li Y, Peng-Peng S, Cui L, Wei-Wei Q, Zhixin L, Hua-Zhen L, Hui S, Ju-Ming Z, Ke-Mei P (2018) Boron affects the development of the kidney through modulation of apoptosis, antioxidant capacity, and nrf2 pathway in the African ostrich chicks. Biol Trace Elem Res 186:226–237
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
This study was conducted in agreement with the precepts of Atatürk University Animal Experimental and Ethical Committee number with 75296309-050.01.04-E.2000066104. The study was approved by the committee in Erzurum, Turkey.
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Alak, G., Ucar, A., Parlak, V. et al. Antioxidant Potential of Ulexite in Zebrafish Brain: Assessment of Oxidative DNA Damage, Apoptosis, and Response of Antioxidant Defense System. Biol Trace Elem Res 199, 1092–1099 (2021). https://doi.org/10.1007/s12011-020-02231-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-020-02231-7