Abstract
Rheumatoid arthritis (RA) is an autoimmune disorder that is a painful health crisis. This study aimed to assess the serum C-reactive protein (CRP), malondialdehyde (MDA), non-enzymatic antioxidant (vitamin C), and trace elements (Zn, Cu, Mn, and Fe) in RA patients, and thereby correlate these parameters with the association of RA. This study included 20 Bangladeshi RA patients and 20 normal healthy volunteers as control subjects. CRP level was determined using a laboratory-based latex agglutination–enhanced immunoassay. The lipid peroxidation level was determined by measurement of the serum level of MDA. Non-enzymatic antioxidant vitamin C was assessed by UV spectrophotometric method. Trace elements were determined by atomic absorption spectroscopy (AAS). Our study observed significantly higher concentrations of CRP (p < 0.001) and MDA (p < 0.001), and significantly lower concentrations of vitamin C (p < 0.001) in the RA patient. The mean values of Zn, Cu, Mn, and Fe were 6.62 ± 0.34, 1.42 ± 0.17, 7.51 ± 0.23, and 29.25 ± 0.41 ppm for the RA patients respectively and 13.57 ± 9.13, 1.15 ± 0.17, 1.59 ± 0.18, and 62.47 ± 5.25 ppm for the control subjects, consequently. There was a significant difference (p < 0.05) in the trace element levels between the RA patients and control subjects. Our study suggests that a higher concentration of CRP and MDA, lower levels of vitamin C, and altered trace elements may be linked to RA.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Rheumatoid arthritis (RA) is a severe autoimmune disorder with various types of immunological and clinical difficulties [1, 2]. Worldwide, it affects 0.5–1% of the population. Among the rural population of Bangladesh, the prevalence of RA is 0.7% and 0.4% in the city dwellers [3]. The prevalence of RA patients in Bangladesh is increasing with time and is a neglected medical problem in this country. RA is becoming a major health crisis in Bangladesh due to the lack of proper management of the disease. It is a progressive autoimmune disease characterized by synovitis, articular damage, swelling of joint, motion impairment, morning stiffness, fatigue, physical disabilities, weight loss, malaise, etc. [4, 5]. It is also associated with cardiovascular, skeletal, pulmonary disorders, and early death [6].
Although it can affect individuals of any age, it typically has a gradual appearance occurring in 80% of patients of 35–50 years of age. It tends to occur in families more in women than in men (3:1) [7, 8]. In many populations around the world, complex genetic and environmental influences are correlated with RA. HLA DRB1*0404 allele is the strongest susceptible factor associated with RA. Though genes only explain half of the genetic effect. Many other factors may be involved [9]. In recent years, several studies are trying to offer new views on the disease mechanism of RA. There are several treatment options that tackle RA, but different patients need different treatment options to benefit. Identifying biomarkers to target the right patients for the right therapies will bring immediate benefits to patients in RA. Many treatment options such as biologic drugs are very costly. Still, now more studies are needed to better understand RA and achieving better therapy for RA [6, 10].
C-reactive protein (CRP) is an acute-phase protein developed by liver hepatocytes. CRP is a precise marker of inflammation [11]. Increase level of CRP is associated with the inflammation [12]. CRP can be used as a pathological tool for the measurement of inflammation in RA. Although the exact disease mechanism of RA is yet to be known, it is believed that reactive oxygen species (ROS) play an important role in the pathogenesis of RA [7]. ROS attacks polyunsaturated fatty acids (PUFA) of cell membranes and induces lipid peroxidation [12]. The elevated level of lipid peroxidation, hydrogen peroxide, and superoxide in plasma and red blood cells (RBC) induces oxidative stress in RA [13, 14]. Cells have several endogenous antioxidants, enzymes, and chemicals to defend from ROS-induced attacks [15]. Malondialdehyde (MDA) is the parameter of oxidative stress and is the lipid peroxidation product that is used to determine the level of damage of cell membrane lipid by ROS. Cell membrane permeability is affected by elevated levels of MDA. The elevated levels of MDA are counteracted by various enzymatic and non-enzymatic antioxidants through the conversion of superoxide free radicals to hydrogen peroxide and oxygen molecule [16,17,18]. Hydrophilic vitamin C is a non-enzymatic antioxidant which undermines the ROS-induced tissue and cell damage and protects it [16, 19]. It works immediately against hydrogen peroxides and ROS [20].
Trace elements in minute amounts are essential for the healthy balance of human health. But, imbalances of the amount of trace elements lead to various hazardous effects. Trace elements are an important component of larger molecules and have definite structural and catalytic functions for life [21]. Zinc (Zn), copper (Cu), manganese (Mn), and iron (Fe) are important trace elements required for healthy regulation of human health. Zn plays a diverse function in human health including regulation of normal growth and cell division, metabolism of protein, wound healing, immune function, metabolic function, and antioxidant activity [22, 23]. Inadequate level of Zn leads the way of intracellular killing, phagocytosis, the formation of cytokines, and injurious effects of B and T cells [21]. Cu is an important component of enzymes and has various functions including oxygen and electron transport; protection of the cell against ROS-induced injury [24, 25]. It also produces hemoglobin; modulates myelin function, melanin, and thyroid normal activity; and functions as antioxidant and pro-oxidant [26, 27]. Moreover, it is essential for cartilage mineralization, elastin, and collagen constitution, cartilage cross-linking and bony structures [28]. Lack of Cu in the human body affects the antioxidant defense system, reduction of hemoglobin, and leads to the generation of the inflammatory response and oxidative stress [21, 29]. Mn is also an important trace element involved in cellular energy production, bone growth, and reproduction. It also associated with blood clotting factors and connective tissue formation [30]. It regulates neurotransmitters and involved in the muscle or nerve dysfunction. Imbalance of Mn is associated with various diseases [31]. Again, Fe is also a major constituent of several metalloproteins and plays a biochemical function in electron transport, oxygen transport, and catalytic activity [32]. It is also a component of hemoglobin and myoglobin. It is essential for DNA synthesis, normal growth, healing, and immune functioning [33]. Deficiency of Fe causes anemia but an elevated level of Fe is toxic which develops ROS [34]. It is also reported that Fe-related oxidative damage is linked to diseases [21]. Although various earlier studies had been carried out to evaluate the effect of oxidative stress in RA, there is lack of data regarding the correlation among oxidative stress, trace element, and aspect of inflammation in RA. Taking the above consideration in mind, for the first time in Bangladesh, this current study aimed to investigate oxidative stress and inflammation and to measure serum Zn, Cu, Mn, and Fe level, and their correlation with other findings in Bangladeshi RA patients.
Materials and Methods
Study Design and Blood Sample Collection
This case-control study is carried out in the Department of Medicine, Dhaka Medical College and Hospital (DMCH), Dhaka. The ethical committee of the DMCH approved the research protocol and gave ethical permission (reference no: MEU-DMC/ECC/2016/160). This study included 20 RA patients and 20 normal healthy volunteers as control subjects based on gender. Patients with synovial inflammation, joint pain, and damage to the joint cartilage or bones reported on their X-ray examination were recruited by a physician for the study. The study included only active RA patients greater than 18 years old. Patients who suffered from other connective tissue diseases and did not wish to take part in the study were excluded. Each RA patient and control subjects gave verbal and written consent about the study and the documents were collected. In all investigations, principles of the Declaration of Helsinki were followed. The patients having respiratory, cardiovascular, and pathologic disorders other than RA were excluded in the study. RA patients were enrolled by physician recommendation. The subjects recruited in the study had not taken any type of antioxidants and trace elements other than RA therapy that may interfere with the level of antioxidants and trace elements. With the help of a predesigned questionnaire, sociodemographic and anthropometric data were collected. Different biological features including age, height, weight, and blood pressure were also collected for both RA and control groups.
Five milliliters of blood was taken from each RA and control group subjects after overnight fasting. Then the blood sample was kept to clot for 1 h at room temperature and after centrifugation at 3000 rpm for 15 min, serum from the blood samples was kept into microtubes and stored at − 80 °C until further analysis. These stored samples were used for measuring the amount of serum concentration of lipid peroxide (MDA), antioxidant vitamin C, CRP, and trace elements.
Chemicals and Reagents
All reagents and chemicals used for the study were of analytical grade from a renowned company. Standards of zinc, copper, iron, and manganese were bought from Buck Scientific, USA. α-Tocopheryl acetate standard applied in this study was obtained from Sigma Chemical Co., USA. HPLC grade chemicals and reagents were purchased from Active Fine Chemicals Limited, Dhaka, Bangladesh, for HPLC-based analysis. Hydrochloric acid (37%) and nitric acid were purchased from Merck, Germany. Other accessories and required chemicals of acceptable grade were provided by the laboratory of the Department of Pharmacy, Noakhali Science and Technology University.
Statistical Analysis
All the values of MDA, antioxidant vitamin C, CRP, and trace elements were showed as the mean ± standard deviation (SD). The values were compared between the RA patients and the controls with independent sample t tests. Pearson’s correlation was also used to correlate among various study parameters and p < 0.05 was considered to be statistically significant. The statistical analysis of data was executed using SPSS statistical software, version 19.0 (Armonk, NY: IBM Corp.)
Determination of CRP
Serum CRP concentrations were determined using a laboratory-based latex agglutination–enhanced immunoassay and also semi-quantitatively by the CRP Wellcotest rapid latex test kit (Wellcome Diagnostics, Dartford, England) following the method described by Richards et al. [34]. Initially, samples were diluted 1/2, 1/4, 1/8, and 1/16 in normal saline for semi-quantitative estimation, as recommended by the manufacturers. Fifty microliters of the diluted serum was then added to one drop of CRP latex from the Wellcotest kit and these were mixed on a glass slide. After 2 min, the presence of any agglutination was observed under artificial light and was recorded and interpreted as a positive result [35].
Quantification of MDA Level
Serum MDA level was measured according to the previously published method [36, 37] using thiobarbituric acid (TBA) reagent. The absorbance of the supernatant was measured spectrophotometrically at the absorbance of 530 nm and the level of MDA was showed as nmol/mL.
Determination of Vitamins C
To measure the vitamin C (ascorbic acid) concentration by a spectrophotometric-based phenyl-hydrazine method was used [38]. The absorbance of 520 nm was used for the sample and standard.
Determination of Trace Elements
The serum level of trace elements was measured by flame atomic absorption spectrometry (FAAS) following the method with a slight change described in the previously published article [39, 40]. A serum sample is diluted with deionized water 1:10 dilution and different concentrations of minerals (0.5, 1.0, 2.0, 5.0, and 10.0 mg/L) were used to prepare the calibration curve. Then the concentrations of trace elements were measured by reading the absorbance’s at 213.9, 324.8, 279.5, and 248.3 nm for Zn, Cu, Mn, and Fe respectively.
Results
This study incorporated RA patients as cases and normal healthy volunteers as controls. Socio-demographic parameters in RA patients and control subjects are shown in the following tables.
Socio-demographic Profile
Table 1 presents the socio-demographic features of both RA patients and controls involved in the study. The average age of the RA patients and control subjects was 36.95 ± 2.22 and 32.20 ± 2.67 years respectively. Women are more prone to RA as we have seen that among the 20 RA patients on the study, 75% (n = 20) were women. In this study, it was also observed that patients from the urban area had higher prevalence of RA than those from the rural area as 65% and 35% of patients were located from an urban and rural area in comparison with that of 60 and 40% for control subjects respectively. This study explored that most of the patients were housewife in both RA patients and control subjects (70% vs 75%) and the remaining small portions were of the student (20% vs 20%), service holder (5% vs 0%), and businessman (5% vs 0%). Analysis of the educational status of the RA explored that 5% of them were illiterate while 10, 70, 10, and 5% continued their study up to primary, secondary, higher secondary, and graduate respectively, whereas in the control subjects, these were 0, 15, 65, 15, and 5% respectively.
Anthropometric, Clinical, and Biochemical Parameters
Anthropometric and biochemical data of the study population are shown in Table 2. The average values of body mass index (BMI) were 22.38 ± 0.84 for RA patients and 23.39 ± 0.36 kg/m2 for the control subjects respectively. The mean values of systolic blood pressure (SBP) and diastolic blood pressure (DBP) were 125.15 ± 4.27 and 82.00 ± 2.89 mmHg for RA patients group while for the control subjects 121.24 ± 2.39 and 78.92 ± 1.14 mmHg respectively.
CRP, MDA, and Antioxidants (Vitamin C) Level
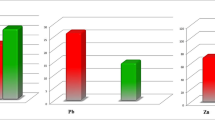
CRP, MDA, and vitamin C levels of the study population are presented in Fig. 1. The serum level of CRP was found 38.12 ± 6.98 and 3.16 ± 0.21 in RA patients and control subjects, respectively, which was statistically significant (p < 0.01). MDA level was found in RA patients 3.85 ± 0.34 nmol/mL which was significantly (p < 0.01) higher in comparison with that of 1.76 ± 0.15 nmol/mL for control subjects. Statistical analysis of vitamin C concentration showed a significantly (p < 0.01) lower level in RA patients compared with control subjects where the mean concentration of vitamin C was found 24.00 ± 3.69 and 31.51 ± 1.64 μmol/L for RA patients and control subjects respectively.
Correlation of Anthropometric and Clinical Characteristics with Serum MDA and Vitamin C
Correlation analysis was performed between variables presented in Table 3. There is a significant (p < 0.05) positive correlation between age and DBP in both RA patients (r = 0.451, p = 0.046) and control subjects (r = 0.831, p = 0.000). In RA patients, there is a significant (p < 0.05) positive correlation between SBP and DBP (r = 0.893, p = 0.000), and CRP and vitamin C (r = 0.513, p = 0.021). In control subjects, there is significant (p < 0.05) positive correlation between DBP and vitamin C (r = 0.479, p = 0.015). All other positive and negative correlation was not statistically significant.
Status of Trace Elements in the Study Population
The mean concentrations of trace elements for RA patients and control subjects are given in Fig. 2; where control subjects showed maximum Zn and Fe value either than RA patients that was 13.57 ± 9.13 ppm and 62.47 ± 5.25 ppm respectively. The difference between the values obtained for these two groups was found statistically significant (p < 0.01). But the mean concentrations of Cu and Mn were significantly (p < 0.05) higher in RA patients than in control subjects that were 1.42 ± 0.17 ppm and 7.51 ± 0.23 ppm, respectively for RA patients.
Effect of Age, BMI, SBP, DBP, CRP, MDA, and Vitamin C on Serum Trace Elements of RA Patients
The data of RA patients were further analyzed to determine the effect of age, BMI, SBP, DBP, and CRP on the serum trace elements level in RA patients (Table 4). We observed a significant (p < 0.05) positive correlation between age and Cu (r = 0.518, p = 0.019), CRP and Cu (r = 0.609, p = 0.004), and vitamin C and Cu (r = 0.451, p = 0.046). We also observed significant (p < 0.05) negative correlation between CRP and Fe (r = − 0.588, p = 0.006), and vitamin C and Fe (r = − 0.469, p = 0.037).
Inter-element Correlations
This study identified inter-element associations between patients with RA and control subjects for the trace elements tested. Table 5 depicts the correlation coefficient and the rate of statistical confidence at which the correlations were calculated. The test showed that patients with RA and control subjects had a negative correlation between Zn and Mn, Cu and Fe, and Mn and Fe. The association between Zn and Cu and Fe was positive in RA patients but negative in control subjects. Only control subjects demonstrate statistically significant (p < 0.05) negative correlation (r = − 0.486, p = 0.014) between Cu and Fe. But for both study classes, all other positive and negative association between inter-element correlations was not considered statistically significant.
Relative Mole Percentage and Mole Percentage Ratios
Figure 3 demonstrates the relative mole percentage of the tested elements. Then, the relative mole percentage ratios of the elements were calculated. Our data exhibited that the element to element mole percentage ratios, namely, Zn/Cu, Zn/Mn, Cu/Fe, Cu/Mn, Cu/Fe, Mn/Fe were dissimilar in the RA patients relative to controls (Table 6).
Discussion
Our study principally focused to identify several biochemical parameters that may play a key role in the pathogenesis of RA. We had also analyzed the socio-demographic characteristics of the study subjects and tried to find out the relationship of these parameters with the disease progression. In this study, we observed an increased level of serum CRP and MDA and decreased level of vitamin C in RA patients than in control subjects which indicates the imbalance of oxidant and antioxidants in the body. It also observed that low serum concentrations of Zn and Fe and higher serum concentrations of Cu and Mn strongly associated with RA patients.
CRP is the marker of systemic inflammation. An elevated level of CRP indicates higher inflammation in the body. It is thought that the increased level of CRP is linked in the inflammation of rheumatoid disorder [41]. Our study observed a significantly higher level of CRP in RA patients than in control subjects (p < 0.001). The previous study also reported a higher level of CRP in RA patients than in controls [42].
Oxygen radical generates a free radical chain reaction which leads to the production of lipid peroxides. Lipid peroxide destroys the essential antioxidant enzymes. The antioxidant helps in the removal of oxygen radicals. MDA is the final product of lipid peroxidation which is responsible for oxidative stress and oxidative stress is the cause of various diseases including rheumatoid arthritis, osteoarthritis, and atherosclerosis. So, the balance between oxygen radicals and antioxidant enzymes is important [7, 36]. The previous study reported an increased level of lipid peroxidation in RA patients [41, 43]. Our study also observed a significantly higher level of lipid peroxidation in RA patients than in control subjects (p < 0.001). In contrast, another study with RA patients found no difference in lipid peroxidation level between RA patients and control subjects [44].
Antioxidants provide defense against oxidative damage by the prevention of lipid peroxidation. It helps in inactivating the oxygen radicals. Vitamin C acts as co-factor in various enzymatic reactions. It works against inflammatory biomarkers [45]. Our study found that the level of antioxidant vitamin C was significantly (p < 0.01) depleted in RA patients when compared with the control subjects. Pearson’s correlation analysis confirmed that there was a negative correlation between the serum level of MDA and vitamin C in both patient and control groups. This may be due to the fact that vitamin C may utilize to a greater extent to counteract lipid peroxidation of cellular components, hence resulting in the depletion of this antioxidant level [46]. The previous study also reported a low level of vitamin C in RA patients than in controls [7, 47].
Trace elements present in a very minute amount in the human body and involve in immune functioning. Numerous reports suggest the involvement of trace elements in RA. An imbalance in trace elements causes harmful effects for the body [37]. In this study, we measured the serum level of Zn, Cu, Mn, and Fe. Zn is an important trace element that has potential antioxidant activity by acting as cofactor many enzymes. It also protects from aging [48]. Alteration of zinc metabolism is associated with RA. Leucocytes and activated phagocytes secrete some interleukins which cause Zn deficiency by interfering transport of Zn from plasma to the liver [49]. The previous study reported a lower level of Zn in RA patients than in controls [50]. These observations are similar to our findings that presented significantly decrease the level of Zn in RA patients than in control subjects (p < 0.001). In contrast, another study with RA patients found no difference in Zn in RA patients and control subjects [51]. An adequate level of Cu is essential for normal regulation of human health. Inflammation is a major problem in RA patients. An elevated level of Cu is associated with inflammation [52]. Our study found a significantly higher level of Cu in RA patients when compared with control subjects (p < 0.001). The previous study also reported higher level of Cu in RA patients than in controls [51]. Our study also found reduced Zn/Cu ratio. This may be the result of a decreased level of Zn and an increased level of Cu. Decrease Zn/Cu ratio is the result of malnutrition. An earlier study reported that alteration of the Zn/Cu ratio causes atherosclerosis [50]. Mn is also an important trace element. Our study found a significantly higher level of Mn in RA patients when compared with control subjects (p < 0.001). In contrast, another previous study also found no significant change of Mn level in RA patients [53]. Zn/Mn ratio is higher in RA patients than in control subjects. Again, Cu/Mn ratio is lower in RA patients than in control subjects. Fe level is essential for immune system functioning. It is also responsible for inflammation and anemia [54]. Our study found a significantly lower level of Fe in RA patients when compared with control subjects (p < 0.001). A lower level of Fe may be associated with the inflammation in RA. The previous study also found a lower level of Fe in RA [54]. Zn/Fe and Mn/Fe ratios are higher in RA patients than in control subjects. Again, Cu/Fe ratio is lower in RA patients than in control subjects.
According to the above discussion presented here, these results depicted that RA is linked with the peroxidation of lipids, elevated CRP, and altered level of vitamin and trace elements during the diurnal diet in all the patients included in our study. It is clear about antioxidant that it can diminish the inflammation in RA patient. So, it is suggested that to take proper dietary antioxidants along with proper intake of trace elements containing food in case of RA is necessary.
Limitations of the Study
We should mention some limitations aside from the importance of our present study. However, in our sample conditions, we did not observe the supplementation of dietary effect; thus, further research may be necessary to determine whether or not nutritional supplements would improve the condition of RA patients. The study was also conducted on a small number of subjects, so it lacks the exact severity of disease scores. The real RA scenario may be depicted by a large-scale analysis with a greater number of samples from different regions of Bangladesh. Although this research still has some limitations, we hope our study will play an important role in creating a new pathological resource for Bangladesh-based RA patients.
Conclusion
Our analysis showed patients with Bangladeshi RA had increased serum levels of CRP and MDA, reduced concentration of vitamin C, and altered concentrations of serum trace elements with control subjects. This study found lower serum levels of Zn and Fe but increased serum levels of Mn and Cu in RA patients as compared with control subjects. Hence, increased serum MDA, altered serum antioxidants, and trace elements will impact RA pathogenesis in Bangladeshi patients. Therefore, proper nutritional intervention with vitamins and trace elements in Bangladeshi RA patients promotes the management of RA and reduces its complications.
Abbreviations
- RA:
-
rheumatoid arthritis
- CRP:
-
C-reactive protein
- MDA:
-
malondialdehyde
- ROS:
-
reactive oxygen species
- BMI:
-
body mass index
- SBP:
-
systolic blood pressure
- DBP:
-
diastolic blood pressure
- FAAS:
-
flame atomic absorption spectrometry
- HPLC:
-
high-performance liquid chromatography
- TBA:
-
thiobarbituric acid
References
Ala S, Shokrzadeh M, Pur SA, Saeedi SS (2009) Zinc and copper plasma concentrations in rheumatoid arthritis patients from a selected population in Iran. Pak J Biol Sci 12:1041–1044
Stejskal V, Reynolds T, Bjørklund G (2015) Increased frequency of delayed type hypersensitivity to metals in patients with connective tissue disease. J Trace Elem Med Biol 31:230–236
Haq SA, Darmawan J, Islam MN, Uddin M, Das BB, Rahman F, Chowdhury MA, Alam MN, Mahmud TA, Chowdhury MR, Tahir M (2005) Prevalence of rheumatic disease and associated outcomes in rural and urban communities in Bangladesh: a COPCORD study. J Rheumatol 32:348–353
Firestein GS (2003) Evolving concepts of rheumatoid arthritis. Nature 423:356–361
Grassi W, De Angelis R, Lamanna G, Cervini C (1998) The clinical features of rheumatoid arthritis. Eur J Radiol 27(S1):S18–S24
Corsiero E, Marrelli A (2018) An update on research advances in rheumatoid arthritis: from clinic to basic science. J Lab Precis Med 3:54
Mateen S, Moin S, Khan AQ, Zafar A, Fatima N (2016) Increased reactive oxygen species formation and oxidative stress in rheumatoid arthritis. PLoS One 11:e0152925
Dennis Wolf, Rheumatoid arthritis, xPharm: the comprehensive pharmacology reference, 2007, 1–11
Silman AJ, Pearson JE (2002) Epidemiology and genetics of rheumatoid arthritis. Arthritis Res Ther 4(S3):S265
Viatte S, Barton A (2017) Genetics of rheumatoid arthritis susceptibility, severity, and treatment response. Semin Immunopathol 39(4):395–408
Gaujoux-Viala C (2013) C-reactive protein versus erythrocyte sedimentation rate in estimating the 28-joint disease activity score. J Rheumatol 40:1785–1787
Uddin MG, Hossain MS, Rahman MA, Uddin AHMM, Bhuiyan MS (2017) Elemental zinc is inversely associated with C- reactive protein and oxidative stress in chronic liver disease. Biol Trace Elem Res 78:189–193
Turi S, Nemeth I, Torkos A, Saghy L, Varga I, Matkovics B et al (1997) Oxidative stress and antioxidant defense mechanism in glomerular diseases. Free Radic Biol Med 22:161–168
Suryaprabha P, Das UN, Ramesh G, Kumar KV, Kumar GS (1991) Reactive oxygen species, lipid peroxides and essential fatty acids in patients with rheumatoid arthritis and systemic lupus erythematosus. Prostaglandins Leukot Essent Fat Acids 43:251–255
Sarban S, Kocyigit A, Yazar M, Isikan UE (2005) Plasma total antioxidant capacity, lipid peroxidation, and erythrocyte antioxidant enzyme activities in patients with rheumatoid arthritis and osteoarthritis. Clin Biochem 38(11):981–986
Vijayakumar D, Suresh K, Manoharan S (2006) Lipid peroxidation and antioxidant status in blood of rheumatoid arthritis patients. Indian J Clin Biochem 21:105
Bagis S, Tamer L, Sahin G, Bilgin R, Guler H, Ercan B, Erdogan C (2005) Free radicals and antioxidants in primary fibromyalgia: an oxidative stress disorder? Rheumatol Int 25:188–190
García-González A, Gaxiola-Robles R, Zenteno-Savín T (2015) Oxidative stress in patients with rheumatoid arthritis. Rev Investig Clin 67:46–53
Ray G, Husain SA (2002) Oxidants, antioxidants and carcinogenesis. Indian J Exp Biol 40:1213–1232
Vincent HK, Taylor AG (2002) Biomarkers and potential mechanisms of obesity-induced oxidant stress in humans. Int J Obes 30:400–418
Ullah Z, Ullah MI, Hussain S, Kaul H, Lone KP (2017) Determination of serum trace elements (Zn, Cu, and Fe) in Pakistani patients with rheumatoid arthritis. Biol Trace Elem Res 175:10–16
Prasad AS (2008) Zinc in human health: effect of zinc on immune cells. Mol Med 14:353–357
Maret W, Sandstead HH (2006) Zinc requirements and the risks and benefits of zinc supplementation. J Trace Elem Med Biol 20:3–18
Harris ED (1983) Copper in human and animal health. Trace Elem Health Ed J 37:145–149
Strecker D, Mierzecki A, Radomska K (2013) Copper levels in patients with rheumatoid arthritis. Ann Agric Environ Med 20:312–316
Harris ED (2001) Copper homeostasis: the role of cellular transporters. Nutr Rev 59:281–285
Araya M, Pizarro F, Olivares M, Arredondo M, Gonzalez M, Mendez M (2006) Understanding copper homeostasis in humans and copper effects on health. Biol Res 39:183–187
Wu G, Gao X, Zhu J, Hu C, Ye G, Liu N (2014) Copper resistance selection and activity changes of antioxidases in the flesh fly Boettcherisca peregrine. J Insect Sci 14:116
Chen H, Huang G, Su T, Gao H, Attieh ZK, McKie AT, Anderson GJ, Vulpe CD (2006) Decreased hephaestin activity in the intestine of copper-deficient mice causes systemic iron deficiency. J Nutr 136:1236–1241
Morello M, Zatta P, Zambenedetti P, Martorana A, D’Angelo V, Melchiorri G, Bernardi G, Sancesario G (2007) Manganese intoxication decreases the expression of manganoproteins in the rat basal ganglia: an immune histochemical study. Brain Res Bull 74:406–415
Chandra RK (1990) Micronutrients and immune functions: an overview. Ann N Y Acad Sci 587:9–16
Aisen P, Enns C, Wessling-Resnick M (2001) Chemistry and biology of eukaryotic iron metabolism. Int J Biochem Cell Biol 33:940–959
Beard JL (2001) Iron biology in immune function, muscle metabolism and neuronal functioning. J Nutr 131:568S–579S
McCord JM (1998) Iron free radicals, and oxidative injury. Semin Hematol 35:5–12
Richards NP, Elliott TS, Powell RJ, O’Callaghan C, Franklin P (1985) Can the rapid semi quantitative estimation of serum C reactive protein be adapted for the management of bacterial infection? J Clin Pathol 38:464–467
Sarwar MS, Sarkar RC, Bhowmick R, Dewan SMR, Ahmed MU, Hasnat A, Rashid M, Islam MS (2015) Effect of socio-economic status and estimation of lipid peroxidation and antioxidant in pre-eclamptic pregnant women: a case–control study. Hypertens Pregnancy 34:125–135
Chowdhury MI, Hasan M, Islam MS, Sarwar MS, Amin MN, Uddin SMN, Rahaman MZ, Banik S, Hussain MS, Yokota K, Hasnat A (2017) Elevated serum MDA and depleted non-enzymatic antioxidants, macro-minerals and trace elements are associated with bipolar disorder. J Trace Elem Med Biol 39:162–168
Rahman MS, Hasan K, Hussain MS, Millat MS, Sen N, Islam MS, Sarwar MS, Noor W, Kar A, Uddin SMN (2018) Pathophysiological status of serum antioxidant, macro-minerals and trace elements in patients with metabolic syndrome in Bangladesh. Int J Pharm Sci 9:1012–1022
Sarwar MS, Ahmed S, Ullah MS, Kabir H, Rahman GK, Hasnat A, Islam MS (2013) Comparative study of serum zinc, copper, manganese, and iron in preeclamptic pregnant women. Biol Trace Elem Res 154:14–20
Amin MN, Liza KF, Sarwar MS, Ahmed J, Adnan MT, Chowdhury MI, Hossain MZ, Islam MS (2015) Effect of lipid peroxidation, antioxidants, macro minerals and trace elements on eczema. Arch Dermatol Res 307:617–623
Xiao Y, Haynes WL, Michalek JE, Russell IJ (2013) Elevated serum high-sensitivity C-reactive protein levels in fibromyalgia syndrome patients correlate with body mass index, interleukin-6, interleukin-8, erythrocyte sedimentation rate. Rheumatol Int 33:1259–1264
Graf J, Scherzer R, Grunfeld C, Imboden J (2009) Levels of C-reactive protein associated with high and very high cardiovascular risk are prevalent in patients with rheumatoid arthritis. PLoS One 4:e6242
Baskol G, Demir H, Baskol M, Kilic E, Ates F, Karakukcu C, Ustdal M (2006) Investigation of protein oxidation and lipid peroxidation in patients with rheumatoid arthritis. Cell Biochem Funct 24:307–311
Kajanachumpol S, Vanichapuntu M, Verasertniyom O, Totemchokchyakarn K, Vatanasuk M (2000) Levels of plasma lipid peroxide products and antioxidant status in rheumatoid arthritis. Southeast Asian J Trop Med Public Health 31:335–338
Mah E, Matos MD, Kawiecki D, Ballard K, Guo Y, Volek JS, Bruno RS (2011) Vitamin C status is related to pro inflammatory responses and impaired vascular endothelial function in healthy, college-aged lean and obese men. J Am Diet Assoc 111:737–743
Sharma JB, Sharma A, Bahadur A, Vimala N (2006) Oxidative stress markers and antioxidant levels in normal pregnancy and pre-eclampsia. Int J Gynecol Obstet 94:23–27
Vijayakumar D, Suresh K, Manoharan S (2006) Lipid peroxidation and antioxidant status in blood of rheumatoid arthritis patients. Indian J Clin Biochem 21:104–108
Frassinetti S, Bronzetti G, Caltavuturo L, Cini M, Croce CD (2006) The role of zinc in life: a review. J Environ Pathol Toxicol Oncol 25:597–610
Svenson KLG, Hallgren R, Johansson E, Lindh U (1985) Reduced zinc in peripheral blood cells from patients with inflammatory connective tissue disease. Inflammation 9:189–199
Sahebari M, Ayati R, Mirzaei H, Sahebkar A, Hejazi S, Saghafi M, Saadati N et al (2016) Serum trace element concentrations in rheumatoid arthritis. Biol Trace Elem Res 171:237
Önal S, Nazıroğlu M, Çolak M, Bulut V, Flores-Arce MF (2011) Effects of different medical treatments on serum copper, selenium and zinc levels in patients with rheumatoid arthritis. Biol Trace Elem Res 142:447–455
Le NT, Xue M, Castelnoble LA, Jackson CJ (2007) The dual personalities of matrix metalloproteinases in inflammation. Front Biosci 12:1475–1487
Arabi SYA, Elmahdi SA, Mohamed SB, Abdrabo AA (2016) Evaluation of chromium and manganese level in rheumatoid arthritis patients. J Biomed Res 2:7–10
Baker JF, Ghio AJ (2009) Iron homoeostasis in rheumatic disease. Rheumatology 48:1339–1344
Acknowledgments
The authors are thankful to all the staff and physicians at the Dhaka Medical College and Hospital (DMCH), Dhaka, Bangladesh, for their generous help during sample collection. The authors are also thankful for the technical and laboratory support provided by Agrochemical and Environmental Research Division, Institute of Food and Radiation Biology, Bangladesh Atomic Energy Research Establishment, Dhaka. The authors express their gratitude to all the staff and faculty members of the Department of Pharmacy, Noakhali Science and Technology University, Bangladesh, for providing laboratory facilities.
Author information
Authors and Affiliations
Contributions
DCD, IJ, and MGU participated in protocol designing and writing. MMH, MAZC, ZF, MMR, AKMHK, SRD, and MABS contributed in drafting and editing. AD checked and approved the overall manuscript.
Corresponding author
Ethics declarations
This case-control study is carried out in the Department of Medicine, Dhaka Medical College and Hospital (DMCH), Dhaka. The ethical committee of the DMCH approved the research protocol and gave ethical permission (reference no: MEU-DMC/ECC/2016/160).
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Das, D.C., Jahan, I., Uddin, M.G. et al. Serum CRP, MDA, Vitamin C, and Trace Elements in Bangladeshi Patients with Rheumatoid Arthritis. Biol Trace Elem Res 199, 76–84 (2021). https://doi.org/10.1007/s12011-020-02142-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-020-02142-7