Abstract
In this modern era, people are paying more attention to adopting healthy lifestyles and suitable nutritional diets. To meet the increasing demand, new food sources are continuously being identified. The present review focuses on underutilized cereal crops, commonly known as pseudo-cereals (Buckwheat, Quinoa, and Amaranth), and their nutritional products. The nutritional properties, amino acid profile, essential amino acid indices, protein efficiency ratio, nutritional index, and biological functions are higher in pseudo-cereals than other true crops. We comprehensively discussed pseudo-cereals’ characteristics and nutritional composition, bioactive components, and functional properties of pseudo-cereals. Also, the processing treatments and applications of pseudo-cereals as dietary food were discussed. Finally, the current challenges in using pseudo-cereals as dietary food supplements were analyzed, and recommendations were made for future studies.
Graphical Abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The greatest challenge of human survival is to meet the food supply demand to the growing population, which is expected to rise 9.8 billion in 2050 (United Nation, 2017). It is also expected that the urbanized area will increase threefold by 2030, which ultimately generate huge pressure on the available production area (d’Amour et al., 2017). Also, the global average daily intake of energy will be increased to 3000 kcal by 2050. Protein as one of the main components of diet played a significant role in the development and physiological functioning of all life forms. Due to increasing pressure to meet the protein demand, animal-based protein consumption is expected to increase, ultimately increasing related environmental problems. This needs identification of new sources and their utilization as food products (Kaur et al., 2022; Langyan et al., 2021a, 2022a).
Plant-based protein sources are extensively searched to meet the protein requirements. From nutritional aspects, the functionality and application of any food primarily depend on proteins. However, their availability in many foods is limited, and the requirements are meet from animal sources (Langyan et al., 2022b; Singh et al., 2022). Food security has been largely dependent on majorly used cereal crops such as rice, wheat, and corn. These grains are an important part of the human diet, yet they lack the essential micronutrients which lead to health concerns (Changan et al., 2017; Chaudhary et al., 2012; Kumar et al., 2014). Also, with an increasing population, it is more challenging to meet the food supply (Langyan et al., 2021b). Hence, the cultivation and utilization of pseudo-cereal crops with a high nutrition profile are of great significance.
Pseudo-cereals, such as buckwheat, amaranth, and quinoa, are considered as the richest sources of high-quality protein, carbohydrates, lipids, vitamins, minerals, and fibers, and also show the presence of bioactive compounds such as phenolic acids, flavonoids, saponins, etc. Pseudo-cereals have beneficial health-promoting effects against cardiovascular diseases, cancer, diabetes, and high blood pressure, and have been used to develop novel functional food products (Priego-Poyato et al., 2021; Thakur & Kumar, 2019; Kalinova & Dadakova, 2009). Additionally, pseudo-cereals have no gluten content and hence widely used in gluten-free formulations. Parameters such as protein efficiency ratio (PER) or net protein use (NPU), bioavailability or digestibility, and index of protein’s nutritional quality, are much higher in pseudo-cereals as compared to cereals (Quan et al., 2018; Upasana & Yadav, 2022).
In today’s era, the globalization of agriculture and its industrialization led to many adverse effects, including high demand for energy, utilization of water, increasing greenhouse gasses (GHG), and climatic disturbances. Moreover, the agricultural land is limited and is under continuous pressure due to different abiotic stresses like temperature, drought, heavy metal, and salinity stress. Additionally, the changes in the climatic conditions also affect food security. However, pseudo-cereals are mainly climate-resilient crops that could be grown in marginal lands not suitable for crops cultivation including low input benefits (Rodríguez et al., 2020).
Although pseudo-cereals are rich in the high level of protein, minerals, amino acids, and non-nutritive components, yet their consumption and commercialization as food products are limited due to gaps in nutritional composition research and limited technologies for their processing and utilization. Also, the supply chain of pseudo-cereals is not well developed, which further limits the availability of these essential foods to the larger population. Hence, the present review comprehensively discusses the current status of these pseudo-cereals in terms of their nutrition and non-nutrition contents, biological functions, food products, processing technologies, applications, and also elucidated the gaps and challenges and provide recommendations for future work.
Pseudocereals: An Overview
Pseudo-cereal is one of any non-grasses plants that are usually used to make bread and other bakery products. The most common pseudo-cereal species known today are Chenopodium quinoa sub sp. Quinoa (quinoa); Fagopyrum esculentum (buckwheat); and Amaranth sps. (amaranth). The seeds of these species mostly resemble true cereals in terms of composition and function, and therefore, they are known as pseudo-cereals (Alvarez-Jubete et al., 2010a).
The South American Andean region (2000–4000 m asl) is known to be the native for quinoa species (Chenopodium quinoa Willd.). The weedy species of quinoa, known as Chenopodium album, is commonly referred to as pigweed in English and “Bathua” in Hindi. This species of quinoa is resistant to frost and grown in regions having less rainfall (300–400 mm). It has smaller seeds with diameter in the range of 1 to 2.5 mm and reaches up to 1 to 3 m high, while the roots dig up to 30 cm into the soil. The stem with a diameter of 3.5 cm is cylindrical in shape, having a branched stem with a variety of colors (white or yellow). The grain is covered with pericarp containing saponins and made up of two layers. Before consuming it as food, the saponins (bitter material) should be removed. Due to excellent adaptability, the quinoa production can be seen in various geographical regions around the world. It has been cultivated in Asia, mainly the Himalayas and Northern India plains produce higher yield. Additionally, the grains of quinoa produced in Japan contains large number of bioactive components as compared to other cereals as well as pseudo-cereals (Dabija et al., 2022; Mir et al., 2018; Pritham et al., 2021). Due to high nutritional composition, quinoa is known as “food for astronauts” (Yasui et al., 2016).
The buckwheat (Fagopyrum esculentum Monch) is originated in China, and then it was moved to Eastern and Central Europe by nomads. During thirteenth century, the buckwheat production has increased in Italy, Austria, and Germany, but thereafter its cultivation was lost due to the introduction of other cereal crops. Nowadays, the cultivation and utilization of buckwheat are continuously increasing due to increasing demand for the gluten-free diet, and hence the global production goes higher. The highest production of buckwheat (1.19 million tonnes) was recorded in Russia, followed by China and Ukraine (FAOSTAT, 2018). In European countries, the buckwheat is cultivated largely in Poland (72,096 MT), followed by France (124,217 MT), and there is very less production in Lithuania, Slovenia, Hungary, and Latvia. Japan is known to be the largest consumer of buckwheat, and it is consumed as the second food crop after rice. The buckwheat can be cultivated in different types of soil, and is considered as the highly nutritious crop containing high protein content (Upasana & Yadav, 2022).
Globally, there is a large biodiversity of amaranth, and among various varieties, Amaranth caudatus, Amaranth cruentus, and Amaranth hypochodriacus are majorly cultivated and grown for their seeds (Kaur et al., 2010). Generally, seeds of amaranth are convex in shape, having weight of 1.3 mg and diameter in a range of 1–1.5 mm. The amaranth crop is heat, drought, and pests resistant and also tolerates poor soils and arid conditions (Mir et al., 2018).
Pseudocereals: Nutritional and Anti-Nutritional Components
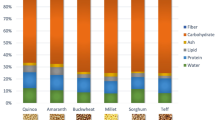
Pseudocereals have recently, attracted the attention of various researchers in food science and technologies as well as nutritionist due to higher nutritional composition. The nutritional composition of the pseudo-cereals (amaranth, quinoa, and buckwheat) is given in the Table 1. The nutritive value of these pseudo cereals is even higher to that of the true cereals. Therefore, a nutritional composition comparison of pseudo cereals with trues cereals (wheat, rice and maize) is presented in the Table 2.
Proteins
With the presence of essential amino acids, one can determine the protein’s nutritional quality. A lack of single amino acid results in diet leads to poor development, growth, and metabolic syndrome in humans and livestock. It has been reported that the protein content in pseudo-cereals is more than in cereals (Pirzadah & Malik, 2020). Interestingly, the lysine that is poorly distributed in cereal proteins is found higher in pseudo-cereals. Amaranth and quinoa also contain a high amount of histidine and arginine, which are good for children's and infants’ nutrition. Recently, it has been observed that proteins present in quinoa can provide many essential amino acids such as histidine (210%), lysine (132.22%), isoleucine (107.33%), methionine + cysteine (157.72%), threonine (177.82%), phenylalanine + tyrosine (247.63%), valine (107.43%), and tryptophan (15.83%) for adult nutrition (Mir et al., 2018). The sulfur-containing amino acids like methionine and cysteine are also present in higher concentrations in pseudo-cereals compared to commonly used cereals like rice, sorghum, and wheat. It has been reported that the leaf of quinoa is considered the best source of protein that can be used in fodder and in pharmaceutical industries (Martínez-Villaluenga et al., 2020). The processing techniques like fermentation and popping showed positive effect on amaranth grains. It has been reported that popping enhances the total lysine content of amaranth grains, which is higher than commonly used cereals. However, there is a limitation of using fermentation and popping methods as they are linked with reducing exogenous factors (e.g., trypsin, phytate, and tannin inhibitors), thereby decreasing the digestibility of proteins. Pseudo-cereals also contain storage proteins of different properties and structures, such as albumins (2S) and globulins (11S and 13S).
Carbohydrates
In carbohydrates, starch, a major energy source, is a biopolymeric component in plant parts (seeds, tubers, and grains) that is generally found in the seed’s perisperm in simple or spherical shapes (Garg et al., 2020). Quinoa contains carbohydrates ranging from 67–74% of its dry weight; however, the content of amylose is lower (11%) than cereals. Starch granules of quinoa are smaller in diameter than maize and wheat, and exhibited higher temperature of gelatinization (e.g., 57–64 °C). Other carbohydrates are usually present in very low amounts. These includes, monosaccharides (2%), disaccharides (2.3%), pentosans (2.9–3.6%), and crude fiber (2.5–3.9%). It has been reported that starch extracted from quinoa is helpful in those applications where breakability reduction and binding improvement are needed (Jan et al., 2016). Compared to amaranth and quinoa, buckwheat contains a large amount of starch and provides higher energy. For example, 100 g of grains of buckwheat gives the energy of 343 cal.
Dietary Fiber
The concept of dietary fibers is less known in pseudo-cereals. Quinoa and amaranth are considered as health-promoting pseudo-cereals due to high nutritional content. In one of the studies, the overall composition of seeds of amaranth contains dietary fiber ranging from 8 to 16%, and out of this, approximately 33–44% were considered as soluble fibers. Compared to quinoa and amaranth, dietary fiber content is higher in the seeds of buckwheat (Alvarez-Jubete et al., 2010b). The composition of monosaccharides in insoluble fibers of amaranth was recorded as 57% glucose, 9% xylose, 22% arabinose, 6% galactose, 4% rhamnose, 2% mannose, and 1% fucose. The insoluble dietary fibers of amaranth and quinoa are composed of homogalacturonans dispersed with RG-I stretch. The amaranth contains a higher fraction of soluble dietary fiber content than quinoa. Soluble fibers of quinoa are mainly consisted of arabinans and homogalacturonans. In contrast, soluble fiber present in amaranth consisted of branched xyloglucans, having side chains of disaccharide and trisaccharide, and has different fermentative and physiological properties (Mir et al., 2018).
Minerals and Vitamins
In general, minerals such as iron, magnesium, and calcium are present in less amounts in gluten-free foods and products. Pseudo-cereals such as quinoa, amaranth, and buckwheat are excellent sources of magnesium; calcium, iron, and other vital minerals (Alvarez-Jubete et al., 2010a). The highest amount of minerals is present in amaranth, followed by quinoa and buckwheat. Vitamins played a major role in almost every physiological function. The content of thiamine is more in amaranth as compared to wheat. Amarnath and quinoa are excellent pseudo-cereals for vitamin C, riboflavin, vitamin E, and folic acid contents, whereas Tartary buckwheat seeds contain higher content of vitamins B2 and B6 (Patil & Jena, 2020).
Fatty Acids
Pseudo-cereals have the highest number of fatty acids compared to other cereal crops, mainly characterized by a large amount of unsaturated fatty acids (linolenic acid). The highest amount of linoleic acid was found in amaranth and quinoa, whereas the oleic acid level and eicosenoic acid were found highest in buckwheat (Dziadek et al., 2016). The level of eicosnoic acid and erucic acid was also reported higher in quinoa. Among saturated fatty acids, palmitic acid was reported higher in amaranth, followed by buckwheat and quinoa (Bock et al., 2021). Polyunsaturated fatty acids improve insulin sensitivity and also cure cardiovascular diseases, osteoporosis, cancer, autoimmune, and inflammatory diseases. A high quantity of squalene was found in amaranth, which is widely used in cosmetic and pharmaceutical applications. The content of lipid present in quinoa and amaranth is 2–3 times higher than wheat and buckwheat (Mir et al., 2018).
Anti-Nutrient Components
Various anti-nutrient components are present in pseudo-cereals. These commonly include polyphenols, which are reported higher in pseudo-cereals than other cereals. Phenolic acid was found higher in buckwheat, followed by quinoa and amaranth. Among the phenolic acids, gallic acid, dihydroxybenzoic acid, vanillic acid, caffeic acid, o-coumaric acid, and rutin were commonly reported in pseudo-cereals (Dziadek et al., 2016). Anthocyanins and flavonoids are other polyphenolic compounds present in pseudo-cereals. The higher content of anthocyanin and flavonoids is present in buckwheat, followed by quinoa, and amaranth (Mir et al., 2018). Pseudo-cereals also contain saponins, phytosterols, phytoecdysteroids, polysaccharides, tannins, oxalates, and phytates (Fig. 1) (Hernández-Ledesma, 2019).
Bioactive Properties of Pseudocereals
Reports have been published on various bioactivities of pseudocereals, which majorly include antitumor, antioxidant, hypoglycaemic, ACE inhibitory, antimicrobial, and hypolipidemic effects (Table 2, Fig. 2). It has been observed that in countries where a high number of cereal crops are consumed, the risk of several diseases related to the metabolic functions has been greatly reduced. Lectins present in pseudo-cereals activated the innate defense mechanism, preventing cancer and obesity. On the other hand, protease inhibitors also have potent ACE inhibitors and anti-inflammatory properties and were closely linked with anti-hypertensive effects (Langyan et al., 2022a). Dietary saponins form insoluble cholesterol complexes, thereby lowering plasma cholesterol levels and decreasing the occurrence of cardiovascular diseases. Also, phenolic compounds like flavonoids, tannins, and phenolic acids, having a significant role in the pigmentation of seeds showed higher antioxidant activity (Giusti et al., 2017). The bioactivities of small peptides present in pseudo-cereals are mainly released from enzymatic hydrolysis by various proteases such as pepsin, trypsin, chymotrypsin, alcalase, papain, pancreatin, thermolysin, and flavourzyme (Awika & Duodu, 2017). These peptides have various bioactivities such as antioxidant, antifungal, antitumoral, and ACE inhibition activity, and are also used for different purposes, like food supplements, functional food ingredients, and nutraceuticals (Table 3) (Awika & Duodu, 2017; Das et al., 2020).
Pseudo-cereals, mainly quinoa, buckwheat, and amaranth, showed antidiabetic, anti-hypertensive, antioxidant, anti-inflammatory, immunoregulatory, anticancer, neuroprotective, and anti-microbial activity (Table 3). These bioactivities are mainly exerted by the protein hydrolysate or the isolated peptides by various mechanisms. For instance, the antidiabetic activity of pseudo-cereals is mainly due to inhibition of dipeptidyl peptidase IV (DPP-IV) and α-amylase and α-glucosidase activity, anti-hypertensive due to angiotensin-I-converting enzyme inhibition, and hypocholesterolemic effect due to inhibition of pancreatic lipase and HMG-CoA reductase activity (Del Hierro et al., 2021; Nongonierma et al., 2015; Sánchez-López et al., 2021; Soares et al., 2015; Vilcacundo et al., 2017; Zieliński et al., 2020).
Pseudo-cereals have also been tested clinically against different disease conditions and/or to fulfill the nutrients requirements. Quinoa and quinoa-based products (e.g., biscuits, crackers, brioche, sponge cake, baguette bread, sliced bread, and pasta) have been tested and found effective in preventing type-2 diabetes mellitus in the diabetic patient with age > 65 years (NCT04529317 dated Sept’ 2016). Also, buckwheat was found effective in lowering the blood glucose level in diabetic adults (NCT00841503). It has also been reported that quinoa effectively reduced weight and other complications (e.g., glycemic index) in overweight persons (NCT02621502). Biscuits made from quinoa were found effective in preventing cardiovascular risk markers in older patients (NCT03291548). The probiotic potential of quinoa milk was investigated in adults and found to change the composition of oral and intestinal flora. Due to its high level of amino acids and free from lactose, gluten, and cholesterol, the fermented quinoa milk provides a more effective probiotic effect (NCT04280731).
Irritable bowel syndrome (IBS) is one of the most common challenges in digestion and after that avoidance of nutritious food products. In a clinical study, the Sourdough wheat bread, regular yeast baked toast bread, and gluten-free diet containing quinoa were tested in adults. It was found that the gluten-free diet containing quinoa prevents IBS and provides a nutritious diet as compared to others (NCT02572908). Chenopodium formosanum and buckwheat extract drink were found effective in preventing the aging effect (NCT04237818). Buckwheat honey was prepared and tested for its preventive effect against cough with acute upper respiratory tract infection in adults. It was found that the buckwheat honey significantly reduced the frequency and severity of cough compared to placebo (NCT01062256). Food prepared with olive, buckwheat, peas, and chestnut flour was found effective in modifying the gut microbiota and the cholesterol metabolism in obese and hypercholesteremic patients (NCT02664428). Amaranth flour improved diet quality and iron intake in children (12–59 months), thus preventing anemia (NCT01224535).
Processing Techniques of Pseudocereals
For improving the nutritional quality of pseudo-cereals and inactivating/eliminating the compounds that interfere with the digestibility of protein, various processing techniques such as cooking, soaking, dehulling, microwave, irradiation, extrusion, and fermentation have been used. During food processing, heat treatment has been widely used for different purposes like sterilization, enhancing texture and flavor, destroying toxic microorganisms, and improving functional and physical properties. The effect of processing techniques on the nutrients (carbohydrates, protein, oil, dietary fiber, etc.) and anti-nutrients (phytates, oxalates, saponins, etc.) in the pseudo cereals is presented in Table 4. These processing methods are useful in improving the protein quality and the digestibility of proteins. However, some adverse effects like protein degradation have also been reported due to thermal processing, affecting the bioavailability of essential amino acids. Several studies postulate the effects of food processing on the digestibility and nutritional properties of proteins from pseudo-cereals. These commonly include conventional grain processing (milling, roasting, drying, cooking, baking) and bioprocessing (enzyme-assisted processing, fermentation, biorefinery). Some of them are used during the processing of pseudo-cereals and are discussed below.
Cooking
Cooking, being one of the most important and common process, influences nutrients bioavailability, nutritional value, and digestibility (Kalpanadevi & Mohan, 2013). With the presence of heat-labile compounds in uncooked proteins, the digestibility of protein is low. Hence, cooking has several effects on the digestibility of proteins like denaturation of proteins or reducing the protein resistance from the enzymatic activity and interacting proteins with non-protein components, which further affect the digestibility. The cooking method also destroys the protease inhibitors, and unfavorable compounds are leaching out and improve digestibility. It also eliminates trypsin inhibitors and reduces the content of phytic acid and tannins ((Fawale et al., 2017). In extrusion cooking, the high temperature and pressure were applied, resulting in high shear forces and very short cooking time. The high heat and pressure cause protein denaturation, while the nutrients are retained due to very low exposure time. A high shear force is required for pseudo-cereals compared to rice and wheat processing due to high lipid and lower amylose content. In amaranth and quinoa, the fat content is higher, and thus it needs to be blended with flour having low-fat content, such as rice or maize. Also, these pseudo-cereals need an additional defatting step before extrusion cooking. In buckwheat, the fat content is lower and could be directly used for extrusion cooking. Maize blended with amaranth or quinoa undergoes extrusion cooking to produce expanded extrudates, with lower tocopherol and fatty acid content, while the phenolic compounds and folate are partially affected (Ramos Diaz et al., 2017). The extrusion cooking also affected the physical and biochemical properties of the pseudo-cereals. For instance, the quinoa under extrusion cooking increased the protein crosslinking and the soluble fiber content, while some essential amino acids such as valine and methionine were slightly reduced (Kuktaite et al., 2021). Due to the presence of aggregated starch compounds in quinoa, the starch is not completely gelatinized after extrusion cooking, which can be overcome by adding sufficient water during the extrusion cooking following drying of the extrudates (Kuktaite et al., 2021). After cooking, the prepared extrudates were milled and showed improved water solubility, protein solubility, oil binding, and foaming properties (Espinosa-Ramírez et al., 2021).
Steaming and boiling are the two most common cooking processes used for most foods, including pseudo-cereals. In a study, both these cooking methods were analyzed for their effect on the mineral content of quinoa, amaranth, buckwheat, and rice. It was found that during steaming quinoa, the manganese, phosphorus, and iron retain 100%. On the other hand, in all the pseudo-cereals, the mineral content in steaming and boiling processes showed no significant differences (Mota et al., 2016). The effect of roasting and boiling on amaranth and quinoa seeds was profound as it increases the bioavailability of minerals, dietary fibers, and phenolics (Repo-Carrasco-Valencia et al., 2010). Also, it was found that roasting improved the gruel viscosity of amaranth as compared to raw and popped grain (Muyonga et al., 2014).
Microwave and Irradiation
Microwave energy utilizes electromagnetic waves having a frequency between 300 MHz and 300 GHz. It is non-ionizing radiation that continuously generates heat because of molecular motion in the product (Divekar et al., 2017). It generally improves functional properties like foaming, emulsifying, and water and oil holding capacity. This method inactivates protease inhibitors, reduces bioactive compounds concentration, and enhances protein quality (Vagadia et al., 2018). Irradiation is a safe method where food is exposed in ionizable radiations with a specific time interval and environment under controllable conditions. It can help avoid various diseases caused by microbes and remove unfavorable compounds, hence improving the protein quality. However, it has a negative impact on protein digestibility as it degrades specific amino acids (like sulfur and aromatic amino acids).
Germination
Germination is the absorption of water by dormant/dry seeds, leading to embryonic axis elongation. During germination, mostly hydrolytic enzymes result in biochemical changes, modify structural properties, and enhance the nutritional value, thereby reduce antinutritional factors by leaching or enzymatic activity. With the improvement of the digestibility of proteins after the germination process, a reduction in phytic acid and polyphenols content in germinated seedlings were recorded along with an increase in the protein solubility (Albarracín et al., 2015).
Water soaking before germination played a major role in the development and growth and the nutritional characteristics of the germinated plant. As such, the slightly acidic electrolyzed water was tested in buckwheat and showed an increase in the GABA, rutin, glutamic acid decarboxylase (GAD), and phenylalanine ammonialyase (PAL) activity (Hao et al., 2016).
Drying
Several drying methods are used for the removal of water molecules present in the food. These methods include hot air-, sun-, freeze-, spray-, and vacuum-drying. Among them, spray-drying is widely used in the food industry to enhance the shelf-life of food. Spray drying mainly uses high temperatures, decrease heat-labile substances within the proteins and affecting the digestibility and functional properties of the protein. Hot air spraying is mostly used to dry liquid foods through moisture vaporization and hence leaves the particulate matter in the form of powder. The freeze-drying process undergoes sublimation, which helps increase aggregations and protein–protein interactions. Also, improve the digestibility of proteins and enhance various functional properties like solubility of the protein, water-holding capacity, foaming, and emulsifying properties (Tontul et al., 2018).
Fermentation
It is the process that utilizes several microorganisms such as yeasts and bacteria that help to improve the nutritional quality of crops. It also enhances the texture, aroma, and flavor of bakery foods. It increases protein availability, functionality, and eliminate undesirable compounds. Fermentation also played a major role in improving the bioavailability and digestibility of nutrients (Fawale et al., 2017). Pseudo-cereals are mainly used for bread making; however, due to lower palatability, it have been fermented with P. pentosaceus, E. faecalis, W. cibaria, L. plantarum, L. salivarius, L. rhamnosus, and L. paracasei (Capuani et al., 2013; Rocchetti et al., 2019a, b; Zieliński et al., 2017, 2019). The lactic acid bacteria fermentation improves the palatability and aroma of pseudo-cereal products. With the help of L. paracasei and L. plantarum, Tartar buckwheat flour was fermented and showed antidiabetic activity (Feng et al., 2018). Also, it has been reported that with the help of lactic acid bacteria, the quinoa pasta product was made, which further improved the vitamin and mineral content in a mouse model (Carrizo et al., 2020). Also, lactic acid bacteria fermentation improves the antioxidant and phenolic content of pseudo-cereals (Bustos et al., 2017). A bread prepared using amaranth (Amaranthus caudatus, grown in India), quinoa (Chenopodium quinoa, grown in Canada), and buckwheat grains (Fagopyrum esculentum Moench, grown in China) and yeast was analyzed for vitamin E content. It was found that the bread prepared using quinoa has the highest content of vitamin E (alpha-tocopherol), whereas amaranth bread contained the highest beta and delta-tocopherol content. Gamma-tocopherol content was highest in both buckwheat and quinoa. It was also recorded that the vitamin E content in the pseudo-cereals made bread was higher than the bread made from wheat (Alvarez-Jubete et al., 2009). Also, the loss in the vitamin E content during high-temperature baking was reported, and pseudo-cereals bread showed lower loss than wheat bread. Among them, the lowest loss in vitamin E content was recorded in quinoa bread.
The bread made from gluten-free pseudo-cereals uses processing techniques such as sourdough technology, high hydrostatic pressure, milling, and non-conventional baking methods (Bender & Schönlechner, 2020; Sciarini et al., 2020). It has been found that sourdough improved the quality of buckwheat and amaranth bread (Bender et al., 2018; Houben et al., 2010). In recent years, ohmic heating has been used in the baking process and has provided significant functionality and digestibility compared to the conventional process Bender et al., 2019. The milling process, including hammer mill followed by cyclonic quinoa and buckwheat, resulted in the production of finest particle size that ultimately increased the quality of the bread (Sciarini et al., 2020).
It has also been reported that sourdough fermentation is suitable for decreasing saponins’ level and improving the rheology and sensory attributes (Bolívar-Monsalve et al., 2018). During fermentation, the amino acid, soluble fibers, phenolics, and antioxidant activity were markedly increased in quinoa sourdough, which later improved the bread quality (Rizzello et al., 2016). Fermentation of pseudo-cereals with Lb. plantarum improves the free amino acid content, gamma-aminobutyric acid (GABA), and phenolic compound while decreasing starch hydrolysis (Coda et al., 2010). The sour dough fermented quinoa flour increases the protein, amino acids, dietary fiber content, antioxidant activity, and tenacity and elasticity of pasta, thereby improved its functional and mechanical quality (Lorusso et al., 2017). The sourdough fermentation of amaranth flour using lactic acid bacteria improve the rheology (viscosity and elasticity) (Houben et al., 2010). The buckwheat sourdough increased the amino acid, magnesium, dietary fibers, phenolic while decreasing the phytic acid and tannins content, thereby extending the shelf-life and increasing the overall quality of the bread (Alvarez-Jubete et al., 2010b; Moroni et al., 2012). The muffin prepared by fermented buckwheat flour showed higher content of macro and micronutrients (WronkoWska, 2016).
In a study, Lactobacillus plantarum 299v® was used for the fermentation of quinoa, canihua, and amaranth grains and flours. It was found that the fermentation process increases the phytate degradation and that too higher in the flours than grains. The addition of Lactobacillus plantarum 299v® increased the lactic acid production during fermentation, and increased the pH and endogenous phytase activity. Also, the bioavailability of minerals was increased during fermentation (Castro-Alba et al., 2019).
Applications of Pseudocereals
The advancement on nutritional value and processing methods, various potential applications of pseudocereals in dietary food have been recognized. Pseudocereals are highly nutritious due to its high nutraceutical potential, and are used in gluten-free product development. Moreover, the proteins present in pseudo-cereals are the richest source of essential amino acids useful in the pharmaceutical and food products. Several studies have showed the potential applications of pseudo-cereals in the developing gluten-free and nutrient-rich products, like bread, confectionary items, pasta, biscuits, etc. Gluten-free bread also contains a higher amount of polyphenol compounds, and hence they impart an antioxidative effect. Whole amaranth flour has also been utilized for the production of gluten-free biscuits having higher protein content. The grains of amaranth and quinoa have been widely utilized in making soups, beverages, porridges, sauces, soufflés, and sweets. The leaf of quinoa is considered the best source of protein that can be used in fodder and food and pharmaceutical industries. Also, the bioactive peptides present in pseudo-cereals are used for different purposes like food supplements, functional food ingredients, and nutraceuticals.
Although many products free from gluten are accessible nowadays from the market; however, a greater number of those products have poor quality in terms of nutrients (Mir et al., 2018). In many studies, it has been reported that the levels of vitamin B, proteins, and fibers are low in gluten-free bakery products compared with gluten-containing bakery products (Thakur et al., 2021a). Thus, there is a need to improvise the development of bakery products free from gluten, matching the nutritional composition with gluten-containing products. Niland and Cash (2018) observed and reported that a diet free from gluten helps in the restoration of health and improves quality of people suffering from celiac diseases. As compared to the normal healthy person, the person with celiac disease, herpetiformis, wheat allergies, dermatitis, gluten sensitivity, and gluten ataxia has a lesser fiber intake from the food, and hence should strictly take a gluten-free diet (Thakur et al., 2021b). People suffering from celiac disease have difficulties in searching for gluten-free products in the market due to high prices of food products, poor sensory features, and shortage and unavailability of a variety of foods, which leads to strictly adhere to a gluten-free diet (Alvarez-Jubete et al., 2010b).
Studies have been conducted to improve the quality of gluten-free products using enzymes, emulsifiers, or hydrocolloids. The flour made from pseudo-cereals (quinoa, amaranth, and buckwheat) was used to produce gluten-free bread compared with those produced from potato and rice starch (Alvarez-Jubete et al., 2010b). Pseudo-cereals enhanced the antioxidant activity and increased fiber, protein, iron, calcium, polyphenol, and vitamin E contents of gluten-free bread. Also, nutrient-dense materials derived from pseudo-cereals have increased nutritional composition and also help in bread preparation with good sensory and physical properties (Capriles & Arêas, 2014). Along with some amount of buckwheat flour to rice-based and corn starch-based flour for the preparation of gluten-free products, the antioxidant capacity and nutritional value were increased (Alvarez-Jubete et al., 2010b). Cheese bread, along with the addition of 10% amaranth flour enhances the levels of dietary fiber, proteins, and iron contents of bread. One of the studies reported that the bread produced from the flour of tartary buckwheat was linked with the reduction in rutin during processing, the concentration of quercetin remained stable, and the product showed overall strong antioxidant activity. The consumption of quinoa increased almost 3 times in the last 8 years (FAOSTAT, 2013). Quinoa and amaranth together are rich in copper, iron, zinc, and manganese, and also reported that the content of magnesium and phosphorus may contribute almost 55% of the daily intake of nutrients from food (do Nascimento et al., 2014).
Challenges and Future Outlook
Instead of the various benefits of pseudo-cereals, yet several challenges related to its cultivation, anti-nutrients and inclusion of these crops into modern food system, includes agronomic factors (yield and growth), social (lack of awareness and low esteem), economic (marketing constraints) as well as technological (seeds processing and genetic factors) challenges are existing (Priego-Poyato et al., 2021). One of the reasons that decline the cultivation of crops is poorly characterized agronomic analysis. Besides agronomic challenges, there is a limitation at the genetic level like self-incompatibility that limits trait improvement and breeding. Similarly, flower abortion and seed shattering are other drawbacks in tartary and common buckwheat that limits their production yield. Other factors like limitation in traditional knowledge reduce cultivation and production of pseudo-cereals (Pirzadah & Malik, 2020).
There are many health concerns linked with the intake of dietary proteins derived from pseudo-cereals. Antinutrients present in plant food are naturally produced by plants and further interfere with absorption, digestion, and utilization of nutrients present in the food (Popova & Mihaylova, 2019). Major anti-nutritional components in quinoa are saponins and, mainly responsible for lowering down the mineral absorption. Another class is phytic acid, forming insoluble chelate with magnesium, iron, zinc, or calcium (Pirzadah & Malik, 2020). Furthermore, oxalates are another group of compounds posing the challenge to accept the pseudo cereals as the food as these can cause irritation in the gastrointestinal tract after ingestion. These include maldigestion of proteins (protease and trypsin inhibitors), and carbohydrates (alpha-amylase inhibitors), malabsorption of minerals (oxalates phytates and tannins), autoimmunity and leaky gut (e.g., some saponins and lectins), inflammation, behavioral effects, and gut dysfunction (when converting cereal gliadins to exorphins), and also exert interference with thyroid iodine uptake (goitrogens) (Popova & Mihaylova, 2019). These adverse effects of antinutrients are generally seen in animals when consumed unprocessed proteins from cereal crops. However, they also exerted beneficial health effects. For instance, at a lower level of lectins, phytates, enzyme inhibitors, saponins, and phenolic compounds, there was a reduction in plasma cholesterol, blood glucose, and triglycerides levels. In addition, saponins have a major role in the functioning of the liver and decrease agglutination of platelets. While some saponins, and also protease inhibitors, phytates, phytoestrogens, and lignans, might help in reducing cancer risk (Popova & Mihaylova, 2019).
Additionally, tannins also have antimicrobial effects. To reduce the concentration of antinutrients in pseudo-cereals and their adverse effects, various processing techniques such as fermentation, soaking, gamma irradiation, sprouting (germination), and heating have been adopted (Popova & Mihaylova, 2019). These processing techniques also remove most of the antinutrients like phytates, glucosinolates, erucic acid, and insoluble fiber from canola proteins that further improve and increase the digestibility and bioavailability (Fleddermann et al., 2013).
Technological interventions are required for generating desirable trait including climate-resilient crops with high nutritional composition, through available genetic resources. Studies on agro-morphological (seed shattering, abiotic, biotic stress), nutritional (trait-specific germplasm), yield related traits, post-harvest technologies are required for further utilization of the information in the crop improvement program. Furthermore, these programs should be easily accessible to the marginal farmers as well as the breeders through a common platform. (Rodríguez et al., 2020). Extension activities and awareness programs for dissemination of knowledge on the nutritional superiority of these pseudo cereals should be conducted and popularized. Market potential of the value-added products of these crops should be explored and documented in order to make pseudo cereals more profitable when compared to other cereal crops. Wider choice through varieties in terms of nutrition, low-input, climate-resilient should be offered through access to the seed. Next, sustainability of these activities can be ensured through community seed bank, capacity building, increasing income and livelihood, social acceptability and synergy starting from production till marketing. These crops should get the place in the national food basket through national policies in order to improve the nutritional surveillance. There is a strong need for the international funding so as to explore the hidden potential of these pseudo-cereals to mitigate the hidden hunger.
Conclusions
The research on pseudo-cereals, especially quinoa, buckwheat, and amaranth has been continuously increasing as gluten-free, nutritious, and functional food products. Along with the gluten-free features, pseudo-cereals also contain high-quality proteins, dietary fibers, minerals like calcium and iron, and phenolic compounds. They also showed bio functional activities as antitumor, antioxidant, hypoglycaemic, anti-hypertensive, antimicrobial, and hypolipidemic. Also, clinical trials revealed their effective uses as health and dietary food supplements. In addition, various processing methods such as germination, cooking, soaking, fermentation, popping, etc., helps in enhancing the nutritional value of grains and make processed gluten-free products like bread, pasta, and confectionary food items. However, in the market, there is still limited availability of gluten-free products. Therefore, more research is needed to exploit and search the functionalities of pseudo-cereals and their food products formulations.
Data Availability
The datasets used in the present study are available from the corresponding author on reasonable request.
References
Albarracín, M., De Greef, D. M., González, R. J., & Drago, S. R. (2015). Germination and extrusion as combined processes for reducing phytates and increasing phenolics content and antioxidant capacity of Oryza sativa L. whole grain flours. International Journal of Food Sciences and Nutrition, 66, 904–911.
Al-Mamun, M. A., Husna, J., Khatun, M., Hasan, R., Kamruzzaman, M., Hoque, K. M. F., et al. (2016). Assessment of antioxidant, anticancer and antimicrobial activity of two vegetable species of Amaranthus in Bangladesh. BMC Complementary Alternative Medicine, 16, 15.
Alonso-Miravalles, L., & O’Mahony, J. A. (2018). Composition, protein profile and rheological properties of pseudocereal-based protein-rich ingredients. Foods, 7(5), 73.
Al-Qabba, M. M., El-Mowafy, M. A., Althwab, S. A., Alfheeaid, H. A., Aljutaily, T., & Barakat, H. (2020). Phenolic profile, antioxidant activity, and ameliorating efficacy of chenopodium quinoa sprouts against CCl4-induced oxidative stress in rats. Nutrients, 12, 2904. https://doi.org/10.3390/nu12102904
Alvarez-Jubete, L., Holse, M., Hansen, Å., Arendt, E. K., & Gallagher, E. (2009). Impact of baking on vitamin E content of pseudocereals amaranth, quinoa, and buckwheat. Cereal Chemistry, 86, 511–515.
Alvarez-Jubete, L., Arendt, E. K., & Gallagher, E. (2010a). Nutritive value of pseudocereals and their increasing use as functional gluten-free ingredients. Trends in Food Science & Technology, 21, 106–113.
Alvarez-Jubete, L., Wijngaard, H., Arendt, E. K., & Gallagher, E. (2010b). Polyphenol composition and in vitro antioxidant activity of amaranth, quinoa buckwheat and wheat as affected by sprouting and baking. Food Chemistry, 119, 770–778. https://doi.org/10.1016/j.foodchem.2009.07.032
Awika, J. M., & Duodu, K. G. (2017). Bioactive polyphenols and peptides in cowpea (Vigna unguiculata) and their health promoting properties: a review. Journal of Functional Foods, 38, 686–697.
Ayala-Nino, A., Rodríguez-Serrano, G. M., Jiménez-Alvarado, R., Bautista-Avila, M., Sánchez-Franco, J. A., González-Olivares, L. G., & Cepeda-Saez, A. (2019). Bioactivity of peptides released during lactic fermentation of amaranth proteins with potential cardiovascular protective effect: an in vitro study. Journal of Medicinal Food, 22, 976–981. https://doi.org/10.1089/jmf.2019.0039
Bai, C. Z., Ji, H. J., Feng, M. L., Hao, X. L., Zhong, Q. M., Cui, X. D., & Wang, Z. H. (2015). Stimulation of dendritic cell maturation and induction of apoptosis in lymphoma cells by a stable lectin from buckwheat seeds. Genetics and Molecular Research, 14, 2162–2175. https://doi.org/10.4238/2015
Bao, T., Wang, Y., Li, Y. T., Gowd, V., Niu, X. H., Yang, H. Y., Chen, L. S., & Chen, W. (2016). Antioxidant and antidiabetic properties of tartary buckwheat rice flavonoids after in vitro digestion. Journal of Zhejiang University. Science. B, 17, 941–951. https://doi.org/10.1631/jzus.B1600243
Bender, D., Fraberger, V., Szepasvári, P., D’Amico, S., Tömösközi, S., Cavazzi, G., Jäger, H., Domig, K. J., & Schoenlechner, R. (2018). Effects of selected lactobacilli on the functional properties and stability of gluten-free sourdough bread. European Food ResEarch and TechnolOgy, 244, 1037–1046.
Bender, D., Gratz, M., Vogt, S., Fauster, T., Wicki, B., Pichler, S., Kinner, M., Jäger, H., & Schoenlechner, R. (2019). Ohmic heating – A novel approach for gluten-free bread baking. Food and Bioprocess TechnolOgy, 12, 1603–1613.
Bender, D., & Schönlechner, R. (2020). Innovative approaches towards improved gluten-free bread properties. Journal of Cereal Science, 91, 102904.
Bock, P. D., Daelemans, L., Selis, L., Raes, K., Vermeir, P., Eeckhout, M., & Van Bockstaele, F. (2021). Comparison of the chemical and technological characteristics of wholemeal flours obtained from Amaranth (Amaranthus sp.), Quinoa (Chenopodium quinoa) and Buckwheat (Fagopyrum sp.) seeds. Foods, 10, 651.
Bolívar-Monsalve, J., Ceballos-González, C., Ramírez-Toro, C., & Bolívar, G. A. (2018). Reduction in saponin content and production of gluten-free cream soup base using quinoa fermented with Lactobacillus plantarum. Journal of Food Processing, 42, e13495.
Borges, J. T. S., Bonomo, R. C., Paula, C. D., Oliveira, L. C., & Cesário, M. C. (2010). Physicochemical and nutritional characteristics and uses of Quinoa (Chenopodium quinoa Willd.). Temas Agrários, 15(1), 9–23.
Bustos, A. Y., Gerez, C. L., Mohtar, L. G., Paz Zanini, V. I., Nazareno, M. A., Taranto, M. P., & Iturriaga, L. B. (2017). Lactic acid fermentation improved textural behaviour, phenolic compounds and antioxidant activity of chia (Salvia hispanica L.) dough. Food Technology and Biotechnology, 55, 381–389.
Capraro, J., De Benedetti, S., Di Dio, M., Bona, E., Abate, A., Antonia, P. C., & Scarafoni, A. (2020). Characterization of chenopodin isoforms from quinoa seeds and assessment of their potential anti-inflammatory activity in caco-2 cells. Biomolecules, 10, 795. https://doi.org/10.3390/biom10050795. PMID: 32455586 PMCID: PMC7277664.
Capriles, V. D., & Arêas, J. A. G. (2014). Novel approaches in gluten-free breadmaking: interface between food science, nutrition, and health. Comprehensive Reviews in Food Science and Food Safety, 13, 871–890. https://doi.org/10.1111/1541-4337.12091
Capuani, A., Behr, J., & Vogel, R. F. (2013). Influence of lactic acid bacteria on redox status and on proteolytic activity of buckwheat (Fagopyrum esculentum Moench) sourdoughs. International Journal of Food Microbiology, 165, 148–155.
Carrizo, S. L., de LeBlanc, A. D. M., LeBlanc, J. G., & Rollán, G. C. (2020). Quinoa pasta fermented with lactic acid bacteria prevents nutritional deficiencies in mice. Food Research International, 127, 108735. https://doi.org/10.1016/j.foodres.2019.108735
Castro-Alba, V., Lazarte, C. E., Perez-Rea, D., Carlsson, N. G., Almgren, A., Bergenståhl, B., & Ranfeldt, Y. (2019). Fermentation of pseudocereals quinoa, canihua, and amaranth to improve mineral accessibility through degradation of phytate. Journal of the Science of Food and Agriculture, 99, 5239–5248. https://doi.org/10.1002/jsfa.9793
Changan, S., Chaudhary, D. P., Kumar, S., Kumar, B., Kaul, J., Guleria, S., Jat, S. L., Singode, A., Tufchi, M., Langyan, S., & Yadav, O. P. (2017). Biochemical characterization of elite maize (Zea mays) germplasm for carotenoids composition. Indian Journal of Agricultural Sciences, 87, 46–50.
Chaudhary, D. P., Mandhania, S., & Sapna, K. R. (2012). Inter-relationship among nutritional quality parameters of maize Zea mays L. genotypes. Indian Journal of Agricultural Sciences, 82, 681–686.
Chauhan, A., Saxena, D., & Singh, S. (2015). Total dietary fibre and antioxidant activity of gluten free cookies made from raw and germinated amaranth (Amaranthus spp.) flour. LWT - Food Science and Technology, 63(2), 939–945. https://doi.org/10.1016/j.lwt.2015.03.115
Choi, J. Y., Lee, J. M., Lee, D. G., Cho, S., Yoon, Y. H., Cho, E. J., & Lee, S. (2015). The n-butanol fraction and rutin from tartary buckwheat improve cognition and memory in an in vivo model of amyloid-β-induced Alzheimer’s disease. Journal of Medicinal Food, 18, 631–641. https://doi.org/10.1089/jmf.2014.3292
Coda, R., Rizzello, C. G., & Gobbetti, M. (2010). Use of sourdough fermentation and pseudocereals and leguminous flours for the making of a functional bread enriched of γ-aminobutyric acid (GABA). International Journal of Food Microbiology, 137(236–245), 68.
Coelho, L. M., Silva, P. M., Martins, J. T., Pinheiro, A. C., & Vicente, A. A. (2018). Emerging opportunities in exploring the nutritional/functional value of amaranth. Food & Function, 9(11), 5499–5512.
Cornejo, F., Novillo, G., Villacrés, E., & Rosell, C. M. (2019). Evaluation of the physicochemical and nutritional changes in two amaranth species (Amaranthus quitensis and Amaranthus caudatus) after germination. Food Research International, 121, 933–939. https://doi.org/10.1016/j.foodres.2019.01.022
d’Amour, C. B., Reitsma, F., Baiocchi, G., Barthel, S., Güneralp, B., Erb, K. H., Haberl, H., Creutzig, F., & Seto, K. C. (2017). Future urban land expansion and implications for global croplands. Proceedings of National Academy of Science, 114, 8939–8944.
Dabija, A., Ciocan, M. E., Chetrariu, A., & Codină, G. G. (2022). Buckwheat and Amaranth as Raw Materials for Brewing, a Review. Plants., 11(6), 756. https://doi.org/10.3390/plants11060756
Dakhili, S., Abdolalizadeh, L., Hosseini, S. M., Shojaee-Aliabadi, S., & Mirmoghtadaie, L. (2019). Quinoa protein: composition, structure and functional properties. Food Chemistry, 299, 125161.
Das, D., Jaiswal, M., Khan, F. N., Ahamad, S., & Kumar, S. (2020). PlantPepDB: a manually curated plant peptide database. Scientific Reports, 10, 1–8.
Del Hierro, J. N., Casado-Hidalgo, G., Reglero, G., & Martin, D. (2021). The hydrolysis of saponin-rich extracts from fenugreek and quinoa improves their pancreatic lipase inhibitory activity and hypocholesterolemic effect. Food Chemistry, 338, 128113. https://doi.org/10.1016/j.foodchem.2020.128113. PMID: 33092009.
Demir, B., & Bilgiçli, N. (2020). Changes in chemical and anti-nutritional properties of pasta enriched with raw and germinated quinoa (Chenopodium quinoa Willd.) flours. Journal of Food Science and Technology, 57(10), 3884–3892. https://doi.org/10.1007/s13197-020-04420-7. Epub 2020 Apr 11. PMID: 32904010; PMCID: PMC7447728.
Divekar, M. T., Karunakaran, C., Lahlali, R., Kumar, S., Chelladurai, V., Liu, X., & Jayas, D. S. (2017). Effect of microwave treatment on the cooking and macronutrient qualities of pulses, Internationa. Journal of Food Properties, 20, 409–422.
do Nascimento, A. B., Fiates, G. M. R., dos Anjos, A., & Teixeira, E. (2014). Gluten-free is not enough–perception and suggestions of celiac consumers. International Journal of Food Science and Nutrition, 65, 394–398. https://doi.org/10.3109/09637486.2013.879286
Dzah, C. S., Duan, Y., Zhang, H., Authur, D. A., & Ma, H. (2020). Ultrasound- subcritical water-and ultrasound assisted subcritical water-derived Tartary buckwheat polyphenols show superior antioxidant activity and cytotoxicity in human liver carcinoma cells. International Food Research Journal, 137, 109598. https://doi.org/10.1016/j.foodres.2020.109598
Dziadek, K., Kopeć, A., Pastucha, E., Piątkowska, E., Leszczyńska, T., Pisulewska, E., Witkowicz, R., & Francik, R. (2016). Basic chemical composition and bioactive compounds content in selected cultivars of buckwheat whole seeds, dehulled seeds and hulls. Journal of Cereal Science, 69, 1–8.
Espinosa-Ramírez, J., Rodríguez, A., De la Rosa-Millán, J., Heredia-Olea, E., Pérez-Carrillo, E., & Serna-Saldívar, S. O. (2021). Shear-induced enhancement of technofunctional properties of whole grain flours through extrusion. Food Hydrocolloids, 111, 106400. https://doi.org/10.1016/j.foodhyd.2020.106400
FAOSTAT. (2013). Retrieved May 14, 2022, from FAOSTAT gateway. http://faostat3.fao.org/browse/C/CC/E
FAOSTAT. (2018). Retrieved October 12, 2021, from http://www.fao.org/faostat/en/#data/QC
Fawale, O. S., Gbadamosi, S. O., Ige, M. M., & Kadiri, O. (2017). Effects of cooking and fermentation on the chemical composition, functional, and antinutritional properties of kariya (Hildergardia barteri) seeds. Food Science and Nutrition, 5, 1106–1115.
Feng, L., Xie, Y., Peng, C., Liu, Y., & Wang, H. (2018). A novel antidiabetic food produced via solid-state fermentation of tartary buckwheat by L. plantarum TK9 and L. paracasei TK1501. Food Technology and Biotechnology, 56, 373–380. https://doi.org/10.17113/ftb.56.03.18.5540
Ferreira, T. A., & Arêas, J. A. G. (2010). Calcium bioavailability of raw and extruded amaranth grains. Food Science and Technology, 30, 532–538. https://doi.org/10.1590/S0101-20612010000200037
Fleddermann, M., Fechner, A., Rößler, A., Bähr, M., Pastor, A., Liebert, F., & Jahreis, G. (2013). Nutritional evaluation of rapeseed protein compared to soy protein for quality, plasma amino acids, and nitrogen balance–A randomized cross-over intervention study in humans. Clinical Nutrition, 32, 519–526. https://doi.org/10.1016/j.clnu.2012.11.005
Fritz, M., Vecchi, B., Rinaldi, G., & Añón, M. C. (2011). Amaranth seed protein hydrolysates have in vivo and in vitro antihypertensive activity. Food Chemistry, 126, 878–884. https://doi.org/10.1016/j.foodchem.2010.11.065
Garg, N. K., Dahuja, A., Singh, A., & Chaudhary, D. P. (2020). Understanding the starch digestibility characteristics of Indian maize hybrids. Indian Journal of Experimental Biology, 58, 738–744.
Giménez-Bastida, J. A., Laparra-Llopis, J. M., Baczek, N., & Zielinski, H. (2018). Buckwheat and buckwheat enriched products exert an anti-inflammatory effect on the myofibroblasts of colon CCD-18Co. Food & Function, 9, 3387–3397. https://doi.org/10.1039/C8FO00193F
Giusti, F., Caprioli, G., Ricciutelli, M., Vittori, S., & Sagratini, G. (2017). Determination of fourteen polyphenols in pulses by high performance liquid chromatography-diode array detection (HPLC-DAD) and correlation study with antioxidant activity and colour. Food Chemistry, 221, 689–697.
Graf, B. L., Poulev, A., Kuhn, P., Grace, M. H., Lila, M. A., & Raskin, I. (2014). Quinoa seeds leach phytoecdysteroids and other compounds with anti-diabetic properties. Food Chemistry, 163, 178–185.
Guardianelli, L. M., Salinas, M. V., & Puppo, M. C. (2019). Chemical and thermal properties of flours from germinated amaranth seeds. Journal of Food Measurement and Characterization, 13(2), 1078–1088. https://doi.org/10.1007/s11694-018-00023-1
Gulpinar, A. R., Orhan, I. E., Kan, A., Senol, F. S., Celik, S. A., & Kartal, M. (2012). Estimation of in vitro neuroprotective properties and quantification of rutin and fatty acids in buckwheat (Fagopyrum esculentum Moench) cultivated in Turkey. International Food Research Journal, 46, 536–543. https://doi.org/10.1016/j.foodres.2011.08.011
Guo, H., Richel, A., Hao, Y., Fan, X., Everaert, N., Yang, X., & Ren, G. (2020). Novel dipeptidyl peptidase-IV and angiotensin-I-converting enzyme inhibitory peptides released from quinoa protein by in silico proteolysis. Food Science and Nutrition, 8, 1415–1422. https://doi.org/10.1002/fsn3.1423
Handoyo, T., Meda, T., Urisu, A., Adachi, T., & Morita, M. (2006). Hypoallergenic buckwheat flour preparation by Rhizopus oligosporus and its application to soba noodle. Food Research International, 39, 598–605. https://doi.org/10.1016/j.foodres.2005.12.003
Hao, J., Wu, T., Li, H., Wang, W., & Liu, H. (2016). Dual effects of slightly acidic electrolyzed water (SAEW) treatment on the accumulation of γ-aminobutyric acid (GABA) and rutin in germinated buckwheat. Food Chemistry, 201, 87–93.
Hernández-Ledesma, B. (2019). Quinoa (Chenopodium quinoa Willd.) as source of bioactive compounds: a review. Bioactive Compounds in Health and Disease, 2, 27–47.
Herrera, T., Navarro, D. H. J., Fornari, T., Reglero, G., & Martin, D. (2019). Inhibitory effect of quinoa and fenugreek extracts on pancreatic lipase and α-amylase under in vitro traditional conditions or intestinal simulated conditions. Food Chemistry, 270, 509–517. https://doi.org/10.1016/j.foodchem.2018.07.145. Epub 2018 Jul 23 PMID: 30174080.
Houben, A., Götz, H., Mitzscherling, M., & Becker, T. (2010). Modification of the rheological behavior of amaranth (Amaranthus hypochondriacus) dough. Journal of Cereal Science, 51, 350–356.
Jan, R., Saxena, D. C., & Singh, S. (2016). Pasting, thermal, morphological, rheological and structural characteristics of Chenopodium (Chenopodium album) starch. LWT-Food Science and Technology, 66, 267–274.
Jan, R., Saxena, D., & Singh, S. (2017). Effect of germination on nutritional, functional, pasting, and microstructural properties of chenopodium (Chenopodium album) flour. Journal of Food Processing and Preservation, 41(3), e12959. https://doi.org/10.1111/jfpp.12959
Jeong, Y. H., Hur, H. J., Lee, A. S., Lee, S. H., & Sung, M. J. (2020). Amaranthus mangostanus inhibits the differentiation of osteoclasts and prevents ovariectomy-induced bone loss. Evidence Based Complementary Alternative Medicine, 2020, 1927017. https://doi.org/10.1155/2020/1927017. PMCID: PMC7029261, PMID: 32089716.
Jin, H. M., & Wei, P. (2011). Anti-fatigue properties of tartary buckwheat extracts in mice. International Journal of Molecular Science, 12, 4770–4780. https://doi.org/10.3390/ijms12084770
Joshi, D. C., Chaudhari, G. V., Sood, S., Kant, L., Pattanayak, A., Zhang, K., & Zhou, M. (2019). Revisiting the versatile buckwheat: reinvigorating genetic gains through integrated breeding and genomics approach. Planta, 250(3), 783–801.
Kalinova, J., & Dadakova, E. (2009). Rutin and total quercetin content in amaranth (Amaranthus spp.). Plant Foods for Human Nutrition, 64, 68–74.
Kalpanadevi, V., & Mohan, V. R. (2013). Effect of processing on antinutrients and in vitro protein digestibility of the underutilized legume, Vigna unguiculata (L.) Walp subsp. Unguiculata. LWT-Food Science and Technology, 51, 455–461.
Kamal, H., Mudgil, P., Bhaskar, B., Fisayo, A. F., Gan, C. Y., & Maqsood, S. (2021). Amaranth proteins as potential source of bioactive peptides with enhanced inhibition of enzymatic markers linked with hypertension and diabetes. Journal of Cereal Science, 101, 103308. https://doi.org/10.1016/j.jcs.2021.103308
Kaur, C., Sethi, M., Devi, V., Chaudhary, D. P., Phagna, R. K., Singh, A., Bhushan, B., Langyan, S., & Rakshit, S. (2022). Optimization of protein quality assay in normal, opaque-2 and quality protein maize. Frontiers in Sustainable Food Systems, 6, 743019. https://doi.org/10.3389/fsufs.2022.743019
Kaur, S., Singh, N., & Rana, J. C. (2010). Amaranthus hypochondriacus and Amaranthus caudatus germplasm: characteristics of plants, grain and flours. Food Chemistry, 123, 1227–1234.
Kuktaite, R., Repo-Carrasco-Valencia, R., de Mendoza, C. C., Plivelic, T. S., Hall, S., & Johansson, E. (2021). Innovatively processed quinoa (Chenopodium quinoa Willd.) food: chemistry, structure and end-use characteristics. Journal of the Science of Food and Agriculture. https://doi.org/10.1002/jsfa.11214
Kumar, S., Sangwan, S., Yadav, R., Langyan, S., & Singh, M. (2014). Maize carotenoid composition and biofortification for provitamin A activity. Maize: nutrition dynamics and novel uses (pp. 83–91). Springer.
Lamothe, L. M., Srichuwong, S., Reuhs, B. L., & Hamaker, B. R. (2015). Quinoa (Chenopodium quinoa W.) and amaranth (Amaranthus caudatus L.) provide dietary fibres high in pectic substances and xyloglucans. Food Chemistry, 167, 490–496.
Langyan, S., Dar, Z. A., Chaudhary, D. P., Shekhar, J. C., Herlambang, S., El-Enshasy, H., Sayyed, R. Z., & Rakshit, S. (2021a). Analysis of nutritional quality attributes and their inter-relationship in maize inbred lines for sustainable livelihood. Sustainability, 13, 1–12. https://doi.org/10.3390/su13116137
Langyan, S., Khan, F. N., Yadava, P., Alhazmi, A., Mahmoud, S. F., Saleh, D. I., Zuan, A. T. K., & Kumar, A. (2021b). In silico proteolysis and analysis of bioactive peptides from sequences of fatty acid desaturase 3 (FAD3) of flaxseed protein. Saudi Journal of Biological Sciences, 28, 5480–5489. https://doi.org/10.1016/j.sjbs.2021.08.027
Langyan, S., Yadava, P., Khan, F. N., Bhardwaj, R., Tripathi, K., Bhardwaj, V., Bhardwaj, R., Gautam, R. K., & Kumar, A. (2022a). Nutritional and food composition survey of major pulses toward healthy, sustainable, and biofortified diets. Frontiers in Sustainable Food Systems, 6, 878269. https://doi.org/10.3389/fsufs.2022.878269
Langyan, S., Yadava, P., Khan, F. N., Dar, Z. A., Singh, R., & Kumar, A. (2022b). Sustaining protein nutrition through plant-based foods. Frontiers in Nutrition, 8, 1237. https://doi.org/10.3389/fnut.2021.772573
Lee, C. C., Shen, S. R., Lai, Y. J., & Wu, S. C. (2013). Rutin and quercetin, bioactive compounds from tartary buckwheat, prevent liver inflammatory injury. Food & Function, 4, 794–802. https://doi.org/10.1039/C3FO30389F
Li, F., Zhang, X., Zheng, S., Lu, K., Zhao, G., & Ming, J. (2016). The composition, antioxidant and antiproliferative capacities of phenolic compounds extracted from tartary buckwheat bran [Fagopyrum tartaricum (L.) Gaerth]. Journal of Functional Foods, 22, 145–155. https://doi.org/10.1016/j.jff.2016.01.027
Liberal, Â., Calhelha, R. C., Pereira, C., Adega, F., Barros, L., Dueñas, M., Santos-Buelga, C., Abreu, R. M., & Ferreira, I. C. (2016). A comparison of the bioactivity and phytochemical profile of three different cultivars of globe amaranth: red, white, and pink. Food & Function, 7, 679–688.
Liu, W., Zhang, Y., Qiu, B., Fan, S., Ding, H., & Liu, Z. (2018). Quinoa whole grain diet compromises the changes of gut microbiota and colonic colitis induced by dextran Sulfate sodium in C57BL/6 mice. Scientific Reports, 8, 14916. https://doi.org/10.1038/s41598-018-33092-9
López, V. R. L., Razzeto, G. S., Giménez, M. S., & Escudero, N. L. (2011). Antioxidant properties of Amaranthus hypochondriacus seeds and their effect on the liver of alcohol-treated rats. Plant Foods for Human Nutrition, 66, 157–162. https://doi.org/10.1007/s11130-011-0218-4
Lorusso, A., Verni, M., Montemurro, M., Coda, R., Gobbetti, M., & Rizzello, C. G. (2017). Use of fermented quinoa flour for pasta making and evaluation of the technological and nutritional features. LWT, 78, 215–221.
Maradini, A. M., Ribeiro, M., Da Silva, J. T., Pinheiro, H. M., Paes, J. B., & Dos Reis, J. S. (2017). Quinoa: nutritional, functional and antinutritional aspects. Critical Reviews in Food Science and Nutrition, 57(8), 1618–1630. https://doi.org/10.1080/10408398.2014.1001811
Martínez-Villaluenga, C., Peñas, E., & Hernández-Ledesma, B. (2020). Pseudocereal grains: nutritional value, health benefits and current applications for the development of gluten-free foods. Food and Chemical Toxicology, 137, 111178.
Mir, N. A., Riar, C. S., & Singh, S. (2018). Nutritional constituents of pseudo cereals and their potential use in food systems: a review. Trends in Food Science & Technology, 75, 170–180.
Moroni, A. V., Zannini, E., Sensidoni, G., & Arendt, E. K. (2012). Exploitation of buckwheat sourdough for the production of wheat bread. European Food Research and Technology, 235, 659–668. https://doi.org/10.1007/s00217-012-1790-z
Moronta, J., Smaldini, P. L., Docena, G. H., & Añón, M. C. (2016). Peptides of amaranth were targeted as containing sequences with potential anti-inflammatory properties. Journal of Functional Foods, 21, 463–473. https://doi.org/10.1016/j.jff.2015.12.022
Mota, C., Nascimento, A. C., Santos, M., Delgado, I., Coelho, I., Rego, A., Matos, A. S., Torres, D., & Castanheira, I. (2016). The effect of cooking methods on the mineral content of quinoa (Chenopodium quinoa), amaranth (Amaranthus sp.) and buckwheat (Fagopyrum esculentum). Journal of Food Composition and Analysis, 49, 57–64. https://doi.org/10.1016/j.jfca.2016.02.006
Mudgil, P., Omar, L. S., Kamal, H., Kilari, B. P., & Maqsood, S. (2019). Multi-functional bioactive properties of intact and enzymatically hydrolysed quinoa and amaranth proteins. LWT, 110, 207–213. https://doi.org/10.1016/j.lwt.2019.04.084
Muyonga, J. H., Andabati, B., & Ssepuuya, G. (2014). Effect of heat processing on selected grain amaranth physicochemical properties. Food Science and Nutrition, 2, 9–16.
NCBI. (2022). Retrieved July 22, 2022, from https://pubchem.ncbi.nlm.nih.gov/
Ng, C. Y., & Wang, M. (2021). The functional ingredients of quinoa (Chenopodium quinoa) and physiological effects of consuming quinoa: a review. Food Frontiers, 2, 329–356. https://doi.org/10.1002/fft2.109
Niland, B., & Cash, B. D. (2018). Health benefits and adverse effects of a gluten-free diet in non–celiac disease patients. Gastroentero Hepatology, 14, 82.
Nongonierma, A. B., Le Maux, S., Dubrulle, C., Barre, C., & FitzGerald, R. J. (2015). Quinoa (Chenopodium quinoa Willd.) protein hydrolysates with in vitro dipeptidyl peptidase IV (DPP-IV) inhibitory and antioxidant properties. Journal of Cereal Science, 65, 112–118.
Obaroakpo, J. U., Liu, L., Zhang, S., Lu, J., Pang, X., & Lv, J. (2019). α-Glucosidase and ACE dual inhibitory protein hydrolysates and peptide fractions of sprouted quinoa yoghurt beverages inoculated with Lactobacillus casei. Food Chemistry, 299, 124985. https://doi.org/10.1016/j.foodchem.2019.124985. Epub 2019 Jun 28. PMID: 31279127.
Odongo, G. A., Schlotz, N., Baldermann, S., Neugart, S., Ngwene, B., Schreiner, M., & Lamy, E. (2018). Effects of Amaranthus cruentus L. on aflatoxin B1-and oxidative stress-induced DNA damage in human liver (HepG2) cells. Food Bioscience, 26, 42–48. https://doi.org/10.1016/j.fbio.2018.09.006
Ontiveros, N., López-Teros, V., de Jesús Vergara-Jiménez, M., Islas-Rubio, A. R., Cárdenas-Torres, F. I., Cuevas-Rodríguez, E. O., Reyes-Moreno, C., Granda-Restrepo, D. M., Lopera-Cardona, S., Ramírez-Torres, G. I., & Cabrera-Chávez, F. (2020). Amaranth-hydrolyzate enriched cookies reduce the systolic blood pressure in spontaneously hypertensive rats. Journal of Functional Foods, 64, 103613. https://doi.org/10.1016/j.jff.2019.103613
Patil, S. B., & Jena, S. (2020). Utilization of underrated pseudocereals of North East India: a systematic review. Nutrition and Food Science, 50, 1229–1240.
Pirzadah, T. B., & Malik, B. (2020). Pseudocereals as super foods of 21st century: recent technological interventions. Journal of Agricultural and Food Research, 2, 100052.
Pongrac, P., Potisek, M., Fraś, A., Likar, M., Budič, B., Myszka, K., Boros, D., Nečemer, M., Kelemen, M., Vavpetič, P., & Pelicon, P. (2016). Composition of mineral elements and bioactive compounds in tartary buckwheat and wheat sprouts as affected by natural mineral-rich water. Journal of Cereal Science, 69, 9–16. https://doi.org/10.1016/j.jcs.2016.02.002
Popova, A., & Mihaylova, D. (2019). Antinutrients in plant-based foods: a review. The Open Biotechnology Journal, 13, 68–76. https://doi.org/10.2174/1874070701913010068
Priego-Poyato, S., Rodrigo-Garcia, M., Escudero-Feliu, J., Garcia-Costela, M., Lima-Cabello, E., Carazo-Gallego, A., & Jimenez-Lopez, J. C. (2021). Current advances research in nutraceutical compounds of legumes, pseudocereals and cereals. In J. C. Jimenez-Lopez (Ed.), Grain and seed proteins functionality. London: IntechOpen. https://doi.org/10.5772/intechopen.97782
Pritham, S. M., Revanna, M. L., Ravindra, U., Kalpana, B., Murthy, N., & Madhusudan. (2021). Physico-chemical, functional and anti-nutritional factors of the white bold quinoa (Chenopodium quinoa willd). International Journal of Chemical Studies, 9(2), 1103–1107. https://doi.org/10.22271/chemi.2021.v9.i2p.11967
Qiu, R., Wang, J., & Parkin, K. L. (2014). Isolation and identification of cytoprotective agents from nonpolar extracts of buckwheat flour. International Food Research Journal, 66, 86–92. https://doi.org/10.1016/j.foodres.2014.08.037
Quan, C., Ferreiro, S., & Cantón, O. (2018). Gluten-free diet: always as easy, useful, and healthy as people think? Journal of Child Science, 08, e75–e81. https://doi.org/10.1055/s-0038-1669381
Quiroga, A. V., Aphalo, P., Nardo, A. E., & Añón, M. C. (2017). In vitro modulation of renin–Angiotensin system enzymes by amaranth (Amaranthus hypochondriacus) protein-derived peptides: alternative mechanisms different from ace inhibition. Journal of Agricultural and Food Chemistry, 65, 7415–7423. https://doi.org/10.1021/acs.jafc.7b02240
Quiroga, A. V., Barrio, D. A., & Añón, M. C. (2015). Amaranth lectin presents potential antitumor properties. LWT-Food Science and Technology, 60, 478–485. https://doi.org/10.1016/j.lwt.2014.07.035
Ramírez-Torres, G., Ontiveros, N., Lopez-Teros, V., Ibarra-Diarte, J. A., Reyes-Moreno, C., Cuevas-Rodríguez, E. O., & Cabrera-Chávez, F. (2017). Amaranth protein hydrolysates efficiently reduce systolic blood pressure in spontaneously hypertensive rats. Molecules, 22, 1905. https://doi.org/10.3390/molecules22111905
Ramos Diaz, J. M., Sundarrajan, L., Kariluoto, S., Lampi, A. M., Tenitz, S., & Jouppila, K. (2017). Effect of extrusion cooking on physical properties and chemical composition of corn-based snacks containing amaranth and quinoa: application of partial least squares regression. Journal of Food Process Engineering, 40, e12320. https://doi.org/10.1111/jfpe.12320
Repo-Carrasco-Valencia, R. A., Encina, C. R., Binaghi, M. J., Greco, C. B., & Ronayne de Ferrer, P. A. (2010). Effects of roasting and boiling of quinoa, kiwicha and kañiwa on composition and availability of minerals in vitro. Journal of the Science of Food and Agriculture, 90, 2068–2073. https://doi.org/10.1002/jsfa.4053. PMID: 20582934.
Rizzello, C. G., Lorusso, A., Montemurro, M., & Gobbetti, M. (2016). Use of sourdough made with quinoa (Chenopodium quinoa) flour and autochthonous selected lactic acid bacteria for enhancing the nutritional, textural and sensory features of white bread. Food Microbiology, 56, 1–13.
Rjeibi, I., Saad, A. B., & Hfaiedh, N. (2016). Oxidative damage and hepatotoxicity associated with deltamethrin in rats: the protective effects of Amaranthus spinosus seed extract. Biomedicine & Pharmacotherapy, 84, 853–860. https://doi.org/10.1016/j.biopha.2016.10.010
Rocchetti, G., Miragoli, F., Zacconi, C., Lucini, L., & Rebecchi, A. (2019a). Impact of cooking and fermentation by lactic acid bacteria on phenolic profile of quinoa and buckwheat seeds. Food Research International, 119, 886–894. https://doi.org/10.1016/j.foodres.2018.10.073
Rocchetti, G., Lucini, L., Rodriguez, J. M. L., Barba, F. J., & Giuberti, G. (2019b). Gluten-free flours from cereals, pseudocereals and legumes: phenolic fingerprints and in vitro antioxidant properties. Food Chemistry, 271, 157–164.
Rodríguez, J. P., Rahman, H., Thushar, S., & Singh, R. K. (2020). Healthy and resilient cereals and pseudo-cereals for marginal agriculture: molecular advances for improving nutrient bioavailability. Frontiers in Genetics, 11, 49. https://doi.org/10.3389/fgene.2020.00049
Ruiz, K. B., Khakimov, B., Søren, B. E., Søren, B., Stefania, B., & Sven-Erik, J. (2017). Quinoa seed coats as an expanding and sustainable source of bioactive compounds: an investigation of genotypic diversity in saponin profiles. Industrial Crops and Products, 104, 156–163. https://doi.org/10.1016/j.indcrop.2017.04.007
Rybicka, I., & Gliszczynska-Swiglo, A. (2017). Gluten-Free flours from different raw materials as the source of vitamin B1, B2, B3 and B6. Journal of Nutritional Science and Vitaminology, 63(2), 125–132.
Sabbione, A. C., Rinaldi, G., Añón, M. C., & Scilingo, A. A. (2016). Antithrombotic effects of Amaranthus hypochondriacus proteins in rats. Plant Foods for Human Nutrition, 71, 19–27. https://doi.org/10.1007/s11130-015-0517-2
Sánchez-López, F., Robles-Olvera, V. J., Hidalgo-Morales, M., & Tsopmo, A. (2021). Angiotensin-I converting enzyme inhibitory activity of Amaranthus hypochondriacus seed protein hydrolysates produced with lactic bacteria and their peptidomic profiles. Food Chemistry, 363, 130320. https://doi.org/10.1016/j.foodchem.2021.130320
Sandoval-Sicairos, E. S., Milán-Noris, A. K., Luna-Vital, D. A., Milán-Carrillo, J., & Montoya-Rodríguez, A. (2021). Anti-inflammatory and antioxidant effects of peptides released from germinated amaranth during in vitro simulated gastrointestinal digestion. Food Chemistry, 343, 128394. https://doi.org/10.1016/j.foodchem.2020.128394
Sciarini, L. S., Steffolani, M. E., Fernández, A., Paesani, C., & Pérez, G. T. (2020). Gluten-free breadmaking affected by the particle size and chemical composition of quinoa and buckwheat flour fractions. Food Science and Technology International, 26, 321–332.
Shreeja, K., Devi, S. S., Suneetha, W. J., & Prabhakar, B. N. (2021). Effect of germination on nutritional composition of common buckwheat (Fagopyrum esculentum Moench). International Research Journal of Pure & Applied Chemistry, 22, 1–7. https://doi.org/10.9734/irjpac/2021/v22i130350
Shukla, A., Srivastava, N., Suneja, P., Yadav, S. K., Hussain, Z., Rana, J. C., & Yadav, S. (2018). Genetic diversity analysis in Buckwheat germplasm for nutritional traits. Indian Journal of Experimental Biology, 56, 827–837.
Sindhu, R., Beniwal, S. K., & Devi, A. (2019). Effect of grain processing on nutritional and physico-chemical, functional and pasting properties of amaranth and quinoa flours. Indian Journal of Traditional Knowledge, 18(3), 500–507.
Singh, R., Langyan, S., Sangwan, S., Rohtagi, B., Khandelwal, A., & Shrivastava, M. (2022). Protein for human consumption from oilseed cakes: a review. Frontiers in Sustainable Food Systems, 6, 856401. https://doi.org/10.3389/fsufs.2022.856401
Sirotkin, A. V., Macejková, M., Tarko, A., Fabova, Z., Alwasel, S., & Harrath, A. H. (2021). Buckwheat, rooibos, and vitex extracts can mitigate adverse effects of xylene on ovarian cells in vitro. Environmental Science and Pollution Research, 28, 7431–7439. https://doi.org/10.1007/s11356-020-11082-7
Siwatch, M., Yadav, R., & Yadav, B. (2019). Chemical, physicochemical, pasting and microstructural properties of amaranth (Amaranthus hypochondriacus) flour as affected by different processing treatments. Quality Assurance and Safety of Crops & Food, 11(1), 3–13. https://doi.org/10.3920/QAS2017.1226
Soares, R. A. M., Mendonça, S., De Castro, L. Í. A., Menezes, A. C. C. C. C., & Arêas, J. A. G. (2015). Major peptides from amaranth (Amaranthus cruentus) protein inhibit HMG-CoA reductase activity. International Journal of Molecular Sciences, 16, 4150–4160.
Stikić, R. I., Milinčić, D. D., Kostić, A. Ž, Jovanović, Z. B., Gašić, U. M., Tešić, ŽL., Djordjević, N. Z., Savić, S. K., Czekus, B. G., & Pešić, M. B. (2020). Polyphenolic profiles, antioxidant, and in vitro anticancer activities of the seeds of Puno and Titicaca quinoa cultivars. Cereal Chemistry, 97, 626–633.
Suárez, S., Aphalo, P., Rinaldi, G., Añón, M. C., & Quiroga, A. (2020). Effect of amaranth proteins on the RAS system. In vitro, in vivo and ex vivo assays. Food Chemistry, 308, 125601. https://doi.org/10.1016/j.foodchem.2019.125601
Świątecka, D., Markiewicz, L. H., & Wroblewska, B. (2013). In vitro evaluation of the effect of the buckwheat protein hydrolysate on bacterial adhesion, physiology and cytokine secretion of Caco-2 cells. Central European Journal of Immunology, 8, 317–327. https://doi.org/10.5114/ceji.2013.37753
Świeca, M., Regula, J., Suliburska, J., Zlotek, U., Gawlik-Dziki, U., & Ferreira, I. M. (2020). Safeness of diets based on gluten-free buckwheat bread enriched with seeds and nuts-effect on oxidative and biochemical parameters in rat serum. Nutrients, 12, 41. https://doi.org/10.3390/nu12010041
Tang, Y., Li, X., Chen, P. X., Zhang, B., Liu, R., Hernandez, M., & Tsao, R. (2016). Assessing the fatty acid, carotenoid, and tocopherol compositions of amaranth and quinoa seeds grown in Ontario and their overall contribution to nutritional quality. Journal of Agricultural and Food Chemistry, 64(5), 1103–1110.
Taniya, M. S., Reshma, M. V., Shanimol, P. S., Krishnan, G., & Priya, S. (2020). Bioactive peptides from amaranth seed protein hydrolysates induced apoptosis and antimigratory effects in breast cancer cells. Food Bioscience, 35, 100588. https://doi.org/10.1016/j.fbio.2020.100588
Tao, T., Pan, D., Zheng, Y. Y., & jun Ma, T. (2019). Optimization of hydrolyzed crude extract from tartary buckwheat protein and analysis of its hypoglycemic activity in vitro. IOP Conference Series: Environmental and Earth Sciences, 295(3), 032065. IOP Publishing.
Thakur, P., & Kumar, K. (2019). Nutritional importance and processing aspects of Pseudocereals. Journal of Agricultural Engineering and Food Technology, 6, 155–160.
Thakur, P., Kumar, K., Ahmed, N., et al. (2021a). Effect of soaking and germination treatments on nutritional, anti-nutritional, and bioactive properties of amaranth (Amaranthus hypochondriacus L.), quinoa (Chenopodium quinoa L.), and buckwheat (Fagopyrum esculentum L.). Current Research in Food Science, 4, 917–925. https://doi.org/10.1016/j.crfs.2021.11.019. PMID: 34927087; PMCID: PMC8646961.
Thakur, P., Kumar, K., & Dhaliwal, H. S. (2021b). Nutritional facts, bio-active components and processing aspects of pseudocereals: a comprehensive review. Food Bioscience, 42, 101170. https://doi.org/10.1016/J.FBIO.2021.101170
Tien, N. N. T., Trinh, L. N. D., Inoue, N., Morita, N., & Hung, P. V. (2018). Nutritional composition, bioactive compounds, and diabetic enzyme inhibition capacity of three varieties of buckwheat in Japan. Cereal Chemistry, 95(5), 615–624.
Tontul, İ, Kasimoglu, Z., Asik, S., Atbakan, T., & Topuz, A. (2018). Functional properties of chickpea protein isolates dried by refractance window drying. International Journal of Biological Macromolecules, 109, 1253–1259.
United Nation. (2017). Retrieved April 12, 2022, from https://www.un.org/development/desa/en/news/population/world-population-prospects-2017.html
Upasana, U., & Yadav, L. (2022). Pseudocereals: a novel path towards healthy eating. In V. Y. Waisundara (Ed.), Pseudocereals. London: IntechOpen. https://doi.org/10.5772/intechopen.103708
USDA. (2022). Retrieved July 18, 2022, from https://fdc.nal.usda.gov/
Vagadia, B. H., Vanga, S. K., Singh, A., Gariepy, Y., & Raghavan, V. (2018). Comparison of conventional and microwave treatment on soymilk for inactivation of trypsin inhibitors and in vitro protein digestibility. Foods, 7, 6.
Velarde-Salcedo, A. J., Barrera-Pacheco, A., Lara-González, S., Montero-Morán, G. M., Díaz-Gois, A., González de Mejia, E., & Barba de la Rosa, A. P. (2013). In vitro inhibition of dipeptidyl peptidase IV by peptides derived from the hydrolysis of amaranth (Amaranthus hypochondriacus L.) proteins. Food Chemistry, 136(2), 758–64. https://doi.org/10.1016/j.foodchem.2012.08.032. Epub 2012 Aug 25 PMID: 23122124.
Vera, E. P., Alca, J. J., Saravia, G. R., Campioni, N. C., & Alpuy, I. J. (2019). Comparison of the lipid profile and tocopherol content of four Peruvian quinoa (Chenopodium quinoa Willd.) cultivars (‘Amarilla de Maranganí’, ‘Blanca de Juli’, INIA 415 ‘Roja Pasankalla’, INIA 420 ‘Negra Collana’) during germination. Journal of Cereal Science, 88, 132–137.
Vilcacundo, R., Martínez-Villaluenga, C., & Hernández-Ledesma, B. (2017). Release of dipeptidyl peptidase IV, α-amylase and α-glucosidase inhibitory peptides from quinoa (Chenopodium quinoa Willd.) during in vitro simulated gastrointestinal digestion. Journal of Functional Foods, 35, 531–539.
WronkoWska, H. Z. M. (2016). Gluten-free muffins based on fermented and unfermented buckwheat flour–content of selected elements. Journal of Food and Nutrition Research, 55, 108–113. https://doi.org/10.1007/s13197-017-2561-4
Xiao, Y., Wu, X., Yao, X., Chen, Y., Ho, C. T., He, C., Li, Z., & Wang, Y. (2021). Metabolite profiling, antioxidant and α-glucosidase inhibitory activities of buckwheat processed by solid-state fermentation with Eurotium cristatum YL-1. Food Research International, 143, 110262. https://doi.org/10.1016/j.foodres.2021.110262
Xu, J. M., Fan, W., Jin, J. F., Lou, H. Q., Chen, W. W., Yang, J. L., et al. (2017). Transcriptome analysis of Al-induced genes in buckwheat (Fagopyrum esculentum Moench) root apex: new insight into Al toxicity and resistance mechanisms in an Al accumulating species. Frontiers in Plant Science, 8, 1141. https://doi.org/10.3389/fpls.2017.01141
Yasui, Y., Hirakawa, H., Oikawa, T., Toyoshima, M., Matsuzaki, C., Ueno, M., et al. (2016). Draft genome sequence of an inbred line of chenopodium quinoa, an allotetraploid crop with great environmental adaptability and outstanding nutritional properties. DNA Research 23, 535–546. https://doi.org/10.1093/dnares/dsw037
Yao, Y., Shi, Z., & Ren, G. (2014). Antioxidant and immunoregulatory activity of polysaccharides from quinoa (Chenopodium quinoa Willd.). International Journal of Molecular Science, 15, 19307–19318. https://doi.org/10.3390/ijms151019307. PMID: 25342323; PMCID: PMC4227275.
Zeyneb, H., Pei, H., Cao, X., Wang, Y., Win, Y., & Gong, L. (2021). In vitro study of the effect of quinoa and quinoa polysaccharides on human gut microbiota. Food Science and Nutrition. https://doi.org/10.1002/fsn3.2540
Zhang, G., Xu, Z., Gao, Y., Huang, X., Zou, Y., & Yang, T. (2015). Effects of germination on the nutritional properties, phenolic profiles, and antioxidant activities of buckwheat. Journal of Food Science, 80(5), H1–H9. https://doi.org/10.1111/1750-3841.12830
Zhang, Q., & Xu, J. G. (2017). Determining the geographical origin of common buckwheat from China by multivariate analysis based on mineral elements, amino acids and vitamins. Scientific Reports, 7(1), 1–8.
Zheng, Y., Wang, X., Zhuang, Y., Li, Y., Tian, H., Shi, P., & Li, G. (2019). Isolation of novel ACE-inhibitory and antioxidant peptides from quinoa bran albumin assisted with an in silico approach: characterization, in vivo antihypertension, and molecular docking. Molecules, 24, 4562. https://doi.org/10.3390/molecules24244562. PMID: 31842519; PMCID: PMC6943578.
Zhou, X., Huang, L., Tang, W., Zhou, Y., Wang, Q., & Li, Z. (2013). A novel buckwheat protein with a beneficial effect in atherosclerosis was purified from Fagopyrum tataricum (L.). Archives of Biological Sciences, 65, 767–772. https://doi.org/10.2298/ABS1302767Z
Zieliński, H., Ciesarová, Z., Kukurová, K., Zielinska, D., Szawara-Nowak, D., Starowicz, M., & Wronkowska, M. (2017). Effect of fermented and unfermented buckwheat flour on functional properties of gluten-free muffins. Journal of Food Science and Technology, 54, 1425–1432.
Zieliński, H., Honke, J., Bączek, N., Majkowska, A., & Wronkowska, M. (2019). Bioaccessibility of D-chiro-inositol from water biscuits formulated from buckwheat flours fermented by lactic acid bacteria and fungi. LWT, 106, 37–43.
Zieliński, H., Szawara-Nowak, D., & Wronkowska, M. (2020). Bioaccessibility of anti-AGEs activity, antioxidant capacity and phenolics from water biscuits prepared from fermented buckwheat flours. LWT, 123, 109051. https://doi.org/10.1016/j.lwt.2020.109051
Acknowledgements
The authors are highly indebted to ICAR (Indian Council of Agricultural Research), New Delhi for support.
Author information
Authors and Affiliations
Contributions
Conceptualization, S.L.; writing S.L, and F.N.K.; editing A.K.
Corresponding author
Ethics declarations
Ethics Approval
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Langyan, S., Khan, F.N. & Kumar, A. Advancement in Nutritional Value, Processing Methods, and Potential Applications of Pseudocereals in Dietary Food: A Review. Food Bioprocess Technol 17, 571–590 (2024). https://doi.org/10.1007/s11947-023-03109-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11947-023-03109-x