Abstract
In this study, the effect of cinnamon essential oil (CEO) at 0.3 and 0.6 mg/mL concentrations loaded in the nanostructured lipid carriers (NLC) against Penicillium citrinum and Penicillium expansum involved in tangerine decay was investigated. The minimum inhibitory concentrations (MIC) of the CEO and CEO-loaded NLC against both P. citrinum and P. expansum were about 0.425 and 1 mg/mL, respectively. Moreover, the minimum fungicidal concentrations (MFC) of the CEO and CEO-loaded NLC were 0.675 and 1.5 mg/mL, respectively, and the values were approximately the same for P. citrinum and P. expansum. According to the chemical and sensory analysis during 25 days of storage at 25 °C, the CEO-loaded NLC reduced the weight loss (from 30.7 to 26.3% and 27.1% at 0.3 and 0.6 mg/mL CEO, respectively) and there was no detrimental effect on the organoleptic and chemical properties such as titratable acidity, pH, TSS, and ascorbic acid content due to the treatment with CEO-loaded NLC. The tangerines were inoculated with P. citrinum and P. expansum spores and the percentage of the infected wounds were evaluated during 25 days of storage at 25 °C. The fungal spoilage of tangerine fruits reduced during storage, significantly (from 100% on the 10th day to 31% and 33% on day 25 for P. citrinum and P. expansum, respectively). Therefore, CEO-loaded NLC has the potential to be introduced as a new treatment for increasing tangerine shelf life.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Half of the world’s fruit and vegetable crops are lost due to postharvest deterioration (Echegoyen & Nerín, 2015). The citrus fruits are an economically important group of crops cultivated extensively in the world. Due to the low pH (Pérez-Alfonso et al., 2012), these fruits are susceptible to numerous postharvest fungal diseases. Penicillium expansum is one of the fungi responsible for postharvest losses of apples, pears, cherries, and citrus fruits. The fruits decayed by P. expansum have a penetrating, pungent, and earthy odor (Mattheis & Roberts, 1992). In addition, the mold fruit decay caused by Penicillium citrinum is difficult to control because of its ability to germinate at temperatures between 0 and 35 °C and proliferate by the mycelial growth (Wang et al., 2011). Although synthetic chemical fungicides can control certain diseases effectively, the use of fungicides to control the postharvest deterioration has been restricted due to their high and acute residual toxicity, long degradation period, environmental pollution, and adverse effects on food and possible side effects on human health (Tao et al., 2014). Hence, current research aims at developing alternative strategies for reducing the use of chemical additives in the food industry. In this context, the environmentally friendly plant extract agents, such as essential oils (EOs), have been shown great potential as alternatives to synthetic fungicides in the disease control or quality maintenance in tomatoes (Soylu et al., 2010), blueberries (Mehra et al., 2013), strawberries (Shao et al., 2013), avocadoes (Sellamuthu et al., 2013), ground beef (Almasi et al., 2020a, 2021), chocolate milk (Divya & Varadaraj, 2012), pork meat products (Bonilla et al., 2014), beef burgers (Ghaderi-Ghahfarokhi et al., 2016), fresh-cut apple (Amiri et al., 2018), fresh-cut orange (Radi et al., 2017a), palm date (Akhavan et al., 2021), sweet cherry fruit (Abdipour et al., 2020), cucumber and strawberry (Almasi et al., 2020b), and citrus (Fan et al., 2014).
Essential oils are volatile, natural, and aromatic oily liquids that can be obtained from several parts of the plants, especially the aerial ones as leaves and flowers (Ribeiro-Santos et al., 2018). Cinnamon (Cinnamomum zeylanicum) EO (CEO) is well known for its antimicrobial activity and has demonstrated high fungicidal activity against Fusarium moniliformae (Rodriguez et al., 2008). Cinnamon EO is rich in cinnamaldehyde (CA) as well as β-caryophyllene, linalool, and other terpenes. Cinnamaldehyde is the major constituent of the cinnamon bark oil and provides the distinctive odor and flavor associated with cinnamon. Cinnamaldehyde, linalool, eugenol, and 1,8 cineol have been reported as active components in inhibiting the growth of Monilia, Botrytis, and Mucor. Cinnamon EO is used worldwide as a food additive and flavoring agent. Cinnamon extract inhibits the growth of Aspergillus parasiticus, Endomyces fibuliger, Penicillium sp., and Pichia anomala (Tzortzakis, 2009).
Although EOs have been proved to be good antifungal agents, their use in maintaining the fruit quality and reducing the decay development is often limited, resulting from their high application costs and such disadvantages as high volatility, strong flavor, and potential toxicity (Amiri et al., 2013). Using carriers such as nanostructured lipid carriers (NLC) is an effective method for solving some of these problems as well as in controlling the fruit fungal disease by lowering the diffusion processes and maintaining high concentrations of the active molecules on the fruit surface (Aloui et al., 2014; Bagheri et al., 2019a).
Nanostructured lipid carriers refer to the nanoscale size particles that are prepared with lipids that remain solid at room temperature (Bilia et al., 2014). For manufacturing NLCs, lipids with very differently structured (sized) molecules can be used. Therefore, these systems are proper mediums for the entrapment of lipophilic compounds (Bagheri et al., 2019b). Nanostructured lipid carriers have good loading capacity because of their heterogeneous lipid structures which hamper crystallization and provide enough space for retaining the active compounds (Pardeike et al., 2009). It has been proved that these nano-capsules can protect labile compounds such as tocotrienol and retinoids from degradation (Ali et al., 2010a). Nanoscale particles provide a large surface area (comparing to micro-particles) which is important for maintaining the balance between effective release and retention of compounds. Several studies also showed that the incorporation of volatile compounds into the nanoscale particles prevents their rapid evaporation (Karrimi Khorrami et al., 2021; Lai et al., 2006). The advantage of using coatings amended with EOs rather than vapor is that there is closer contact between the EO and the fruit surface, allowing exposure of each fruit to similar concentrations of the inhibitor over a longer period (du Plooy et al., 2009). du Plooy et al. (2009) revealed that Lippia scaberrima EO (at 0.25%) in combination with carnauba tropical wax has provided either 100% (preventive treatment) or 95% (curative treatment) of the disease control against the citrus fruit P. digitatum infection.
To the best of our knowledge, there has been no published report regarding the use of NLC as the carrier of CEO in the field of postharvest (except for one on date palm fruit performed by Akhavan et al. (2021)) and moreover the effect of CEO to inactivate P. citrinum and P. expansum. The NLC systems have been extensively studied in pharmaceutical field for several years but there are a limited number of studies in food field. This is mostly because of the very limited number of ingredients which can be used in the formulation of NLCs and are legally permitted in foods. By introducing NLCs in food science in recent years, an increasing number of researches would be performed in near future. Therefore, the aim of this study was to determine the effect of CEO loaded in NLC against P. citrinum and P. expansum (as two of the major postharvest pathogens of the citrus fruits) in order to increase the tangerine shelf life with an emphasis on the potential use of this system as an alternative antifungal compound in the future.
Materials and Methods
Materials
Cinnamon plant barks were obtained from a local market in Shiraz. Phenolphthalein, ascorbic acid, metaphosphoric acid, NaOH, and all other chemicals were purchased from Merck (Darmstadt, Germany).
Fungal Strain
The pathogen Penicillium citrinum (PTCC 5304) was purchased from the Persian Type Culture Collection (PTCC) and the spores of P. expansum were isolated from an infected tangerine fruit and routinely cultured on the potato dextrose agar (PDA) for 7 days at 25 ± 2 °C. Spores suspensions were prepared using sporulating 3-week-old cultures in sterile distilled water and its concentration was determined with a hemocytometer and adjusted at the concentration of 105 CFU/mL for inoculation experiments.
Preparation of CEO
The cinnamon bark powder was distilled in a Clevenger-type steam distillation apparatus for 2 h. The obtained EO was separated from the aqueous phase solution, dried with sodium sulfate, and stored at 4 °C until used (Lai et al., 2006).
Preparation of NLC
For the preparation of NLC, the CEO was dissolved in the melted purified edible tallow (30% w/w) at 85 °C and the CEO-loaded lipid (2%) was dispersed in a hot Tween 60 aqueous solution of 5% (w/w). The mixtures were stirred with an Ultra-Turrax (T18, IKA, Germany) for 5 min at 8000 rpm. The obtained pre-emulsion was then homogenized (Lab-60 high-pressure homogenizer, APVGaulin, Germany) for three cycles at 90 °C (Lai et al., 2006) and the pressure of 800 bars. The final aqueous concentration of obtained NLC was 2% containing 0.6 mg/mL CEO. This dispersion was diluted with distilled water to the concentration of 1% NLC which corresponds to 0.3 mg/mL CEO.
Particle Size Analysis of NLC
The particle size analysis of NLC was measured based on the dynamic light scattering technique using a nano-particle size analyzer instrument (LB-550, Horiba, Japan) (Mozaffar et al., 2021).
Scanning Electron Microscopy (SEM)
The CEO-loaded NLC sample (1%) was diluted 1000 times with distilled water for better observation of individual particles. Afterward, the sample was placed on a glass lamella (1.5 × 1.5 cm) and allowed to dry at room temperature (25 °C). It was then sputter-coated with gold and examined under a field emission scanning electron microscope (WEGA3 SB, TSCAN, Czech Republic) operating at an accelerating voltage of 20 kV and the magnification of 35,000 × (Akhavan et al., 2021).
The Minimum Inhibitory Concentration (MIC) and Minimum Fungicidal Concentration (MFC) of CEO and CEO-Loaded NLC
The MIC and MFC of the CEO and CEO-loaded NLC against P. citrinum and P. expansum were determined by broth dilution method in the test tubes as follows: 50 μL from the various dilutions of CEO and CEO-loaded NLC was added to 5 mL of the sterile yeast extract sucrose broth tubes containing 105 CFU/mL. The tubes were then incubated on an incubator shaker (Benchtop, Oj-Azma Plast, Tehran, Iran) at 25 °C for 24 h. The highest dilution (the lowest concentration), showing no visible growth, was regarded as MIC. The tubes showing no growth were sub-cultured on potato dextrose agar plates to determine if the inhibition was reversible or permanent. Minimum fungicidal concentration was determined as the highest dilution (the lowest concentration) at which no growth occurred on the plates (Rammanee & Hongpattarakere, 2011). Trials were performed to find the ranges of effective dilutions; after which the ranges were focused twice for precise determination of MIC and MFC.
Plant Materials and Treatments Conditions
Tangerine fruits (Jahromi variety), uniform in size, ripeness, and color, free of physical injury, and signs of infection, were harvested from a local orchard, washed with sodium hypochlorite solution (20 mg/L), and allowed to dry at room temperature. The treatments were performed the day after harvest. Four treatments were considered, including the control (without any treatment), plain CEO at the concentration of 0.3 mg/mL, CEO-loaded NLC containing 0.3 mg/mL of CEO, and CEO-loaded NLC containing 0.6 mg/mL of CEO. The treatment of tangerines with plain CEO at the concentration of 0.6 mg/mL was not performed due to its severe and improper odor created in the fruits. Therefore, the fruits were dipped into CEO and NLC solutions for 1 min and after dripping, allowed to dry at room temperature. For each treatment, the fruits were divided into three parts. One part was inoculated with P. citrinum, the other was inoculated with P. expansum spores for evaluation of infection percentages, and the third part included the intact fruits with no inoculation for evaluation of physicochemical characteristics.
To perform inoculation, the fruits were wounded (2 mm deep and 2 mm wide) with a sterile nail, with three equatorial wounds per fruit. Then the wounded sites were inoculated with aliquots (20 µL) of spore suspensions (1 × 105 CFU/mL) and allowed to dry at room temperature. The fruits were then examined for infections, chemical, and sensory characterization during 25 days of storage at 25 ± 1 °C and 92 ± 2% relative humidity. Each treatment comprised four replicate boxes, each containing 40 fruits.
Weight Loss
The weight loss was calculated by using Eq. 1:
where A is the initial weight of the fruits and B is the fruit weight after the storage period (Amiri et al., 2021).
Degree of Fruit Spoilage
The number of infected or spoiled fruits was recorded periodically to assess the effect of CEO-loaded NLC on retarding fruit spoilage and the results were reported as the percentage of infected wounds (infected wounds of more than 3 mm in diameter were considered positive) (Eq. 2) (Radi et al., 2010):
Determination of the Titratable Acidity, pH, Total Soluble Solids (TSS), and Ascorbic Acid Content
The titratable acidity was assessed by titration with sodium hydroxide (0.1 N) and the results were expressed as the percentage of total acids. For pH measurements, 50 g of tangerine fruits was mixed with deionized water (1:20 w/v) and homogenized for 1 min. Then pH value was measured using a digital pH meter (MA235 model, Mettler-Toledo International Inc., Switzerland) at 20 °C. Total soluble solids were measured using a digital refractometer (Abe model Atago, NAR-3 T, Japan) at 20 °C. The ascorbic acid content was measured by titration using 2, 6-dichlorophenol-indophenol (Amiri et al., 2021).
Sensory Analysis
Sensory analysis was carried out by 33 selected panelists using a 5-point hedonic scale. Undamaged fruits (free of physical injury and signs of infection) were randomly selected and served on the white plates. The comparison was performed among the treatments (control, free CEO, NLC + 0.3 g/L CEO, and NLC + 0.6 g/L CEO) on days 0, 10, and 25. The sensory quality of fruits was evaluated for peel color, tangerine odor with peel, tangerine taste without peel, and overall acceptability (Marpudi et al., 2011). The peel color was scored from deep orange (score 5) to pale yellow (score 1). The whole tangerine odor, flesh taste, and overall acceptability were scored from like extremely (score 5) to dislike extremely (score 1).
Statistical Analysis
The SPSS 16.0 software (Team EQX, USA) was used for data analysis. The mean values were calculated and reported as the mean ± standard deviation (SD). The data were analyzed by the one-way analysis of variance (ANOVA) and further by Duncan’s multiple range test and differences at P < 0.05 were considered as significant.
Results and Discussion
Morphology and Particle Size Analysis of NLC
The particle size distribution of NLC is shown in Fig. 1. The particle sizes of NLC ranged from 16 to 220 nm with an average size of 94.9 nm and the span of 1.154. This result was very close to the reports of Karrimi Khorrami et al. (2021) and Bagheri et al., (2019a, 2019b) on NLC, Akhavan et al. (2021) on CEO-loaded NLC, and Almasi et al. (2021) on thyme oil-loaded microemulsion. The scanning electron micrograph of CEO-loaded NLC (Fig. 2) showed spherical nanoparticles with a smooth surface and a relatively narrow size distribution (according to DLS measurement). The lipid nanoparticles give the EO the opportunity to be homogeneously distributed throughout the interior of a film or a food medium as well as on the desired surfaces unlike the free EO (Karrimi Khorrami et al., 2021). Karrimi Khorrami et al. (2021) reported that the spherical structure of NLCs created greater surface roughness and porous structure in the alginate films.
The results of this study were also confirmed by Akhavan et al. (2021), Wang et al. (2012), and Tiyaboonchai et al. (2007). The micrograph was consistent with the result of particle size analysis.
MIC and MFC of CEO and CEO-Loaded NLC
The minimum inhibitory concentration and MFC values of CEO and NLC are shown in Table 1. The MFC values were significantly greater than the MIC values. The obtained MIC and MFC values of CEO and CEO-Loaded NLC were not significantly different for P. citrinum and P. expansum, respectively. The MIC and MFC values were 0.425 and 0.675 mg/mL, respectively, for CEO. These values for CEO-loaded NLC were more than two folds higher than free CEO for both molds (about 1.0 and 1.5 mg/mL for MIC and MFC, respectively). Cinnamon EO antifungal activity may be due to the bioactivity of CA (Xu et al., 2011), and also eugenol (Kouassi et al., 2012), as the main compounds of CEO are cinnamaldehyde (42–82%), eugenol (1–11%), cinnamic alcohol (8%), cinnamic acid (10%), cinnamyl acetate, o-methoxycinnamaldehyde, benzyl benzoate, linalool, and safrole (up to 2%) (Kouassi et al., 2012). Aldehydes are known to possess powerful antimicrobial activity. It has been proposed that an aldehyde group conjugated to a carbon-to-carbon double bond is a highly electronegative arrangement. Such electronegative compounds may interfere in biological processes involving electron transfer and react with vital nitrogen components, e.g., proteins and nucleic acids, and therefore inhibit the growth of the microorganism (Ranasinghe et al., 2003). The inhibitory activity of CEO against fungi has also been proved by other researchers. For filamentous fungi (three Aspergillus spp. and one Fusarium sp.), the MICs of CEO and CA from the Chinese medicinal herb Cinnamomum cassia bloom were ranged from 75 to 150 μg/mL (Ooi et al., 2006). Tzortzakis (2009) showed that CEO reduced the spore germination and the germ tube length in Colletotrichum coccodes, Botrytis cinerea, and Rhizopus stolonifer. This researcher declared that these antifungal effects were dependent on the oil concentration. Cinnamon leaf volatile oil, found by Singh et al. (2007), was 100% effective against Aspergillus niger, A. flavus, Fusarium moniliforme, Fusarium graminearum, P. citrinum, and Penicillium viridicatum.
As described above, the MIC and MFC values for CEO-loaded NLC were more than two folds higher than the plain CEO. This shows that CEO was efficiently entrapped in the solid lipid matrix of nanoparticles and all of the EO was not available at the experiment time. The entrapped EO can act as a reservoir during the storage period of fruits and may compensate the vaporized or diffused CEO, maintaining the required concentration for inhibiting the growth of molds. Shi et al. (2012) loaded frankincense and myrrh EOs into NLCs and stated that the evaporation loss of the active components was reduced in the NLCs. In another research, the rapid evaporation of Artemisia arborescens L. EO was reduced as the result of the incorporation of the EO into NLC (Lai et al., 2006).
The Effect of the CEO-Loaded NLC on Weight Loss During Storage
The postharvest weight changes in fruits and vegetables are usually due to the loss of water and the consumption of carbohydrates through transpiration and respiration processes (Amiri et al., 2021). The loss of water can lead to wilting and shriveling, which both reduce a commodity’s marketability (Fawole et al., 2012). The results of weight loss are shown in Fig. 3. The CEO-loaded NLC showed a significant effect on the weight loss of the tangerine fruits in comparison with the control and CEO samples. The reduction percentages of weight loss at 0.3 mg/mL CEO concentration were 22.9, 17.1, 4.5, and 14.2%, respectively, on the storage days of 10, 15, 20, and 25 in comparison with the control sample. The weight loss of 0.6 mg/mL and 0.3 mg/mL samples did not exhibit a significant difference during the storage time. Maximum percentages of weight loss were related to the control and CEO samples throughout storage (Fig. 3) and no significant differences were observed between these two samples (p ≥ 0.05).
Coatings form a semi-permeable barrier to water vapor and gas exchange, leading to weight loss reduction, respiration rate modification, and senescence delay of the coated produce (Hosseinifarahi et al., 2020). It is well known that lipid-based coatings such as waxes are effective against weight loss due to their apolar nature and good water barrier property (Garcia et al., 2000). Subsequently, the incorporation of sunflower oil as an apolar compound into a starch-based film (Garcia et al., 2000) or walnut oil into a whey protein isolate film (Galus & Kadzińska, 2016) reduced the water vapor permeability of coatings. Therefore, the incorporation of EOs as a source of non-polar and volatile compounds into the especially lipid-based coatings can keep high concentrations of EOs for extended periods of time from one side and gives better results in decreasing the water vapor permeability of coatings as well as reducing the levels of blue and green rots than that of the free EO at the same concentration from the other side (Kouassi et al., 2012). Acevedo-Fani et al. (2015) incorporated sage EO nanoemulsion into an alginate-based film and reported that the water vapor resistance of the film was improved. In a study conducted by Istúriz-Zapata et al. (2020), nanostructured coatings of chitosan containing CEO and CA could effectively reduce the weight loss of cucumber. Akhavan et al. (2021) declared that the application of CA-loaded NLC on the date palm fruit effectively reduced the weight loss of the samples. The weight losses of banana and papaya fruits treated with 0.4% cinnamon oil were 25.89 and 28.56%, respectively, after 28 days of cold storage, while control sample weight losses of banana and papaya were 32.14 and 38.12%, respectively (Maqbool et al., 2011).
On the other hand, it is said that ultra-fine materials such as nanoparticles exhibit a distinct adhesiveness to surfaces. The particles adhering to the surface may lead to film formation and therefore, to an occlusion effect. The occlusion can increase by decreasing the particle size (Muller et al., 2007). It is confirmed that nanoparticles are 15-folds more occlusive than microparticles. This higher occlusion may result in lower water loss which encourages the researchers to use lipid nanoparticles-containing formulations for dermal application and skin hydration (Pardeike & Muller, 2006). The SEM micrograph of CEO-loaded NLC (Fig. 2) showed that the particles made a good coverage (although the NLC solution was diluted 1000 times for microscopy). Subsequently, the small size of NLC particles ensures close and wide contact to the surface of the tangerines and may decrease the amount of water loss.
Effect of CEO-Loaded NLC on Fruit Spoilage During Storage
The effect of CEO-loaded NLC on the postharvest decay of tangerine fruits inoculated by P. citrinum and P. expansum was investigated and the results are shown in Fig. 3. Control samples showed 87.3 and 92.6% decay on day 5 for P. citrinum and P. expansum respectively, while on day 10, the decay percentage reached 100%. The CEO and CEO-loaded NLC at two used concentrations decreased the decay percentages significantly (p < 0.05). At 0.6 mg/mL CEO concentration, the spoilage percentages of inoculated fruits by P. citrinum were 0.0, 2.2, 15.6, 24.5, and 31.1% on storage days of 5, 10, 15, 20, and 25, respectively. On the same days, spoilage percentages were 8.9, 42.3, 71.2, 77.8, and 93.4, respectively, for 0.3 mg/mL CEO concentration. The higher CEO concentration was significantly effective in decreasing spoilage and increasing tangerine shelf life.
Also, the spoilage percentages of P. expansum inoculated fruits which were treated with 0.6 mg/mL CEO-loaded NLC were 0.0, 4.5, 17.8, 31.1, and 33.4%, respectively, on the storage days of 5, 10, 15, 20, and 25, while for the fruits treated with 0.3 mg/mL CEO-loaded NLC, the values were 11.2, 42.2, 82.2, 97.8, and 100% on the same storage days. Like the effect of CEO-loaded NLC on inoculated fruits by P. citrinum, the higher CEO concentration was more effective in the reduction of P. expansum spoilage. Moreover, the CEO-loaded NLC was more potent in reducing the infection caused by P. citrinum than P. expansum. Regarding free CEO, the spoilage percentages of the sample were initially quite similar to that of the 0.3 mg/mL CEO-loaded NLC (up to the 10th day). But after that, the spoilage percentages of CEO sample were higher than 0.3 mg/mL CEO-loaded NLC for both examined molds and were less than the control sample until the 15th day. This may be due to the controlled gradual release of EO in the NLC samples over time compared to the free CEO sample (Akhavan et al., 2021). A look back to the MIC and MFC results (Table 1) may support this hypothesis, as the CEO loaded in NLC particles showed higher MIC and MFC than free CEO but better results obtained by using 0.3 mg/mL CEO-loaded NLC than the free form. This was while it could be anticipated that the free CEO be more effective. This can be related to the loss of CEO as the result of evaporation, diffusion into the fruit cells, and also non-uniform distribution of CEO micro-droplets on fruit surface in comparison with NLC nanoparticles.
Wang et al. (2005) reported that CA has a conjugated double bond and a long CH chain outside the ring, resulting in higher antifungal activity. Besides, the hydroxyl groups in the antimicrobial compounds could form hydrogen bonds with active enzymes affecting the biosynthesis of mycotoxins, resulting in deactivation (Xu et al., 2011). According to Roller and Seedhar (2002), CA was very effective in reducing the viable counts of the natural microflora of kiwifruit when used at 0.15–0.75 mg/mL in a dipping solution. Ali et al. (2014) demonstrated that CEO and propolis extract decreased the severity score and disease incidence in chilli. Istúriz-Zapata et al. (2020) demonstrated that chitosan coating containing CEO or CA enhanced the postharvest quality of cucumber by indicating antifungal activity against Fusarium solani. Xing et al. (2012) reported that the antifungal activity of clove oil on orange decay caused P. citrinum was improved with increasing the oil concentration. The antifungal activities of cinnamon extract were evaluated on banana crown rot fungi (Colletotrichum musae, Fusarium spp., and Lasiodiplodia theobromae) in vitro. Cinnamon extract completely inhibited conidial germination and mycelial growth of all fungi at 5.0 mg/mL (Win et al., 2007). Perumal et al. (2017) confirmed the inhibitory effect of thyme, clove, and cinnamon EOs against Colletotrichum gloeosporioides and Lasiodiplodia theobromae (the major fungal diseases of mango). It has been reported that the mixtures of clove and cinnamon leaves successfully inhibited the growth of L. monocytogenes (Cava-Roda et al., 2012). Leaf and seed extracts of huamuchil (Pithecellobium dulce) had fungicidal effects on sporulation and mycelial growth of Botrytis cinerea, Penicillium digitatum, and Rhizopus stolonifer of strawberry fruit (Bautista-Baños et al., 2003). Akhavan et al. (2021) reported that the mold count in the treated CA-loaded NLC dates reduced by about 3.5 log CFU/g compared with the control. Zhu et al. (2013) controlled green mold decay of citrus fruit by using the combination of Rhodosporidium paludigenum and sodium bicarbonate. Chitosan–CEO coating delayed the appearance of sweet peppers’ surface decay in comparison to uncoated sweet peppers. The decay percentage of coated peppers was below 5% at the end of storage, whereas the uncoated samples showed the highest decay incidence (34%) (Xing et al., 2011). There are many other reports declaring the influence of EOs on reducing postharvest decay of fruits and vegetables, but there is a lack of research in using NLC as carriers of EOs in this regard.
Titratable Acidity, pH, and TSS Changes During Storage
Titratable acidity, pH, and TSS were measured during storage and the results are shown in Fig. 4. The acidity of fruits is an important characteristic to determine their quality and acceptability. Very high or very low acidity values are not recommended for qualified fruits (Sophia et al., 2014). Titratable acidity (Fig. 4a) decreased in all samples during storage and reached from 0.83 to 0.50 after 25 days of storage. Free CEO and the CEO-loaded NLCs showed no significant effect on titratable acidity. Reduction in fruits’ titratable acidity during storage is due to the conversion of acids into sugars and their further utilization in the metabolic processes of the fruits (Sophia et al., 2014). Tangerine fruits’ pH values (Fig. 4b) were increased in all of the samples during storage as a result of decreasing acidity and reached from ~ 3.20 on day 0 to about 3.85 on day 25. In this regard, no significant difference was observed among the samples (p ≥ 0.05).
A gradual significant increase of TSS was observed for all of the treatments (from 11.2 to about 15.0%) but there was not any significant difference among the samples (Fig. 4c) during the storage time (p ≥ 0.05). Total soluble solid changes may be related to respiration, ripeness, and water loss during storage (Jiang et al., 2005). Respiration in the fruit tissue causes a continuous decrease in the oxygen concentration and an increase in the carbon dioxide content over time; meanwhile, organic acids are consumed and converted to simple sugars during respiration, resulting in a decrease in TA and an increase in TSS and pH (Amiri et al., 2021). This was in line with Ranasinghe et al. (2003), who reported no significant difference between pH, total acidity, and TSS of treated bananas with cinnamon extract and control samples after 21 days of storage at 14 °C. Chitosan coatings incorporated with lemon EO did not show a significant effect in terms of the acidity, pH, and TSS of strawberries throughout storage (Perdones et al., 2012). Meanwhile, the date palm fruit treated with CA-loaded NLC showed lower pH, titratable acidity, and TSS compared to the control sample (Akhavan et al., 2021). Yin et al. (2019) showed that the mango fruits coated with the chitosan- or alginate-based coatings containing CEO could effectively inhibit TSS and titratable acidity decrease and decrease pH and the weight loss.
Ascorbic Acid Changes During Storage
Ascorbic acid is an important vitamin for human nutrition that is supplied by fruits (especially citrus and some tropical) and vegetables (Hernández et al., 2006). The effect of CEO and CEO-loaded NLC on the ascorbic acid content in tangerine fruit is shown in Fig. 5. Figure 6 reveals that a significant decrease in the ascorbic acid content of tangerine fruits during storage has occurred. No significant difference was observed between the control sample and the samples treated with CEO-loaded NLC in both concentrations which means that CEO-loaded NLC has no effect on decreasing the rate of AA reduction. According to Xing et al. (2011), the treatment of chitosan coating containing CEO (7.5 mg/mL) could reduce the loss of ascorbic acid in jujube fruits. However, the lower levels of ascorbic acid were achieved when CEO was used at higher concentrations (10 and 20 mg/mL). According to Jayaprakasha et al. (2007), chitosan–CEO coating could inhibit ascorbic acid loss due to the protection caused by phenolic antioxidants in the CEO. Shafiee et al. (2010) declared that the application of a nutrient solution containing salicylic acid resulted in no change in the ascorbic acid content of the strawberry fruit. Radi et al. (2017a) and Radi et al. (2017b) declared that the coating of orange slices with pectin-based coatings enriched with orange peel EO micro/nanoemulsions and gelatin incorporated with aloe vera, respectively, resulted in higher retention of ascorbic acid. Xu et al. (2020) declared that CEO nanoemulsion in combination with ascorbic acid decreased the degradation rate of ascorbic acid.
Ascorbic acid is more sensitive to destruction when the commodity is subjected to adverse handling and storage conditions. Losses are enhanced by extended storage, higher temperatures, low relative humidity, physical damage, and chilling injury. Also, L-ascorbic acid is easily oxidized and converted to L-dehydroascorbic acid (Lee & Kader, 2000). Moreover, the ascorbic acid reduction could be due to metabolizing and converting it to sugars, like the other acids mentioned before.
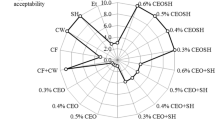
Effect of CEO-Loaded NLC on Sensory Properties of Fruits During Storage
One of the limiting factors for the application of coatings containing EOs on fruits and vegetables is their influence on the sensory characteristics of the coated products, mainly due to the high amounts of volatile compounds which alter the natural odor and flavor of fruits and vegetables (Sánchez-González et al., 2011). Therefore, sensory properties including color, odor (before and after peeling), and overall acceptability of the control sample and the treated tangerine fruits were evaluated at days 0, 10, and 25 of storage and the results are shown in Fig. 6. Color scores (Fig. 6a) of tangerine fruits were decreased gradually throughout the storage time (p < 0.05). There was no significant difference between the color scores of tangerine fruits at day 0 immediately after performing the treatments. At days 10 and 25, the highest color scores were related to tangerine fruits treated with NLC containing 0.6 mg/mL CEO (4.75 and 3.50, respectively). This treatment showed a statistically significant difference with free CEO, 0.3 mg/mL CEO-loaded NLC, and the control samples, but the difference between these three samples was not significant (p ≥ 0.05). It seems that NLC containing CEO can protect the color of tangerine fruit and higher CEO concentrations have more protective effect. Coatings applied to fruits could influence the fruit respiration rate and volatile levels by acting as a barrier that alters permeability to gases and could reduce the fungal spoilage and consequently slow down the external and internal color change of the fruits (Fagundes et al., 2014; Sophia et al., 2014). In the study of Xing et al. (2011), color changes of coated sweet peppers with the mixture of chitosan-CEO were negligible and they were still green at the end of storage in comparison with the uncoated sample. It has been described that the application of gum arabic, zein, alginate, and HPMC coatings was able to delay the color changes in tomatoes during storage at 20 °C by creating a modified atmosphere in the fruit (Ali et al., 2010b; Rong-yu & Yao-wen, 2003; Zapata et al., 2008). Changes in skin color of the grapes treated with grapefruit seed extract (GSE) or GSE plus chitosan increased slower than the uncoated fruits (Xu et al., 2007).
Odor evaluation of whole tangerine fruits before peeling (Fig. 6b) showed that the odor scores decreased during storage in all samples and CEO and CEO-containing NLC had an adverse effect on this parameter. On days 0, 10, and 25, the highest odor scores (4.83, 4.17, and 3.50, respectively) were related to the control sample and the lowest scores belonged to the free CEO. Regarding CEO-containing NLC, the odor score was decreased significantly with CEO concentration. In this regard, the lowest scores after the plain CEO were related to tangerine fruits treated with NLC containing 0.6 mg/mL CEO (p < 0.05).
The taste scores of tangerine fruits (Fig. 6c) were reduced throughout storage in all samples, but there was no significant difference between treated samples and untreated ones. It showed that treating with CEO and CEO-containing NLC had no adverse effect on edible parts taste of tangerine fruits.
The scores of overall acceptability (Fig. 6d) decreased throughout the storage in all samples. No significant differences were observed between the treated and untreated tangerine fruits. It showed that CEO and NLC containing CEO at both studied concentrations had no undesirable effect on sensory characteristics of tangerine fruit. It can be noted that although the application of CEO reduced the odor scores, its contribution to the overall acceptability was not significant, because the fruit odor before peeling was not important to the panelists. Akhavan et al. (2021) declared that the application of free CA and CA-loaded NLC improved the sensory attributes of date palm fruit stored at 4 °C for 180 days. According to Xu et al. (2011), chitosan coatings enriched with CEO at the concentrations of 0 to 0.75% did not produce undesirable sensory properties in jujube fruits. However, the higher concentrations at the range of 1.0 to 2.0% showed lower sensory acceptability. In the study of Ranasinghe et al. (2003), they reported no significant difference in odor and overall acceptability of CEO-treated banana compared to the control and benomyl treatment after 21 days, stored at 14 °C. Meanwhile, Nath et al. (2013) declared that the application of Ocimum sanctum not only was effective in inhibiting the Penicillium brevicompactum spore germination in Khasi mandarin but also created acceptable sensory properties.
Conclusion
Cinnamon essential oil–loaded NLC reduced significantly the weight loss of tangerine fruits during storage and had a strong antifungal effect against P. citrinum and P. expansum, while reducing tangerine infection from 100% at day 10 (control) to 31–33% at day 25 (0.6 mg/mL CEO-Loaded NLC) (p < 0.05). Higher CEO concentration was significantly effective in decreasing spoilage (p < 0.05). Titratable acidity, pH, ascorbic acid content, and total solids contents decreased during storage (p < 0.05), but free CEO and CEO-loaded NLC showed no significant effect on these chemical parameters (p ≥ 0.05). Nanostructured lipid carriers containing CEO (0.6 mg/mL) protected the color of tangerine fruits. The odor scores of intact fruits before peeling showed the adverse effect of applying CEO-loaded NLC, but it had no undesirable effect on flesh taste and overall acceptability of tangerines. The retention of quality and the extension of tangerine shelf life by CEO-loaded NLC revealed that such a method could be considered as a safe tangerine treatment method on a commercial scale during the storage and marketing process.
Data Availability
Data will be available if requested.
References
Abdipour, M., Sadat Malekhossini, P., Hosseinifarahi, M., & Radi, M. (2020). Integration of UV irradiation and chitosan coating: A powerful treatment for maintaining the postharvest quality of sweet cherry fruit. Scientia Horticulturae, 264, 109197.
Acevedo-Fani, A., Salvia-Trujillo, L., Rojas-Graü, M. A., & Martín-Belloso, O. (2015). Edible films from essential-oil-loaded nanoemulsions: Physicochemical characterization and antimicrobial properties. Food Hydrocolloids, 47, 168–177.
Akhavan, H. R., Hosseini, F. S., Amiri, S., & Radi, M. (2021). Cinnamaldehyde-loaded nanostructured lipid carriers extend the shelf life of date palm fruit. Food and Bioprocess Technology, 14(8), 1478–1489.
Ali, A., Chow, W. L., Zahid, N., & Kying, M. (2014). Efficacy of propolis and cinnamon oil coating in controlling post-harvest anthracnose and quality of chilli (Capsicum annuum L.) during cold storage. Food and Bioprocess Technology, 7, 2742–2748.
Ali, H., Shirode, A. B., Sylvester, P. W., & Nazzal, S. (2010a). Preparation and in vitro antiproliferative effect of tocotrienol loaded lipid nanoparticles. Colloids and Surfaces a: Physicochemical and Engineering Aspects, 353, 43–51.
Ali, A., Maqbool, M., Ramachandran, S., & Alderson, P. G. (2010b). Gum arabic as a novel edible coating for enhancing shelf-life and improving postharvest quality of tomato (Solanum lycopersicum L.) fruit. Postharvest Biology and Technology, 58, 42–47.
Almasi, L., Radi, M., & Amiri, S. (2020a). The release rate and antimicrobial activity of calcium‐alginate films containing self‐microemulsifying Thymus vulgaris essential oil against Escherichia coli and Staphylococcus aureus. Journal of Food Safety, 40, e12828.
Almasi, L., Radi, M., Amiri, S., & McClements, D. J. (2021). Fabrication and characterization of antimicrobial biopolymer films containing essential oil-loaded microemulsions or nanoemulsions. Food Hydrocolloids, 117, 106733.
Almasi, L., Radi, M., Amiri, S., & Torri, L. (2020b). Fully dilutable Thymus vulgaris essential oil:acetic or propionic acid microemulsions are potent fruit disinfecting solutions. Food Chemistry, 343, 128411.
Aloui, H., Khwaldia, K., Licciardello, F., Mazzaglia, A., Muratore, G., Hamdi, M., & Restuccia, C. (2014). Efficacy of the combined application of chitosan and Locust Bean Gum with different citrus essential oils to control postharvest spoilage caused by Aspergillus flavus in dates. International Journal of Food Microbiology, 170, 21–28.
Amiri, S., Abbasi, S., Ezzatpanah, H., & Hosseini, E. (2013). Nanocapsulation of orange peel oil using microemulsion technique. Agro Food Industry Hi Tech, 24(2), 72–75.
Amiri, S., Akhavan, H. R., Zare, N., & Radi, M. (2018). Effect of gelatin-based edible coatings incorporated with Aloe vera and green tea extracts on the shelf-life of fresh-cut apple. Italian Journal of Food Science, 30(1), 61–74.
Amiri, S., Nicknam, Z., Radi, M., Sayadi, M., Bagheri, F., Karimi Khorrami, N., & Abedi, E. (2021). Postharvest quality of orange fruit as influenced by salicylic acid, acetic acid, and carboxymethyl cellulose coating. Journal of Food Measurement and Characterization, 19 pages.
Bagheri, F., Radi, M., & Amiri, S. (2019a). Evaluating the physical, mechanical and morphological properties of sodium alginate nanocomposite film containing solid lipid nano-particles. Iranian Journal of Food Science and Technology, 16, 263–271.
Bagheri, F., Radi, M., & Amiri, S. (2019b). Drying conditions highly influence the characteristics of glycerol-plasticized alginate films. Food Hydrocolloids, 90, 162–171.
Bautista-Baños, S., Garcıa-Domınguez, E., Barrera-Necha, L., Reyes-Chilpa, R., & Wilson, C. (2003). Seasonal evaluation of the postharvest fungicidal activity of powders and extracts of huamuchil (Pithecellobium dulce): Action against Botrytris cinerea, Penicillium digitatum and Rhizopus stolonifer of strawberry fruit. Postharvest Biology and Technology, 29, 81–92.
Bilia, A. R., Guccione, C., Isacchi, B., Righeschi, C., Firenzuoli, F., & Bergonzi, M. C. (2014). Essential oils loaded in nanosystems: A developing strategy for a successful therapeutic approach. Evidence-Based Complementary and Alternative Medicine. https://doi.org/10.1155/2014/651593
Bonilla, J., Vargas, M., Atarés, L., & Chiralt, A. (2014). Effect of chitosan essential oil films on the storage-keeping quality of pork meat products. Food and Bioprocess Technology, l, 7, 2443–2450.
Cava-Roda, R. M., Taboada-Rodríguez, A., Valverde-Franco, M. T., & Iniesta, F. M. (2012). Antimicrobial activity of vanillin and mixtures with cinnamon and clove essential oils in controlling Listeria monocytogenes and Escherichia coli O157:H7 in milk. Food and Bioprocess Technology, 5, 2120–2131.
Divya, K. H., & Varadaraj, M. C. (2012). Response surface plots for the behavioral pattern of Yersinia enterocolitica in chocolate milk as affected by trans-cinnamaldehyde, a spice essential oil constituent. Food and Bioprocess Technology, 5, 498–507.
du Plooy, W., Regnier, T., & Combrinck, S. (2009). Essential oil amended coatings as alternatives to synthetic fungicides in citrus postharvest management. Postharvest Biology and Technology, 53, 117–122.
Echegoyen, Y., & Nerín, C. (2015). Performance of an active paper based on cinnamon essential oil in mushrooms quality. Food Chemistry, 170, 30–36.
Fagundes, C., Palou, L., Monteiro, A. R., & Pérez-Gago, M. B. (2014). Effect of antifungal hydroxypropyl methylcellulose-beeswax edible coatings on gray mold development and quality attributes of cold-stored cherry tomato fruit. Postharvest Biology and Technology, 92, 1–8.
Fan, F., Tao, N., Jia, L., & He, X. (2014). Use of citral incorporated in postharvest wax of citrus fruit as a botanical fungicide against Penicillium digitatum. Postharvest Biology and Technology, 90, 52–55.
Fawole, O., Arowora, K., & Nwaubani, S. (2012). Effects of edible coatings from aloe vera gel on quality and postharvest physiology of Ananas comosus (L.) fruit during ambient storage. Global Journal of Science Frontier Research, 12.
Galus, S., & Kadzińska, J. (2016). Whey protein edible films modified with almond and walnut oils. Food Hydrocolloids, 52, 78–86.
Garcia, M., Martino, M., & Zaritzky, N. (2000). Lipid addition to improve barrier properties of edible starch-based films and coatings. Journal of Food Science, 65, 941–947.
Ghaderi-Ghahfarokhi, M., Barzegar, M., Sahari, M. A., & Azizi, M. H. (2016). Nanoencapsulation approach to improve antimicrobial and antioxidant activity of thyme essential oil in beef burgers during refrigerated storage. Food and Bioprocess Technology, 9, 1187–1201.
Hernández, Y., Lobo, M. G., & González, M. (2006). Determination of vitamin C in tropical fruits: A comparative evaluation of methods. Food Chemistry, 96, 654–664.
Hosseinifarahi, M., Jamshidi, E., Amiri, S., Kamyab, F., & Radi, M. (2020). Quality, phenolic content, antioxidant activity, and the degradation kinetic of some quality parameters in strawberry fruit coated with salicylic acid and Aloe vera gel. Journal of Food Processing and Preservation, 44(9), e14647.
Isturiz-Zapataa, M. A., Hernandez-Lopeza, M., Correa-Pachecob, Z. N., & Barrera-Nechaa, N. N. (2020). Quality of cold-stored cucumber as affected by nanostructured coatings of chitosan with cinnamon essential oil and cinnamaldehyde. LWT - Food Science and Technology, 123, 109089.
Jayaprakasha, G., Negi, P., Jena, B., & Rao, L. J. M. (2007). Antioxidant and antimutagenic activities of Cinnamomum zeylanicum fruit extracts. Journal of Food Composition and Analysis, 20, 330–336.
Jiang, Y., Li, J., & Jiang, W. (2005). Effects of chitosan coating on shelf life of cold-stored litchi fruit at ambient temperature. LWT-Food Science and Technology, 38, 757–761.
Karimi Khorrami, N., Radi, M., Amiri, S., & McClements, D. J. (2021). Fabrication and characterization of alginate-based films functionalized with nanostructured lipid carriers. International Journal of Biological Macromolecules, 182, 373–384.
Kouassi, K. H. S., Bajji, M., & Jijakli, H. (2012). The control of postharvest blue and green molds of citrus in relation with essential oil–wax formulations, adherence and viscosity. Postharvest Biology and Technology, 73, 122–128.
Lai, F., Wissing, S. A., Müller, R. H., & Fadda, A. M. (2006). Artemisia arborescens L essential oil-loaded solid lipid nanoparticles for potential agricultural application: Preparation and characterization. An Official Journal of the American Association of Pharmaceutical Scientists, 7, E10–E18.
Lee, S. K., & Kader, A. A. (2000). Preharvest and postharvest factors influencing vitamin C content of horticultural crops. Postharvest Biology and Technology, 20, 207–220.
Maqbool, M., Ali, A., Alderson, P. G., Mohamed, M. T. M., Siddiqui, Y., & Zahid, N. (2011). Postharvest application of gum arabic and essential oils for controlling anthracnose and quality of banana and papaya during cold storage. Postharvest Biology and Technology, 62, 71–76.
Marpudi, S. L., Abirami, L., Pushkala, R., & Srividya, N. (2011). Enhancement of storage life and quality maintenance of papaya fruits using Aloe vera based antimicrobial coating. Indian Journal of Biotechnology, 10, 83–89.
Mattheis, J., & Roberts, R. (1992). Identification of geosmin as a volatile metabolite of Penicillium expansum. Applied and Environmental Microbiology, 58, 3170–3172.
Mehra, L., MacLean, D., Shewfelt, R., Smith, K., & Scherm, H. (2013). Effect of postharvest biofumigation on fungal decay, sensory quality, and antioxidant levels of blueberry fruit. Postharvest Biology and Technology, 85, 109–115.
Mozaffar, Sh., Radi, R., Amiri, S., & McClements, D.J. (2021). A new approach for drying of nanostructured lipid carriers (NLC) by spray-drying and using sodium chloride as the excipient. Journal of Drug Delivery Science and Technology, 61, 102212.
Muller, R., Hommoss, A., Pardeike, J., & Schmidt, C. (2007a). Lipid nanoparticles (NLC) as novel carrier for cosmetics: Special features & state of commercialisation. SÖFW-Journal, 133.
Nath, A., Barman, K., Chandra, S., & Baiswar, P. (2013). Effect of plant extracts on quality of Khasi mandarin (Citrus reticulata Blanco) fruits during ambient storage. Food and Bioprocess Technology, 6, 470–474.
Ooi, L. S., Li, Y., Kam, S. L., Wang, H., Wong, E. Y., & Ooi, V. E. (2006). Antimicrobial activities of cinnamon oil and cinnamaldehyde from the Chinese medicinal herb Cinnamomum cassia Blume. The American Journal of Chinese Medicine, 34, 511–522.
Pardeike, J., & Muller, R. (2006). In vivo skin hydration properties of a coenzyme Q10 containing cream with nanostructured lipid carriers (NLC). AAPS J 8, 001660.
Pardeike, J., Hommoss, A., & Muller, R. H. (2009). Lipid nanoparticles (SLN, NLC) in cosmetic and pharmaceutical dermal products. International Journal of Pharmaceutics, 366, 170–184.
Perdones, A., Sánchez-González, L., Chiralt, A., & Vargas, M. (2012). Effect of chitosan–lemon essential oil coatings on storage-keeping quality of strawberry. Postharvest Biology and Technology, 70, 32–41.
Pérez-Alfonso, C., Martínez-Romero, D., Zapata, P., Serrano, M., Valero, D., & Castillo, S. (2012). The effects of essential oils carvacrol and thymol on growth of Penicillium digitatum and P. italicum involved in lemon decay. International Journal of Food Microbiology, 158, 101–106.
Perumal, A. B., Sellamuthu, P. S., Nambiar, R. B., & Sadiku, E. R. (2017). Effects of essential oil vapour treatment on the postharvest disease control and different defence responses in two mango (Mangifera indica L.) cultivars. Food and Bioprocess Technology, 10, 1131–1141.
Radi, M., Afshari Jouybari, H., Mesbahi, G., Farahnaky, H., & Amiri, S. (2010). Effect of hot acetic acid solutions on postharvest decay caused by Penicillium expansum on Red Delicious apples. Scientia Horticulturae, 126, 421–425.
Radi, M., Akhavan-Darabi, S., Akhavan, H. R., & Amiri, S. (2017a). The use of orange peel essential oil microemulsion and nanoemulsion in pectin-based coating to extend the shelf life of fresh-cut orange. Journal of Food Processing and Preservation, 9 pages.
Radi, M., Firouzi, E., Akhavan, H., & Amiri, S. (2017b). Effect of gelatin based edible coatings incorporated with Aloe vera and black and green tea extracts on the shelf life of fresh-cut oranges. Journal of Food Quality, 2017, 1–10.
Rammanee, K., & Hongpattarakere, T. (2011). Effects of tropical citrus essential oils on growth, aflatoxin production, and ultrastructure alterations of Aspergillus flavus and Aspergillus parasiticus. Food and Bioprocess Technology, 4, 1050–1059.
Ranasinghe, L., Jayawardena, B., & Abeywickrama, K. (2003). Use of waste generated from cinnamon bark oil (Cinnamomum zeylanicum Blume) extraction as a post harvest treatment for Embul banana. Journal of Food, Agriculture, and Environment, 1, 340–344.
Ribeiro-Santos, R., Andrade, M., Sanches-Silva, A., & de Melo, N. R. (2018). Essential oils for food application: Natural substances with established biological activities. Food and Bioprocess Technology, 11, 43–71.
Rodriguez, A., Nerin, C., & Batlle, R. (2008). New cinnamon-based active paper packaging against Rhizopusstolonifer food spoilage. Journal of Agricultural and Food Chemistry, 56, 6364–6369.
Roller, S., & Seedhar, P. (2002). Carvacrol and cinnamic acid inhibit microbial growth in fresh-cut melon and kiwifruit at 4° and 8° C. Letters in Applied Microbiology, 35, 390–394.
Rong-yu, Z., & Yao-wen, H. (2003). Influence of hydroxypropyl methylcellulose edible coating on fresh-keeping and storability of tomato. Journal of Zhejiang University Science, 4, 109–113.
Sánchez-González, L., Vargas, M., González-Martínez, C., Chiralt, A., & Cháfer, M. (2011). Use of essential oils in bioactive edible coatings: A review. Food Engineering Reviews, 3, 1–16.
Sellamuthu, P. S., Sivakumar, D., Soundy, P., & Korsten, L. (2013). Essential oil vapours suppress the development of anthracnose and enhance defence related and antioxidant enzyme activities in avocado fruit. Postharvest Biology and Technology, 81, 66–72.
Shafiee, M., Taghavi, T. S., & Babalar, M. (2010). Addition of salicylic acid to nutrient solution combined with postharvest treatments (hot water, salicylic acid, and calcium dipping) improved postharvest fruit quality of strawberry. Scientia Horticulturae, 124, 40–45.
Shao, X., Wang, H., Xu, F., & Cheng, S. (2013). Effects and possible mechanisms of tea tree oil vapor treatment on the main disease in postharvest strawberry fruit. Postharvest Biology and Technology, 77, 94–101.
Shi, F., Zhao, J. H., Liu, Y., Wang, Z., Zhang, Y. T., & Feng, N. P. (2012). Preparation and characterization of solid lipid nanoparticles loaded with frankincense and myrrh oil. International Journal of Nanomedicine, 7, 2033–2043.
Singh, G., Maurya, S., & Catalan, C. A. (2007). A comparison of chemical, antioxidant and antimicrobial studies of cinnamon leaf and bark volatile oils, oleoresins and their constituents. Food and Chemical Toxicology, 45, 1650–1661.
Sophia, O., Robert, G. M., & Ngwela, W. J. (2014). Effect of Aloe vera gel coating on postharvest quality and shelf life of mango (Mangifera indica L.) fruits Var.‘Ngowe’. Journal of Horticulture and Forestry, 7(1), 1–7.
Soylu, E. M., Kurt, Ş, & Soylu, S. (2010). In vitro and in vivo antifungal activities of the essential oils of various plants against tomato grey mould disease agent Botrytis cinerea. International Journal of Food Microbiology, 143, 183–189.
Tao, N., Jia, L., & Zhou, H. (2014). Anti-fungal activity of Citrus reticulata Blanco essential oil against Penicillium italicum and Penicillium digitatum. Food Chemistry, 153, 265–271.
Tiyaboonchai, W., Tungpradit, W., & Plianbangchang, P. (2007). Formulation and characterization of curcuminoids loaded solid lipid nanoparticles. International Journal of Pharmaceutics, 337, 299–306.
Tzortzakis, N. G. (2009). Impact of cinnamon oil-enrichment on microbial spoilage of fresh produce. Innovative Food Science and Emerging Technologies, 10, 97–102.
Wang, K., Jin, P., Cao, S., Rui, H., & Zheng, Y. (2011). Biological control of green mould decay in postharvest Chinese bayberries by Pichia membranaefaciens. Journal of Phytopathology, 159, 417–423.
Wang, S. Y., Chen, P. F., & Chang, S. T. (2005). Antifungal activities of essential oils and their constituents from indigenous cinnamon (Cinnamomum osmophloeum) leaves against wood decay fungi. Bioresource Technology, 96, 813–818.
Wang, X. F., Zhang, S. L., Zhu, L. Y., Xie, S. Y., Dong, Z., Wang, Y., & Zhou, W. Z. (2012). Enhancement of antibacterial activity of tilmicosin against Staphylococcus aureus by solid lipid nanoparticles in vitro and in vivo. Veterinary Journal, 191, 115–120.
Win, N. K. K., Jitareerat, P., Kanlayanarat, S., & Sangchote, S. (2007). Effects of cinnamon extract, chitosan coating, hot water treatment and their combinations on crown rot disease and quality of banana fruit. Postharvest Biology and Technology, 45, 333–340.
Xing, Y., Li, X., Xu, Q., Yun, J., Lu, Y., & Tang, Y. (2011). Effects of chitosan coating enriched with cinnamon oil on qualitative properties of sweet pepper (Capsicum annuum L.). Food Chemistry, 124, 1443–1450.
Xing, Y., Xu, Q., Li, X., Che, Z., & Yun, J. (2012). Antifungal activities of clove oil against Rhizopus nigricans, Aspergillus flavus and Penicillium citrinum in vitro and in wounded fruit test. Journal of Food Safety, 32, 84-93.
Xu, J., Zhou, L., Miao, J., Yu, W., Zou, L., Zhou, W., Liu, Ch., & Lui, W. (2020). Effect of cinnamon essential oil nanoemulsion combined with ascorbic acid on enzymatic browning of cloudy apple juice. Food and Bioprocess Technology, 13, 860–870.
Xu, Q., Che, Z., Li, X., & Li, W. (2011). Effects of chitosan-oil coating on blue mold disease and quality attributes of jujube fruits. Food and Function, 2, 466–474.
Xu, W. T., Huang, K. L., Guo, F., Qu, W., Yang, J. J., Liang, Z. H., & Luo, Y. B. (2007). Postharvest grapefruit seed extract and chitosan treatments of table grapes to control Botrytis cinerea. Postharvest Biology and Technology, 46, 86–94.
Yin, Ch., Huang, Ch., Wang, J., Liu, Y., Lu, P., & Huang, L. (2019). Effect of chitosan- and alginate-based coatings enriched with cinnamon essential oil microcapsules to improve the postharvest quality of mangoes. Materials, 12, 2039.
Zapata, P. J., Guillén, F., Martínez-Romero, D., Castillo, S., Valero, D., & Serrano, M. (2008). Use of alginate or zein as edible coatings to delay postharvest ripening process and to maintain tomato (Solanum lycopersicon Mill) quality. Journal of the Science of Food and Agriculture, 88, 1287–1293.
Zhu, R., Lu, L., Guo, J., Lu, H., Abudureheman, N., & Yu, T. (2013). Postharvest control of green mold decay of citrus fruit using combined treatment with sodium bicarbonate and Rhodosporidium paludigenum. Food and Bioprocess Technology, 6, 2925–2930.
Funding
We would like to thank Islamic Azad University of Yasooj for the financial support of this research.
Author information
Authors and Affiliations
Contributions
Mohsen Radi: Conceptualization, Methodology, and Writing — original draft and editing. Hanieh Ahmadi: Methodology, Visualization, and Writing. Sedigheh Amiri: Conceptualization, Methodology, and Writing — original draft and editing.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Radi, M., Ahmadi, H. & Amiri, S. Effect of Cinnamon Essential Oil-Loaded Nanostructured Lipid Carriers (NLC) Against Penicillium Citrinum and Penicillium Expansum Involved in Tangerine Decay. Food Bioprocess Technol 15, 306–318 (2022). https://doi.org/10.1007/s11947-021-02737-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11947-021-02737-5