Abstract
Purpose of review
Routine screening for colorectal cancer (CRC) in adults > 50 years of age has led to overall reductions in CRC incidence and CRC-related mortality. Yet CRC incidence among young adults age < 50 continues to increase without a clear explanation. This review examines the changing epidemiology of CRC and emerging evidence regarding the influence of genetic and lifestyle factors on risk for colorectal neoplasia.
Recent findings
Young-onset CRC (yCRC), defined as CRC diagnosed in individuals younger than age 50, is a heterogeneous disease. Approximately, one in every five individuals affected with yCRC carries a pathogenic germline variant in genes associated with predisposition to cancer. However, most have no clinically identifiable risk factors. Analyses of birth cohorts estimate CRC risk among millennials to be 2–4 times higher than their grandparents’, suggesting that changes in health behaviors and environmental factors are having an impact on CRC risk. Young individuals with CRC tend to be diagnosed at later stages and often present with metastatic disease. yCRC tumors arise predominantly in the distal colon and are more likely than older-onset tumors to exhibit microsatellite and chromosome stable (MACS) phenotypes. Although yCRC patients are more likely than their older counterparts to be treated with multimodality chemotherapy regimens, more aggressive treatments have not yielded measurable survival gains. Since one in ten new CRC diagnoses involve individuals age < 50, recent guidelines have proposed lowering the age for average risk CRC screening from 50 to 45; however, further studies are needed to evaluate testing strategies based on individuals’ age and risk.
Summary
Significant shifts in CRC epidemiology and diversity of tumor phenotypes support genetic and environmental factors as modifiers of cancer risk. Emerging data correlating tumor molecular features with outcomes justify further investigation into mechanisms of carcinogenesis to elucidate how specific factors (inherited and/or acquired) might stimulate young-onset colorectal neoplasia.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Colorectal cancer (CRC) is the third leading cause of cancer-related mortality in the USA [1]. Implementation of CRC screening in the mid-1990s has significantly decreased CRC incidence and mortality in individuals over the age of 50 [1,2,3]. Paradoxically, a measurable increase in CRC incidence among individuals under the age of 50 years has been observed, which can be traced back to 1990 [2, 4]. While a number of possible explanations for this trend have been suggested, a clear causative factor has yet to be elucidated. Since most individuals affected with yCRC would otherwise be categorized as average risk, some groups have recommended that screening be expanded to decrease the burden of yCRC. Understanding the reasons underlying the recent increase in yCRC is necessary to guide the development of effective interventions. Here, we review the changing epidemiology of yCRC, tumor molecular features, and potential explanations for the increasing incidence of colorectal neoplasia in young individuals, which impact approaches to yCRC treatment and prevention.
Epidemiology
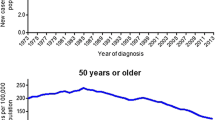
Since the implementation of routine screening for CRC in the 1990s, there have been significant and persistent overall decreases in both CRC incidence and mortality [1,2,3, 4, 5•, 6, 7]. Unfortunately, this trend pertains only to individuals over the age of 50, while CRC incidence among young individuals age < 50 continues to rise with annual increases approaching 1.4% for colon cancer and 2.4% in rectal cancer as measured from 2005 to 2014 [1]. The changing epidemiology of CRC is well illustrated by several studies analyzing data from the US Surveillance, Epidemiology, and End Results (SEER) program demonstrating that CRC incidence increased by 1.4% per year among individuals aged < 50 while decreasing by 3.1% per year among those > 50 years old [3, 5, 8, 9•, 10, 11]. Along with the estimated 51% relative increase in CRC incidence among individuals age < 50 [12••], there has also been an increase in yCRC mortality, up 13% from 2000 to 2014, which stands in stark contrast to the 34% decrease in CRC mortality observed among individuals age > 50 years [3]. Interestingly, the sharpest rise in CRC mortality is seen among young non-Hispanic White individuals [12••]. Although overall CRC mortality rates have declined over time, racial disparities persist with Blacks with yCRC experiencing worse outcomes compared to non-Hispanic Whites and Hispanics at every disease stage, but most pronounced among those with stage II colon cancer and stage III rectal cancer [13].
Approximately, 10% of incident CRCs are now diagnosed in individuals aged < 50 [3] with the largest burden occurring in those aged 40–49, accounting for 75% of the yCRC cases [9•, 14]. Comparison of CRC incidence patterns over time demonstrates a dramatic increase from 8.3 to 11.8 per 100,000 from 1990 to 2014, with age-specific incidence rates increasing in successive birth cohorts, supporting hypotheses that changes in lifestyle (e.g., diet, patterns of antibiotic use) are having an impact on CRC risk [4].
Clinical features of young-onset CRC
CRCs often present differently in young adults. Multiple studies have noted that a higher proportion of yCRCs arise in the sigmoid colon and rectum [3, 7, 15,16,17,18]. When compared to their older counterparts, young CRC patients tend to have a higher rate of red flag symptoms, such as rectal bleeding, obstruction, or abdominal pain [16, 19]. Failure to consider CRC as a potential diagnosis often delays the diagnostic process, with patients under the age of 50 years having a 1.4-fold increase in time to diagnosis than their older counterparts [20]. Consequently, a higher proportion of yCRCs present at a later stage [21, 22]. One multicenter retrospective study comparing CRC in patients 18–44 years and > 44 years found 61.2 and 44.5% had metastatic disease at diagnosis [23•]. It has been suggested that tumors in patients under 40 years old seem to have more aggressive histologic subtypes compared to their older counterparts, with 13 versus 1% of samples showing signet ring histology, 29 versus 11% showing perineural invasion, and 22 versus 6% showing venous invasion [24].
Molecular subtypes of CRC
Although data on molecular characteristics of yCRC tumors are limited, reports suggest that there are underlying differences in the biology of older onset vs yCRC.
CRCs, as a whole, encompass a heterogenous group of cancers with the majority of CRCs falling into a limited number of molecular subtypes. While a comprehensive review of the molecular subtypes of CRC is not provided here, it can be referenced elsewhere [25,26,27]. Briefly, of the three main molecular subtypes, the majority of CRCs exhibit the chromosomal instability (CIN) subtype, which typically displays chromosomal aneuploidy and a stepwise accumulation of mutations in specific genes including APC, KRAS, TP53, and BRAF. The microsatellite instability (MIN) subtype is characterized by microsatellite instability (MSI) due to loss of DNA mismatch repair (MMR) and can arise sporadically or be associated with germline alterations in MMR genes (Lynch syndrome) resulting in hypermutated tumors. Finally, the CpG island methylator phenotype (CIMP) displays widespread methylation changes resulting in epigenetic silencing. These subtypes can additionally display some overlap and are not mutually exclusive, with a minority of CRCs exhibiting both microsatellite and chromosome stability (MACS).
Interestingly, preliminary studies suggest differences in the proportion of molecular tumor subtypes between older-onset and yCRC. Among older-onset CRCs, a majority of cases exhibit CIN (51%), while MACS (34%) and MSI (13%) account for only a minority. However, among yCRCs, most exhibit MACS (45%) and MSI (21%) phenotypes, with lower proportions of CIN subtypes (32%) [28]. The higher prevalence of MSI in the yCRC group has been attributed to the increased prevalence of Lynch syndrome, the most common of the hereditary colorectal cancer syndromes.
Although data are limited, the MACS subtype which appears to be more common in the yCRC group and also appears to be associated with worse disease-free survival [28, 29]. LINE-1 hypomethylation, a surrogate marker for global hypomethylation, has also been found to be more prevalent in yCRCs and is associated with poorer prognosis [30].
The differences in molecular subtypes in yCRC come with significant clinical implications. For example, CRCs exhibiting MMR deficiency typically have poor response to fluorouracil-based adjuvant chemotherapy [31]. Interestingly, however, patients with MMR-deficient tumors tend to exhibit better overall survival, attributed to the more immunogenic nature of MIN subtype [32]. Although assessment of tumor MSI phenotype is recommended for all CRCs with implications for treatment, prognosis, and for diagnosing Lynch syndrome, in clinical practice, MSI testing is performed in only a minority of yCRC patients [10].
Treatment of CRC in young patients
Evidence regarding outcomes and effectiveness of specific treatment regimens in yCRC remains scarce. Currently, no evidence-based age-specific treatment regimens exist for CRC, though some recent reports do suggest differences in treatment outcomes between yCRC and older-onset CRC.
Several studies have reported that young patients are more likely to be treated with adjuvant chemotherapy regardless of the presence of distant metastases [33]. Kneuertz et al. investigated a nationwide cohort of CRC cases diagnosed age 18–49 years and found that compared to those age 65–75 years, younger patients were more likely to receive systemic chemotherapy and these more intensive regimens were not associated with increased survival in stage I or stage II cancers and offered only marginal benefit in stage III or stage IV diseases [34•]. Similarly, Manjelievskaia et al. investigated the differences in chemotherapy use and outcomes in colon cancer patients in ages 18–49 (young), 50–64 (middle-aged), and 65–75 (older) and found that young and middle-aged patients were more likely to receive adjuvant chemotherapy but did not show a significant improvement in survival compared to their older counterparts [35••]. Based on these retrospective observational studies, it is unclear whether current clinical practice tends toward overtreatment of yCRC patients compared to their older counterparts (with marginal benefit) or whether the underlying biology of yCRCs may be more aggressive, requiring more aggressive treatment to match outcomes. Currently, no guidelines recommend modifying CRC treatments based on young age and additional prospective studies and randomized clinical trials are needed to determine the optimal treatment regiments for younger patients.
Future directions regarding specific treatment options are currently being explored. Recent observations also highlight a significant difference in tumor mutation rates involved in histone methylation and demethylation which play an important role in CRC pathogenesis [36, 37]. With a better understanding of the molecular differences between yCRC and their older counterparts, it may be possible to exploit specific molecular alterations for the treatment of yCRC.
The role of genetic predisposition syndromes
It is important to consider the possibility of an underlying genetic predisposition to cancer in every individual affected with yCRC. Next-generation sequencing multigene panel genetic tests identify pathogenic germline variants in 16–20% of yCRC cases, involving genes associated with high and moderate penetrance cancer syndromes (Table 1) [38, 41••]. Lynch syndrome and familial adenomatous polyposis are the most prevalent syndromes, associated with early onset of colorectal neoplasia requiring colonoscopy beginning at age 20–25 and 10–12, respectively [39]. However, the variability in clinical presentations and potential for phenotypic overlap justifies the recommendation for genetic testing using a multigene panel for all individuals with yCRC [40].
Whereas earlier reports suggested genetic testing would be low yield in the absence of family history of CRC, genetic testing uncovers genetic diagnoses in approximately one in every five yCRC cases, with only half of germline mutation carriers reporting a family history of CRC [41••]. Making the diagnosis of a hereditary cancer syndrome can have an impact on surgical approaches (e.g., subtotal colectomy instead of segmental resection), selection of chemotherapy regimen, colonoscopy surveillance intervals, and surveillance for extracolonic cancers [39]. Furthermore, predictive genetic testing can be used to identify at-risk relatives who would benefit from early colonoscopy.
Possible explanations for the rising incidence of yCRC
The underlying cause(s) for the rise in incidence of yCRC remains elusive. A number of theories have been put forth which invoke both genetic and lifestyle factors. Although germline genetic alterations can be implicated in one in five individuals with yCRC, hereditary syndromes account for only a minority of cases. Since the prevalence of pathogenic variants in a population does not change significantly over time, genetic factors alone would not explain the recent increase in CRC incidence [16, 42]. Potential interactions between genetic risk alleles and comorbidities such as obesity [43, 44], diabetes [45], and lifestyle factors such as consumption of processed meat, alcohol, and tobacco use [42, 46, 47] are being explored through risk modeling [48]. Metabolic changes in insulin-like growth factor-1 (IGF-1), interleukin-6 (IL-6), and cyclooxygenase-2 (COX-2) have been associated with pro-carcinogenic effects [49]. A recent study investigated risk factors of CRC in young adults from ages 20–39 and found metabolic syndrome (obesity, elevated blood pressure, abdominal obesity), smoking status, and alcohol intake were independently associated with a higher risk of CRC, while tobacco use and alcohol intake were not significant risk factors in the very young age group (20–29) [50]. Birth cohort analyses of CRC incidence data suggest rates of yCRC had been decreasing just prior to the recent sharp increases with an inflection point in the mid-1990s, suggesting that recent changes in exposures that might alter the gastrointestinal microbiome (e.g., diet, antibiotic use) warrant further investigation [4].
Additional studies linking health behaviors with disease phenotypes will be important for assessing the potential influence of modifiable risk factors on CRC risk and developing novel approaches to CRC treatment and prevention.
Strategies for primary and secondary CRC prevention in young adults
In this review, we define yCRC as cases diagnosed at age < 50. While most reports in the literature use a similar definition, this age cutoff is artificial, based primarily on the current age at which it has been recommended for average-risk adults to begin CRC screening [51, 52]. Individuals who are above average risk for CRC (due to family history of colorectal neoplasia or personal history of inflammatory bowel disease) are recommended to commence CRC screening earlier [51]. The American College of Gastroenterology stood out from peer organizations by recommending earlier screening for Black individuals starting at age 45 [53] based on observed racial disparities in CRC incidence and outcomes [9•, 10].
Current CRC screening recommendations were based on CRC incidence data from 1975 to 1979 and do not account for the more recent increases in yCRC incidence. A recent modeling analysis adjusted CRC screening models using updated incidence data [54••] concluded that in order to maintain a similar burden to benefit ratio as previous recommendations, average risk screening should start at age 45 rather than 50. Based on this, the American Cancer Society issued an updated guideline with qualified recommendation to lower the age for average-risk screening to 45 [12••]. This change has generated considerable debate given the paucity of data about the effectiveness of the various CRC screening tests (endoscopic vs stool-based) in younger patients [55]. A recent analysis concluded that screening individuals age 45–49 years would prevent 900 CRC-related deaths at a cost of $5.5 billion [56].
There is a clear need for more effective ways to stratify individuals’ CRC risk. However, a significant proportion of yCRC cases could be prevented by identifying individuals eligible for earlier screening by current guidelines. Clinicians should review family history of cancer that includes all diagnoses in first and second-degree relatives to determine whether a patient’s risk for colorectal neoplasia is average, moderate, or high. In addition, the use of genetic risk models (e.g., PREMM5 www.premm.dfci.harvard.edu) can be helpful for identifying individuals who meet criteria for genetic testing [57].
Conclusions
Incidence of CRC has been increasing among individuals age < 50 for unclear reasons. Existing data suggest that yCRC patients represent a heterogeneous group. Young-onset tumors differ from older-onset CRCs in their clinical characteristics, pathologic features, molecular profiles, and potentially, response to treatment. Additional studies are needed to elucidate the etiology of the rising incidence of yCRC in order to determine how best to implement screening based on risk and target treatments to the molecular mechanisms which drive colorectal neoplasia in young adults.
References and Recommended Readings
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Cancer facts & figures 2018. Atlanta, Georgia: American Cancer Society2018. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-andstatistics/annual-cancer-facts-and-figures/2018/cancer-facts-and-figures-2018.pdf.
Colorectal cancer facts & figures 2017-2019. Atlanta, Georgia: American Cancer Society2017. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-andstatistics/colorectal-cancer-facts-and-figures/colorectal-cancer-facts-and-figures-2017-2019.pdf.
Siegel RL, Miller KD, Fedewa SA, Ahnen DJ, Meester RGS, Barzi A, et al. Colorectal cancer statistics, 2017. CA Cancer J Clin. 2017;67(3):177–93. https://doi.org/10.3322/caac.21395.
Murphy CC, Singal AG, Baron JA, Sandler RS. Decrease in incidence of young-onset colorectal cancer before recent increase. Gastroenterology. 2018;155:1716–1719.e4. https://doi.org/10.1053/j.gastro.2018.07.045.
• Siegel RL, Fedewa SA, Anderson WF, Miller KD, Ma J, Rosenberg PS, et al. Colorectal cancer incidence patterns in the United States, 1974-2013. J Natl Cancer Inst. 2017;109(8):djw322. https://doi.org/10.1093/jnci/djw322 This analysis highlights the significant differences in CRC incidence across age groups as well as annual percentage change in incidence by age.
Bhandari A, Woodhouse M, Gupta S. Colorectal cancer is a leading cause of cancer incidence and mortality among adults younger than 50 years in the USA: a SEER-based analysis with comparison to other young-onset cancers. J Investig Med. 2017;65(2):311–5. https://doi.org/10.1136/jim-2016-000229.
Sia CS, Paul E, Wale RJ, Lynch AC, Heriot AG, Warrier SK. No increase in colorectal cancer in patients under 50 years of age: a Victorian experience from the last decade. Color Dis. 2014;16(9):690–5. https://doi.org/10.1111/codi.12648.
Yeo H, Betel D, Abelson JS, Zheng XE, Yantiss R, Shah MA. Early-onset colorectal cancer is distinct from traditional colorectal cancer. Clin Colorectal Cancer. 2017;16(4):293–9 e6. https://doi.org/10.1016/j.clcc.2017.06.002.
• Murphy CC, Lund JL, Sandler RS. Young-onset colorectal cancer: earlier diagnoses or increasing disease burden? Gastroenterology. 2017;152(8):1809–12 e3. https://doi.org/10.1053/j.gastro.2017.04.030 This study investigates the association between use of colonoscopy over the past two decades and its correlation with the increased incidence of yCRC. It finds that while initial rates of colonoscopy were increasing in parallel with the increasing incidence of yCRC, these two trends diverge around 2009 implying that increasing colonoscopy use does not explain the increasing yCRC incidence.
Murphy CC, Sanoff HK, Stitzenberg KB, Baron JA, Lund JL, Sandler RS. Patterns of sociodemographic and clinicopathologic characteristics of stages II and III colorectal cancer patients by age: examining potential mechanisms of young-onset disease. J Cancer Epidemiol. 2017;2017:4024580–10. https://doi.org/10.1155/2017/4024580.
Siegel RL, Miller KD, Jemal A. Colorectal cancer mortality rates in adults aged 20 to 54 years in the United States, 1970-2014. JAMA. 2017;318(6):572–4. https://doi.org/10.1001/jama.2017.7630.
•• Wolf AMD, Fontham ETH, Church TR, Flowers CR, Guerra CE, LaMonte SJ, et al. Colorectal cancer screening for average-risk adults: 2018 guideline update from the American Cancer Society. CA Cancer J Clin. 2018;68(4):250–81. https://doi.org/10.3322/caac.21457 This guideline recommended lowering the CRC screening age for asymptomatic average risk individuals from 50 to 45, generating debate.
Holowatyj AN, Ruterbusch JJ, Rozek LS, Cote ML, Stoffel EM. Racial/ethnic disparities in survival among patients with young-onset colorectal cancer. J Clin Oncol. 2016;34(18):2148–56. https://doi.org/10.1200/JCO.2015.65.0994.
You YN, Xing Y, Feig BW, Chang GJ, Cormier JN. Young-onset colorectal cancer: is it time to pay attention? Arch Intern Med. 2012;172(3):287–9. https://doi.org/10.1001/archinternmed.2011.602.
Davis DM, Marcet JE, Frattini JC, Prather AD, Mateka JJ, Nfonsam VN. Is it time to lower the recommended screening age for colorectal cancer? J Am Coll Surg. 2011;213(3):352–61. https://doi.org/10.1016/j.jamcollsurg.2011.04.033.
Myers EA, Feingold DL, Forde KA, Arnell T, Jang JH, Whelan RL. Colorectal cancer in patients under 50 years of age: a retrospective analysis of two institutions’ experience. World J Gastroenterol. 2013;19(34):5651–7. https://doi.org/10.3748/wjg.v19.i34.5651.
Jones HG, Radwan R, Davies M, Evans M, Khot U, Chandrasekaran TV, et al. Clinicopathological characteristics of colorectal cancer presenting under the age of 50. Int J Color Dis. 2015;30(4):483–9. https://doi.org/10.1007/s00384-015-2166-1.
Segev L, Kalady MF, Church JM. Left-sided dominance of early-onset colorectal cancers: a rationale for screening flexible sigmoidoscopy in the young. Dis Colon Rectum. 2018;61(8):897–902. https://doi.org/10.1097/DCR.0000000000001062.
Riaz R, Masood N, Benish A. Red flag symptoms: detailed account of clinicopathological features in young-onset colorectal cancer. Intest Res. 2017;15(2):203–7. https://doi.org/10.5217/ir.2017.15.2.203.
Chen FW, Sundaram V, Chew TA, Ladabaum U. Advanced-stage colorectal cancer in persons younger than 50 years not associated with longer duration of symptoms or time to diagnosis. Clin Gastroenterol Hepatol. 2017;15(5):728–37. e3. https://doi.org/10.1016/j.cgh.2016.10.038.
Fu J, Yang J, Tan Y, Jiang M, Wen F, Huang Y, et al. Young patients (≤ 35 years old) with colorectal cancer have worse outcomes due to more advanced disease: a 30-year retrospective review. Medicine (Baltimore). 2014;93(23):e135. https://doi.org/10.1097/MD.0000000000000135.
Silla IO, Rueda D, Rodriguez Y, Garcia JL, de la Cruz VF, Perea J. Early-onset colorectal cancer: a separate subset of colorectal cancer. World J Gastroenterol. 2014;20(46):17288–96. https://doi.org/10.3748/wjg.v20.i46.17288.
• Rho YS, Gilabert M, Polom K, Aladashvili A, Kopeckova K, Megdanova V, et al. Comparing clinical characteristics and outcomes of young-onset and late-onset colorectal cancer: an international collaborative study. Clin Colorectal Cancer. 2017;16(4):334–42. https://doi.org/10.1016/j.clcc.2017.03.008 This international, multicenter analysis looked at differences in use of adjuvant treatments for yCRC.
Chang DT, Pai RK, Rybicki LA, Dimaio MA, Limaye M, Jayachandran P, et al. Clinicopathologic and molecular features of sporadic early-onset colorectal adenocarcinoma: an adenocarcinoma with frequent signet ring cell differentiation, rectal and sigmoid involvement, and adverse morphologic features. Mod Pathol. 2012;25(8):1128–39. https://doi.org/10.1038/modpathol.2012.61.
Fearon ER. Molecular genetics of colorectal cancer. Annu Rev Pathol. 2011;6:479–507. https://doi.org/10.1146/annurev-pathol-011110-130235.
Rodriguez-Salas N, Dominguez G, Barderas R, Mendiola M, Garcia-Albeniz X, Maurel J, et al. Clinical relevance of colorectal cancer molecular subtypes. Crit Rev Oncol Hematol. 2017;109:9–19. https://doi.org/10.1016/j.critrevonc.2016.11.007.
Cancer Genome Atlas N. Comprehensive molecular characterization of human colon and rectal cancer. Nature. 2012;487(7407):330–7. https://doi.org/10.1038/nature11252.
Ballester V, Rashtak S, Boardman L. Clinical and molecular features of young-onset colorectal cancer. World J Gastroenterol. 2016;22(5):1736–44. https://doi.org/10.3748/wjg.v22.i5.1736.
Banerjea A, Hands RE, Powar MP, Bustin SA, Dorudi S. Microsatellite and chromosomal stable colorectal cancers demonstrate poor immunogenicity and early disease recurrence. Color Dis. 2009;11(6):601–8. https://doi.org/10.1111/j.1463-1318.2008.01639.x.
Antelo M, Balaguer F, Shia J, Shen Y, Hur K, Moreira L, et al. A high degree of LINE-1 hypomethylation is a unique feature of early-onset colorectal cancer. PLoS One. 2012;7(9):e45357. https://doi.org/10.1371/journal.pone.0045357.
Ribic CM, Sargent DJ, Moore MJ, Thibodeau SN, French AJ, Goldberg RM, et al. Tumor microsatellite-instability status as a predictor of benefit from fluorouracil-based adjuvant chemotherapy for colon cancer. N Engl J Med. 2003;349(3):247–57. https://doi.org/10.1056/NEJMoa022289.
Li P, Xiao ZT, Braciak TA, Ou QJ, Chen G, Oduncu FS. Impact of age and mismatch repair status on survival in colorectal cancer. Cancer Med. 2017;6(5):975–81. https://doi.org/10.1002/cam4.1007.
Schellerer VS, Merkel S, Schumann SC, Schlabrakowski A, Fortsch T, Schildberg C, et al. Despite aggressive histopathology survival is not impaired in young patients with colorectal cancer : CRC in patients under 50 years of age. Int J Color Dis. 2012;27(1):71–9. https://doi.org/10.1007/s00384-011-1291-8.
• Kneuertz PJ, Chang GJ, Hu CY, Rodriguez-Bigas MA, Eng C, Vilar E, et al. Overtreatment of young adults with colon cancer: more intense treatments with unmatched survival gains. JAMA Surg. 2015;150(5):402–9. https://doi.org/10.1001/jamasurg.2014.3572 Analysis of hospital system data found yCRC patients were significantly more likely to receive high intensity multimodality chemotherapy without associated surival gains.
•• Manjelievskaia J, Brown D, Mc Glynn KA, Anderson W, Shriver CD, Zhu K. Chemotherapy use and survival among young and middle-aged patients with colon cancer. JAMA Surg. 2017;152(5):452–9. https://doi.org/10.1001/jamasurg.2016.5050 This study highlights differences in the treatment approaches for yCRC. It describes an increase in use of adjuvant chemotherapy for yCRC patients compared to oldercounterparts with no differences in outcomes. The authors propose a potential overuse of chemotherapy among young patients with CRC.
Puccini A, Lenz HJ, Marshall JL, Arguello D, Raghavan D, Korn WM, Weinberg BA, Poorman K, Heeke AL, Philip PA, Shields AF, Goldberg RM, Salem ME Impact of patient age on molecular alterations of left-sided colorectal tumors. The Oncologist 2018. https://doi.org/10.1634/theoncologist.2018-0117, theoncologist.2018, theoncologist.0117.
Gargalionis AN, Piperi C, Adamopoulos C, Papavassiliou AG. Histone modifications as a pathogenic mechanism of colorectal tumorigenesis. Int J Biochem Cell Biol. 2012;44(8):1276–89. https://doi.org/10.1016/j.biocel.2012.05.002.
Pearlman R, Frankel WL, Swanson B, Zhao W, Yilmaz A, Miller K, et al. Prevalence and spectrum of germline cancer susceptibility gene mutations among patients with early-onset colorectal Cancer. JAMA Oncol. 2017;3(4):464–71. https://doi.org/10.1001/jamaoncol.2016.5194.
Syngal S, Brand RE, Church JM, Giardiello FM, Hampel HL, Burt RW. American College of Gastroenterology ACG clinical guideline: genetic testing and management of hereditary gastrointestinal cancer syndromes. Am J Gastroenterol. 2015;110(2):223–62; quiz 63. https://doi.org/10.1038/ajg.2014.435.
Genetic/familial high-risk assessment: Colorectal. In: NCCN Clinical Practice Guidelines in Oncology. 2018. https://www.nccn.org/professionals/physician_gls/pdf/genetics_colon.pdf. Accessed 11/3/2018 2018.
•• Stoffel EM, Koeppe E, Everett J, Ulintz P, Kiel M, Osborne J, et al. Germline genetic features of young individuals with colorectal cancer. Gastroenterology. 2018;154(4):897–905 e1. https://doi.org/10.1053/j.gastro.2017.11.004 This analysis of outcomes of germline sequencing in patients referred to a cancer genetics clinic found 1 in 5 yCRC patients had pathogenic germline variants in common CRC genes, many of whom reported no family history of CRC.
Rosato V, Bosetti C, Levi F, Polesel J, Zucchetto A, Negri E, et al. Risk factors for young-onset colorectal cancer. Cancer Causes Control. 2013;24(2):335–41. https://doi.org/10.1007/s10552-012-0119-3.
Hidayat K, Yang CM, Shi BM. Body fatness at an early age and risk of colorectal cancer. Int J Cancer. 2018;142(4):729–40. https://doi.org/10.1002/ijc.31100.
Botma A, Nagengast FM, Braem MG, Hendriks JC, Kleibeuker JH, Vasen HF, et al. Body mass index increases risk of colorectal adenomas in men with Lynch syndrome: the GEOLynch cohort study. J Clin Oncol. 2010;28(28):4346–53. https://doi.org/10.1200/JCO.2010.28.0453.
de Kort S, Masclee AAM, Sanduleanu S, Weijenberg MP, van Herk-Sukel MPP, Oldenhof NJJ, et al. Higher risk of colorectal cancer in patients with newly diagnosed diabetes mellitus before the age of colorectal cancer screening initiation. Sci Rep. 2017;7:46527. https://doi.org/10.1038/srep46527.
Cavestro GM, Mannucci A, Zuppardo RA, Di Leo M, Stoffel E, Tonon G. Early onset sporadic colorectal cancer: worrisome trends and oncogenic features. Dig Liver Dis. 2018;50(6):521–32. https://doi.org/10.1016/j.dld.2018.02.009.
Botma A, Vasen HF, van Duijnhoven FJ, Kleibeuker JH, Nagengast FM, Kampman E. Dietary patterns and colorectal adenomas in Lynch syndrome: the GEOLynch cohort study. Cancer. 2013;119(3):512–21. https://doi.org/10.1002/cncr.27726.
Hsu L, Jeon J, Brenner H, Gruber SB, Schoen RE, Berndt SI, et al. A model to determine colorectal cancer risk using common genetic susceptibility loci. Gastroenterology. 2015;148(7):1330–9. e14. https://doi.org/10.1053/j.gastro.2015.02.010.
Zheng J, Zhao M, Li J, Lou G, Yuan Y, Bu S, et al. Obesity-associated digestive cancers: a review of mechanisms and interventions. Tumour Biol. 2017;39(3):1010428317695020. https://doi.org/10.1177/1010428317695020.
Kim NH, Jung YS, Yang HJ, Park SK, Park JH, Park DI, et al. Prevalence of and risk factors for colorectal neoplasia in asymptomatic young adults (20-39 years old). Clin Gastroenterol Hepatol. 2018;17:115–22. https://doi.org/10.1016/j.cgh.2018.07.011.
Rex DK, Boland CR, Dominitz JA, Giardiello FM, Johnson DA, Kaltenbach T, et al. Colorectal cancer screening: recommendations for physicians and patients from the U.S. multi-society task force on colorectal cancer. Gastroenterology. 2017;153(1):307–23. https://doi.org/10.1053/j.gastro.2017.05.013.
Bibbins-Domingo K, Grossman DC, Curry SJ, Davidson KW, Epling JW Jr, Garcia FAR, et al. Screening for colorectal cancer: US preventive services task force recommendation statement. JAMA. 2016;315(23):2564–75. https://doi.org/10.1001/jama.2016.5989.
Rex DK, Johnson DA, Anderson JC, Schoenfeld PS, Burke CA, Inadomi JM, et al. American College of Gastroenterology guidelines for colorectal cancer screening 2009 [corrected]. Am J Gastroenterol. 2009;104(3):739–50. https://doi.org/10.1038/ajg.2009.104.
•• Peterse EFP, Meester RGS, Siegel RL, Chen JC, Dwyer A, Ahnen DJ, et al. The impact of the rising colorectal cancer incidence in young adults on the optimal age to start screening: microsimulation analysis I to inform the American Cancer Society colorectal cancer screening guideline. Cancer. 2018;124(14):2964–73. https://doi.org/10.1002/cncr.31543 This analysis incoorporates contemporary CRC incidence data to model outcomes of CRC screening to justify offering screening for CRC at an earlier age.
Corley DA, Peek RM Jr. When should guidelines change? A clarion call for evidence regarding the benefits and risks of screening for colorectal cancer at earlier ages. Gastroenterology. 2018;155(4):947–9. https://doi.org/10.1053/j.gastro.2018.08.040.
Bretthauer M, Kalager M, Weinberg DS. From colorectal cancer screening guidelines to headlines: beware! Ann Intern Med. 2018;169(6):405–6. https://doi.org/10.7326/M18-1720.
Kastrinos F, Uno H, Ukaegbu C, Alvero C, McFarland A, Yurgelun MB, et al. Development and validation of the PREMM5 model for comprehensive risk assessment of Lynch syndrome. J Clin Oncol. 2017;35(19):2165–72. https://doi.org/10.1200/JCO.2016.69.6120.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Anand Venugopal declares that he has no conflict of interest.
Elena Stoffel declares that she has no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Colon
Rights and permissions
About this article
Cite this article
Venugopal, A., Stoffel, E.M. Colorectal Cancer in Young Adults. Curr Treat Options Gastro 17, 89–98 (2019). https://doi.org/10.1007/s11938-019-00219-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11938-019-00219-4