Abstract
Purpose of Review
Myocardial infarction, which is the most important clinical sign of coronary artery disease, is one of the leading causes of global mortality, despite current treatment methods. Stem cell transplantation, which fastens on the regeneration of damaged tissue, has been suggested as an alternative approach in cardiac regenerative medicine; however, complications such as low survival in ischemic conditions, immune rejection, and teratoma formation have limited the routine use of stem cells in clinical treatments.
Recent Findings
Exosomes, which have been shown to play an essential role in intracellular communication and carry protein, lipid, and nucleic acid-based rich cargo content, have emerged as a new potential in the diagnosis and treatment of many diseases such as neurodegenerative diseases, cancer, and cardiovascular diseases in recent years. First findings have brought into the open that the exosomes secreted by local cells in the myocardium layer and stem cells have an essential role in the repair of cardiac damage because they involve pro-angiogenic, pro-survival, anti-fibrotic, or anti-apoptotic molecules.
Conclusion
This review has comprehensively discussed the rich cargo content of somatic and stem cell-derived exosomes, their regulatory mechanisms in amelioration of the pathophysiology of coronary artery disease, molecular interactions of exosomal cargo contents, strengths, and limitations of exosomal strategies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Opinion statement
Exosome-based therapeutic strategies hold great promise in addressing limitations such as low survival rate of cells, teratoma formation, and immune rejection, especially when compared to traditional stem cell therapies. Research on cardiac tissue repair using exosomes derived from various cell types has underscored the significance of the source cell in determining exosomal cargo content, which indirectly impacts repair efficiency. The precise definition of exosomal cargo is essential, and modifications may be necessary to ensure cargo stability. Stem cell-derived exosomes, in particular, offer favorable platforms for cardiac repair compared to somatic cells. However, the variability in cargo content among stem cells calls for fine-tuned approaches. Fortunately, advancements in molecular sciences provide methods for manipulating stem cell-derived exosomal cargo, such as extrinsic conditioning or intrinsic genome editing. On the other hand, maintaining the stability of exosomes under circulation conditions and their activity in specific regions is another important challenge. One promising solution involves conjugating exosomes with markers that facilitate targeted cellular delivery. While our understanding of exosome roles and cargo content in cardiac repair has significantly advanced in the past decade, comprehensive approaches are still under development. Clinical applications will ultimately depend on large-scale and thorough analyses of exosomes with diverse cargo content in therapeutic contexts.
Introduction
Cardiovascular diseases with a high prevalence are the leading cause of mortality and morbidity all over the world. According to the data of the Global Burden of Disease study, approximately 20.5 million people died due to cardiovascular diseases in 2021, and approximately 9.44 million of these deaths were due to coronary artery disease (CAD) [1, 2].
Myocardial infarction (MI), characterized as cardiac necrosis due to prolonged ischemia, is the most important clinical sign of CAD [3]. Current treatments aim to reopen occluded arteries and restore blood flow to the heart but do not ensure effective repair of the damaged myocardial tissue. Stem cell transplantation, a promising approach for tissue repair, has encountered limitations, including low survival rate of cells in ischemic conditions, issues specific to certain stem cell types like teratoma formation, limited resources, and immune rejection [4, 5]. Initially, early transplantation studies suggested that transplanted stem cells directly differentiated into cardiac cells, providing therapeutic benefits. However, recent research into the mechanisms of stem cell therapy in cardiac repair has revealed that these therapeutic effects primarily result from paracrine factors and their intercellular transfer via exosomes [6,7,8]. In this review, the contents of exosomes released from cells in the cardiac tissue microenvironment and stem cell-derived exosomes that can be used for therapeutic intervention in order to reverse the pathophysiological process of CAD and improve the prognosis of patients have been discussed in detail, and new strategies for future studies have been pointed out.
Extracellular vesicles (EVs)
Intercellular cross-talk is one of the most prominent properties of multicellular organisms. Many molecules that control mechanisms such as cell migration, proliferation, differentiation, and apoptosis are transmitted by direct cell–cell connections or by the transfer of secreted molecules [9]. Recently, EV has been proposed as an additional cell–cell cross-talk mechanism in which cells change their proteins, lipids, and genetic material [10, 11]. EVs are cell-derived membranous structures with a phospholipid bilayer that arise directly or indirectly from the plasma membrane [12]. They can be classified differently according to their size, the cargo contents they carry, and their formation from the plasma membrane.
Ectosomes (shedding microvesicles) are EVs of 50–2000 nm in size, containing protein and lipid, that is secreted out of the cell by budding directly from the plasma membrane [13]. The molecular composition of ectosomes differs according to the cell type.
Apoptotic bodies (apoptosomes) are heterogeneous vesicle structures with sizes between 50 and 5000 nm, which are formed as a result of apoptosis of cells, known as programmed cell death, by condensation and budding of the contents in a region close to the membrane [14].
Retro-virus-like particles (RLP), whose origins continue to be an active area of research, are EVs with a size of 90–100 nm, which are thought to have a different dynamic than microvesicles, although they are formed by budding from the plasma membrane. Some studies have suggested that these particles are composed of transcription of human endogenous retrovirus sequences [14, 15].
Smallest known active group of EVs: exosomes
Exosomes are structures that vary between 30 and 100 nm in size and are formed indirectly from the plasma membrane [16]. In 1983, these vesicles released out of the cell were identified during the studies on the loss of transferrin from reticulocytes during the maturation of reticulocytes to erythrocytes [17, 18], and in 1987, these small extruded vesicles were named “exosomes” [19]. Exosomes, which have many physiological functions in the body, can be found in many fluids, such as blood, urine, saliva, amniotic fluid, synovial fluid, nasal secretions, and breast milk, and can be named dexosome (dendritic cell-derived exosomes) [20], oncosome/texosome (cancer cell-derived exosomes) [21], and prostasome (prostate cancer cell-derived exosomes) [22] according to the cell types they originate from [23].
The interest in these vesicles, which have been proven to exist about 30 years ago and are thought to be primarily waste shedding from the plasma membrane, is increasing day by day. The reason for this is the realization that these vesicles, which are found in many biological fluids, can be used as new tools in the diagnosis, progression, and treatment of many diseases such as cancer, neurodegenerative diseases, and cardiovascular diseases, thanks to the cargo contents they carry. Recent studies have shown that some of the cargo contents carried by exosomes are specific to the cell type they originate from, can change in disease and health conditions, and some are invariable parts of exosomes. In general terms, cargo contents; integrins, immunoglobulins, many adhesion proteins, cytoskeletal proteins (actin, cytokeratin, myosin, tubulin), endosomal separation complex proteins required for transport (ESCRT) (alix, TSG101, vps-28, vps-22), heat-shock proteins (Hsp70, Hsp90), CD9, CD81, and CD63 involved in endosomal vesicle traffic, Annexin and Rab protein family, which regulate fusion events and cytoskeleton, apart from these, they consist of products such as nucleic acids, mRNA, microRNA (miRNA), circular RNA (circRNA), long non-coding RNA (lncRNA) and can protect these products from the extracellular environment during transport [24, 25].
In order to be recognized by the cells they target, exosomes carry membrane lipids such as serine membrane lipids, cholesterol, sphingolipid, and sphingomyelin, as well as transmembrane proteins, tetraspanins, and receptor molecules found in the plasma membrane of the cell they originate from [26]. It is thought that these multi-molecule messengers, which act in autocrine and paracrine signaling, can be taken into the cell by three known mechanisms, although they have not been fully elucidated:
-
1.
Exosomes can fuse with the plasma membrane of the target cell and transfer their contents to the cell (direct fusion) [27].
-
2.
Cellular internalization of exosomes can occur by an actin-cytoskeleton and phosphatidylinositol 3-kinase-dependent phagocytosis [28].
-
3.
Proteases in the plasma membrane of the target cell are associated with the exosome membrane proteins (receptor-ligand association) [29]. Thus, the cargo in the exosome is released and the contents are taken into the cell lumen depending on the selectively permeable membrane.
Role and function of exosomes in the pathophysiology of CAD
The mammalian heart includes cardiomyocytes (CMs), endothelial cells (ECs), fibroblasts (FBs), immune cells such as macrophage, DC, and T lymphocyte, telocytes (TCs), and CSCs whose presence has recently been proven [30]. A well-organized and efficient cross-talk between these cell types is essential for the heart, the main organ of the cardiovascular system, to maintain its integrity and pump the blood needed by the body.
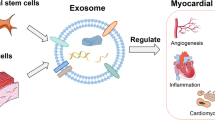
Accumulating evidence indicates that exosomes, defined as multimolecular messengers with autocrine and paracrine effects, also play a significant role in cardiac cell–cell cross-talk (Figs. 1 and 2) [31, 32]. The cargo contents of these exosomes, which vary according to the cell types they originate from, may also vary depending on the normal or pathological condition of the cell [24, 33]. Therefore, the contents and roles of exosomes released from local cells in the myocardial layer and stem cell-derived exosomes to be used for therapeutic intervention should be well understood in order to prevent developing pathophysiological processes and improve the prognosis of patients.
Somatic cells-derived exosomes
CMs-derived exosomes
Exosomes derived from CMs have a bilayer membrane structure, contain the markers caveolin 3 and flotilin 1 on their surface, and enable cross-talk with other cells in the heart (Fig. 1) [34]. Studies have shown that cargo contents consist of various proteins, enzymes, and their inhibitors and that these contents can change under various stress factors (hypoxia, glucose deprivation, alcohol, etc.) [35,36,37,38,39].
Angiogenesis is of vital importance to meet the nutrient and oxygen demand of the myocardial layer and to maintain myocardial contraction under ischemic conditions [40]. The interaction and underlying mechanism between ECs, which play an active role in this important process, and exosomes released from CMs have been subjected to many studies (Fig. 1) [38, 41,42,43]. The first study, it has been showed that the relatively most abundant miRNAs in exosomes released from ischemic CMs are miR-222 and miR-143 and that these exosomes support the proliferation of ECs, stimulate the formation of capillary-like structures, and strengthen adhesion complexes [38]. Other miRNAs whose levels increase in CM-derived exosomes under stress have been identified as miR-200c-3p and miR-19a-3p [41, 42]. Both in vitro and in vivo, CMs containing these miRNAs have been shown to cross-talk with ECs, suppress the proliferation of ECs, and reduce their angiogenic capacity. In the in vitro study, the use of GW4869, an exosome release inhibitor, in culture, reversed this effect on ECs [41], confirming that cross-talk between cells occurs in the direction of an exosomal signal. Another recent study has researched the presence and mechanism of miR-29a in CMs-derived exosomes [43], which has been previously reported to play an important role in regulating angiogenesis and cardiac hypertrophy [44]. This study on cardiac microvascular ECs (CMECs) has shown that the target gene of exosomal miR-29a is VEGFA and that it inhibits the angiogenic capacity of ECs by inhibiting the expression of this gene. It is known that one of the cargo contents of exosomes derived from CMs is circRNAs, one of the classes of non-coding RNAs. A recent study showed that silencing of exosomal circHIPK3 released from hypoxic CMs resulted in increased apoptosis in CMECs [45]. circHIPK3 acts as an endogenous miR-29a sponge to inhibit miR-29a activity. Further analysis by the researchers has revealed that this circRNA first inhibits miR-29a activity, leading to increased VEGFA expression and subsequently promoting the proliferation and migration of cardiac ECs [46].
Autophagy and apoptosis are two different types of cell death regulated by genes, especially in cardiac cells under ischemic conditions [47]. In studies on exosomes derived from CMs, the effects of cargo contents of exosomes on these two mechanisms have also been identified [35, 45, 46, 48,49,50,51,52]. Hsps are defined as proteins whose amount increases in the cell in which they are located during stress. Extracellular Hsp60 activates toll-like receptor-4 (TLR-4), causing apoptosis of CMs and myocardial inflammation [48]. On the other hand, it has been shown that CM-derived exosomal Hsp20 activates the AKT signaling pathway that supports survival, suppresses TNF-α and IL-1β factors, and alleviates MI by reducing the myocardial apoptosis index [50]. In addition to its survival effect, CM-derived exosomal Hsp20 has also been shown to protect against cardiac reverse remodeling and increase left ventricular ejection fraction [53, 54]. Yang et al. have reported that exosomal miR-30a can be transferred between CMs via exosomes in hypoxic conditions and increased the expression of core autophagy regulators beclin-1, Atg12, and LC3II/LC3I that contribute to the maintenance of autophagic response in CMs [51].
One of the mechanisms of action of exosomes derived from CMs is cardiac fibrosis. It has been revealed that miR-208a is upregulated in CMs and transferred to FBs via exosomes. Inhibiting miR-208a in vivo, whose target gene was determined to be Dyrk2, which is involved in cell growth, reduced post-MI cardiac fibrosis, and improved cardiac functions in rats. Transfusion of exosomes containing miR-208a into normal rats has resulted in impaired cardiac functions [55]. miR-217 is another ncRNA that has a role in cardiac fibrosis. CM-derived exosomal miR-217 has been shown to increase the proliferation of FBs, with its in vivo overexpression exacerbating cardiac fibrosis, hypertrophy, and cardiac dysfunction [56]. Besides miRNAs, circular RNAs have also been found to play a role in cross-talk between CM-FB. CM-derived exosomal circ_0036176 has been shown to result from the re-splicing of exon 2 of the Myo9a gene into exon4, increase its number in the myocardium of patients with heart failure, and inhibit FB proliferation. Interestingly, circ_0036176 has been found to contain a 627 nt ORF encoding a 208 amino acid protein (Myo9a-208). Myo9a-208 has been shown to mediate the inhibitory effect of circ_0036176 on FB proliferation [57]. These results suggest that circ_0036176 may be a potential therapeutic target in cardiac fibrosis.
Under normal conditions, macrophages are present in the heart with the anti-inflammatory M2 phenotype. After MI, macrophages have been shown to be in the M1 phenotype and release cytokines and proteases that induce apoptosis of CMs [58]. Cross-talk with macrophages has been also demonstrated in studies of CM-derived exosomes (Fig. 1) [59,60,61,62]. When the underlying mechanism has been examined, it has been shown that CM exosomal miR-106b-3p via the Wnt signaling pathway [60] and miR-146a-5p via TRAF6 [61] can stimulate M1 macrophage polarization and thus they can cause pathological changes in MI. In another study, it has been revealed that exosomal miR-155 released from hypertrophic CMs regulates cardiac hypertrophy by triggering the secretion of inflammatory cytokines IL-6 and IL-8 [62].
ECs-derived exosomes
Another group of cells found in the heart is ECs. These cells have been shown to be involved in the pathological mechanisms of CAD by interacting with various cells (Fig. 2). In a study, it has been shown that human umbilical vein ECs (HUVECs) exposed to ischemia–reperfusion (I/R) release 2 times more exosomes and reduce the apoptosis of CMs compared to the control group [63]. In an in vivo study with HUVECs-derived exosomes, injection of these exosomes into the infarct region inhibited CM apoptosis via the PI3K/AKT signaling pathway and significantly improved cardiac functions [64]. Another study has focused on pyroptosis, which is cell death as a result of inflammation. In this study, Zhang et al. have shown that miR-27b-3p is overexpressed in exosomes derived from hypoxic CMEC, and this miRNA decreases the development of cardiac damage after I/R by inhibiting oxidative stress and pyroptosis induced by FOXO1/GSDMD signaling [65].
Krüppel-like factor 2 (KLF2) is a transcription factor involved in the regulation of EC metabolism. Activation of KLF2 in ECs induces the vasodilating effect and prevents the development of atherosclerosis thanks to its many regulated genes [66]. Hergenreider et al. have found that the miR-143/145 cluster, which plays an important role in vascular modulation, is significantly upregulated in the exosomes of KLF2-transformed HUVECs [67]. These exosomes have been observed to reduce the formation of atherosclerotic lesions in the aorta of atherosclerosis model mice. In another study, the reason for the decrease in these lesions was associated with the decrease in inflammation-related expression [68].
Gollmann-Tepeköylü et al. have used shockwave therapy on muscles to trigger exosome release from ECs in ischemic muscle and to investigate endogenous repair mechanisms in CAD [69]. Exosomal miR-19a-3p released from ECs induced angiogenesis, reduced myocardial fibrosis, and improved left ventricular function after myocardial ischemia by activating Akt and ERK. In a study on cardiac contraction, it has been shown that the 16 kDa fragment of N-terminal prolactin, which can be enzymatically cleaved in many target tissues, induces the expression of exosomal mir-146a in ECs. Exosomal transfer of this mir-146a to CMs has been observed to improve cardiac functions by targeting erb-b2 receptor tyrosine kinase 4 (ERBB4), notch1, and interleukin-1 receptor-associated kinase 1 genes (IRAK1) [70].
FBs-derived exosomes
In studies examining the cross-talk of cardiac FBs with ECs (Fig. 2), it has been shown that FBs are activated by TGF-β, and exosomes derived from active FBs cause dysfunction in ECs [71, 72]. Further analysis has revealed that miR-200a-3p is highly expressed in the cargo content of these exosomes, resulting in decreased gene expression such as VEGFA, and angiopoietin1 in ECs, and a consequent reduction in angiogenesis capacity. Inhibition of active FBs miR-200a-3p has attenuated this dysfunction observed in ECs [72].
The effects of exosomes derived from FBs on different types of cell death of CMs under pathological conditions have been also studied. In a study on apoptosis, it has been shown in vivo that exosomes derived from FBs reduce apoptosis through the PI3K/AKT pathway after MI, thereby improving cardiac functions [73]. Liu et al. have shown that miR-133a, which increases in the exosomes of FBs under I/R conditions, reduces the pyroptosis of CMs by suppressing embryonic lethal abnormal vision-like 1, which has a proinflammatory function [74]. Ferroptosis, which can be a form of death of CMs during MI, is characterized by iron-dependent lipid peroxide accumulation [75]. The researchers have set up an in vitro pacing model to examine the effect of exosomes derived from FBs on ferroptosis and have found that exosomes of pacing group cardiac FBs exacerbate ferroptosis of CMs. The reason for this has been determined to be increased miR-23a-3p in cargo content. However, inhibition of miR-23a-3p has protected CMs from ferroptosis with upregulation of its target, solute carrier family 7 member 11 (SLC7A11) gene [76]. Bang et al. have shown that miR-21-3p transferred to CMs in exosomes derived from FBs induces cardiac hypertrophy by reducing SH3 domain-containing protein 2 (SORBS2) and PDZ and LIM domain protein 5 (PDLIM5) [77]. Silencing this miRNA in the mouse model of hypertrophy has suppressed the hypertrophic growth of CMs.
Immune cells-derived exosomes
Inflammatory responses in the heart play critical roles in clearing the infarct area from injured and dead cells and ECM residual and in the formation of scar tissue [78]. miR-155 expression has been found to be increased in the mouse heart after MI, and it was primarily expressed in macrophages of the damaged heart and presented to FBs. When its effect on FBs has been examined, it has been observed that it inhibited the proliferation of FBs and supported cardiac inflammation by down-regulating the Son of Sevenless 1 (SoS1) gene [79]. A further study has shown that exosomal miR-155 was transferred from M1 macrophages to ECs after MI. On the other hand, ECs have been observed to have an anti-angiogenic effect by down-regulating these genes, including Rac family small GTPase 1 (RAC1) and p21-activated kinase 2 (PAK2), which also lead to impaired cardiac functions [80]. Besides miR-155, exosomal circRNA has been found in macrophage-FB cross-talk. It has been shown that circUbe3a in exosomes derived from the M2 macrophage phenotype after MI promotes the proliferation and migration of FBs, exacerbating post-MI cardiac fibrosis [81].
The effects of macrophage-derived exosomes on cell death in their cross-talk with CMs have been researched [82,83,84]. In these studies on M2 phenotype macrophages, it has been shown that by down-regulating SRY-Box Transcription Factor 6 (SOX6), the target of miR-1271-5p, it reduced apoptosis of CMs in hypoxic conditions and attenuated cardiac damage in acute MI [82]. The identified miR-148a and miR-145-5p have been shown to inhibit CM pyroptosis by targeting the TLR-4 protein [83].
In studies on DCs, which migrate to the infarct border area and play a role in wound healing, it has been shown that dexosomes activate CD4+ T cells and provide improvement in cardiac functions by inducing the expression of IL-4 and IL-10 [85, 86]. Dexosomes have also been shown to play a role in post-MI angiogenesis. Angiogenesis-related miR-16-5p, 23a-3p, 150-5p, 126-3p, and 494‑3p have been found to be upregulated in the exosomes of DCs cultured with supernatants of necrotic CMs relative to the control group. Co-culture of these exosomes with CMECs significantly has upregulated VEGF expression and increased tube formation by CMECs [87, 88].
In the heart, the persistence of inflammation is controlled by CD4+ T cells. The use of immune therapeutics that inhibit CD4+ T cells has been found to prevent cardiac fibrosis and hypertrophy in the ischemic heart, and these cells are associated with pathological cardiac remodeling [89,90,91]. In a study on the underlying mechanism, it has been found that miR-142-3p in the exosomes of CD4+ T cells inhibited the Wnt signaling pathway, thus triggering myofibroblast activation and fibrogenesis. Exosomal miR-142-3p has been identified as a potential target to treat cardiac remodeling [92]. In addition, the effects of CD4+ T cells-derived exosomes on the activation of CMECs have been also examined. CD4+ T cell exosome treatment has exacerbated mitochondrial reactive oxygen species generation, decreased nitric oxide levels, and increased the proliferation of CMECs [93, 94].
TCs-derived exosomes
Another group of cells in the heart is TCs. These cells differ from other cells in that they have long cytoplasmic projections called telopods [95]. The presence of measurable amounts of angiogenic miRNAs (10a, 21, 27b, 100, 126-3p, 130a, 143, 155, 503) in exosomes derived from TCs has been shown [96]. This angiogenic effect has been subsequently demonstrated in vitro on ECs and in vivo in the MI rat model. In addition, increased angiogenesis, decreased cardiac fibrosis, and improved cardiac functions have been observed in the MI rat model [97]. In a recent study, it has been shown that TC exosomal miR-21-5p silenced cell death by targeting p53 target 1 gene (Cdip1) in CMECs and, thus down-regulate activated caspase-3. This mechanism has facilitated angiogenesis and regeneration after MI by inhibiting apoptosis of CMECs under ischemic and hypoxic conditions [98].
Stem cells-derived exosomes
ESCs-derived exosomes
Exosomes derived from embryonic stem cells (ESCs) can stimulate the proliferation of endogenous cardiac progenitor cells (CPCs) and upregulate the expression of CM genes in these cells, through miR-294 [99]. In addition, a decrease in neovascularization, apoptosis of CMs, fibrosis, and improvement in cardiac functions have been observed due to the cardiac proliferative effect after the administration of exosomes derived from ESCs. ESCs can also serve as the main source of exosome secretion by differentiating into cardiac progenitors. For example, treatment of EVs released from CPCs differentiated from human ESCs in a model of post-infarct heart injury found that 927 upregulated genes were specifically associated with cardiac remodeling pathways [100]. On the other hand, in the study to evaluate the cardioprotective potential of lncRNA content of ESCs-derived exosomes, it has been emphasized that lncRNAs GM4890 and XLOC01990 may play a crucial role in cardiac repair mechanisms following MI [101]. Of note, the use of ESCs-derived exosomes, which exert activity through reduced proinflammatory control, has been proposed to inhibit the cytotoxic effects of chemotherapeutics on cardiac tissue [102]. Wu et al. have shown that miR-497 increased the survival of neonatal rat CMs and tube formation on HUVECs in infarcted myocardium, in addition to high expression of lncRNA MALAT1 in exosomes derived from ESC-derived cardiovascular progenitor cells [103].
MSCs-derived exosomes
Mesenchymal stem cells (MSCs) play a pivotal role in maintaining normal cardiac function through interactions within the cardiac niche. Leveraging their exosome-rich cargo, MSCs hold promise for therapeutic interventions addressing cardiac tissue abnormalities, including the treatment of atherosclerotic plaque development. Takafuji et al. have stimulated TNF-α using exosomes released from adipose-derived MSCs (ADMSCs) and showed that cell adhesion molecules and macrophage accumulation were reduced in human aortic ECs (HAOECs) through the MAPK and NFκB pathway [104]. Bian et al. assessed the exosomes with an average diameter of 100 nm from bone marrow MSCs (BMMSCs) in a hypoxic acute MI model. They found these exosomes enhanced proliferation and tubule formation in HUVECs. Additionally, injecting BMMSC-derived EVs into the infarct border region reduced infarct size and improved cardiac function [105]. Similarly, it has been found that it mediated angiogenesis and showed anti-inflammatory activity, especially by reducing T cell proliferation [106]. In a study using human MSCs-derived exosomes, injected into mice via the tail vein, a significant reduction in infarct size was observed within 24 h [107]. In addition, increased cardiac function has been reported in animals treated with exosomes after a 28-day period compared to the control group. Exosomes from ADMSCs have been shown to reduce I/R injury-induced myocardial apoptosis via Bcl-2 upregulation, Bax downregulation, and Caspase-3 inhibition in MI-induced mice via the Wnt/β-catenin signaling pathway [108]. In another study with BMMSCs, exosomes from these cells reduced mouse CM apoptosis through AMPK/mTOR and Akt/mTOR signaling pathways, by upregulating LC3B expression [109].
Research into stem cell-derived exosomes has advanced methods to modify their contents through cell conditioning or gene transfer. Deng et al.’s study shows that irisin-pretreated BMMSCs release exosomes that mitigate pyroptosis and oxidative stress in CMs via NLRP3, suggesting their potential for MI treatment [110]. Another approach is a manipulation of MSCs with lentiviral CXCR4 to control the contents of exosomes released from MSCs to protect cardiac tissue following MI [111]. Decreased Caspase-3 with increased insulin-like growth factor-1 (IGF-1)α and p-Akt expression has been reported in CMs, and increased angiogenesis and decreased infarct size mediated by CXCR4 have been reported to modulate cardiac remodeling. In another study that proposed to use the cardioprotective activity of Akt signaling pathways as a therapeutic mediator, transfection of human umbilical cord MSCs (hucMSCs) with adenovirus Akt has been performed [112]. It has been observed that the expression of PDGF-D was significantly increased, and tubule-like structure formation and angiogenesis were restored. Another approach is the use of hypoxia-conditioned stem cells to trigger secretion mechanisms of stem cells through enrichment of exosomal cargo content or stimulation of vesicle pathways [113, 114]. For example, proteomic analysis of exosomes derived from hypoxia-pre-conditioned BMMSCs has shown that the high redox protein profile may regulate increased angiogenesis [115].
One of the players of the exosomal regulators is ncRNAs. ncRNAs that are not directly converted to protein products interfere with cellular functions by post-transcriptional mechanisms for the cell [116]. Nakamura et al. have examined the miRNA content of exosomes while investigating the role of adiponectin in the release of MSC-derived exosomes. The researchers have reported that MSCs-derived exosomes have high levels of let7, miR-21, 100, 48a, 10, 26, and 199 and emphasized that the targets of these miRNAs were critical in heart function, especially in fibrogenic and adhesion pathways, and the cellular metabolic processes [117]. Applying MSCs-derived exosomes in vivo, Li et al. have shown that let7 reduced the area of athreosclerotic plaque in ApoE − / − mice and inhibited M2 macrophage polarization and infiltration via let7/HMGA2/NF-κB and let7/IGF2BP1/PTEN, respectively [118]. It has also been reported that MSCs-derived exosomes reduce the development of atherosclerotic plaques by regulating M2 macrophage polarization with KLF6 inhibition and macrophage infiltration with inhibiting ERK1/2 signaling pathway via miR-21a-5p [119]. Yang et al. have observed that the rich miR-145 content of MSCs-derived exosomes inhibited JAM-A in vivo, thereby reducing the formation of atherosclerotic plaques via the transendothelial migration pathway [120]. It has been shown that inflammation and abdominal aortic aneurysm can be inhibited by targeting TXNIP and NLRP3 by miR-17-5p in exosomes from ADMSCs [121].
Elucidating the roles of the interaction between ncRNAs and coding RNAs in exosomal pathways on cardiac tissue pathophysiology has also been the driving force for the development of therapeutic options for MI, in addition to atherosclerosis (Table 1) [144]. Feng et al. have reported that exosomes secreted from mouse BMMSCs after ischemic preconditioning contain a high amount of miR-22 and they observed MECP2-mediated antiapoptopic effect after internalization of the exosomes [125]. Wang et al. have compared the cardioprotective effects of human BMMSCs, ADMSCs, and endometrium-derived MSCs (EnMSCs) in a rat model of acute MI [145]. It has been found that miR-21 in EnMSCs-derived exosomes, assumed the main regulatory role by increasing Bcl-2 and VEGF. It has been shown that exosomal miR-21 can be used in C-kit+ CSCs-mediated ischemic myocardium therapy via the same pathways [127]. In addition, the researchers revealed that these exosomes contain miR-19a and it may show a cardioprotective effect as a result of decreased PTEN expression and activation of antiapoptopic Akt and ERK. In a study evaluating the efficacy of HIF-1α overexpression on human dental pulp MSCs exosome secretion, overexpression of 10 miRNAs, including, miR-15, 16, 17, 31, 126, 145, 221, 222, 320a, and 424, were associated with vesicular pathways, secretion, and capillary development. It has been also emphasized that Notch signaling and angiogenesis pathways can be modulated by exosomal cargo content [113]. On the contrary, the contents of stem cell-derived exosomes may also have negatory effects on MI therapy. Ning et al. have underlined that the miR-153-3p content of BMSCs-derived exosomes supports hypoxia-induced myocardial damage via VEGF/PI3K/Akt/eNOS signal pathway [146].
Although the miRNA contents of MSCs-derived exosomes vary, miRNA targets in MI therapeutic applications are generally associated with apoptotic signaling pathways, particularly the PTEN/Akt signaling (Table 1). miRNAs serve as mediators of indirect regulation mechanisms of exosomal lncRNAs and circRNAs as well as post-transcriptional regulation of coding genes directly. Exosomal transport of lncRNAs is more inconvenient because they are larger molecules compared to miRNAs. For these reasons, MSCs-derived exosomal lncRNA/circRNA therapeutic approaches are limited compared to miRNA studies (Fig. 3) [147, 148]. It has been reported that lncRNA fetal-lethal non-coding developmental regulatory RNA (FENDRR) regulates the miR-28/TEAD1 axis through exosomes derived from MSCs and reduces the progression of athrosclerosis plaque through apoptosis, oxidative stress, and inflammatory response [149]. Researchers who performed an MSCs-derived exosomal approach of si-LOC100129516 to silencing lncRNAs, which are known to contribute to the progression of atheroscrelosis in CAD with its upregulation, demonstrated that intracellular lipid accumulation is reduced by the stimulation of the PPARγ/LXRα/ABCA1 pathway [150]. It has been shown that lncRNA XIST found in exosomes derived from ADMSCs, reduces the miR-214-3p repressive effect on Arl2, thereby suppressing myocardial pyroptosis in atrial fibrillation [151]. Although the roles of extracellular circRNAs in the development of atherosclerosis have been defined [152], further studies are needed for the therapeutic use of stem cell-derived exosomal circRNAs.
MSCs-derived lncRNA KLF3-AS1 controls regulation of the SIRT1 gene through miR-138-5p. KLF3-AS1 overexpression causes a decrease in the infarct area and downregulation of apoptosis and pyroptosis pathways [153]. Similarly, it has been reported that the lncRNA UCA1 participates in the miR-873-5p/XIAP axis and increases the expression of the anti-apoptotic Bcl-2 protein, causing the reduction in the repressive effect of miR-873-5p on XIAP [154]. It has been shown that rat MSCs-derived exosomal lncRNA H19 content was upregulated by atorvastatin, which is frequently used in coronary diseases, it has been found that angiogenesis and pro-cardioprotective effect were achieved by modulating the miR-675/VEGF-ICAM1 axis [155]. The cardioprotective effect of exosomal lncRNA NEAT1 obtained from MSCs treated with macrophage migration inhibitory factor (MIF) has been mediated through inhibition of apoptosis via the miR-142-3p/FOXO1 pathway [156]. Exosomal lncRNA Mir9-3hg from BMMSCs has been shown to directly bind with pumilio RNA binding family member 2 (Pum2) to downregulate its intracellular expression [157]. It has been found that the expression of peroxiredoxin 6 (PRDX6) increased due to the decreased expression of Pum2, and accordingly, the cardiac function improved with the inhibition of the CM ferroptosis pathway. With the discovery that circRNAs, apart from lncRNAs, can also play a crucial role in miRNA/mRNA-mediated cardiac pathways, approaches to the therapeutic use of exosomal circRNAs in cardiovascular diseases have emerged [158, 159]. One of the early studies has indicated that increased circ_0001273 expression in exosomes from hucMSCs has the potential for myocardial repair and regeneration after MI [160]. A recent study has revealed the circ_0002113/miR-188-3p/RUNX1 axis with exosomes derived from BMMSCs. It has been reported that suppression of MI is regulated via the USP7/p53 apoptotic pathway via circ_0002113 in the exosome [161]. CircRTN4 in MSCs-derived exosomes has been subjected to reduce myocardial injury in sepsis-induced myocardial cells through the miR-497-5p/MG53 axis. In particular, it has been reported that cell survival was increased and apoptosis was suppressed [162]. In a recent report, exosomal circ_0001747 from ADMSCs has been found to inhibit apoptosis after hypoxia/reoxygenation injury via the miR-199-3p/MCL1 axis [163].
CPCs- and CDCs-derived exosomes
Analyzing EVs derived from CPCs from normal and pathological adult human hearts, Romano et al. have used them to create a conditioned medium and examined their activity on cardiac cells [164]. Proteomic analyses have shown that CPC-derived EVs contain growth factors that modulate cell migration, proliferation, and differentiation processes. Initial studies have reported that upregulation of miR-210, 132, and 146a-3p expression of CPC-derived exosomes inhibited CMs apoptosis after MI [6]. They have also evaluated the effect of CPC-derived exosomes on reducing CM apoptosis through IGF-1 receptor activation in subsequent studies [165]. Off note, it has been emphasized that CPC exosomes could be a miR-146a-5p-mediated solution not only to cardiovascular diseases but also to cardiotoxicity problems caused by chemotherapeutics used for the treatment of cancer [166]. Gray et al. compared exosomes from CPCs under hypoxic and normal conditions, finding higher levels of pro-angiogenic miRNAs like miRNA-132 and miRNA-146a in hypoxic CPC-derived exosomes [167]. These hypoxic exosomes promoted tube formation in ECs, reduced fibrosis, and improved cardiac function in vivo. It has been shown that exosomes derived from mouse CPCs reduce CM apoptosis both in vivo and in vitro after MI [168]. The injections of human CPCs-derived exosomes after MI have resulted in decreasing infarct size and fibrosis, in addition to increasing vessel density [6]. Also, it has been reported that the highest anti-fibrotic and antiapoptopic effects of CPCs-derived exosomes are observed when injected into the mouse infarct border region at the time of MI [165, 169]. Cardiosphere-derived cells (CDCs) from the cardiac progenitor cell group may also contribute to cardiac tissue repair mechanisms through exosomal interactions. For example, Hypoxic conditioned CDCs release exosomes with increased levels of pro-angiogenic miRNAs (miR-126, 130a, and 210), potentially promoting cardioprotective effects [170].
One of the most promising studies has found evidence that clinical applications of CDCs are possible. In a clinical study involving five children with dilated cardiomyopathy, CDCs-derived exosomes containing miR-146a-5p showed potential therapeutic effects by reducing proinflammatory cytokines and myocardial fibrosis [171]. Analysis of exosomes derived from CDCs has highlighted the significance of miR-146 and miR-210 in the prevention of CMs apoptosis. These findings not only provide valuable insights into the potential of RNA-based exosomal therapeutics but also underscore the promise of CDC-derived exosomes in cardiac tissue repair mechanisms.
iPSCs-derived exosomes
It has been shown in comprehensive studies with dermal or cardiac reprogrammed induced pluripotent stem cells (iPSCs)-derived exosomes are more reliable and efficient for enhancing cardioprotective effect, proliferation, tube formation, and myocardial evolution or reducing apoptosis (Fig. 4a). It has been observed that iPSCs-derived EVs are rich in pro-angiogenic and cardioprotective miRNA (> 200) and proteins (such as BMP-4, PDGFα, TDGF1, thrombospondin-1, and VEGF-C) [172]. In in vivo studies, it has been stated that iPSC and iPSCs-derived EVs have similar cardioprotective effects on living myocardium; EVs are more effective in inhibiting apoptosis compared to cell therapy; and teratoma formation that occurs in iPSC application has not been observed. In a study conducted with exosomes derived from iPSCs obtained from mouse cardiac FBs, it has been shown that a cardioprotective effect was observed in CMs thanks to miR-21 and miR-210 in exosomes [173].
Gao et al. have shown the exosome-mediated cardiac disease therapeutic function of cardiac tissue cells differentiated from human iPSCs [174••]. It has been emphasized that increased myocardial recovery was achieved according to in vitro and in vivo results. Evaluating the potential of cardiac- and dermal- FB reprogrammed iPSCs for cardiac tissue repair, Kurtzwald-Josefson et al. have reported that reduced expression of miR-22 contributes to the cardiac remodeling process [175•]. Harane et al. assessed the therapeutic potential of EVs derived from cardiovascular progenitors and CMs derived from iPSCs in a murine model. These EVs, enriched with 16 highly conserved miRNAs, significantly enhanced EC migration, proliferation, and tube formation (Fig. 4b) [176]. In parallel, the efficacy of EVs derived from iPSCs and iPSCs-derived CMs has been researched in vivo by application of hydrogel to the infarcted rat heart [177]. It has been reported that the cargo content of vesicles secreted from CM-differentiated iPSCs exhibits a cardiac tissue-specific profile. Distinct miRNA profiles between EVs secreted from iPSCs and iPSCs-derived CMs were particularly striking. Increased expression of 7 miRNAs including miR-122, 1, 143, 127b, 200b, 99b, and 30d, and decreased expression of miR-302a, 92a, and 302b in iPSC differentiated CMs have been observed (Fig. 4c). This unique miRNA profile indicates that EVs from iPSC-derived CMs are more appropriate for therapeutic applications aimed at reducing infarct size and cell hypertrophy. Results from exosomes of iPSCs-derived CMs have clearly indicated that pathways in the recovery of cardiac function can be mediated by various miRNAs. On the other hand, transcriptomic profiles of exosomes of CMs differentiated from ESC and iPSC suggest that the specificity of the exosomal cargo content is significantly correlated with the reprogrammed cell type [178].
Challenges in coordinating the effect of the cargo contents of exosomes on cells
Understanding the cardiac tissue repair mechanisms of exosomes and developing therapeutic options using these mechanisms is one of the most remarkable issues for today's scientific authorities. Although we have a much deeper understanding of the role of exosome and cargo contents in cardiac repair than a decade ago studies are still needed to develop comprehensive approaches [179]. Most exosomal approaches focus on miRNA-mediated regulation mechanisms. When examining the extensive miRNA expression profiles of exosomal cargo contents, function-specific mapping, and identification are challenging due to the wide diversity of miRNAs. At the same time, miRNA target interactions identified to date indicate that miRNA regulatory functions are modulated by multiple, layered, and fine-tuning interactions.
On the other hand, unlike miRNAs, lncRNAs and circRNAs show higher tissue- and function- specificity. However, the layered interaction networks of all these ncRNAs are the main challenge in the development of exosomal RNA approaches. For example, the lncRNA Mir9-hg, which has been reported to improve cardiac function, may also contribute to the development of lung squamous cell carcinoma by cross-talking the miR-138-5p/LIMK1 axis [180]. Although the regulatory role of circRNAs, which is one of the critical molecular modulators in many tissues, in cardiac tissues has been strikingly demonstrated, there is limited report in the use of exosomal circRNAs originating from different cell types for cardiovascular therapeutic purposes [181]. For these reasons, comprehensive and fine-tuned approaches are needed for the use of exosomal RNAs as cardiac therapeutics. Clinical applications will only be possible as a result of large-scale and comprehensive analyses of the therapeutic applications of exosomes with rich cargo content.
Conclusion and future perspective
The comprehensive review on CAD and MI which is the most important clinical symptom of CAD shows that clinical challenges have not been overcome yet, although various treatment approaches have been applied for the regeneration of tissue damage after CAD. Stem cell studies, promising for CAD treatment, face limitations such as low cell survival under ischemic conditions and disadvantages associated with different stem cell types. Recent evidence indicates that the regenerative effects of these cells, such as cytoprotection, inflammation control, and angiogenesis, result from the exosomes they secrete rather than direct tissue integration.
Exosomes, vital for intracardiac communication, are emerging as a promising platform in cardiac regenerative medicine due to their rich cargo content. Exosome-based therapies show potential to overcome limitations like cell survival, teratoma formation, and immune rejection compared to stem cell therapy. However, this evolving strategy requires further research before practical use in cardiac repair. Cargo content must be tailored to specific biological functions in target tissues or cells. Manipulating exosomal molecules and conjugating markers for precise cell targeting can offer effective treatment strategies in cardiac repair.
Availability of data and material
Not applicable.
Code availability
Not applicable.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Lindstrom M, DeCleene N, Dorsey H et al. Global burden of cardiovascular diseases and risks collaboration, 1990–2021.
Vaduganathan M, Mensah GA, Turco JV, Fuster V, Roth GA. The global burden of cardiovascular diseases and risk: a compass for future health: American College of Cardiology Foundation Washington DC, 2022:2361–71.
Alpert JS, Thygesen K, Antman E, Bassand J. Myocardial infarction redefined–a consensus document of The Joint European Society of Cardiology/American College of Cardiology Committee for the redefinition of myocardial infarction. J Am Coll Cardiol. 2000;36(3):959–69.
Singla DK. Embryonic stem cells in cardiac repair and regeneration. Antioxid Redox Signal. 2009;11(8):1857–63.
Aslan A, Allahverdiyev AM, Bagirova M, Abamor ES. Problems in stem cell therapy for cardiac repair and tissue engineering approaches based on graphene and its derivatives. Curr Stem Cell Res Ther. 2018;13(6):447–57. https://doi.org/10.2174/1574888X13666180510110055.
Barile L, Lionetti V, Cervio E, et al. Extracellular vesicles from human cardiac progenitor cells inhibit cardiomyocyte apoptosis and improve cardiac function after myocardial infarction. Cardiovasc Res. 2014;103(4):530–41.
Ailawadi S, Wang X, Gu H, Fan G-C. Pathologic function and therapeutic potential of exosomes in cardiovascular disease. Biochimica et Biophysica Acta (BBA)-Mol Basis Dis. 2015;1852(1):1–11.
Mayourian J, Ceholski DK, Gorski P et al. Exosomal microRNA-21–5p mediates mesenchymal stem cell paracrine effects on human cardiac tissue contractility. Circ Res. 2018;118.312420.
Raposo G, Stoorvogel W. Extracellular vesicles: exosomes, microvesicles, and friends. J Cell Biol. 2013;200(4):373–83.
Balaj L, Lessard R, Dai L, et al. Tumour microvesicles contain retrotransposon elements and amplified oncogene sequences. Nat Commun. 2011;2:180.
Cossetti C, Iraci N, Mercer TR, et al. Extracellular vesicles from neural stem cells transfer IFN-γ via Ifngr1 to activate Stat1 signaling in target cells. Mol Cell. 2014;56(2):193–204.
van Niel G, D’Angelo G, Raposo G. Shedding light on the cell biology of extracellular vesicles. Nature Rev Mol Cell Biol. 2018.
Meldolesi J. Ectosomes and exosomes-two extracellular vesicles that differ only in some details. Biochem Mol Biol J. 2016;2(1).
Akers JC, Gonda D, Kim R, Carter BS, Chen CC. Biogenesis of extracellular vesicles (EV): exosomes, microvesicles, retrovirus-like vesicles, and apoptotic bodies. J Neurooncol. 2013;113(1):1–11.
Bronson DL, Fraley EE, Fogh J, Kalter S. Induction of retrovirus particles in human testicular tumor (Tera-1) cell cultures: an electron microscopic study. J Natl Cancer Inst. 1979;63(2):337–9.
Edgar JR. Q&A: What are exosomes, exactly? BMC Biol. 2016;14(1):46.
Harding C, Heuser J, Stahl P. Receptor-mediated endocytosis of transferrin and recycling of the transferrin receptor in rat reticulocytes. J Cell Biol. 1983;97(2):329–39.
Pan B-T, Teng K, Wu C, Adam M, Johnstone RM. Electron microscopic evidence for externalization of the transferrin receptor in vesicular form in sheep reticulocytes. J Cell Biol. 1985;101(3):942–8.
Johnstone RM, Adam M, Hammond J, Orr L, Turbide C. Vesicle formation during reticulocyte maturation. Association of plasma membrane activities with released vesicles (exosomes). J Biol Chem. 1987;262(19):9412–20.
Le Pecq J-B. Dexosomes as a therapeutic cancer vaccine: from bench to bedside. Blood Cells Mol Dis. 2005;35(2):129–35.
Hosseini HM, Soleimanirad J, Aghdam EM, Amin M, Fooladi AAI. Texosome-anchored superantigen triggers apoptosis in original ovarian cancer cells. Med Oncol. 2015;32(1):409.
Kelly R, Holland P, Skibinski G, et al. Extracellular organelles (prostasomes) are immunosuppressive components of human semen. Clin Exp Immunol. 1991;86(3):550–6.
Simpson R, Mathivanan S. Extracellular microvesicles: the need for internationally recognised nomenclature and stringent purification criteria. 2012.
Théry C, Boussac M, Véron P, et al. Proteomic analysis of dendritic cell-derived exosomes: a secreted subcellular compartment distinct from apoptotic vesicles. J Immunol. 2001;166(12):7309–18.
Schorey JS, Bhatnagar S. Exosome function: from tumor immunology to pathogen biology. Traffic. 2008;9(6):871–81.
Azmi AS, Bao B, Sarkar FH. Exosomes in cancer development, metastasis, and drug resistance: a comprehensive review. Cancer Metastasis Rev. 2013;32(3–4):623–42.
Clayton A, Turkes A, Dewitt S, Steadman R, Mason MD, Hallett MB. Adhesion and signaling by B cell-derived exosomes: the role of integrins. FASEB J. 2004;18(9):977–9.
Feng D, Zhao WL, Ye YY, et al. Cellular internalization of exosomes occurs through phagocytosis. Traffic. 2010;11(5):675–87.
Blanchard N, Lankar D, Faure F, et al. TCR activation of human T cells induces the production of exosomes bearing the TCR/CD3/ζ complex. J Immunol. 2002;168(7):3235–41.
Ieda M. Heart development and regeneration via cellular interaction and reprogramming. Keio J Med. 2013;62(4):99–106.
Barile L, Gherghiceanu M, Popescu LM, Moccetti T, Vassalli G. Ultrastructural evidence of exosome secretion by progenitor cells in adult mouse myocardium and adult human cardiospheres. BioMed Res Int. 2012;2012.
Giricz Z, Varga ZV, Baranyai T, et al. Cardioprotection by remote ischemic preconditioning of the rat heart is mediated by extracellular vesicles. J Mol Cell Cardiol. 2014;68:75–8.
Théry C, Zitvogel L, Amigorena S. Exosomes: composition, biogenesis and function. Nat Rev Immunol. 2002;2(8):569.
Waldenström A, Gennebäck N, Hellman U, Ronquist G. Cardiomyocyte microvesicles contain DNA/RNA and convey biological messages to target cells. PLoS ONE. 2012;7(4): e34653.
Gupta S, Knowlton AA. HSP60 trafficking in adult cardiac myocytes: role of the exosomal pathway. Am J Physiol Heart Circ Physiol. 2007;292(6):H3052–6. https://doi.org/10.1152/ajpheart.01355.2006.
Yu X, Deng L, Wang D, et al. Mechanism of TNF-α autocrine effects in hypoxic cardiomyocytes: initiated by hypoxia inducible factor 1α, presented by exosomes. J Mol Cell Cardiol. 2012;53(6):848–57.
Malik ZA, Kott KS, Poe AJ, et al. Cardiac myocyte exosomes: stability, HSP60, and proteomics. Am J Physiol Heart Circ Physiol. 2013;304(7):H954–65. https://doi.org/10.1152/ajpheart.00835.2012.
Ribeiro-Rodrigues TM, Laundos TL, Pereira-Carvalho R, et al. Exosomes secreted by cardiomyocytes subjected to ischaemia promote cardiac angiogenesis. Cardiovasc Res. 2017;113(11):1338–50. https://doi.org/10.1093/cvr/cvx118.
Garcia NA, Ontoria-Oviedo I, González-King H, Diez-Juan A, Sepúlveda P. Glucose starvation in cardiomyocytes enhances exosome secretion and promotes angiogenesis in endothelial cells. PLoS ONE. 2015;10(9): e0138849.
Hsieh PC, Davis ME, Lisowski LK, Lee RT. Endothelial-cardiomyocyte interactions in cardiac development and repair. Annu Rev Physiol. 2006;68:51–66.
Ottaviani LM, Juni RP, Sansonetti M et al. Cardiomyocyte-derived Mir-200c-3p in exosomes affects endothelial angiogenic capacity and impairs cardiac function. Circ Res. 2019;125(Suppl_1):A896-A96.
Gou L, Xue C, Tang X, Fang Z. Inhibition of Exo-miR-19a-3p derived from cardiomyocytes promotes angiogenesis and improves heart function in mice with myocardial infarction via targeting HIF-1α. Aging (Albany NY). 2020;12(23):23609.
Li G, Qiu Z, Li C, et al. Exosomal MiR-29a in cardiomyocytes induced by angiotensin II regulates cardiac microvascular endothelial cell proliferation, migration and angiogenesis by targeting VEGFA. Curr Gene Ther. 2022;22(4):331–41. https://doi.org/10.2174/1566523222666220303102951.
Bonauer A, Carmona G, Iwasaki M, et al. MicroRNA-92a controls angiogenesis and functional recovery of ischemic tissues in mice. Science. 2009;324(5935):1710–3. https://doi.org/10.1126/science.1174381.
Wang Y, Zhao R, Liu W, et al. Exosomal circHIPK3 released from hypoxia-pretreated cardiomyocytes regulates oxidative damage in cardiac microvascular endothelial cells via the miR-29a/IGF-1 pathway. Oxid Med Cell Longev. 2019;2019:7954657. https://doi.org/10.1155/2019/7954657.
Wang Y, Zhao R, Shen C, et al. Exosomal CircHIPK3 released from hypoxia-induced cardiomyocytes regulates cardiac angiogenesis after myocardial infarction. Oxid Med Cell Longev. 2020;2020:8418407. https://doi.org/10.1155/2020/8418407.
Ye J, Huang Y, Que B, et al. Interleukin-12p35 knock out aggravates doxorubicin-induced cardiac injury and dysfunction by aggravating the inflammatory response, oxidative stress, apoptosis and autophagy in mice. EBioMedicine. 2018;35:29–39. https://doi.org/10.1016/j.ebiom.2018.06.009.
Kim SC, Stice JP, Chen L, et al. Extracellular heat shock protein 60, cardiac myocytes, and apoptosis. Circ Res. 2009;105(12):1186–95. https://doi.org/10.1161/CIRCRESAHA.109.209643.
Tian J, Guo X, Liu X-M, et al. Extracellular HSP60 induces inflammation through activating and up-regulating TLRs in cardiomyocytes. Cardiovasc Res. 2013;98(3):391–401.
Yu DW, Ge PP, Liu AL, Yu XY, Liu TT. HSP20-mediated cardiomyocyte exosomes improve cardiac function in mice with myocardial infarction by activating Akt signaling pathway. Eur Rev Med Pharmacol Sci. 2019;23(11):4873–81. https://doi.org/10.26355/eurrev_201906_18075.
Yang Y, Li Y, Chen X, Cheng X, Liao Y, Yu X. Exosomal transfer of miR-30a between cardiomyocytes regulates autophagy after hypoxia. J Mol Med. 2016;94:711–24.
Zhang C, Gan X, Liang R, Jian J. Exosomes derived from epigallocatechin gallate-treated cardiomyocytes attenuated acute myocardial infarction by modulating microRNA-30a. Front Pharmacol. 2020;11:126. https://doi.org/10.3389/fphar.2020.00126.
Wang X, Gu H, Huang W, et al. Hsp20-mediated activation of exosome biogenesis in cardiomyocytes improves cardiac function and angiogenesis in diabetic mice. Diabetes. 2016;65(10):3111–28. https://doi.org/10.2337/db15-1563.
Wang X, Gu H, Huang W, Wang Y, Fan G-C. Hsp20-reprogrammed exosomes derived from cardiomyocytes provide protection against diabetic cardiomyopathy in mice. Circulation 2014;130(suppl_2):A12638-A38.
Yang J, Yu X, Xue F, Li Y, Liu W, Zhang S. Exosomes derived from cardiomyocytes promote cardiac fibrosis via myocyte-fibroblast cross-talk. Am J Transl Res. 2018;10(12):4350–66.
Nie X, Fan J, Li H, et al. miR-217 promotes cardiac hypertrophy and dysfunction by targeting PTEN. Mol Ther Nucleic Acids. 2018;12:254–66. https://doi.org/10.1016/j.omtn.2018.05.013.
Guo J, Chen LW, Huang ZQ, et al. Suppression of the inhibitory effect of circ_0036176-translated Myo9a-208 on cardiac fibroblast proliferation by miR-218-5p. J Cardiovasc Transl Res. 2022;15(3):548–59. https://doi.org/10.1007/s12265-022-10228-x.
Poon IK, Lucas CD, Rossi AG, Ravichandran KS. Apoptotic cell clearance: basic biology and therapeutic potential. Nat Rev Immunol. 2014;14(3):166–80. https://doi.org/10.1038/nri3607.
Zhang Z, Xu Y, Cao C, et al. Exosomes as a messager to regulate the crosstalk between macrophages and cardiomyocytes under hypoxia conditions. J Cell Mol Med. 2022;26(5):1486–500.
Sun S, Wu Y, Maimaitijiang A, Huang Q, Chen Q. Ferroptotic cardiomyocyte-derived exosomes promote cardiac macrophage M1 polarization during myocardial infarction. PeerJ. 2022;10: e13717. https://doi.org/10.7717/peerj.13717.
Chen C, Cai S, Wu M, et al. Role of cardiomyocyte-derived exosomal microRNA-146a-5p in macrophage polarization and activation. Dis Markers. 2022;2022:2948578. https://doi.org/10.1155/2022/2948578.
Yu H, Qin L, Peng Y, Bai W, Wang Z. Exosomes derived from hypertrophic cardiomyocytes induce inflammation in macrophages via miR-155 mediated MAPK pathway. Front Immunol. 2020;11: 606045. https://doi.org/10.3389/fimmu.2020.606045.
Zheng Y, Vicencio JM, Yellon DM, Davidson SM. 27 exosomes released from endothelial cells are cardioprotective. Heart. 2014;100(Suppl 1):A10–A10.
Liu W, Feng Y, Wang X, et al. Human umbilical vein endothelial cells-derived exosomes enhance cardiac function after acute myocardial infarction by activating the PI3K/AKT signaling pathway. Bioengineered. 2022;13(4):8850–65. https://doi.org/10.1080/21655979.2022.2056317.
Zhang B, Sun C, Liu Y, Bai F, Tu T, Liu Q. Exosomal miR-27b-3p derived from hypoxic cardiac microvascular endothelial cells alleviates rat myocardial ischemia/reperfusion injury through inhibiting oxidative stress-induced pyroptosis via Foxo1/GSDMD signaling. Oxid Med Cell Longev. 2022;2022.
Dabravolski SA, Sukhorukov VN, Kalmykov VA, Grechko AV, Shakhpazyan NK, Orekhov AN. The role of KLF2 in the regulation of atherosclerosis development and potential use of KLF2-targeted therapy. Biomedicines. 2022;10(2):254. https://doi.org/10.3390/biomedicines10020254.
Hergenreider E, Heydt S, Tréguer K, et al. Atheroprotective communication between endothelial cells and smooth muscle cells through miRNAs. Nat Cell Biol. 2012;14(3):249–56.
He S, Wu C, Xiao J, Li D, Sun Z, Li M. Endothelial extracellular vesicles modulate the macrophage phenotype: Potential implications in atherosclerosis. Scand J Immunol. 2018;87(4): e12648. https://doi.org/10.1111/sji.12648.
Gollmann-Tepeköylü C, Pölzl L, Graber M, et al. miR-19a-3p containing exosomes improve function of ischaemic myocardium upon shock wave therapy. Cardiovasc Res. 2020;116(6):1226–36.
Halkein J, Tabruyn SP, Ricke-Hoch M, et al. MicroRNA-146a is a therapeutic target and biomarker for peripartum cardiomyopathy. J Clin Investig. 2013;123(5):2143–54.
Ranjan P, Kumari R, Krishnamurthy P, Kishore R, Verma SK. Cardiac fibroblast-derived exosomes mediate endothelial dysfunction and heart failure. Circ Res. 2019;125(Suppl_1):A256-A56.
Ranjan P, Kumari R, Goswami SK, et al. Myofibroblast-derived exosome induce cardiac endothelial cell dysfunction. Front Cardiovasc Med. 2021;8: 676267. https://doi.org/10.3389/fcvm.2021.676267.
Li Q, Shen X, Wen S, Li Y, Xie D-M. Transplantation of exosomes derived from CD90 positive fibroblasts reduce apoptosis of cardiomyocytes in mice after acute myocardial infarction. 2021.
Liu N, Xie L, Xiao P, et al. Cardiac fibroblasts secrete exosome microRNA to suppress cardiomyocyte pyroptosis in myocardial ischemia/reperfusion injury. Mol Cell Biochem. 2022;477(4):1249–60. https://doi.org/10.1007/s11010-021-04343-7.
Wu X, Li Y, Zhang S, Zhou X. Ferroptosis as a novel therapeutic target for cardiovascular disease. Theranostics. 2021;11(7):3052.
Liu D, Yang M, Yao Y, et al. Cardiac fibroblasts promote ferroptosis in atrial fibrillation by secreting exo-miR-23a-3p targeting SLC7A11. Oxid Med Cell Longev. 2022;2022:3961495. https://doi.org/10.1155/2022/3961495.
Bang C, Batkai S, Dangwal S, et al. Cardiac fibroblast-derived microRNA passenger strand-enriched exosomes mediate cardiomyocyte hypertrophy. J Clin Investig. 2014;124(5):2136–46.
Frangogiannis NG. Pathophysiology of myocardial infarction. Compr Physiol. 2011;5(4):1841–75.
Wang C, Zhang C, Liu L, et al. Macrophage-derived mir-155-containing exosomes suppress fibroblast proliferation and promote fibroblast inflammation during cardiac injury. Mol Ther. 2017;25(1):192–204. https://doi.org/10.1016/j.ymthe.2016.09.001.
Liu S, Chen J, Shi J, et al. M1-like macrophage-derived exosomes suppress angiogenesis and exacerbate cardiac dysfunction in a myocardial infarction microenvironment. Basic Res Cardiol. 2020;115(2):22. https://doi.org/10.1007/s00395-020-0781-7.
Wang Y, Li C, Zhao R, et al. CircUbe3a from M2 macrophage-derived small extracellular vesicles mediates myocardial fibrosis after acute myocardial infarction. Theranostics. 2021;11(13):6315–33. https://doi.org/10.7150/thno.52843.
Long R, Gao L, Li Y, et al. M2 macrophage-derived exosomes carry miR-1271-5p to alleviate cardiac injury in acute myocardial infarction through down-regulating SOX6. Mol Immunol. 2021;136:26–35.
Dai Y, Wang S, Chang S, et al. M2 macrophage-derived exosomes carry microRNA-148a to alleviate myocardial ischemia/reperfusion injury via inhibiting TXNIP and the TLR4/NF-κB/NLRP3 inflammasome signaling pathway. J Mol Cell Cardiol. 2020;142:65–79.
Wei L, Zhao D. M2 macrophage-derived exosomal miR-145-5p protects against the hypoxia/reoxygenation-induced pyroptosis of cardiomyocytes by inhibiting TLR4 expression. Ann Transl Med. 2022;10(24):1376. https://doi.org/10.21037/atm-22-6109.
Liu H, Gao W, Yuan J, et al. Exosomes derived from dendritic cells improve cardiac function via activation of CD4(+) T lymphocytes after myocardial infarction. J Mol Cell Cardiol. 2016;91:123–33. https://doi.org/10.1016/j.yjmcc.2015.12.028.
Zhang Y, Gao W, Yuan J, et al. CCR7 mediates dendritic-cell-derived exosome migration and improves cardiac function after myocardial infarction. Pharmaceutics. 2023;15(2):461. https://doi.org/10.3390/pharmaceutics15020461.
Liu H, Yu W, Wang C, et al. GW26-e0461 Exosomes secreted from dendritic cells induce angiogenesis by cardiac microvascular endothelial cells after myocardial infarction. J Am Coll Cardiol. 2015;66(16S):C63–C63.
Liu H, Zhang Y, Yuan J, et al. Dendritic cell-derived exosomal miR-494-3p promotes angiogenesis following myocardial infarction. Int J Mol Med. 2021;47(1):315–25.
Laroumanie F, Douin-Echinard V, Pozzo J, et al. CD4+ T cells promote the transition from hypertrophy to heart failure during chronic pressure overload. Circulation. 2014;129(21):2111–24.
Bansal SS, Ismahil MA, Goel M et al. Activated T lymphocytes are essential drivers of pathological remodeling in ischemic heart failure. Circ Heart Fail 2017;10(3):e003688.
Zhao X, Wang J, He J, Tian X, Zhu D, Cai L. Effects of activated CD4+ T cell-derived exosomes on cardiac remodeling after myocardial infarction. Zhonghua Wei Zhong Bing ji jiu yi xue. 2021;33(11):1332–6.
Cai L, Chao G, Li W, et al. Activated CD4+ T cells-derived exosomal miR-142-3p boosts post-ischemic ventricular remodeling by activating myofibroblast. Aging (Albany NY). 2020;12(8):7380.
Rolski F, Czepiel M, Weglarczyk K, Siedlar M, Kania G, Błyszczuk P. Autoreactive T lymphocytes activate cardiac endothelium independently of Tnf-α and cause endothelial dysfunction through exosomes in experimental autoimmune myocarditis. Circulation 2020;142(Suppl_3):A14723-A23.
Rolski F, Czepiel M, Tkacz K et al. T lymphocyte-derived exosomes transport MEK1/2 and ERK1/2 and induce NOX4-dependent oxidative stress in cardiac microvascular endothelial cells. Oxid Med Cell Longev. 2022;2022.
Wang J, Jin M, Ma W-h, Zhu Z, Wang X. The history of telocyte discovery and understanding. Telocytes: Connecting Cells 2016:1–21.
Manole C, Cismaşiu V, Gherghiceanu M, Popescu L. Experimental acute myocardial infarction: telocytes involvement in neo-angiogenesis. J Cell Mol Med. 2011;15(11):2284–96.
Yang J, Li Y, Xue F, Liu W, Zhang S. Exosomes derived from cardiac telocytes exert positive effects on endothelial cells. Am J Transl Res. 2017;9(12):5375.
Liao Z, Chen Y, Duan C, et al. Cardiac telocytes inhibit cardiac microvascular endothelial cell apoptosis through exosomal miRNA-21-5p-targeted cdip1 silencing to improve angiogenesis following myocardial infarction. Theranostics. 2021;11(1):268.
Khan M, Nickoloff E, Abramova T, et al. Embryonic stem cell-derived exosomes promote endogenous repair mechanisms and enhance cardiac function following myocardial infarction. Circ Res. 2015;117(1):52–64. https://doi.org/10.1161/CIRCRESAHA.117.305990.
Kervadec A, Bellamy V, El Harane N, et al. Cardiovascular progenitor-derived extracellular vesicles recapitulate the beneficial effects of their parent cells in the treatment of chronic heart failure. J Heart Lung Transplant. 2016;35(6):795–807. https://doi.org/10.1016/j.healun.2016.01.013.
Huang G. Abstract 139: Systemic analysis and discovery of embryonic stem cell-derived exosomal long non-coding RNAs as potential therapeutic modulators of myocardial repair. Circ Res. 2019;125(Suppl_1):A139-A39. https://doi.org/10.1161/res.125.suppl_1.139p.
Dargani ZT, Singla DK. Embryonic stem cell-derived exosomes inhibit doxorubicin-induced TLR4-NLRP3-mediated cell death-pyroptosis. Am J Physiol-Heart Circ Physiol. 2019;317(2):H460–71.
Wu Q, Wang J, Tan WLW, et al. Extracellular vesicles from human embryonic stem cell-derived cardiovascular progenitor cells promote cardiac infarct healing through reducing cardiomyocyte death and promoting angiogenesis. Cell Death Dis. 2020;11(5):354. https://doi.org/10.1038/s41419-020-2508-y.
Takafuji Y, Hori M, Mizuno T, Harada-Shiba M. Humoral factors secreted from adipose tissue-derived mesenchymal stem cells ameliorate atherosclerosis in Ldlr-/- mice. Cardiovasc Res. 2019;115(6):1041–51. https://doi.org/10.1093/cvr/cvy271.
Bian S, Zhang L, Duan L, Wang X, Min Y, Yu H. Extracellular vesicles derived from human bone marrow mesenchymal stem cells promote angiogenesis in a rat myocardial infarction model. J Mol Med (Berl). 2014;92(4):387–97. https://doi.org/10.1007/s00109-013-1110-5.
Teng X, Chen L, Chen W, Yang J, Yang Z, Shen Z. Mesenchymal stem cell-derived exosomes improve the microenvironment of infarcted myocardium contributing to angiogenesis and anti-inflammation. Cell Physiol Biochem. 2015;37(6):2415–24. https://doi.org/10.1159/000438594.
Arslan F, Lai RC, Smeets MB, et al. Mesenchymal stem cell-derived exosomes increase ATP levels, decrease oxidative stress and activate PI3K/Akt pathway to enhance myocardial viability and prevent adverse remodeling after myocardial ischemia/reperfusion injury. Stem Cell Res. 2013;10(3):301–12. https://doi.org/10.1016/j.scr.2013.01.002.
Cui X, He Z, Liang Z, Chen Z, Wang H, Zhang J. Exosomes from adipose-derived mesenchymal stem cells protect the myocardium against ischemia/reperfusion injury through wnt/beta-catenin signaling pathway. J Cardiovasc Pharmacol. 2017;70(4):225–31. https://doi.org/10.1097/FJC.0000000000000507.
Liu L, Jin X, Hu CF, Li R, Zhou Z, Shen CX. Exosomes derived from mesenchymal stem cells rescue myocardial ischaemia/reperfusion injury by inducing cardiomyocyte autophagy via AMPK and Akt pathways. Cell Physiol Biochem. 2017;43(1):52–68. https://doi.org/10.1159/000480317.
Deng J, Zhang T, Li M, et al. Irisin-pretreated BMMSCs secrete exosomes to alleviate cardiomyocytes pyroptosis and oxidative stress to hypoxia/reoxygenation injury. Curr Stem Cell Res Ther. 2022. https://doi.org/10.2174/1574888X18666221117111829.
Kang K, Ma R, Cai W, et al. Exosomes secreted from CXCR4 overexpressing mesenchymal stem cells promote cardioprotection via Akt signaling pathway following myocardial infarction. Stem Cells Int. 2015;2015: 659890. https://doi.org/10.1155/2015/659890.
Ma J, Zhao Y, Sun L, et al. Exosomes derived from Akt-modified human umbilical cord mesenchymal stem cells improve cardiac regeneration and promote angiogenesis via activating platelet-derived growth factor D. Stem Cells Transl Med. 2017;6(1):51–9. https://doi.org/10.5966/sctm.2016-0038.
Gonzalez-King H, Garcia NA, Ontoria-Oviedo I, Ciria M, Montero JA, Sepulveda P. Hypoxia inducible factor-1alpha potentiates jagged 1-mediated angiogenesis by mesenchymal stem cell-derived exosomes. Stem Cells. 2017;35(7):1747–59. https://doi.org/10.1002/stem.2618.
Zhu J, Lu K, Zhang N, et al. Myocardial reparative functions of exosomes from mesenchymal stem cells are enhanced by hypoxia treatment of the cells via transferring microRNA-210 in an nSMase2-dependent way. Artif Cells Nanomed Biotechnol. 2018;46(8):1659–70. https://doi.org/10.1080/21691401.2017.1388249.
Xu CM, Karbasiafshar C, Brinck Teixeira R, et al. Proteomic assessment of hypoxia-pre-conditioned human bone marrow mesenchymal stem cell-derived extracellular vesicles demonstrates promise in the treatment of cardiovascular disease. Int J Mol Sci. 2023;24(2):1674–774. https://doi.org/10.3390/ijms24021674.
Beermann J, Piccoli MT, Viereck J, Thum T. Non-coding RNAs in development and disease: background, mechanisms, and therapeutic approaches. Physiol Rev. 2016;96(4):1297–325. https://doi.org/10.1152/physrev.00041.2015.
Nakamura Y, Kita S, Tanaka Y, et al. Adiponectin stimulates exosome release to enhance mesenchymal stem-cell-driven therapy of heart failure in mice. Mol Ther. 2020;28(10):2203–19. https://doi.org/10.1016/j.ymthe.2020.06.026.
Li J, Xue H, Li T, et al. Exosomes derived from mesenchymal stem cells attenuate the progression of atherosclerosis in ApoE(-/-) mice via miR-let7 mediated infiltration and polarization of M2 macrophage. Biochem Biophys Res Commun. 2019;510(4):565–72. https://doi.org/10.1016/j.bbrc.2019.02.005.
Ma J, Chen L, Zhu X, Li Q, Hu L, Li H. Mesenchymal stem cell-derived exosomal miR-21a-5p promotes M2 macrophage polarization and reduces macrophage infiltration to attenuate atherosclerosis. Acta Biochim Biophys Sin (Shanghai). 2021;53(9):1227–36. https://doi.org/10.1093/abbs/gmab102.
Yang W, Yin R, Zhu X, et al. Mesenchymal stem-cell-derived exosomal miR-145 inhibits atherosclerosis by targeting JAM-A. Mol Ther Nucleic Acids. 2021;23:119–31. https://doi.org/10.1016/j.omtn.2020.10.037.
Hu J, Jiang Y, Wu X, et al. Exosomal miR-17-5p from adipose-derived mesenchymal stem cells inhibits abdominal aortic aneurysm by suppressing TXNIP-NLRP3 inflammasome. Stem Cell Res Ther. 2022;13(1):349. https://doi.org/10.1186/s13287-022-03037-1.
Luther KM, Haar L, McGuinness M, et al. Exosomal miR-21a-5p mediates cardioprotection by mesenchymal stem cells. J Mol Cell Cardiol. 2018;119:125–37. https://doi.org/10.1016/j.yjmcc.2018.04.012.
Lin F, Zhang S, Liu X, Wu M. Mouse bone marrow derived mesenchymal stem cells-secreted exosomal microRNA-125b-5p suppresses atherosclerotic plaque formation via inhibiting Map4k4. Life Sci. 2021;274: 119249. https://doi.org/10.1016/j.lfs.2021.119249.
Gao H, Yu Z, Li Y, Wang X. miR-100-5p in human umbilical cord mesenchymal stem cell-derived exosomes mediates eosinophilic inflammation to alleviate atherosclerosis via the FZD5/Wnt/beta-catenin pathway. Acta Biochim Biophys Sin (Shanghai). 2021;53(9):1166–76. https://doi.org/10.1093/abbs/gmab093.
Feng Y, Huang W, Wani M, Yu X, Ashraf M. Ischemic preconditioning potentiates the protective effect of stem cells through secretion of exosomes by targeting Mecp2 via miR-22. PLoS ONE. 2014;9(2): e88685. https://doi.org/10.1371/journal.pone.0088685.
Yu B, Kim HW, Gong M, et al. Exosomes secreted from GATA-4 overexpressing mesenchymal stem cells serve as a reservoir of anti-apoptotic microRNAs for cardioprotection. Int J Cardiol. 2015;182:349–60. https://doi.org/10.1016/j.ijcard.2014.12.043.
Shi B, Wang Y, Zhao R, Long X, Deng W, Wang Z. Bone marrow mesenchymal stem cell-derived exosomal miR-21 protects C-kit+ cardiac stem cells from oxidative injury through the PTEN/PI3K/Akt axis. PLoS ONE. 2018;13(2):e0191616-e191716.
Zhu W, Wang Q, Zhang J et al. Exosomes derived from miR-214–3p overexpressing mesenchymal stem cells promote myocardial repair. 2021. https://doi.org/10.21203/rs.3.rs-721088/v2.
Wen Z, Mai Z, Zhu X, et al. Mesenchymal stem cell-derived exosomes ameliorate cardiomyocyte apoptosis in hypoxic conditions through microRNA144 by targeting the PTEN/AKT pathway. Stem Cell Res Ther. 2020;11(1):36. https://doi.org/10.1186/s13287-020-1563-8.
Peng Y, Zhao JL, Peng ZY, Xu WF, Yu GL. Exosomal miR-25-3p from mesenchymal stem cells alleviates myocardial infarction by targeting pro-apoptotic proteins and EZH2. Cell Death Dis. 2020;11(5):317. https://doi.org/10.1038/s41419-020-2545-6.
Chen G, Wang M, Ruan Z, Zhu L, Tang C. Mesenchymal stem cell-derived exosomal miR-143-3p suppresses myocardial ischemia-reperfusion injury by regulating autophagy. Life Sci. 2021;280: 119742. https://doi.org/10.1016/j.lfs.2021.119742.
Wang W, Peng X, Zhao L, Zhao H, Gu Q. Extracellular vesicles from bone marrow mesenchymal stem cells inhibit apoptosis and autophagy of ischemia-hypoxia cardiomyocyte line in vitro by carrying miR-144-3p to inhibit ROCK1. Curr Stem Cell Res Ther. 2023;18(2):247–59. https://doi.org/10.2174/1574888X17666220503192941.
Cheng H, Chang S, Xu R, et al. Hypoxia-challenged MSC-derived exosomes deliver miR-210 to attenuate post-infarction cardiac apoptosis. Stem Cell Res Ther. 2020;11(1):224. https://doi.org/10.1186/s13287-020-01737-0.
Song BW, Lee CY, Kim R, et al. Multiplexed targeting of miRNA-210 in stem cell-derived extracellular vesicles promotes selective regeneration in ischemic hearts. Exp Mol Med. 2021;53(4):695–708. https://doi.org/10.1038/s12276-021-00584-0.
Ma T, Chen Y, Chen Y, et al. MicroRNA-132, delivered by mesenchymal stem cell-derived exosomes, promote angiogenesis in myocardial infarction. Stem Cells Int. 2018;2018:3290372. https://doi.org/10.1155/2018/3290372.
Chen Q, Liu Y, Ding X, et al. Bone marrow mesenchymal stem cell-secreted exosomes carrying microRNA-125b protect against myocardial ischemia reperfusion injury via targeting SIRT7. Mol Cell Biochem. 2020;465(1–2):103–14. https://doi.org/10.1007/s11010-019-03671-z.
Zhu LP, Tian T, Wang JY, et al. Hypoxia-elicited mesenchymal stem cell-derived exosomes facilitates cardiac repair through miR-125b-mediated prevention of cell death in myocardial infarction. Theranostics. 2018;8(22):6163–77. https://doi.org/10.7150/thno.28021.
Zhao J, Li X, Hu J, et al. Mesenchymal stromal cell-derived exosomes attenuate myocardial ischaemia-reperfusion injury through miR-182-regulated macrophage polarization. Cardiovasc Res. 2019;115(7):1205–16. https://doi.org/10.1093/cvr/cvz040.
Sun C, Li W, Li Y, et al. MiR-182-5p Mediated by exosomes derived from bone marrow mesenchymal stem cell attenuates inflammatory responses by targeting TLR4 in a mouse model of myocardial infraction. Immune Netw. 2022;22(6): e49. https://doi.org/10.4110/in.2022.22.e49.
Yue R, Lu S, Luo Y, et al. Mesenchymal stem cell-derived exosomal microRNA-182-5p alleviates myocardial ischemia/reperfusion injury by targeting GSDMD in mice. Cell Death Discov. 2022;8(1):202. https://doi.org/10.1038/s41420-022-00909-6.
Sun L, Zhu W, Zhao P et al. Down-regulated exosomal microRNA-221–3p derived from senescent mesenchymal stem cells impairs heart repair. Front Cell Develop Biol. 2020.
Wang Y, Shen Y. Exosomal miR-455-3p from BMMSCs prevents cardiac ischemia-reperfusion injury. Hum Exp Toxicol. 2022;41:9603271221102508. https://doi.org/10.1177/09603271221102508.
Sun XH, Wang X, Zhang Y, Hui J. Exosomes of bone-marrow stromal cells inhibit cardiomyocyte apoptosis under ischemic and hypoxic conditions via miR-486-5p targeting the PTEN/PI3K/AKT signaling pathway. Thromb Res. 2019;177:23–32. https://doi.org/10.1016/j.thromres.2019.02.002.
Liu J, Wu J, Li L, Li T, Wang J. The role of exosomal non-coding RNAs in coronary artery disease. Front Pharmacol. 2020.
Wang K, Jiang Z, Webster KA, et al. Enhanced cardioprotection by human endometrium mesenchymal stem cells driven by exosomal microRNA-21. Stem Cells Transl Med. 2017;6(1):209–22. https://doi.org/10.5966/sctm.2015-0386.
Ning W, Li S, Yang W, et al. Blocking exosomal miRNA-153-3p derived from bone marrow mesenchymal stem cells ameliorates hypoxia-induced myocardial and microvascular damage by targeting the ANGPT1-mediated VEGF/PI3k/Akt/eNOS pathway. Cell Signal. 2021;77: 109812. https://doi.org/10.1016/j.cellsig.2020.109812.
Huang Y. Exosomal lncRNAs from mesenchymal stem cells as the novel modulators to cardiovascular disease. Stem Cell Res Ther. 2020;11:1–2.
Liu W, Higashikuni Y, Sata M. Linking RNA dynamics to heart disease: the lncRNA/miRNA/mRNA axis in myocardial ischemia-reperfusion injury. Hypertens Res. 2022;45(6):1067–9. https://doi.org/10.1038/s41440-022-00905-4.
Zhang N, Luo Y, Zhang H, Zhang F, Gao X, Shao J. Exosomes derived from mesenchymal stem cells ameliorate the progression of atherosclerosis in ApoE(-/-) mice via FENDRR. Cardiovasc Toxicol. 2022;22(6):528–44. https://doi.org/10.1007/s12012-022-09736-8.
Sun L, He X, Zhang T, Han Y, Tao G. Knockdown of mesenchymal stem cell-derived exosomal LOC100129516 suppresses the symptoms of atherosclerosis via upregulation of the PPARgamma/LXRalpha/ABCA1 signaling pathway. Int J Mol Med. 2021;48(6):208–308. https://doi.org/10.3892/ijmm.2021.5041.
Yan B, Liu T, Yao C, Liu X, Du Q, Pan L. LncRNA XIST shuttled by adipose tissue-derived mesenchymal stem cell-derived extracellular vesicles suppresses myocardial pyroptosis in atrial fibrillation by disrupting miR-214-3p-mediated Arl2 inhibition. Lab Invest. 2021;101(11):1427–38. https://doi.org/10.1038/s41374-021-00635-0.
Li M, Ding W, Liu G, Wang J. Extracellular circular RNAs act as novel first messengers mediating cell cross-talk in ischemic cardiac injury and myocardial remodeling. J Cardiovasc Transl Res. 2022;15(3):444–55. https://doi.org/10.1007/s12265-022-10219-y.
Mao Q, Liang XL, Zhang CL, Pang YH, Lu YX. LncRNA KLF3-AS1 in human mesenchymal stem cell-derived exosomes ameliorates pyroptosis of cardiomyocytes and myocardial infarction through miR-138-5p/Sirt1 axis. Stem Cell Res Ther. 2019;10(1):393. https://doi.org/10.1186/s13287-019-1522-4.
Sun L, Zhu W, Zhao P, et al. Long noncoding RNA UCA1 from hypoxia-conditioned hMSC-derived exosomes: a novel molecular target for cardioprotection through miR-873-5p/XIAP axis. Cell Death Dis. 2020;11(8):696. https://doi.org/10.1038/s41419-020-02783-5.
Huang P, Wang L, Li Q, et al. Atorvastatin enhances the therapeutic efficacy of mesenchymal stem cells-derived exosomes in acute myocardial infarction via up-regulating long non-coding RNA H19. Cardiovasc Res. 2020;116(2):353–67. https://doi.org/10.1093/cvr/cvz139.
Chen H, Xia W, Hou M. LncRNA-NEAT1 from the competing endogenous RNA network promotes cardioprotective efficacy of mesenchymal stem cell-derived exosomes induced by macrophage migration inhibitory factor via the miR-142-3p/FOXO1 signaling pathway. Stem Cell Res Ther. 2020;11(1):31. https://doi.org/10.1186/s13287-020-1556-7.
Zhang JK, Zhang Z, Guo ZA, et al. The BMSC-derived exosomal lncRNA Mir9-3hg suppresses cardiomyocyte ferroptosis in ischemia-reperfusion mice via the Pum2/PRDX6 axis. Nutr Metab Cardiovasc Dis. 2022;32(2):515–27. https://doi.org/10.1016/j.numecd.2021.10.017.
Ma J, Lei P, Chen H, et al. Advances in lncRNAs from stem cell-derived exosome for the treatment of cardiovascular diseases. Front Pharmacol. 2022;13: 986683. https://doi.org/10.3389/fphar.2022.986683.
Hu X, Qin H, Yan Y, et al. Exosomal circular RNAs: biogenesis, effect, and application in cardiovascular diseases. Front Cell Dev Biol. 2022;10: 948256. https://doi.org/10.3389/fcell.2022.948256.
Li CX, Song J, Li X, Zhang T, Li ZM. Circular RNA 0001273 in exosomes derived from human umbilical cord mesenchymal stem cells (UMSCs) in myocardial infarction. Eur Rev Med Pharmacol Sci. 2020;24(19):10086–95. https://doi.org/10.26355/eurrev_202010_23228.
Tian T, Li F, Chen R, Wang Z, Su X, Yang C. Therapeutic potential of exosomes derived from circRNA_0002113 lacking mesenchymal stem cells in myocardial infarction. Front Cell Develop Biol. 2022.
Li J, Jiang R, Hou Y, Lin A. Mesenchymal stem cells-derived exosomes prevent sepsis-induced myocardial injury by a CircRTN4/miR-497-5p/MG53 pathway. Biochem Biophys Res Commun. 2022;618:133–40. https://doi.org/10.1016/j.bbrc.2022.05.094.
Zhou D, Dai Z, Ren M, Yang M. Adipose-derived stem cells-derived exosomes with high amounts of Circ_0001747 alleviate hypoxia/reoxygenation-induced injury in myocardial cells by targeting MiR-199b-3p/MCL1 Axis. Int Heart J. 2022;63(2):356–66. https://doi.org/10.1536/ihj.21-441.
Romano V, Belviso I, Sacco AM, et al. Human cardiac progenitor cell-derived extracellular vesicles exhibit promising potential for supporting cardiac repair in vitro. Front Physiol. 2022;13: 879046. https://doi.org/10.3389/fphys.2022.879046.
Barile L, Cervio E, Lionetti V, et al. Cardioprotection by cardiac progenitor cell-secreted exosomes: role of pregnancy-associated plasma protein-A. Cardiovasc Res. 2018;114(7):992–1005. https://doi.org/10.1093/cvr/cvy055.
Milano G, Biemmi V, Lazzarini E, et al. Intravenous administration of cardiac progenitor cell-derived exosomes protects against doxorubicin/trastuzumab-induced cardiac toxicity. Cardiovasc Res. 2020;116(2):383–92. https://doi.org/10.1093/cvr/cvz108.
Gray WD, French KM, Ghosh-Choudhary S, et al. Identification of therapeutic covariant microRNA clusters in hypoxia-treated cardiac progenitor cell exosomes using systems biology. Circ Res. 2015;116(2):255–63. https://doi.org/10.1161/CIRCRESAHA.116.304360.
Chen L, Wang Y, Pan Y, et al. Cardiac progenitor-derived exosomes protect ischemic myocardium from acute ischemia/reperfusion injury. Biochem Biophys Res Commun. 2013;431(3):566–71. https://doi.org/10.1016/j.bbrc.2013.01.015.
Ibrahim AG, Cheng K, Marban E. Exosomes as critical agents of cardiac regeneration triggered by cell therapy. Stem Cell Reports. 2014;2(5):606–19. https://doi.org/10.1016/j.stemcr.2014.04.006.
Namazi H, Mohit E, Namazi I, et al. Exosomes secreted by hypoxic cardiosphere-derived cells enhance tube formation and increase pro-angiogenic miRNA. J Cell Biochem. 2018;119(5):4150–60. https://doi.org/10.1002/jcb.26621.
Hirai K, Ousaka D, Fukushima Y, et al. Cardiosphere-derived exosomal microRNAs for myocardial repair in pediatric dilated cardiomyopathy. Sci Transl Med 2020;12(573):eabb3336-eabb36 doi: https://doi.org/10.1126/scitranslmed.abb3336.
Adamiak M, Cheng G, Bobis-Wozowicz S, et al. Induced pluripotent stem cell (iPSC)-derived extracellular vesicles are safer and more effective for cardiac repair than iPSCs. Circ Res. 2018;122(2):296–309. https://doi.org/10.1161/CIRCRESAHA.117.311769.
Wang Y, Zhang L, Li Y, et al. Exosomes/microvesicles from induced pluripotent stem cells deliver cardioprotective miRNAs and prevent cardiomyocyte apoptosis in the ischemic myocardium. Int J Cardiol. 2015;192:61–9. https://doi.org/10.1016/j.ijcard.2015.05.020.
•• Gao L, Wang L, Wei Y et al. Exosomes secreted by hiPSC-derived cardiac cells improve recovery from myocardial infarction in swine. Sci Transl Med 2020;12(561):eaay1318-eaay18. https://doi.org/10.1126/scitranslmed.aay1318. A comprehensive study about the comparison of iPSCs-derived exosomes and iPSCs, in cardiac repair. In vivo findings demonstrate the potential of iPSCs-derived exosomes to prevent complications from cell transplantation.
• Kurtzwald-Josefson E, Zeevi-Levin N, Rubchevsky V et al. Cardiac fibroblast-induced pluripotent stem cell-derived exosomes as a potential therapeutic mean for heart failure. Int J Mol Sci 2020;21(19):7215–15. https://doi.org/10.3390/ijms21197215. Diversities the in cargo content of iPSCs-derived exosomes which are programmed from fibroblasts that are isolated from distinct tissues have been shown to affect cardiac regeneration potential.
El Harane N, Kervadec A, Bellamy V, et al. Acellular therapeutic approach for heart failure: in vitro production of extracellular vesicles from human cardiovascular progenitors. Eur Heart J. 2018;39(20):1835–47. https://doi.org/10.1093/eurheartj/ehy012.
Liu B, Lee BW, Nakanishi K, et al. Cardiac recovery via extended cell-free delivery of extracellular vesicles secreted by cardiomyocytes derived from induced pluripotent stem cells. Nat Biomed Eng. 2018;2(5):293–303. https://doi.org/10.1038/s41551-018-0229-7.
Lee WH, Chen WY, Shao NY, et al. Comparison of non-coding RNAs in exosomes and functional efficacy of human embryonic stem cell- versus induced pluripotent stem cell-derived cardiomyocytes. Stem Cells. 2017;35(10):2138–49. https://doi.org/10.1002/stem.2669.
Li H, Gu J, Sun X, Zuo Q, Li B, Gu X. Isolation of swine bone marrow lin-/CD45-/CD133 + cells and cardio-protective effects of its exosomes. Stem Cell Rev Rep. 2023;19(1):213–29. https://doi.org/10.1007/s12015-022-10432-x.
Xiong Y, Yang C, Yang X, Ding C, Wang Q, Zhu H. LncRNA MIR9-3HG enhances LIMK1 mRNA and protein levels to contribute to the carcinogenesis of lung squamous cell carcinoma via sponging miR-138-5p and recruiting TAF15. Pathol Res Pract. 2022;237: 153941. https://doi.org/10.1016/j.prp.2022.153941.
Yang T, Long T, Du T, Chen Y, Dong Y, Huang ZP. Circle the cardiac remodeling with circRNAs. Front Cardiovasc Med. 2021;8: 702586. https://doi.org/10.3389/fcvm.2021.702586.
Author information
Authors and Affiliations
Contributions
These authors contributed equally to this work.
Corresponding author
Ethics declarations
Conflict of Interest
Selcen Ari Yuka declares that she has no conflict of interest. Ayca Aslan declares that she has no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yuka, S.A., Aslan, A. Current Approaches in Cardiac Repair: Somatic and Stem Cell Exosomes. Curr Treat Options Cardio Med 25, 689–714 (2023). https://doi.org/10.1007/s11936-023-01021-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11936-023-01021-3