Abstract
Purpose of Review
In this review we aim to summarize the latest findings on the network of molecules produced by muscle and bone under physiological and pathological conditions.
Recent Findings
The concomitant onset of osteoporosis and sarcopenia is currently one of the main threats that can increase the risk of falling fractures during aging, generating high health care costs due to hospitalization for bone fracture surgery. With the growing emergence of developing innovative therapies to treat these two age-related conditions that often have common onset, a broader understanding of molecular messengers regulating the communication between muscle and bone tissue became imperative.
Summary
Recently it has been highlighted that two muscle-derived signals, such as the myokines Irisin and L-BAIBA, positively affect bone tissue. In parallel, there are signals derived from bone that affect either positively the skeletal muscle, such as osteocalcin, or negatively, such as RANKL.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Given their anatomical proximity, the closely coupled muscle and bone tissues communicate through mechanical interaction and via a finely tuned network of molecules released by both tissues in physiological and pathological conditions [1, 2].
The mechanical load, which is simply applied in an upright posture, reaches its maximum benefit while exercising. Regular physical activity, due to synchronous reinforcement of bone and muscle mass, improves overall health and can prevent or delay several diseases, including osteoporosis, sarcopenia, diabetes, and obesity [3, 4]. Numerous prospective studies have confirmed that mortality can be reduced by 20–40% by walking or running activities [5, 6] slowing down the progression of age-related conditions [3].
Since it is well documented that increased exercise, hence mechanical loading, has anabolic effects on muscle and bone, as well as states of unloading or immobility cause catabolic responses, the scientific community is currently making efforts to investigate the molecular mediators of the bone–muscle crosstalk.
The first two muscle-secreted factors identified during the last decade of research and termed myokines were myostatin and interleukin 6. It has been shown that myostatin, a suppressor of skeletal muscle mass and development [7], also negatively regulates bone mass. Myostatin knockout mice display improved bone strength following physical activity [8] and increased differentiation of bone marrow mesenchymal cells into osteoblasts [9]. Interleukin 6 (IL-6), highly expressed in skeletal muscle [10], enhances bone resorption, most likely by increasing RANKL gene expression in osteoblasts [11] but, at the same time, increases differentiation of early osteoblasts [12].
First reports showing that bone cells produce factors that affect muscle are instead more recently dated. Among these, prostaglandin E2 and Wnt 3a, two molecules produced by osteocytes in response to shear stress, were found capable of stimulating myogenesis and muscle function [13].
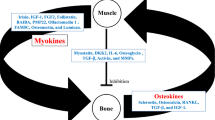
In this review, we will focus on two novel muscle-derived signals, such as the myokines Irisin [14] and L-BAIBA [15••] that can directly affect bone. We will also discuss very recent evidence regarding signals derived from bone that can affect skeletal muscle with positive effects, such as osteocalcin [16••], and with negative effects, such as RANKL [17••] (Fig. 1).
Schematic representation of the network of molecules produced by skeletal muscle and bone tissues. Some muscle-derived signals, such as the myokines Irisin and L-BAIBA, which positively (+) affect bone tissue by increasing the activity of osteoblasts (OBs) and preventing the apoptosis of osteocytes (OTs). In parallel, there are signals derived from bone that affect either positively the skeletal muscle, such as osteocalcin, which improves muscle functions during exercise, or negatively (−), such as RANKL, which reduces muscle function and strength, and glucose uptake, We thank Servier Medical Art (https://smart.servier.com/) for providing free image software to build the figure
A better understanding of molecular mechanisms underlying the actions of these messengers would make possible in the future to develop innovative therapies, especially to treat the two twin conditions of aging that commonly affect both tissues, such as osteoporosis and sarcopenia.
Crosstalk from Muscle to Bone
Irisin
The myokine irisin is produced under skeletal muscle contraction in the form of the precursor, the fibronectin type III domain-containing protein 5 (FNDC5), which is then cleaved and released into the bloodstream. Irisin stimulates trans-differentiation of white adipose tissue into a brown adipose tissue-like, by increasing cellular mitochondrial density and expression of uncoupling protein-1 to promote thermogenesis and energy expenditure [18]. Our studies demonstrated that a weekly irisin dose lower than that required for WAT trans-differentiation increases cortical bone mass and strength in mice, thus suggesting that the bone was the primary target organ of the action of Irisin [19].
Both young healthy mice and osteoporotic murine models have been studied to rule out the effects of treatment with exogenous recombinant irisin (rec-irisin). In young mice rec-irisin clearly recapitulated the effects of exercise by improving cortical bone geometry, increasing bending strength and resistance to fracture [19]. In hindlimb unloaded mice, a murine model which mimics adverse effects on musculoskeletal system caused by physical immobility or microgravity [20, 21], intermittent administration of rec-irisin preserved cortical and trabecular bone mineral density, bone volume fraction (BV/TV), and Fractal dimension [22••]. Molecular studies showed that this effect was mainly mediated through inhibition of unloading-induced sclerostin increase and osteoprotegerin decrease [22••]. More recently, we also showed that irisin prevented disuse-induced reduction of viable osteocytes and Caspase-9 and Caspase-3 activations in cortical bone of hindlimb unloaded mice [23]. This murine model is also characterized by muscular atrophy, whose onset was fully prevented by rec-Irisin treatment which preserved muscle integrity, fiber size and the expression of myosin Type II (MyHC II), nuclear respiratory factor 1 (NRF1), and mitochondrial transcription factor A (TFAM) [22••].
We have previously shown that the highest expression of the Irisin precursor, FNDC5, was detectable in muscle tissue and that mice treated with irisin showed a higher number of FNDC5 positive fibers than control mice, suggesting that its autocrine action may amplify its synthesis [19]. The autocrine action of irisin has been also studied in vitro on C2C12 myotubes, in which the a 24-h treatment with rec-irisin significantly increased the expression of the proliferator-activator peroxisome γ receptor-activator-1α (PGC-1α), NRF1, and TFAM, leading to an increase in mitochondrial content and oxygen consumption [24]. Other in vitro studies have shown that an enhancement of FNDC5 mRNA expression and irisin secretion occurs during myogenic differentiation and that treatment with irisin increased insulin-like growth factor 1 (IGF-1) and decreased myostatin gene expression via the ERK pathway [25].
In humans, we and other authors observed a positive correlation between irisin and bone mineral density in young athletes [26, 27]. In soccer players, we found higher linear association at the lumbar vertebrae, right arm, and head than at the femurs. As these bone segments have a lower impact from mechanical loading in soccer, the surprising result indicated that the effect of irisin on bone might be systemic, rather than strictly connected to bone sites where load is applied [27]. We also found in 96 children diagnosed with childhood type 1 diabetes mellitus under insulin replacement therapy that irisin serum levels were positively associated with bone quality and the improvement of glycemic control [28]. Soininen et al. showed that irisin was one of the determinants of bone mineral density in a population of 6–8 years old children [29] In line with this, we found in healthy children that irisin positively correlated with circulating osteocalcin, and negatively with DKK1, one of the bone anabolic inhibitors of the WNT pathway [30]. Additionally, multivariate regression analysis showed that Irisin was a greater determinant of bone mineral status than bone alkaline phosphatase [31]. In adults, irisin was negatively associated with serum sclerostin levels in patients with prediabetes [32] and with vertebral fragility fractures [33, 34] and sarcopenia [35] in post-menopausal women. We also observed reduced irisin levels in osteoporotic women with hyperparathyroidism compared with controls [36••]. In vitro data further supported this observation showing that Teriparatide (1–34 PTH) treatment decreased the expression of FNDC5 in C2C12 myotubes by acting on PTH receptor, which in turn activates Erk1/2 phosphorylation, most likely through the increase of intracellular cAMP [36••]. On the other hand, rec-irisin reduced PTH receptor expression on osteoblasts by 50% compared with untreated cells, suggesting that irisin can exert its anabolic effect on bone, not only by stimulating osteoblast activity but also by inhibiting the catabolic action of PTH on these cells [36••].
Despite debates and controversies since its discovery, overall the scientific consensus has now unanimously recognized that irisin plays a crucial role in bone metabolism; therefore, Irisin could be a possible serum marker of bone status and a therapeutic option to treat bone diseases in the future.
L-BAIBA
In 2014, Roberts et al. described BAIBA as a small molecule (103.6-Da) produced by skeletal muscle during exercise. BAIBA may be present in the form of two enantiomers: L-BAIBA and D-BAIBA. Using amino-acid L-valine as an energy source, L-BAIBA is produced through the control of the transcriptional co-activator PGC-1α. It has been demonstrated that L-BAIBA is a mediator of the beneficial effect of exercise from skeletal muscle to other organs, especially bone, in an endocrine manner [37].
Experimental evidence showed that BAIBA is involved in the β-oxidation pathway of fatty acids in the liver, is implicated in the browning of white adipose tissue [37], prevents diet-induced obesity [38], plays a role in protecting against metabolic disorders in type 2 diabetes [39], and increases insulin resistance and inflammation of skeletal muscle [40].
In a recent study, L-BAIBA has been described as a molecule that protects osteocytes from cell death. Kitase et al. have shown that only the enantiomer L-BAIBA is produced by contracted muscle independently of skeletal muscle type, sex, and age. In this study, the authors demonstrated that treatment with L-BAIBA on osteocytes in vitro protects against apoptosis induced by exposure to reactive oxygen species [15••]. In hindlimb unloaded mice, in which bone loss is characterized by osteocyte apoptosis [41], it has been observed a reduction in trabecular bone volume and increased osteocyte apoptosis when mice received normal drinking water, whereas in mice receiving drinking water supplemented with L-BAIBA (100 mg/kg/day), the bone volume and viability of osteocytes were preserved [15••]. Osteoblasts appear to be the main mediators of the molecule effect on bone volume, whereas no changes in the number of osteoclasts and their activity as well as changes in the levels of the nuclear factor receptor activator κΒ (RANK)-ligand have been observed in male or female mice following treatment with L-BAIBA.
L-BAIBA has been shown to bind to glycine receptors and to the MRGPRD, which is also known to be a receptor for β-alanine and gamma-Aminobutyric acid (GABA). Kitase et al. found that MU6840, an antagonist of MRGPRD, blocked the beneficial effect of L-BAIBA on ROS-induced cell death [42]. Potentially L-BAIBA may have a direct effect on osteoblasts, but since osteocytes express much higher levels of MRGPRD, the effects on osteoblasts may be indirect and mediated by osteocytes [15••]. Furthermore, the MRGPRD receptor is strongly expressed on osteocytes in young mice, but its expression is reduced in old mice, implying that osteocytes lose their response to BAIBA with aging, despite muscles of young and old mice both produce L-BAIBA under contraction [15••]. Data currently suggest that L-BAIBA-mediated effects of exercise can increase bone mass and strength in younger subjects, but this positive effect on bones would be significantly reduced with age.
L-BAIBA also plays autocrine functions on skeletal muscle. It has been reported that this myokine mitigates insulin resistance and inflammation and stimulates fatty acid oxidation via the AMP-activated protein kinase (AMPK) and peroxisome proliferator-activated receptor δ (PPARδ) signaling pathway in skeletal muscle [40]. It has been also shown that L-BAIBA increased the muscle contractile strength of the extensor digitorum longus and soleus muscles in male mice but not in females. Possible gender differences in response to L-BAIBA have been hypothesized, as musculoskeletal responses to exercise can change between males and females [43].
In summary, L-BAIBA is a novel muscle-derived molecule that can exert both direct and indirect effects on bone cells, also preserving muscle strength especially in male mice [15••].
Crosstalk from Bone to Muscle
RANKL
The increased levels of the nuclear factor kappa-B ligand receptor activator (RANKL) in menopausal women play a primary role in the development of osteoporosis [44], as this molecule, by binding to its receptor (RANK), activates the differentiation, activity, and survival of bone-resorbing cells, the osteoclasts [45]. On the other hand, osteoprotegerin (OPG), a soluble receptor for RANKL, prevents its binding to RANK, thus inhibiting osteoclastogenesis. Denosumab (Dmab), a RANKL-blocking antibody mimicking the OPG action, has been shown to reduce the risk of fracture and is widely used for the treatment of osteoporosis [46]. Recently, it has been observed that the fall rate was also reduced in patients receiving Dmab compared with placebo, but this effect has remained for a long time only an unexplained observation [47, 48].
The receptor for RANKL is also expressed in skeletal muscle and its activation mainly inhibits myogenic differentiation, resulting in skeletal muscle dysfunction [49, 50]. In turn, exogenous OPG administration has been shown to reduce inflammation and to restore skeletal muscle function in mdx mice, a mouse model of Duchenne muscular dystrophy [51, 52].
A recent study showed that in post-menopausal osteoporotic women treated with Dmab or Bisphosphonates, both treatment increased bone mineral density at lumbar spine; however, only Dmab significantly increased appendicular lean mass and improved handgrip strength. In transgenic mice over-expressing RANKL, bone loss was associated with impairment of muscle function and strength, and glucose absorption [17••]. These alterations were coupled with increased expression of factors inhibiting muscle growth and function, such as myostatin [17••]. In line with these data, other findings showed that conditional RANK deletion in muscle prevented muscle atrophy and dysfunction induced by denervation in mice [53]. Taken together, these data established that the RANK/RANKL/OPG system also plays a crucial role in muscle metabolism and open new opportunities to further investigate benefits of RANKL inhibition for the treatment of sarcopenia.
OSTEOCALCIN
Osteocalcin is a protein secreted in a carboxylated form solely by osteoblasts [54]. It is a key factor responsible for the mineralization of extracellular matrix and its serum levels are clinically used as marker of osteoblastic bone formation [55].
In 1996, Ducy et al. observed that mice carrying deletion of the osteocalcin gene (osteocalcin knockout mice) had more visceral fat, were poor breeders, and generated litters with significantly fewer pups than wild-type littermates. To date, several roles of osteocalcin have been showed by both in vitro and in vivo studies revealing that the undercarboxylated (bioactive) form of osteocalcin controls numerous physiological processes in an endocrine fashion [56]. Karsenty’s group observed that bioactive osteocalcin induces insulin production in pancreatic islets as well as adiponectin expression in adipocytes [57,58,59]. Moreover, it has been observed that deletion in osteoblasts of the insulin receptor mimics the phenotype observed in osteocalcin knockout mice. These findings suggested that the insulin-mediated osteocalcin expression in osteoblasts could further stimulate insulin secretion in pancreatic islet cells, thus forming an endocrine pancreatic bone loop [60]. In line with this, it has been demonstrated that glucose tolerance in mice on a high-fat diet was restored by daily injections of osteocalcin [61, 62].
In 2016, the scientific evidence that osteocalcin levels increased in mice and humans during physical activity and at the same time its levels decreased during aging led to the discovery of a remarkable function of this bone-derived hormone [16]. Mera et al. showed that treatment with exogenous osteocalcin, whether administered acutely or chronically, increased the exercise capacity of 3-month-old mice and restored the exercise capacity of 9-, 12- and 15-month-old mice. The authors showed that osteocalcin promotes the expression of fatty acid transporters, stimulates β-oxidation, and the translocation of the GLUT4 glucose transporter to the plasma membrane by promoting the absorption of glucose and stimulating catabolism in the skeletal muscle [63]. Osteocalcin also stimulates the synthesis of IL-6 [1], a myokine whose circulating levels increase during exercise, which in turn promotes adaptation to exercise by stimulating the production of osteocalcin in bone [63].
More recently, it has been observed that osteocalcin is necessary for brain development and functions. Its absence makes the brain, particularly the hippocampus, smaller and less developed and, in addition, mice with osteocalcin deficiency are less active and suffer from anxiety and memory impairment compared with wild-type mice [64]. The regulation of cognitive functions by osteocalcin, together with the observation that its circulating levels decrease in mid-life compared with adolescence in all tested species, suggested that osteocalcin may be an anti-geronic hormone effective in preventing age-related cognitive decline [64]. The numerous functions that osteocalcin regulates in the brain during development and after childbirth have long required identification of its receptor, the G protein-coupled receptor 158, which has recently been discovered [65]. Remarkably, the same group hypothesized that bone evolved, in part, to improve the ability of bone vertebrates to escape dangerous situations. In 2019, it has been reported that in rodents and humans exposed to various types of stress, there is a rapid wave of circulating bioactive osteocalcin which mediated acute stress response by inhibiting parasympathetic tone [66]. More specifically, the authors reported that when the amygdala in brain perceives danger, it transmits a signal to osteoblasts to secrete bioactive osteocalcin in the bloodstream. In turn osteocalcin reduces the activity of the nerve fibers of the parasympathetic nervous system, triggers the body’s stress response and the release of adrenaline resulting in surges in both heart rate and breathing, as reaction to the threat. These results would explain why in adrenalectomized rodents, lacking both adrenal steroid hormones and adrenal-derived catecholamine, and in glucocorticoid-deficient patients, the acute stress response to dangerous situation is still observed [66].
Although the many functions of osteocalcin seem unrelated, its ability to improve muscle function during exercise, promote memory and facilitate the acute response to stress, suggest that this hormone of exclusive bone derivation gives a survival advantage which, from a broader point of view, could result in a slowdown in the onset of age-related diseases.
Conclusion
Throughout life, from embryogenesis to aging, bone and muscle tissue cooperate mechanically and biochemically as a single unit. During aging, it is known that osteoporosis often coexists with sarcopenia, creating a negative loop between muscle and bone responsible for reducing quality of life and increasing mortality. However, it is not yet clear if the pathological conditions develop at the same time or if one precedes the other. From a mechanical point of view, it is more likely that the decline in muscle function, resulting in decreased load on the skeleton, is responsible for the loss of bone mass. However, there are osteoporotic patients who have not been diagnosed with sarcopenia. The most likely hypothesis currently under investigation is that age-dependent reduction in regenerative capacity of both tissues could be a shared mechanism for sarcopenia and osteoporosis. The decrease of regenerative capacity of both tissues implies a parallel dysregulation of their biochemical communication through the musculoskeletal secretome. Hopefully a better understanding of these molecular entities involved in the crosstalk between these tissues can shift the paradigm for the treatment of osteoporosis and sarcopenia simultaneously.
References
Papers of particular interest, published recently, have been highlighted as: •• Of major importance
Pedersen BK, Febbraio MA. Muscles, exercise and obesity: skeletal muscle as a secretory organ. Nat Rev Endocrinol. 2012;8:457–65.
Pedersen BK, Akerström TC, Nielsen AR, Fischer CP. Role of myokines in exercise and metabolism. J Appl Physiol. 2007;103:1093–8.
Dunstan D. Diabetes: exercise and T2DM-move muscles more often! Nat Rev Endocrinol. 2011;7:189–90.
Egan B, Zierath JR. Exercise metabolism and the molecular regulation of skeletal muscle adaptation. Cell Metab. 2013;17:162–84.
Arem H, Moore SC, Patel A, Hartge P, Berrington de Gonzalez A, Visvanathan K, et al. Leisure time physical activity and mortality: a detailed pooled analysis of the dose-response relationship. JAMA Intern Med. 2015;175:959–67.
O’Donovan G, Lee IM, Hamer M, Stamatakis E. Association of “weekend warrior” and other leisure time physical activity patterns with risks for all-cause, cardiovascular disease, and Cancer mortality. JAMA Intern Med. 2017;177:335–42.
McPherron AC, Lawler AM, Lee SJ. Regulation of skeletal muscle mass in mice by a new TGF-β superfamily member. Nature. 1997;387:83–90.
Hamrick MW, Samaddar T, Pennington C, McCormick J. Increased muscle mass with myostatin deficiency improves gains in bone strength with exercise. J Bone Miner Res. 2006;21:477–83.
Hamrick MW, Shi X, Zhang W, Pennington C, Thakore H, Haque M, et al. Loss of myostatin (GDF-8) function increases osteogenic differentiation of bone marrow-derived stem cells but the osteogenic effect is ablated with unloading. Bone. 2007;40:1544–53.
Jonsdottir IH, Schjerling P, Ostrowski K, Asp S, Richter EA, Pedersen BK. Muscle contractions induce interleukin-6 mRNA production in rat skeletal muscles. J Physiol. 2000;528(Pt 1):157–63.
Udagawa N, Takahashi N, Katagiri T, Tamura T, Wada S, Findlay DM, et al. Interleukin (IL)-6 induction of osteoclast differentiation depends on IL-6 receptors expressed on osteoblastic cells but not on osteoclast progenitors. J Exp Med. 1995;182(5):1461–8.
Hiscock N, Chan MH, Biscci T, Darby IA, Febbraio MA. Skeletal myocytes are a source of interleukin-6 mRNA expression and protein release during contraction: evidence of fiber type specificity. FASEB J. 2004;18:992–4.
Mo C, Romero-Suarez S, Bonewald L, Johnson M, Brotto M. Prostaglandin E2: from clinical applications to its potential role in bone-muscle crosstalk and myogenic differentiation. Recent Pat Biotechnol. 2012;6:223–9.
Colaianni G, Sanesi L, Storlino G, Brunetti G, Colucci S, Grano M. Irisin and bone: from preclinical studies to the evaluation of its circulating levels in different populations of human subjects. Cells. 2019;8:5–451.
•• Kitase Y, Vallejo JA, Gutheil W, Vemula H, Jähn K, Yi J, et al. β-aminoisobutyric Acid, l-BAIBA, Is a Muscle-Derived Osteocyte Survival Factor. Cell Rep. 2018;22:1531–44 This work identifies a new muscle-derived molecule with anabolic effect on bone mass.
•• Mera P, Laue K, Ferron M, Confavreux C, Wei J, Galán-Díez M, et al. Osteocalcin Signaling in myofibers is necessary and sufficient for optimum adaptation to exercise. Cell Metab. 2016;23(6):1078–92 This is the first study showing that the bone-derived hormone osteocalcin is involved in muscle metabolism and improves muscle functions during exercise.
•• Bonnet N, Bourgoin L, Biver E, Douni E, Ferrari S. RANKL Inhibition Improves Muscle Strength and Insulin Sensitivity and Restores Bone Mass. J Clin Invest. 2019;129(8):3214–23 This work provides evidence that RANKL is a negative factor affecting skeletal muscle integrity and function.
Boström P, Wu J, Jedrychowski MP, Korde A, Ye L, Lo JC, et al. A PGC1-α-dependent myokine that drives brown-fat-like development of white fat and thermogenesis. Nature. 2012;481:463–8.
Colaianni G, Cuscito C, Mongelli T, Pignataro P, Buccoliero C, Liu P, et al. The myokine irisin increases cortical bone mass. Proc Natl Acad Sci U S A. 2015;112:12157–62.
Allen MR, Bloomfield SA. Hindlimb unloading has a greater effect on cortical compared with cancellous bone in mature female rats. J Appl Physiol. 2003;94:642–50.
Swift JM, Nilsson MI, Hogan HA, Sumner LR, Bloomfield SA. Simulated resistance training during hindlimb unloading abolishes disuse bone loss and maintains muscle strength. J Bone Miner Res. 2010;25:564–74.
•• Colaianni G, Mongelli T, Cuscito C, Pignataro P, Lippo L, Spiro G, et al. Irisin prevents and restores bone loss and muscle atrophy in hind-limb suspended mice. Sci Rep. 2017;7:2811 This study demonstrated that intermittent treatment with low dose of recombinant Irisin can prevent the unload-induced onset of osteoporosis and muscular atrophy in mice.
Storlino G, Colaianni G, Sanesi L, Lippo L, Brunetti G, Errede M, et al. Irisin prevents disuse-induced osteocyte apoptosis. J Bone Miner Res. 2019;35:766–75. https://doi.org/10.1002/jbmr.3944.
Vaughan RA, Gannon NP, Mermier CM, Conn CA. Irisin, a unique non-inflammatory myokine in stimulating skeletal muscle metabolism. J Physiol Biochem. 2015 Dec;71(4):679–89.
Huh JY, Dincer F, Mesfum E, Mantzoros CS. Irisin stimulates muscle growth-related genes and regulates adipocyte differentiation and metabolism in humans. Int J Obes. 2014 Dec;38(12):1538–44.
Singhal V, Lawson EA, Ackerman KE, Fazeli PK, Clarke H, Lee H, et al. Irisin levels are lower in young amenorrheic athletes compared with eumenorrheic athletes and non-athletes and are associated with bone density and strength estimates. PLoS One. 2014;9:e100218.
Colaianni G, Notarnicola A, Sanesi L, Brunetti G, Lippo L, Celi M, et al. Irisin levels correlate with bone mineral density in soccer players. J Biol Regul Homeost Agents. 2017;31:21–8.
Faienza MF, Brunetti G, Sanesi L, Colaianni G, Celi M, Piacente L, et al. High irisin levels are associated with better glycemic control and bone health in children with type 1 diabetes. Diabetes Res Clin Pract. 2018;141:10–7.
Soininen S, Sidoroff V, Lindi V, Mahonen A, Kröger L, Kröger H, et al. Body fat mass, lean body mass and associated biomarkers as determinants of bone mineral density in children 6–8 years of age—the physical activity and nutrition in children (PANIC) study. Bone. 2018;108:106–14.
Faienza MF, Ventura A, Delvecchio M, Fusillo A, Piacente L, Aceto G, et al. High Sclerostin and Dickkopf-1 (DKK-1) serum levels in children and adolescents with type 1 diabetes mellitus. J Clin Endocrinol Metab. 2017;102:1174–81.
Colaianni G, Faienza MF, Sanesi L, Brunetti G, Pignataro P, Lippo L, et al. Irisin serum levels positively correlate with bone mineral status in a population of healthy children. Pediatr Res. 2019;85:484–8.
Klangjareonchai T, Nimitphong H, Saetung S, Bhirommuang N, Samittarucksa R, Chanprasertyothin S, et al. Circulating sclerostin and Irisin are related and interact with gender to influence adiposity in adults with prediabetes. Int J Endocrinol. 2014;2014:261545.
Palermo A, Strollo R, Maddaloni E, Tuccinardi D, D’Onofrio L, Briganti SI, et al. Irisin is associated with osteoporotic fractures independently of bone mineral density, body composition or daily physical activity. Clin Endocrinol. 2015;82:615–9.
Anastasilakis AD, Polyzos SA, Makras P, Gkiomisi A, Bisbinas I, Katsarou A, et al. Circulating Irisin is associated with osteoporotic fractures in postmenopausal women with low bone mass but is not affected by either teriparatide or denosumab treatment for 3 months. Osteoporos Int. 2014;25:1633–42.
Park HS, Kim HC, Zhang D, Yeom H, Lim SK. The novel myokine irisin: clinical implications and potential role as a biomarker for sarcopenia in postmenopausal women. Endocrine. 2019;64:341–8.
•• Palermo A, Sanesi L, Colaianni G, Tabacco G, Naciu AM, Cesareo R, et al. A novel interplay between irisin and PTH: From basic studies to clinical evidence in hyperparathyroidism. J Clin Endocrinol Metab. 2019;104(8):3088–96 This work identifies the existence of a negative interplay between PTH and Irisin biology.
Roberts LD, Boström P, O’Sullivan JF, Schinzel RT, Lewis GD, Dejam A, et al. β-Aminoisobutyric acid induces browning of white fat and hepatic β-oxidation and is inversely correlated with cardiometabolic risk factors. Cell Metab. 2014;19:96–108.
Begriche K, Massart J, Abbey-Toby A, Igoudjil A, Lettéron P, Fromenty B. Beta-aminoisobutyric acid prevents diet-induced obesity in mice with partial leptin deficiency. Obesity. 2008;16:2053–67.
Shi CX, Zhao MX, Shu XD, Xiong XQ, Wang JJ, Gao XY, et al. β-aminoisobutyric acid attenuates hepatic endoplasmic reticulum stress and glucose/lipid metabolic disturbance in mice with type 2 diabetes. Sci Rep. 2016;6:21924.
Jung TW, Hwang HJ, Hong HC, Yoo HJ, Baik SH, Choi KM. BAIBA attenuates insulin resistance and inflammation induced by palmitate or a high fat diet via an AMPK-PPARδ-dependent pathway in mice. Diabetologia. 2015;58:2096–105.
Aguirre JI, Plotkin LI, Stewart SA, Weinstein RS, Parfitt AM, Manolagas SC, et al. Osteocyte apoptosis is induced by weightlessness in mice and precedes osteoclast recruitment and bone loss. J Bone Miner Res. 2009;21:605–15. https://doi.org/10.1359/jbmr.060107.
Uno M, Nishimura S, Fukuchi K, Kaneta Y, Oda Y, Komori H, et al. Identification of physiologically active substances as novel ligands for MRGPRD. J Biomed Biotechnol. 2012;2012:816159.
Wallace JM, Rajachar RM, Allen MR, Bloomfield SA, Robey PG, Young MF, et al. Exercise-induced changes in the cortical bone of growing mice are bone- and gender-specific. Bone. 2007;40(4):1120–7.
Eghbali-Fatourechi G, Khosla S, Sanyal A, Boyle WJ, Lacey DL, Riggs BL. Role of RANK ligand in mediating increased bone resorption in early postmenopausal women. J Clin Invest. 2003;111(8):1221–30.
Lacey DL, Boyle WJ, Simonet WS, Kostenuik PJ, Dougall WC, Sullivan JK, et al. Bench to bedside: elucidation of the OPG–RANK–RANKL pathway and the development of denosumab. Nat Rev Drug Discov. 2012;11(5):401–19.
McCloskey EV, Johansson H, Oden A, Austin M, Siris E, Wang A, et al. Denosumab reduces the risk of osteoporotic fractures in postmenopausal women, particularly in those with moderate to high fracture risk as assessed with FRAX. J Bone Miner Res. 2012;27(7):1480–6.
Huang J, Hsu YH, Mo C, Abreu E, Kiel DP, Bonewald LF, et al. METTL21C is a potential pleiotropic gene for osteoporosis and sarcopenia acting through the modulation of the NF-κB signaling pathway. J Bone Miner Res. 2014;29(7):1531–40.
Lewiecki EM. Safety and tolerability of denosumab for the treatment of postmenopausal osteoporosis. Drug Healthc Patient Saf. 2011;3:79–91.
Langen RC, Schols AM, Kelders MC, Wouters EF, Janssen-Heininger YM. Inflammatory cytokines inhibit myogenic differentiation through activation of nuclear factor-kappaB. FASEB J. 2001;15(7):1169–80.
Lee D, Goldberg AL. Muscle wasting in fasting requires activation of NF-κB and inhibition of AKT/mechanistic target of rapamycin (mTOR) by the protein acetylase, GCN5. J Biol Chem. 2015;290(51):30269–79.
Dufresne SS, Dumont NA, Bouchard P, Lavergne É, Penninger JM, Frenette J. Osteoprotegerin protects against muscular dystrophy. Am J Pathol. 2015;185(4):920–6.
Dufresne SS, Dumont NA, Boulanger-Piette A, Fajardo VA, Gamu D, Keke-Guena SA, et al. Muscle RANK is a key regulator of Ca2+ storage, SERCA activity, and function of fast-twitch skeletal muscles. Am J Physiol Cell Physiol. 2016;310(8):C663–72.
Dufresne SS, Boulanger-Piette A, Bossé S, Frenette J. Physiological role of receptor activator nuclear factor-kB (RANK) in denervation-induced muscle atrophy and dysfunction. Receptors Clin Investig. 2016;3(2):e13231–6.
Hauschka PV, Lian JB, Cole DE, Gundberg CM. Osteocalcin and matrix Gla protein: vitamin K-dependent proteins in bone. Physiol Rev. 1989;69:990–1047.
Delmas PD, Eastell R, Garnero P, Seibel MJ, Stepan J, Committee of Scientific Advisors of the International Osteoporosis Foundation. The use of biochemical markers of bone turnover in osteoporosis. Committee of Scientific Advisors of the international Osteoporosis Foundation. Osteoporos Int. 2000;11(Suppl. 6):S2–17.
Ducy P, Desbois C, Boyce B, Pinero G, Story B, Dunstan C, et al. Increased bone formation in osteocalcin-deficient mice. Nature. 1996;382:448–52.
Lee NK, Sowa H, Hinoi E, Ferron M, Ahn JD, Confavreux C, et al. Endocrine regulation of energy metabolism by the skeleton. Cell. 2007;130:456–69.
Oury F, Sumara G, Sumara O, Ferron M, Chang H, Smith CE, et al. Endocrine regulation of male fertility by the skeleton. Cell. 2011;144:796–809.
Wei J, Hanna T, Suda N, Karsenty G, Ducy P. Osteocalcin promotes β-cell proliferation during development and adulthood through Gprc6a. Diabetes. 2014;63:1021–31.
Fulzele K, Riddle RC, DiGirolamo DJ, Cao X, Wan C, Chen D, et al. Insulin receptor signaling in osteoblasts regulates postnatal bone acquisition and body composition. Cell. 2010;142:309–19.
Ferron M, McKee MD, Levine RL, Ducy P, Karsenty G. Intermittent injections of osteocalcin improve glucose metabolism and prevent type 2 diabetes in mice. Bone. 2012;50:568–75.
Huang L, Yang L, Luo L, Wu P, Yan S. Osteocalcin improves metabolic profiles, body composition and arterial stiffening in an induced diabetic rat model. Exp Clin Endocrinol Diabetes. 2017;125:234–40.
Mera P, Laue K, Wei J, Berger JM, Karsenty G. Osteocalcin is necessary and sufficient to maintain muscle mass in older mice. Mol Metab. 2016;5:1042–7.
Oury F, Khrimian L, Denny CA, Gardin A, Chamouni A, Goeden N, et al. Maternal and offspring pools of osteocalcin influence brain development and functions. Cell. 2013;155:228–41.
Khrimian L, Obri A, Ramos-Brossier M, Rousseaud A, Moriceau S, Nicot A, et al. Gpr158 mediates osteocalcin's regulation of cognition. J Exp Med. 2017;214(10):2859–73.
Berger JM, Singh P, Khrimian L, Morgan DA, Chowdhury S, Arteaga-Solis E, et al. Mediation of the Acute Stress Response by the Skeleton. Cell Metab. 2019;30(5):890–902.e8.
Funding
This work was supported by Ministero dell’Istruzione, dell’Università e della Ricerca, PRIN 2015JSWLTN_003 (Progetto di Ricerca d’Interesse Nazionale, Grant 2015), by ERISTO (ESA) and TecnoMed Puglia grants to MG.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that there is no conflict of interest regarding the publication of this paper. All authors approved the final version of the submitted manuscript and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Graziana Colaianni, Silvia Colucci, and Maria Grano report a patent irisin for care and prevention of osteoporosis issued. Giuseppina Storlino and Lorenzo Sanesi declare no conflict of interest.
Human and Animal Rights and Informed Consent
All studies by the authors involving animal and/or human subjects were performed after approval by the appropriate institutional review boards. When required, written informed consent was obtained from all participants.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Muscle and Bone
Rights and permissions
About this article
Cite this article
Colaianni, G., Storlino, G., Sanesi, L. et al. Myokines and Osteokines in the Pathogenesis of Muscle and Bone Diseases. Curr Osteoporos Rep 18, 401–407 (2020). https://doi.org/10.1007/s11914-020-00600-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11914-020-00600-8