Abstract
Purpose of Review
The aim of this review is to focus on the recent advances in the molecular knowledge of small cell lung cancer (SCLC) and potential promising new treatment strategies, like targeting the DNA damage pathway, epigenetics, angiogenesis, and oncogenic drivers.
Recent Findings
In the last few years, the addition of immunotherapy to chemotherapy has led to significant improvements in clinical outcomes in this complex neoplasia. Nevertheless, the prognosis remains dismal. Recently, numerous genomic alterations have been identified, and they may be useful to classify SCLC into different molecular subtypes (SCLC-A, SCLC-I, SCLC-Y, SCLC-P).
Summary
SCLC accounts for 10-20% of all lung cancers, most patients have an extensive disease at the diagnosis, and it is characterized by poor prognosis. Despite the progresses in the knowledge of the disease, efficacious targeted treatments are still lacking. In the near future, the molecular characterisation of SCLC will be fundamental to find more effective treatment strategies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Small cell lung cancer (SCLC) accounts for about 10–20% of all lung cancers and it is characterized by a high rate of proliferation, early metastases, and poor prognosis [1, 2]. As many as 98% of patients are smokers providing direct evidence that tobacco carcinogens are responsible for the initiation of SCLC [2, 3]. About 70% of patients have extensive disease (ED-) at diagnosis with a 2-year survival rate of approximately 2% [4]. For more than 30 years, chemotherapy (CT) with platinum and etoposide (PE) has been the standard front-line therapy [5, 6]. The introduction of immune checkpoint inhibitors (ICIs) set a new step in clinical practice for patients with ED-SCLC [7••].
As demonstrated by IMpower133 and CASPIAN, beyond others first-line trials conducted in asiatic population such as CAPSTONE-1 and ASTRUM-005, the addition of ICI to first-line platinum-based chemotherapy significantly improved overall survival (OS) with a reduction of 25 to 30% of the relative risk of death [7••, 8, 9••, 10]. Nevertheless, more research is needed to further improve this dismal benefit. Although SCLC is characterized by numerous genomic alterations, effective targeted therapies are still lacking. Indeed, over the past decade, the complete genomic profile of SCLC has highlighted a broad and complex genetic landscape of this tumour, including somatic mutations on transcription factors, receptor tyrosine kinases genes, and epigenetic changes in chromatin modifiers enzymes [2, 11,12,13,14]. However, all the trials to date have failed to demonstrate a survival benefit from using targeted agents. A deeper study of molecular aberrations could lead to the identification of new therapeutic targets [9••, 15].
Toward a New Molecular Classification
Despite almost all cases of SCLC have total genomic loss of function of both TP53 and RB1, and are treated as a single disease, they are characterized by high heterogeneity [2]. Several authors have tried to classify SCLC into different subgroups based on molecular features.
Towards this end, Carney and colleagues identified in vitro two subtypes of SCLC, a classic (70%) and a variant subtype (30%). The classic phenotype cell lines express L-dopa decarboxylase, bombesin-like immunoreactivity, neuron-specific enolase, and the brain isozyme of creatine kinase. The variant subtype lack the expression of either L-dopa decarboxylase or bombesin-like immunoreactivity. The variant subtype is the most aggressive and is characterized by shorter doubling time and higher resistance to chemotherapy [16]. Furthermore, Poirier and colleagues identified two genes, achaete-scute homologue 1 (ASCL1) and neurogenic differentiation factor 1 (NEUROD1), both involved in neuroendocrine cells differentiation, but differentially expressed in SCLC. These genes are mutually exclusive in SCLC cell lines. ASCL1 has lower expression in variant SCLC compared to classic SCLC, and the authors proposed ASCL1 as the best candidate for differentiating SCLC subtypes [17]. ASCL1 and NEUROD1 regulate different genes, but ASCL1 is the leading cause of tumour formation. ASCL1 targets oncogenes such as MYCL1, SOX2, RET, and BCL2, suggesting it is required for SCLC survival. In addition, ASCL1 regulates NOTCH pathway genes, such as Delta-like ligand 3 protein (DLL3), which may represent a possible therapeutic target in SCLC. NEUROD1 targets the oncogene MYC, and cMYC protein expression is a predictive biomarker for multiple Aurora kinase inhibitors (AURKi). Therefore, ASCL1-positive and NEUROD1-positive SCLC cells have different origins and create two distinct subgroups of SCLC [18•].
Another gene identified in SCLC cells and potentially helpful to stratify SCLC into subgroups is the yes-associated protein 1 (YAP1). YAP1 is a key step in the tumour-suppressive Hippo pathway, which controls cell proliferation, apoptosis, and organ size. When Hippo signalling is active, YAP1 is inactivated and isolated in the cytoplasm for degradation. When Hippo signalling is inactive, YAP1 promotes pro-survival gene expression, proliferation, and tissue growth. RB1 is co-expressed with YAP1 in SCLC cell lines, thus it can be considered a surrogate of YAP1. Wild-type RB1 is present in about 25% of SCLC patients and is associated with decreased survival compared to patients with mutant RB1 [19].
POU class 2 homeobox 3 (POU2F3), also known as SKN-1a/OCT-11, is a transcription factor required for the generation of a chemosensory cell type of the gastrointestinal and respiratory tracts. POU2F3 is expressed in about 18% of SCLC and is mutually exclusive with ASCL1 and NEUROD1. Thus, POU2F3 identifies a unique subgroup of SCLC characterized by low expression of neuroendocrine markers. Interestingly, POU2F3-expressing cells are dependent on the tyrosine kinase receptor insulin-like growth factor 1 (IGF-1R) and therefore potentially sensitive to inhibitors of IGF-1R.
Based on these findings, Rudin et al. proposed a molecular classification of SCLC tumours. They identified 4 molecular subtypes called SCLC-A, SCLC-N, SCLC-Y and SCLC-P, according to the expression of transcription factors required for neuroendocrine (ASCL1 or NEUROD1) or non-neuroendocrine (YAP1 or POU2F3) differentiation. The last letters (in SCLC-?) indicate the transcription factor strongly associated with each subtype [20•].
Subsequent in vivo studies analysed the expression of ASCL1, NEUROD1, POU2F3 and YAP1 by immunohistochemistry (IHC). Baine’s research group observed that 69% of cases are ASCL1 dominant and 17% are NEUROD1 dominant. They confirmed that POU2F3 expression (7% of SCLC) is mutually exclusive of ASCL1 and NEUROD1. In addition, ASCL1/NEUROD1 double-negative tumours (14% SCLC) are a distinct subtype of SCLC characterized by low expression of neuroendocrine markers. Conversely, 7% of ASCL1/NEUROD1 double-negative cases have no identified transcription factors. Notably, YAP1 is absent or expressed at low levels in SCLC cells, compared with non-small cell lung cancer (NSCLC) cells where it is strongly expressed [21, 22••]. YAP1 protein expression seems also to correlate with the stage of disease, with highest expression in limited stage SCLC, an inflamed tumour microenvironment, and a better prognosis 23•]. Thus, it is not clear if YAP1-positive SCLC tumours represent a distinct subtype.
A similar study validated the molecular classification by IHC and showed intratumoral heterogeneity in SCLC tumours, which can contribute to chemo-resistance. In fact, Qu and colleagues observed that SCLC tumours can be positive for two (17.6%) or three (2.8%) subtype markers. Tumours that are predominantly positive for MYC are in either POU2F3 or YAP1 subgroups, while MYC negative tumours are mostly in ASCL1 and NEUROD1 subtypes. Moreover, tumour-associated CD8 + T cells are interconnected with molecular subtype; the non-ASCL1/NEUROD1 subtypes have significantly more CD8 + T cells than those of the ASCL1/NEUROD1 subtypes, thus they can benefit more from immunotherapy. Finally, Qu and colleagues also showed that a small percentage (6.3%) of SCLC tumours are negative for all four subtype markers [20•].
More recently, Gay and colleagues identified a SCLC subtype negative for all transcription factors, named SCLC-inflamed (SCLC-I). SCLC-I exhibits an inflamed phenotype that may make it more sensitive to immunotherapy. Indeed, it is characterized by high levels of immune checkpoints, MHC genes, and interferon-γ signalling pathway components and high levels of immune cell populations, such as T cells, natural killer cells, and macrophages [24].
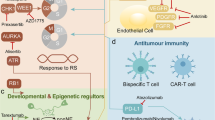
Therefore, a molecular classification of SCLC represents the starting point to improve the current standard of care, overcome resistance to chemo-immunotherapy and to develop biomarker-guided therapies. The main characteristics of the molecular subtypes are reported in Fig. 1 and Table 1.
Targeting the DNA Damage Repair Pathway
Interest in DNA damage and repair (DDR) has rapidly increased since it emerged that aggressive tumours, including SCLC, have a DDR pathway alteration.
Impairment in DDR pathway is a well-known predictive biomarker of platinum sensitivity. Furthermore, it is a predictive biomarker of ICIs benefit due to its positive correlation with tumour mutation burden (TMB).
There are five major DNA damage repair pathways. The base excision repair (BER) mechanism repairs single strand breaks. The homologous recombination repair (HRR) and the non-homologous end joining (NHEJ) fix double-strand breaks. The mismatch repair (MMR) mechanism repairs replication errors. Finally, the nucleotide excision repair (NER) repairs platinum and UV radiation damage [25, 26].
PARP Inhibitors
Poly (ADP-ribose) polymerase (PARP) inhibitors target a family of proteins called PARP involved in several processes including DNA repair (using BER, HRR and NHEJ) and apoptosis. PARP1 and PARP2 are key proteins that are activated when DNA damage occurs, which they detect and then send signals to other proteins to repair it.
There are tumours in which one of the DNA repair pathways, HRR, is no longer functional due to mutations in one of its main components, such as BRCA1 or BRCA2 proteins. Therefore, the DNA damage, originating from the genetic instability of the tumour, can neither be repaired either by PARP- 1 nor inhibited by the drug, or by the alternative pathway of homologous recombination, whose functionality is intrinsically compromised in these types of tumours. The simultaneous mutation (or inhibition) of a pair of genes or biochemical pathway, unlike the mutation (or inhibition) of only one of these, causes cell death and is called synthetic lethality.
SCLC is sensitive to DNA damage, and PARP1 is highly expressed in SCLC. Combination strategies with PARP inhibitors (PARPis) have been studied previously in second line, and then in first line, with only modest results.
Following an initial phase I/II study that established the recommended phase II study dose of olaparib in combination with temozolomide (TMZ), a phase II study was conducted to evaluate clinical activity in relapsed SCLC. The combination demonstrated a 41.7% overall response rate (ORR), median progression-free survival (PFS) of 4.2 months and median overall survival (OS) of 8.5 months [27].
Likewise, a phase II study evaluated the combination of veliparib and TMZ versus placebo and TMZ (1:1) in recurrent SCLC. It showed an increased ORR (39% v 14%; p = 0.016), but neither an increase in PFS (p = 0.19) nor in median OS (8.2 months versus 7.0 months; p = 0.50). Moreover, grade 3/4 thrombocytopenia and neutropenia were more common in the combination arm. Notably, a statistically and clinically significant improvement of PFS (5.7 v 3.6 months; P = 0.009) and OS (12.2 v 7.5 months; P = 0.014) was observed in patients with SLFN11-positive tumours treated with veliparib plus TMZ [28].
Based on these results, PARPis were tested in earlier lines.
Olaparib was evaluated as a maintenance monotherapy in patients with SCLC in partial or complete response following first-line treatment or chemo-radiation therapy. Patients were randomized 2:2:1: olaparib 300 mg twice a day (BD), olaparib 200 mg three times a day (TDS), placebo BD, or placebo TDS. There was no significant difference in either PFS or OS between olaparib and placebo [29].
First-line veliparib was tested in combination with cisplatin plus etoposide chemotherapy in patients with ED-SCLC. Patients were randomized 1:1: chemotherapy plus veliparib versus chemotherapy plus placebo. The experimental combination showed a significant benefit in PFS (median PFS 6.1 vs 5.5 months, observed stratified PFS HR 0.63, one-sided P = 0.01), but neither in OS nor ORR. No predictive biomarkers were identified [30].
Conversely, in another study SLFN11 was identified as a potential predictive biomarker of benefit from PARPis in patients with untreated ED-SCLC. In this phase 2 study, randomization was 1:1:1: veliparib plus chemotherapy with carboplatin plus etoposide (CE) followed by veliparib maintenance, veliparib plus CE followed by placebo or placebo plus CE followed by placebo (control arm). The first arm combination improved PFS compared with the control arm (HR 0.67; 80% confidence interval (CI). 0.50–0.88; p = 0.059), although the difference was not clinically significant (median PFS 5.8 versus 5.6 months), with a trend in SLFN11 positive patients (HR, 0.6; 80% CI: 0.36–0.97). Moreover, there was no significant benefit in OS [31].
Another PARPi, niraparib, was tested as a maintenance strategy in Asian patients with ED-SCLC with partial or complete response to first line chemotherapy in a randomized, double-blind, phase 3 study. Likewise, niraparib showed a statistically significant improvement in PFS (HR 0.66; 95% CI: 0.46–0.95; p = 0.0242), even though clinically modest (median PFS 1.54 versus 1.36 months) [32].
Therefore, despite the fact that the DDR pathway represents a potential therapeutic target, DDR alterations seem not to correlate with platinum chemotherapy outcomes in SCLC, in contrast to ovarian or breast cancers.
Another area of research is the use of PARPi to overcome ICI resistance. Preclinical studies demonstrated that inhibition of DDR proteins such PARP enables the anti-tumour immune response of PD-L1 inhibition through T cell-mediated effects. These pre-clinical studies provided a rationale for combining PARP inhibitors with immunotherapies in SCLC [33, 34]. A phase 2 study of durvalumab plus olaparib in patients with relapsed SCLC did not meet its primary endpoint of PFS, but it did demonstrate the importance of appropriate patient selection on the basis of tumours and tumor microenviroment (TME) biological characteristics [35]. Several trials studying combination of PARP inhibitor plus ICIs are ongoing, for both pre-treated ED-SCLC (phase 2 NCT04701307, phase 1/2 NCT04728230), and for the maintenance/consolidation therapy following first-line treatment (phase 2 NCT04782089, phase 2 NCT04334941, phase 1b/2 NCT03830918).
Lurbinectedin
Lurbinectedin is a drug that prevents oncogenic transcription activity in cancer cells. It inhibits RNA formation by preventing the binding of transcription factors to their promoters, thus promoting DNA double-strand breaks and inducing cell death [36, 37]. Moreover, lurbinectedin reduces inflammation of the tumour microenvironment and decreases transcription within tumour-associated macrophages. This induces tumour cell death, reduces angiogenesis, and enhances anti-tumour immunity [38]. Lurbinectedin showed increased beneficial activity in tumours with defects in DNA mismatch repair, including SCLC.
Initially, a phase II single-arm basket trial evaluated patients with ED SCLC pre-treated with only one line of treatment. Patients were treated with lurbinectedin 3.2 mg/m2 every 3 weeks until disease progression or unacceptable toxicity. An ORR of 35.2%, median PFS of 3.5 months and OS of 9.3 months were reported [39]. Based on these results, lurbinectedin has been granted by FDA as orphan drug status for the treatment of patients that progress after first-line platinum-based chemotherapy.
Moreover, combination strategies were tested to improve efficacy. A phase I study investigated the association of lurbinectedin 2.0 mg/m2 and doxorubicin 40 mg/m2. The combination achieved an ORR of 91.7% in second-line patients with sensitive disease (platinum-free interval ≥ 90 days) and an ORR of 33.3% with resistant disease (platinum-free interval < 90 days). In third-line setting patients achieved an ORR of 20% in all subgroups [40].
Following these encouraging results, the Phase III study ATLANTIS tested the same combination compared to topotecan or CAV (cyclophosphamide, doxorubicin, and vincristine) in over 600 patients progressing to first-line platinum-containing chemotherapy. The combination did not improve OS compared to the control arm. Furthermore, an OS difference was not observed in either patients with or without central nervous system involvement. Despite this, the combination showed a better toxicity profile compared to the control arm [41].
Targeting Angiogenesis
The angiogenesis process is crucial for tumorigenesis and remains active during cancer growth. The vascular endothelial growth factor (VEGF) pathway is over-active in a lot of tumours, including SCLC [42]. It has been demonstrated that the hypoxia-inducible factor 1a (HIF-1a) can promote SCLC growth and angiogenesis and, therefore, be another potential therapeutic target [43].
Bevacizumab is a humanised monoclonal antibody targeting VEGF, approved for several malignancies, such as colon-rectal, kidney, and ovarian cancer [44]. Bevacizumab is the most studied antiangiogenic therapy in SCLC, with inconsistent results.
In metastatic SCLC a number of studies have been conducted, mainly phase II studies.
Regarding relapsed pre-treated-patients, bevacizumab was evaluated in combination with chemotherapy (topotecan or paclitaxel) in two phase II studies. Results showed good tolerability, but few efficacy advantages compared to historical controls [45, 46].
Recently, a phase I study evaluating lurbinectedin plus paclitaxel with or without bevacizumab in advanced solid tumours, including SCLC, showed no major interactions between the drugs [47].
Most of the trials evaluated bevacizumab in combination with chemotherapy in a first line setting. Two phase II studies focused on the combination of a platinum agent plus irinotecan and bevacizumab, which showed similar results. In fact, median PFS was 7 months (95% CI, 6.4–8.4 months) with cisplatin, irinotecan and bevacizumab, and median time to progression was 9.13 months (95% CI, 7.36–9.46 months) with carboplatin, irinotecan and bevacizumab. Likewise, median OS was 11.6 months (95% CI, 10.5–15.1 months) and 12.1 months (95% CI, 9.6–13.5 months), respectively [48, 49]. Bevacizumab was also evaluated in the first line maintenance setting in the phase II studies showing an improvement in PFS and OS compared to historical controls for those patients who received cisplatin and etoposide without bevacizumab maintenance [50]. The SALUTE trial was the first placebo-controlled, double-blind, randomized phase II trial that assessed bevacizumab in this therapy setting. It showed a small PFS improvement (5.5 months in the Bevacizumab arm versus 4.4 months in the placebo arm, HR 0.53) without an OS advantage [51].
More recently, two phase III trials have been conducted to evaluate the efficacy of adding bevacizumab to chemotherapy as first-line treatment. They confirmed a favourable toxicity profile of the combination, although without either a clinically significant PFS or an OS improvement [52, 53].
Regarding limited-disease SCLC, only one phase II study investigating the activity of bevacizumab as maintenance therapy after chemo-radiotherapy has been conducted, and reported a median OS of 15 months, ORR and 2-year PFS of 80% and 54% respectively [54]. However, safety represents a concern for the high incidence of tracheoesophageal fistulae.
Pazopanib is an orally administered tyrosine kinase inhibitor (TKI) targeting vascular endothelial growth factor receptor (VEGFR), platelet-derived growth factor receptors (PDGFR), and c-kit. It has been studied in SCLC as maintenance therapy following platinum and etoposide chemotherapy, in the study KCSG-LU12-07, which showed a statistically significant PFS improvement (3.7 months in pazopanib group versus 1.8 months in the placebo group, p < 0.0001). However, pazopanib did not show a good toxicity profile with 51.2% interruption rate due to adverse events [55]. Pazopanib has also been evaluated in second line of therapy by the Hellenic Oncology Research Group. In this phase II study, patients with platinum-sensitive and platinum-resistant SCLC were enrolled and received pazopanib. Albeit small, the study showed promising activity in patients with platinum-sensitive disease, with a DCR of 48.3% [56].
Sunitinib is an oral TKI that binds VEGFR, PDGFR, Flt-3, and Kit [44]. As with pazopanib, sunitinib has been studied as maintenance therapy following CT in SCLC in two trials. In a phase II, non-randomized trial, it was evaluated following CT with irinotecan and carboplatin, and showed a 1-year OS of 54% with rare adverse events [57]. In the CALGB 30504 a randomized, placebo-controlled, phase II trial, patients were randomized to receive sunitinib or a placebo after etoposide plus platinum-based chemotherapy. Sunitinib was found to be safe and effective. Indeed, median PFS was 2.1 months for placebo and 3.7 months for sunitinib arm (p = 0.02) [58].
Nintedanib is a potent TKI that links to VEGFR1-3, FGFR 1–3, and PDGFR α and β [59]. A phase 2 study was conducted to evaluate its efficacy and safety in relapsed/platinum-refractory SCLC. Nintedanib had a manageable toxicity profile but very poor efficacy in these patients’ setting. Indeed, ORR was 5%, with 1 month of PFS [59].
Anlotinib is an orally administered TKI directed to VEGFR, fibroblast growth factor receptor (FGFR), PDGFR, and c-kit [44]. Several studies have been developed in China, where anlotinib is approved for patients with pre-treated SCLC to evaluate the safety and the efficacy of this drug [44]. It has been studied alone as a third or fourth line of treatment [60], and in combination with standard CT as a first line of therapy [61, 62], showing good tolerability and promising clinical efficacy.
Another drug studied mainly in China is apatinib, a VEGF2 inhibitor. Likewise, some studies evaluating apatinib as maintenance or in second and further line therapies have been developed. As with anlotinib, apatinib showed a safe toxicity profile and interesting clinical activity [63,64,65,66].
Targeting Proteins and Oncogenic Drivers
Oncogenic drivers are genes with acquired mutations that are causally linked to cancer initiation and progression [67]. In NSCLC, a number of oncogenic driver mutations have been determined, enabling a molecular targeted treatment approach. Conversely, in SCLC the main driver mutations are loss of function of suppressor genes such as RB1 (60–90%) and TP53 (75–90%) [11, 68]. RB and TP53 mutations lose function and as a result are un-targetable. Nevertheless, a few activating mutations have been identified in SCLC, introducing the initial concept of “oncogene-addiction” in this aggressive tumour.
DLL-3
DLL-3 (delta-like protein 3) is a cell surface protein that inhibits the tumor suppressor gene NOTCH-1 and consequently upregulates the expression of Achaete-scute homolog 1 (ASH-1), a transcription factor driving SCLC oncogenesis [69]. DLL-3 is highly expressed in SCLC and other high-grade neuroendocrine tumours but not in normal lung tissue, suggesting that this protein may play an important role in neuroendocrine tumorigenesis and can be a vector for delivering cytotoxic agents to DLL3-positive cells [70,71,72]. Rovalpituzumab tesirine (Rova-T) is a first-in-class, antibody–drug conjugate (ADC) composed of an IgG1 monoclonal antibody that targets DLL3 linked to a toxic DNA agent, pyrrolobenzodiazepine (PBD), and a protease-cleavable linker [73]. Phase I and phase I–II studies have been conducted to assess safety and pharmacokinetics [74,75,76]. These seemingly promising results led to the development of phase II trials, like the Trinity Study, in which DLL-3 high SCLC demonstrated a 14.3% ORR with a median OS of 5.7 months [77]. These disappointing results have since been confirmed by a few Phase III trials. The THAOE study, a randomized trial, compared Rova-T with topotecan in second-line therapy in DLL3-high metastatic SCLC, but did not show any benefits in terms of OS [78]. Likewise, the MERU trial, a phase III study evaluating Rova-T in maintenance therapy after first-line platinum chemotherapy did not meet the primary endpoint. Thus, the lack of survival benefit led to an early discontinuation of the study. Moreover, Rova-T was associated with higher, unique, and unacceptably adverse events (AEs) like pleural and pericardial effusions, peripheral oedema and photosensitivity [79]. Therefore, the development of Rova-T has been discontinued. Nevertheless, new generation anti-DLL-3 drugs are currently under investigation. AMG 757 (tarlatamab) is a first-in-class bispecific T cell engager that binds DLL-3 and CD3 domains of the T cell receptor, leading to T cell-mediated tumour lysis. In vitro, an interesting killing activity has been shown, which led to the development of Phase I trials, currently ongoing [80,81,82]. Moreover, a phase II trial (NCT05060016) is evaluating safety and efficacy in the pre-treated SCLC population. Another drug, AMG 119, a chimeric antigen receptor T cell (CAR-T), showed promising results in vitro and in vivo, but the Phase I trial has been suspended for the time being [80].
CD56
CD56, also known as NCAM1, is an adhesion molecule involved in nervous system differentiation and immune surveillance. CD56 aberrant expression is evident in many solid tumours with neuroendocrine origin, like SCLC. For this reason, it could be an appropriate molecular target [83]. Lorvotuzumab mertansine (LM, IMGN901) is an ADC with an anti-CD56 antibody linked to a microtubule inhibitor, DM1 [69]. A phase I–II study evaluated LM in association with carboplatin/etoposide regimen in ED-SCLC, but this showed no evidence of efficacy improvement in the combination regimen as well as resulting in an increased number of AEs. In fact, 21 patients (63.6%) had a treatment-related adverse event (TRAEs) resulting in death [84]. Another Phase I study showed 96.9% of TRAEs, mostly grade 1 or 2 [83]. Therefore, further investigations are needed to better understand safety and efficacy of LM.
TROP 2
TROP2 (trophoblast cell surface antigen-2) is a transmembrane glycoprotein member of the EpCAM family, overexpressed in many tumours (e.g., breast cancer and NSCLC), acting either like an oncogene driver or an onco-suppressor. Non-tumour-tissues rarely express TROP2, so it can be a reliable therapeutic target [69]. Sacituzumab govitecan (SG) is a Trop2-directed antibody linked to SN-38, an active metabolite of irinotecan [85, 86]. The IMMU-132–01 phase I–II basket trial evaluated the efficacy and safety of SG in several tumours, including sixty-two SCLC patients. In this population, an ORR of 17.7% was shown, with a median OS of 7.1 months (95% CI, 5.6–8.1 months) and a median PFS of 5.5 months (95% CI, 3.6–7.6 months) [86]. A phase I–II clinical trial in previously treated ED-SCLC patients showed an ORR of 14%, a median OS of 7.5 months (95% CI, 6.2–8.8) and a median PFS of 3.7 months (95% CI, 2.1–4.3) [87]. These results are encouraging but need to be confirmed in larger prospective studies. Trials evaluating SG in combination with PARP inhibitors are currently ongoing [69].
SOX2
SOX2 is a pluripotency factor and a key regulator of neuroendocrine cells. A SOX2 amplification, regulated by the Hedgehog cascade, is associated with SCLC growth [11]. A phase I trial of sonidegib (Hedgehog inhibitor) in combination with cisplatin and etoposide for ED-SLCLC treatment showed an ORR of 79% (95% CI, 49–95%) and 6/15 TRAEs [88]. Another study evaluating vismodegib, another Hedgehog inhibitor, in combination with cisplatin and etoposide did not show any benefit in terms of either OS or PFS [89].
Bcl-2
Bcl-2 is an anti-apoptotic protein frequently overexpressed in SCLC [90]. A phase I and a phase II study with navitoclax, a selective inhibitor of Bcl-2 and Bcl-x, showed limited activity in advanced and recurrent SCLC with a high rate of serious thrombocytopenia [91, 92]. To mitigate this relevant side effect navitoclax was re-engineered as venetoclax. Venetoclax selectively binds Bcl-2 without causing thrombocytopenia, and showed promising preclinical results [90, 93]. A phase II study with another Bcl-1 inhibitor, obatoclax, in addition to carboplatin and etoposide demonstrated a good safety profile, but no clinical benefit in terms of either ORR, OS or PFS [94]. Similarly, AT-101 is an oral, pan-Bcl2 inhibitor, evaluated in a phase I-II trial in combination with topotecan. The combination is safe, but no clinical benefit was achieved in the early phases, so the enrolment closed [95].
AURK
AURKs (aurora kinases) are a family of kinases that play a key role in the cell cycle, in particular in the cell duplication. Aurora kinase A (AURKA) promotes mitosis through activation of checkpoint kinase 1 (CHK1), and is highly expressed in SCLC, thus representing a potential therapeutic target. An AURK hyperexpression is a pro-tumorigenic pathway in many cancer types, including SCLC. For this reason, a number of AURK inhibitors (AURK-is) have been developed in the last few years [96]. AURK-is action mechanism has been tested in mice [97], and this led to Phase I and Phase II clinical trials in pre-treated patients. Alisertib is an oral AURKA inhibitor that, as a single agent, showed beneficial activity in a phase II trial in terms of ORR (21%, 10/48 pts) in relapsed or refractory SCLC [98]. Following these encouraging findings, alisertib was studied in combination with weekly paclitaxel compared to placebo plus weekly paclitaxel in relapsed or refractory SCLC. The combination proved a PFS advantage (HR 0.77, 95% CI: 0.557–1.067, p 0.113), while no benefit in OS was observed. Interestingly, as previously reported in another study [99], Myc was revealed to be a potential predictive biomarker. In fact, although the number of patients is small (33), a greater advantage for PFS was observed in patients with c-Myc expression (4.64 vs 2.27 months, HR 0.29, 95% CI: 0.12–0.72) compared to c-Myc-negative patients [100].
Danusertib, a multi AURK-i, was tested in 24-h infusion in solid tumours in a Phase I study. It included 2 SCLC patients of which one had an ORR of 23 weeks [101]. A Phase II trial with danusertib showed a median PFS of 8.1 weeks (95% CI, 7.1–8.9) and a median OS of 11.4 months (95% CI, 4.5–n.r.) in the SCLC cohort [102].
Finally, prexasertinib, a selective CHK1 inhibitor, was investigated as a single-agent in a phase II trial in patients with pre-treated ED-SCLC, but it failed to demonstrate beneficial activity [103].
MYC
MYC gene amplifications have been identified in 6–25% of SCLC, especially in the SCLC-N subtype, and are associated with poorer outcomes and treatment resistance [97]. Indeed, data suggests that MYC promotes a subset of more aggressive SCLC subtype with lower expression of neuroendocrine markers [96]. A preclinical trial showed that arginine depletion with pegylated arginine deiminase (ADI-PEG 20) suppresses tumour growth in mice that had MYC-driven tumours [99]. A Clinical trial to evaluate ADI-Peg 20 is ongoing (NCT03449901). A few cases have been described in which MYC amplification can make cells sensitive to AURK-is [104], and this is supported by preclinical data [97, 105].
Targeting Epigenetics
Epigenetic refers to modulation of gene expression profiling without alteration of the DNA sequence [106]. Several studies demonstrated a critical role of epigenetic alterations during SCLC development and progression [107,108,109,110,111].
DNA methylation is one of the epigenetic processes, and it is involved in key SCLC genes’ regulation [112,113,114].
DNA methyltransferases (DNMTs) are the effectors of this process, and DNMTs inhibitors are small molecules that when used in low doses, can induce the expression of silenced genes, while when used in high doses can directly kill cancer cells [115, 116].
Another important epigenetic mechanism consists of histone modifications, in particular methylation and acetylation. Histone methyltransferases (HTMs) and histone demethylases (HDMs) are the effectors of histone methylation, while acetylation is regulated by histone acetyltransferases (HATs) and histone deacetylases (HDACs) [117]. A few studies showed that these mechanisms are a cause of SCLC pathogenesis and development and of chemotherapy sensitivity/resistance [118,119,120,121].
Several inhibitors of histone modifications have been tested for SCLC treatment. When considering HDAC inhibitors, preclinical studies demonstrated that different molecules suppress SCLC cell proliferation and promote chemotherapy anti-tumour effects in SCLC cell lines [122,123,124,125,126]. Many phase I/II trials are currently ongoing. Furthermore, little, and inconsistent data is available. One single-centre phase I trial showed that the combination of the HDAC inhibitor belinostat with cisplatin plus etoposide is safe and active in SCLC and other neuroendocrine cancers [127]. Nevertheless, a phase II trial evaluating the HDAC inhibitor panobinostat and a phase II trial investigating the HDAC inhibitor romidepsin showed a lack of activity of these two compounds [128, 129].
Also, HMTs and HDMs inhibitors showed activity against SCLC lines in preclinical studies. For example, the HMT enhancer of zeste homolog 2 (EZH2) is highly expressed in SCLC, and preclinical evidence showed that its inhibition can overcome SCLC chemoresistance [130]. At present no data derived from clinical trials is available for EZH2 inhibitors.
Lysine specific demethylase-1 (LSD1) is a highly expressed HDM in various haematological and solid tumours, including SCLC [131]. Its inhibitors, ORY-1001 (Iadademstat) and GSK2879552, repressed SCLC tumorigenesis and growth in preclinical studies [132, 133]. At present the findings from clinical studies are discouraging and inconsistent with pre-clinical analysis. A phase I multicentre, open-label study that investigated the safety, pharmacokinetics, pharmacodynamics, and clinical activity of the LSD1 inhibitor GSK2879552 in patients with relapsed or refractory SCLC was terminated due to a high incidence of adverse events and poor disease control [134]. Nevertheless, there is a lot of interest in epigenetic drugs, in particular LSD-1 inhibitors, and many phase I or II clinical trials are ongoing (e.g., NCT05420636, NCT03850067, NCT05268666, NCT03460977).
Finally, multitarget epigenetic molecules are also currently under investigation. Very recently, JBI-802 has been identified as a dual LSD1/HDAC6/8 inhibitor, which displayed huge antiproliferative effects in SCLC and others haematological and solid tumours [135]. Moreover, combinations with DNMT and HDAC inhibitors are being researched only in preclinical settings to date [136].
Therefore, all this data underlines the crucial role of epigenetics in carcinogenesis and proliferation of tumor cells.
Furthermore, recent studies have demonstrated the crucial role of epigenetic machinery in modulating immune cell functions and antitumor immune response, modifying tumour immunogenicity as well as affecting immune cells [137].
Epigenetic modifications regulate the antigen processing and presentation, the maturation and differentiation of dendritic cells, the activation, trafficking and infiltration of T cells, and the development of Treg [137, 138]. Moreover, it is demonstrated that the expression of PD-1 and PD-L1 in SCLC is partly related to an upregulation of DNA methyltransferase 1 (DNMT1) [139].
Considering in particular the antigen presentation phase, preclinical data showed that SCLC has a low intrinsic expression of MHC class I and II molecules [140, 141], and one of the underlying mechanisms behind this consists of epigenetic modifications, as demonstrated by the restoration of MHC class I expression as well as the T cell-mediated killing of tumour cells due to the pharmacological inhibition of EZH2 in SCLC cell lines [142].
Conversely, considering the lack of function of T cells in the TME, recent studies found that DNMT inhibitors and histone modifications inhibitors can reverse tumour immune evasion. This is in addition to being able to modulate T cell exhaustion state towards effector and memory T cell phenotypes in mouse models of NSCLC, ovarian cancer and melanoma cell lines, thus sensitizing to anti-CTLA4 and anti-PD1 therapy [143,144,145]. In this context, the LSD1 HDM plays a critical role due to its ability to suppress endogenous double stranded RNA (dsRNA) levels and interferon (IFN) responses in tumour cells, as demonstrated by a preclinical study that showed that LSD1 inhibition in tumour cells causes intracellular dsRNA stress and resultant IFN activation and anti-tumour T cell immunity promotion. Moreover, the authors demonstrated that LSD1 depletion converts tumours resistant to PD-1 blockade to cells responsive to ICIs [146]. Based on these findings, clinical trials on LSD1 inhibitors in combination with ICIs are ongoing for various solid tumours, including SCLC. A phase I/II trial is evaluating bomedemstat in combination with maintenance atezolizumab for ED-SCLC in first-line following induction chemo-immunotherapy (NCT05191797). A phase IImulti-cohort study is ongoing to assess safety and efficacy of CC-90011, an oral LSD1 inhibitor, in combination with nivolumab in pre-treated SCLC patients (NCT04350463).
Epigenetic mechanisms also have a role in the evasion of innate immunity. In particular, it has been demonstrated that SCLC aggressiveness and metastasis is partially related to a low expression of NK-activating ligands (NKG2DL) due to an epigenetic silencing with consequent loss of NK cell recognition. Restoring NKG2DL using HDAC inhibitors in preclinical models suppressed tumour growth and metastasis by inducing infiltration and activation of NK and T cells [147]. Research on the potential role of these epigenetic drugs to overcome ICI resistance is at a very early stage, but expectations are high for the future.
Overcoming the ICI Resistance
After more than 20 years without innovation in SCLC management, ICIs targeting PD-1/PD-L1 axis have changed treatment algorithm for this disease [7••, 9••]. Moreover, there is a compelling rationale for ICI efficacy based on the high immunogenicity of SCLC related to a high genomic instability and consequent high tumour mutational burden (TMB) [148]. Nevertheless, ICI efficacy remains modest, and there is a significant focus on overcoming the ICI resistance in SCLC in first-line and relapsed settings.
The main strategy to increase ICI therapeutic activity consists of combinations of different immunotherapy approaches, which co-target immune molecules highly expressed in SCLC. Indeed, recent studies on SCLC biology showed a low expression of PD-L1 and tumour-infiltrating lymphocytes (TILs) in SCLC samples in contrast to a higher expression of other immune inhibitory proteins, such as B7-H3, the T cell immunoreceptor with immunoglobulin and ITIM domain (TIGIT), the T cell immunoglobulin mucin receptor 3 (TIM3), the lymphocyte activation gene 3 (LAG3), and others [149, 150].
TIGIT
TIGIT is transmembrane protein expressed by a variety of immune cells that, when activated, induces a tolerogenic microenvironment. In particular, it competes with CD226 (a transmembrane protein that enhances lymphocytes cytotoxicity mechanisms) on activated T cells for binding to CD155, CD112, and CD113 ligands, and it also acts on Natural Killer cells. Moreover, in various cancers, the TIGIT expression profile correlates with the expression of other immune inhibitors receptors, including LAG3, CTLA4, and PD-16–7. Anti-TIGIT monoclonal antibodies are tested in several ongoing trials, which have enrolled patients affected by various solid tumors. Considering limited-stage SCLC, a phase II-3-arm study (NCT04952597) is examining the combination of the anti-TIGIT monoclonal antibody ociperlimab (BGB-A1217) plus tislelizumab (anti-PD1) concurrent with chemoradiotherapy (CRT). Another phase II study (NCT04308785) is evaluating atezolizumab with or without the anti-TIGIT tiragolumab as consolidation therapy following CRT.
Turning to untreated ED-SCLC, the phase III SKYSCRAPER-02 (NCT04256421) is ongoing: 490 patients have been enrolled and randomized to receive standard care (atezolizumab plus carboplatin and etoposide) either with or without tiragolumab. Unfortunately, at the interim analysis at a median follow up of 14.3 months this study failed to meet its co-primary endpoints of PFS improvement (median PFS was 5.4 months (95% CI 4.7–5.5) with tiragolumab vs 5.6 months without this agent). Nevertheless, the study will continue as planned until final OS analysis [151, 152].
LAG-3
LAG-3 is an inhibitory immunoreceptor expressed on immune cells including activated T, T-regulatory, NK. and plasmacytoid dendritic cells. Its best-characterized ligand is the major histocompatibility complex class II (MHC-II), but there are other ligands, such as fibrinogen-like protein 1 (FGL-1). Sustained T cell activation in a chronic inflammatory environment, such as TME, increases LAG-3 co-expression with co-inhibitory receptors including PD-1 and, when activated, LAG-3 contributes to T cell suppression and subsequent immune dysfunction [153]. Several LAG-3–targeting molecules are currently in early stages of clinical development with early results suggesting a modest benefit when used as single agents, but dual LAG-3/PD-1 blockade has a significant role in reducing tumour growth by increasing the proportion of effector T cells in the tumour [154, 155].
To date, patients with SCLC have been recruited only in basket trials such as a phase 1/2 study evaluating LAG525 (anti-LAG3) in combination with spartalizumab (anti-PD-1). This SCLC cohort met the criteria for expansion on the basis of the clinical benefit rate [156]. The ongoing phase 1 trial is evaluating tebotelimab, which is a monoclonal antibody engineered to bind PD-1 and LAG-3 concomitantly or independently (NCT03219268).
TIM-3
TIM-3 is an inhibitory receptor with a crucial role in both innate and adaptive immune responses, and it is often co-expressed with PD-1 [156, 157]. TIM-3 also has a role in resisting PD-1 blockade, and preclinical studies demonstrated that dual TIM-3 and PD-1 blockade is more effective than targeting either pathway alone [158, 159]. Further, Anti-TIM3 drugs, like the anti-LAG3 drugs mentioned above, are at a very early stage of development. At the moment, the available data for SCLC patients derives from the small numbers of a phase 1 basket trial [160], so it will still take a long time to understand the real potential of these treatments.
Other strategies to overcome ICI resistance include combinations of ICIs with inhibitors of proteins that are highly or selectively expressed in SCLC, including PARP, already discussed in previous paragraph, and fucosyl-GM1.
Fucosyl-GM1
Fucosyl-GM1 is a monosialoganglioside with limited expression in normal tissue, but highly expressed on SCLC cells. Encouraging preclinical results of BMS-986012 [161], a monoclonal antibody that binds to Fucosyl-GM1 with high affinity and specificity, led to a phase 1/2 clinical trial. In this trial the safety and preliminary efficacy of BMS-986012, both as a monotherapy and in combination with nivolumab, in patients with relapsed or refractory SCLC were evaluated [162]. Considering that the study enrolled pre-treated patients, albeit some without ICIs, the BMS-986012 plus nivolumab combination showed encouraging results in this first-in-human study with an ORR of 38% and a median OS of 18.7 months. Based on these results, a phase 2 study is currently evaluating the safety and efficacy of BMS-986012 combined with carboplatin, etoposide, and nivolumab in first line (NCT04702880).
Finally, one of the major areas of study and interest in the field of immunotherapy is chimeric antigen receptor (CAR) T cells, which are genetically modified T lymphocytes with a transgenic receptor capable of binding a specific antigen as well as an intracellular signalling domain that triggers the cytolytic effects. Initially, CAR T cells therapy was developed for blood malignancies but in recent years its use is being trialled for solid tumours, with encouraging results, even if they are less significant than haematological malignancies, which is due to tumour heterogeneity and hostile TME[163]. Different CAR-T cells have been developed against SCLC with promising results in pre-clinical studies. DLL3-CAR T cells showed preclinical positive results in term of safety and efficacy [164]. Anti-CD56 CAR T cells significantly reduced tumour burden in animal models of neuroblastoma and SCLC, but they only had a modest effect on survival; further investigations to limit toxicity related to on-target, off-tissue effects are needed [165]. AC133-specific CAR T cells exhibited an important cytotoxicity, and prolonged survival in a humanized orthotopic SCLC model, with a modest activity in monotherapy and a higher efficacy when combined with PD-1-inhibition and CD73-inhibition [166]. GD2-specific CAR T cells reduced the tumour burden in vitro and in vivo in xenograft models of GD2-expressing lung tumours (SCLC and NSCLC), and the susceptibility of tumours to this treatment was enhanced by pre-treatment with tazemetostat (EZH2 inhibitor), which can upregulate GD2 expression in tumour cells [167].
A phase 1 study to assess the tolerability and safety of the AMG 119, an anti-DLL-3 CAR T, was initiated in 2018, but it has been suspended for the time being (NCT03392064). A phase 1 first-in-human study with DLL3-targeted chimeric antigen receptor T cells (LB2102) and a phase 1 with autologous CAR T cells against the GD2 antigen in patients with advanced lung cancer, NSCLC and SCLC, are being planned (NCT05680922 and NCT05620342). So far, CAR-T cells have shown promising results in preclinical studies, but many more studies in-humans are needed before we can consider them an innovative therapeutic strategy in SCLC.
Conclusions
Recently, many studies have led to the identification of the specific genetic abnormalities that characterise SCLC. The growing dataset in gene expression profiling in SCLC seems to be suggesting a new direction. A deeper understanding of SCLC at molecular level is at the base for the development of new, effective, and safe treatments. Several encouraging therapeutic approaches have been reported, the most promising of which target angiogenesis and epigenetics. Finally, therapies targeting proteins or oncogenic drivers show positive preliminary results (Table 2). Further studies based on molecular classification of SCLC and updated data on these novel therapies will hopefully improve outcomes in SCLC. Nonetheless, more research is needed to face the currently dismal prognosis and prospects of SCLC.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Van Meerbeeck JP, Fennell DA, De Ruysscher DKM. Small-cell lung cancer. Lancet. 2011;378:1741–55.
George J, Lim JS, Jang SJ, Cun Y, Ozretić L, Kong G, et al. Comprehensive genomic profiles of small cell lung cancer. Nature. 2015;524(7563):47–53.
Govindan R, Page N, Morgensztern D, Read W, Tierney R, Vlahiotis A, et al. Changing epidemiology of small-cell lung cancer in the United States over the last 30 years: analysis of the surveillance, epidemiologic, and end results database. J Clin Oncol. 2006;24:4539–44.
Ortega-Franco A, Ackermann C, Paz-Ares L, Califano R. First-line immune checkpoint inhibitors for extensive stage small-cell lung cancer: clinical developments and future directions. ESMO Open. 2021;6:100003.
Früh M, De Ruysscher D, Popat S, Crinò L, Peters S, Felip E. Small-cell lung cancer (SCLC): ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Annal Oncol. 2013;24:99–105.
Lally BE, Urbanic JJ, Blackstock AW, Miller AA, Perry MC. Small cell lung cancer: have we made any progress over the last 25 years? Lear Object Oncol. 2007;12:1096–104.
Horn L, Mansfield AS, Szczęsna A, Havel L, Krzakowski M, Hochmair MJ, et al. First-line atezolizumab plus chemotherapy in extensive-stage small-cell lung cancer. N Engl J Med. 2018;379:2220–9. This study results’ have been practice changing, leading to the introduction of a new standard of therapy.
Wang J, Zhou C, Yao W, Wang Q, Min X, Chen G, et al. Adebrelimab or placebo plus carboplatin and etoposide as first-line treatment for extensive-stage small-cell lung cancer (CAPSTONE-1): a multicentre, randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2022;23:739–47.
Paz-Ares L, Dvorkin M, Chen Y, Reinmuth N, Hotta K, Trukhin D, et al. Durvalumab plus platinum–etoposide versus platinum–etoposide in first-line treatment of extensive-stage small-cell lung cancer (CASPIAN): a randomised, controlled, open-label, phase 3 trial. Lancet. 2019;394:1929–39. This study results’ have been practice changing, leading to the introduction of a new standard of therapy.
Cheng Y, Han L, Wu L, Chen J, Sun H, Wen G, et al. Effect of first-line serplulimab vs placebo added to chemotherapy on survival in patients with extensive-stage small cell lung cancer: The ASTRUM-005 Randomized Clinical Trial. JAMA. 2022;328:1223–32.
Rudin CM, Durinck S, Stawiski EW, Poirier JT, Modrusan Z, Shames DS, et al. Comprehensive genomic analysis identifies SOX2 as a frequently amplified gene in small-cell lung cancer. Nat Genet. 2012;44:1111–6.
Gazdar AF, Bunn PA, Minna JD. Small-cell lung cancer: what we know, what we need to know and the path forward Nat Rev Cancer. Nat Publ Group. 2017;17(12):725–37.
Peifer M, Fernández-Cuesta L, Sos ML, George J, Seidel D, Kasper LH, et al. Integrative genome analyses identify key somatic driver mutations of small-cell lung cancer. Nat Genet. 2012;44:1104–10.
Arcaro A. Targeted therapies for small cell lung cancer: Where do we stand? Crit Rev Oncol Hematol. Elsevier Ireland Ltd. 2015;95:154–64.
Bunn PA, Minna JD, Augustyn A, Gazdar AF, Ouadah Y, Krasnow MA, et al. Small cell lung cancer: can recent advances in biology and molecular biology be translated into improved outcomes? Journal of Thoracic Oncology. Lippincott Williams and Wilkins; 2016. 453–74.
Carney DN, Gazdar AF, Bepler G, et al. Establishment and identification of small cell lung cancer cell. Cancer Res. 1985;45:2913–23.
Poirier JT, Dobromilskaya I, Moriarty WF, Peacock CD, Hann CL, Rudin CM. Selective tropism of Seneca Valley virus for variant subtype small cell lung cancer. J Natl Cancer Inst. 2013;105:1059–65.
Borromeo MD, Savage TK, Kollipara RK, He M, Augustyn A, Osborne JK, et al. ASCL1 and NEUROD1 reveal heterogeneity in pulmonary neuroendocrine tumors and regulate distinct genetic programs. Cell Rep. 2016;16:1259–72. This study describes a new molecular subtype.
McColl K, Wildey G, Sakre N, Lipka MB, Behtaj M, Kresak A, et al. Reciprocal expression of INSM1 and YAP1 defines subgroups in small cell lung cancer. Oncotarget. 2017;8:73745–56.
Qu S, Fetsch P, Thomas A, Pommier Y, Schrump DS, Miettinen MM, et al. Molecular subtypes of primary SCLC tumors and their associations with neuroendocrine and therapeutic markers. J Thorac Oncol. 2022;17:141–53 This study describes a new molecular subtype.
Baine MK, Hsieh MS, Lai WV, Egger JV, Jungbluth AA, Daneshbod Y, et al. SCLC subtypes defined by ASCL1, NEUROD1, POU2F3, and YAP1: a comprehensive immunohistochemical and histopathologic characterization. J Thorac Oncol. 2020;15:1823–35.
Pearsall SM, Humphrey S, Revill M, Morgan D, Frese KK, Galvin M, et al. The Rare YAP1 Subtype of SCLC Revisited in a Biobank of 39 circulating tumor cell patient derived explant models: A Brief Report. J Thorac Oncol. 2020;15:1836–43. This study summarizes the SCLC classification.
Owonikoko TK, Dwivedi B, Chen Z, Zhang C, Barwick B, Ernani V, et al. YAP1 Expression in SCLC defines a distinct subtype with T-cell-inflamed phenotype. J Thorac Oncol. 2021;16:464–76. This study describes a new molecular subtype.
Gay CM, Stewart CA, Park EM, Diao L, Groves SM, Heeke S, et al. Patterns of transcription factor programs and immune pathway activation define four major subtypes of SCLC with distinct therapeutic vulnerabilities. Cancer Cell. 2021;39:346-360.e7.
O’Connor MJ. Targeting the DNA Damage Response in Cancer. Mol Cell. 2015;60:547–60.
Pearl LH, Schierz AC, Ward SE, Al-Lazikani B, Pearl FMG. Therapeutic opportunities within the DNA damage response. Nat Rev Cancer. 2015;15:166–80.
Farago AF, Yeap BY, Stanzione M, Hung YP, Heist RS, Marcoux JP, et al. Combination olaparib and temozolomide in relapsed small-cell lung cancer. Cancer Discov. 2019;9:1372–87.
Pietanza MC, Waqar SN, Krug LM, Dowlati A, Hann CL, Chiappori A, et al. Randomized, double-blind, phase ii study of temozolomide in combination with either veliparib or placebo in patients with relapsed-sensitive or refractory small-cell lung cancer. J Clin Oncol. 2018;36:2386–94.
Woll P, Gaunt P, Danson S, Steele N, Ahmed S, Mulatero C, et al. Olaparib as maintenance treatment in patients with chemosensitive small cell lung cancer (STOMP): A randomised, double-blind, placebo-controlled phase II trial. Lung Cancer. 2022;171:26–33.
Owonikoko TK, et al. Randomized phase II trial of cisplatin and etoposide in combination with veliparib or placebo for extensive-stage small-cell lung cancer: ECOG-ACRIN 2511 study. J Clin Oncol. 2019;37(3):222–9. https://doi.org/10.1200/JCO.18.00264.
Byers LA, Bentsion D, Gans S, Penkov K, Son C, Sibille A, et al. Veliparib in combination with carboplatin and etoposide in patients with treatment-naïve extensive-stage small cell lung cancer: a phase 2 randomized study. Clin Cancer Res. 2021;27:3884–95.
Ai X, Pan Y, Shi J, Yang N, Liu C, Zhou J, et al. efficacy and safety of niraparib as maintenance treatment in patients with extensive-stage sclc after first-line chemotherapy: a randomized, double-blind, phase 3 study. J Thorac Oncol. 2021;16:1403–14.
Sen T, Rodriguez BL, Chen L, Della Corte CM, Morikawa N, Fujimoto J, et al. Targeting DNA damage response promotes antitumor immunity through STING-mediated T-cell activation in small cell lung cancer. Cancer Discov. 2019;9:646–61.
Zhang N, Gao Y, Huang Z, Dai P, Luo Y, Wu Q, et al. PARP inhibitor plus radiotherapy reshapes an inflamed tumor microenvironment that sensitizes small cell lung cancer to the anti-PD-1 immunotherapy. Cancer Lett. 2022;545:215852.
Thomas A, Vilimas R, Trindade C, Erwin-Cohen R, Roper N, Xi L, et al. Durvalumab in combination with olaparib in patients with relapsed sclc: results from a phase II study. J Thorac Oncol. 2019;14:1447–57.
Nuñez GS, Robles CMG, Giraudon C, Martínez-Leal JF, Compe E, Coin F, et al. Lurbinectedin specifically triggers the degradation of phosphorylated RNA polymerase II and the formation DNA breaks in cancer cells. Mol Cancer Ther. 2016;15:2399–412.
Singh S, Jaigirdar AA, Mulkey F, Cheng J, Hamed SS, Li Y, et al. FDA approval summary: lurbinectedin for the treatment of metastatic small cell lung cancer. Clin Cancer Res. 2021;27:2378–82. Available from: https://pubmed.ncbi.nlm.nih.gov/33288660/
Belgiovine C, Bello E, Liguori M, Craparotta I, Mannarino L, Paracchini L, et al. Lurbinectedin reduces tumour-associated macrophages and the inflammatory tumour microenvironment in preclinical models. Br J Cancer. 2017;117:628–38.
Sala MA, et al. Lurbinectedin as second-line treatment for patients with small-cell lung cancer: a single-arm, open-label, phase 2 basket trial. Lancet Oncol. 2020;21:645–54.
Calvo E, Moreno V, Flynn M, Holgado E, Olmedo ME, Criado MPL, et al. Antitumor activity of lurbinectedin (PM01183) and doxorubicin in relapsed small-cell lung cancer: results from a phase I study. Ann Oncol. 2017;28:2559–66.
Aix SP, Ciuleanu TE, Navarro A, Cousin S, Bonanno L, Smit EF, et al. Combination lurbinectedin and doxorubicin versus physician’s choice of chemotherapy in patients with relapsed small-cell lung cancer (ATLANTIS): a multicentre, randomised, open-label, phase 3 trial. Lancet Respir Med. 2023;11:74–86.
Yuan M, Zhao Y, Arkenau HT, Lao T, Chu L, Xu Q. Signal pathways and precision therapy of small-cell lung cancer. Springer Nature: Signal Transduct Target Ther; 2022.
Wan J, Chai H, Yu Z, Ge W, Kang N, Xia W, et al. HIF-1a effects on angiogenic potential in human small cell lung carcinoma. 2011. http://www.jeccr.com/content/30/1/77
Montanino A, Manzo A, Carillio G, Palumbo G, Esposito G, Sforza V, et al. Angiogenesis inhibitors in small cell lung cancer. Front Oncol. Frontiers Media S.A.; 2021.
Spigel DR, Waterhouse DM, Lane S, Legenne P, Bhatt K. Efficacy and safety of oral topotecan and bevacizumab combination as second-line treatment for relapsed small-cell lung cancer: an open-label multicenter single-arm phase II study. Clin Lung Cancer. 2013;14:356–63.
Mountzios G, Emmanouilidis C, Vardakis N, Kontopodis E, Hatzidaki D, Popis E, et al. Paclitaxel plus bevacizumab in patients with chemoresistant relapsed small cell lung cancer as salvage treatment: a phase II multicenter study of the Hellenic Oncology Research Group. Lung Cancer. 2012;77:146–50.
Calvo E, Sessa C, Harada G, de Miguel M, Kahatt C, Luepke-Estefan XE, et al. Phase I study of lurbinectedin in combination with weekly paclitaxel with or without bevacizumab in patients with advanced solid tumors. Invest New Drugs. 2022;40:1263–73.
Ready NE, Dudek AZ, Pang HH, Hodgson LD, Graziano SL, Green MR, et al. Cisplatin, irinotecan, and bevacizumab for untreated extensive-stage small-cell lung cancer: CALGB 30306, a phase II study. J Clin Oncol. 2011;29:4436–41.
Spigel DR, Greco FA, Zubkus JD, Murphy PB, Saez RA, Farley C, et al. Phase II trial of irinotecan, carboplatin, and bevacizumab in the treatment of patients with extensive-stage small-cell lung cancer. J Thorac Oncol. 2009;4:1555–60.
Horn L, Dahlberg SE, Sandler AB, Dowlati A, Moore DF, Murren JR, et al. Phase II study of cisplatin plus etoposide and bevacizumab for previously untreated, extensive-stage small-cell lung cancer: Eastern cooperative oncology group study E3501. J Clin Oncol. 2009;27:6006–11.
Spigel DR, Townley PM, Waterhouse DM, Fang L, Adiguzel I, Huang JE, et al. Randomized phase II study of bevacizumab in combination with chemotherapy in previously untreated extensive-stage small-cell lung cancer: results from the SALUTE trial. J Clin Oncol. 2011;29:2215–22.
Pujol JL, Lavole A, Quoix E, Molinier O, Souquet PJ, Barlesi F, et al. Randomized phase II-III study of bevacizumab in combination with chemotherapy in previously untreated extensive small-cell lung cancer: results from the IFCT-0802 trial†. Ann Oncol. 2015;26:908–14.
Tiseo M, Boni L, Ambrosio F, Camerini A, Baldini E, Cinieri S, et al. Italian, Multicenter, Phase III, Randomized Study of Cisplatin Plus Etoposide With or Without Bevacizumab as First-Line Treatment in Extensive-Disease Small-Cell Lung Cancer: The GOIRC-AIFA FARM6PMFJM Trial. J Clin Oncol. 2017;35:1281–7.
Patton JF, Spigel DR, Greco FA, Liggett WH, Zubkus JD, Baskette M, et al. Irinotecan (I), carboplatin (C), and radiotherapy (RT) followed by maintenance bevacizumab (B) in the treatment (tx) of limited-stage small cell lung cancer (LS-SCLC): Update of a phase II trial of the Minnie Pearl Cancer Research Network. J Clin Oncol. 2006;24:7085–7085.
Sun JM, Lee KH, Kim BS, Kim HG, Min YJ, Yi SY, et al. Pazopanib maintenance after first-line etoposide and platinum chemotherapy in patients with extensive disease small-cell lung cancer: a multicentre, randomised, placebo-controlled Phase II study (KCSG-LU12-07). Br J Cancer. 2018;118:648–53.
Koinis F, Agelaki S, Karavassilis V, Kentepozidis N, Samantas E, Peroukidis S, et al. Second-line pazopanib in patients with relapsed and refractory small-cell lung cancer: a multicentre phase II study of the Hellenic Oncology Research Group. Br J Cancer. 2017;117:8–14.
Spigel DR, Greco FA, Rubin MS, Shipley D, Thompson DS, Lubiner ET, et al. Phase II study of maintenance sunitinib following irinotecan and carboplatin as first-line treatment for patients with extensive-stage small-cell lung cancer. Lung Cancer. 2012;77:359–64.
Ready NE, Pang HH, Gu L, Otterson GA, Thomas SP, Miller AA, et al. Chemotherapy with or without maintenance sunitinib for untreated extensive-stage small-cell lung cancer: a randomized, double-blind, placebo-controlled phase II study-CALGB 30504 (Alliance). J Clin Oncol. 2015;33:1660–5.
Han JY, Kim HY, Lim KY, Hwangbo B, Lee JS. A phase II study of nintedanib in patients with relapsed small cell lung cancer. Lung Cancer. 2016;96:108–12.
Cheng Y, Wang Q, Li K, Shi J, Liu Y, Wu L, et al. Anlotinib vs placebo as third- or further-line treatment for patients with small cell lung cancer: a randomised, double-blind, placebo-controlled Phase 2 study. Br J Cancer. 2021;125:366–71.
Kong T, Chen L, Zhao X, Duan F, Zhou H, Wang L, et al. Anlotinib plus etoposide and cisplatin/carboplatin as first-line therapy for extensive-stage small cell lung cancer (ES-SCLC): a single-arm, phase II study. Invest New Drugs. 2022;40:1095–105.
Liu C, Liao J, Wu X, Zhao X, Sun S, Wang H, et al. A phase II study of anlotinib combined with etoposide and platinum-based regimens in the first-line treatment of extensive-stage small cell lung cancer. Thorac Cancer. 2022;13:1463–70.
Teng F, Xing P, Yang K, Gao L, Tian Z, Li J. Apatinib as maintenance therapy following standard first-line chemotherapy in extensive disease small cell lung cancer: A phase II single-arm trial. Thorac Cancer. 2022;13:557–62.
Luo H, Zhang L, Yang B, Feng Y, Xiong Y, Zhang S, et al. A randomized phase 2 trial of apatinib vs observation as maintenance treatment following first-line induction chemotherapy in extensive- stage small cell lung cancer. Invest New Drugs. 2020;38:148–59.
Xu Y, Huang Z, Lu H, Yu X, Li Y, Li W, et al. Apatinib in patients with extensive-stage small-cell lung cancer after second-line or third-line chemotherapy: a phase II, single-arm, multicentre, prospective study. Br J Cancer. 2019;121:640–6.
Xu Y, Wang X, Sun C, Gao Z, He H, Qiu S, et al. A phase II study of antiangiogenic therapy (Apatinib) plus chemotherapy as second-line treatment in advanced small cell lung cancer. Cancer Med. 2023;12:3.
Waks Z, Weissbrod O, Carmeli B, Norel R, Utro F, Goldschmidt Y. Driver gene classification reveals a substantial overrepresentation of tumor suppressors among very large chromatin-regulating proteins. Sci Rep. 2016;6:1.
Chen J, Guanizo A, Luong Q, Jayasekara WSN, Jayasinghe D, Inampudi C, et al. Lineage-restricted neoplasia driven by Myc defaults to small cell lung cancer when combined with loss of p53 and Rb in the airway epithelium. Oncogene. 2022;41:138–45.
Desai A, Abdayem P, Adjei AA, Planchard D. Antibody-drug conjugates: a promising novel therapeutic approach in lung cancer. Lung Cancer. 2022;163:96–106.
Owen DH, Giffin MJ, Bailis JM, Smit MD, Carbone DP, He K. DLL3: an emerging target in small cell lung cancer. J Hematol Oncol. 2019;12(1):61.
Furuta M, Kikuchi H, Shoji T, Takashima Y, Kikuchi E, Kikuchi J, et al. DLL3 regulates the migration and invasion of small cell lung cancer by modulating Snail. Cancer Sci. 2019;110:1599–608.
Uprety D, Remon J, Adjei AA. All that glitters is not gold: the story of rovalpituzumab tesirine in SCLC. J Thorac Oncol. 2021;16(9):1429–33.
Rudin CM, Pietanza MC, Bauer TM, Ready N, Morgensztern D, Glisson BS, et al. Rovalpituzumab tesirine, a DLL3-targeted antibody-drug conjugate, in recurrent small-cell lung cancer: a first-in-human, first-in-class, open-label, phase 1 study. Lancet Oncol. 2017;18:42–51.
Malhotra J, Nikolinakos P, Leal T, Lehman J, Morgensztern D, Patel JD, et al. A PHASE 1–2 study of rovalpituzumab tesirine in combination with nivolumab plus or minus ipilimumab in patients with previously treated extensive-stage SCLC. J Thorac Oncol. 2021;16:1559–69.
Udagawa H, Akamatsu H, Tanaka K, Takeda M, Kanda S, Kirita K, et al. Phase I safety and pharmacokinetics study of rovalpituzumab tesirine in Japanese patients with advanced, recurrent small cell lung cancer. Lung Cancer. 2019;135:145–50.
Hann CL, Burns TF, Dowlati A, Morgensztern D, Ward PJ, Koch MM, et al. A phase 1 study evaluating rovalpituzumab tesirine in frontline treatment of patients with extensive-stage SCLC. J Thorac Oncol. 2021;16:1582–8.
Morgensztern D, Besse B, Greillier L, Santana-Davila R, Ready N, Hann CL, et al. Efficacy and safety of rovalpituzumab tesirine in third-line and beyond patients with DLL3-expressing, relapsed/refractory small-cell lung cancer: Results from the phase II TRINITY study. Clin Cancer Res. 2019;25:6958–66.
Blackhall F, Jao K, Greillier L, Cho BC, Penkov K, Reguart N, et al. Efficacy and safety of rovalpituzumab tesirine compared with topotecan as second-line therapy in dll3-high sclc: results from the phase 3 TAHOE study. J Thorac Oncol. 2021;16:1547–58.
Johnson ML, Zvirbule Z, Laktionov K, Helland A, Cho BC, Gutierrez V, et al. Rovalpituzumab tesirine as a maintenance therapy after first-line platinum-based chemotherapy in patients with extensive-stage–SCLC: results from the phase 3 MERU Study. J Thorac Oncol. 2021;16:1570–81.
Cortinovis DL, Colonese F, Abbate MI, Sala L, Meazza Prina M, Cordani N, et al. Harnessing DLL3 inhibition: from old promises to new therapeutic horizons. Front Med (Lausanne). 2022;9:989405.
Owonikoko T, Boyer M, Johnson M, Govindan R, Rodrigues L, Blackhall F, et al. OA11.03 A Phase 1 study of AMG 757, half-life extended bispecific T-cell engager (BiTE®)immune therapy against DLL3, in SCLC. J Thorac Oncol. 2021;16:126.
Paz-Ares L, Champiat S, Lai WV, Izumi H, Govindan R, Boyer M, et al. Tarlatamab, a first-in-class DLL3-targeted bispecific T cell engager, in recurrent small-cell lung cancer: an open-label, phase 1 study. Journal of Clinical Oncology. 2023;. https://doi.org/10.1200/JCO.22.02823
Shah MH, Lorigan P, O’Brien ME, Fossella FV, Moore KN, Bhatia S, et al. Phase I study of IMGN901, a CD56-targeting antibody-drug conjugate, in patients with CD56-positive solid tumors. Invest New Drugs. 2016;34(3):290–9.
Socinski MA, Kaye FJ, Spigel DR, Kudrik FJ, Ponce S, Ellis PM, et al. Phase 1/2 study of the CD56-targeting antibody-drug conjugate lorvotuzumab mertansine (IMGN901) in combination with carboplatin/etoposide in small-cell lung cancer patients with extensive-stage disease. Clin Lung Cancer. 2017;18:68-76.e2.
Goldenberg DM, Sharkey RM. Antibody-drug conjugates targeting TROP-2 and incorporating SN-38: a case study of anti-TROP-2 sacituzumab govitecan. InMAbs. 2019;11:987–95.
Bardia A, Messersmith WA, Kio EA, Berlin JD, Vahdat L, Masters GA, et al. Sacituzumab govitecan, a Trop-2-directed antibody-drug conjugate, for patients with epithelial cancer: final safety and efficacy results from the phase I/II IMMU-132-01 basket trial. Ann Oncol. 2021;32:746–56.
Gray JE, Heist RS, Starodub AN, Camidge DR, Kio EA, Masters GA, et al. Therapy of small cell lung cancer (SCLC) with a topoisomerase-I-inhibiting antibody-drug conjugate (ADC) targeting trop-2, sacituzumab govitecan. Cancer Therapy: Clinical. 2017;23.
Pietanza MC, Litvak AM, Varghese AM, Krug LM, Fleisher M, Teitcher JB, et al. A phase I trial of the Hedgehog inhibitor, sonidegib (LDE225), in combination with etoposide and cisplatin for the initial treatment of extensive stage small cell lung cancer. Lung Cancer. 2016;99:23–30.
Belani CP, Dahlberg SE, Rudin CM, Fleisher M, Chen HX, Takebe N, et al. Vismodegib or cixutumumab in combination with standard chemotherapy for patients with extensive-stage small cell lung cancer: A trial of the ECOG-ACRIN cancer research group (E1508). Cancer. 2016;122:2371–8.
Mak DWS, Li S, Minchom A. Challenging the recalcitrant disease—developing molecularly driven treatments for small cell lung cancer. Eur J Cancer. 2019;119:132–50.
Rudin CM, Hann CL, Garon EB, Ribeiro De Oliveira M, Bonomi PD, Camidge DR, et al. Phase II study of single-agent navitoclax (ABT-263) and biomarker correlates in patients with relapsed small cell lung cancer. Clin Cancer Res. 2012;18:3163–9.
Gandhi L, Camidge DR, de Oliveira MR, Bonomi P, Gandara D, Khaira D, et al. Phase I study of Navitoclax (ABT-263), a novel Bcl-2 family inhibitor, in patients with small-cell lung cancer and other solid tumors. J Clin Oncol. 2011;29:909–16.
Lochmann TL, Floros KV, Naseri M, Powell KM, Cook W, March RJ, et al. Venetoclax is effective in small-cell lung cancers with high BCL-2 expression. Clin Cancer Res. 2018;24:360–9.
Langer CJ, Albert I, Ross HJ, Kovacs P, Blakely LJ, Pajkos G, et al. Randomized phase II study of carboplatin and etoposide with or without obatoclax mesylate in extensive-stage small cell lung cancer. Lung Cancer. 2014;85:420–8.
Heist RS, Fain J, Chinnasami B, Khan W, Molina JR, Sequist LV, et al. Phase I/II study of AT-101 with topotecan in relapsed and refractory small cell lung cancer. J Thoracic Oncol. 2010;5(10):1637–43.
Stefani A, Piro G, Schietroma F, Strusi A, Vita E, Fiorani S, et al. Unweaving the mitotic spindle: a focus on aurora kinase inhibitors in lung cancer. Front Oncol. Frontiers Media S.A.; 2022.
Mollaoglu G, Guthrie MR, Böhm S, Brägelmann J, Can I, Ballieu PM, et al. myc drives progression of small cell lung cancer to a variant neuroendocrine subtype with vulnerability to aurora kinase inhibition. Cancer Cell. 2017;31:270–85.
Melichar B, Adenis A, Lockhart AC, Bennouna J, Dees EC, Kayaleh O, et al. Safety and activity of alisertib, an investigational aurora kinase A inhibitor, in patients with breast cancer, small-cell lung cancer, non-small-cell lung cancer, head and neck squamous-cell carcinoma, and gastro-oesophageal adenocarcinoma: a five-arm phase 2 study. Lancet Oncol. 2015;16:395–405.
Chalishazar MD, Wait SJ, Huang F, Ireland AS, Mukhopadhyay A, Lee Y, et al. MYC-driven small-cell lung cancer is metabolically distinct and vulnerable to arginine depletion. Clin Cancer Res. 2019;25:5107–21.
Owonikoko TK, Niu H, Nackaerts K, Csoszi T, Ostoros G, Mark Z, et al. Randomized Phase II Study of paclitaxel plus alisertib versus paclitaxel plus placebo as second-line therapy for SCLC: primary and correlative biomarker analyses. J Thorac Oncol. 2020;15:274–87.
Cohen RB, Jones SF, Aggarwal C, von Mehren M, Cheng J, Spigel DR, et al. A phase I dose-escalation study of danusertib (PHA-739358) administered as a 24-hour infusion with and without granulocyte colony-stimulating factor in a 14-day cycle in patients with advanced solid tumors.
Schöffski P, Besse B, Gauler T, de Jonge MJA, Scambia G, Santoro A, et al. Efficacy and safety of biweekly i.v. administrations of the Aurora kinase inhibitor danusertib hydrochloride in independent cohorts of patients with advanced or metastatic breast, ovarian, colorectal, pancreatic, small-cell and non-small-cell lung cancer: A multi-tumour,multi-institutional phase II study. Annal Oncol. 2015;26:598–607.
Byers LA, Navarro A, Schaefer E, Johnson M, Özgüroğlu M, Han JY, et al. A phase II trial of prexasertib (LY2606368) in patients with extensive-stage small-cell lung cancer. Clin Lung Cancer. 2021;22:531–40.
Kolla BC, Racila E, Patel MR. Deep and prolonged response to aurora a kinase inhibitor and subsequently to nivolumab in MYCL1-driven small-cell lung cancer: case report and literature review. Case Rep Oncol Med. 2020;2020:1–6.
Hook KE, Garza SJ, Lira ME, Ching KA, Lee NV, Cao J, et al. An integrated genomic approach to identify predictive biomarkers of response to the aurora kinase inhibitor PF-03814735. Mol Cancer Ther. 2012;11:710–9.
Chen Y, Hong T, Wang S, Mo J, Tian T, Zhou X. Epigenetic modification of nucleic acids: from basic studies to medical applications. Chem Soc Rev. 2017;46(10):2844–72.
Tlemsani C, Pongor L, Elloumi F, Girard L, Huffman KE, Roper N, et al. SCLC-CellMiner: a resource for small cell lung cancer cell line genomics and pharmacology based on genomic signatures. Cell Rep. 2020;33(3):108296.
Zhao Z, Szczepanski AP, Tsuboyama N, Abdala-Valencia H, Goo YA, Singer BD, et al. Pax9 determines epigenetic state transition and cell fate in cancer. Cancer Res. 2021;81:4696–708.
Zheng Y, Wang Z, Wei S, Liu Z, Chen G. Epigenetic silencing of chemokine CCL2 represses macrophage infiltration to potentiate tumor development in small cell lung cancer. Cancer Lett. 2021;499:148–63.
Cao J, Song Y, Bi N, Shen J, Liu W, Fan J, et al. DNA methylation-mediated repression of miR-886-3p predicts poor outcome of human small cell lung cancer. Cancer Res. 2013;73:3326–35.
Fang S, Shen Y, Chen B, Wu Y, Jia L, Li Y, et al. H3K27me3 induces multidrug resistance in small cell lung cancer by affecting HOXA1 DNA methylation via regulation of the lncRNA HOTAIR. Ann Transl Med. 2018;6:440–440.
Zhao L, Wu X, Zheng J, Dong D. DNA methylome profiling of circulating tumor cells in lung cancer at single base-pair resolution. Oncogene. 2021;40:1884–95.
Kalari S, Jung M, Kernstine KH, Takahashi T, Pfeifer GP. The DNA methylation landscape of small cell lung cancer suggests a differentiation defect of neuroendocrine cells. Oncogene. 2013;32:3559–68.
Poirier JT, Gardner EE, Connis N, Moreira AL, De Stanchina E, Hann CL, et al. DNA methylation in small cell lung cancer defines distinct disease subtypes and correlates with high expression of EZH2. Oncogene. 2015;34:5869–78.
Cao J, Yan Q. Cancer epigenetics, tumor immunity, and immunotherapy. Trends Cancer. 2020;6(7):580–92.
Ahuja N, Sharma AR, Baylin SB. Epigenetic therapeutics: a new weapon in the war against cancer. Annu Rev Med. 2016;67:73–89.
Audia JE, Campbell RM. Histone modifications and cancer. Cold Spring Harb Perspect Biol. 2016;8(4):e019521.
Jia D, Augert A, Kim DW, Eastwood E, Wu N, Ibrahim AH, et al. Crebbp loss drives small cell lung cancer and increases sensitivity to HDAC inhibition. Cancer Discov. 2018;8:1422–37.
Qiu Z, Zhu W, Meng H, Tong L, Li X, Luo P, et al. CDYL promotes the chemoresistance of small cell lung cancer by regulating H3K27 trimethylation at the CDKN1C promoter. Theranostics. 2019;9:4717–29.
Oser MG, Sabet AH, Gao W, Chakraborty AA, Schinzel AC, Jennings RB, et al. The KDM5A/RBP2 histone demethylase represses NOTCH signaling to sustain neuroendocrine differentiation and promote small cell lung cancer tumorigenesis. Genes Dev. 2019;33(24):1718–38.
Zheng M, Niu Y, Bu J, Liang S, Zhang Z, Liu J, Guo L, Zhang Z, Wang Q. ESRP1 regulates alternative splicing of CARM1 to sensitize small cell lung cancer cells to chemotherapy by inhibiting TGF-β/Smad signaling. Aging. 2021;13(3):3554–72.
Sun L, He Q, Tsai C, Lei J, Chen J, Makcey LV, et al. HDAC inhibitors suppressed small cell lung cancer cell growth and enhanced the suppressive effects of receptor-targeting cytotoxins via upregulating somatostatin receptor II. Am J Transl Res. 2018.
Pan CH, Chang YF, Lee MS, Wen BC, Ko JC, Liang SK, et al. Vorinostat enhances the cisplatin-mediated anticancer effects in small cell lung cancer cells. BMC Cancer. 2016;16.
Crisanti MC, Wallace AF, Kapoor V, Vandermeers F, Dowling ML, Pereira LP, et al. The HDAC inhibitor panobinostat (LBH589) inhibits mesothelioma and lung cancer cells in vitro and in vivo with particular efficacy for small cell lung cancer. Mol Cancer Ther. 2009;8:2221–31.
Tsurutani J, Soda H, Oka M, Suenaga M, Doi S, Nakamura Y, et al. Antiproliferative effects of the histone deacetylase inhibitor FR901228 on small-cell lung cancer lines and drug-resistant sublines. Int J Cancer. 2003;104:238–42.
Platta CS, Greenblatt DY, Kunnimalaiyaan M, Chen H. The hdac inhibitor trichostatin a inhibits growth of small cell lung cancer cells. J Surg Res. 2007;142:219–26.
Balasubramaniam S, Redon CE, Peer CJ, Bryla C, Lee MJ, Trepel JB, et al. Phase I trial of belinostat with cisplatin and etoposide in advanced solid tumors, with a focus on neuroendocrine and small cell cancers of the lung. Anticancer Drugs. 2018;29:457–65.
DeMarinis F, Atmaca A, Tiseo M, Giuffreda L, Rossi A, Gebbia V, D’antonio C, Dal Zotto L, Al-Batran SE, Marsoni S, Wolf M. Phase II study of the histone deacetylase inhibitor panobinostat (LBH589) in pretreated patients with small-cell lung cancer. J Thorac Oncol. 2013;8(8):1091–4.
Otterson GA, Hodgson L, Pang H, Vokes EE, Cancer and Leukemia Group B. Phase II study of the histone deacetylase inhibitor Romidepsin in relapsed small cell lung cancer (Cancer and Leukemia Group B 30304). J Thorac Oncol. 2010;5:1644–8.
Gardner EE, Lok BH, Schneeberger VE, Desmeules P, Miles LA, Arnold PK, et al. Chemosensitive Relapse in small cell lung cancer proceeds through an EZH2-SLFN11 Axis. Cancer Cell. 2017;31:286–99.
Noce B, Di Bello E, Fioravanti R, Mai A. LSD1 inhibitors for cancer treatment: focus on multi-target agents and compounds in clinical trials. Front Pharmacol. Frontiers Media S.A.; 2023.
Augert A, Eastwood E, Ibrahim AH, Wu N, Grunblatt E, Basom R, et al. Targeting NOTCH activation in small cell lung cancer through LSD1 inhibition. Sci Signal. 2019;12.
Stewart CA, Byers LA. Altering the course of small cell lung cancer: targeting cancer stem cells via LSD1 inhibition. Cancer Cell. 2015;28(1):4–6.
Bauer TM, Besse B, Martinez-Marti A, Trigo JM, Moreno V, Garrido P, et al. Phase I, open-label, dose-escalation study of the safety, pharmacokinetics, pharmacodynamics, and efficacy of GSK2879552 in relapsed/refractory SCLC. J Thorac Oncol. 2019;14:1828–38.
Sivanandhan D, Rajagopal S, Nair S, Dhkar R, Viswakarma S, et al. Abstract 1756: JBI-802, novel dual inhibitor of LSD1-HDAC6 for treatment of cancer. Cancer Res. 2020;80:1756–1756.
Luszczek W, Cheriyath V, Mekhail TM, Borden EC. Combinations of DNA methyltransferase and histone deacetylase inhibitors induce DNA damage in small cell lung cancer cells: correlation of resistance with IFN-stimulated gene expression. Mol Cancer Ther. 2010;9:2309–21.
Hogg SJ, Beavis PA, Dawson MA, Johnstone RW. Targeting the epigenetic regulation of antitumour immunity. Nat Rev Drug Discov. 2020;10(11):776–800.
Chen X, Pan X, Zhang W, Guo H, Cheng S, He Q, et al. Epigenetic strategies synergize with PD-L1/PD-1 targeted cancer immunotherapies to enhance antitumor responses. Acta Pharm Sin B. 2020;10(5):723–33.
Yan F, Pang J, Peng Y, Molina JR, Yang P, Liu S. Elevated cellular PD1/PD-L1 expression confers acquired resistance to cisplatin in small cell lung cancer cells. PLoS One. 2016;11(9):0162925.
Doyle A, Martin WJ, Funa K, Gazdar A, Carney D, Martin SE, et al. Markedly decreased expression of class i histocompatibility antigens, protein, and mrna in human small-cell lung cancer. J Exp Med. 1985;161(5):1135–51.
Yazawa T, Kamma H, Fujiwara M, Matsui M, Horiguchi H, Satoh H, et al. Lack of class II transactivator causes severe deficiency of HLA-DR expression in small cell lung cancer. J Pathol. 1999;187:191–9.
Burr ML, Sparbier CE, Chan KL, Chan YC, Kersbergen A, Lam EYN, et al. An evolutionarily conserved function of polycomb silences the MHC class I antigen presentation pathway and enables immune evasion in cancer. Cancer Cell. 2019;36:385-401.e8.
Topper MJ, Vaz M, Chiappinelli KB, DeStefano Shields CE, Niknafs N, Chiu Yen R-W, et al. Epigenetic therapy ties MYC depletion to reversing immune evasion and treating lung cancer. Cell. 2017;171:1284–300. https://doi.org/10.1016/j.cell.
Chiappinelli KB, Strissel PL, Desrichard A, Li H, Henke C, Akman B, et al. Inhibiting DNA Methylation causes an interferon response in cancer via dsRNA including endogenous retroviruses. Cell. 2015;162:974–86.
Ghoneim HE, Fan Y, Moustaki A, Abdelsamed HA, Dash P, Dogra P, et al. De Novo Epigenetic programs inhibit PD-1 blockade-mediated T cell rejuvenation. Cell. 2017;170:142-157.e19.
Sheng W, LaFleur MW, Nguyen TH, Chen S, Chakravarthy A, Conway JR, et al. LSD1 Ablation stimulates anti-tumor immunity and enables checkpoint blockade. Cell. 2018;174:549-563.e19.
Zhu M, Huang Y, Bender ME, Girard L, Kollipara R, Eglenen-Polat B, et al. Evasion of innate immunity contributes to small cell lung cancer progression and metastasis. Cancer Res. 2021;81:1813–26.
Calles A, Aguado G, Sandoval C, Álvarez R. The role of immunotherapy in small cell lung cancer. Clinical and Translational Oncology. Springer-Verlag Italia s.r.l.; 2019; 961–76.
Carvajal-Hausdorf D, Altan M, Velcheti V, Gettinger SN, Herbst RS, Rimm DL. Expression and clinical significance of PD-L1, B7–H3, B7–H4 and TILs in human small cell lung Cancer (SCLC). J Immunother Cancer. 2019;7(1):65.
Dora D, Rivard C, Yu H, Bunn P, Suda K, Ren S, et al. Neuroendocrine subtypes of small cell lung cancer differ in terms of immune microenvironment and checkpoint molecule distribution. Mol Oncol. 2020;14:1947–65.
Annese T, Tamma R, Ribatti D. Update in TIGIT immune-checkpoint role in cancer. Front Oncol. Frontiers Media S.A.; 2022.
Rudin CM, Liu S V, Lu S, Soo RA, Hong MH, Lee J-S, et al. SKYSCRAPER-02: Primary results of a phase III, randomized, double-blind, placebo-controlled study of atezolizumab (atezo) + carboplatin + etoposide (CE) with or without tiragolumab (tira) in patients (pts) with untreated extensive-stage small cell lung cancer (ES-SCLC). 2022.
Andrews LP, Marciscano AE, Drake CG, Vignali DAA. LAG3 (CD223) as a cancer immunotherapy target. Immunol Rev. 2017;1:80–96.
Woo SR, Turnis ME, Goldberg MV, Bankoti J, Selby M, Nirschl CJ, et al. Immune inhibitory molecules LAG-3 and PD-1 synergistically regulate T-cell function to promote tumoral immune escape. Cancer Res. 2012;72:917–27.
Puhr HC, Ilhan-Mutlu A. New emerging targets in cancer immunotherapy: The role of LAG3. ESMO Open: BMJ Publishing Group; 2019.
Schöffski P, Tan DSW, Martín M, Ochoa-De-Olza M, Sarantopoulos J, Carvajal RD, et al. Phase I/II study of the LAG-3 inhibitor ieramilimab (LAG525) ± anti-PD-1 spartalizumab (PDR001) in patients with advanced malignancies. J Immunother Cancer. 2022;10.
Du W, Yang M, Turner A, Xu C, Ferris RL, Huang J, Kane LP, Lu B. TIM-3 as a target for cancer immunotherapy and mechanisms of action. Int J Mol Sci. 2017;18(3):645.
Sakuishi K, Apetoh L, Sullivan JM, Blazar BR, Kuchroo VK, Anderson AC. Targeting Tim-3 and PD-1 pathways to reverse T cell exhaustion and restore anti-tumor immunity. J Exp Med. 2010;207:2187–94.
Koyama S, Akbay EA, Li YY, Herter-Sprie GS, Buczkowski KA, Richards WG, et al. Adaptive resistance to therapeutic PD-1 blockade is associated with upregulation of alternative immune checkpoints. Nat Commun. 2016;7.
Curigliano G, Gelderblom H, Mach N, Doi T, Tai D, Forde PM, et al. Phase I/Ib clinical trial of sabatolimab, an anti–TIM-3 antibody, alone and in combination with spartalizumab, an anti–PD-1 antibody, in advanced solid tumors. Clin Cancer Res. 2021;27:3620–9.
Ponath P, Menezes D, Pan C, Chen B, Oyasu M, Strachan D, et al. A novel, fully human anti-fucosyl-GM1 antibody demonstrates potent in vitro and in vivo antitumor activity in preclinical models of small cell lung cancer. Clin Cancer Res. 2018;24:5178–89.
Chu Q, Leighl NB, Surmont V, van Herpen C, Sibille A, Markman B, et al. BMS-986012, an Anti–fucosyl-GM1 monoclonal antibody as monotherapy or in combination with nivolumab in relapsed/refractory SCLC: results from a first-in-human phase 1/2 study. JTO Clin Res Rep. 2022;3:100400.
Guzman G, Reed MR, Bielamowicz K, Koss B, Rodriguez A. CAR-T therapies in solid tumors: opportunities and challenges. Curr Oncol Rep [Internet]. 2023 [cited 2023 Jun 12];25. Available from: https://pubmed.ncbi.nlm.nih.gov/36853475/
Zhang Y, Tacheva-Grigorova SK, Sutton J, Melton Z, Mak YSL, Lay C, et al. Allogeneic CAR T cells targeting DLL3 are efficacious and safe in preclinical models of small cell lung cancer. Clin Cancer Res. 2023;29:971–85.
Crossland DL, Denning WL, Ang S, Olivares S, Mi T, Switzer K, et al. Antitumor activity of CD56-chimeric antigen receptor T cells in neuroblastoma and SCLC models. Oncogene. 2018;37:3686–97.
Taromi S, Firat E, Simonis A, Braun LM, Apostolova P, Elze M, et al. Enhanced AC133-specific CAR T cell therapy induces durable remissions in mice with metastatic small cell lung cancer. Cancer Lett. 2022;538:215697.
Reppel L, Tsahouridis O, Akulian J, Davis IJ, Lee H, Fucà G, et al. Targeting disialoganglioside GD2 with chimeric antigen receptor-redirected T cells in lung cancer. J Immunother Cancer. 2022;10.
Funding
Open access funding provided by Università degli Studi di Milano - Bicocca within the CRUI-CARE Agreement.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Canova, S., Trevisan, B., Abbate, M.I. et al. Novel Therapeutic Options for Small Cell Lung Cancer. Curr Oncol Rep 25, 1277–1294 (2023). https://doi.org/10.1007/s11912-023-01465-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11912-023-01465-7