Abstract
Purpose of RevSiew
Evidence is scaling up for sex differences in heart failure; however, clinical relevance of sex-specific differential thresholds for biomarkers is not clearly known. Current ambiguity warrants a further look into the sex-specific studies on cardiac biomarkers and may facilitate understanding of phenotypic presentations, clinical manifestations, and pathophysiologic pathway differences in men and women.
Recent Findings
Recent studies have confirmed the fact that females have differential threshold for biomarkers, with lower troponin and higher NT proBNP levels. Ambiguity continues to exist in the clinical relevance of ST-2, Galectin 3, and other biomarkers.
Novel biomarkers, proteomic biomarkers, and circulating micro RNAs with machine learning are actively being explored. Biomarkers in HFpEF patients with higher female representation are evolving. In recent clinical trials, sex-related difference in biomarkers is not seen despite therapeutic intervention being more effective in females compared to males.
Summary
Sex-related difference exists in the expression of biomarkers in health and in various disease states of heart failure. However, this differentiation has not effectively translated into the clinical practice in terms of diagnostic studies or prognostication. Active exploration to bridge the knowledge gap and novel technologies can shed more light in this area.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Biomarkers of heart failure (HF) are broadly characterized into markers of myocardial injury, myocardial stretch, myocardial inflammation, or fibrosis. Biomarkers facilitate the diagnosis, track the progression of the disease, help define response to therapy, and assist in prognostication (Table 1). Despite the growing evidence of sexual dimorphism in expression of biomarkers in patients with HF, the underlying mechanisms for differential expression remain to be understood. Sex-specific differential expression of HF biomarkers may be attributable to a wide range of factors including anthropometry, adiposity, tissue distribution, sex hormones, genetic, epigenetic, greater incidence of comorbidities, and environmental factors.
HF is a global epidemic and understanding of sexand race related differences is crucial for improving care. Prevalence of heart failure with preserved ejection fraction (HFpEF) is higher in women than men while the risk of heart failure with reduced ejection (HFrEF) is greater in men than women. In Olmsted County study for incident HF, age adjusted all-cause mortality is similar among men and women despite higher risk of cardiovascular death in men compared to women [1]. The mortality and the HF-related hospitalizations were lower in women compared with men regardless of the left ventricular systolic function in the Swedish Heart Failure Registry after adjusting all confounders [2•]. However, women tend to be more symptomatic with worse quality of life and greater exercise limitation despite the observed survival advantage [2•, 3]. Sex-specific regulation of mitochondrial function and energy metabolism has been postulated to contribute to sexual dimorphism of HF [4]. Ischemia is the prominent etiology of HF in men, whereas co-morbid conditions such as HF and diabetes mellitus drive the risk of HF in women.
Estrogen offers cardio-protection and incidence of ischemic heart disease is more commonly seen in men compared to women under the age of 60. In contrast, ischemic heart disease in women confers higher risk of HF in comparison to men. Cardioprotective effects of estrogen are diminished as women attain menopause and sex-based differences in the concentrations of the HF biomarkers may enrich the understanding of the variations in the pathophysiology, mechanistic pathways, and clinical manifestations of HF among men and women. In health, women tend to have elevated levels of biomarkers associated with adiposity, cardiac stretch, and fibrosis, whereas men are more susceptible to have elevated levels of markers associated with cardiac injury and inflammation [5]. This suggests biological sex as a variable in the development of HF and understanding which may provide the underpinnings of sex-specific pathobiology pathways for HF.
Natriuretic Peptides
Natriuretic peptide (NP) are biomarkers of myocardial stretch, with BNP and NTproBNP being the most used for evaluation, prognostication, and management of patients with suspected or proven HF. [6] These serve a counterregulatory function and have diuretic, natriuretic, and vasodilator properties. [7] There levels are elevated in the presence of HF and used for diagnostic evaluation in acute decompensated HF. Furthermore, these elevated NP concentrations have a prognostic role as predictors of mortality and cardiovascular events across different stages of heart failure, and their levels can be used to titrate therapy in chronic HF [8, 9]. NT-proBNP is considered to be more stable than BNP because of its relatively long half-life. Besides heart failure their levels can also be elevated in valvular disorders, infiltrative heart disease, ischemic heart disease, critical illness, kidney disease, and pulmonary embolism. NPs values are affected by age, sex, body mass index ( BMI), etc. In healthy individuals, females exhibit higher levels of NT-proBNP compared to males [10]. Sex hormones are thought to be playing a role in this difference; however, there is no conclusive evidence so far [11]. Change et al. reported that androgens are inversely associated with NT-proBNP levels. The inverse relationship of testosterone and NPs may be attributed to up‐regulation of neprilysin activity by testosterone and explains both the sex-based differences observed in premenopausal women and younger men, and the post-menopausal change in NP levels in women. Menopause in women alters the cardiovascular risk; thereby it is imperative to consider sex differences in biomarkers in the context of life cycle changes in women. The robust sex-based differences seen with NT-proBNP are exclusive to premenopausal women [12]. Studies have shown that postmenopausal women have lower NT-proBNP levels than premenopausal women [12, 13]. It is still not fully understood if loss of NP difference plays a role in narrowing the differences in cardiovascular risk after women attains menopause.
There is mixed data about sex-specific differences in prognostic information provided by NPs. There are studies that did not show any sex-specific differences in levels of NPs in incident HF [14•, 15]. In a multicenter study, a non-significant trend toward lower NT-proBNP levels in men with no mortality difference was noted; however, at very high levels of NT-proBNP, higher mortality trend was noted in women when compared with men with similar levels [15]. A Korean report from registry database suggested NT-proBNP in men to have a better prediction of long-term mortality and HF readmission than in women [16]. Similar sex-specific interaction was observed in another prospective study where NT-proBNP was noted to be a stronger predictor of HF risk in men than in women [17]. A subgroup analysis showed significant impact of sex- and race-based differences in NT-proBNP levels in predicting HF risk at a given level of NT-proBNP, thereby highlighting the importance of considering sex-based differences while prognosticating [18•].
Cardiac Troponins
Cardiac troponins are specific markers of cardiac injury and are primarily markers of myocardial ischemia. However, troponin levels may be elevated in HF and is attributed to multiple mechanisms like ischemia caused by coronary artery or microvascular disease, inflammation, neurohormonal overactivation, myocardial apoptosis, infiltrative processes, etc. Since the advent of high-sensitivity assays, circulatory troponins (cTn) are noted to be elevated in most patients with HF [19]. In healthy individuals, cTn levels tend to be higher in men than women [20]. This has been attributed to differences in left ventricular mass and pathophysiology of myocardial ischemia in men and women, protective antioxidant role of estrogens, and sex-specific prothrombotic tendency. High-sensitivity troponin (hs-TnT) emerged as a strong independent predictor of outcome in chronic heart failure [21], but the data on sex-specific differences in HF is still not concrete with most studies not suggestive of any sex-specific differences [22]. Nevertheless, in the sex-specific analyses by Suthahar et al., levels of cTns were higher in men and remained significantly associated with HF in men [14•]. Another study stratified these sex-differences with phenotypes and noted a stronger predictive association of hsTnT with outcomes in men with HFpEF than in women, but no significant sex-specific difference was noted in HFrEF in same analysis [23].
Soluble Suppression of Tumorgenicity 2 (ST2)
Another novel biomarker that is a marker of ventricular remodeling and fibrosis is ST2, a member of IL1 family. The ST2 gene encodes two primary isoforms: transmembrane form (ST2L) and secreted soluble form (sST2), the latter being mechanically induced in cardiomyocytes in response to myocardial stretch [24]. sST2 acts as a “decoy” receptor for IL-33 and promotes myocardial damage by inhibiting the cardioprotective effects of IL33-ST2L interaction [25]. sST2 levels are elevated in adult males when compared with adult females [26]. Similar trends with higher levels of ST2 in males is noted in HF [27, 28]. sST2 levels were not associated with any specific sex differences [14•]. Lew et al. in their multimarker study did not demonstrate significant sex associations overall but noted significantly lower levels in postmenopausal, but not premenopausal women when compared with age-matched men [12]. Whether the sex-specific differences in sST2 measurements translate into a differential outcome in HF in men and women remains largely unknown. Furthermore, the higher levels of sST2 are not explained by sex-hormones, and the pathophysiology of sex-specific differences in healthy individuals and in patients with HF is not clear [29, 30].
Galectin-3 (Gal-3)
Galectin-3 (Gal-3) is a β-galactosidase binding lectin and plays a significant role in pathogenesis of various diseases including heart failure by promoting fibrosis and myocardial remodeling. Although Gal-3 expression is essential for tissue repair after initial injury, persistent elevation engenders inflammatory fibrosis, tissue heterogeneity, and adverse cardiac remodeling [31]. Nonetheless, it is less variable with alterations in the loading conditions of the heart and has acquired a class II indication as diagnostic and prognostic marker for heart failure patients in ACC/AHA/HFSA guidelines for management of HF [32, 33].
In animal studies, Gal-3 expression was significantly upregulated in failing hearts of homozygous transgenic male rats. [31]. Although preferential expression of Gal-3 is noted in women in several population studies, sex-specific differences in Gal-3 levels are inconsistent in HF and the impact of these sex related differences on management of HF is nebulous [14•]. The differential expression of Gal-3 is attributed to dichotomy in body composition and fat mass distribution with dissimilitude comorbid profiles of the participant sexes rather than sex hormones [34, 35]. While the baseline Gal-3 is higher in women, the incident heart failure risk with elevated Gal-3 is similar in both sexes in the FINRISK cohort [36]. However, Dekelva et al. reported a higher degree of diastolic dysfunction along with upregulation of Gal-3 expression with associated increase in circulation Gal-3 in female subjects in a cohort of myocardial infarction patients treated with percutaneous intervention [37]. There was higher incidence of heart failure and left ventricular hypertrophy in females in this cohort (70% vs 44.6%, p = 0.034 and amp; 35% vs 19.3%, p = 0.02) (37). Sex-specific longitudinal variability of Gal-3 in predicting incident HF or disease progression is yet to be determined [14•, 38]. Also, kinetics of Gal-3 is influenced by kideny function and predictive value of Gal-3 was narrowed in HF after adjusting to kidney function [14•, 35].
Insulin-Like Growth Factor Binding Protein-7 (IGFBP-7)
Sex-specific differences in the maladaptive remodeling pathways of the heart in patients with HF are yet to be understood completely. Cardiac aging affects men and women differently due to lifecycle changes in women such as menopause leading to alterations in mitochondrial biogenesis, sex-specific regulation of mitochondrial function, and regulation of inflammation [39]. Dysregulations of mitochondrial function, autophagy, oxidative stress, and inflammation may trigger accelerated senescence in cardiac myocytes which in turn may lead to myocardial dysfunction and fibrosis. Mitochondrial dysfunction and senescence-associated secreting phenotype (SASP) are among the key characteristics of cardiomyocyte senescence [40]. Preclinical studies demonstrated IGFBP-7 plays a key role in modulating cardiomyocyte senescence and garnered attention as a potential therapeutic target [41]. IGFBP-7 is a marker of senescence and a member of SASP family and is recognized as a novel biomarker both in HFpEF and HFrEF. IGFBP-7 modulates cellular senescence through regulation of insulin-like growth factor-1 (IGF-1). Older age, higher NT-proBNP values, worse NYHA functional class, and lower eGFR were reported to be associated with higher IGFBP-7 levels by Motiwala et al. [42]. Elevations of IGFB-7 demonstrated strong correlation with echocardiographic parameters of diastolic dysfunction both in HFrEF and HFpEF patients [43,44,45]. IGFBP-7 was observed to have incremental additive value in combination with other clinical variables, NT-proBNP and hsTnT in post hoc analysis of DAPA-HF trail [46]. IGFBP-7 was noted in higher concentrations in patients with HFrEF in comparison to HFpEF and was associated with worse clinical outcomes [47]. Although sex-specific data is limited, Hage et al. reported no significant differences in the levels of IGFBP-7 between men and women with both HFpEF and HFrEF. However, a community-based study in Italy noted a strong association with age and women were in smaller portion in highest tertile of IGFBP-7 [48]. Similar findings were noted in the post hoc analysis of the DAPA-HF study. It demonstrated independent predictive value in estimating mortality and hospitalizations for HF.
Special Considerations
Obesity
Obesity is an important factor that can alter the biomarker levels. Higher BMI is associated with lower NP levels. [49]. Albeit sex-specific data regarding obesity-associated reduction of NPs remains scarce. Suthahar et al. noted upon sex-stratification, obese males had slightly lower NT-proBNP levels, but this trend was not seen in obese females [50]. In the general population, higher BMI is not associated with NT-proBNP levels, but males with higher BMI were noted to have higher NT-proBNP levels and females did not exhibit this trend. Low NP levels in abdominal obesity are mostly found in women than men, as visceral fat can increase testosterone levels that lower NP levels. [50, 51]. Thus, we see that lower NT‐proBNP levels in obese healthy individuals are better explained by sex than by obesity warranting sex-specific cut points for NP levels for diagnosis of HF [52]. However, in patients with HF, obesity seems to have a prominent role on NP levels and more data is needed to assess the sex-specific differences of NPs in obese patients with HF [53]. Cardiac troponin is elevated in patients with obesity and increases with increasing BMI and has no known sex-differences in obese individuals with HF [54]. Associations of sST2 that was initially lower in women was completely attenuated after accounting for body composition.
Female Specific Disease States
Takotsubo cardiomyopathy predominantly affects post-menopausal females with only 4–12% male involvement. Females do better overall with good prognosis and less severe disease than males. Patients with takotsubo Cardiomyopathy have lower troponin levels and higher BNP/NT-proBNP levels compared to ischemic cardiomyopathy patients. However, sex-specific difference in the biomarkers like BNP has not been observed. In the Tokyo registry among the various biomarkers, only C-reactive protein was lower in females and had prognostic impact, higher levels predicting higher mortality [55].
In post-partum cardiomyopathy sensitive markers of heart failure BNP/NTproBNP could be non-specific and elevated from various other conditions like pulmonary embolism, pre-eclampsisa, ischemic disease, etc. Various other biomarkers like prolactin, cathepsin D, micro RNA-146, soluble fms-like tyrosine kinase-1, intelukins, and placental growth factor are associated with post-partum cardiomyopathy [56]. Higher baseline NT-proBNP predicts failure to improve cardiac function in the 6 months. In the Nationwide Danish Cohort of women, biomarkers like NT-proBNP, copeptin, soluble fms-like tyrosine kinase 1, and placental growth factors were elevated up to 7 years, and the levels correlated with recovery of LV function [57].
In Adriamycin induced cardiomyopathy, premenopausal females are at lower risk of cardiotoxicity and prepubertal females at higher risk with no clear increased risk noted in post-menopausal women [58]. Monitoring of cardiotoxicity by troponin and NTproBNP levels aids early detection of cardiotoxicity [59]. In a large prospective study of breast cancer patients undergoing Adriamycin and or tratstuzumab therapy, troponin, NT proBNP, Myeloperoxidase, placental growth factor, and growth differentiation factor 15 were elevated. In this cohort, hs‐cTnT levels > 14 ng/l at anthracycline completion were associated with a twofold higher risk of cardiotoxicity. They also noted decline in ejection fraction by about 1% for every doubling of NT-proBNP [60].
Heart Failure with Preserved Ejection Fraction
The biomarkers in HFpEF can be attributed to myocardial stretch and injury as is described in the HFrEF population but can also be related to inflammation, fibrosis, endothelial dysfunction, and other co-morbidities like renal dysfunction, obesity, and anemia [61, 62]. BNP and NT-proBNP levels correlate well with left ventricular end-diastolic pressure, volume, and all diastolic filling patterns. NT-proBNP levels are generally lower in patients with HFpEF (sometimes extending to a normal range) than those with HFrEF. Women have higher NT-proBNP levels than men, although higher levels portend poor prognosis equally in both sexes [63]. Cunningham et al. showed that baseline NT-proBNP strongly predicted total HF hospitalizations and cardiovascular death in the PARAGON-HF trial cohort [64•]. However, caution needs to be exercised in the interpretation of the absolute value of NT-proBNP as co-morbidities have statistically significant interactions with the measured levels. A larger burden of atrial fibrillation, lower BMI and advanced stages of chronic kidney disease are associated with higher NT-proBNP levels [65]. Women generally tend to be older, have higher blood pressure and have more comorbidities like diabetes and kidney disease than men [66]. Initiation of sacubitril/valsartan results in a 19–23% reduction in NT-proBNP [64•, 67] with a marginal increase later in the course of the disease. However, there is no interaction with respect to sex in terms of response to sacubitril/valsartan.
Circulating cardiac troponins are also powerful predictors of adverse outcomes in HFpEF. High-sensitivity troponin I (hsTnI) and T (hsTnT) predict outcomes including all-cause mortality and first HF hospitalization equally in men and women. High-sensitivity troponin I is more strongly associated with adverse events for HFpEF in men than in women with a hazard ratio of 3.33 vs 1.35, respectively [68]. A threshold of hsTnI < 4 ng/l or < 6 ng/l is indicative of low risk of developing clinical HF in women and men, respectively, with a level > 10 ng/l and > 12 ng/l suggestive of higher risk [69]. Tromp et al. demonstrated that after correcting for covariates like age, sex, and blood pressure, comparatively higher levels of high-sensitivity CRP are present in patients with HFpEF while greater. Levels of NT-proBNP are associated with HFrEF [70]. Galectin-3 levels can predict the development of new onset HFpEF in both sexes with a change in its level being a strong predictor when controlling for other biomarkers [71]. Adipocyte-derived serum fatty acid binding protein 4 (FABP4) is an independent predictor of left ventricular mass and reduced longitudinal fractional shortening in obese women and may serve as a risk predictor for diastolic dysfunction and cardiac remodeling in this population [72, 73]. FABP4 is noted to be higher in women than in men and has a direct correlation with the presence of atrial fibrillation, inflammatory markers like IL-6 and TNFα 13.
Recent studies have explored the association of proteomic signatures with the development of HF. One study with a 54% female representation identified 5 protein biomarkers of HFpEF after adjusting for age and sex, namely NT-proBNP, growth differentiation factor-15 [GDF-15], adrenomedullin, un-carboxylated matrix Gla protein, and C-reactive protein, representing pathways of inflammation, cardiac stress, and vascular stiffness [74]. Coronary microvascular dysfunction in HFpEF is related to inflammation mediated chemokine and cytokine signaling pathway in men and P13-kinase and transforming growth factor-beta signaling pathway in women [75]. Endotrophin, a collage VI-derived peptide, is a novel risk marker for all-cause mortality and multimorbidity in elderly women. Plasma levels of endotrophin predict risk of future HF hospitalizations and death, particularly in the HFpEF population, surpassing the MAGGIC score and NT-proBNP [76]. A shared pathogenic background for pre-eclampsia and HFpEF is suggested owning to the presence of common biomarkers including fatty acid-binding protein 4, adrenomedullin, mid-region pro adrenomedullin, and cancer antigen 125 [77]. A promising strategy for the early identification and risk stratification of patients with HFpEF may lie in leveraging proteomics, metabolomics, and circulating microRNA along with machine-learning [78].
Future Directions
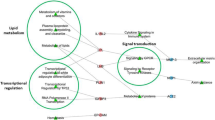
Micro RNAs are small non coding RNAs that play a crucial role in post transcriptional gene regulation. The dysregulation of miRNAs in cardiomyopathy suggests their potential use as biomarkers for diagnosis, prognosis, and monitoring of disease progression. X-chromosome encodes several miRNAs and estrogen influences miRNA regulation and transcription. Sex-based miRNA are potential mediators of the sex-specific cardiovascular pathophysiology in HFpEF [79]. Therapeutic interventions to the miRNA expression in time could allow development of female specific therapies in HF.
Artificial intelligence and machine learning in heart failure is evolving and has varied applications in diagnosis, classification, and prognosis of HF. Woolley et al. used machine learning to cluster HEpEF patients into four subgroups based on biomarkers [80].They used 363 different biomarkers in their study consisting of cardiovascular, inflammatory, and oncology markers. Clusters based on biomarkers had distinct pathophysiologic and clinical outcome. Although this study had significant female representation, they did not comment on sex differences. Kobayashi et al. applied machine learning-derived echocardiographic phenotypes to predict HF incidence in asymptomatic individuals [81]. In one phenotype with significant female presentation inflammatory markers like FABP4, interleukin-6 was elevated and predicted 2 × HF or cardiovascular death. On the contrary phenotype with male predominance remodeling biomarkers like troponin, ST2, and C type natriuretic peptide were elevated and the risk increased by fivefold.
Precision medicine with the use of genomics and single nucleotide polymorphism can help identify genomic phenotypes and potential therapeutic targets and the field is evolving as a promising future biomarker tool (Fig. 1).
Conclusion
While the concept of sex- and gender-based variability in pathophysiology, clinical presentation, and clinical outcomes is well recognized, the guidelines for gender- or sex-specific care have not been well delineated. Contrary to the predominant view of biological sex as a binary genotype of sex chromosomes, gender is a multidimensional construct and is a dynamic variable and hormonal profiles might not fit the construct of biological sex.
Recognition of sex-specific heart failure risk factors, incorporation of sex-specific diagnostic criteria, and tailoring management accordingly may help facilitate the equitable care for women. There is also growing evidence that a combined multi-marker approach may provide a better risk stratification and risk prediction models than any single marker. Further research is warranted to shine light on clinical applications of sex-specific thresholds of heart failure biomarkers with a goal to personalize the treatment strategies and improve clinical outcomes.
Abbreviations
- HF:
-
Heart failure
- HFpEF:
-
Heart failure with preserved ejection fraction
- HFrEF:
-
Heart failure with reduced ejection fraction
- NP:
-
Natriuretic peptide
- cTn:
-
Circulatory troponins
- hs-TnT:
-
High-sensitivity troponin
- ST2:
-
Soluble suppression of tumorgenicity 2
- Gal-3:
-
Galectin-3
- IGFBP-7:
-
Insulin-like growth factor binding protein-7
- SASP:
-
Senescence-associated secreting phenotype
- BMI:
-
Body mass index
- FABP4:
-
Adipocyte-derived serum fatty acid binding protein 4
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
Gerber Y, Weston SA, Redfield MM, Chamberlain AM, Manemann SM, Jiang R, Killian JM, Roger VL. A contemporary appraisal of the heart failure epidemic in Olmsted County, Minnesota, 2000 to 2010. JAMA Intern Med. 2015;175(6):996–1004. https://doi.org/10.1001/jamainternmed.2015.0924.
• Stolfo D, Uijl A, Vedin O, Strömberg A, Faxén UL, Rosano GMC, Sinagra G, Dahlström U, Savarese G. Sex-based differences in heart failure across the ejection fraction spectrum: phenotyping, and prognostic and therapeutic implications. JACC Heart Fail. 2019;7(6):505–15. https://doi.org/10.1016/j.jchf.2019.03.011. (Erratum.In:JACCHeartFail.2020Apr;8(4):345-346).
Lam CS, Carson PE, Anand IS, Rector TS, Kuskowski M, Komajda M, McKelvie RS, McMurray JJ, Zile MR, Massie BM, Kitzman DW. Sex differences in clinical characteristics and outcomes in elderly patients with heart failure and preserved ejection fraction: the Irbesartan in Heart Failure with Preserved Ejection Fraction (I-PRESERVE) trial. Circ Heart Fail. 2012;5(5):571–8. https://doi.org/10.1161/CIRCHEARTFAILURE.112.970061.
Regitz-Zagrosek V. Sex and gender differences in heart failure. Int J Heart Fail. 2020;2(3):157–81. https://doi.org/10.36628/ijhf.2020.0004.
Lau ES, Binek A, Parker SJ, Shah SH, Zanni MV, Van Eyk JE, Ho JE. Sexual dimorphism in cardiovascular biomarkers: clinical and research implications. Circ Res. 2022;130(4):578–92. https://doi.org/10.1161/CIRCRESAHA.121.319916.
Januzzi J. Natriuretic peptides as biomarkers in heart failure. J Investig Med. 2013;61(6):950–5. https://doi.org/10.2310/JIM.0b013e3182946b69.
Bhardwaj A, Januzzi JL. Natriuretic peptide guided management of acutely destabilized heart failure. Rationale and treatment algorithm. Critical Pathways in cardiology. 2009. https://doi.org/10.1097/HPC.0b013e3181c4a0c6.
Zile, et al. Prognostic value of brain natriuretic peptide vs history of heart failure hospitalization in a large real-world population. Clin Cardiol. 2020. https://doi.org/10.1002/clc.23468.
Januzzi JL Jr, Rehman SU, Mohammed AA, Bhardwaj A, Barajas L, Barajas J, Kim HN, Baggish AL, Weiner RB, Chen-Tournoux A, Marshall JE, Moore SA, Carlson WD, Lewis GD, Shin J, Sullivan D, Parks K, Wang TJ, Gregory SA, Uthamalingam S, Semigran MJ. Use of amino-terminal pro-B-type natriuretic peptide to guide outpatient therapy of patients with chronic left ventricular systolic dysfunction. J Am Coll Cardiol. 2011;58(18):1881–9. https://doi.org/10.1016/j.jacc.2011.03.072.
Fradley MG, Larson MG, Cheng S, McCabe E, Coglianese E, Shah RV, Levy D, Vasan RS, Wang TJ. Reference limits for N-terminal-pro-B-type natriuretic peptide in healthy individuals (from the Framingham Heart Study). Am J Cardiol. 2011;108:1341–5. https://doi.org/10.1016/j.amjcard.2011.06.057.
Chang AY, Abdullah SM, Jain T, Stanek HG, Das SR, McGuire DK, Auchus RJ, de Lemos JA. Associations among androgens, estrogens, and natriuretic peptides in young women: observations from the Dallas Heart Study. J Am Coll Cardiol. 2007;49:109–16. https://doi.org/10.1016/j.jacc.2006.10.040.
Lew J, Sanghavi M, Ayers CR, et al. Sex-based differences in cardiometabolic biomarkers. Circulation. 2017;135:544–55. https://doi.org/10.1161/CIRCULATIONAHA.116.023005.
Maffei S, Del Ry S, Prontera C, Clerico A. Increase in circulating levels of cardiac natriuretic peptides after hormone replacement therapy in postmenopausal women. Clin Sci (Lond). 2001;101:447–53.
• Suthahar, et al. Sex-specific associations of cardiovascular risk factors and biomarkers with incident heart failure. J Am Coll Cardiol. 2020;76(12):1455–65. https://doi.org/10.1016/j.jacc.2020.07.044. This study assessed associations of individual biomarkers with incident heart failure and none of the biomarkers displayed a significant interaction with sex for heart failure outcome.
Franke J, Lindmark A, Hochadel M, Zugck C, Koerner E, Keppler J, Ehlermann P, Winkler R, Zahn R, Katus HA, Senges J, Frankenstein L. Gender aspects in clinical presentation and prognostication of chronic heart failure according to NT-proBNP and the heart failure survival score. Clin Res Cardiol. 2015;104:334–41. https://doi.org/10.1007/s00392-014-0786-z.
Kim HL, Kim MA, Choi DJ, Han S, Jeon ES, Cho MC, Kim JJ, Yoo BS, Shin MS, Seong IW, Ahn Y, Kang SM, Kim YJ, Kim HS, Chae SC, Oh BH, Lee MM, Ryu KH. Korean Heart Failure Registry. Gender difference in the prognostic value of N-terminal pro-B type natriuretic peptide in patients with heart failure – a report from the Korean Heart Failure Registry (KorHF). Circ J. 2017;81:1329–36. https://doi.org/10.1253/circj.CJ-16-1345.
Magnussen C, Niiranen et al. Sex-specific epidemiology of heart failure risk and mortality in Europe: results from the BiomarCaRE consortium. JACC: Heart Failure. 2019, 204–213. https://doi.org/10.1016/j.jchf.2018.08.008.
• Myhre PL, et al. Sex and race differences in n-terminal pro–B-type natriuretic peptide concentration and absolute risk of heart failure in the community. JAMA Cardiol. 2022;7(6):623–31. https://doi.org/10.1001/jamacardio.2022.0680. In this study, sex- and race-based differences in NT-proBNP was noted and absolute risk associated with a given value of NT-proBNP varied substantially by sex and race. Consideration of NT-proBNP values in the context of sex and race allows for more uniform prediction of absolute risk across important demographic subgroups.
Januzzi JL, Filippatos G, Nieminen M, Gheorghiade M. Troponin elevation in patients with heart failure: on behalf of the third Universal Definition of Myocardial Infarction Global Task Force: heart failure section. Eur Heart J. 2012;33:2265–71. https://doi.org/10.1093/eurheartj/ehs191.
Romiti GF, Cangemi R, Toriello F, Ruscio E, Sciomer S, Moscucci F, Vincenti M, Crescioli C, Proietti M, Basili S, Raparelli V. Sex-specific cut-offs for high-sensitivity cardiac troponin: is less more? Cardiovasc Ther. 2019;2019:1–12. https://doi.org/10.1155/2019/9546931.
Aimo, et al. Prognostic value of high-sensitivity troponin t in chronic heart failure: an individual patient data meta-analysis. Circulation. 2018;137(3):286–97. https://doi.org/10.1161/CIRCULATIONAHA.117.031560.
Evans JDW, Dobbin SJH, Pettit SJ, di Angelantonio E, Willeit P. High-sensitivity cardiac troponin and new-onset heart failure: a systematic review and meta-analysis of 67,063 patients with 4,165 incident heart failure events. JACC Heart Fail. 2018;6:187–97. https://doi.org/10.1016/j.jchf.2017.11.003.
Gohar A, Chong JPC, Liew OW, den Ruijter H, de Kleijn DPV, Sim D, Yeo DPS, Ong HY, Jaufeerally F, Leong GKT, Ling LH, Lam CSP, Richards AM. The prognostic value of highly sensitive cardiac troponin assays for adverse events in men and women with stable heart failure and a preserved vs reduced ejection fraction. Eur J Heart Fail. 2017;19:1638–47. https://doi.org/10.1002/ejhf.911.
Bhardwaj A, Januzzi JL. ST2: a novel biomarker for heart failure. Expert Rev Mol Diagn. 2010;10(4):459–64. https://doi.org/10.1586/erm.10.25.
Bayés-Genis A, González A, Lupón J. ST2 in heart failure. Circ Heart Fail. 2018;11:e005582. https://doi.org/10.1161/CIRCHEARTFAILURE.118.005582.
Dieplinger B, Januzzi JL, Steinmair M, Gabriel C, Poelz W, Haltmayer M, Mueller T. Analytical and clinical evaluation of a novel high-sensitivity assay for measurement of soluble ST2 in human plasma – the Presage ST2 assay. Clin Chim Acta. 2009;409:33–40. https://doi.org/10.1016/j.cca.2009.08.010.
Meyer S, van der Meer P, van Deursen VM, Jaarsma T, van Veldhuisen DJ, van der Wal MHL, Hillege HL, Voors AA. Neurohormonal and clinical sex differences in heart failure. Eur Heart J. 2013;34:2538–47. https://doi.org/10.1093/eurheartj/eht152.
Anand IS, Rector TS, Kuskowski M, Snider J, Cohn JN. Prognostic value of soluble ST2 in the Valsartan Heart Failure Trial. Circ Heart Fail. 2014;7:418–26. https://doi.org/10.1161/CIRCHEARTFAILURE.113.001036.
Coglianese EE, Larson MG, Vasan RS, Ho JE, Ghorbani A, McCabe EL, et al. Distribution and clinical correlates of the interleukin receptor family member soluble ST2 in the Framingham heart study. Clin Chem. 2012;58:1673–81. https://doi.org/10.1373/clinchem.2012.192153.
Dieplinger B, Egger M, Poelz W, Gabriel C, Haltmayer M, Mueller T. Soluble ST2 is not independently associated with androgen and estrogen status in healthy males and females. Clin Chem Lab Med. 2011;49:1515–8. https://doi.org/10.1515/CCLM.2011.239. Epub 2011 Jun 10 PMID: 21663467.
Sharma UC, Pokharel S, van Brakel TJ, van Berlo JH, Cleutjens JP, Schroen B, André S, Crijns HJ, Gabius HJ, Maessen J, Pinto YM. Galectin-3 marks activated macrophages in failure-prone hypertrophied hearts and contributes to cardiac dysfunction. Circulation. 2004;110(19):3121–8. https://doi.org/10.1161/01.CIR.0000147181.65298.4D.
Yancy CW, Jessup M, Bozkurt B, Butler J, Casey DE Jr, Colvin MM, Drazner MH, Filippatos GS, Fonarow GC, Givertz MM, Hollenberg SM, Lindenfeld J, Masoudi FA, McBride PE, Peterson PN, Stevenson LW, Westlake C. 2017 ACC/AHA/HFSA focused update of the 2013 ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Failure Society of America. Circulation. 2017;136(6):e137–61. https://doi.org/10.1161/CIR.0000000000000509.
Chow SL, Maisel AS, Anand I, Bozkurt B, de Boer RA, Felker GM, Fonarow GC, Greenberg B, Januzzi JL Jr, Kiernan MS, Liu PP, Wang TJ, Yancy CW, Zile MR. Role of biomarkers for the prevention, assessment, and management of heart failure: a scientific statement from the American Heart Association. Circulation. 2017;135(22):e1054–91. https://doi.org/10.1161/CIR.0000000000000490.
Pang J, Nguyen VT, Rhodes DH, Sullivan ME, Braunschweig C, Fantuzzi G. Relationship of galectin-3 with obesity, IL-6, and CRP in women. J Endocrinol Invest. 2016;39(12):1435–43. https://doi.org/10.1007/s40618-016-0515-8.
Lau ES, Liu E, Paniagua SM, Sarma AA, Zampierollo G, López B, Díez J, Wang TJ, Ho JE. Galectin-3 Inhibition with modified citrus pectin in hypertension. JACC Basic Transl Sci. 2021;6(1):12–21. https://doi.org/10.1016/j.jacbts.2020.10.006.
Jagodzinski A, Havulinna AS, Appelbaum S, Zeller T, Jousilahti P, Skytte-Johanssen S, Hughes MF, Blankenberg S, Salomaa V. Predictive value of galectin-3 for incident cardiovascular disease and heart failure in the population-based FINRISK 1997 cohort. Int J Cardiol. 2015;1(192):33–9. https://doi.org/10.1016/j.ijcard.2015.05.040.
M Dekleva and others, Sex related difference in heart failure development in patients after first myocardial infarction; role of galectine-3, European Heart Journal, Volume 43, Issue Supplement_2, October 2022, ehac544.816, https://doi.org/10.1093/eurheartj/ehac544.816.
Motiwala SR, Szymonifka J, Belcher A, Weiner RB, Baggish AL, Sluss P, Gaggin HK, Bhardwaj A, Januzzi JL. Serial measurement of galectin-3 in patients with chronic heart failure: results from the ProBNP Outpatient Tailored Chronic Heart Failure Therapy (PROTECT) study. Eur J Heart Fail. 2013;15(10):1157–63. https://doi.org/10.1093/eurjhf/hft075.
Barcena de Arellano ML, Pozdniakova S, Kühl AA, Baczko I, Ladilov Y, Regitz-Zagrosek V. Sex differences in the aging human heart: gen sirtuins, pro-inflammatory shift and reduced anti-oxidative defense. Aging (Albany NY). 2019;11(7):1918–33. https://doi.org/10.18632/aging.101881.
Bracun V, van Essen B, Voors AA, van Veldhuisen DJ, Dickstein K, Zannad F, Metra M, Anker S, Samani NJ, Ponikowski P, Filippatos G, Cleland JGF, Lang CC, Ng LL, Shi C, de Wit S, Aboumsallem JP, Meijers WC, Klip IT, van der Meer P, de Boer RA. Insulin-like growth factor binding protein 7 (IGFBP7), a link between heart failure and senescence. ESC Heart Fail. 2022;9(6):4167–76. https://doi.org/10.1002/ehf2.14120.
Zhang L, Smyth D, Al-Khalaf M, et al. Insulin-like growth factor-binding protein-7(IGFBP7) links senescence to heart failure. Nat Cardiovasc Res. 2022;1:1195–214. https://doi.org/10.1038/s44161-022-00181-y.
Motiwala SR, Szymonifka J, Belcher A, Weiner RB, Baggish AL, Gaggin HK, Bhardwaj A, Januzzi JL Jr. Measurement of novel biomarkers to predict chronic heart failure outcomes and left ventricular remodeling. J Cardiovasc Transl Res. 2014;7(2):250–61. https://doi.org/10.1007/s12265-013-9522-8.
Hage C, Bjerre M, Frystyk J, Gu HF, Brismar K, Donal E, Daubert JC, Linde C, Lund LH. Comparison of prognostic usefulness of serum insulin-like growth factor-binding protein 7 in patients with heart failure and preserved versus reduced left ventricular ejection fraction. Am J Cardiol. 2018;121(12):1558–66. https://doi.org/10.1016/j.amjcard.2018.02.041.
Gandhi PU, Gaggin HK, Redfield MM, Chen HH, Stevens SR, Anstrom KJ, Semigran MJ, Liu P, Januzzi JL Jr. Insulin-like growth factor-binding protein-7 as a biomarker of diastolic dysfunction and functional capacity in heart failure with preserved ejection fraction: results from the RELAX trial. JACC Heart Fail. 2016;4(11):860–9. https://doi.org/10.1016/j.jchf.2016.08.002.
Gandhi PU, Gaggin HK, Sheftel AD, Belcher AM, Weiner RB, Baggish AL, Motiwala SR, Liu PP, Januzzi JL Jr. Prognostic usefulness of insulin-like growth factor-binding protein 7 in heart failure with reduced ejection fraction: a novel biomarker of myocardial diastolic function? Am J Cardiol. 2014;114(10):1543–9. https://doi.org/10.1016/j.amjcard.2014.08.018.
Adamson C, Welsh P, Docherty KF, de Boer RA, Diez M, Drożdż J, Dukát A, Inzucchi SE, Køber L, Kosiborod MN, Ljungman CEA, Martinez FA, Ponikowski P, Sabatine MS, Morrow DA, Lindholm D, Hammarstedt A, Boulton DW, Greasley PJ, Langkilde AM, Solomon SD, Sattar N, McMurray JJV, Jhund PS. IGFBP-7 and outcomes in heart failure with reduced ejection fraction: findings from DAPA-HF. JACC Heart Fail. 2023;11(3):291–304. https://doi.org/10.1016/j.jchf.2022.09.004.
Tan ES, Chan SP, Choi YC, Chris JP, Troughton R, Poppe K, Lund M, Devlin G, Doughty RN, Richards AM. Regional handling and prognostic performance of circulating insulin-like growth factor binding protein-7 in heart failure. JACC: Heart Failure. 2023;11(6):662–74. https://doi.org/10.1016/j.jchf.2023.01.016.
Meessen JMTA, Cesaroni G, Mureddu GF, Boccanelli A, Wienhues-Thelen UH, Kastner P, Ojeda-Fernandez L, Novelli D, Bazzoni G, Mangiavacchi M, Agabiti N, Masson S, StaszewskyL LR. PREDICTOR Investigators. IGFBP7 and GDF-15, but not P1NP, are associated with cardiac alterations and 10-year outcome in an elderly community-based study. BMC Cardiovasc Disord. 2021;21(1):328. https://doi.org/10.1186/s12872-021-02138-8.
Das SR, et al. Impact of body mass and body composition on circulating levels of natriuretic peptides: results from the Dallas Heart Study. Circulation. 2005;112:2163–8. https://doi.org/10.1161/CIRCULATIONAHA.105.555573.
Suthahar N, Meijers WC, Ho JE, Gansevoort RT, Voors AA, van der Meer P, Bakker SJL, Heymans S, van Empel V, Schroen B, van der Harst P, van Veldhuisen DJ, de Boer RA. Sex-specific associations of obesity and N-terminal pro-B-type natriuretic peptide levels in the general population. Eur J Heart Fail. 2018;20(8):1205–14. https://doi.org/10.1002/ejhf.1209.
Cediel G, Codina P, Spitaleri G, Domingo M, Santiago-Vacas E, Lupón J, et al. Gender-related differences in heart failure biomarkers. Front Cardiovasc Med. 2021;7:617705. https://doi.org/10.3389/fcvm.2020.617705.
Mueller C, McDonald K, de Boer RA, Maisel A, Cleland JGF, Kozhuharov N, Coats AJS, Metra M, Mebazaa A, Ruschitzka F, Lainscak M, Filippatos G, Seferovic PM, Meijers WC, Bayes-Genis A, Mueller T, Richards M, Januzzi JL. Heart Failure Association of the European Society of Cardiology. Heart Failure Association of the European Society of Cardiology practical guidance on the use of natriuretic peptide concentrations. Eur J Heart Fail. 2021;21:715–31. https://doi.org/10.1002/ejhf.1494.
Suthahar N, Meems, et al. Sex-related differences in contemporary biomarkers for heart failure: a review. Eur J Heart Fail. 2020;22(5):775–88. https://doi.org/10.1002/ejhf.1771.
Ndumele CE, Coresh J, Lazo M, Hoogeveen RC, Blumenthal RS, Folsom AR, Selvin E, Ballantyne CM, Nambi V. Obesity, subclinical myocardial injury, and incident heart failure. JACC Heart Fail. 2014;2:600–7. https://doi.org/10.1016/j.jchf.2014.05.017.
Arao K, Yoshikawa T, Isogai T, Imori Y, Mochizuki H, Sakata K, Takaoka Y, Yamaguchi T, Nagao K, Yamamoto T, Takayama M. A study of takotsubo syndrome over 9 years at the Tokyo Cardiovascular Care Unit Network Registry. J Cardiol. 2023;82(2):93–9. https://doi.org/10.1016/j.jjcc.2022.12.011.
Azibani F, Sliwa K. Peripartum cardiomyopathy: an update. Curr Heart Fail Rep. 2018;15(5):297–306. https://doi.org/10.1007/s11897-018-0404-x.
Ersbøll AS, Goetze JP, Johansen M, Hauge MG, Sliwa K, Vejlstrup N, Gustafsson F, Damm P. Biomarkers and their relation to cardiac function late after peripartum cardiomyopathy. J Card Fail. 2021;27(2):168–75. https://doi.org/10.1016/j.cardfail.2021.01.002.
Wilcox NS, Rotz SJ, Mullen M, Song EJ, Ky Hamilton B, Moslehi J, Armenian SH, Wu JC, Rhee JW, Ky B. Sex-specific cardiovascular risks of cancer and its therapies. Circ Res. 2022;130(4):632–51. https://doi.org/10.1161/CIRCRESAHA.121.319901.
Stone JR, Kanneganti R, Abbasi M, Akhtari M. Monitoring for chemotherapy-related cardiotoxicity in the form of left ventricular systolic dysfunction: a review of current recommendations. JCO Oncol Pract. 2021;17(5):228–36. https://doi.org/10.1200/OP.20.00924.
Demissei BG, Hubbard RA, Zhang L, Smith AM, Sheline K, McDonald C, Narayan V, Domchek SM, DeMichele A, Shah P, Clark AS, Fox K, Matro J, Bradbury AR, Knollman H, Getz KD, Armenian SH, Januzzi JL, Tang WHW, Liu P, Ky B. Changes in cardiovascular biomarkers with breast cancer therapy and associations with cardiac dysfunction. J Am Heart Assoc. 2020;9(2):e014708. https://doi.org/10.1161/JAHA.119.014708.
Bayes-Genis A, Cediel G, Domingo M, et al. Biomarkers in heart failure with preserved ejection fraction. Card Fail Rev. 2022;8:e20. https://doi.org/10.15420/cfr.2021.37.
Lubien E, DeMaria A, Krishnaswamy P, et al. Utility of B-natriuretic peptide in detecting diastolic dysfunction: comparison with Doppler velocity recordings. Circulation Circulation. 2002;105:595–601. https://doi.org/10.1161/hc0502.103010.
Faxén UL, Lund LH, Orsini N, et al. N-terminal pro-B-type natriuretic peptide in chronic heart failure: the impact of sex across the ejection fraction spectrum. Int J Cardiol. 2019;287:66–72. https://doi.org/10.1016/j.ijcard.2019.04.023.
• Cunningham JW, Vaduganathan M, Claggett BL, et al. Effects of sacubitril/valsartan on N-terminal pro-B-type natriuretic peptide in heart failure with preserved ejection fraction. JACC Heart Failure. 2020;8:372–81. https://doi.org/10.1016/j.jchf.2020.03.002. Baseline NT-proBNP predicted HF events and sacubitril/valsartan reduced NT-proBNP consistently in men and women and in patients with lower or higher EF.
Januzzi JL, Myhre PL. The challenges of NT-proBNP Testing in HFpEF: shooting arrows in the wind. JACC: Heart Failure. 2020;8:382–5. https://doi.org/10.1016/j.jchf.2020.03.003.
Hopper I, Kotecha D, Chin KL, et al. Comorbidities in heart failure: are there gender differences? Curr Heart Fail Rep. 2016;13:1–12. https://doi.org/10.1007/s11897-016-0280-1.
Solomon SD, Zile M, Pieske B, et al. The angiotensin receptor neprilysin inhibitor LCZ696 in heart failure with preserved ejection fraction: a phase 2 double-blind randomised controlled trial. The Lancet. 2012;380:1387–95. https://doi.org/10.1016/S0140-6736(12)61227-6.
Gohar A, Chong JPC, Liew OW, et al. (2017) The prognostic value of highly sensitive cardiac troponin assays for adverse events in men and women with stable heart failure and a preserved vs. reduced ejection fraction. Eur J Heart Fail. 2017;19:1638–47. https://doi.org/10.1002/ejhf.911.
Morfino P, Aimo A, Castiglione V, et al. Biomarkers of HFpEF: natriuretic peptides, high-sensitivity troponins and beyond. J Cardiovasc Dev Dis 2022;9. https://doi.org/10.3390/jcdd9080256.
Tromp J, Khan MAF, Klip IT, et al. Biomarker profiles in heart failure patients with preserved and reduced ejection fraction. J Am Heart Assoc. 2017;6:e003989. https://doi.org/10.1161/JAHA.116.003989.
Watson CJ, Gallagher J, Wilkinson M, et al. Biomarker profiling for risk of future heart failure (HFpEF) development. J Transl Med. 2021;19:61. https://doi.org/10.1186/s12967-021-02735-3.
Engeli S, Utz W, Haufe S, et al. Fatty acid binding protein 4 predicts left ventricular mass and longitudinal function in overweight and obese women. Heart. 2013;99:944–8. https://doi.org/10.1136/heartjnl-2013-303735.
Harada T, Sunaga H, Sorimachi H, et al. Pathophysiological role of fatty acid-binding protein 4 in Asian patients with heart failure and preserved ejection fraction. ESC Heart Fail. 2020;7:4256–66. https://doi.org/10.1002/ehf2.13071.
Takvorian KS, Wang D, Courchesne P, et al. The association of protein biomarkers with incident heart failure with preserved and reduced ejection fraction. Circulation: Heart Failure. 2023;16:e009446. https://doi.org/10.1161/CIRCHEARTFAILURE.121.009446.
Chandramouli C, Ting TW, Tromp J, et al. Sex differences in proteomic correlates of coronary microvascular dysfunction among patients with heart failure and preserved ejection fraction. Eur J Heart Failure. 2022;24:681–4. https://doi.org/10.1002/ejhf.2435.
Chirinos Julio A, Zhao L, Reese-Petersen Alexander L, et al. Endotrophin, a collagen VI formation–derived peptide, in heart failure. NEJM Evidence 2022;1:EVIDoa2200091. https://doi.org/10.1056/EVIDoa2200091.
Alma LJ, Bokslag A, Maas AHEM, et al. Shared biomarkers between female diastolic heart failure and pre-eclampsia: a systematic review and meta-analysis. ESC Heart Failure. 2017;4:88–98. https://doi.org/10.1002/ehf2.12129.
Chirinos JA, Orlenko A, Zhao L, et al. Multiple plasma biomarkers for risk stratification in patients with heart failure and preserved ejection fraction. J Am Coll Cardiol. 2020;75:1281–95. https://doi.org/10.1016/j.jacc.2019.12.069.
Florijn BW, Bijkerk R, van der Veer EP, van Zonneveld AJ. Gender and cardiovascular disease: are sex-biased microRNA networks a driving force behind heart failure with preserved ejection fraction in women? Cardiovasc Res. 2018;114(2):210–25. https://doi.org/10.1093/cvr/cvx223.
Woolley RJ, Ceelen D, Ouwerkerk W, Tromp J, Figarska SM, Anker SD, Dickstein K, Filippatos G, Zannad F, Metra M, Ng L, Samani N, van Veldhuisen DJ, Lang C, Lam CS, Voors AA. Machine learning based on biomarker profiles identifies distinct subgroups of heart failure with preserved ejection fraction. Eur J Heart Fail. 2021;23(6):983–91. https://doi.org/10.1002/ejhf.2144.
Kobayashi M, Huttin O, Magnusson M, et al. Machine learning-derived echocardiographic phenotypes predict heart failure incidence in asymptomatic individuals. J Am Coll Cardiol Img. 2022;15(2):193–208. https://doi.org/10.1016/j.jcmg.2021.07.004.
Author information
Authors and Affiliations
Contributions
All authors have contributed significantly to the manuscript preparation. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Conflict of Interest
Roopa Rao, Anju Bhardwaj, Mrudula Munnagala, Sonu Abraham, Sanjana Adig, Eman Hamad declare no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rao, R.A., Bhardwaj, A., Munagala, M. et al. Sex Differences in Circulating Biomarkers of Heart Failure. Curr Heart Fail Rep 21, 11–21 (2024). https://doi.org/10.1007/s11897-023-00634-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11897-023-00634-w