Abstract
Colorectal cancer (CRC) is the third most common cancer diagnosis and cause of cancer-related death in the USA. It accounts for more than one million cases diagnosed each year worldwide. In the past 10 years, new drugs have been approved, but the survival times are still modest. Alternative therapeutic strategies are clearly needed. A large number of tumor antigens and epitopes recognized by cytotoxic T lymphocytes (CTLs) have been identified in CRC. Cancer vaccines, designed to activate immune effectors (T cells and antibodies), to prevent recurrence, or to treat advanced cancers, have demonstrated clinical benefit in melanoma, prostate cancer, and lymphoma. Immunotherapy that targets tumor-associated antigens (TAAs) on cancer cells or tumor stroma is under investigation. This review discusses CRC immune responsiveness, current status of CRC vaccines, challenges, and future directions. New therapeutic modalities hold a promise to improve the patient’s clinical outcome.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Colorectal cancer (CRC) is a common and lethal disease. It is estimated that 136,830 new CRC patients are diagnosed annually in the USA [1]. Although mortality from this disease has been progressively declining since 1990 at a rate of about 3 % per year, it still remains to be the third most common cause of cancer death [2]. Modest results have been reported with the advent of the new-targeted therapies approved for the treatment of CRC [3, 4]. Novel targeted strategies are of crucial need. Of these strategies, immunotherapy has gained lots of attention specifically after the advances proven in melanoma and renal cell cancer. CRC cells express antigenic peptides. In addition, tumor-associated lymphocytes are thought to play a role in the development of immune activity against cancer cells. Immune surveillance has been observed early in the neoplastic process when immune responses lead to production of tumor-specific antibodies and activation of T cells in subjects with premalignant colorectal adenomas [5]. Many candidate tumor-associated antigens (TAAs) have been identified as potential targets for CRC vaccines [6–9]. This review will discuss the basic principles of CRC immunity, highlighting the current and future immune targets and peptide CRC vaccines.
Immunology and Immune Evasion in CRC
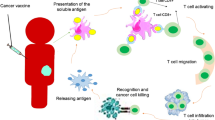
The interaction between cancer cells and its microenvironment promotes proliferation and metastasis. CRC grows in immune-competent hosts by evasion of recognition and elimination by the host’s immune system. CRCs are often infiltrated by lymphocytes that are themselves activated, leading to the assumption that the immune response evoked by tumors is not sufficient to prevent tumor growth. Cancer microenvironment is complex and capable of inactivating and suppressing the immune response against itself through multiple mechanisms, such as human leukocyte antigen (HLA) loss, explaining failure of immune surveillance. Understanding these mechanisms may help in identifying targets that promote the immune system to recognize cancer and to potentiate the effect of cytotoxicity against it.
A key step is the generation of adaptive immunity. Adaptive immunity is the generation of diverse T and B cell repertoires by antigen-specific responses. This involves the antigen presentation by antigen-presenting cells (APCs) to T helper (Th) cells. The activation of the Th cell through production of cytokines promotes cellular (cytotoxic T lymphocytes) and humoral (B lymphocyte-producing antibodies) immune activation systems. These signals can either be stimulatory or inhibitory. These regulatory cells suppress the immunity to maintain tolerance to self and limit the immune damage to normal tissues. T cells are regulated through different signals. The major histocompatibility complex (MHC) is one of the main stimulators for antigen presentation to be recognized by the T cells. There are two types of MHC. MHC class I is expressed on all cell types and serves as an antigen-presenting molecule for CD8+ T cells, and MHC class II is present on APCs and tumor cells. The MHC class II is recognized by CD4+ T cells (T helper 1 response) through a host APC or when the peptide is endogenously produced in a tumor cell expressing MHC class II, leading to direct lysis of the tumor cells and activation of the cytotoxic T cell and macrophage immune response. Peptide fragments should complex with the MHC molecules to have a T cell response to specific epitopes of antigens through polymorphisms of the MHC molecules. This specific antigen/epitope-MHC complexing is known as MHC restriction [10, 11]. Other co-stimulatory signals and adhesion molecules play a role in the T cell regulatory response, side by side with the T cell co-inhibitory responses. The CD28/cytotoxic T lymphocyte antigen 4 (CTLA4)/B7-1/B7-2 receptor/ligand grouping, member of the immunoglobulin superfamily, plays a role as a checkpoint to suppress immune activity against the self. Regulatory T cells (Tregs) play a central role in recognizing self-cells from tumor cells through the CD4/CD25/FOXP3+ [12••, 13]. The infiltration of these cells in CRC appears to carry a better prognosis than other tumors with the same infiltration [14, 15]. The immune escape mechanisms are many; these involve T lymphocytes, dendritic, macrophage, natural killer (NK), and myeloid-derived suppressor cells. Cancer cells are characterized by molecular instability and clonal heterogeneity, allowing mutations that help in developing resistance to immune recognition, leading to proliferation. Resistant tumor cells escape from the immune system checkpoint, leading to a phenomenon called immune editing which is the switch of the antigenic peptides detected by the host immunity to peptides that are not detected by the immune system, thus, evasion [16, 17]. This is a complex process and involves multiple other pathways that interact with the stimulation and inhibition of the immune system. Figure 1 summarizes the pathway by which a peptide induces an immune response.
T cells recognize antigens presented to the T cell receptor (TCR) as antigen peptides within the major histocompatibility complex of the antigen-presenting cell (APC) and tumor cells. Tumor cell usually stimulate a MHC class I-CD8+ T cell response. Both the APC and tumor cell engage a co-stimulatory effect of the T cell through the CTLA4 and CD28 binding to the APC ligands (CD80 also called B7-H or PD1 and CD86). Another dependent stimulatory effect is the intercellular adhesion molecule 1 (ICAM1) and leukocyte function-associated antigen (LFA3) on the APC that bind to the LFA1 and CD2, respectively. The formation of PD1/PD-L1 or B7-1 receptor/PD-L1 ligand complex transmits an inhibitory signal which reduces the proliferation of the CD8+ T cells. Engagement of PD-L1 with its receptor PD1 on T cells delivers a signal that inhibits TCR-mediated activation of IL-2 production and T cell proliferation. Regulatory T cells (Tregs) serve as checkpoints and suppress expansion of T cells directed against self-antigens
Peptide and MHC Complexes
Each MHC allelic variant binds to a specific subset of the peptides that share conserved amino acid residues, defining the peptide-binding motif. The MHC-peptide complex is recognized by the T cell receptor (TCR), a step that is needed to initiate an immunologic response. Another immunogenicity pathway is TAAs which are either expressed as differentiation antigens (found on normal tissue of origin) or as tumor-restricted antigens. TAAs are unique to cancer cells that induce cytotoxic T cell response. Usually TAAs are complexed to MHC class I, initiating CD8+ T cell activation. Some TAA epitopes induce CD4+ T cell activation. All TAAs are targets to vaccine therapies but are still underdeveloped in terms of their clinical use. This could be due to the fact that the majority of TAA epitopes are derived from normal proteins, leading to immune tolerance, preventing to mount an immune response. A search for cancer-specific TAAs from histologically different tumors that have peptide epitopes recognized by T cells in the context of less frequent HLA alleles remains crucial.
Cancer Stimulating the Immune System (Th1 to Th2 Shift)
CRC harbors immunogenic proteins. Some TAA- and MHC-restricted epitopes derived from tumor antigens can be targeted by T cell-mediated adaptive immune response [18, 19]. The immune stimulation leads to production and proliferation of antigen-specific memory T cells in the cancer tissue [20], inhibition of primary tumor growth, and attenuation of the metastatic potential [21], resulting in better outcomes [22–24]. Cancer cells inhibit the antigenic immune response system recognition by shifting from Th1 to Th2, downregulating HLA class I antigen processing, and inducing defective dendritic cell function. This leads to T cell loss of signaling molecules, escaping death receptors, impairing NK activity, Tregs, and complement decay acceleration [25]. Through an unknown mechanism, CRC cells activate Th2, suppressing Th1 response. IL-10 overexpression in CRC [26] is thought to be the underlying mechanism explaining the inhibition of Th1 cytokine production as well as antigen presentation to Th1 cells, creating a Th2 to Th1 imbalance. IL-10 is a negative prognostic factor for treatment response as well as survival [8].
Dendritic Cells
Dendritic cells (DCs) play a pivotal role in antigen recognition and presentation of antigens to other immune cells. CRC patients have impaired DC activation within the tumor. CRC patients also have low numbers of peripheral DC and altered T cell stimulatory capability [27]. Immune-suppressive factors secreted by cancer cells like vascular endothelial growth factor (VEGF), IL-10, transforming growth factor (TGF), and platelet growth factor (PGF) disable DC differentiation, maturation, migration, and function, hence interfering with the whole adaptive immune cascade [28, 29].
Tregs
Regulatory T cells (CD4+ CD25+ Treg cells), known as suppressor cells, are T cells that suppress the immune system activation. Tregs maintain immune homeostasis and tolerance to host antigens. They are widely present in CRC tissue [30]. Tregs play a role in the suppression of TAA immunity through IL-10 and TGF-β [31, 32], leading to inhibition of antigen-specific CD4+ and CD8+ cytokine production. Moreover, Tregs inhibit NK and DC [33]. Treg depletion has been proven to enhance antigen-specific immune responses to cancer vaccines in CRC [34]. Targeting Tregs in CRC has been an area of interest to vaccine development.
Peptides and Peptide Vaccines
Antigen Targets in Colon Cancer
Immunotherapy is an active therapeutic approach designed to trigger immune responses to tumor-specific antigens. Immunotherapy strategies include the use of peptides derived from TAA, whole tumor cells, in vitro-generated DCs, or viral vector-based cancer vaccines [35••].
A peptide vaccine is based on the identification and synthesis of epitopes that induce TAA-specific antitumor immune responses. TAAs have been the most popular targets for vaccine therapy in CRC. CRC express TAAs such as carcinoembryonic antigen (CEA), mucin 1, epidermal growth factor receptor (EGFR), squamous cell carcinoma antigen recognized by T cell 3 (SART3), beta-human chorionic gonadotropin (β-hCG), Wilms’ tumor antigen 1 (WT1), survivin-2B, MAGE3, p53, or KRAS. Multiple small clinical trials have been performed, analyzing the safety and efficacy of different peptide vaccines. Frequently reported side effects are ulceration at the injection site, fever, fatigue, nausea, and anorexia. Serious adverse reactions are very rare, which make vaccine immunotherapy in CRC an important therapy to study.
RAS
A group at the National Institute of Health (NIH) evaluated in a phase II trial the use of a RAS peptide vaccine in the adjuvant setting for resected pancreatic cancer and CRC patients [36]. Patients were vaccinated with 13-mer mutant RAS peptide, corresponding to the tumor’s detected RAS mutations. The RAS target developed specific immune responses to the relevant mutant RAS peptide. This is confirmed by measuring IFN-gamma messenger ribonucleic acid (mRNA) expression using quantitative real-time PCR. Five of 11 patients showed a positive immune response. The seven CRC patients had a mean disease-free survival of 27.2 months and an overall survival (OS) of 41.5 months. No serious acute or delayed systemic side effects were seen. A phase III study of Imprime PGG combined with cetuximab in recurrent or progressive wild-type KRAS CRC subjects (PRIMUS) is actively recruiting (NCT01309126).
Microsatellite Instability
Microsatellite instability (MSI) constitutes 15 % of all CRC patients [37]. A Scandinavian study described T cell reactivity against several Th cell epitopes, representing a common frame shift mutation in TGF-βRII in patients with MSI tumors [38]. A specific peptide sequence was recognized by T cells from two of three CRC patients and from three of three patients with hereditary non-polyposis colon cancer (HNPCC). The study identified frameshift peptides derived from TGF-βRII and BAX that carry several HLA class II-binding specific motifs, and used these to stimulate T cells from patients and normal donors. These epitopes are attractive targets for cancer vaccines, including a prophylactic vaccine for individuals carrying a genetic predisposition for HNPCC.
Ring Finger Protein 43 and Translocase of the Outer Mitochondrial Membrane 34 in HLA-A2402
Ring finger protein 43 (RNF43) and the 34-kDa translocase of the outer mitochondrial membrane 34 (TOMM34) have been identified as cancer-specific peptides in CRC using genome-wide expression profile analysis by cDNA microarray technique. VEGF receptors 1 and 2 are essential targets to tumor angiogenesis, and peptides derived from these receptors significantly induce the effective tumor-specific cytotoxic T lymphocyte (CTL) response in vitro and in vivo. A Japanese group developed a novel peptide vaccine derived from RNF43 and TOMM34 administered to 21 metastatic CRC patients in combination with oral tegafur-uracil (a prodrug of 5-FU) and leucovorin for 4 weeks, followed by 1 week of rest with chemotherapy [39]. In this phase I clinical trial, all patients were HLA-A2402-positive. The CTL responses against RNF43 and TOMM34 in peripheral lymphocytes were assessed. The vaccinations were well tolerated, and CTL responses were induced against both antigens in 8 patients and against one antigen in 12 patients; one patient had no CTL response. The rate of stable disease was 83 %. The group with CTL responses against both antigens had better survival, followed by the group showing CTL responses against one antigen (p = 0.0079). The patient with no CTL response had the worst survival. In an effort to improve clinical efficacy, the same group formulated a seven-peptide vaccine which they gave to advanced CRC patients [40]. The peptides were derived from proteins RNF43, TOMM34, forkhead box M1 (FOXM1), maternal embryonic leucine zipper kinase (MELK), Holliday junction-recognizing protein (HJURP), VEGF receptor (VEGFR)-1 and VEGFR-2. Thirty patients were enrolled; all were HLA-A2402-positive. Partial response was achieved in 3 patients, stable disease in 15, and progression in 12. Patients that exhibited positive CTL responses to all seven peptides had better survival.
This multiple peptide approach was also used in the development of a vaccine consisting of five novel HLA-A2402-restricted peptides: three peptides derived from oncoantigens, RNF43, TOMM34, and insulin-like growth factor II mRNA-binding protein 3 (KOC1), and the remaining two from angiogenesis factors, VEGFR-1 and VEGFR-2 [41]. Eighteen HLA-A2402-positive metastatic CRC patients were enrolled. Enzyme-linked immunospot (ELISPOT) assay (highly sensitive and accurate detection of rare antigen-specific T cells) for monitoring cellular immune responses was performed before and after vaccinations. Dose-dependent induction of peptide-specific CTL was observed. Complete response is achieved in one patient and stable disease in six patients for 4 to 7 months. The OS was 13.5 months. Patients in whom induction of CTL specific to three or more peptides was detected had an OS of 27.8 months. Patients with poor immune responses had a significantly lower OS of 3.7 months (p = 0.032).
EP2101
EP2101, a vaccine targeting 10 different peptide antigens, is designed to activate the immune system against tumor cells in order to delay or prevent the recurrence of cancer. An ongoing study is recruiting patients to test the safety of the vaccine in CRC patients. The trial includes biomarker correlative study intended to measure the level of immune-stimulating capability of EP2101 in CRC patients.
Ephrin Type-A Receptor 2
Ephrin type-A receptor 2 (EphA2) is a member of a large tyrosine kinase receptor family. Eph receptors play an important role in oncogenesis [42] and angiogenesis [43]. It is overexpressed in CRC [44]. Its utility as a tumor antigen has been evaluated in a liver metastasis mouse model transfected with EphA2-positive CRC. A study utilizing EphA2-derived peptide in combination with amphiphilic nanoparticles in a murine model resulted in activity against CRC liver metastases following immunization [42].
SART3
The tumor rejection antigen SART3 possesses two antigenic epitopes (SART3109−118 and SART3315−323) capable of inducing HLA-A24-restricted and tumor-specific cytotoxic lymphocytes. SART3 is expressed in more than 70 % of CRC patients [43]. SART3 vaccine was administered to 12 advanced CRC patients in a phase 1 trial [44]. There were significant levels of increased cellular immune responses to HLA-A24+ CRC cells. Of the 11 patients who were eligible for evaluation, nine had stable disease and two had progressive disease at the first clinical evaluation at 5 weeks. SART3 increased cellular immunity.
p53
The tumor-associated self-antigen p53 is commonly overexpressed in CRC and can serve as a target for immunotherapy. In a phase I/II trial, 10 CRC patients were vaccinated twice with a set of 10 overlapping p53 synthetic long peptides (SLPs) [45]. p53-specific T cell responses were induced in nine of 10 CRC patients as measured by IFN-gamma ELISPOT. In six of nine patients, p53-specific T cell reactivity persisted for at least 6 months. However, multiparameter flow cytometry revealed that only a minor population of the p53-specific CD4+ T cells was optimally polarized. The same group of investigators combined the p53-SLP vaccine together with interferon-α [46]. Eleven CRC patients were enrolled after being treated with the p53-SLP vaccine alone. Toxicity was limited to swelling at the vaccination site. Compared to the previous trial, the addition of interferon-α significantly improved the frequency of p53-specific T cells. Moreover, in this trial, p53-specific T cells were detectable in blood samples of all patients in a direct ex vivo multiparameter flow cytometric assay, opposed to only two of 10 patients vaccinated with p53-SLP alone. Patients in this trial displayed a broader p53-specific immunoglobulin-G response, indicating an overall better p53-specific Th response.
Survivin
Survivin is present during fetal development, but undetectable in terminally differentiated normal adult tissues [47]. It is a member of the inhibitor of apoptosis protein. It is identified as a CRC peptide. It is expressed on both cancer and endothelial cells of the tumor vasculature. Spontaneous CTL responses against different survivin epitopes in cancer patients underline the relevance of survivin-directed immunological trials [48]. The HLA-A24-restricted antigenic peptide, survivin-2B80-88, recognized by CD8+ CTL has shown efficacy in CRC patients [49]. The investigators developed two protocols for therapy: survivin-2B80-88 plus incomplete Freund’s adjuvant (IFA) and survivin-2B80-88 plus IFA and interferon-α. The effect of survivin-2B80-88 plus IFA was not significantly different from that with survivin-2B80-88 alone, but treatment with the vaccination protocol of survivin-2B80-88 plus IFA and interferon-α resulted in clinical improvement and enhanced immunological responses. Tetramer analysis demonstrated that survivin-2B80-88 peptide-specific CTLs were increased by at least twofold after vaccination with this protocol in four of eight patients. A peptide vaccine is being studied with HLA-A1-, HLA-A2-, and HLA-B35-restricted survivin epitopes in an ongoing trial utilizing Montanide ISA-51 for patients with stage IV melanoma and advanced pancreatic, colon, and cervical carcinoma and actively recruiting patients to study the safety of this vaccine.
MUC1 Glycoprotein
Ever since the first characterization of MUC1 as a tumor antigen and successful cloning of the muc1 gene, MUC1 has been a promising candidate for vaccine-based interventions against human adenocarcinomas [50]. MUC1 glycoprotein neoplastic cells express high levels of the hypoglycosylated form of MUC1 and lack luminal expression. Low-level luminal or apical expression of the glycosylated MUC1 is found on normal colonic epithelial cells [51] and accounts for the mucus that is normally present on the lining of the colon. MUC1 is highly expressed in a modified form on adenomatous polyps and CRC and is thought to be a part of the process of progression from adenomas to cancer. This abnormal expression induces humoral and cellular immune responses [52]. Abnormal expression of MUC1 is also found on premalignant colorectal adenomas where it promotes malignant transformation by interacting with β-catenin, RAS, and other tumor-promoting signaling pathways [50]. Many different MUC1 vaccines such as MUC1 peptides with adjuvants, MUC1-loaded dendritic cells, or MUC1 DNA expressed in viral vectors have been tested in phase I/II trials in cancer patients who had failed standard therapy [9]. These therapeutic vaccines were well tolerated, but only mildly immunogenic. In contrast, many of these same vaccines tested in the prophylactic setting in animal models [51] were highly immunogenic and resulted in immune protection against spontaneous or xenografted MUC1-positive tumors. MUC1 100-mer peptide is a vaccine studied in metastatic castrate-resistant prostate cancer and advanced colonic adenomas at risk for developing colon cancer. The MUC1-poly-ICLC vaccine is being tested in persons with a history of advanced adenomatous polyps to prevent recurrence of polyps. The vaccine goal is to help the immune system identify changes in MUC1 that accompany cancer progression and to eliminate the abnormal cells that make abnormal MUC1 [6].
Anti-Programmed Cell Death Protein 1 Antibody (CT-011)
Programmed cell death protein 1 (PD1) is an inhibitory receptor that belongs to CD28-B7 family. PD1 binds to two ligands PD-L1 and PD-L2 to downregulate T cell immune responses. PD1 is expressed on T, B, and NK cells. The overexpression of PD1 ligand (PD-L1) in tumors is associated with immune suppression and poor prognosis. A promising avenue of clinical research in CRC is the use of immune checkpoint inhibitors. These target molecules serve as brakes that stop the immune response. Blocking these inhibitory molecules leads to enhancement of preexisting anticancer immune responses. A phase I trial using tremelimumab, an anti-CTLA4, and MEDI4736, an anti-PD-L1, used in combination for patients with advanced solid tumors is currently recruiting patients (NCT01975831).
CEA
DCs are the professional antigen-presenting cells of the immune system. As such, they are currently used in clinical vaccination protocols in cancer patients. Ability of mature DCs pulsed with CEA peptide or electroporated with CEA mRNA to induce CEA-specific T cell responses in patients with resectable liver metastases from CRC [7, 53, 54]. A CRC mouse model was used to evaluate CEA as a target for peptide vaccine and in combination with an antibody targeted to CEA epitope. Transgenic mice carrying tumors positive for CEA and HLA-A2 were vaccinated with DCs having the epitope of CEA. Complete response was reported in 25 % of mice. No recurrence was observed for 90 days. This was associated with production of INF-γ by CD4+, thought to improve cancer MHC class I expression [55].
GA733
Human CRC antigen GA733 is a transmembrane glycoprotein and functions as a cell-cell adhesion molecule (CO17-1A/KS1-4/KSA/EpCAM). Its extracellular domain has been a target in CRC vaccination trials, where recombinant protein produced tumor-specific humoral and cellular immune responses [56].
Human Chorionic Gonadotropin
hCG is produced by CRC. hCG is expressed on CRC cells in up to 52 % of patients, and elevated levels of circulating hCG have been observed in up to 41 % [57–61]. A COOH terminal peptide of β-hCG vaccine conjugated to diphtheria toxoid targeting hCG by enhancing an antibody specific production was tested in 77 patients with metastatic CRC [62]. Immunizations were well tolerated, and anti-hCG antibodies were detected in 56 of the 77 patients. OS was 34 weeks. Patients who developed anti-hCG antibody levels higher than the median value exhibited better OS (45 weeks) as compared to OS of patients (24 weeks) who had levels lower than the median (p = 0.0002).
Growth Factors and Vaccines
TAA peptides have been administered to cancer patients in conjunction with granulocyte macrophage colony-stimulating factor (GM-CSF), which induces the recruitment of DCs at the site of vaccination and promotes their differentiation, a process that favors TAA processing and presentation [63].
Other TAAs
Other clinical trials that are recruiting now are targeting different TAAs. A phase II adjuvant chemotherapy ± FANG in CRC with liver metastases (FANG-CLM) (NCT01505166), a phase I study of guanylyl cyclase C vaccine in stage I/II colon cancer (NCT01972737) and a phase I study of vaccine therapy with or without sirolimus in treating patients with NY-ESO-1 expressing solid tumors (NCT01522820) are actively recruiting patients to enroll in these trials. Table 1 summarizes all the ongoing trials involving peptide vaccines in CRC.
Monitoring Response
All responses are based on Response Evaluation Criteria In Solid Tumors (RECIST) criteria and clinical outcomes. It has been difficult to establish outcome correlation to cancer vaccines through evaluating immunologic responses. The ability to monitor the induction of vaccine- and tumor-specific immune responses and to correlate the presence and magnitude of vaccine-induced immune responses to clinical outcomes is essential to vaccine development. Immune monitoring is important to check efficacy of the vaccine in generating antigen cascade responses, to develop potencies to compare different vaccines, to evaluate vaccine-induced immune responses, and to possibly select specific populations, by checking biomarkers that might benefit from specific targeted vaccines [64]. Assays fail to define surrogate markers that could be used as predictors of clinical response and thus serve to advance vaccine development. Many trials have evaluated cancer immunotherapy through assays designed to detect cytokines, proliferation of cancer cells, antibodies, and TAAs, but to date, these assays have failed to have any clinical outcome correlation [65–68]. Further studies are needed to evaluate the difficulties establishing correlations between T cell function and clinical efficacy. Standardizing the efforts used to monitor responses is essential.
Conclusion
Peptide vaccines are gaining much attention because of the reported safety data from all the trials. The main side effects are local swelling at the injection sites and mild fatigue. Failure mechanisms include the quality of T cell response and its function (TAA tolerance), heterogeneity of the tumor cells, escape mechanisms of the cancer cells because of their genetic instability leading to the loss or reduced expression of the peptides targeted by the T cells, access to these vaccines, and side effects. The main drawbacks to the peptide vaccination strategy suggested by Koido et al. [69•] are limitations due to the patient’s HLA type, ineffectiveness of CD8+ CTLs due to the downregulation of certain antigens and MHC class I molecules, impaired DC function in patients with advanced cancer, and tumor microenvironment, where immune suppressive cells such as Tregs exist. Nagorsen and Thiel performed a systematic review of all CRC vaccines (not only peptide vaccines) [70]. The responses were poor with only five patients out of 527 achieving either a partial or complete response.
Advantages of peptide vaccines lay in the inexpensive production, ease of administration, specific TAA targets, and improvement of patients’ immune response. Future directions in peptide vaccines need to focus on improving immunogenicity, designing specific TAA targeted epitopes, and sensitizing the immune system to detect these epitopes through peptide-HLA epitope complexes. In addition, identifying molecular pathways of epitope expression and their ability to modulate these processes is critical in designing future vaccines. Targeting multiple epitopes with one vaccine and utilizing combination treatments with chemotherapy agents and growth factors may represent other venues to improve outcomes. With the advent of genomic sequencing, TAA and other cancer-specific proteins can be identified and targeted.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Siegel R et al. Cancer statistics, 2014. CA Cancer J Clin. 2014;64(1):9–29.
Jemal A et al. Annual report to the nation on the status of cancer, 1975–2009, featuring the burden and trends in human papillomavirus (HPV)-associated cancers and HPV vaccination coverage levels. J Natl Cancer Inst. 2013;105(3):175–201.
Andre T, Chibaudel B. [Aflibercept (Zaltrap(®)) approved in metastatic colorectal cancer]. Bull Cancer. 2013;100(10):1023–5.
Grothey A et al. Regorafenib monotherapy for previously treated metastatic colorectal cancer (CORRECT): an international, multicentre, randomised, placebo-controlled, phase 3 trial. Lancet. 2013;381(9863):303–12.
Sallusto F, Lanzavecchia A. Heterogeneity of CD4+ memory T cells: functional modules for tailored immunity. Eur J Immunol. 2009;39(8):2076–82.
Kimura T et al. MUC1 vaccine for individuals with advanced adenoma of the colon: a cancer immunoprevention feasibility study. Cancer Prev Res (Phila). 2013;6(1):18–26.
Lesterhuis WJ et al. Vaccination of colorectal cancer patients with CEA-loaded dendritic cells: antigen-specific T cell responses in DTH skin tests. Ann Oncol. 2006;17(6):974–80.
Asadullah K, Sterry W, Volk HD. Interleukin-10 therapy—review of a new approach. Pharmacol Rev. 2003;55(2):241–69.
Schimanski CC et al. LICC: L-BLP25 in patients with colorectal carcinoma after curative resection of hepatic metastases: a randomized, placebo-controlled, multicenter, multinational, double-blinded phase II trial. BMC Cancer. 2012;12:144.
Thibodeau J, Bourgeois-Daigneault MC, Lapointe R. Targeting the MHC class II antigen presentation pathway in cancer immunotherapy. Oncoimmunology. 2012;1(6):908–16.
Dickgreber N et al. Targeting antigen to MHC class II molecules promotes efficient cross-presentation and enhances immunotherapy. J Immunol. 2009;182(3):1260–9.
Protti MP, Monte LD, Lullo GD. Tumor antigen-specific CD4+ T cells in cancer immunity: from antigen identification to tumor prognosis and development of therapeutic strategies. Tissue Antigens. 2014;83(4):237–46. The authors reviewed the studies that first identified the tumor-specific CD4+ T cells in cancer, the techniques used to identify the tumor antigens recognized, the role of the different CD4+ T cell subsets in tumor immunity and in cancer prognosis, and the development of therapeutic strategies aimed at activating efficient antitumor CD4+ T cell effectors.
Bailey SR et al. Th17 cells in cancer: the ultimate identity crisis. Front Immunol. 2014;5:276.
Loddenkemper C et al. Regulatory (FOXP3+) T cells as target for immune therapy of cervical intraepithelial neoplasia and cervical cancer. Cancer Sci. 2009;100(6):1112–7.
Sobhani I, Le Gouvello S. Critical role for CD8+ FoxP3+ regulatory T cells in colon cancer immune response in humans. Gut. 2009;58(6):743–4.
Peggs KS, Quezada SA, Allison JP. Cell intrinsic mechanisms of T-cell inhibition and application to cancer therapy. Immunol Rev. 2008;224:141–65.
Abele R, Tampe R. The TAP translocation machinery in adaptive immunity and viral escape mechanisms. Essays Biochem. 2011;50(1):249–64.
Shawler DL et al. Antigenic and immunologic characterization of an allogeneic colon carcinoma vaccine. Clin Exp Immunol. 2002;129(1):99–106.
Chan CC et al. Multiple serological biomarkers for colorectal cancer detection. Int J Cancer. 2010;126(7):1683–90.
Sallusto F, Geginat J, Lanzavecchia A. Central memory and effector memory T cell subsets: function, generation, and maintenance. Annu Rev Immunol. 2004;22:745–63.
Pages F et al. Effector memory T cells, early metastasis, and survival in colorectal cancer. N Engl J Med. 2005;353(25):2654–66.
Baier PK et al. Analysis of the T cell receptor variability of tumor-infiltrating lymphocytes in colorectal carcinomas. Tumour Biol. 1998;19(3):205–12.
Diederichsen AC et al. Prognostic value of the CD4+/CD8+ ratio of tumour infiltrating lymphocytes in colorectal cancer and HLA-DR expression on tumour cells. Cancer Immunol Immunother. 2003;52(7):423–8.
Galon J et al. Type, density, and location of immune cells within human colorectal tumors predict clinical outcome. Science. 2006;313(5795):1960–4.
Evans C et al. The correlation between colorectal cancer rates of proliferation and apoptosis and systemic cytokine levels; plus their influence upon survival. Br J Cancer. 2006;94(10):1412–9.
Oc’Hara RJ et al. Advanced colorectal cancer is associated with impaired interleukin 12 and enhanced interleukin 10 production. Clin Cancer Res. 1998;4(8):1943–8.
Della Porta M et al. Dendritic cells and vascular endothelial growth factor in colorectal cancer: correlations with clinicobiological findings. Oncology. 2005;68(2–3):276–84.
Malmberg KJ. Effective immunotherapy against cancer: a question of overcoming immune suppression and immune escape? Cancer Immunol Immunother. 2004;53(10):879–92.
Yang L, Carbone DP. Tumor-host immune interactions and dendritic cell dysfunction. Adv Cancer Res. 2004;92:13–27.
Misra N et al. Cutting edge: human CD4+ CD25+ T cells restrain the maturation and antigen-presenting function of dendritic cells. J Immunol. 2004;172(8):4676–80.
Curiel TJ et al. Specific recruitment of regulatory T cells in ovarian carcinoma fosters immune privilege and predicts reduced survival. Nat Med. 2004;10(9):942–9.
Maloy KJ, Powrie F. Regulatory T cells in the control of immune pathology. Nat Immunol. 2001;2(9):816–22.
Wolf AM et al. Increase of regulatory T cells in the peripheral blood of cancer patients. Clin Cancer Res. 2003;9(2):606–12.
Morse MA et al. Depletion of human regulatory T cells specifically enhances antigen-specific immune responses to cancer vaccines. Blood. 2008;112(3):610–8.
Buhrman JD, Slansky JE. Improving T cell responses to modified peptides in tumor vaccines. Immunol Res. 2013;55(1–3):34–47. The authors identified epitopes based on stimulation of the naturally responding T cell repertoire and proved that this dramatically improved the efficacy of peptide vaccination.
Toubaji A et al. Pilot study of mutant ras peptide-based vaccine as an adjuvant treatment in pancreatic and colorectal cancers. Cancer Immunol Immunother. 2008;57(9):1413–20.
Vilar E, Gruber SB. Microsatellite instability in colorectal cancer—the stable evidence. Nat Rev Clin Oncol. 2010;7(3):153–62.
Saeterdal I et al. Frameshift-mutation-derived peptides as tumor-specific antigens in inherited and spontaneous colorectal cancer. Proc Natl Acad Sci U S A. 2001;98(23):13255–60.
Okuno K et al. Phase I clinical trial of a novel peptide vaccine in combination with UFT/LV for metastatic colorectal cancer. Exp Ther Med. 2011;2(1):73–9.
Okuno K et al. Clinical trial of a 7-peptide cocktail vaccine with oral chemotherapy for patients with metastatic colorectal cancer. Anticancer Res. 2014;34(6):3045–52.
Hazama S et al. A phase I study of combination vaccine treatment of five therapeutic epitope-peptides for metastatic colorectal cancer; safety, immunological response, and clinical outcome. J Transl Med. 2014;12:63.
Yamaguchi S et al. EphA2-derived peptide vaccine with amphiphilic poly(gamma-glutamic acid) nanoparticles elicits an anti-tumor effect against mouse liver tumor. Cancer Immunol Immunother. 2010;59(5):759–67.
Sasatomi T et al. Expression of tumor rejection antigens in colorectal carcinomas. Cancer. 2002;94(6):1636–41.
Miyagi Y et al. Induction of cellular immune responses to tumor cells and peptides in colorectal cancer patients by vaccination with SART3 peptides. Clin Cancer Res. 2001;7(12):3950–62.
Speetjens FM et al. Induction of p53-specific immunity by a p53 synthetic long peptide vaccine in patients treated for metastatic colorectal cancer. Clin Cancer Res. 2009;15(3):1086–95.
Zeestraten EC et al. Addition of interferon-alpha to the p53-SLP(R) vaccine results in increased production of interferon-gamma in vaccinated colorectal cancer patients: a phase I/II clinical trial. Int J Cancer. 2013;132(7):1581–91.
Tanaka T et al. Potential survival benefit of anti-apoptosis protein: survivin-derived peptide vaccine with and without interferon alpha therapy for patients with advanced or recurrent urothelial cancer—results from phase I clinical trials. Clin Dev Immunol. 2013;2013:262967.
Tsuruma T et al. Phase I clinical study of anti-apoptosis protein, survivin-derived peptide vaccine therapy for patients with advanced or recurrent colorectal cancer. J Transl Med. 2004;2(1):19.
Kameshima H et al. Immunogenic enhancement and clinical effect by type-I interferon of anti-apoptotic protein, survivin-derived peptide vaccine, in advanced colorectal cancer patients. Cancer Sci. 2011;102(6):1181–7.
Byrd JC, Bresalier RS. Mucins and mucin binding proteins in colorectal cancer. Cancer Metastasis Rev. 2004;23(1–2):77–99.
Ohyabu N et al. An essential epitope of anti-MUC1 monoclonal antibody KL-6 revealed by focused glycopeptide library. J Am Chem Soc. 2009;131(47):17102–9.
Wang YQ et al. Correlation between auto-antibodies to survivin and MUC1 variable number tandem repeats in colorectal cancer. Asian Pac J Cancer Prev. 2012;13(11):5557–62.
Figdor CG et al. Dendritic cell immunotherapy: mapping the way. Nat Med. 2004;10(5):475–80.
Lesterhuis WJ et al. Immunogenicity of dendritic cells pulsed with CEA peptide or transfected with CEA mRNA for vaccination of colorectal cancer patients. Anticancer Res. 2010;30(12):5091–7.
Saha A et al. Therapy of established tumors in a novel murine model transgenic for human carcinoembryonic antigen and HLA-A2 with a combination of anti-idiotype vaccine and CTL peptides of carcinoembryonic antigen. Cancer Res. 2007;67(6):2881–92.
Maruyama H et al. Cancer vaccines: single-epitope anti-idiotype vaccine versus multiple-epitope antigen vaccine. Cancer Immunol Immunother. 2000;49(3):123–32.
Lundin M et al. Tissue expression of human chorionic gonadotropin beta predicts outcome in colorectal cancer: a comparison with serum expression. Int J Cancer. 2001;95(1):18–22.
Braunstein GD et al. Ectopic production of human chorionic gonadotropin in Ugandan patients with hepatocellular carcinoma. Cancer. 1973;32(1):223–6.
Goldstein DP, Kosasa TS, Skarim AT. The clinical application of a specific radioimmunoassay for human chorionic gondotropin in trophoblastic and nontrophoblastic tumors. Surg Gynecol Obstet. 1974;138(5):747–51.
Gailani S et al. Human chorionic gonadotrophins (hCG) in non-trophoblastic neoplasms. Assessment of abnormalities of hCG and CEA in bronchogenic and digestive neoplasms. Cancer. 1976;38(4):1684–6.
Marcillac I et al. Free human chorionic gonadotropin beta subunit in gonadal and nongonadal neoplasms. Cancer Res. 1992;52(14):3901–7.
Moulton HM et al. Active specific immunotherapy with a beta-human chorionic gonadotropin peptide vaccine in patients with metastatic colorectal cancer: antibody response is associated with improved survival. Clin Cancer Res. 2002;8(7):2044–51.
Parmiani G et al. Cancer immunotherapy with peptide-based vaccines: what have we achieved? Where are we going? J Natl Cancer Inst. 2002;94(11):805–18.
Butterfield LH et al. Recommendations from the iSBTc-SITC/FDA/NCI workshop on immunotherapy biomarkers. Clin Cancer Res. 2011;17(10):3064–76.
Le Poole IC, Gerberi MA, Kast WM. Emerging strategies in tumor vaccines. Curr Opin Oncol. 2002;14(6):641–8.
Nagorsen D et al. Immunological monitoring of cancer vaccine therapy. Expert Opin Biol Ther. 2004;4(10):1677–84.
Shafer-Weaver K et al. Application of the granzyme B ELISPOT assay for monitoring cancer vaccine trials. J Immunother. 2006;29(3):328–35.
Whiteside TL et al. Immunologic monitoring of cellular immune responses in cancer vaccine therapy. J Biomed Biotechnol. 2011;2011:370374.
Koido S et al. Immunotherapy for colorectal cancer. World J Gastroenterol. 2013;19(46):8531–42. The authors review the effect of immunotherapy for inducing cytotoxic T lymphocytes and the major immunotherapeutic approaches for CRC that are currently in clinical trials, including peptide vaccines, dendritic cell-based cancer vaccines, whole tumor cell vaccines, viral vector-based cancer vaccines, adoptive cell transfer therapy, antibody-based cancer immunotherapy, and cytokine therapy.
Nagorsen D, Thiel E. Clinical and immunologic responses to active specific cancer vaccines in human colorectal cancer. Clin Cancer Res. 2006;12(10):3064–9.
Compliance with Ethics Guidelines
Conflict of Interest
Walid Shaib declares that he has no conflict of interest. Daniel Goldstein declares that he has no conflict of interest. Bassel F. El-Rayes has received research support from Synta Pharmaceuticals, Novartis, Oncolytics Biotech, AVEO Pharmaceuticals, Bristol-Myers Squibb, Pfizer, Kyowa Hakko Kirin, and Genentech.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shaib, W., Goldstein, D. & El-Rayes, B.F. Peptide Vaccines for Treatment of Colon Cancer: Have We Made Progress?. Curr Colorectal Cancer Rep 10, 477–486 (2014). https://doi.org/10.1007/s11888-014-0250-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11888-014-0250-5