Abstract
Purpose of Review
This review aims to summarize our current understanding and management strategies of acute cardiorenal syndrome (CRS).
Recent Findings
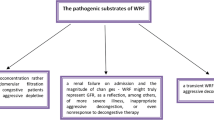
The definition of acute CRS remains debated, in part due to the lack of reliable insights into salt and water handling of the kidneys beyond impairment in glomerular filtration. Protocolized use of loop diuretics to ensure adequate delivery to their target of action, as well as segmental tubular blockade with adjunctive use of thiazide diuretics, acetazolamide, amiloride, or sodium-glucose transporter 2 (SGLT2) inhibitors, may result in more effective natriuresis in patients with acute CRS who exhibit diuretic resistance. Other strategies, such as modulating renal sodium avidity with the use of hypertonic saline, reduction of intra-abdominal pressure, or device-based salt and volume removal, are promising and warrant further investigation.
Summary
Acute CRS remains a significant contributor of morbidity and mortality for the acute heart failure population. New strategies have challenged current dogmas in our understanding of its pathophysiology, which may lead to potential new treatment approaches.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The concept of “cardiorenal” disease was first described in 1914 by Dr. Alfred Stengel [1]. He proposed the term “cardiorenal” that referred to “cases of combined cardiovascular and renal disease without such manifest predominance of either as to justify a prompt determination of the one element as primary and important and the other as secondary and unimportant.” Dr. Stengel presented an empiric framework categorizing these patients into three groups that would each require different treatment strategies: (1) those with primary heart failure (HF) leading to secondary renal failure, (2) atherosclerotic vascular disease leading to both secondary HF and renal failure, and (3) primary renal failure leading to secondary HF [1]. By proposing this framework, Stengel’s goal was to help clinicians better identify and treat the primary insult in any given patient presenting with “cardiorenal” disease. It is therefore important to appreciate that different and complex pathophysiologic processes leading to the disruption of the intricate interdependence of the heart and the kidneys have long been recognized for over a century, prior to any effective treatment strategies or diagnostic tests.
Today, the term “cardiorenal syndrome” (CRS) refers to any disease state where HF and renal dysfunction happen to be present simultaneously, which became too broad and non-specific. A widely circulated classification scheme proposed by the Acute Dialysis Quality Initiative (ADQI) workgroup in 2008 categorized CRS from types 1 to 5 and was created in an effort to highlight the bidirectional nature of heart-kidney interactions [2]. However, such a classification scheme has yet to better define treatment strategies for CRS in clinical practice as it has provided limited incremental insights into what pathophysiologic mechanisms clinicians may target [3•]. This lack of progress over the century since Stengel’s treatise, in part, has been due to the following: (1) the obscure and somewhat misleading terminologies used over the years in describing cardiorenal dysregulation across patients with CRS; (2) the lack of insights into underlying causes and contributing factors of renal insufficiency or diuretic ineffectiveness when treating patients with CRS; and (3) the lack of innovation and novel treatment approaches beyond loop diuretics despite testing of a wide range of proposals and randomized clinical trials. Without more precise and quantifiable definitions of CRS, its true prevalence and incidence can be difficult to ascertain.
It is very important to distinguish acute CRS as a defined subset of acute HF syndromes. In clinical practice, acute CRS in patients with heart failure is commonly described as “an extreme form of cardio-renal dysregulation in which therapy to relieve congestive symptoms of HF is limited by further decline in renal function” [4]. Previously known as “resistant edema” in the era of mercurial diuretics, this definition is far narrower than that of the ADQI classification scheme (restricting only to their “Type 1” category), and thus may not encompass the full spectrum of heart-kidney crosstalk. Yet, it captures the challenges commonly seen when escalating diuretic use for volume removal in the presence of clinical congestion (so-called diuretic resistance, which refers to the relatively insufficient responses to diuretic therapy) leads to progressive compromise in renal function patients with heart failure. The demonstration that impaired cardiac output can directly diminish renal perfusion pressures since the 1990s had dominated the “cardiocentric view” of acute CRS [5]. With new drug or device therapies available to tackle this morbid condition, we are now challenging the prevailing dogmas surrounding acute CRS. In this review, we will outline the recent advances in our contemporary approaches that include new insights into (1) pathophysiologic contributions of acute CRS; (2) adequacy and effectiveness of diuretic therapy; and (3) hemodynamic, neurohormonal, and metabolic interdependence.
Dogma #1: Acute Cardiorenal Syndrome Is Primarily Driven by Acute Kidney Injury Due to Cardiac Insufficiency
The primary driver in a large majority of cases of acute CRS may not be acute impairment of cardiac output or intrinsic kidney injury, but rather a complex set of perturbations in systemic and regional hemodynamic and/or neurohormonal responses that can be overcome by better appreciation of their pathophysiologic triggers. It is imperative that clinicians develop a better understanding of the trajectories of renal function that are relevant in the care of patients with heart failure, and appreciate the strengths and limitations of various bedside metrics that have been summarized in a recent consensus statement [6•].
Worsening Renal Function as a Flawed Metric in the Setting of Acute CRS
Originally thought to be primarily caused by arterial underfilling leading to impaired renal perfusion due to acute cardiac insufficiency [7], we now recognize that acute CRS often develops in the presence of adequate cardiac output (so-called warm and wet profile) [8, 9]. Nevertheless, acute CRS often presents with “worsening renal failure” (WRF) that manifests as a rise in serum creatinine of ≥ 0.3–0.5 mg/dL and/or a drop in estimated glomerular filtration rate (eGFR)—both have been associated with poorer clinical outcomes especially in the absence of adequate diuresis or decongestion. It is important to understand, however, that this metric was derived from a large observational cohort as the optimal “cut-off” between sensitivity and specificity in predicting in-hospital mortality [10•]. Whether or not a rise in serum creatinine (or a drop in eGFR) truly reflects a decline in renal function has been debated over the past decade, since different underlying causes of WRF determine different clinical courses. Theoretically in the setting of acute HF, a rise in serum creatinine may reflect a drop in glomerular filtration (as a result of decreased renal perfusion) or azotemia (accumulation of uremic solutes and nitrogen-rich compounds), both contributing to disease progression and adverse outcomes. However, patients who have advanced heart failure and have lower muscle mass may also have lower circulating creatinine (as a result with falsely lower serum creatinine or creatinine clearance), even though more specific markers of glomerular filtration have provided only marginally incremental prognostic values in both acute and chronic HF settings [11, 12].
Several recent observations have challenged the notion that WRF is clinically relevant in the absence of its clinical context. In the setting of adequate diuresis, a rise in creatinine was associated with better rather than worse long-term outcomes, since this may be driven by hemoconcentration as a result of successful diuresis (thereby not necessarily reflecting true damage to the renal tubules) [13•, 14,15,16]. Therefore, adequacy of diuresis in the congested state may serve as a more clinically relevant metric of therapeutic success. Indeed, this concept has been demonstrated in a post hoc analysis of the DOSE-AHF trial, where brisk diuresis during acute HF admissions with or without WRF was associated with better rather than worse long-term clinical outcomes, whereas “improvement” in renal function (represented by drop in serum creatinine) paradoxically tracked with worse clinical outcomes [17]. Furthermore, WRF as a result of initiation of drugs with known benefits (such as neurohormonal antagonists) have also been associated with better rather than worse outcomes. In contrast, reducing WRF has not been found to be an effective therapeutic target as drugs targeting WRF have not improved clinical outcomes [18].
Lack of True Acute Kidney Injury in Diuretic-Responsive Patients with Acute CRS
To go a step further, not all WRF reflects true “worsening”, meaning intrinsic kidney injury and clinical deterioration. We should all remember that contemporary clinical criteria for AKI in the nephrology literature include both rising serum creatinine and diminishing urine output [19]. In the acute HF setting, the majority of patients are diuretic responsive—their urine outputs are often robust in response to aggressive diuretic therapy despite fluctuating creatinine and blood urea nitrogen (BUN). Therefore, the propensity to develop a rise in creatinine following aggressive diuresis may simply reflect a lower circulatory reserve to maintain adequate renal perfusion to support aggressive diuresis (so-called effective arterial blood volume, effective circulating volume, or plasma refill rate in the nephrology literature). In fact, the kidneys have evolved to counter intravascular volume depletion by fully activating mechanisms to safely preserve salt and water for survival.
Early proponents of WRF as a therapeutic target have touted that a rise in circulating novel AKI biomarkers may indicate intrinsic renal damage that may be linked to poorer prognosis in acute CRS [20]. Certainly, true AKI may occur in the setting of acute CRS, but it is not common despite a third or more patients developing WRF following aggressive diuresis. In fact, acute CRS is likely driven by processes beyond “cardiogenic” alterations in renal hemodynamics, and often presents without clear evidence of intrinsic damage such as proteinuria or urinary sediments. Plasma and urinary biomarker levels often reflect different compartments of the body, as the large majority of biomarker filtered through the glomeruli are often reabsorbed at the proximal renal tubules, where direct injury at the renal tubular level can produce such biomarkers in the urine that may indicate AKI [21].
The lack of injury in the majority of WRF in acute HF was first reported by Dupont and colleagues when urinary neutrophil gelatinase-associated lipocalin (NGAL) was not elevated in those with acute HF treated with aggressive diuretic therapy despite experiencing WRF [22]. There is also clear evidence that blood and urine AKI biomarkers behave differently and reflect different aspects of renal dysfunction [21]. Several contemporary studies have now confirmed that there is no clear evidence that a general rise in AKI biomarkers was associated with worsening clinical outcomes upon aggressive diuresis, even in the setting of WRF or underlying chronic kidney disease [23, 24].
Volume Redistribution and Compartmentalization May Contribute to Acute CRS
One of the biggest paradigm shifts in understanding the pathophysiology of acute CRS has been the recognition that acute HF may in part be exacerbated by volume redistribution rather than solely the consequences of excessive and overwhelming fluid retention [25•]. Patients presenting with acute HF syndromes may have different degrees and distributions of “congestion,” and not everyone responds in the same manner to loop diuretics. We should not assume that the kidneys are failing when WRF develops, while in fact they are appropriately responding to intravascular volume depletion and effectively protecting the body from excessive dehydration. Utilizing blood volume analyses, Miller and colleagues have challenged the notion that all acute HF patients have excessive volume overload and need aggressive diuresis, demonstrating that there are a substantial number of patients seen in the emergency department with minimal increase in blood volume, which may indicate that these patients have a vascular component of increased filling pressure that is independent of volume expansion [26]. To bypass such natural counter-regulatory measures with force, mechanical volume removal can further exacerbate dyshomeostasis. This has been best illustrated in CARRESS-AHF, where mechanical removal of volume by ultrafiltration (UF) was directly compared with goal-directed stepped pharmacologic therapy in patients with acute HF presenting with WRF, and was associated with more rather than less WRF and hyponatremia [27•, 28, 29]. Hence, the subjective perception of central congestion (even in the presence of peripheral edema) in these patients may have led to unnecessary diuresis that can lead to intravascular volume depletion. Fortunately, such cases are easily corrected by stepping down diuretic therapy. However, this also emphasizes the need to have an accurate assessment of volume status, and confirmation of clinical suspicion by blood testing or invasive hemodynamic assessment if in doubt or with unexpected treatment responses.
The causal relationship of increased renal venous pressure leading to decreased GFR has previously been described in animal models, which clearly illustrated that impaired “forward” cardiac output is only one of several factors leading to acute CRS [30,31,32,33]. Several groups have revived this concept over the past decade by demonstrating the association of venous congestion with WRF and impairment of diuretic responses in patients with acute HF [9, 34]. This has also been observed in the chronic HF setting [34]. Recently, Nijst and colleagues provided direct evidence in a series of mechanistic human studies, confirming the direct contributions of volume expansion on blunting natriuresis and renal venous flow in the kidneys [35, 36]. In addition, the potential contribution of gut edema and ascites leading to raised intra-abdominal pressure (IAP) has been theorized as a contributing factor to CRS [37]. In animal models, increased IAP can precipitate renal injury in acute HF [38]. Indeed, a subset of patients admitted with advanced decompensated HF may exhibit abnormally elevated IAP as measured by bladder catheterization [39•]. While the majority of patients improve their IAP following effective diuresis, those that did not achieve adequate decongestion demonstrated persistently elevated IAP. The mechanism by which elevated IAP leads to worsening renal function is likely mediated by its direct impact on renal venous congestion from visceral edema due to increased blood volume across the splanchnic circulation—a phenomenon that has been recognized since the 1950s [40]. There is currently only anecdotal evidence to support the use of interventions directed towards elevated IAP (i.e., paracentesis) in the setting of acute HF [41], and thus, both retrospective and prospective studies are still needed to confirm benefit. In support of this approach, direct splanchnic nerve block to transiently relieve visceral congestion has demonstrated short-term improvement in hemodynamics and provided symptomatic relief in patients with acute and decompensated HF [42, 43]. However, understanding the long-term benefits and risks of such therapeutic approaches in acute CRS warrants further investigation.
Dogma #2: Decongestive Strategies with Loop Diuretics Exacerbate Acute Cardiorenal Syndrome
For a long time, overzealous use of loop diuretics has been faulted as being one of the contributors to acute CRS. For example, higher doses of loop diuretics has been associated with poorer long-term outcomes in a post hoc analysis from the ESCAPE trial [44]. As hypotension serves as a primary driver of WRF in advanced decompensated HF above and beyond central hemodynamics [45], intravascular volume depletion from aggressive use of loop diuretics has been considered a primary driver of acute CRS. However, in the CARRESS-AHF study in patients with persistent congestion and WRF, achieving net negative urine output regardless of treatment strategy was not associated with progressive AKI in the majority of patients with HF [29, 46]. This suggests that effective decongestion plays an important role in overcoming rather than precipitating perceived renal insufficiency that contributes to acute CRS, and that the large majority of patients are indeed “diuretic responsive” rather than “diuretic resistant.” Furthermore, the need to tailor diuretic use in this vulnerable population requires more in-depth understanding of the role these drugs play in acute CRS [47].
Adequate Amounts of Loop Diuretics Should Be Administered in Persistent Congestion
Since their discovery in the 1950s, loop diuretics have become the mainstay of acute heart failure treatment largely because of their effective natriuretic and chlorouric properties [48]. In the setting of acute HF with fluid overload, patients are often admitted to receive intravenous loop diuretics boluses and may occasionally convert to continuous infusions or be augmented with thiazides and even vasoactive drugs [49]. The primary purpose is to deliver adequate inhibition of sodium reabsorption at the loop of Henle. This allows effective blockade of urinary sodium reabsorption, such that the downstream sodium reabsorptive mechanisms are overwhelmed, thereby achieving net negative sodium balance without excessive kaliuresis [50]. Fortunately, loop diuretics have a broad therapeutic window yet have a relatively short half-life to maintain their diuretic efficacies. One must therefore ensure that adequate loop diuretics are being delivered to maintain the patient above the threshold levels required in the upsloping portion of the loop diuretic dose-response curve (Fig. 1). Strategies to maintain adequate drug exposure in the therapeutic window (that can be affected by absorption, protein-binding in transport, secretion via proximal convoluted tubules, and renal clearance) include (1) giving higher doses; (2) giving more frequent dosing; (3) changing from oral to intravenous route of administration, or switching to more bioavailable loop diuretics; and (4) changing from bolus to continuous infusions [50].
Intravenous loop diuretic pharmacokinetics and dose-response curve in heart failure. In heart failure, the diuretic threshold is shifted upwards (from solid to shaded circle), thereby reducing the effective range of therapy (i.e., area under the curve) as well as increasing the amount of intravenous diuretics required to produce a natriuretic response. The development of acute CRS shifts the dose-response curve further to the right as a result of the renal secretory defect, requiring even higher diuretic doses for the same response. Meanwhile, the maximal natriuretic response is also diminished upon reaching the diuretic ceiling (open circle)
One common cause of “acute CRS” is simply the inadequate use of loop diuretics or adjunctive drugs to produce adequate natriuresis. The DOSE-AHF study was designed to address the research question as to whether continuous and higher-dose intravenous loop diuretics are superior to bolus/standard dosing in a heterogenous patient population with substantial home oral loop diuretic use (≥ 80 mg furosemide equivalent) [51•]. The results have prompted much debate, as contrary to popular belief all groups showed similar clinical outcomes with the high-dose group achieving statistically significant better symptom relief without increased incidence of WRF [51•]. However, a post hoc analysis of the same study has perhaps shed some important mechanistic insight into the findings [52]. Notably, the design of the DOSE-AHF study compared 2.5-times the home dose vs the home dose, but in a patient population that was randomized with a wide range of home doses. As such, there is a large heterogeneity of doses delivered, some likely adequate and others potentially inadequate. In fact, almost a third of the standard dose group received total intravenous loop diuretic doses higher than those in the high-dose group [52]. When adjusted for the amount of volume of diuresis achieved, those in the high-dose group portend better outcomes than those in the low-dose group. These findings suggest that the different groups may be unequally distributed in their diuretic dosing after all as the randomization did not overcome their disparate diuretic efficiencies.
Prompt Assessment of Diuretic Responses May Guide Diuretic Dosing
The classic teaching in the diagnostic workup of acute renal failure has been to avoid measuring urine electrolytes due to the interference of diuretics. However, there has been increasing recognition that urine sodium and other electrolytes are key metrics to assess diuretic response, and should be considered in the setting of acute CRS to determine whether the ineffective volume removal is a result of inadequate diuretic prescription and/or delivery. This concept has first been suggested by Singh and colleagues, upon assessing both urine sodium and furosemide levels in a cohort of patients receiving continuous intravenous furosemide therapy in the intensive care unit. They observed the discrepancies between natriuretic responses to loop diuretics and glomerular filtration rates, as well as the poor prognostication of impaired natriuresis despite adequate furosemide delivery (as directly measured by urine furosemide levels and calculating the relatively low urine sodium-to-furosemide ratios) [53]. Subsequently, the use of clinical parameters such as urine output or weight per set amounts of diuretics (e.g., 40 mg furosemide equivalent) has been proposed by several groups as a quantifiable metric to identify diuretic resistance and the need to adjust or augment diuretic therapy [54, 55]. This important point has been highlighted in the latest European Society of Cardiology Heart Failure Association Cardio-Renal Working Group consensus statement on the use of diuretics in acute HF (summarized in Fig. 2) [56], and are now supported by observations from multiple studies [57,58,59,60].
Goal-Directed Stepwise Pharmacologic Therapy May Overcome Inadequate Diuretic Responses
It has been recognized for decades that if diuretic responses are deemed insufficient with loop diuretics alone, additional drugs can be used to augment natriuresis to provide some short-term relief [61]. The concept of sequential nephron blockade builds on the premise that increase in sodium reabsorption occurs at the distal convoluted tubule in the setting of upstream Na-K-2Cl symporter inhibition by loop diuretics [62]. In most cases, the distal nephron has been the primary target, since targeting compensatory increases in sodium reabsorption at the distal convoluting tubules in response to loop diuretic use may overcome diuretic resistance [63].
Blocking distal renal tubular sodium reabsorption has been achieved by thiazide diuretics as illustrated in the stepwise pharmacologic arm of the CARRESS-AHF study (Table 1) [46]. Often used as a second-line strategy for persistent congestion despite high-dose loop diuretics, oral metolazone and hydrochlorothiazide or intravenous chlorothiazide have been added. In post hoc analyses of several studies, add-on thiazide use can produce greater diuresis and weight loss compared with loop diuretics alone in the setting of WRF [64]. Head-to-head comparisons, however, have yet to reveal significant advantages of one over another [65,66,67,68]. Vasopressin receptor antagonists have also been considered as an adjunctive therapy with or without hyponatremia, but randomized controlled trials did not show incremental clinical benefit [69].
Natriuretic doses of mineralocorticoid receptor antagonists (MRA) have spurred some interest in this arena especially given their established clinical benefits in patients with chronic HF as well as other edematous states like liver cirrhosis. Earlier single-center studies have suggested improvement in natriuresis with the addition of MRA in acute HF [60, 70]. However the ATHENA-AHF study did not show incremental clinical benefits when adding natriuretic-dose MRA (100 mg daily) to high-dose intravenous furosemide even in the setting of diuretic resistance [71, 72]. This may be explained by lower than anticipated concentrations of active metabolites of spironolactone (including the main metabolite canrenone, which often takes a few days to accumulate) measured in the MRA group, suggesting that either there was a potential pharmacokinetic issue with the study drug or the potential benefits of MRA may not be fully realized when administered acutely [73].
Dogma #3: Withholding Neurohormonal Antagonists and Sodium Restriction Are Key Strategies to Restore Renal Function in Acute Cardiorenal Syndrome
In the setting of WRF (as well as hypotension or hyperkalemia), it is not uncommon for clinicians to avoid nephrotoxins, optimize renal perfusion, and improve total body sodium balance. Inhibitors of the renin-angiotensin-aldosterone system (RAAS) have long been associated with reducing glomerular filtration rates via their vasodilatory actions from blockade of angiotensin II-induced constriction of the efferent arteriole [47]. Once acute CRS ensues, clinicians often withhold RAAS inhibitors and advocate salt restriction while administrating high-dose loop diuretics, even though these strategies have very limited supportive evidence [74]. However, the “low-salt syndrome” in refractory heart failure can also contribute to diuretic resistance, rendering a reconsideration of the importance of electrolyte homeostasis in this vulnerable population.
Besides hyponatremia, other often-overlooked electrolyte derangements have been associated with acute CRS. Chloride is the key electrolyte for regulating both reabsorption of tubular electrolytes and water in the kidney through the RAAS and distribution of body fluid in each compartment of the body. In the era of mercurial diuretics, lysine chloride had been used to treat refractory fluid retention [75]. Recognition of low serum chloride as a marker of poor prognosis and revisiting the chloride-sensing renin release as part of tubuloglomerular feedback has also prompted the concept that electrolyte depletion may exacerbate diuretic resistance [76,77,78]. As hypochloremia is intimately associated with diuretic resistance [79], careful selection and combination of various diuretics and their doses with careful considerations of not only sodium/volume but also chloride homeostasis (largely targeting the proximal renal tubule) could become an important therapeutic option for acute CRS.
Withholding and Rechallenging Neurohormonal Antagonists in Acute CRS
Inhibition of the RAAS and sympathetic nervous system is a cornerstone of HF management in improving long-term clinical outcomes [49]. However, most of the landmark trials that support their use have excluded patients with acute CRS, making the benefit of these agents uncertain in this population [80]. That being said, WRF associated with initiation of RAAS inhibitors have not been associated with worse outcomes, and in fact showed improvement in long-term outcomes [81, 82]. As discussed earlier, rise in serum creatinine alone is not associated with worse clinical outcomes, and therefore should not be a justifiable reason to withhold neurohormonal blocking agents which have proven mortality benefit [83]. Hyperkalemia on the other hand does occur in the setting of intravascular depletion in some patients, and can be countered by lowering doses or adding oral potassium binders [83]. Randomized controlled trials in this area are lacking. Nevertheless, post hoc analyses from contemporary studies have shown that use of RAAS inhibitors following hospital discharge from acute HF was associated with better clinical outcomes [84]. That being said, maximizing RAAS blockade with addition of aliskiren (renin inhibitors) did not show incremental benefits in patients admitted for acute HF [85], yet treatment with sacubitril/valsartan confer clinical benefits when compared with enalapril [86]. These findings are concordant with the fact that guideline-directed medical therapy that has been shown to improve the clinical trajectory of patients with heart failure should be maintained throughout the clinical course or re-administered as soon as possible if being withheld. Judicious rechallenging of these drugs is warranted, and if intolerance is encountered, it is imperative that patients are followed closely to consider restarting these medications following discharge.
Targeting Proximal Renal Tubules as a Novel Treatment Strategy for Acute CRS
The proximal renal tubules (PCT) have been largely overlooked in the cause and progression of acute CRS, in part due to the myopic focus on enhancing the effectiveness of loop diuretics and the assumption that PCT may contribute very little in the overall natriuretic potential, due to the belief that the distal nephron serves as the key driver of diuretic resistance [63]. Meanwhile, drugs that specifically target the PCT (such as acetazolamide) often produce a weak diuretic effect, and have often been used to modify acid-base status in specific conditions like high-altitude pulmonary edema. The potential “loop diuretic-sparing” role of acetazolamide, a carbon anhydrase inhibitor targeting the PCT, has been revisited in two pilot studies that compared add-on intravenous or oral acetazolamide with loop diuretics alone in patients admitted with acute HF. Both studies observed statistically significant increases in natriuresis with the addition of acetazolamide when adjusted for loop diuretic dose [87, 88]. Meanwhile in a retrospective case series of ambulatory patients with advanced heart failure, use of oral acetazolamide was also associated with an improvement in functional class and on surrogates of fluid overload [89]. While it is unclear whether acute administration of acetazolamide can alter the natural history of acute CRS, there is evidence that acetazolamide can preserve chloride homeostasis [90]. It is conceivable that early administration of acetazolamide may act synergistically with loop diuretics to improve decongestion in acute HF and clinical outcomes, which is the subject of an ongoing multicenter clinical trial [91].
Sodium-glucose transporter-2 (SGLT2) inhibitors have emerged as an important disease-modifying drug therapy in heart failure [92]. SGLT2 inhibitors exert their glycosuric and natriuretic effects through inhibition of glucose and sodium cotransport in the PCT. In several major cardiovascular outcome trials, SGLT2 inhibitors were found to significantly reduce progression of renal dysfunction [93,94,95]. One of the proposed mechanisms for renal protection by SGLT2 inhibitors is that the enhanced natriuresis results in increased sodium delivery to the macula densa, resulting in afferent arteriolar vasoconstriction and thereby reduced glomerular pressure [96]. Another hypothesis is that inhibition of SGLT2 activity reduces the oxygen demand of the nephron, rendering it less susceptible to oxidative stress injury in the setting of hypoxia or impaired renal perfusion [97,98,99]. Early insights suggested that SGLT2 inhibitor use in the acute HF population is safe and may improve overall diuresis [100,101,102]. Interestingly, empagliflozin has been shown to produce an additive natriuretic effect over time when given prior to bumetanide that is independent of glycosuria, and may be associated with increased erythropoietin levels and uric acid excretion [103]. SGLT2 inhibitors also appeared to preserve serum chloride levels in diabetic patients [104]. Yet, their role in acute CRS remains to be determined.
Direct Modulation of Sodium Avidity with Saline to Promote Excretion
Saline administration during aggressive diuretic therapy has long been considered as a controversial treatment strategy for acute CRS. The proposed mechanism is that salt restriction leads to a reduction in chloride sensing by the macula densa in the distal nephron, which leads to a sodium-avid state in the kidney [105]. Beyond the initial reports from Italy [106], several groups have also demonstrated the potential benefits of concomitant saline infusion during aggressive diuresis during acute HF admissions [107,108,109,110]. However, a small single-center, pilot randomized trial in patients with underlying advanced chronic kidney disease failed to demonstrate incremental benefits of hypertonic saline use [111]. Recently, Griffin and colleagues reported their single-center experience on the use of hypertonic saline solution in patients with advanced HF and diuretic resistance [112]. In this non-randomized retrospective cohort study, daily administration of a 150-mL bolus of 3% hypertonic saline via a standardized protocol was associated with improved diuresis without significant change in respiratory status, adverse neurologic effects, or sodium levels [112]. It is of interest that concomitant saline infusion with loop diuretic therapy has been tested in a device-based algorithm-driven approach, with a goal-directed dose adjustments based on natriuretic/diuretic feedback (so-called controlled decongestion), showing safety and potential effectiveness [113].
Conclusions and Future Perspectives
Our current understanding of acute CRS remains limited in part because of the lack of a uniform clinical definition as a result of the lack of a unifying pathophysiologic mechanism. Clinicians and investigators have been distracted by largely inaccurate assertions that the underlying renal pathophysiology parallels other forms of oliguric AKI and that our indiscriminatory use of intravenous loop diuretics without mechanistic insights can effectively alter the natural history of what has triggered the congestion to begin with. Loop diuretics remain the mainstay of any congested state although their effective usage could potentially circumvent a subset of otherwise (real or conceived) iatrogenic situations leading to acute CRS. Meanwhile, new treatment strategies have the potential of providing adjunctive support or even directly targeting the pathologic states of congestion or organ perfusion that may be independent of loop diuretic use.
Further research is needed to better elucidate the underlying mechanisms of cardiorenal syndrome and potential targets for treatment. From a diagnostic standpoint, we need a better definition of cardiorenal syndrome that, when used, prompts the clinician to approach the case in a different manner from the routine care of an acute HF patient. From a therapeutic standpoint, novel therapeutic strategies targeting PCT or upfront segmental nephron blockade of sodium reabsorption and those that modify renal sodium avidity should be further investigated. Meanwhile, exciting new device-based therapies aimed at improving cardiorenal hemodynamics and salt/volume homeostasis are currently under intensive investigation in pilot first-in-human studies [114,115,116,117,118,119]. These novel strategies provide unique regional modulations that may offer promising avenues of therapy for selected patients with acute CRS in the future while providing important mechanistic insights into the complex pathophysiology of acute CRS [120].
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
Stengel A. Cardiorenal disease: the clinical determination of cardiovascular and renal responsibility, respectively, in its disturbance. JAMA. 1914;63(17):1463–9.
Ronco C, Haapio M, House AA, Anavekar N, Bellomo R. Cardiorenal syndrome. J Am Coll Cardiol. 2008;52(19):1527–39.
• Rangaswami J, Bhalla V, Blair JEA, Chang TI, Costa S, Lentine KL, et al. Cardiorenal syndrome: classification, pathophysiology, diagnosis, and treatment strategies: a scientific statement from the American Heart Association. Circulation. 2019;139(16):e840–e78 Updated scientific statement with extensive summary of cardio-renal syndrome across the spectrum of heart failure and renal failure.
National Heart L, Institute B. NHLBI working group: cardio-renal connections in heart failure and cardiovascular disease, 2004. 2014.
Ljungman S, Laragh JH, Cody RJ. Role of the kidney in congestive heart failure. Relationship of cardiac index to kidney function. Drugs. 1990;39(Suppl 4):10–21 discussion 2-4.
• Mullens W, Damman K, Testani JM, Martens P, Mueller C, Lassus J, et al. Evaluation of kidney function throughout the heart failure trajectory - a position statement from the Heart Failure Association of the European Society of Cardiology. Eur J Heart Fail. 2020;22(4):584–603 Key position statement that provides guidance on how to effectively use diuretics in heart failure, outlining someone of the key advances including adequate diuretics dosing and prompt assessment of treatment responses with urine output or spot urine sodium.
Schrier RW, Abraham WT. Hormones and hemodynamics in heart failure. N Engl J Med. 1999;341(8):577–85.
Hanberg JS, Sury K, Wilson FP, Brisco MA, Ahmad T, Ter Maaten JM, et al. Reduced cardiac index is not the dominant driver of renal dysfunction in heart failure. J Am Coll Cardiol. 2016;67(19):2199–208.
Mullens W, Abrahams Z, Francis GS, Sokos G, Taylor DO, Starling RC, et al. Importance of venous congestion for worsening of renal function in advanced decompensated heart failure. J Am Coll Cardiol. 2009;53(7):589–96.
• Gottlieb SS, Abraham W, Butler J, Forman DE, Loh E, Massie BM, et al. The prognostic importance of different definitions of worsening renal function in congestive heart failure. J Card Fail. 2002;8(3):136–41 Paper that established the definiton of “worsening renal function” as a metric for cardio-renal syndrome for the acute heart failure population.
Tang WH, Dupont M, Hernandez AF, Voors AA, Hsu AP, Felker GM, et al. Comparative assessment of short-term adverse events in acute heart failure with cystatin C and other estimates of renal function: results from the ASCEND-HF trial. JACC Heart Fail. 2015;3(1):40–9.
Dupont M, Wu Y, Hazen SL, Tang WH. Cystatin C identifies patients with stable chronic heart failure at increased risk for adverse cardiovascular events. Circ Heart Fail. 2012;5(5):602–9.
• Testani JM, Chen J, McCauley BD, Kimmel SE, Shannon RP. Potential effects of aggressive decongestion during the treatment of decompensated heart failure on renal function and survival. Circulation. 2010;122(3):265–72 Landmark paper showing worsening renal function in the setting of effective diuresis does not lead to poor long-term outcomes in acute heart failure.
McCallum W, Tighiouart H, Testani JM, Griffin M, Konstam MA, Udelson JE, et al. Acute kidney function declines in the context of decongestion in acute decompensated heart failure. JACC Heart Fail. 2020;8(7):537–47.
Metra M, Cotter G, Senger S, Edwards C, Cleland JG, Ponikowski P, et al. Prognostic significance of creatinine increases during an acute heart failure admission in patients with and without residual congestion: a post hoc analysis of the PROTECT data. Circ Heart Fail. 2018;11(5):e004644.
Metra M, Davison B, Bettari L, Sun H, Edwards C, Lazzarini V, et al. Is worsening renal function an ominous prognostic sign in patients with acute heart failure? The role of congestion and its interaction with renal function. Circ Heart Fail. 2012;5(1):54–62.
Brisco MA, Zile MR, Hanberg JS, Wilson FP, Parikh CR, Coca SG, et al. Relevance of changes in serum creatinine during a heart failure trial of decongestive strategies: insights from the DOSE trial. J Card Fail. 2016;22(10):753–60.
Massie BM, O’Connor CM, Metra M, Ponikowski P, Teerlink JR, Cotter G, et al. Rolofylline, an adenosine A1-receptor antagonist, in acute heart failure. N Engl J Med. 2010;363(15):1419–28.
Kidney Disease: Improving global outcomes (KDIGO) Acute Kidney Injury WorkGroup. KDIGO Clinical Practice Guideline for Acute Kidney Injury. Kidney Int Suppl. 2012;2:1–138.
Maisel AS, Mueller C, Fitzgerald R, Brikhan R, Hiestand BC, Iqbal N, et al. Prognostic utility of plasma neutrophil gelatinase-associated lipocalin in patients with acute heart failure: the NGAL EvaLuation along with B-type NaTriuretic peptide in acutely decompensated heart failure (GALLANT) trial. Eur J Heart Fail. 2011;13(8):846–51.
Shrestha K, Shao Z, Singh D, Dupont M, Tang WH. Relation of systemic and urinary neutrophil gelatinase-associated lipocalin levels to different aspects of impaired renal function in patients with acute decompensated heart failure. Am J Cardiol. 2012;110(9):1329–35.
Dupont M, Shrestha K, Singh D, Awad A, Kovach C, Scarcipino M, et al. Lack of significant renal tubular injury despite acute kidney injury in acute decompensated heart failure. Eur J Heart Fail. 2012;14(6):597–604.
Ahmad T, Jackson K, Rao VS, Tang WHW, Brisco-Bacik MA, Chen HH, et al. Worsening renal function in patients with acute heart failure undergoing aggressive diuresis is not associated with tubular injury. Circulation. 2018;137(19):2016–28.
Rao VS, Ahmad T, Brisco-Bacik MA, Bonventre JV, Wilson FP, Siew ED, et al. Renal effects of intensive volume removal in heart failure patients with preexisting worsening renal function. Circ Heart Fail. 2019;12(6):e005552.
• Fallick C, Sobotka PA, Dunlap ME. Sympathetically mediated changes in capacitance: redistribution of the venous reservoir as a cause of decompensation. Circ Heart Fail. 2011;4(5):669–75 Important paper that outlined the concept of volume redistribution as underlying cause of congetion in some patients with heart failure.
Miller WL, Mullan BP. Understanding the heterogeneity in volume overload and fluid distribution in decompensated heart failure is key to optimal volume management: role for blood volume quantitation. JACC Heart Fail. 2014;2(3):298–305.
• Bart BA, Goldsmith SR, Lee KL, Givertz MM, O’Connor CM, Bull DA, et al. Ultrafiltration in decompensated heart failure with cardiorenal syndrome. N Engl J Med. 2012;367(24):2296–304 Landmark trial showing equivalence of stepwise pharmacologic therapy with ultrafiltration in patients with acute heart failure and worsening renal function.
Kitai T, Grodin JL, Kim YH, Tang WH. Impact of ultrafiltration on serum sodium homeostasis and its clinical implication in patients with acute heart failure, congestion, and worsening renal function. Circ Heart Fail. 2017;10(2):e003603.
Grodin JL, Carter S, Bart BA, Goldsmith SR, Drazner MH, Tang WHW. Direct comparison of ultrafiltration to pharmacological decongestion in heart failure: a per-protocol analysis of CARRESS-HF. Eur J Heart Fail. 2018;20(7):1148–56.
Blake WD, Wegria R. Effect of increased renal venous pressure on renal function. Am J Phys. 1949;157(1):1–13.
Burnett JC, Knox FG. Renal interstitial pressure and sodium excretion during renal vein constriction. Am J Phys. 1980;238(4):F279–82.
Dilley JR, Corradi A, Arendshorst WJ. Glomerular ultrafiltration dynamics during increased renal venous pressure. Am J Phys. 1983;244(6):F650–8.
Doty JM, Saggi BH, Sugerman HJ, Blocher CR, Pin R, Fakhry I, et al. Effect of increased renal venous pressure on renal function. J Trauma. 1999;47(6):1000–3.
Damman K, van Deursen VM, Navis G, Voors AA, van Veldhuisen DJ, Hillege HL. Increased central venous pressure is associated with impaired renal function and mortality in a broad spectrum of patients with cardiovascular disease. J Am Coll Cardiol. 2009;53(7):582–8.
Nijst P, Martens P, Dupont M, Tang WHW, Mullens W. Intrarenal flow alterations during transition from euvolemia to intravascular volume expansion in heart failure patients. JACC Heart Fail. 2017;5(9):672–81.
Nijst P, Verbrugge FH, Martens P, Dupont M, Tang WHW, Mullens W. Renal response to intravascular volume expansion in euvolemic heart failure patients with reduced ejection fraction: mechanistic insights and clinical implications. Int J Cardiol. 2017;243:318–25.
Verbrugge FH, Dupont M, Steels P, Grieten L, Malbrain M, Tang WH, et al. Abdominal contributions to cardiorenal dysfunction in congestive heart failure. J Am Coll Cardiol. 2013;62(6):485–95.
Abu-Saleh N, Aronson D, Khamaisi M, Khoury EE, Awad H, Kabala A, et al. Increased intra-abdominal pressure induces acute kidney injury in an experimental model of congestive heart failure. J Card Fail. 2019;25(6):468–78.
• Mullens W, Abrahams Z, Skouri HN, Francis GS, Taylor DO, Starling RC, et al. Elevated intra-abdominal pressure in acute decompensated heart failure: a potential contributor to worsening renal function? J Am Coll Cardiol. 2008;51(3):300–6 Demonstration of intra-abdominal hypertension as contributor to acute cardio-renal syndrome.
Rapaport E, Weisbart MH, Levine M. The splanchnic blood volume in congestive heart failure. Circulation. 1958;18(4 Part 1):581–7.
Mullens W, Abrahams Z, Francis GS, Taylor DO, Starling RC, Tang WH. Prompt reduction in intra-abdominal pressure following large-volume mechanical fluid removal improves renal insufficiency in refractory decompensated heart failure. J Card Fail. 2008;14(6):508–14.
Fudim M, Ganesh A, Green C, Jones WS, Blazing MA, DeVore AD, et al. Splanchnic nerve block for decompensated chronic heart failure: splanchnic-HF. Eur Heart J. 2018;39(48):4255–6.
Fudim M, Jones WS, Boortz-Marx RL, Ganesh A, Green CL, Hernandez AF, et al. Splanchnic nerve block for acute heart failure. Circulation. 2018;138(9):951–3.
Hasselblad V, Gattis Stough W, Shah MR, Lokhnygina Y, O’Connor CM, Califf RM, et al. Relation between dose of loop diuretics and outcomes in a heart failure population: results of the ESCAPE trial. Eur J Heart Fail. 2007;9(10):1064–9.
Dupont M, Mullens W, Finucan M, Taylor DO, Starling RC, Tang WH. Determinants of dynamic changes in serum creatinine in acute decompensated heart failure: the importance of blood pressure reduction during treatment. Eur J Heart Fail. 2013;15(4):433–40.
Bart BA, Goldsmith SR, Lee KL, Givertz MM, O’Connor CM, Bull DA, et al. Ultrafiltration in decompensated heart failure with cardiorenal syndrome. N Engl J Med. 2012;367(24):2296–304.
Oster JR, Materson BJ. Renal and electrolyte complications of congestive heart failure and effects of therapy with angiotensin-converting enzyme inhibitors. Arch Intern Med. 1992;152(4):704–10.
Stason WB, Cannon PJ, Heinemann HO, Laragh JH. Furosemide. A clinical evaluation of its diuretic action. Circulation. 1966;34(5):910–20.
Yancy CW, Jessup M, Bozkurt B, Butler J, Casey DE Jr, Drazner MH, et al. 2013 ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2013;62(16):e147–239.
Felker GM, Ellison DH, Mullens W, Cox ZL, Testani JM. Diuretic therapy for patients with heart failure: JACC state-of-the-art review. J Am Coll Cardiol. 2020;75(10):1178–95.
• Felker GM, Lee KL, Bull DA, Redfield MM, Stevenson LW, Goldsmith SR, et al. Diuretic strategies in patients with acute decompensated heart failure. N Engl J Med. 2011;364(9):797–805 Landmark trial showing equivalence of high versus low-dose as well as bolus vs continuous dose of loop diuretics in acute heart failure.
Hanberg JS, Tang WHW, Wilson FP, Coca SG, Ahmad T, Brisco MA, et al. An exploratory analysis of the competing effects of aggressive decongestion and high-dose loop diuretic therapy in the DOSE trial. Int J Cardiol. 2017;241:277–82.
Singh D, Shrestha K, Testani JM, Verbrugge FH, Dupont M, Mullens W, et al. Insufficient natriuretic response to continuous intravenous furosemide is associated with poor long-term outcomes in acute decompensated heart failure. J Card Fail. 2014;20(6):392–9.
Testani JM, Brisco MA, Turner JM, Spatz ES, Bellumkonda L, Parikh CR, et al. Loop diuretic efficiency: a metric of diuretic responsiveness with prognostic importance in acute decompensated heart failure. Circ Heart Fail. 2014;7(2):261–70.
Ter Maaten JM, Valente MA, Damman K, Cleland JG, Givertz MM, Metra M, et al. Combining diuretic response and hemoconcentration to predict rehospitalization after admission for acute heart failure. Circ Heart Fail. 2016;9(6):e002845.
Mullens W, Damman K, Harjola VP, Mebazaa A, Brunner-La Rocca HP, Martens P, et al. The use of diuretics in heart failure with congestion - a position statement from the Heart Failure Association of the European Society of Cardiology. Eur J Heart Fail. 2019;21(2):137–55.
Biegus J, Zymlinski R, Sokolski M, Todd J, Cotter G, Metra M, et al. Serial assessment of spot urine sodium predicts effectiveness of decongestion and outcome in patients with acute heart failure. Eur J Heart Fail. 2019;21(5):624–33.
Collins SP, Jenkins CA, Baughman A, Miller KF, Storrow AB, Han JH, et al. Early urine electrolyte patterns in patients with acute heart failure. ESC Heart Fail. 2019;6(1):80–8.
Damman K, Ter Maaten JM, Coster JE, Krikken JA, van Deursen VM, Krijnen HK, et al. Clinical importance of urinary sodium excretion in acute heart failure. Eur J Heart Fail. 2020; published online on February 22, 2020. https://doi.org/10.1002/ejhf.1753.
Ferreira JP, Girerd N, Medeiros PB, Santos M, Carvalho HC, Bettencourt P, et al. Spot urine sodium excretion as prognostic marker in acutely decompensated heart failure: the spironolactone effect. Clin Res Cardiol. 2016;105(6):489–507.
Leiter L. Combinations of diuretics in the treatment of edema. Am Heart J. 1970;80(3):422–6.
Knauf H, Mutschler E. Sequential nephron blockade breaks resistance to diuretics in edematous states. J Cardiovasc Pharmacol. 1997;29(3):367–72.
Rao VS, Planavsky N, Hanberg JS, Ahmad T, Brisco-Bacik MA, Wilson FP, et al. Compensatory distal reabsorption drives diuretic resistance in human heart failure. J Am Soc Nephrol. 2017;28(11):3414–24.
Grodin JL, Stevens SR, de Las FL, Kiernan M, Birati EY, Gupta D, et al. Intensification of medication therapy for cardiorenal syndrome in acute decompensated heart failure. J Card Fail. 2016;22(1):26–32.
Brisco-Bacik MA, Ter Maaten JM, Houser SR, Vedage NA, Rao V, Ahmad T, et al. Outcomes associated with a strategy of adjuvant metolazone or high-dose loop diuretics in acute decompensated heart failure: a propensity analysis. J Am Heart Assoc. 2018;7(18):e009149.
Cardinale M, Altshuler J, Testani JM. Efficacy of intravenous chlorothiazide for refractory acute decompensated heart failure unresponsive to adjunct metolazone. Pharmacotherapy. 2016;36(8):843–51.
Cox ZL, Hung R, Lenihan DJ, Testani JM. Diuretic strategies for loop diuretic resistance in acute heart failure: the 3T trial. JACC Heart Fail. 2020;8(3):157–68.
Shulenberger CE, Jiang A, Devabhakthuni S, Ivaturi V, Liu T, Reed BN. Efficacy and safety of intravenous chlorothiazide versus oral metolazone in patients with acute decompensated heart failure and loop diuretic resistance. Pharmacotherapy. 2016;36(8):852–60.
Konstam MA, Gheorghiade M, Burnett JC Jr, Grinfeld L, Maggioni AP, Swedberg K, et al. Effects of oral tolvaptan in patients hospitalized for worsening heart failure: the EVEREST outcome trial. JAMA. 2007;297(12):1319–31.
Verbrugge FH, Martens P, Ameloot K, Haemels V, Penders J, Dupont M, et al. Spironolactone to increase natriuresis in congestive heart failure with cardiorenal syndrome. Acta Cardiol. 2019;74(2):100–7.
Butler J, Anstrom KJ, Felker GM, Givertz MM, Kalogeropoulos AP, Konstam MA, et al. Efficacy and safety of spironolactone in acute heart failure: the ATHENA-HF randomized clinical trial. JAMA Cardiol. 2017;2(9):950–8.
Greene SJ, Felker GM, Giczewska A, Kalogeropoulos AP, Ambrosy AP, Chakraborty H, et al. Spironolactone in acute heart failure patients with renal dysfunction and risk factors for diuretic resistance: from the ATHENA-HF trial. Can J Cardiol. 2019;35(9):1097–105.
de Denus S, Leclair G, Dube MP, St-Jean I, Zada YF, Oussaid E, et al. Spironolactone metabolite concentrations in decompensated heart failure: insights from the ATHENA-HF trial. Eur J Heart Fail. 2020; published online on April 1, 2020. https://doi.org/10.1002/ejhf.1802.
Krantz MJ, Ambardekar AV, Kaltenbach L, Hernandez AF, Heidenreich PA, Fonarow GC, et al. Patterns and predictors of evidence-based medication continuation among hospitalized heart failure patients (from get with the guidelines-heart failure). Am J Cardiol. 2011;107(12):1818–23.
Rubin AL, Spritz N, Mead AW, Herrmann RA, Braveman WS, Luckey EH. The use of L-lysine monomydrochloride in combination with mercurial diuretics in the treatment of refractory fluid retention. Circulation. 1960;21:332–6.
Grodin JL, Mullens W, Dupont M, Taylor DO, McKie PM, Starling RC, et al. Hemodynamic factors associated with serum chloride in ambulatory patients with advanced heart failure. Int J Cardiol. 2018;252:112–6.
Grodin JL, Simon J, Hachamovitch R, Wu Y, Jackson G, Halkar M, et al. Prognostic role of serum chloride levels in acute decompensated heart failure. J Am Coll Cardiol. 2015;66(6):659–66.
Grodin JL, Sun JL, Anstrom KJ, Chen HH, Starling RC, Testani JM, et al. Implications of serum chloride homeostasis in acute heart failure (from ROSE-AHF). Am J Cardiol. 2017;119(1):78–83.
Hanberg JS, Rao V, Ter Maaten JM, Laur O, Brisco MA, Perry Wilson F, et al. Hypochloremia and diuretic resistance in heart failure: mechanistic insights. Circ Heart Fail. 2016;9(8):e003180.
Damman K, Tang WH, Felker GM, Lassus J, Zannad F, Krum H, et al. Current evidence on treatment of patients with chronic systolic heart failure and renal insufficiency: practical considerations from published data. J Am Coll Cardiol. 2014;63(9):853–71.
Testani JM, Kimmel SE, Dries DL, Coca SG. Prognostic importance of early worsening renal function after initiation of angiotensin-converting enzyme inhibitor therapy in patients with cardiac dysfunction. Circ Heart Fail. 2011;4(6):685–91.
Beldhuis IE, Streng KW, Ter Maaten JM, Voors AA, van der Meer P, Rossignol P, et al. Renin-angiotensin system inhibition, worsening renal function, and outcome in heart failure patients with reduced and preserved ejection fraction: a meta-analysis of published study data. Circ Heart Fail. 2017;10(2): e003588.
Oliveros E, Oni ET, Shahzad A, Kluger AY, Lo KB, Rangaswami J, et al. Benefits and risks of continuing angiotensin-converting enzyme inhibitors, angiotensin II receptor antagonists, and mineralocorticoid receptor antagonists during hospitalizations for acute heart failure. Cardiorenal Med. 2020;10(2):69–84.
Vader JM, LaRue SJ, Stevens SR, Mentz RJ, DeVore AD, Lala A, et al. Timing and causes of readmission after acute heart failure hospitalization-insights from the heart failure network trials. J Card Fail. 2016;22(11):875–83.
Gheorghiade M, Bohm M, Greene SJ, Fonarow GC, Lewis EF, Zannad F, et al. Effect of aliskiren on postdischarge mortality and heart failure readmissions among patients hospitalized for heart failure: the ASTRONAUT randomized trial. JAMA. 2013;309(11):1125–35.
Velazquez EJ, Morrow DA, DeVore AD, Duffy CI, Ambrosy AP, McCague K, et al. Angiotensin-Neprilysin inhibition in acute decompensated heart failure. N Engl J Med. 2019;380(6):539–48.
Imiela T, Budaj A. Acetazolamide as add-on diuretic therapy in exacerbations of chronic heart failure: a pilot study. Clin Drug Investig. 2017;37(12):1175–81.
Verbrugge FH, Martens P, Ameloot K, Haemels V, Penders J, Dupont M, et al. Acetazolamide to increase natriuresis in congestive heart failure at high risk for diuretic resistance. Eur J Heart Fail. 2019;21:1415–22.
Nunez J, Heredia R, Paya A, Sanchis I, Del Prado S, Minana G, et al. Use of acetazolamide in the treatment of patients with refractory congestive heart failure. Cardiovasc Ther. 2018;36(6):e12465.
Kataoka H. Acetazolamide as a potent chloride-regaining diuretic: short- and long-term effects, and its pharmacologic role under the ‘chloride theory’ for heart failure pathophysiology. Heart Vessel. 2019;34(12):1952–60.
Mullens W, Verbrugge FH, Nijst P, Martens P, Tartaglia K, Theunissen E, et al. Rationale and design of the ADVOR (acetazolamide in decompensated heart failure with volume overload) trial. Eur J Heart Fail. 2018;20(11):1591–600.
McMurray JJV, Solomon SD, Inzucchi SE, Kober L, Kosiborod MN, Martinez FA, et al. Dapagliflozin in patients with heart failure and reduced ejection fraction. N Engl J Med. 2019;381(21):1995–2008.
Wiviott SD, Raz I, Bonaca MP, Mosenzon O, Kato ET, Cahn A, et al. Dapagliflozin and cardiovascular outcomes in type 2 diabetes. N Engl J Med. 2019;380(4):347–57.
Zinman B, Wanner C, Lachin JM, Fitchett D, Bluhmki E, Hantel S, et al. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med. 2015;373(22):2117–28.
Neal B, Perkovic V, Mahaffey KW, de Zeeuw D, Fulcher G, Erondu N, et al. Canagliflozin and cardiovascular and renal events in type 2 diabetes. N Engl J Med. 2017;377(7):644–57.
Heerspink HJ, Perkins BA, Fitchett DH, Husain M, Cherney DZ. Sodium glucose cotransporter 2 inhibitors in the treatment of diabetes mellitus: cardiovascular and kidney effects, potential mechanisms, and clinical applications. Circulation. 2016;134(10):752–72.
Gilbert RE. SGLT2 inhibitors: beta blockers for the kidney? Lancet Diabetes Endocrinol. 2016;4(10):814.
Kimura Y, Kuno A, Tanno M, Sato T, Ohno K, Shibata S, et al. Canagliflozin, a sodium-glucose cotransporter 2 inhibitor, normalizes renal susceptibility to type 1 cardiorenal syndrome through reduction of renal oxidative stress in diabetic rats. J Diabetes Investig. 2019;10(4):933–46.
Yang CC, Chen YT, Wallace CG, Chen KH, Cheng BC, Sung PH, et al. Early administration of empagliflozin preserved heart function in cardiorenal syndrome in rat. Biomed Pharmacother. 2019;109:658–70.
Damman K, Beusekamp JC, Boorsma EM, Swart HP, Smilde TDJ, Elvan A, et al. Randomized, double-blind, placebo-controlled, multicentre pilot study on the effects of empagliflozin on clinical outcomes in patients with acute decompensated heart failure (EMPA-RESPONSE-AHF). Eur J Heart Fail. 2020;22(4):713–22.
Kambara T, Shibata R, Osanai H, Nakashima Y, Asano H, Murohara T, et al. Importance of sodium-glucose cotransporter 2 inhibitor use in diabetic patients with acute heart failure. Ther Adv Cardiovasc Dis. 2019;13:1753944719894509.
Griffin M, Riello R, Rao VS, Ivey-Miranda J, Fleming J, Maulion C, et al. Sodium glucose cotransporter 2 inhibitors as diuretic adjuvants in acute decompensated heart failure: a case series. ESC Heart Fail. 2020; published online on May 31, 2020. https://doi.org/10.1002/ehf2.12759.
Griffin M, Rao VS, Ivey-Miranda J, Fleming J, Mahoney D, Maulion C, et al. Empagliflozin in heart failure: diuretic and cardio-renal effects. Circulation. 2020 published online on May 15, 2020. https://doi.org/10.1161/CIRCULATIONAHA.120.045691.
Kataoka H, Yoshida Y. Enhancement of the serum chloride concentration by administration of sodium–glucose cotransporter-2 inhibitor and its mechanisms and clinical significance in type 2 diabetic patients: a pilot study. Diabetol Metab Syndr. 2020;12:5.
Mullens W, Verbrugge FH, Nijst P, Tang WHW. Renal sodium avidity in heart failure: from pathophysiology to treatment strategies. Eur Heart J. 2017;38(24):1872–82.
Paterna S, Fasullo S, Parrinello G, Cannizzaro S, Basile I, Vitrano G, et al. Short-term effects of hypertonic saline solution in acute heart failure and long-term effects of a moderate sodium restriction in patients with compensated heart failure with New York Heart Association class III (Class C) (SMAC-HF Study). Am J Med Sci. 2011;342(1):27–37.
Issa VS, Bacal F, Mangini S, Carneiro RM, Azevedo CH, Chizzola PR, et al. Hypertonic saline solution for renal failure prevention in patients with decompensated heart failure. Arq Bras Cardiol. 2007;89(4):251–5.
Gandhi S, Mosleh W, Myers RB. Hypertonic saline with furosemide for the treatment of acute congestive heart failure: a systematic review and meta-analysis. Int J Cardiol. 2014;173(2):139–45.
Morisawa D, Hirotani S, Oboshi M, Sugahara M, Fukui M, Ando T, et al. Combination of hypertonic saline and low-dose furosemide is an effective treatment for refractory congestive heart failure with hyponatremia. J Cardiol Cases. 2014;9(5):179–82.
Lafreniere G, Beliveau P, Begin JY, Simonyan D, Cote S, Gaudreault V, et al. Effects of hypertonic saline solution on body weight and serum creatinine in patients with acute decompensated heart failure. World J Cardiol. 2017;9(8):685–92.
Engelmeier RS, Le TT, Kamalay SE, Utecht KN, Nikstad TP, Kaliebe JW, et al. Radomized trial of high dose furosemide-hypertonic saline in acute decompensated heart failure with advanced renal disease. J Am Coll Cardiol. 2019;59(13 Supplement):E958.
Griffin M, Soufer A, Goljo E, Colna M, Rao VS, Jeon S, et al. Real world use of hypertonic saline in refractory acute decompensated Heart failure: a U.S. center’s experience. JACC Heart Fail. 2020;8(3):199–208.
Biegus J, Zymlinski R, Siwolowski P, Testani J, Szachniewicz J, Tycinska A, et al. Controlled decongestion by Reprieve therapy in acute heart failure: results of the TARGET-1 and TARGET-2 studies. Eur J Heart Fail. 2019;21(9):1079–87.
Rao VS, Turner JM, Griffin M, Mahoney D, Asher J, Jeon S, et al. First-in-human experience with peritoneal direct sodium removal using a zero-sodium solution: a new candidate therapy for volume overload. Circulation. 2020;141(13):1043–53.
Feld Y, Hanani N, Costanzo MR. Hydrostatic pressure gradient ultrafiltration device: a novel approach for extracellular fluid removal. J Heart Lung Transplant. 2018;37(6):794–6.
Kapur NK, Karas RH, Newman S, Jorde L, Chabrashvili T, Annamalai S, et al. First-in-human experience with occlusion of the superior vena cava to reduce cardiac filling pressures in congestive heart failure. Catheter Cardiovasc Interv. 2019;93(7):1205–10.
Vora AN, Schuyler Jones W, DeVore AD, Ebner A, Clifton W, Patel MR. First-in-human experience with Aortix intraaortic pump. Catheter Cardiovasc Interv. 2019;93(3):428–33.
Dierckx R, Vanderheyden M, Heggermont W, Goethals M, Verstreken S, Bartunek J. Treatment of diuretic resistance with a novel percutaneous blood flow regulator: concept and initial experience. J Card Fail. 2019;25(11):932–4.
Regamey J, Barras N, Rusca M, Hullin R. A role for the Reitan catheter pump for percutaneous cardiac circulatory support of patients presenting acute congestive heart failure with low output and renal dysfunction? Futur Cardiol. 2020;16(3):159–64.
Rosenblum H, Kapur NK, Abraham WT, Udelson J, Itkin M, Uriel N, et al. Conceptual considerations for device-based therapy in acute decompensated heart failure: DRI2P2S. Circ Heart Fail. 2020;13(4):e006731.
Funding
Dr. Tang is partially supported by grants from the National Institutes of Health and the Office of Dietary Supplements (R01HL103931, R01DK106000, R01HL126827).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Alan Kiang declares that he has no conflict of interest.
W.H. Wilson Tang is a consultant for Sequana Medical A.G., has received honorarium from Springer Nature for authorship/editorship, and has received personal fees from the American Board of Internal Medicine.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Heart Failure
Rights and permissions
About this article
Cite this article
Tang, W.H.W., Kiang, A. Acute Cardiorenal Syndrome in Heart Failure: from Dogmas to Advances. Curr Cardiol Rep 22, 143 (2020). https://doi.org/10.1007/s11886-020-01384-0
Published:
DOI: https://doi.org/10.1007/s11886-020-01384-0