Abstract
Dosimetry models for the estimation of particle deposition in the human respiratory tract (RT) in conjunction with clearance transport models are vital components to relate human exposure with internal dose in a quantitative manner. The current work highlights knowledge and modelling approaches on particle deposition and translocation in the human body in an effort to determine health risks in respect to different particle physicochemical properties and human physiology parameters. These include breathing conditions, variability of the geometry of the RT, chemical composition and size of deposits. Different dosimetry modelling approaches have been studied including empirical formulations, one-dimensional flow modelling and computational fluid dynamic methods (CFD). The importance of a realistic modelling of hygroscopicity has been also investigated. A better understanding of the relationship between health effects and inhaled particle dose may be elaborated using dosimetry and clearance modelling tools. A future required approach is to combine dosimetry models with physiologically based pharmacokinetic models (PBPK) to simulate the transport and cumulative dose of particle-bound chemical species in different organs and tissues of the human body.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Airborne particulate matter (PM) comprises of particles with different physicochemical characteristics and morphology, and their dynamics are related to their size. Emissions of mainly anthropogenic particles in urban areas have been associated with human health effects (Pope et al., 2009; World Health Organization (WHO), 2021; Hinds, 1999; Gruzieva et al., 2022). The PM variable chemical composition is also in direct relation with the source origin and inherent characteristics of the ambient air. Epidemiological and toxicological studies have studied the effects that PM concentrations have on human health (Pope et al., 2009; Dominici et al., 2005; Gruzieva et al., 2022; Robinson et al., 2022; Bakand and Hayes, 2016; World Health Organization (WHO), 2021; Oberdorster et al., 1994). Specifically, exposure to PM was implicated to respiratory problems (Farokhi et al., 2018) and systemic inflammation, cancer and atherosclerosis among other health effects (Deng et al., 2018; Schwartz, 2004; Perez-Crespo et al., 2022). The European Environmental Agency (EEA) (European Environmental Agency (EEA), 2020) recently reported almost 0.3 million premature deaths in EU-27 during 2019 due to PM exposure.
The quantification of human health risk as a result from exposure to PM requires the study of a sequence of processes, from concentration characteristics to internal dose of ambient particles (USEPA, 2018; Lazaridis, 2011). The scientific challenge is to relate measurements of particle size, number and mass concentrations; human exposure; inhaled and internal dose; synergistic processes; and, finally, potential adverse health effects. In this health assessment, it is crucial to understand the deposition characteristics of particles in the human respiratory tract (RT) in relation to their size and chemical composition, toxicant-target interactions and tissue response (USEPA, 2018, 2019; Housiadas and Lazaridis, 2010). In this research framework, the understanding of particle deposition patterns in the RT and combined effects of different chemical compounds bound to particles may provide scientific evidence in the quantification of potential risk for health effects through inhalation exposure (USEPA, 2019; Darquenne, 2012, Hofmann, 2011; Lazaridis et al., 2001; Stapleton et al. 2000; Chalvatzaki et al., 2022a).
The physicochemical characteristics of the deposited particles (size distribution, density and shape factor) define the deposition site in the RT. Additional factors which affect the deposition profile are physiological (inhalation rate, tidal volume, volumetric flow rate of inspired air of breathing, breathing pattern) and human anatomical parameters (ICRP 1994; Aleksandropoulou and Lazaridis 2013; Brown 2015).
In the respiratory tract modelling, it is necessary to incorporate realistic particle clearance mechanisms modelling for particles from the nasal passage, tracheobronchial and alveolar regions. Materials accumulated in the tracheobronchial airways are mainly transported on the surface of the mucus layer towards the larynx, where it is swallowed and moved into the gastrointestinal tract (GIT) (ICRP, 1994; Gradon et al., 1996; Housiadas and Lazaridis, 2010; Farkas, 2020). Deposited particles are also subject to slow clearance such as their uptake by airway macrophages, epithelial transcytosis, as well as their temporary storage in the periciliary liquid layer and subsequent uptake from the mucus layer. Furthermore, the mechanisms of translocation and disintegration are mainly responsible for the clearance of particles deposited in small nonciliated airways and alveoli. The elimination pathways include sweat, urine, biliary and feces excretion.
Variability in the particle deposition profile and clearance in the human respiratory tract arising from differences in the subject age, sex and health status adds another difficulty in the assessment of human exposure to particles (ICRP, 1994; USEPA, 2018). Research to reduce uncertainty related to the estimation of particle dose to humans will deliver crucial data to health researchers studying the adverse health effects of particles. In this framework, the Εxposome concept studies both environmental exposures and health characteristics to determine human health (Gruzieva et al., 2022; Galveias et al., 2022; Trumble and Finch, 2019).
An important research endeavor is to combine dosimetry models with physiologically based pharmacokinetic models (PBPK) to study the transport of chemical species bound in the particle phase in different organs and tissues of the human body (Jeong, et al., 2022; Kolli et al., 2019). The calculation of target tissue dose through respiratory dosimetry and PBPK modelling, metabolism and translocation of particle dose in body fluids may provide valuable information for assessing the exposure–dose relationships including dosimetry and PBPK approaches (Georgopoulos et al., 2014; Chalvatzaki et al., 2022a; Chalvatzaki and Lazaridis, 2015). Ongoing developments in toxicodynamic modelling include also the inhalation pharmacokinetic models for assessing absorption/bioavailability, such as the AstraZeneca “LungSim” model (Tehler et al., 2018), the Pfizer “PulmoSim” model (Borghardt et al., 2015), the Mimetikos Preludium (Eriksson et al., 2020) dosimetry model and other in silico models for xenobiotic metabolism (Kirchmair et al., 2015; Enlo-Scott et al., 2021; Christou et al., 2020).
In the current paper, we aim to give an overview of modelling approaches of particle deposition and clearance including PBPK models in the human body. Mainly “routinely operational” models are studied and a short presentation of an emerging important literature on CFD modelling approaches was given.
Human respiratory tract
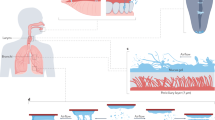
The anatomical structure of the human respiratory system is presented in Fig. 1. Three main anatomic regions can be observed which are common to all mammalian species with the exception of the respiratory bronchioles region (USEPA, 2018). The human respiratory tract comprised from three main regions, the extrathoracic or upper respiratory tract (ET), the tracheobronchial region (TB) and the alveolar–interstitial region (AI) (ICRP, 1994; USEPA, 2018). The main deposition mechanisms are impaction, sedimentation and diffusion. Other deposition mechanisms include interception, turbulent mixing and electrostatic precipitation (Hinds, 1999; Finlay, 2001). In the ET region, the main mechanism is the impaction; in the TB region, a combination of sedimentation and impaction, whereas diffusion dominates in the AI region (ICRP, 1994; Chalvatzaki et al., 2022b).
The term extrathoracic (ET) region refers to the upper airways and incorporates the extrathoracic airways ET1 (anterior nose) and ET2 (posterior nasal passages, larynx, pharynx and mouth). The purpose of this region is to humidify and warm the inhaled air. The regions bronchial (BB) and the bronchiolar (bb) refer to the tracheo-bronchial region and include airways which conduct air from the upper RT to the gas exchange region. The alveolar-interstitial region (AI) is where gas exchange takes place with the blood and it comprises the respiratory bronchioles, the alveolar ducts, the alveoli and the interstitial connective tissue (Housiadas and Lazaridis, 2010).
Particle inhalability in the respiratory tract
Inhalability (I) refers to the intake efficiency of ambient particles to enter the mouth and nose of a human subject. This efficiency depends on both air flow close to the subject and the particle size. Different analytical equations have been proposed in the scientific literature for the definition of the inhalability in respect to wind velocity and particle size (ICRP, 1994; Hinds, 1999; Housiadas and Lazaridis, 2010).
Reliable simulations for the determination of particle deposition patterns in the human RT need the use of a morphometric model for the respiratory system. ICRP (1994) introduced a simple anatomical lung model for the human airways using the symmetric model of Weibel (Weibel A model; Weibel, 1963) (ICRP, 1994; Lazaridis, 2011). The model accounts for 24 airway generations, with 23 symmetrical levels of bifurcations, having trachea as the generation zero and alveoli generation 23. The model considers the different generations as a sequence of filters with specific efficiency to determine the deposition fraction. The dimensions of the airways are variable depending on factors such as age, gender, race and weight. Empirical functions were used to determine the deposition profile in each region of the RT.
The Weibel model is a simplification of the real-world anatomy of the RT and does not account for population variability as well as for time-dependent generation characteristics and asymmetric bifurcations (Finlay, 2001). Therefore, even though the main characteristics and structure of the human respiratory system are known, there are no available detailed models with specific geometry of the different parts. Measurements of casts of normal lungs have been used to determine the dimensions of the different airways (e.g. Weibel, 1963; Weibel, 1991). A simplification of the real structure of the RT was introduced with modelling the airways as cylindrical structures with a defined length and diameter.
An inter-subject variability of extrathoracic morphometry, the tracheobronchial airways and alveolar dimensions have to be studied in the evaluation of the population variability using recent data on imagining technology (Tay et al., 2021) and from in vivo studies (Hussain et al., 2011). Breathing patterns for healthy subjects have also to be examined as another source of inter subject variability.
Dosimetry tools have to be implemented in conjunction with geometry reconstruction of the human respiratory system arising from computed tomography/magnetic resonance imaging (CT/MRI) scans. This will allow for a better characterization of the human dose at different parts of the respiratory tract and targeted organ dose for specific chemical species. Modelled toxicokinetic profiles such as plasma and organ time-dependent concentrations require also to be used to determine associations between dose and toxicodynamic responses.
Deposition and clearance mechanisms
Particle deposition of particles in the RT
Particle deposition depends on their size and physicochemical properties as well as on the human subject’s physiology. The total deposition is calculated as a sum of independent deposition efficiencies of the different deposition mechanisms (Hinds. 1999; Finlay. 2001; Housiadas and Lazaridis, 2010). The particle size and chemical composition are the major determinants for the deposition profile in RT with the larger particles to deposit at the head airways (USEPA, 2018). Particles smaller than 10 μm in size are able to follow the air flow and deposit to the alveolar region with particles smaller than 2.5 nm to be associated stronger with health effects (Schwartz and Neas, 2000). An important aspect of particles is related to their chemical composition since specific components are more toxic such as metals (World Health Organization (WHO), 2021).
The dose of particles in each part of the RT is calculated using the following expression:
where H is the dose-rate (μg/h), n0 is the inhalability ratio, cA is the aerosol concentration in air, B is the ventilation rate, α is the fine mode mass fraction, nfine.i is the retention in region i of lungs for fine particles and ncoarse.i is the retention in region i of lungs for coarse particles (ICRP, 1994).
Different deposition mechanisms are important in the RT with the inertial impaction to be dominant in the upper RT, where particles mainly larger than 0.5 μm fail to follow the flow lines at locations that air changes abruptly direction (International Commission on Radiological Protection (ICRP), 2002). In addition, gravitational settling is important for deposition in small airways and the alveolar region (Lazaridis, 2011). Brownian motion also is a dominant mechanism at the bronchioli and the alveolar region due to low values of the air velocity and long residence time. Besides, Brownian diffusion for particles smaller than 0.01 μm dominates at the head airways due to high flow in the region (International Commission on Radiological Protection (ICRP), 2002; Housiadas and Lazaridis, 2010). Εlectrostatic attraction is also a deposition mechanism with importance mainly in the alveolar region for ultrafine particles (USEPA. 2018; Housiadas and Lazaridis, 2010). For fiber particles, the deposition mechanism of interception is also important (ICRP, 1994).
Model simulations using the ICRP model to determine size-dependent particle deposition at different parts of the RT for various subjects is presented in Fig. 2. At the upper RT, there is higher deposition of particles at sizes lower than 0.2 μm for men due to higher inhalation rates compared to women but for larger particles, there is no noticeable difference. The deposition fraction for a child is higher for particles above 0.2 μm at the upper airways compared to adults but for smaller particles the deposition fraction resembles that of an adult. Specific differences in the deposition characteristics occur between male and female subjects as shown in Fig. 2f with a total deposition larger for male individuals for the fine particle fraction and almost equal between male and female for the coarse fraction (Lazaridis, 2011).
Mass particle percentage which deposits in the a ET1 and b ET2 thoracic regions, e alveolar region, and f total deposition of particles in the RT for different subjects in respect to the aerodynamic particle diameter (Adapted from Lazaridis, 2011).
Furthermore, for a child even though the deposition fraction for the alveolar region is small compared to an adult, the surface density of deposits is large (USEPA. 2018). An interesting situation occurs for people with health problems such as elderly and adults with chronic obstructive pulmonary disease (COPD) who have higher deposition fraction in the RT compared to healthy individuals (USEPA, 2018).
Particle clearance in the human respiratory tract
The human respiratory tract incorporates different particle clearance mechanisms and particle removal from the nasal passage, bronchial tree and the alveolar region. (ICRP, 1994; Housiadas and Lazaridis, 2010; USEPA, 2018; Chalvatzaki and Lazaridis, 2015; Ermund et al., 2021; Phalen et al., 2021). The Human Respiratory Tract Model (HRTM) is a widely adopted model for the calculation of particle clearance (ICRP, 1994; Lazaridis, 2011). The physicochemical properties of the particles (e.g. solubility, particle size, chemical composition), their mass/number dose and the region of the deposition and physiological parameters (metabolic rate, health condition, age, gender) determine the clearance rate of each mechanism (Housiadas and Lazaridis, 2010).
The movement of metals from the blood into the tissues can be described as a blood-flow-limited model by a PBPK model (Reddy et al., 2005; Chou et al., 2009; Sharma et al., 2005; Schroeter et al., 2011; O’Flaherty et al., 2001; Chalvatzaki and Lazaridis, 2015). An overview of a respiratory tract clearance model which is based on ICRP (2012) publication is given in Fig. 3.
An overview of a compartment model showing the time-dependent particle transport from each respiratory tract region (Paquet et al., 2016). Rates shown alongside arrows are reference values in units of day–1, mj,k is the mechanical movement rate of particles from compartment j to k, Rj (t) is the mass of particles in compartment j of the respiratory tract after time t and s is the rate of absorption into blood (adapted from Chalvatzaki and Lazaridis (2015))
There are a number of factors which affect the transport of particles in the RT such as their physicochemical characteristics, physiological and respiration conditions and health status of the individual subject (Smith et al., 2008; USEPA, 2018; Housiadas and Lazaridis, 2010). The clearance mechanisms include mainly particle transport to the gastrointestinal tract via the pharynx and to lymph nodes via lymphatic channels and abrorption into blood (ICRP, 1994 USEPA, 2018; Lazaridis, 2011). These mechanisms have different rates. For example, in the alveolar region, the clearance can be fast (rate close to 0.02 day−1), medium (rate close to 0.001 day−1) or slow (rate close to 0.0001 day−1) in relation to the actual clearance mechanism (ICRP, 1994; Schlesinger, 1988).
The retention of particles in each region is given by the continuity equation
where λi(t) is the instantaneous clearance rate of the deposit in compartment i and Ri(t) the retained mass after time t. The transport is the summation of two rates, the mechanical movement and absorption into blood λi(t) = mi(t) + si(t).
According to the ICRP (2015), the respiratory tract clearance model consists of 13 compartments. The retained dose of particles in each compartment j and the dose to the oesophagus, lymph nodes and blood was estimated by Eqs. 3–4 for rapidly and slowly dissolving particles:
where mk,j is the mechanical movement rate of particles from compartment k to j, mj,k is the mechanical movement rate of particles from compartment j to k, fr is the fraction of particles dissolved rapidly, sr is the rapid dissolution rate, ss is the slow dissolution rate, Hj is the deposited dose in the compartment j, Ij and Ik are the retained doses of particles dissolving relatively rapidly in the compartment j and k respectively and Tj and Tk are the retained doses of particles dissolving slowly in the compartment j and k respectively. The absorption of particles in the blood was assumed to be moderate. The default values for fr, sr and ss during moderate absorption were based on the ICRP. Therefore, fr, sr and ss were set equal to 0.2 day−1, 3 days−1 and 0.005 day−1 respectively.
An example of clearance of deposited PM10 particles from the human respiratory tract estimated from concentrations at a suburban background location (Chania, Greece) is presented in Fig. 4 (Chalvatzaki et al., 2022a). The majority of the deposited particles in the human respiratory tract was transferred to the oesophagus (51 %), a finding that is associated with the high deposited dose in the ET region due to the high contribution of coarse particles to PM10. In the ET region, particles are transferred mainly to the GI tract (oesophagus) via mucociliary clearance while the deposited particles in the TB and AI regions remain in the respiratory tract and absorbed to the blood.
a Dose (deposited and retained) in the human respiratory tract, oesophagus and blood, and b dose in the lymph nodes (dapted from Chalvatzaki et al., 2022a).
Physiologically based pharmacokinetic models
The use of specific dose–response functions for determining the health effects of humans from exposure to particles require a realistic determination of delivered doses of substances. Therefore, there is a need to develop reliable PBPK models for a mixture of particles. The PBPK models simulate the time course of absorption, distribution, metabolism and excretion of chemicals that enter the body. The general concept of PBPK modelling is to mathematically describe relevant physiological, physicochemical and biochemical processes that determine the pharmacokinetic behaviour of a compound. The use of specific dose–response functions for determining the health effects of humans from exposure to particles require a realistic determination of delivered doses of substances.
In PBPK modelling, there are several compartments which correspond to organs or tissues and mathematical formulations which represent physiological processes have been applied to describe the interconnections through blood flow (Jeong et al., 2022; Chou et al., 2009; Sharma et al., 2005; Reddy et al., 2005). Additional chemical compounds have also to be integrated in the existing kinetic models including carbonaceous particles (Nemmar et al., 2002).
The kinetic equations describing the accumulation and clearance of metals (As, Pb, Mn) in each tissue group i can be described (Chalvatzaki and Lazaridis, 2015) as:
where Ai,j is the dose of chemical j in tissue group i (μg), Qi is the blood flow rate to tissue group i (L h−1), Ca,j is the arterial concentration (μg L−1) of chemical j and Cv,j is the venous concentration of chemical j (μg L−1).
Computational dosimetry methods
In the scientific literature, the focus is on the calculation of particle deposition efficiencies using compartmental modelling approaches (USEPA, 2018; Housiadas and Lazaridis, 2010; Yeh and Schum, 1980; Zhang and Kleinstreuer, 2001; Zhang et al., 2002a,b). In respect to the use of one-dimensional numerical methodologies for the determination of the particle deposition in the RT, there was the development of trumpet models (Housiadas and Lazaridis, 2010). In this approach, the human lung is modelled as a chamber shaped like a trumpet with variable cross-sectional area (Nixon and Egan, 1987; Darquenne and Paiva, 1994; Darquenne et al., 2022). Other models include a stochastic approach to account for the lung asymmetry (Koblinger and Hofmann, 1990) and a Markov chain approach to describe the particle movement in the respiratory tract (Sonnenberg et al., 2020; Lin et al., 2021).
Dosimetry models were also used to estimate the deposition of pharmaceutical aerosols in the human respiratory tract by dry powder inhalers (DPIs) (Chalvatzaki et al., 2020; Mellat et al. 2017). Reliable estimation of the pharmaceutical aerosol deposition in the human respiratory tract is vital for a better effective treatment for lung diseases (Rangaraj et al., 2019; Farkas et al. 2019; Kolanjiyil et al. 2017; Chalvatzaki et al., 2020; Mellat et al., 2017).
Computational fluid dynamics (CFD) models have been also applied to simulate the airflow patterns and detailed deposition hot spots in the human RT (Martonen et al., 2005; Koullapis et al., 2021). Recent approaches are aiming to simulate the whole lung combing different modelling approaches such as CFD models for the upper airways with 1D or 0D approaches at the distal lung (Kuprat et al., 2021; Koullapis et al., 2020; Koullapis et al., 2021). These models may be effective tools for a realistic simulation of the particle deposition at specific sites.
Classification of models
The determination of particle deposition in the RT can be accomplished using a variety of in silico methods. One can divide the in silico approaches in two main modelling categories, the empirical and mechanistic ones. In the empirical approaches, the equations which govern the deposition in the RT are based on numerical simulations and experimental data. However, the algebraic expressions used for the determination of deposition fraction do not directly consider fluid flow characteristics, particle processes and differences in physiological geometrical morphologies. The most well-known standard model recommended by the International Commission on Radiological Protection is the ICRP66 model (ICRP, 1994). The model is based on empirical expressions which were derived from a specific database of particle deposition experiments and a morphometric anatomical structure. A model which is based on the ICRP66 methodology is the LUDEP program which simulates the deposition of radionuclides particles in the human respiratory tract using a user friendly software interface. Even though the ICRP66 is based on a specific dataset of experimental data, it is a reliable and easy to use dosimetry model. In addition, the Bioaerosol Adaptation of the International Committee on Radiological Protection’s (ICRP) Lung deposition model (BAIL) (Guha et al., 2014) and alternative approaches (Sturm and Hofmann, 2009) have been used for the prediction of microorganism deposition in the respiratory tract. BAIL assumes a Gaussian distribution of particle sizes, a use of a shape-factor of elongated particles and an alternative approach to determine breathing parameters.
A model which is based on the ICRP approach (1994; 2016) is the ExDoM2 (Chalvatzaki and Lazaridis 2015) which uses modified values for the deposition in the upper respiratory tract and the clearance mechanisms and dose to the blood, oesophagus and lymph nodes. The ExDoM2 model has the feature to simulate particle size distribution data at specific time intervals (Chalvatzaki et al., 2022a, b). Furthermore, the determination of a chemical resolved particle deposition profile in the human respiratory tract using the ExDoM2 model was the focus of the work of Aleksandropoulou and Lazaridis (2013). The study used data derived from indoor/outdoor studies in Oslo, Norway (Lazaridis et al., 2008) and concluded that the dose was enhanced for the carbonaceous fine fraction of particles in the alveolar region of the lung whereas the dose of crustal material dominated in the extrathoracic region.
Another widely used model is the multiple-path particle dosimetry (MPPD V 3.04) model which was developed by Applied Research Associates (https://www.ara.com/mppd/) with the contribution of several agencies for calculation of deposition of airborne particles (Miller et al., 2016).
Mechanistic models on the other hand consider explicitly the particle processes and the actual anatomical details of the individual subjects (Finlay. 2001; Nixon and Egan, 1987; Housiadas and Lazaridis, 2010; Lazaridis et al., 2001; Mitsakou et al., 2005). Even though there is an extensive literature in respect to mechanistic models, mainly the whole-RT 1D (one-dimensional) models and the CFD-based models are discussed.
The whole-RT 1D mechanistic models solve the convective aerosol transport along the flow pathway in one dimension in the RT. CFD models simulate deposition patterns at specific bifurcations in the RT using a detailed velocity field inside the RT and a precise description of the human lung geometry (Lambert et al., 2011; Koullapis et al., 2016; Hofemeier and Sznitman, 2015). Table 1 describes the different model categories accordingly and presents their characteristics.
Empirical compartmental modelling: the ICRP model
A number of empirical models have been presented in the literature (Chan and Lippmann, 1980; Yu and Diu, 1982; Chang et al., 1991; Aleksandropoulou and Lazaridis, 2013; ICRP, 1994).
A simplified set of equations for average exercise level fitted to the ICRP model can be used for determining the deposition fraction at different particle diameters (dp, μm) (Hinds, 1999). These expressions for the deposition fraction (DF) at different parts of RT are shown in Eqs. 6–9 for, respectively, the head airways (regions ET1 + ET2), the tracheobronchial region (BB + bb), the alveolar region (AI) and total deposition.
In the above expressions, IF is the inhalable fraction (inhalability) and is given by Eq. 8 below. The ICRP model gives the expression below for the inhalable fraction in relation to the particle aerodynamic diameter da (expressed in μm) and wind speed U (in m s−1)
Whole-RT 1D mechanistic modelling—comparison with experimental results
Deterministic and stochastic modelling approaches have been implemented in whole-RT 1D mechanistic models using Eulerian and Lagrangian modelling approaches to study the flow patterns in the lung (Housiadas and Lazaridis, 2010). In the stochastic models, Monte Carlo techniques were implemented to simulate lung asymmetry and path variations since variability is an important feature in humans (Asgharian et al., 2001).
In addition, Lagrangian and Eulerian models have been implemented. In the Lagrangian framework, a moving frame of reference was adopted (Anjilvel and Asgharian, 1995; Finlay and Stapleton, 1995; Darquenne and Paiva, 1994; Koblinger and Hofmann, 1990). A detailed description of the Lagrangian modelling framework was given by Housiadas and Lazaridis (2010).
The Eulerian models use fixed frame of reference and are capable to determine deposition fluxes and concentrations (Mitsakou et al. 2005; Lazaridis et al., 2001; Nixon and Egan, 1987; Darquenne and Paiva, 1994). As an example, Fig. 5 shows RT-1D model predictions of deposition in the tracheobronchial and the alveolar regions respectively versus data from different studies (Lazaridis et al., 2001).
Predicted a tracheobronchial deposition and b alveolar deposition in comparison with experimental data (Adapted from Lazaridis et al., 2001)
CFD-based mechanistic modelling
The use of CFD modelling is coming more pronounced the last years using 3-D simulations describing site specific deposition patterns, such as in the nasopharyngeal region, in an alveolated duct and variable geometries of the human respiratory tract including anatomies of subjects with respiratory diseases (Longest et al., 2016; Lambert et al., 2011; Koullapis et al., 2016) delivering at the same time reliable results. The methodology is computationally very intensive and therefore the simulations are mainly limited to the first 10 airway generations (Koullapis et al., 2018) or they cover the terminal alveolar region (Hofemeier and Sznitman, 2015). Therefore, the simulations cannot cover the whole respiratory tract but focus on specific processes such as the effect of local structural features on the velocity field and particle deposition at specific locations of the RT. A detailed discussion on airflow characteristics inside the respiratory tract is given by Housiadas and Lazaridis (2010).
Recent advances combine simulations in a small number of upper generation airways with the alveolar region which was simulated by using spherical alveoli to the distal bifurcation airways, therefore developing a whole lung CFD models (Kolanjiyil and Kleinstreuer, 2013; Longest et al., 2016; Koullapis et al., 2020).
Aerosol processes—hygroscopicity
Aerosol processes such as the hygroscopic growth inside the RT have to be examined in the mechanistic models. The potential of hygroscopic particles to grow in the humid respiratory system has been studied in the scientific literature since this affects directly their size (Löndahl et al. 2009; Winkler-Heil et al. 2014; Chalvatzaki and Lazaridis, 2018; Vu et al. 2015). The relative humidity inside the lungs has been estimated to be 99.5% (Löndahl et al. 2009; Youn et al. 2016). Chemical composition and particle size are the two parameters that influence the hygroscopic growth (Petters and Kreidenweis, 2007). In addition, aerosol mixing and aerosol properties such as size, density, shape significantly influence their deposition profile in the human respiratory (Ching and Kajino, 2018). In particular, aggregates emitted from diesel combustion have higher deposition fractions in comparison to spherical particles with the same mobility diameter (Vu et al., 2018).
The hygroscopic growth can be modelled using the κ-Köhler theory for taking into account the chemical composition (e.g. NaCl) of particles. The hygroscopic growth factor (Gf) is determined using the equation (Petters and Kreidenweis 2007):
where, da is the diameter of particles (μm) at a % RH and dp is the dry diameter (μm).
The hygroscopic growth factor (Gf) may be calculated using the following equation (Petters and Kreidenweis 2007; Carrico et al., 2008):
where κ is the hygroscopicity parameter κ and aw is the water activity.
The water activity was calculated with the following equation (Chalvatzaki and Lazaridis, 2018; Vu et al. 2015):
where RH is the relative humidity and Ck is the Kelvin curvature correction factor.
The growth factor and the diameter of particles at 99.5% RH (Vu et al. 2015) were calculated using Eqs. (6) and (7) respectively:
where da is the diameter of particles at a% RH (before inhalation).
The model results showed that the hygroscopicity plays a more significant role in the tracheobronchial (TB) and alveolar-interstitial (AI) regions of the respiratory tract. In particular, the hygroscopicity increases the deposition of particles for dae ≥ 0.3 μm with the maximum effect to be observed for the size range 0.3 μm ≤ dae ≤ 2.5 μm. In addition, the hygroscopicity leads to a significant decrease in deposition for the size range 0.02 μm ≤ dae ≤ 0.25 μm (Chalvatzaki and Lazaridis, 2018; Vu et al. 2015).
Conclusions
Dosimetry models have been widely used for the calculation of particle deposition due to environmental human exposure and found also applications in medical industry for determining the dose of pharmaceutical particles at specific target sites in the human respiratory tract.
The use of state-of-the-art dosimetry models for determining the particle deposition patterns would elucidate lung burdens and a more accurate prediction of health effects arising from human exposure. In this context, the use of real-world data derived from different indoor microenvironments and outdoors in conjunction with mechanistic tools for particle dosimetry will fill a knowledge gap to estimate actual internal doses from exposure of ambient particles in variable microenvironments and outdoors. This will be also a first step towards a development of dose–response relationships for specific particle species.
There are currently available dosimetry models of varying complexity ranging from empirical to detailed CFD models. Besides, the clearance mechanisms of deposited particles in the RT have been extensively studied and clearance rates have been derived. Especially for specific chemical components in particles such as metals there are available kinetic equations describing their accumulation and clearance at different tissue groups in the body.
It is evident the need of the combined use of dosimetry and PBPK models for determining the internal dose of specific chemical species and for examining the clearance mechanisms of particles and toxicological synergistic effects of chemical mixtures. Therefore, realistic modelling of combined dosimetry and clearance work is expected to transform the way that we understand drivers of potential health effects from exposure to particles. In this direction, it is needed to integrate aerosol dynamic processes such as hygroscopic growth in the calculations of particle deposition.
Data availability
The datasets generated during the current study are available from the corresponding author (lazaridi@mred.tuc.gr) on reasonable request.
References
Aleksandropoulou V, Lazaridis M (2013) Development and application of a model (ExDoM) for calculating the respiratory tract dose and retention of particles under variable exposure conditions. Air Qual Atmos Health 6:13–26. https://doi.org/10.1007/s11869-010-0126-z
Anjilvel S, Asgharian B (1995) A multiple-path model of particle deposition in the rat lung. Fundam Appl Toxicol 28:41–50
Asgharian B, Hofmann W, Miller FJ (2001) Mucociliary clearance of insoluble particles from the tracheobronchial airways of the human lung. J Aerosol Sci 32:817–832
Bakand S, Hayes A (2016) Toxicological considerations, toxicity assessment, and risk management of inhaled nanoparticles. Int J Mol Sci 17(6):929
Borghardt JM, Weber B, Staab A, Kloft C (2015) Pharmacometric models for characterizing the pharmacokinetics of orally inhaled drugs. AAPS J 17:853–870. https://doi.org/10.1208/s12248-015-9760-6
Brown JS (2015) Chapter 27-Deposition of particles. In: Parent JA (ed) Comparative biology of the normal lung, 2nd edn. Academic Press, Cambridge, pp 513–536. https://doi.org/10.1016/B978-0-12-404577-4.00027-8
Carrico CM, Petters MD, Kreidenweis SM, Collett JL Jr, Engling G, Malm WC (2008) Aerosol hygroscopicity and cloud droplet activation of extracts of filters from biomass burning experiments. J Geophys Res Atmos 113(D8). https://doi.org/10.1029/2007JD009274
Chalvatzaki E, Lazaridis M (2015) Development and application of a dosimetry model (ExDoM2) for calculating internal dose of specific particle-bound metals in the human body. Inhal Toxicol 27(6):308–320
Chalvatzaki E, Lazaridis M (2018) A dosimetry model of hygroscopic particle growth in the human respiratory tract. Air Qual Atmos Health 11(4):471–482
Chalvatzaki E, Chatoutsidou SE, Lazaridis M (2020) Deposition of pharmaceutical aerosols in the human respiratory tract by dry powder inhalers (DPIs). J Drug Deliv Sci Technol 59:101915
Chalvatzaki E, Chatoutsidou SE, Lazaridis M (2022a) Regional deposited dose in the human respiratory tract using different particulate metrics. J Environ Expo Assess 1:18
Chalvatzaki E, Katsivela E, Raisi L, Lazaridis M (2022b) Assessment of personal deposited dose of bioaerosols and particles in a wastewater treatment plant facility. Air Qual Atmos Health 16(1):165–181. https://doi.org/10.1007/s11869-022-01264-2
Chan TL, Lippmann M (1980) Experimental measurements and empirical modelling of the regional deposition of inhaled particles in humans. Am Ind Hyg Assoc J 41:399–409
Chang I, Griffith W, Shyr L, Yeh H, Cuddihy G, Seiler A (1991) Software for the draft NCRP respiratory tract dosimetry model. Radiat Prot Dosim 38(1/3):193–199
Ching J, Kajino M (2018) Aerosol mixing state matters for particles deposition in human respiratory system. Sci Rep 8:8864
Chou WC, Chio CO, Liao CM (2009) Assessing airborne PM-bound arsenic exposure risk in semiconductor manufacturing facilities. J Hazard Mater 167:976–986
Christou S, Chatziathanasiou T, Angeli S, Koullapis P, Stylianou F, Sznitman J, Guo HH, Kassinos C (2020) Anatomical variability in the upper tracheobronchial tree: sex-based differences and implications for personalized inhalation therapies. J Appl Physiol 130:678–707
Darquenne C (2012) Aerosol deposition in health and disease. J Aerosol Med Pulm Drug Deliv 25(3):140–147
Darquenne C, Paiva M (1994) One-dimensional simulation of aerosol transport and deposition in the human lung. J Appl Physiol 77:2889–2898
Darquenne C, Borojeni AAT, Colebank MJ, Forest MG, Madas BG, Tawhai M, Jiang Y (2022) Aerosol transport modelling: the key link between lung infections of individuals and populations. Front Physiol 13:923945. https://doi.org/10.3389/fphys.2022.923945
Deng Q, Ou C, Chen J, Xiang Y (2018) Particle deposition in tracheobronchial airways of an infant, child and adult. Sci Total Environ 612:339–346
Dominici F, McDermott A, Daniels M, Zeger SL, Samet JM (2005) Revised analyses of the national morbidity, mortality, and air pollution study: mortality among residents of 90 cities. J Toxicol Environ Health A 68:1071–1092
Enlo-Scott Z, Backstrom E, Mudway I, Forbes B (2021) Drug metabolism in the lungs: opportunities for optimising inhaled medicines. Expert Opin Drug Metab Toxicol 17(5):611–625
Eriksson J, Thorn H, Lennernas H, Sjogren E (2020) Pulmonary drug absorption and systemic exposure in human: predictions using physiologically based biopharmaceutics modeling. Eur J Pharm Biopharm 156:191–202
Ermund A, Meiss LN, Dolan B, Jaudas F, Ewaldsson L, Bahr A, Klymiuk N, Hansson GC (2021) Mucus threads from surface goblet cells clear particles from the airways. Respir Res 22:303. https://doi.org/10.1186/s12931-021-01898-3
European Environmental Agency (EEA) (2020). https://www.eea.europa.eu.
Farkas Á (2020) Simulation of the effect of mucociliary clearance on the bronchial distribution of inhaled radon progenies and related cellular damage using a new deposition and clearance model for the lung. Radiat Environ Biophys 59:651–661. https://doi.org/10.1007/s00411-020-00868-5
Farkas Á, Lizal F, Jedelsky J, Elcner J, Horváth A, Jicha M (2019) Simulation of airway deposition of an aerosol drug in COPD patients. Pharmaceutics 11:153. https://doi.org/10.3390/pharmaceutics11040153
Farokhi A, Heederik D, Smit LAM (2018) Respiratory health effects of exposure to low levels of airborne endotoxin—a systematic review. Environ Health 17(1):1–20
Finlay WH (2001) The mechanics of inhaled pharmaceutical aerosols an introduction. Academic Press, San Diego, CA
Finlay WH, Stapleton KW (1995) The effect on regional lung deposition of coupled heat and mass transfer between hygroscopic droplets and their surrounding phase. J Aerosol Sci 26:655–670
Galveias A, Ribeiro H, Guimaraes F, Costa MJ, Rodrigues P, Costa AR, Abreu I, Antunes CM (2022) Differential Quercus spp. pollen-particulate matter interaction is dependent on geographical areas. Sci Total Environ 832:154892
Georgopoulos PG, Brinkerhoff CJ, Isukapalli S, Dellarco M, Landrigan PJ, Lioy PJ (2014) A Tiered framework for risk-relevant characterization and ranking of chemical exposures: applications to the national children's study (NCS). Risk Anal 34(7):1299–1316
Gradon L, Pratsinis E, Prodgorski A, Scott S, Panda S (1996) Modeling retention of inhaled particles in rat lungs including toxic and overloading effects. J Aerosol Sci 27(3):487–503
Gruzieva O, Jeong A, He S, Yu Z, de Bont J, Pinho MG, Eze IC, Kress S, Wheelock CE, Peters A, Vlaanderen J, de Hoogh K, Scalbert A, Chadeau-Hyam M, Vermeulen RCH, Gehring U, Probst-Hensch N, Melén E (2022) Air pollution, metabolites and respiratory health across the life-course. Eur Respir Rev 31:220038. https://doi.org/10.1183/16000617.0038-2022
Guha S, Hariharan P, Myers M (2014) Enhancement of ICRP's lung deposition model for pathogenic bioaerosols. Aerosol Sci Technol 48(12):1226–1235
Hinds WC (1999) Aerosol technology, properties, behavior, and measurements of airborne particles. Wiley Interscience, p 504
Hofemeier P, Sznitman J (2015) Revisiting pulmonary acinar particle transport: convection, sedimentation, diffusion, and their interplay. J Appl Physiol 39:1375–1385
Hofmann W (2011) Modelling inhaled particle deposition in the human lung—a review. J Aerosol Sci 42(10):693–724
Housiadas C, Lazaridis M (2010) Inhalation dosimetry modelling. In: Lazaridis M, Colbeck I (eds) Human Exposure to Pollutants via Dermal Absorption and Inhalation. Springer
Hussain M, Renate W-H, Hofmann W (2011) Effect of intersubject variability of extrathoracic morphometry, lung airways dimensions and respiratory parameters on particle deposition. J Thorac Dis 3:156–170
ICRP (1994) ICRP Publication 66: Human respiratory tract model for radiological protection. Ann ICRP 24(4):482 Pergamon, Oxford
ICRP (2015) ICRP Publication 130: Occupational intakes of radionuclides: Part 1. Ann ICRP 44(2):5–188. https://doi.org/10.1177/0146645315577539
International Commission on Radiological Protection (ICRP) (2002) Guide for the practical application of the ICRP human respiratory tract model. Ann ICRP 32(1-2):17–28 Pergamon, Oxford
Jeong SH, Jang JH, Lee YB (2022) Drug delivery to the brain via the nasal route of administration: exploration of key targets and major consideration factors. J Pharm Investig 24:1–34. https://doi.org/10.1007/s40005-022-00589-5
Kirchmair J, Göller AH, Lang D, Kunze J, Testa B, Wilson ID, Glen RC, Schneider G (2015) Predicting drug metabolism: experiment and/or computation? Nat Rev Drug Discov 14:387–404
Koblinger L, Hofmann W (1990) Monte Carlo modeling of aerosol deposition in human lungs. Part I: simulation of particle transport in a stochastic lung structure. J Aerosol Sci 21:661–674
Kolanjiyil AV, Kleinstreuer C (2013) Nanoparticle mass transfer from lung airways to systemic regions—Part I: whole-lung aerosol dynamics. J Biomech Eng 135:121003
Kolanjiyil AV, Kleinstreuer C, Sadikot RT (2017) Computationally efficient analysis of particle transport and deposition in a human whole-lung-airway model. Part II: Dry powder inhaler application. Comput Biol Med 84:247–253
Kolli AR, Kuczaj AK, Martin F, Hayes AW, Peitsch MC, Hoeng J (2019) Bridging inhaled aerosol dosimetry to physiologically based pharmacokinetic modeling for toxicological assessment: nicotine delivery systems and beyond. Crit Rev Toxicol 49:725–741
Koullapis PG, Kassinos SC, Bivolarova MP, Melikov AK (2016) Particle deposition in a realistic geometry of the human conducting airways: effects of inlet velocity profile, inhalation flowrate and electrostatic charge. J Biomech 49(11):2201–2212
Koullapis PG, Nicolaou L, Kassinos SC (2018) In silico assessment of mouth-throat effects on regional deposition in the upper tracheobronchial airways. J Aerosol Sci 117:164–188
Koullapis PG, Stylianou FS, Sznitman J, Olsson B, Kassinos SC (2020) Towards whole-lung simulations of aerosol deposition: a model of the deep lung. J Aerosol Sci 144:105541
Koullapis P, Stylianou F, Lin C-L, Kassinos S, Sznitman J (2021) In silico methods to model dose deposition. Chapter 7. In: Kassinos S, Backman P, Conway J, Hickey AJ (eds) In inhaled medicines: optimizing development through integration of in silico, in vitro and in vivo approaches. Academic Press
Kuprat AP, Jalali M, Jan T, Corley RA, Asgharian B, Price O et al (2021) Efficient bi-directional coupling of 3D computational fluid-particle dynamics and 1D multiple path particle dosimetry lung models for multiscale modeling of aerosol dosimetry. J Aerosol Sci 151:105647. https://doi.org/10.1016/j.jaerosci.2020.105647
Lambert AR, Oshaughnessy PT, Tawhai MH, Hoffman EA, Lin C (2011) Regional deposition of particles in an image-based airway model: large-eddy simulation and left-right lung ventilation asymmetry. Aerosol Sci Technol 45(1):11–25
Lazaridis M (2011) First principles of meteorology and air pollution. In: Environmental pollution. Springer, p 362
Lazaridis M, Broday DM, Hov Ø, Georgopoulos PG (2001) Integrated exposure and dose modeling and analysis System. 3. Deposition of inhaled particles in the human respiratory tract. Environ Sci Technol 35:3727–3734
Lazaridis M, Aleksandropoulou V, Hanssen JE, Dye C, Eleftheriadis K, Katsivela E (2008) Inorganic and carbonaceous components in indoor/outdoor particulate matter in two residential houses in Oslo, Norway. J Air Waste Manage Assoc 58(3):346–356
Lin CL, Hoffman EA, Kassinos S (2021) Machine learning and silico methods. In: Kassinos S, Backman P, Conway J, Hickey AJ (eds) In inhaled medicines: optimizing development through integration of In Silico, In Vitro and In Vivo approaches. Academic Press
Löndahl J, Massling A, Swietlicki E, Bräuner EV, Ketzel M, Pagels J, Loft S (2009) Experimentally determined human respiratory tract deposition of airborne particles at a busy street. Environ Sci Technol 43:4659–4664
Longest PW, Tian G, Khajeh-Hosseini-Dalasm N, Hindle M (2016) Validating whole-airway CFD predictions of DPI aerosol deposition at multiple flow rates. J Aerosol Med Pulm Drug Deliv 29:461–481
Martonen TB, Rosati JA, Isaacs KK (2005) Modeling deposition of inhaled particles. In: Ruzer LS, Harley NH (eds) In the CRC Aerosols Hanbook. CRC Press, pp 129–172
Mellat M, Borojeni AAM, Sahebkar A, Ghanei M (2017) Adapting the ICRP model to predict regional deposition of the pharmaceutical aerosols inhaled through DPIs and nebulizers. J Drug Deliv Sci Technol 37:81–87
Miller FJ, Asgharian B, Schroeter JD, Price OT (2016) Improvements and additions to the multiple path particle dosimetry model. J Aerosol Sci 99:14–26. https://doi.org/10.1016/j.jaerosci.2016.01.018
Mitsakou C, Helmis C, Housiadas C (2005) Eulerian modeling of lung deposition with sectional representation of aerosol dynamics. J Aerosol Sci 36:75–94
Nemmar A, Hoet PM, Vanquickenborne B, Dinsdale D, Thomeer M, Hoylaerts MF, Vanbilloen H, Mortelmans L, Nemery B (2002) Passage of inhaled particles into the blood circulation in humans. Circulation 105:411–414
Nixon W, Egan MJ (1987) Modeling study of regional deposition of inhaled aerosols with special reference to effects of ventilation asymmetry. J Aerosol Sci 18(5):563–579
Oberdorster G, Ferin J, Lehnert BE (1994) Correlation between particle size, in vivo particle persistence, and lung injury. Environ Health Perspect 102(Suppl 5):173–179
O’Flaherty EJ, Kerger BD, Hays SM, Paustenbach DJ (2001) A physiologically based model for the ingestion of chromium (III) and Chromium (VI) by Humans. Toxicol Sci 60:196–213
Paquet F, Bailey MR, Leggett RW, Lipsztein J, Fell TP, Smith T, Nosske D, Eckerman KF, Berkovski V, Ansoborlo E, Giussani A (2016) ICRP Publication 134: Occupational intakes of radionuclides: Part 2. Ann ICRP 45(3/4):7–349
Perez-Crespo L, Kusters MSW, Lopez-Vicente M, Lubczynska M, Foraster M, White T, Hoek G, Tiemeier H, Muetzel RL, Guxens M (2022) Exposure to traffic-related air pollution and noise during pregnancy and childhood, and functional brain connectivity in preadolescents. Environ Int 164:107275
Petters MD, Kreidenweis SM (2007) A single parameter representation of hygroscopic growth and cloud condensation nucleus activity. Atmos Chem Phys 7:1961–1971
Phalen RF, Hoover MD, Oldham MJ, Jarabek AM (2021) Inhaled aerosol dosimetry: research-related needs and recommendations. J Aerosol Sci 155:105755
Pope CA, Ezzati M, Dockery DW (2009) Fine particulate air pollution and life expectancy in the United States. New Eng J Med 360:376–386
Rangaraj N, Pailla SR, Sampathi S (2019) Insight into pulmonary drug delivery: mechanism of drug deposition to device characterization and regulatory requirements. Pulm Pharmacol Ther 54:1–21
Reddy MB, Yang RSH, Clewell HJ, Andersen ME (2005) Physiologically based pharmacokinetic (PBPK) modeling. In: Science and application. John Wiley & Sons, Inc., Hoboken, New Jersey
Robinson PD, Salimi F, Cowie CT, Clifford S, King GG, Thamrin C, Hardaker K, Mazaheri M, Morawska L, Toelle BG, Marks GB (2022) Ultrafine particle exposure and biomarkers of effect on small airways in children. Environ Res 214:113860
Schlesinger R (1988) Biological disposition of airborne particles: basic principles and application to vehicular emissions. In: Air Pollution, the Automobile, and Public Health. National Academy Press, Washington DC, p 704
Schroeter JD, Nong A, Yoon M, Taylor MD, Dorman DC, Andersen ME, Clewell HJ III (2011) Analysis of manganese tracer kinetics and target tissue dosimetry in monkeys and humans with multi-route physiologically based pharmacokinetic models. Toxicol Sci 120:481–498
Schwartz J (2004) Air pollution and children's health. Pediatrics 113:1037–1043
Schwartz J, Neas LM (2000) Fine particles are more strongly associated than coarse particles with acute respiratory health effects in schoolchildren. Epidemiol 11:6–10
Sharma M, Maheshwari M, Morisawa S (2005) Dietary and inhalation intake of lead and estimation of blood lead levels in adults and children in Kanpur, India. Risk Anal 25:1573–1588
Smith JRH, Bailey MR, Etherington G, Shutt AL, Youngman MJ (2008) Effect of particle size on slow particle clearance from the bronchial tree. Exp Lung Res 34:287–312
Sonnenberg AH, Herrmann J, Grinstaff MW, Suki B (2020) A Markov chain model of particle deposition in the lung. Sci Rep 10:13573. https://doi.org/10.1038/s41598-020-70171-2
Stapleton K, Guentsch E, Hoskinson MK, Finlay W (2000) On the suitability of k-ε turbulence modeling for aerosol deposition in the mouth and throat: a comparison with experiment. J Aerosol Sci 31:739–749
Sturm R, Hofmann W (2009) A theoretical approach to the deposition and clearance of fibers with variable size in the human respiratory tract. J Hazard Mater 170:210–218
Tay YX, Kothan S, Kada S, Cai S, Lai CWK (2021) Challenges and optimization strategies in medical imaging service delivery during COVID-19. World J Radiol 13(5):102–121
Tehler U, Fransson R, Thorn H, Franek F, Westergren J (2018) Lung-Sim: a physiologically based biopharmaceutical prediction tool. J Aerosol Med Pulm Drug Deliv
Trumble BC, Finch CE (2019) The exposome in human evolution: from dust to diesel. Q Rev Biol 94:333–394
USEPA (2018). Integrated science assessment for particulate matter EPA/600/R-18/179.
USEPA (2019). Guidelines for human exposure assessment. EPA/100/B-19/001
Vu TV, Delgado-Saborit JM, Harrison RM (2015) A review of hygroscopic growth factors of submicron aerosols from different sources and its implication for calculation of lung deposition efficiency of ambient aerosols. Air Qual Atmos Health 8:429–440
Vu TV, Zauli-Sajani S, Poluzzi V, Harrison RM (2018) Factors controlling the lung dose of road traffic-generated sub-micrometre aerosols from outdoor to indoor environments. Air Qual Atmos Health 11:615–625
Weibel ER (1963) Morphometry of the human lung. Academic Press, New York
Weibel ER (1991) Fractal geometry—a design principle for living organisms. Am J Physiol 261(6):L361–L369
Winkler-Heil R, Ferron G, Hofmann W (2014) Calculation of hygroscopic particle deposition in the human lung. Inhal Toxicol 26(3):193–206
World Health Organization (WHO) (2021) WHO air quality guidelines for particulate matter, ozone, nitrogen dioxide and sulfur dioxide. WHO Press, Geneva, Switzerland
Yeh HC, Schum GM (1980) Models of human lung airways and their application to inhaled particle deposition. Bull Math Biol 42:461–480
Youn JS, Csavina J, Rine KP, Shingler T, Taylor MP, Sáez AE, Betterton EA, Sorooshian A (2016) Hygroscopic properties and respiratory system deposition behavior of particulate matter emitted by mining and smelting operations. Environ Sci Technol 50:11706–11713
Yu CP, Diu CK (1982) A comparative study of aerosol deposition in different lung models. Am Ind Hyg Assoc J 43:54–65
Zhang Z, Kleinstreuer C (2001) Effect of particle inlet distributions on deposition in a triple bifurcation lung airway model. J Aerosol Med 14:13–29
Zhang Z, Kleinstreuer C, Kim CS (2002a) Cyclic micron-size particle inhalation and deposition in a triple bifurcation lung airway model. J Aerosol Sci 33:257–281
Zhang Z, Kleinstreuer C, Kim CS (2002b) Aerosol deposition efficiencies and upstream release positions for different inhalation modes in the upper bronchial airway model. Aerosol Sci Technol 36:828–844
Acknowledgements
The author gratefully acknowledges the financial support of the European Commission under grant INCHILDHEALTH, which was funded from the H2020 RTD Framework Programme of the European Union (Grant agreement no: 101056883).
Funding
Open access funding provided by HEAL-Link Greece. The author gratefully acknowledges the financial support of the European Commission under grant INCHILDHEALTH, which was funded from the H2020 RTD Framework Programme of the European Union (Grant agreement no: 101056883).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
All authors consent for the publication of the manuscript and the materials incorporated.
Competing interests
The author declares no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lazaridis, M. Modelling approaches to particle deposition and clearance in the human respiratory tract. Air Qual Atmos Health 16, 1989–2002 (2023). https://doi.org/10.1007/s11869-023-01386-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11869-023-01386-1