Abstract
Background
Interstitial lung disease (ILD) is a common pulmonary manifestation of Sjögren’s syndrome (SjS) and associated with an increased risk of death. Early detection and treatment of ILDs and knowing the risk factors are very important for prognosis in rheumatic diseases.
Aims
This study was performed to determine ILD and associated factors in patients with SjS.
Methods
Four hundred three SjS patients were evaluated in this cross-sectional cohort study. Clinical, laboratory, serological, and imaging features were compared of patients with and without pulmonary involvement. Logistic regression analyses were used to identify risk factors for lung involvement and to identify independent risk factors.
Results
Thirty-five (8.7%) of SjS patients had ILD and 368 (91.3%) had no ILD. The presence of Raynaud’s phenomenon was significantly more common in ILD. The geriatric age group over the age of 65 years (OR 8198; 95% CI 3788–17,742; p < 0.001), Raynaud’s phenomenon (OR 17,852; 95% CI 6155–51,779; p < 0.001), and smoking (OR 3598; 95% CI 1495–8657; p = 0.003) were risk factors to be associated for ILD in the multivariable analysis. The most common abnormality was non-specific interstitial pneumonia in 20 patients (57.1%) and usual interstitial pneumonia in 15 (42.9%) patients.
Conclusions
The distribution of male patients compared to female patients was higher in patients with lung involvement than in patients without lung involvement. This may be related to older age, higher smoking rate, and longer nicotine consumption in men. Age, smoking, and severity of lung involvement are more important than inflammation status and autoantibodies for prognosis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Sjögren’s syndrome (SjS) is a chronic multisystem inflammatory autoimmune disease characterized by dry eyes, mouth, and lymphocytic infiltration of the lacrimal and salivary glands [1, 2]. Glandular and extra-glandular involvement may be observed in patients [3]. As glandular findings, dry eyes and/or mouth are the most common symptoms of SS patients. Moreover, itching due to dry skin, vulvovaginal dryness, dyspareunia, difficulty in swallowing, persistent dry cough, hoarseness, and nasal dryness and crusting may occur. Approximately one-third of the patients have extra glandular symptoms such as synovitis, vasculitis, and skin, renal, neurological, and lung involvement. Skin lesions such as annular erythema, urticarial vasculitis, tubulointerstitial changes in the kidney, and neurological findings such as peripheral neuropathy may be observed [4, 5].
Interstitial lung disease (ILD) is the most common pulmonary involvement in SjS and has been reported in 10–20% of patients [6]. Ten to fifteen percent of patients may develop ILD years before the onset of SjS. The most common pattern of ILD is non-specific interstitial pneumonia (NSIP), followed by usual interstitial pneumonia (UIP) and organizing pneumonia (OP) [7]. ILD leads to poor prognosis and life-threatening complications, including respiratory failure and secondary pulmonary hypertension [6, 7]. Hypergammaglobulinemia, lymphopenia, rheumatoid factor (RF), anti-Ro/SS-A and anti-La/SS-B antibodies, smoking, male gender, advanced age, and longer disease duration are defined as risk factors for lung disease in SjS [2, 7, 8]. Considering that patients with primary SjS-ILD may have different clinical courses, a change in pulmonary function and lung HRCT findings are likely to predict response to treatment and prognosis.
In this study, we aimed to evaluate the relationship between clinical, laboratory, and serological findings and lung involvement in patients with SjS.
Methods
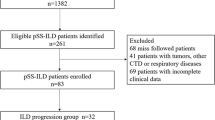
This cross-sectional study included 403 patients aged 18 years or above, fulfilling the 2016 ACR/EULAR Classification Criteria for SjS and attending the rheumatology outpatient clinic of our faculty from January 2016 and July 2022 over a period of 6 months [9]. Patients with infection, malignancy, autoimmune rheumatic diseases other than SjS, and comorbidities that could cause respiratory symptoms were not included in our study. The cross-sectional retrospective cohort study was approved by the Faculty of Medicine Ethics Committee (approval number: 216/09).
Demographic characteristics, clinical features, and serological profiles of the patients were evaluated from electronic health records in the hospital database, retrospectively. Dry mouth, dry eyes, Raynaud’s phenomenon, arthritis/arthralgia, cough, sputum, dyspnea, Schirmer test, lymphopenia, erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), anti-nuclear antibody (ANA), anti-SS-A/Ro52, and rheumatoid factor (Rf) were noted. Findings of lung involvement such as ground glass opacities, basal and peripheral reticular opacities, honeycomb appearance, and traction bronchiectasis were defined by high-resolution computed tomography (HRCT). The forced vital capacity (FVC), forced expiratory volume in one second (FEV1), FEV1/FVC, forced expiratory flow (FEF 25–50-75), and carbon monoxide diffusion capacity (DLCO) were recorded.
Statistical Package for the Social Sciences (SPSS) version 21 (IBM Corporation, Armonk, NY) was used for data analysis. Descriptive statistics were given as frequency, median (25–75p) and mean ± standard deviation. The Kolmogorov–Smirnov test was used to decide whether a variable had a normal distribution. The chi-square test and Fisher’s exact test were used to compare categorical variables. The Mann–Whitney U test was used to compare the means of two independent groups with normal distribution, and the T-test was used to compare the means of two continuous data groups with parametric analysis conditions. Logistic regression analysis was used to identify risk factors for lung involvement in SjS. A p value of < 0.05 was considered statistically significant.
Results
The mean age of 403 primary SjS patients (25 males, 378 females) included in the study was 55.4 ± 13.3 years. The female/male ratio was 15.1/1, and the mean disease duration was 34.5 ± 23.1 months. Of all patients, 252 (62.5%) had dry mouth, 289 (71.7%) dry eyes, 16 (4%) Raynaud’s phenomenon, and 189 (46.9%) arthralgia/arthritis. Schirmer’s test below 5 mm was detected in at least one eye of 349 (86.6%) patients. The presence of chronic sialoadenitis in salivary gland biopsy was detected in 215 (53.3%) patients. There were 18 (4.5%) patients with pulmonary hypertension detected by echocardiography. ANA positivity was 337 (83.6%), and Rf positivity was 79 (19.6%) in all patients. Anti-CCP positivity was not detected in the patients. Anti-SSA/anti-Ro-52 positivity was 68.6% in SjS patients with pulmonary involvement.
Patients with ILD were older (65.8 ± 9.2 vs 54.4 ± 13.2 years; p < 0.001) and had longer disease duration (35.9 ± 20.0 vs 33.7 ± 22.3 months; p > 0.05) compared to patients without ILD. Female rates were lower in SjS patients with ILD than without ILD (71.4% vs 94.6%). 71.4% of patients with ILD and 23.4% of patients without ILD were over 65 years of age (p < 0.001). The distribution of male patients compared to female patients was higher in patients with lung involvement than in patients without lung involvement. It may be the more frequent proportion of smokers in men and due to higher age longer nicotine consumption. Among the respiratory system findings of SjS patients with ILD, cough was detected in 30 (85.7%), sputum in 11 (31.4%), and shortness of breath in 25 (71.4%) patients. Raynaud’s phenomenon was statistically significantly higher in the group with ILD (p = 0.001) (Table 1).
Considering the laboratory and serological characteristics of the patients, sedimentation and CRP values in patients with lung involvement were 39 ± 17.7 mm/h and 2 mg/dl [2,3,4,5, 9], respectively. ANA was positive in 33 (94.3%) and Rf in 9 (25.7%) patients. The rates were 82.6% and 19% in patients without ILD, respectively. Anti-SSA/Ro-52 rates were 68.6% in patients with ILD and 56% in patients without ILD. Anti-CCP positivity was not detected in SjS patients. The presence of lymphopenia was present in 3 (8.6%) patients with ILD, and the Schirmer test was < 5 mm in 28 (80%) patients in at least one eye. The rate of smoking was significantly higher in patients with ILD compared to those without ILD. The demographic and clinical features of the patients with SjS are shown in Table 1.
The values of FVC, FEV1, FEV1/FVC, FEF25, FEF50, FEF75, and DLCO were evaluated in all patients with lung involvement. The mean of FEV1/FVC, FEF25, FEF50, FEF75, and DLCO was lower in ILD with UIP patent than NSIP pattern. The mean of FEV1 was 78.8 ± 18.1, FVC was 78.5 ± 17.9, and FEV1/FVC was 82.8 ± 8.9 (Table 2). In patients with lung involvement, radiological findings on HRCT showed ground glass opacities, traction bronchiectasis, honeycomb appearance, and fibrosis. The most common abnormality was ground glass opacities (57.1%). Those with lung involvement had 20 (57.1%) NSIP and 15 (42.9%) UIP. Lymphocytic interstitial pneumonia (LIP) and OP were not detected in any patient.
Multivariate logistic regression analysis was performed for risk factors that could predict the development of ILD in pSS patients. In the multivariable logistic regression analyses, the geriatric age group over the age of 65 years (OR 8198; 95% CI 3788–17,742; p < 0.001), Raynaud’s phenomenon (OR 17,852; 95% CI 6155–51,779; p < 0.001), and smoking (OR 3598; 95% CI 1495–8657; p = 0.003) were found as risk factors to be associated for ILD in SjS patients. Corticosteroids, azathioprine, mycophenolate mofetil, and rituximab were used as treatment options for SjS patients with ILD. All patients have corticosteroid use. Azathioprine was used in 17 (48.5%) of SjS patients with ILD, mycophenolate mofetil in 3 (8.5%), and rituximab in 2 (5.7%) patients. During follow-up, 5 SjS patients with ILD had lung progression evaluated with the change in ESSDAI. In these patients, immunosuppressive therapy (rituximab and mycophenolate mofetil) combined with high-dose steroids was given. No deaths occurred in the patients’ follow-up.
Discussion
SjS is an autoimmune rheumatic disease with systemic manifestations affecting many organs and systems. Lung involvement, defined by symptoms and pulmonary function test or radiographic abnormalities, occurs in approximately 10 to 20% of patients [10]. In our study, the rate of those with lung involvement was found to be 8.7% among all patients. The distribution of male patients compared to female patients was higher in patients with lung involvement than in patients without lung involvement (28.6% vs 4.1%; p < 0.001). Patients with ILD were older and had longer disease duration compared to patients without ILD.
Advanced age, smoking, ANA, RF, and higher CRP levels are potential risk factors for the development of ILD in SjS [2]. The frequency of ILD has increased in patients over 65 years of age [11]. ILD was found to be more likely to be male, older, and smokers compared to the group without ILD [12, 13]. In our study, the mean age of patients with ILD was 65.8 ± 9.2 years, and the number of smokers was 8 (22.9%). There were no significant differences in smoking in both groups. Smoking causes loss of airspace wall tissue in areas distant from macroscopic lesions and contributes to lung fibrosis by causing a net increase in collagen mass. Links between aging and lung fibrosis, including telomere wear, genomic variability, and epigenetic changes, have been reported [12]. Also, studies have found a relationship between Raynaud’s syndrome and lung involvement in SjS [7, 14]. In the multivariable logistic regression analyses, the geriatric age group over the age of 65 years, Raynaud’s phenomenon, and smoking were significantly found as risk factors to be associated with ILD in SjS patients.
A higher prevalence of extra-glandular symptoms was observed in patients with positive anti-Ro/La antibodies [15]. Anti-Ro52 antibodies are known as a risk factor for ILD in patients with SjS [16, 17]. Davidson et al. reported that pulmonary complications occur predominantly in patients with positive anti-SSA antibodies [18]. In a study, ANA positivity was found to be higher in the group with ILD [12,13,14]. The presence of lymphopenia was also reported as a potential risk factor for ILD [2, 7, 13]. In the studies performed, no significant difference was found between CRP, C3, and C4 levels between the groups with and without ILD [7, 12, 13]. In our study, the rate of ANA and anti-SSA/Ro52 positivity was found to be higher in the group with lung involvement. Cough, sputum, and shortness of breath are commonly reported symptoms in patients with SjS pulmonary involvement [7, 14]. The prevalence of cough is estimated to be between 41 and 50% and has a significant impact on quality of life [2]. In our study, cough was detected in 30 (85.7%), sputum in 11 (31.4%), and dyspnea in 23 (71.4%) SjS patients with ILD.
HRCT is considered a useful and sensitive method to detect lung abnormalities in patients with SjS [7]. The abnormalities are ground glass opacities, traction bronchiectasis, honeycomb appearance, and fibrosis. According to the HRCT pattern classification, NSIP was the most detected (30%) [17, 19]. In a study evaluating 201 patients, NSIP (n = 72, 45.5%), UIP (n = 16, 10.10%), and LIP (n = 13, 8.2%) were reported in primer SjS [12]. In HRCT, we found the NSIP pattern in 20 (57.1%) and the UIP pattern in 15 (42.9%) SjS patients. As a result of the development of ILD in patients with a diagnosis of primary SjS, deterioration in respiratory function capacities and a decrease in DLCO are observed [2]. In a study, the mean of FEV1 and FVC values was found to be significantly lower in patients with parenchymal lesions, and the severity of lung disease was correlated with a decrease in FVC and FEV1 [13]. In a study of 165 primary SjS with lung involvement, impairment in diffusion capacity was the most common and serious complication among pulmonary function tests. 48.6% of the patients had respiratory dysfunction; 21.57% had a restrictive disease pattern, and 19.61% had small airway dysfunction [17].
Conclusion
The distribution of male patients compared to female patients was higher in patients with lung involvement than in patients without lung involvement. This may be related to older age, higher smoking rate, and longer nicotine consumption in men. Age, smoking, and severity of lung involvement are more important than inflammation status and autoantibodies for prognosis.
References
Negrini S, Emmi G, Greco M et al (2022) Sjögren’s syndrome: a systemic autoimmune disease. Clin Exp Med 22(1):9–25. https://doi.org/10.1007/s10238-021-00728-6
Natalini JG, Johr C, Kreider M (2019) Pulmonary involvement in Sjögren syndrome. Clin Chest Med 40(3):531–544. https://doi.org/10.1016/j.ccm.2019.05.002
Chung A, Wilgus ML, Fishbein G, Lynch JP 3rd (2019) Pulmonary and bronchiolar involvement in Sjogren’s syndrome. Seminars in respiratory and critical care medicine 40(2):235–254. https://doi.org/10.1055/s-0039-1688448
André F, Böckle BC (2022) Sjögren’s syndrome. Journal der Deutschen Dermatologischen Gesellschaft = Journal of the German Society of Dermatology : JDDG 20(7):980–1002. https://doi.org/10.1111/ddg.14823
Vivino FB, Bunya VY, Massaro-Giordano G et al (2019) Sjogren’s syndrome: an update on disease pathogenesis, clinical manifestations and treatment. Clin immunol (Orlando, Fla.) 203:81–121. https://doi.org/10.1016/j.clim.2019.04.009
Manfredi A, Sebastiani M, Cerri S et al (2017) Prevalence and characterization of non-sicca onset primary Sjögren syndrome with interstitial lung involvement. Clin Rheumatol 36(6):1261–1268. https://doi.org/10.1007/s10067-017-3601-1
Lin W, Xin Z, Zhang J et al (2022) Interstitial lung disease in primary Sjögren’s syndrome. BMC Pulm Med 22(1):73. https://doi.org/10.1186/s12890-022-01868-5
He C, Chen Z, Liu S et al (2020) Prevalence and risk factors of interstitial lung disease in patients with primary Sjögren’s syndrome: a systematic review and meta-analysis. Int J Rheum Dis 23(8):1009–1018. https://doi.org/10.1111/1756-185X.13881
Shiboski CH, Shiboski SC, Seror R et al (2017) 2016 American College of Rheumatology/European League against rheumatism classification criteria for primary Sjögren’s syndrome: a consensus and data-driven methodology involving three international patient cohorts. Ann Rheum Dis 76(1):9–16. https://doi.org/10.1136/annrheumdis-2016-210571
Kreider M, Highland K (2014) Pulmonary involvement in Sjögren syndrome. Semin Respir Crit Care Med 35(2):255–264. https://doi.org/10.1055/s-0034-1371529
Lee KA, Choi W, Kim J et al (2021) Elderly-onset primary Sjögren’s syndrome focused on clinical and salivary gland ultrasonographic features. Jt Bone Spine 88(4):105132. https://doi.org/10.1016/j.jbspin.2021.105132
Wang Y, Hou Z, Qiu M, Ye Q (2018) Risk factors for primary Sjögren syndrome-associated interstitial lung disease. J Thorac Dis 10(4):2108–2117. https://doi.org/10.21037/jtd.2018.03.120
Yazisiz V, Arslan G, Ozbudak IH et al (2010) Lung involvement in patients with primary Sjögren’s syndrome: what are the predictors? Rheumatol Int 30(10):1317–1324. https://doi.org/10.1007/s00296-009-1152-8
Roca F, Dominique S, Schmidt J et al (2017) Interstitial lung disease in primary Sjögren’s syndrome. Autoimmun Rev 16(1):48–54. https://doi.org/10.1016/j.autrev.2016.09.017
Ramos-Casals M, Solans R, Rosas J et al (2008) Primary Sjögren syndrome in Spain: clinical and immunologic expression in 1010 patients. Medicine 87(4):210–219. https://doi.org/10.1097/MD.0b013e318181e6af
Kuwana M, Gil-Vila A, Selva-O’Callaghan A (2021) Role of autoantibodies in the diagnosis and prognosis of interstitial lung disease in autoimmune rheumatic disorders. Ther Adv Musculoskelet Dis 13:1759720X211032457. https://doi.org/10.1177/1759720X211032457
Gao H, Zhang XW, He J et al (2018) Prevalence, risk factors, and prognosis of interstitial lung disease in a large cohort of Chinese primary Sjögren syndrome patients: a case-control study. Medicine 97(24):e11003. https://doi.org/10.1097/MD.0000000000011003
Davidson BK, Kelly CA, Griffiths ID (2000) Ten year follow up of pulmonary function in patients with primary Sjögren’s syndrome. Ann Rheum Dis 59(9):709–712. https://doi.org/10.1136/ard.59.9.709
Kamiya Y, Fujisawa T, Kono M et al (2019) Prognostic factors for primary Sjögren’s syndrome-associated interstitial lung diseases. Respir Med 159:105811. https://doi.org/10.1016/j.rmed.2019.105811
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sargın, G., Baygin, H., Cildag, S. et al. Interstitial lung disease and associated factors in patients with Sjögren’s syndrome. Ir J Med Sci 193, 1385–1389 (2024). https://doi.org/10.1007/s11845-024-03629-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11845-024-03629-1