Abstract
Background
Overweight and obesity epidemic is still expanding, and it is affecting women of childbearing age. Multiple studies have shown unmatched results concerning the effect of body mass index (BMI) besides gestational weight gain (GWG) on pregnancy and neonatal outcomes. This study aims to determine the effect of each of the two anthropometric indicators: pre-gestational BMI and gestational weight gain on the course of pregnancy, and neonatal outcomes.
Methods
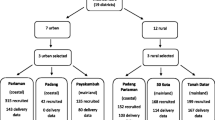
A retrospective study was conducted at Notre Dame de Secours University Hospital (CHU-NDS) Jbeil-Lebanon. The data was collected from the hospital archive. Out of 804 deliveries during 2020, 583 women were included after randomly choosing their files and eliminating those with exclusion criteria or incomplete data.
Results
Underweight/healthy BMI mothers had a higher chance of having low GWG (45.5%), vaginal delivery (51.3%), and a baby of appropriate size (78.6%) or small size for gestational age (10.4%). Obese women had a higher risk of excessive GWG (49.3%), delivery via C-section. (69.3%), and large for gestational age babies (26.7%). Mothers who had low GWG were at a higher risk of having babies of appropriate size (80.1%) or small size for gestational age (13.1%). Mothers who had high GWG had a higher risk of having baby boys (58.9%), large for their gestational age (26.1%), with hypoglycemia at birth (20.6%).
Conclusion
Both extremes of BMI and GWG are linked to adverse neonatal outcomes. This highlights the importance of weight monitoring even during pregnancy to prevent its negative impact on neonates.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Obesity has emerged as one of the twenty-first century’s most pressing public health challenges, for as being the second most common preventable cause of death worldwide [1]. The excess of fat accumulation defines obesity, and its prevention benefits children’s health and well-being immediately and throughout adulthood [2]. The World Health Organization defines obesity as a body mass index of 30 and above [3]. Between 1975 and 2016, the prevalence of obesity nearly tripled [3]. In the USA, the National Health and Nutrition Examination Survey (NHANES) found that for citizens aged 20 and over, 42.5% are obese, with 9% having severe obesity, and 31.1% being overweight [4].
There is a relationship between obesity and a dozen distinct forms of cancer [5,6,7], coronary artery disease [8, 9], stroke [1], chronic kidney disease [10, 11], impaired cognition [12, 13], diabetes mellitus type 2 [6, 14, 15], fatty liver disease [16], gallbladder disease [17, 18], hypertension [19], lung disorders [20, 21], metabolic syndrome [22, 23], musculoskeletal disorders [24, 25], psychosis [26], infertility [27,28,29,30], and mortality [3, 31].
In women of childbearing age, increased BMI was linked to a variety of adverse effects during pregnancy: gestational diabetes mellitus, hypertensive events, and a higher risk of maternal mortality. It also resulted in an increased risk of perinatal mortality, prenatal macrosomia, certain birth abnormalities, and childhood metabolic disorders [32]. Hence, it is necessary to evaluate the burden of high BMI among pregnant women, given its impact on mothers and newborns. Low or high pre-pregnancy BMI and insufficient or excessive weight gain during pregnancy have been related to a high risk of poor consequences on the newborn. Large for gestational age newborns, preterm birth, and neonatal mortality have been linked to overweight and obese BMI and high gestational weight gain, whereas preterm birth and small for gestational age infants have been linked to underweight BMI and insufficient weight gain during pregnancy [33].
The Eastern Mediterranean area is not excluded, as data show that obesity is on the rise in the Arab world. There is an expansion in the proportion of young Lebanese women with a high BMI, exceeding the stated global average of 0.5 kg/m2 each decade in women [34]. Since multiple studies demonstrated unmatched results, and since there are few studies done in the region concerning this particular topic, it is critical to investigate the effects of this increased weight gain on mothers and newborns in a sample of Lebanese women. Hence, the two objectives of this study are to determine the effect of pre-gestational BMI and gestational weight gain on the course of pregnancy and neonatal outcomes.
Methods
Study design
A retrospective study was conducted at Notre Dame des Secours University Hospital Center (CHU-NDS), Jbeil-Lebanon, to determine the effect of each of the two anthropometric indicators: pre-gestational BMI and gestational weight gain first, on the course of pregnancy, and second on neonatal outcomes. Data was collected from the CHU-NDS Hospital archive (electronic and paper-based), and out of 804 deliveries during the year 2020, 583 women were included in the study after eliminating those with missing data and those with the following exclusion criteria: (1) Perinatal mortality, (2) twin pregnancy, (3) in vitro fertilization, (4) weight loss during pregnancy, and (5) COVID-19 infection during pregnancy based on a positive PCR.
Minimal sample size calculation
Based on the fact that BMI is associated with higher odds of cesarean delivery (OR = 1.74) and a 22.2% frequency of cesarean delivery in underweight women according to a study published by Ratnasiri et al. [49], the Epi-info software v 7.2 calculated a minimal sample size of 520 to have enough statistical power with 5% alpha error and 80% power.
Questionnaire and variables
The following variables were studied
For neonatal, the following variables were collected: gender, size for gestational age (based on the Fenton curve of 2013 [35]), preterm birth [36], Apgar score at 1 min and 5 min [37], icterus, the need for ICU admission (NICU), and glycemia at birth (according to the American Academy of Pediatrics [38]).
For pregnant women, information about age, pre-pregnancy BMI [39, 40], level of education, employment, chronic illnesses, consumption of tobacco, alcohol, and coffee during pregnancy were collected. Information regarding the course of pregnancy (the threat of premature labor, vaginal bleed, pregnancy-induced HTN [41], preeclampsia and eclampsia [42], gestational diabetes based on the WHO criteria [22], total gestational weight gain [43], gestational weeks, type of delivery) were also gathered.
Statistical analysis
The Statistical Package for Social Science (SPSS) version 22 was used to analyze the data. The Chi-square test was used to compare two categorical variables, whereas the ANOVA test was used to compare three means. P value < 0.05 was considered statistically significant.
Results
The results of the bivariate analysis of factors associated with the pre-pregnancy BMI categories are summarized in Table 1. Underweight/healthy BMI mothers had a higher chance of having a university level of education, gaining weight less than it is recommended during pregnancy, having a vaginal delivery, and delivering a baby with appropriate size or small size for his gestational age. Overweight mothers had a higher chance of having an appropriate weight gain during pregnancy. Obese mothers had a higher chance of having a secondary level of education or less, gaining weight more than is recommended, delivering via C-section, and having large for gestational age babies.
The results of the bivariate analysis of factors associated with the total weight gain during pregnancy categories are summarized in Table 2. Mothers who had deficient GWG had a higher chance of not having chronic illnesses and having babies of appropriate size or small size for gestational age. Those who had appropriate GWG had a higher chance of not smoking during gestation, having baby girls, and having infants with normal glycemia at birth. Mothers who had excessive GWG had a higher chance of having chronic illnesses, smoking tobacco during pregnancy, and having baby boys who are large for their gestational age with hypoglycemia at birth.
Discussion
Pre-pregnancy BMI association with maternal factors and pregnancy outcomes
In our study, underweight/healthy BMI mothers had a higher chance of having a university level of education, gaining weight less than it is recommended during pregnancy, and having a vaginal delivery. Overweight mothers had a higher chance of having an appropriate weight gain during pregnancy. Obese mothers had a higher chance of having a secondary level of education or less, gaining weight more than it is recommended, and delivering their baby via C-section.
Concerning education level, Sun et al. in Korea demonstrated that women who are uneducated or who have any education degree until high school were risk factors to keep normal pre-pregnancy BMI compared with those who have a college or a university degree [44]. The discordance with our results may be linked to our small sample size, the ethnicity of the pregnant women (583 Lebanese vs 3172 Chinese women in this study), and the diet of each country that is mainly based on rice and wheat in China, in contrast to the healthy Mediterranean diet that is based on vegetables, fruits, whole beans, grains, and dairy products. In contrast, our results are consistent with those of Wahabi et al. with a higher percentage of high BMI among illiterate women [45]. Hermann et al. studied in a multi-center prospective cohort study the relation of education with BMI and waist circumference in 10 countries [46]. They concluded that in comparison to women with the lowest education level, those with university education had a 2.1 kg/m2 lower BMI due to the lower energy intake from food and sports practice.
Concerning GWG, Magdalena Nowak et al. in Poland demonstrated that there is no association between insufficient GWG and low or normal pre-pregnancy BMI [47]. The difference with our results may be due to the higher percentage of underweight women; 43 out of 474 = 9% and normal weight women; 313 out of 474 = 66% in this study compared to ours; and 22 out of 583 = 4.8% and 323 out of 583 = 55.4%, respectively. In addition, it may be because the previously named study was done in Poland, a country where the underweight/normal BMI women are not living in a low-income country that warrants them access to healthcare and a healthy diet. Thus, during their pregnancy, they had a medical follow-up besides a healthy and rich diet to gain a recommended weight. In addition, this same study besides other studies concluded that high BMI, whether overweight or obese, correlates with excessive GWG [34, 47, 48]. These results are incompatible with ours; it may be due to the exclusion of women with chronic illnesses in the previously named study, whereas in our study, they were included. In addition, probably our overweight participants have chronic illnesses (such as polycystic ovarian syndrome, hypothyroidism, and diabetes) that promote them to consult a dietitian during their pregnancy to track their gestational weight gain. Besides the tendency of obese women to consume high caloric food, binge eating, not exercising, and according to the IOM recommendations, this category has the lowest gestational weight gain cut-offs compared to other BMI categories.
Concerning the delivery type, our results are consistent with Ratnasiri et al. [49], Yesilçiçek Çalik et al. [50], Choi et al. [51], and Wang et al. [52] who demonstrated that the rate of cesarean delivery is lower in underweight and normal weight mothers than in those overweight or obese. This can be explained by the fact that high BMI mothers have criteria of metabolic syndrome like hyperinsulinemia due to insulin resistance and resultant hyperglycemia. In addition, as maternal glucose will cross the placenta, the fetus will have hyperinsulinemia that will store glucose as fat. As a result, we will have a high risk of fetal macrosomia, which increases the need for cesarean delivery.
Pre-pregnancy BMI association with neonatal outcomes
In our study, we found that being underweight/healthy BMI is associated with a higher risk of delivering a baby with appropriate size or small size for gestational age. Obese mothers had a higher chance of having babies large for gestational age. Liu et al. [53] besides another study done in California by Ratnasiri et al. [49] revealed that decreased maternal BMI resulted in babies being small for their gestational age, and increased maternal BMI resulted in babies being large for their gestational age. The relation between small for gestational age neonates and underweight mothers can be explained by the baby being constitutionally small; it may be related to a family history of being small for their gestational age, thus having a small weight and/or height. High BMI mothers have peripheral insulin resistance that results in hyperglycemia leading to large for his gestational age neonates by the mechanism previously explained. In contrast, Rong Peng et al. concluded that there is no relation between BMI and high neonatal weight [54]. The discordance with our results can be explained by the fact that it was a case–control study where the cases were the mothers who delivered normal birth weight babies and the control group was formed of the mothers who delivered high birth weight infants (> 4 kg). In contrast, in our study, we used the Fenton curve where the babies that are large for their gestational age had a larger weight and/or height relatively according to their gestational age.
Total gestational weight gain association with maternal factors and pregnancy outcomes
We found that having a low GWG is associated with a higher chance of not having chronic illnesses. A high GWG is associated with a higher risk of having chronic illnesses and smoking tobacco during gestation.
Many revised articles from the literature have excluded from their study women with chronic diseases (MINA cohort study in Qatar and Lebanon [55], prospective cohort study in Poland [56], a study conducted by Magdalena Nowak et al. [47]). In our study, we did not exclude chronic illnesses, and we found a link with GWG. It can be explained by the fact that chronic illnesses cause chronic inflammation and elevation of stress hormone which is cortisol, resulting in excessive weight gain.
Concerning smoking, Kominiarek et al.’s results are coherent with ours, and it was found that the lowest percentage of women who smoked is among those who had adequate GWG over 25 hospitals in the USA [57]. In contrast, a MINA cohort study done in Lebanon and Qatar concluded that smoking during pregnancy did not affect GWG [55]. The results are inconsistent with ours, and many studies have shown that smoking interferes with our metabolism by promoting the burn of calories proportionally to the number of cigarettes. Hence, quitting smoking will result in a rebound effect leading to weight gain.
Total weight gain during pregnancy association with neonatal outcomes
We found that mothers who had deficient GWG had a higher chance of having babies of appropriate size or small size for gestational age. Mothers who had appropriate GWG had a higher chance of having baby girls and having infants with normal glycemia at birth. Those who had excessive GWG had a higher chance of having baby boys who are large for their gestational age with hypoglycemia at birth.
Concerning the female gender relation with GWG, it was not discussed in the articles reviewed in the literature. This association can be explained by the fact that adequate and healthy caloric intake leads to appropriate weight gain. Besides that on the Fenton curve, the female neonate cut-offs concerning the weight and the height are lower than those of baby boys. In contrast, concerning the male gender, a retrospective cohort study done in Turkey found no relationship between male gender and GWG [58]. The discordance in the results may be due to the lower percentage of women with excessive GWG in the Turkish study (80 out of 572) versus our study (180 out of 583) where they are better represented. Besides, it may be linked to the fact that boys have higher cut-offs of weight and size compared to girls on the Fenton curve, which can explain the higher gestational weight gain.
Concerning the babies’ size for their gestational age, Yin Sun et al. demonstrated that high BMI and excessive weight gain during gestation were related to large for gestational age infants [44]. A MINA cohort study done in Lebanon and Qatar demonstrated that excessive weight gain during gestation was linked to having babies large for their gestational age [55]. Kominiarek et al. results are also coherent with the previously cited studies with macrosomia in the case of high GWG [48]. These results can be explained by the excessive caloric intake that can lead to gestational diabetes leading to maternal hyperglycemia, neonatal hyperinsulinemia, and hence excessive neonatal fat deposition. In addition, low gestational weight gain besides low maternal caloric intake and poor protein diet is positively correlated with small for gestational age babies.
Concerning neonatal glycemia, Kominiarek et al. concluded that adequate GWG is associated with normal neonatal glycemia, whereas excessive gestational weight gain is associated with neonatal hypoglycemia [48]. Besides, Stotland et al. concluded that excessive GWG was associated with a higher risk of hypoglycemia [59]. This is consistent with our results; hence, being on the right track, gaining weight adequately means the healthy consumption of calories. This will decrease the risk of the development of gestational diabetes. Hence, the mother will have normal glycemia, besides a normal insulin production, and a peripheral insulin sensitivity warranting high and excessive maternal glucose concentration to cross the placenta. Therefore, the baby will have normal glucose concentration, resulting in a normal level of insulin production. Mothers with high excessive weight gain during pregnancy will lead to neonatal hyperinsulinemia as previously explained. After birth, the neonate still has an excessive insulin release by the pancreas despite that he is not receiving anymore excessive maternal glucose, which will result in hypoglycemia.
Limitations
The sample was collected from one center in Jbeil area and maybe not be representative on a national level. Besides, it is a retrospective study, instead of being a prospective study that will follow the pregnant patients from the beginning of their pregnancy until the date of their delivery, to record the data concerning the neonates (Apgar score, the reason of NICU admission), gestation course, and the pregnant women (screening of gestational diabetes and gestational hypertension, recording BMI and GWG, the causes of cesarean delivery). Moreover, no data about medications was collected.
Despite these limitations, our study has multiple strengths. We studied the relation between pre-pregnancy BMI and GWG, besides the effect of each one of them on the pregnancy and the neonate. Two previous studies were indeed done in Beirut, but our study concentrated on both pregnancy course and neonatal variables, and we relied on the Fenton curve to analyze the gestational age of neonates. In addition, we studied the threat of premature labor, vaginal bleed, neonatal icterus, and the consumption of alcohol and coffee; the relation of these variables with pre-pregnancy BMI and GWG was not discussed in the articles reviewed for the literature review, besides the relations of neonatal glycemia at birth with pre-pregnancy BMI and female gender with GWG.
Conclusion
In our study, we found that there is a significant association between the different pre-pregnancy BMI classes and maternal education, total gestational weight gain, type of delivery, and size of the baby. Moreover, we found that there is a significant association between total GWG and maternal chronic illnesses, tobacco smoking during pregnancy, gender of the newborn, size of the baby, and neonatal glycemia at birth. Hopefully, the results of this study will be helpful in the gynecology and pediatrics medical fields. By insisting on the importance of maintaining the BMI range within normal values, many pregnancies are unplanned; thus, this study also provides the necessity of weight gain monitoring during gestation to prevent negative neonatal and delivery outcomes.
Data availability
The datasets generated and/or analyzed during the current study are not publicly available due to restrictions from the ethics committee but are available from the corresponding author on reasonable request.
Abbreviations
- BMI:
-
Body mass index
- GWG:
-
Gestational weight gain
- HTN:
-
Hypertension
- NICU:
-
Neonatal intensive care unit
References
Quiñones-Ossa GA, Lobo C, Garcia-Ballestas E et al (2021) Obesity and stroke: does the paradox apply for stroke? Neurointervention 16(1):9–19. https://doi.org/10.5469/neuroint.2020.00108
World Health Organization. Obesity and overweight. Available from: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight
World Health Organization. Obesity. Accessed February 28, 2022. https://www.who.int/health-topics/obesity#tab=tab_1
Ogden CL, Fryar CD, Martin CB et al (2020) Trends in obesity prevalence by race and Hispanic origin - 1999–2000 to 2017–2018. JAMA - J Am Med Assoc 324(12):1208–1210. https://doi.org/10.1001/jama.2020.14590
Obesity and Cancer | Harvard Medical School. Accessed February 28, 2022. https://hms.harvard.edu/news/obesity-cancer
Health risks | Obesity prevention source | Harvard T.H. Chan School of Public Health. Accessed February 28, 2022. https://www.hsph.harvard.edu/obesity-prevention-source/obesity-consequences/health-effects/
Obesity and cancer fact sheet - National Cancer Institute. Accessed February 28, 2022. https://www.cancer.gov/about-cancer/causes-prevention/risk/obesity/obesity-fact-sheet
Ades PA, Savage PD (2017) Obesity in coronary heart disease: an unaddressed behavioral risk factor. Prev Med (Baltim) 104:117–119. https://doi.org/10.1016/j.ypmed.2017.04.013
Neeland IJ (2021). Obesity and cardiovascular disease Published online. https://doi.org/10.1161/CIR.0000000000000973
Ejerblad E, Fored CM, Lindblad P, Fryzek J, McLaughlin JK, Nyren O (2006) Obesity and risk for chronic renal failure. J Am Soc Nephrol 17:1695–1702
Eknoyan G (2011) Obesity and chronic kidney disease. Nefrologia 31(4):397–403. https://doi.org/10.3265/NEFROLOGIA.PRE2011.MAY.10963
Wang C, Chan JSY, Ren L (2016) Yan JH 2473081:2016
Obesity associated with a higher risk for dementia, new study finds | National Institute on Aging. Accessed February 28, 2022. https://www.nia.nih.gov/news/obesity-associated-higher-risk-dementia-new-study-finds
CDC (2020) National Diabetes Statistics Report 2020. Estimates of diabetes and its burden in the United States
Centers for Disease Control and Prevention. Diabetes and Obesity Maps. Accessed February 28, 2022. https://www.cdc.gov/diabetes/data/center/slides.html
Polyzos SA, Kountouras J, Mantzoros CS (2019) Obesity and nonalcoholic fatty liver disease: from pathophysiology to therapeutics. Metabolism 92:82–97. https://doi.org/10.1016/J.METABOL.2018.11.014
Jeong SU, Lee SK (2012) Obesity and gallbladder diseases. Korean J Gastroenterol 59(1):27–34. https://doi.org/10.4166/kjg.2012.59.1.27
Everyday Health. The link between gallstones, obesity, and weight loss | Everyday Health. Accessed February 28, 2022. https://www.everydayhealth.com/gallbladder/symptoms/link-between-gallstones-obesity-weight-loss/
Jiang SZ, Lu W, Zong XF et al (2016) Obesity and hypertension. Exp Ther Med 12(4):2395–2399. https://doi.org/10.3892/etm.2016.3667
Brock JM, Billeter A, Müller-Stich BP, Herth F (2020) Obesity and the lung: what we know today. Respiration 99(10):856–866. https://doi.org/10.1159/000509735
Dixon AE, Peters U (2018) The effect of obesity on lung function. Expert Rev Respir Med 12(9):755–767. https://doi.org/10.1080/17476348.2018.1506331
Engin A (2017) The definition and prevalence of obesity and metabolic syndrome. Adv Exp Med Biol 960:1–17. https://doi.org/10.1007/978-3-319-48382-5_1
Al-Hamad D, Raman V (2017) Metabolic syndrome in children and adolescents. Transl Pediatr 6(4):397–407. https://doi.org/10.21037/tp.2017.10.02
Onyemaechi NOC, Anyanwu GE, Obikili EN et al (2016) Impact of overweight and obesity on the musculoskeletal system using lumbosacral angles. Patient Prefer Adherence 10:291–296. https://doi.org/10.2147/PPA.S90967
Wang C, Chan JSY, Ren L, Yan JH (2016) Obesity reduces cognitive and motor functions across the lifespan. Neural Plast. https://doi.org/10.1155/2016/2473081
Kassem M, Haddad C, Daccache C et al (2021) Factors associated with overweight and obesity in Lebanese male patients with schizophrenia. Perspect Psychiatr Care 57(3):1347–1355. https://doi.org/10.1111/ppc.12697
Giviziez CR, Sanchez EGM, Approbato MS et al (2016) Obesity and anovulatory infertility: a review. J Bras Reprod Assist 20(4):240–245. https://doi.org/10.5935/1518-0557.20160046
Jungheim ES, Travieso JL, Carson KR, Moley KH (2012) Obesity and reproductive function. Obstet Gynecol Clin North Am 39(4):479–493. https://doi.org/10.1016/j.ogc.2012.09.002
Katib A (2015) Mechanisms linking obesity to male infertility. Cent Eur J Urol 68(1):79–85. https://doi.org/10.5173/ceju.2015.01.435
Chambers TJ, Anderson RA (2015) The impact of obesity on male fertility. Hormones 14(4):563–568. https://doi.org/10.14310/HORM.2002.1621
Obesity - Our World in Data. Accessed February 28, 2022. https://ourworldindata.org/obesity
Chen C, Xu X, Yan Y (2018) Estimated global overweight and obesity burden in pregnant women based on panel data model. PLoS One 13(8). https://doi.org/10.1371/journal.pone.0202183
Dzakpasu S, Fahey J, Kirby RS et al (2015) Contribution of prepregnancy body mass index and gestational weight gain to adverse neonatal outcomes: population attributable fractions for Canada. BMC Pregnancy Childbirth 15(1). https://doi.org/10.1186/s12884-015-0452-0
Papazian T, Tayeh GA, Sibai D et al (2017) Impact of maternal body mass index and gestational weight gain on neonatal outcomes among healthy Middle-Eastern females. PLoS One 12(7). https://doi.org/10.1371/journal.pone.0181255
Growth parameters in neonates - pediatrics - MSD Manual Professional Edition. Accessed February 28, 2022. https://www.msdmanuals.com/professional/pediatrics/perinatal-problems/growth-parameters-in-neonates
Quinn JA, Munoz FM, Gonik B et al (2016) Preterm birth: case definition & guidelines for data collection, analysis, and presentation of immunisation safety data. Vaccine 34(49):6047–6056. https://doi.org/10.1016/j.vaccine.2016.03.045
Watterberg KL, Aucott S, Benitz WE et al (2015) The apgar score. Pediatrics 136(4):819–822. https://doi.org/10.1542/peds.2015-2651
Hosagasi NH, Aydin M, Zenciroglu A et al (2018) Incidence of hypoglycemia in newborns at risk and an audit of the 2011 American academy of pediatrics guideline for hypoglycemia. Pediatr Neonatol 59(4):368–374. https://doi.org/10.1016/j.pedneo.2017.11.009
Definitions, classification, and epidemiology of obesity. Accessed February 28, 2022. https://www.ncbi.nlm.nih.gov/books/NBK279167/?report=reader
Dwyer JT, Melanson KJ, Sriprachy-anunt U et al (2015) Dietary treatment of obesity. Endotext. Published online February 28, 2015. Accessed March 19, 2022. https://www.ncbi.nlm.nih.gov/books/NBK278991/
Braunthal S, Brateanu A (2019) Hypertension in pregnancy: pathophysiology and treatment. SAGE Open Med 7:205031211984370. https://doi.org/10.1177/2050312119843700
Preeclampsia: practice essentials, overview, pathophysiology. Accessed February 28, 2022. https://emedicine.medscape.com/article/1476919-overview
Gilmore LA, Redman LM (2015) Weight gain in pregnancy and application of the 2009 IOM guidelines: toward a uniform approach. Obesity 23(3):507–511. https://doi.org/10.1002/oby.20951
Sun Y, Shen Z, Zhan Y et al (2020) Effects of pre-pregnancy body mass index and gestational weight gain on maternal and infant complications. BMC Pregnancy Childbirth 20(1):1–13. https://doi.org/10.1186/s12884-020-03071-y
Wahabi H, Esmaeil S, Fayed A (2021) Maternal prepregnancy weight and pregnancy outcomes in Saudi women: Subgroup analysis from riyadh mother and baby cohort study (RAHMA). Biomed Res Int. https://doi.org/10.1155/2021/6655942
Hermann S, Rohrmann S, Linseisen J et al (2011) The association of education with body mass index and waist circumference in the EPIC-PANACEA study. Published online
Nowak M, Kalwa M, Oleksy P et al (2019) The relationship between pre-pregnancy BMI, gestational weight gain and neonatal birth weight: a retrospective cohort study. Ginekol Pol 90(1):50–54. https://doi.org/10.5603/GP.2019.0008
Lakhani CM, Tierney BT, Manrai AK et al (2017) Department, Kuschner 乳鼠心肌提取 HHS Public Access. Physiol Behav 176(3):139–148. https://doi.org/10.1097/AOG.0000000000002854
Ratnasiri AWG, Lee HC, Lakshminrusimha S et al (2019) Trends in maternal prepregnancy body mass index (BMI) and its association with birth and maternal outcomes in California, 2007–2016: a retrospective cohort study. PLoS One 14(9). https://doi.org/10.1371/journal.pone.0222458
Yeşilçiçek Çalik K, Korkmaz Yildiz N, Erkaya R (2018) Effects of gestational weight gain and body mass index on obstetric outcome. Saudi J Biol Sci 25(6):1085–1089. https://doi.org/10.1016/j.sjbs.2018.02.014
Choi S-K, Park I-Y, Shin J (2011) The effects of pre-pregnancy body mass index and gestational weight gain on perinatal outcomes in Korean women: a retrospective cohort study. Reprod Biol Endocrinol 9(1):6. https://doi.org/10.1186/1477-7827-9-6
Wang T, Li L, Wu C et al (2021) Body mass index and gestational weight gain are associated with maternal and neonatal outcomes based on Chinese women. J Diabetes Res. https://doi.org/10.1155/2021/4542367
Liu L, Ma Y, Wang N et al (2019) Maternal body mass index and risk of neonatal adverse outcomes in China: a systematic review and meta-analysis. BMC Pregnancy Childbirth 19(1). https://doi.org/10.1186/s12884-019-2249-z
Xie YJ, Peng R, Han L, Zhou X, Xiong Z, Zhang Y, Li J, Yao R, Li T, Zhao Y (2016) Associations of neonatal high birth weight with maternal pre-pregnancy body mass index and gestational weight gain: a case-control study in women from Chongqing, China. BMJ Open 6(8):e010935. https://doi.org/10.1136/bmjopen-2015-010935
Abdulmalik MA, Ayoub JJ, Mahmoud A et al (2019) Pre-pregnancy BMI, gestational weight gain and birth outcomes in Lebanon and Qatar: results of the MINA cohort. PLoS One 14(7). https://doi.org/10.1371/journal.pone.0219248
Lewandowska M, Więckowska B, Sajdak S (2020) Pre-pregnancy obesity, excessive gestational weight gain, and the risk of pregnancy-induced hypertension and gestational diabetes mellitus. J Clin Med 9(6):1–13. https://doi.org/10.3390/jcm9061980
Kominiarek MA, Grobman W, Adam E, Buss C, Culhane J, Entringer S, Simhan H, Wadhwa PD, Kim KY, Keenan-Devlin L, Borders A (2018) Stress during pregnancy and gestational weight gain. J Perinatol 38(5):462–467. https://doi.org/10.1038/s41372-018-0051-9
Eraslan Sahin M, Col Madendag I (2019) Effect of gestational weight gain on perinatal outcomes in low risk pregnancies with normal prepregnancy body mass index. Biomed Res Int. https://doi.org/10.1155/2019/3768601
Stotland NE, Cheng YW, Hopkins LM, Caughey AB (2006) Gestational weight gain and adverse neonatal outcome among term infants. Obstet Gynecol 108(3):635–643. https://doi.org/10.1097/01.AOG.0000228960.16678.bd
Acknowledgements
The authors would like to thank all participants.
Author information
Authors and Affiliations
Contributions
SR and GN conceived and designed the survey. SR performed the data collection and entry. SH involved in the statistical analysis and data interpretation. SR wrote the manuscript. All authors read the manuscript, critically revised it for intellectual content, and approved the final version.
Corresponding author
Ethics declarations
Ethical approval and consent to participate
The ethics committee at the Notre-Dame des Secours University Hospital (CHUNDS) hospital approved the study protocol. The need for written informed consent was waived by the Notre-Dame des Secours University Hospital ethics committee due to the retrospective nature of the study. All methods were performed in accordance with the relevant guidelines and regulations (Declaration of Helsinki).
Consent to publish
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rouhana, S., Hallit, S. & Nicolas, G. The association of maternal pre-pregnancy Body Mass Index and gestational weight gain with pregnancy and neonatal outcomes. Ir J Med Sci 193, 303–312 (2024). https://doi.org/10.1007/s11845-023-03472-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11845-023-03472-w