Abstract
Background
Hyponatremia is the most common electrolyte disorder in lung cancer, and it particularly occurs in small cell lung cancer (SCLC) patients. The prognostic significance of hyponatremia has been reported in several studies with controversial results.
Aims
We aimed in this study to investigate hyponatremia and evaluate its prognostic value in SCLC patients.
Methods
The data of 373 SCLC patients were analyzed retrospectively. Serum sodium concentrations were measured from blood samples taken from all patients before treatment. Hyponatremia was defined as a serum sodium concentration below 135 mmol/L and then assigned into two groups: mild (130 to 134 mmol/L) and severe (below ≤ 129 mmol/L) hyponatremia.
Results
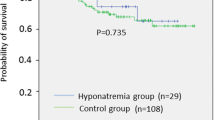
Hyponatremia was detected in 85 (22.8%) patients (mild hyponatremia in 51 (13.7%) and severe hyponatremia in 34 (9.1%) patients). Furthermore, 26% (63 of 242) of ED-SCLC patients and 16.8% (22 of 131) of LD-SCLC patients had hyponatremia. While no clinical parameter was statistically associated with serum sodium concentrations in LD-SCLC patients, hyponatremic ED-SCLC patients were more frequently associated with weight loss (p = 0.04) and liver metastasis (p = 0.04). In LD-SCLC, the overall survival (OS) rates of patients with hyponatremia were similar to those with normonatremia (p = 0.6). Likewise, hyponatremic and normonatremic ED-SCLC patients had similar life expectancies (p = 0.1). Moreover, the severity of hyponatremia did not affect OS in either LD-SCLC (p = 0.3) or ED-SCLC (p = 0.1).
Conclusion
Serum sodium concentration did not have an impact on survival in SCLC patients; thus, we concluded that neither the presence nor the severity of hyponatremia affected the outcome of these patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hyponatremia is the most common electrolyte disorder in lung cancer, particularly small cell lung cancer (SCLC) [1, 2]. Hyponatremia occurs in nearly 15% of SCLC patients [3]. It is not frequently life-threatening but will often cause a decrease in patient performance and quality of life, resulting in prolonged hospitalization and delay in planned chemotherapy applications [1].
So far, the prognostic significance of low sodium levels has been inconclusive in several studies with a small number of patients: some of them advocated that hyponatremia was a negative prognostic factor [3,4,5,6,7,8], and yet, others disagreed [9, 10]. In two systemic reviews, analyzing the independent prognostic role of hyponatremia in lung cancer including SCLC, the authors identified hyponatremia as an independent risk factor in poor survival in only half of the studies [11, 12]. Because of controversial results, we aimed in this study to investigate hyponatremia and evaluate its prognostic value in SCLC patients.
Material and methods
Patients
The data of 373 patients with pathologically confirmed SCLC were enrolled in the study and analyzed retrospectively. They were treated and followed up at the Institute of Oncology and the medical oncology outpatient clinic of Chest Diseases and Thoracic Surgery Training and Research Hospital between 2013 and 2021.
The VA Lung Study Group’s two-stage classification scheme has been used to define the extent of disease in patients with SCLC. Limited disease (LD-SCLC) is a disease confined to the ipsilateral hemithorax, which can be safely encompassed within a radiation field, and extended disease (ED-SCLC) is a disease beyond the ipsilateral hemithorax, including malignant pleural or pericardial effusion or hematogenous metastases. Patients were treated with various therapeutic approaches including surgery, radiotherapy, and various platinum-based chemotherapy and followed up according to standard guidelines, such as NCCN and ESMO Guidelines. The study was reviewed and approved by our Regional Ethical Committee.
Serum sodium concentrations were measured from blood samples taken from all patients before treatment. Hyponatremia was defined as a serum sodium concentration below 135 mmol/L and then assigned into two groups: mild (130 to 134 mmol/L) and severe (below ≤ 129 mmol/L) hyponatremia.
The medical records were retrieved from the cancer registry for review of the clinical features and outcomes.
Statistical analyses
The chi-square tests were used to evaluate the impact of clinical parameters on serum sodium levels. Kaplan–Meier analysis was used for estimation of survival of patients, and differences in survival were assessed by the log-rank statistics. Univariate analyses were carried out using Cox proportional hazards models. Overall survival (OS) was determined from the date of pathologic diagnosis to death resulting from any cause. Statistical analysis was carried out using SPSS 21.0 software (SPSS Inc., Chicago, Illinois, USA). A p value ≤ 0.05 was considered significant.
Results
Patients
A total of 373 SCLC patients were enrolled in the study. The median age of the patients was 63 years, ranging from 40 to 90 years, and the group was male dominant (n = 312, 83.6%). The majority of the patients had ED-SCLC (n = 242, 64.9%). Other demographic and clinical characteristics are shown in Table 1.
The median (range) of serum sodium concentration was 138 mmol/L (111–149) in all SCLC patients. Hyponatremia was detected in 85 (22.8%) patients [median (range), 132 mmol/L (111–134)], and 51 patients (13.7%) had mild hyponatremia [median (range), 133 mmol/L (130–134)]; on the other hand, 34 patients (9.1%) had severe hyponatremia [median (range), 125.5 mmol/L (111–129)].
Associations with clinical parameters
Hyponatremia was detected in 26% (63 of 242) of ED-SCLC patients and in 16.8% (22 of 131) of LD-SCLC patients. While no clinical parameter was statistically associated with serum sodium concentrations in LD-SCLC patients, hyponatremic ED-SCLC patients were more frequently associated with weight loss (p = 0.04) and liver metastasis (p = 0.04) (Table 1). However, distributions of other prognostic clinical factors — demographic (age and gender), patient-related (body mass index, performance status, smoking habits), pathologic (Ki-67), and clinical (tumor size, localization, responses to chemotherapy, relapse) — between serum sodium concentrations were found not significant (p > 0.05).
Survival
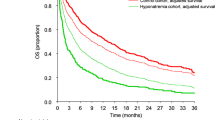
In LD-SCLC, the OS rates of patients with hyponatremia were similar to those with normonatremia; 1-year OS rates were 80.0% vs 71.2%, respectively (p = 0.6) (Fig. 1). Likewise, hyponatremic and normonatremic ED-SCLC patients had similar life expectancies (p = 0.1; 1-year OS rates were 15.7% vs 31.6%, respectively) (Fig. 2). Moreover, the severity of hyponatremia did not affect OS in either LD-SCLC (p = 0.3) or ED-SCLC (p = 0.1) (Figs. 3 and 4).
Univariate analysis
It was shown that neither the presence nor the severity of hyponatremia in both LD-SCLC and ED-SCLC affected OS. The effects of clinical variables on OS in patients at both stages are shown in Table 2.
Discussion
Among different types of cancer, SCLC is the one most frequently associated with hyponatremia: retrospective studies showed an incidence of about 20–44% in this setting [1, 2]. In our study, we found that the rate of hyponatremia was 22.8% in all SCLC patients, of which 9.1% was severe, and it was more frequent in ED-SCLC patients than in LD-SCLC patients (26% vs 16.8%, respectively). These findings were consistent with the results of the previous studies [3,4,5,6,7,8]. In a retrospective study (n = 453), serum sodium concentration was less than 125 mEq/L and 126–135 mEq/L in 11% and 33% of the SCLC patients, retrospectively [4]. Likewise, Hermes et al. found that hyponatremia (serum sodium concentration < 135 mmol/l) was present in 18.9% of 395 SCLC patients, 24% and 11% of whom had ED-SCLC and LD-SCLC, respectively [5]. They also reported that 8.8% of these patients had severe hyponatremia (serum sodium concentration < 129 mmol/l). The clinical data from 564 patients enrolled in 6 prospective topotecan iv studies showed that hyponatremia (< 135 mEq/L) was present in 17.9% of patients (< 125 mEq/L in 2.8%, 126–130 mEq/L in 2%, and 130–134 mEq/L in 13.1% of the patients) [6]. Moreover, Wang et al. reported that hyponatremia was present in 16.3% of all 631 patients, and severe hyponatremia was detected in 8.1% [3]; on the other hand, another study showed that 26% of 1171 patients had hyponatremia [7]. In a recent meta-analysis, the prevalence of hyponatremia in SCLC patients varied between 3 and 46.6% with an average of 24% [12]. However, Yang et al. found that the incidence of hyponatremia was 46.56% of 320 SCLC patients [8].
So far, the prognostic significance of hyponatremia has been inconclusive in several studies, yet some of them advocated that hyponatremia was a negative prognostic factor [3,4,5,6,7,8]. In a retrospective study from China, hyponatremic SCLC patients were associated with significantly shorter median survival than the normal group: 11.4 vs 14.5 months, p < 0.001 [3]. Hansen et al. concluded that hyponatremia was a significant prognostic factor associated with poor outcomes, with a median survival of 11.2 months in patients with normal sodium concentrations as opposed to 7.1 months in patients with subnormal values (p = 0.0001) [4]. Similarly, a retrospective analysis showed that hyponatremia was an independent predictor of mortality in patients with both LD- and ED-SCLC patients [5]. Hyponatremia was also shown as an independent prognostic factor for patients with SCLC treated with topotecan in a second-line setting [6]. However, we disagree with these previous conclusions: we found that neither the presence nor the severity of hyponatremia in both LD- and ED-SCLC affected survival. We believe that this result is not surprising, because we found that hyponatremia was not associated with such factors as performance status, sedimentation rate, serum LDH level, and response to chemotherapy that were prognostic for SCLC and found significant in univariate analysis.
Furthermore, in a systemic review, 6 of 13 studies investigating the prognostic power of hyponatremia in SCLC found that hyponatremia was a poor prognostic factor for survival, and in another meta-analysis, 6 of 12 studies showed low serum sodium as an independent poor prognostic factor [11, 12]. However, these studies have limitations, such as differences in detection time of hyponatremia (before or together with treatment), vague survival times and rates, and a lack of multivariate analyses in most studies; therefore, we believe that the interpretation of these results might be misleading.
The retrospective design of this study is the most significant limitation. Furthermore, the data solely depends on the accuracy of the patient records at the centers, and some data on clinical features are missing. Therefore, we believe that future prospective studies with greater numbers of patients that include, evaluate, and interpret all these details will provide more accurate conclusions in this regard.
In conclusion, we did not ascertain the impact of serum sodium concentration on survival in SCLC patients, and we found that neither the presence nor the severity of hyponatremia affected the outcome in this patient group.
References
Fiordoliva I, Meletani T, Baleani MG et al (2017) Managing hyponatremia in lung cancer: latest evidence and clinical implications. Ther Adv Med Oncol 9:711–719
Fuca G, Mariani L, Vullo SL et al (2019) Weighing the prognostic role of hyponatremia in hospitalized patients with metastatic solid tumors: the HYPNOSIS study. Sci Rep 9:12993
Wang W, Song Z, Zhang Y (2016) Hyponatremia in small cell lung cancer is associated with a poorer prognosis. Transl Cancer Res 5:36–43
Hansen O, Sørensen P, Hansen KH (2010) The occurrence of hyponatremia in SCLC and the influence on prognosis: a retrospective study of 453 patients treated in a single institution in a 10-year period. Lung Cancer 68:111–114
Hermes A, Waschki B, Reck M (2012) Hyponatremia as prognostic factor in small cell lung cancer–a retrospective single institution analysis. Respir Med 106:900–904
Tiseo M, Buti S, Boni L et al (2014) Prognostic role of hyponatremia in 564 small cell lung cancer patients treated with topotecan. Lung Cancer 86:91–95
Sandfeld-Paulsen B, Aggerholm-Pedersen N, Winther-Larsen A (2021) Hyponatremia in lung cancer: incidence and prognostic value in a Danish population-based cohort study. Lung Cancer 153:42–48
Yang Y, Sun N, Sun P, Zhang L (2017) Clinical characteristics and prognosis of elderly small cell lung cancer patients complicated with hyponatremia: a retrospective analysis. Anticancer Res 37:4681–4686
Allan SG, Stewart ME, Love S et al (1990) Prognosis at presentation of small cell carcinoma of the lung. Eur J Cancer 26:703–705
Umemura S, Segawa Y, Ueoka H et al (2007) Serum level of arginine-vasopressin influences the prognosis of extensive disease small-cell lung cancer. J Cancer Res Clin Oncol 133:519–524
Castillo JJ, Vincent M, Justice E (2012) Diagnosis and management of hyponatremia in cancer patients. Oncologist 17:756–765
Bartalis E, Gergics M, Tinusz B et al (2021) Prevalence and prognostic significance of hyponatremia in patients with lung cancer: systemic review and meta-analysis. Front Med 8:671951
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tas, F., Ozturk, A. & Erturk, K. Neither the presence nor the severity of hyponatremia affected the outcome of the patients with small cell lung cancer. Ir J Med Sci 192, 1613–1619 (2023). https://doi.org/10.1007/s11845-022-03199-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11845-022-03199-0