Abstract
Larval parasitoids can substantially reduce the population density of the pollen beetle [Brassicogethes aeneus (Fabricius), syn. Meligethes aeneus (Fabricius)]. The most abundant tersilochine parasitoids of pollen beetle are Tersilochus heterocerus, Phradis interstitialis and P. morionellus. The main activity of these parasitoids was observed in the period shortly before flowering to full flowering of oilseed rape. Insecticide applications during this period may have negative effects on parasitoids. In the present study, the effects of the insecticides Biscaya (a.i. thiacloprid), Mavrik (a.i. tau-fluvalinate) and Karate Zeon (a.i. lambda-cyhalothrin) applied during the bud or flowering stage of winter oilseed rape on parasitization of pollen beetle larvae by T. heterocerus were studied in 12 field trials at different locations in Germany in 2013–2015. The effects on parasitism by Phradis spp. were assessed in 2015. Parasitism of pollen beetle larvae by T. heterocerus was found in all field trials in all experimental years, but in most trials not before full flowering. Maximum percentage of parasitized larvae at different locations ranged between 3.4 and 16.8% in 2013, 8.3 and 22.4% in 2014 and from 11.1 to 29.1% in 2015. Levels of parasitism were not significantly different between the untreated control and insecticide treatments within each location. In contrast to T. heterocerus, Phradis spp. was not detected at all locations and not before flowering declining. In field trials at Lucklum and Puch, the maximum level of parasitism by Phradis spp. was 9.4 and 18.3%, respectively. No significant effect of insecticide application on parasitism by Phradis spp. was observed between the treatments. The results of this study showed that the insecticides used in the field trials did not affect parasitization of pollen beetle larvae by T. heterocerus and Phradis spp., regardless whether applied at the bud stage, at the beginning of flowering or full flowering.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Several hymenopteran endoparasitoids attack the egg and larval stages of pollen beetle [Brassicogethes aeneus (Fabricius), syn. Meligethes aeneus (Fabricius)] and may have significant impact on the population density (Nilsson and Andreasson 1987; Thies et al. 2008). Parasitism levels exceeding 40% have been reported by Nitzsche (1998), Büchi (2002), Nilsson (2003), Hokkanen (2006), Williams (2006), Ulber et al. (2010a) and Kaasik et al. (2014a, b). The most abundant larval parasitoids of pollen beetle are the ichneumonid parasitoids Phradis interstitialis (Thomson), P. morionellus (Holmgren) and Tersilochus heterocerus (Thomson), the latter being often predominant in crops of winter oilseed rape (Nitzsche 1998; Nilsson 2003) and P. morionellus in spring oilseed rape (Nilsson 2003; Veromann et al. 2006a; Kaasik et al. 2014b). These univoltine species are widely distributed throughout Europe (Nilsson 2003), with T. heterocerus and P. interstitialis predominating in Germany (Ulber et al. 2010a). The braconid Diospilus capito is also widely distributed throughout northern Europe, but it is more common in spring oilseed rape (Nilsson 2003; Veromann et al. 2006b; Kaasik et al. 2014b).
In Germany, P. interstitialis colonizes oilseed rape crops from mid of April onwards, whereas T. heterocerus and P. morionellus commonly occur 1–2 weeks later at the end of April or beginning of May (Nitzsche 1998; Johnen et al. 2010; Neumann 2010). Females are sexually mature when they emerge from their overwintering sites (Nilsson 2003). The females of P. interstitialis lay their eggs through the bud walls into the eggs of pollen beetle (Osborne 1960; Williams and Cook 2010; Berger et al. 2015). P. morionellus oviposits into larvae within green and yellow buds and open flowers (Williams and Cook 2010; Berger et al. 2015). T. heterocerus prefers L2-larvae for oviposition, which are feeding in open flowers (Nilsson and Andreasson 1987; Williams and Cook 2010) but also smaller host larvae are used for oviposition (Berger et al. 2015). The first larval instar of Phradis spp. hatches shortly after oviposition (Nilsson 2003), whereas larvae of T. heterocerus hatch from the egg only when the host larva is fully developed and ready to drop down to the soil for pupation (Osborne 1960; Nitzsche 1998). The parasitoid larvae complete their development in the soil and kill their hosts before they pupate in the cocoon of their hosts. Approximately 1 month after pupation, the parasitoids develop into adults, which stay in diapause until the next spring (Nilsson 2003; Ulber et al. 2010a).
The application of insecticides may be an important factor affecting parasitoid populations (Hanson et al. 2015). Adult parasitoids emerging in spring from crops of wheat or barley succeeding oilseed rape can be exposed to insecticide treatments targeted to control cereal aphids (Nilsson and Andreasson 1987; Jansen and San Martin y Gomez 2014). The main activity of adult tersilochine parasitoids was observed in the period shortly before flowering to full flowering of oilseed rape (Ulber and Nitzsche 2006). In consequence, applications of insecticides during this period may have negative effects on parasitoids searching for hosts (Johnen and Ulber 2004). Parasitoids can be exposed to insecticides by direct contact with the spray mist, by contact to insecticidal residues on the leaves while seeking for food or hosts, or by uptake of contaminated food material (Croft and Brown 1975; Longley and Jepson 1996). In addition to lethal effects, pesticides may have sublethal effects on natural enemies; for example by influencing host searching behaviour, oviposition or fertility. Further, pesticides may have repellent effects (Wright and Verkerk 1995). The aim of the present study was to analyze the effects of the insecticides Biscaya (a.i. thiacloprid), Mavrik (a.i. tau-fluvalinate) and Karate Zeon (a.i. lambda-cyhalothrin) applied at different growth stages between the bud stage and full flowering of winter oilseed rape on the parasitism rates of pollen beetle larvae by T. heterocerus and Phradis spp. in field trials in Germany in 2013–2015.
Materials and methods
Effects of the neonicotinoid Biscaya (72 g thiacloprid ha−1) and the pyrethroids Mavrik (48 g tau-fluvalinate ha−1) and Karate Zeon (7.5 g lambda-cyhalothrin ha−1) on parasitism rates of pollen beetle larvae were studied at four locations in Germany per year in Mecklenburg-Western Pomerania, Lower Saxony and Bavaria in 2013–2015 (Table 1). Conventional fields with regional land management were used as experimental fields. Pyrethroids were applied before the start of the trials at BBCH 19–30 if threshold values of the cabbage stem weevil (Ceutorhynchus pallidactylus) or rape stem weevil (C. napi) were exceeded. Only few pollen beetles were present in the fields at the time of controlling weevils. The experiments were established in randomized block design and each treatment with four replicated plots of 36–1080 m2 each. The controls were arranged as included controls according to EPPO PP 1/152(4) (2012) and there were no additional isolation zones within the trial site. All samplings were done in the centre of each plot leaving sufficient distance to adjacent plots. Different winter oilseed rape cultivars were used for the experiments. The BBCH code of Lancashire et al. (1991) was used to characterize the growth stages of winter oilseed rape. Insecticide applications were conducted at the bud stage or at the flowering stage with recommended product dose rates in Germany.
To collect pollen beetle larvae dropping from plants, 10 plastic bowls (17 cm × 12.1 cm) per plot filled with a 10% sodium benzoate water solution for conservation were placed on the ground in the centre of each plot (approximately 1 m distance to tramlines) during the bud stage (BBCH 53–55) in the field trials near Wendhausen (2013), Sickte (2014) and Lucklum (2015). The plastic bowls were put out into the field trials early in the bud stage not to miss the beginning of the larval dropping. They were checked regularly and emptied weekly after first larvae dropped down from BBCH 62 until BBCH 80. At all other locations, four plastic bowls (18.3 cm × 13.6 cm) per plot were used. In 2013, the plastic bowls were set up at BBCH 61 (Puch), BBCH 63 (Stöckendrebber) and BBCH 70 (Cramonshagen). In 2014 and 2015, the plastic bowls were established at all trial sites in the bud stage not to miss the beginning of the larval dropping.
The collected larvae were stored in 70% ethanol, later separated by length to larval instar according to Osborne (1964) and counted under the binocular microscope (8–10-fold magnification). For determination of parasitism rates by T. heterocerus, all collected larvae were carefully inspected under the binocular microscope to count the dark brown to black pigmented eggs, which can be easily seen through the cuticle of the host larvae (Osborne 1960). Superparasitism and multiparasitism of larvae were not recorded separately.
In addition, in 2015 in the field trial in Lucklum, a random subsample of 60 L2-pollen beetle larvae per treatment and date (as long as sufficient larvae were available) was dissected. The larvae were collected during four 1 week-periods between BBCH 65 and 76. Larvae were fixed with a needle on dark coloured wax within a petri dish and subsequently covered with water containing a detergent. The abdomen and thorax of the larvae were opened longitudinal by using a needle under a binocular microscope (10–12-fold magnification) to identify the eggs of T. heterocerus within the host body. The dissection showed slightly higher parasitism rates by T. heterocerus compared to determination of parasitism rates by external inspection. The presented data for parasitism rates of larvae by T. heterocerus were obtained from the much easier method of external inspection.
To examine the parasitism rates of pollen beetle larvae by Phradis spp. in field trials 2015, the L2-larvae were dissected as described above. The eggs of Phradis spp. are colourless and verification of the eggs is difficult (Osborne 1960). Because of these difficulties, only the larvae of Phradis spp., which are creamy-white with a slightly sclerotized but unpigmented head (Osborne 1960) were recorded. Random samples of 60 L2-larvae per treatment and date (depending on availability) were dissected on two to four sampling periods per location between BBCH 65 and 76. Parasitoid larvae were summarized as Phradis spp. because a morphological separation of P. interstitialis and P. morionellus is only possible by using adults.
Statistical analyses
In the field trials of 2013, the data on parasitism of L1- and L2-larvae were pooled. Results are presented for both development stages together. In the following years, parasitized larvae were separated into L1- and L2-larvae. Statistical analyses were carried out using the software R, version 3.1.2 (R Core Team 2014; packages: lme4 Bates et al. 2015, multcomp Hothorn et al. 2008, effects Fox 2003, MASS Venables and Ripley 2002, lsmeans Lenth 2015). Data of each year and field trial location were analyzed separately, because of a high variability in infestation levels and weather conditions. A generalized linear model (GLM) was used to analyze the differences in the parasitism level of pollen beetle larvae by T. heterocerus in the different treatments. A negative binomial model was used because the Poisson model revealed overdispersion. The treatment and date were included into the model as main effects and their interaction was tested. To compare total numbers of parasitized larvae in the different treatments accumulated over the collection periods, a linear model was used and the confidence intervals obtained from model estimates were used for interpretation of effects.
Parasitism rates of larvae by Phradis spp. in the field trials of 2015 were analyzed by using GLM (binomial for proportion data). The treatment and the date were included into the model as main effects and their interaction was tested. The treatments were compared pairwise for each day of assessment with the package lsmeans for post hoc testing. p values were adjusted with the Hochberg method (Blakesley et al. 2009).
Results
The pollen beetle larvae dropping from plants were collected with plastic bowls from BBCH 62 to 76–78. The cumulative number of L1-larvae is not presented because in all field trials in 2014 and 2015, parasitization of L1-larvae of pollen beetle by T. heterocerus was either not present or only on a low level. The first L2-larvae dropped down from BBCH 62 onwards. The most massive dropping period of L2-larvae was observed in all experimental years at BBCH 65–69 at petal fall. But even after the end of flowering, several L2-larvae were found in the plastic trays until BBCH 76–78. The cumulated sum of L2-larvae of the complete period of migration resulted in a lower number of L2-larvae in plots treated with Biscaya and Mavrik (Table 2). Applications of Karate Zeon increased the number of L2-larvae compared to the control.
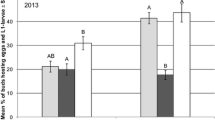
In 2013, parasitism of larvae by T. heterocerus was detected in all field trials (Fig. 1). The lowest level of parasitism was recorded in Stöckendrebber with a maximum percentage of 3.4% parasitized larvae in Karate Zeon treated plots. In Wendhausen, parasitized larvae were recorded from BBCH 63 onwards with a maximum of 8.0% of parasitized larvae in the control. In Puch, first parasitized larvae were detected during BBCH 61–65, but the parasitism rate was low at this time. For the total collection period, a maximum percentage of 13.2% parasitized larvae was found in Karate Zeon treated plots in Puch. However, the plastic bowls were removed too early (BBCH 75) in Puch because the end of larval migration of pollen beetle was missed. In the field trial in Cramonshagen 2013, the plastic bowls were set up too late (ca. at BBCH 70) to record the start of larval dropping and the end of larval migration was also missed because the bowls were retracted too early (BBCH 78). According to own observations, larvae that did not finish development before petal fall wait for rain that facilitates burying into the soil for pupation and often curl on young pods. Also, Burkhardt and von Lengerken (1920) observed larvae curled on pods. In Cramonshagen, a maximum percentage of 16.8% parasitized larvae was found in Karate Zeon treated plots. Within each location, the parasitism rates of larvae did not differ significantly between different insecticide treatments and the untreated control (GLM, p > 0.05). In the field trial in Wendhausen, the application of Biscaya at the beginning of flowering (BBCH 60) and at full flowering (BBCH 65) resulted in minimal reduced parasitism level of larvae compared to application of Biscaya at the bud stage (BBCH 53). These differences were not significant (GLM, p > 0.05).
Level of parasitism (%) of L1- and L2-pollen beetle larvae by T. heterocerus (± SE) following application of insecticides in winter oilseed rape field trials of 2013 in Germany near Wendhausen (collection period 2 May–21 June, BBCH 60–80), Stöckendrebber (14 May–12 June, BBCH 63–79), Cramonshagen (28 May–18 June, BBCH ca. 70–78) and Puch (6 May–5 June, BBCH 61–75). Biscaya = 72 g thiacloprid ha−1, Karate Zeon = 7.5 g lambda-cyhalothrin ha−1, Mavrik = 48 g tau-fluvalinate ha−1. GLM, p ≤ 0.05; n.s. no significant difference between treatments within each location, n.a. treatment not tested
In 2014 and 2015, the larval stages of parasitized pollen beetle larvae were differentiated. In 2014, parasitism of L1-larvae by T. heterocerus was detected in field trials in Bavaria and Mecklenburg-Western Pomerania, but not in Lower Saxony. The maximum level of parasitism of L1-larvae was 5.4% in Biscaya treated plots in Puch and 2.6% in Mavrik treated plots in Kleefeld. No significant differences between the treatments within each location were recorded (GLM, p > 0.05). Parasitized L1-larvae were not detected before BBCH 65.
Parasitism of L2-larvae by T. heterocerus was observed in all field trials in 2014. Maximum percentage of parasitized L2-larvae was 8.3% in untreated plots in Stöckendrebber, 9.4% in untreated plots in Kleefeld, 17.4% in untreated plots in Sickte and 22.4% in Karate Zeon treated plots in Puch (Fig. 2). At all locations, parasitized L2-larvae were first recorded from BBCH 65 onwards but not in earlier collection periods. Between insecticide treated and untreated plots within each location parasitism rates of L2-larvae were not significantly different (GLM, p > 0.05). Application of Biscaya and Mavrik at early flowering (BBCH 62) in the field trial in Sickte resulted in slightly lower parasitism rates of larvae compared to an application of these insecticides at the bud stage at BBCH 55. Parasitism rates in plots treated with Biscaya at BBCH 62 were reduced by 1.6% compared to treatment at the bud stage. Treatment with Mavrik at BBCH 62 resulted in 5.2% lower parasitism level compared to the treatment at BBCH 55. All observed differences were not significant (GLM, p > 0.05).
Level of parasitism (%) of L2-pollen beetle larvae by T. heterocerus (± SE) following application of insecticides in winter oilseed rape field trials of 2014 in Germany near Sickte (collection period 16 April–21 May, BBCH 62–76), Stöckendrebber (15 April–19 May, BBCH 62–79), Kleefeld (22 April–26 May, BBCH 64–80) and Puch (23 April–14 May, BBCH 65–69). Biscaya = 72 g thiacloprid ha−1, Karate Zeon = 7.5 g lambda-cyhalothrin ha−1, Mavrik = 48 g tau-fluvalinate ha−1. GLM, p ≤ 0.05; n.s. no significant difference between treatments within each location, n.a. treatment not tested
In 2015, parasitism of L1-larvae by T. heterocerus was detected in all field trials, except Niedernstöcken. The maximum level of parasitism of L1-larvae was 2.1% in plots treated with Mavrik at BBCH 62 in Lucklum, 10.6% in untreated plots in Kleefeld and 4.7% in Biscaya treated plots in Puch. No significant differences between the treatments within each location were recorded (GLM, p > 0.05). As in 2014, no parasitized L1-larvae were detected before BBCH 65.
Parasitism of L2-larvae by T. heterocerus was detected in all field trials in 2015, but again not before BBCH 65. Maximum percentage of L2-larvae parasitized by T. heterocerus was 11.1% in Biscaya treated plots in Niedernstöcken, 14.1% in untreated plots in Puch, 15.2% in plots treated with Biscaya at BBCH 55 in Lucklum and 29.1% in Biscaya treated plots in Kleefeld (Fig. 3). As in 2014, no significant differences were observed between different insecticide treatments and the control within each location (GLM, p > 0.05). Again, insecticide applications of Biscaya and Mavrik at early flowering (BBCH 62) in the field trial in Lucklum resulted in slightly lower parasitism rates of L2-larvae compared to applications of the insecticides at the bud stage at BBCH 55. Parasitism rates in plots treated with Biscaya at BBCH 62 were reduced by 6.6% compared to treatment of Biscaya at the bud stage. Treatment with Mavrik at BBCH 62 resulted in 5.2% lower parasitism rate compared to the treatment at BBCH 55. All differences were not significant (GLM, p > 0.05).
Level of parasitism (%) of L2-pollen beetle larvae by T. heterocerus (± SE) following application of insecticides in winter oilseed rape field trials of 2015 in Germany near Lucklum (collection period 4 May–9 June, BBCH 65–78), Niedernstöcken (29 April–10 June, BBCH 62–79), Kleefeld (11 May–22 June, BBCH 63–80) and Puch (29 April–10 June, BBCH 65–79). Biscaya = 72 g thiacloprid ha−1, Karate Zeon = 7.5 g lambda-cyhalothrin ha−1, Mavrik = 48 g tau-fluvalinate ha−1. GLM, p ≤ 0.05; n.s. no significant difference between treatments within each location, n.a. treatment not tested
The parasitoids Phradis spp. were not detected in all field trials in 2015, in contrast to T. heterocerus. By dissection of L2-pollen beetle larvae from the field trials in Mecklenburg-Western Pomerania and Niedernstöcken collected between BBCH 65 and 67 and between BBCH 67 and 70, no larvae parasitized by Phradis spp. were found. The subsequent dissection of pollen beetle larvae from the field trial in Niedernstöcken collected between BBCH 70 and 75 showed a parasitism rate of 4.2% for larvae from the untreated plots and 1.7% for larvae from plots treated with Biscaya, with no significant difference between the treatments (GLM, p > 0.05).
In Lucklum also no Phradis spp. were detected before BBCH 69. In the collection period between BBCH 69 and 71 in all treatments 3.3% of the larvae were parasitized, except larvae from the plots treated with Biscaya at BBCH 62, where no parasitism was found (Fig. 4). One week later, the dissection of the larvae collected between BBCH 71 and 76 showed that parasitism of larvae collected in untreated plots was 9.4%. In Biscaya treated plots, separately treated at BBCH 55 or 62, parasitism of larvae was 1.7%, respectively. Larvae from plots treated with Mavrik (regardless of the application date) showed a parasitism rate of 3.3%. Between all treatments, no significant difference was observed (GLM, p > 0.05).
Level of parasitism (%) of L2-pollen beetle larvae by Phradis spp. (± SE) following application of insecticides in winter oilseed rape field trials of 2015 in Germany near Lucklum and Puch at different collection periods. Biscaya = 72 g thiacloprid ha−1, Karate Zeon = 7.5 g lambda-cyhalothrin ha−1, Mavrik = 48 g tau-fluvalinate ha−1. GLM, p ≤ 0.05; n.s. no significant difference between treatments within each location, n.a. treatment not tested
In addition, a random sample of L2-larvae from the field trial in Bavaria collected between BBCH 67 and 69 was dissected. In all treatments, parasitism rates were 3.3%, except larvae from plots treated with Mavrik at BBCH 57 with 8.3% being parasitized, but differences were not significant (GLM, p > 0.05; Fig. 4). One week later, the dissection of the larvae collected between BBCH 69 and 75 showed 8.3% parasitism rate of the larvae from untreated plots. The highest parasitism rate was recorded from plots treated with Biscaya at BBCH 57 (18.3%), followed by plots treated with Mavrik at BBCH 57 (13.3%). Dissection of larvae from Karate Zeon treated plots resulted in a parasitism rate of 8.3%, similar to the control. Observed differences between all treatments did not differ significantly (GLM, p > 0.05).
Discussion
In the present study, effects of the insecticides Biscaya, Mavrik and Karate Zeon applied at different growth stages of winter oilseed rape between the green bud stage and full flowering on the parasitism rates of pollen beetle larvae by T. heterocerus and Phradis spp. were analyzed in field trials in 2013–2015. In all field trials in 2014 and 2015, parasitization of L1-larvae of pollen beetle by T. heterocerus was either not present or only on a low level. This corresponds with observations by Nilsson and Andreasson (1987), who found that large L2-larvae are preferred by T. heterocerus for oviposition.
Two different methods were used for the determination of parasitism rates of L2-pollen beetle larvae by T. heterocerus. The dissection of L2-larvae showed a slightly higher parasitism rate by T. heterocerus compared to determination of dark pigmented eggs by external inspection. This was analyzed only for the larvae of the last two collection periods from BBCH 69 onwards. According to Nilsson and Andreasson (1987), this error is negligible.
In this study, the parasitism rates of pollen beetle larvae in Biscaya and Mavrik treated plots were lower in some trials than in the control, but without significant differences. In all field trials, the application of Biscaya, regardless whether applied at the bud stage or at flowering, reduced the number of L2-larvae compared to untreated plots (Table 2). For example, in plots treated with Biscaya at BBCH 55 in Lucklum 2015, the number of L2-larvae dropping to the ground was reduced by 64% compared to the control (total sum of L2-larvae m−2 in the control 1716.1 ± 179.3). In most experiments, these effects of Biscaya were significant. In plots treated with Mavrik, the number of L2-larvae was reduced to a lesser extent than in Biscaya treated plots. In contrast, application of Karate Zeon in 2013 in all field trials resulted in a higher number of L2-larvae compared to the control. For example, the abundance of larvae increased up to 29% in Cramonshagen or up to 37% in Puch. In 2014 and 2015, Karate Zeon was tested only near Puch in Bavaria where the number of L2-larvae increased up to 31 and 42%, respectively, compared to untreated plots. In consequence in Biscaya and Mavrik treated plots, the number of host larvae available for parasitoids was lower in the untreated plots, whereas in Karate Zeon treated plots, the highest number occurred.
Parasitoids may show a density-dependent response to host populations (Hanson et al. 2015). Contrasting results on density-dependence of parasitism have been reported in literature: Longley and Jepson (1996) found that wheat plants infested by low numbers of Sitobion avenae were less favoured or avoided by female parasitoids of Aphidius spp. than plants with higher aphid infestation. Similarly, parasitoids of pollen beetle were found to aggregate on plots with a higher host density (Zaller et al. 2009). A positive correlation between the density of the host population and the parasitism rate was also detected by Neumann (2010) in one of three experimental years. However, Nilsson and Andreasson (1987) did not found a correlation between the number of pollen beetle larvae and parasitism.
Herbivore-induced plant volatiles can attract natural enemies to infested plants (Mattiacci et al. 1994; Dicke and Baldwin 2009; Heil 2014). Volatiles of oilseed rape released after insect feeding have major impact on the searching behaviour of T. heterocerus and Phradis spp. (Jönsson et al. 2005). Because infestation by beetles and larvae was lower in Biscaya and Mavrik treated plots, lower amounts of synomones may have been released in these plots and parasitoids may have visited these plots less frequently.
In 2014 and 2015, parasitization of L1- and L2-larvae by T. heterocerus was not found in the samples before BBCH 65. This is in accordance with Nitzsche (1998) and Johnen et al. (2010), who observed that in Germany, depending on annual weather conditions, the parasitoid does not occur before the beginning of flowering at the end of April or beginning of May. In all experimental years, insecticides applied in the bud stage had no significant effect on T. heterocerus. This is in accordance with Nilsson (1985), who stated that pesticide applications during the bud stage will hardly affect these parasitoids, but could have an effect on P. interstitialis. It can be assumed that T. heterocerus was not present in the fields at the time of application at the bud stage and immigration occurred not until the beginning of flowering, which started for example in Wendhausen (2013), Sickte (2014) and Lucklum (2015) at least 10 days after application. Therefore, the insecticides used seem to have a low persistent efficacy on these beneficials.
In the region of Goettingen, in the centre of Germany, the main activity of tersilochine parasitoids was found in the period shortly before flowering until full flowering (Ulber and Nitzsche 2006). In consequence application of insecticides in this activity phase, as occurred in the field trials in Wendhausen, Sickte and Lucklum at BBCH 60–65, might have negative effects on parasitoids. Insecticide applications of Biscaya at BBCH 60, 62 or 65 and of Mavrik at BBCH 62 resulted in slightly lower parasitism rates of larvae by T. heterocerus in all years compared to applications of these insecticides at the bud stage (BBCH 53–55), but these differences were not significant and again host larvae occurred less frequent in treated plots.
In 2015, the dissection of L2-larvae to determine parasitism rates showed that parasitism by Phradis spp. did not occur in all field trials (no detection in Mecklenburg-Western Pomerania) and at other locations were not detected before BBCH 67. In Niedernstöcken and Lucklum larvae parasitized by Phradis spp. were only detected from BBCH 70 to 75 (Niedernstöcken) and BBCH 69 to 71 (Lucklum) onwards. The earliest parasitization was recorded in the field trial in Bavaria where larvae were parasitized from BBCH 67 onwards. P. interstitialis predominates in winter oilseed rape in Germany (Ulber et al. 2010a), whereas P. morionellus is more abundant in spring oilseed rape in northern Europe (Nilsson 2003; Veromann et al. 2006a; Ulber et al. 2010a; Kaasik et al. 2014b) and was rarely found on winter oilseed rape in Germany by Ulber et al. (2010a). So it is likely that most of the Phradis larvae found in the field trials in 2015 were P. interstitialis. To discriminate between P. interstitialis and P. morionellus rearing of adults from the larvae would have been necessary which was not possible.
The immigration of P. interstitialis into oilseed rape fields started from mid of April onwards (Nitzsche 1998; Johnen et al. 2010). Insecticide applications during the bud stage in mid-April, as in the present field trials (BBCH 53 to 57–59), might have a harmful effect on P. interstitialis. The females are sexually mature at this time (Nilsson 2003) and oviposit through the wall of the buds into the eggs of pollen beetle (Osborne 1960; Williams and Cook 2010; Berger et al. 2015). The development time from egg laying of pollen beetle to hatch of L1-larvae takes approximately 10 days in winter oilseed rape, depending on temperature (Nilsson 1988). The first larval instar period lasts approximately 5–10 days (Burkhardt and von Lengerken 1920; Nilsson 1988). The first Phradis spp. larvae were found in L2-pollen beetle larvae not before BBCH 67 (mid of May 2015). Subtracting the development time of the host larvae of about 15–20 days, it can be assumed that P. interstitialis did not migrate into the fields before end of April or beginning of May 2015 (BBCH 62–65). This means that the parasitoids occurred 2–3 weeks after the applications in the field trials and were little exposed to insecticide residuals. Consequently, no significant effects of insecticides on Phradis spp. were observed. In our field trials, the start of migration of the parasitoids was not assessed by yellow water traps as described by Nitzsche (1998) or Williams et al. (2003). Application of Biscaya and Mavrik at BBCH 62, as conducted in Lucklum in 2015, did not have significant effects on P. interstitialis.
The insecticides used in the present field trials in 2013–2015 did not significantly affect parasitization of pollen beetle larvae by T. heterocerus and Phradis spp., regardless whether applied at the bud stage, at the beginning of flowering or at full flowering. In contrast, examples of negative effects of insecticides on parasitoids have been reported in literature. Veromann et al. (2006b) described reduced parasitism rates of pollen beetle larvae by D. capito and Phradis spp. in winter and spring oilseed rape after application of the pyrethroids Fastac (a.i. alpha-cypermethrin) and Karate (lambda-cyhalothrin) at BBCH 51 and 67 in 1 year field trials in Estonia. It has to be taken into account that application of the insecticides reduced number of overwintered pollen beetles. Consequently, the number of eggs and finally the number of larvae are expected to be lower in treated plots and plots with reduced egg and larval density may be less attractive to the parasitoids.
The impact of various insecticides on parasitization of pollen beetle larvae has been reported by Jansen and San Martin y Gomez (2014). In a 1-year trial, the authors tested the insecticides Mavrik 2F (tau-fluvalinate), Biscaya (thiacloprid), Boravi WG (Phosmet), Plenum (Pymetrozine) and Pyrinex (Chlorpyriphos-ethyl) applied at the end of the bud stage. All insecticide treatments caused a significant reduction of the adult population of parasitic Hymenoptera and decreased the parasitism rate of pollen beetle larvae. The strongest reduction was caused by Biscaya and Pyrinex, but only Biscaya reduced the larval parasitism significantly. The clear effect of Biscaya in the field trial of Jansen and San Martin y Gomez (2014) is in contrast to the results of the present study. It has to be considered, that Jansen and San Martin y Gomez (2014) as well as Veromann et al. (2006b) did not take into account the significant reduction of overwintered pollen beetles due to the insecticide treatments and consequently, a lower larval density in treated plots. In the field trial of Jansen and San Martin y Gomez (2014) in Biscaya and Pyrinex treated plots, 84 and 93% less larvae were recorded. This is in accordance with the present field trials, where application of Biscaya, regardless whether treated at the bud stage or at beginning of flowering, resulted in a reduction of L2-larvae. Consequently as described above, a density-dependent reaction of parasitoids is possible and plots with low larval density might be visited less frequently by parasitoids. Further, it is important to note that Jansen and San Martin y Gomez (2014) did not separate between the different parasitoids, which occur in spring at different times (Nitzsche 1998; Johnen et al. 2010) and prefer different development stages of pollen beetle (Osborne 1960; Williams and Cook 2010; Berger et al. 2015).
The effects of lambda-cyhalothrin and tau-fluvalinate on key parasitoids of oilseed rape pests have also been studied in a project in Poland in 2 years (Ulber et al. 2010b). The insecticides were applied at BBCH 54/56 and 65/68. Applications of insecticides resulted in mortality of parasitoids. Lambda-cyhalothrin showed a more persistent effect compared to tau-fluvalinate, which reduced parasitism level of host larvae less. The total number of pollen beetle larvae available for the parasitoids in different treatments is not mentioned in the reference. Treatment of insecticides may have reduced larval density. In the period of the field trial (2002 and 2004), the pyrethroid resistance of pollen beetles was not as far developed as nowadays, so it has to be expected that Karate reduced pollen beetle density as well as Mavrik. Consequently, a lower number of larvae might have been available for the parasitoids which may respond density-dependent.
The field results obtained in Poland are in accordance with laboratory tests using adult parasitoids by Jackowski et al. (2008). In a tarsal plate test effects of lambda-cyhalothrin and tau-fluvalinate, both tested at full and reduced application rates, were observed on Phradis spp., with Mavrik being less toxic than Karate. A 50% reduced application rate of Mavrik showed no effect on mortality of Phradis spp. compared to 100%. In contrast, the reduction of the application rate to 50% of Karate resulted in a lower parasitoid mortality than 100% dose rate.
In 3-year-field trials at Goettingen (Germany), the effects of lambda-cyhalothrin, tau-fluvalinate and thiacloprid on the parasitoids T. heterocerus and P. interstitialis were analyzed (Neumann 2010). Insecticides were applied with full and half recommended dose rate at BBCH 61/62 and BBCH 65/69. In contrast to the studies of Jackowski et al. (2008) and Ulber et al. (2010b), no significant effects of lambda-cyhalothrin on parasitism of pollen beetle larvae were obtained consistently over 3 years, regardless of 50 or 100% dose rate and of applications at early flowering or full flowering. Tau-fluvalinate applied with 50 and 100% dose rate at early flowering resulted in significant reduced parasitism although the application was conducted 9 days before parasitoid immigration. 50% dose rate of tau-fluvalinate at full flowering, parallel to the activity peak of the parasitoids, also resulted in significant reduced parasitism, whereas the full dose rate of tau-fluvalinate at full flowering did not significantly reduce parasitism.
Tau-fluvalinate was tested only in the first year by Neumann (2010) and was replaced by thiacloprid in the second and third year. For thiacloprid, no clear trend was detectable. One year thiacloprid significantly reduced the number of pollen beetle larvae when applied with full dose rate at the beginning of flowering and at full flowering with 50 and 100% dose rate. No significant differences were observed concerning parasitism. This is in accordance with the results of the present field trials. In the following year, Neumann (2010) observed that Biscaya had a significant effect on parasitoids if applied with full dose rate at the beginning of flowering and with both application rates at full flowering. But it has to be considered that the number of host larvae per plant was very low in this year (2 larvae per plant).
Finally, it can be concluded that no clear and consistent effects of lambda-cyhalothrin, tau-fluvalinate and thiacloprid on parasitism rates of pollen beetle larvae are reported in literature. Effects of individual insecticides differ between authors and between years within one paper. More information on effects of insecticides on parasitoids is urgently needed to protect parasitoids in the crop and support natural control of pests. Therefore, trials over several years with different locations are necessary to test such side effects of insecticides more detailed. It would be important to monitor the immigration and the activity peak of the different parasitoids, to determine whether there may be long term effects of insecticides applied before immigration of parasitoids or whether the application was coincident with the activity peak of the parasitoids. To analyze parasitized larvae, it has to be considered that pollen beetle larvae should be collected over a longer period and not only during one or two collection dates, as our data show that eggs or larvae of parasitoids occur in most cases not before BBCH 65 (T. heterocerus) or BBCH 67 (Phradis spp.) in higher numbers and still occur in larvae collected after the end of flowering. In addition, the total number of adult pollen beetle and host larvae needs to be monitored to separate between direct toxic and indirect effects.
References
Bates D, Maechler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67(1):1–48
Berger J, Jönsson M, Hedlund K, Anderson P (2015) Niche separation of pollen beetle parasitoids. Front Ecol Evol 3:45. https://doi.org/10.3389/fevo.2015.00045
Blakesley RE, Mazumdar S, Dew MA, Houck PR, Tang G, Reynolds CF III, Butters MA (2009) Comparisons of methods for multiple hypothesis testing in neuropsychological research. Neuropsychology 23:255–264
Büchi R (2002) Mortality of pollen beetle (Meligethes spp.) larvae due to predators and parasitoids in rape fields and the effect of conservation strips. Agric Ecosyst Environ 90:255–263
Burkhardt F, von Lengerken H (1920) Beiträge zur Biologie des Rapsglanzkäfers (Meligethes aeneus Fabr.). Z angew Entomol 6:270–295
Croft BA, Brown AWA (1975) Response of arthropod natural enemies to insecticides. Annu Rev Entomol 20:285–335
Dicke M, Baldwin IT (2009) The evolutionary context for herbivore-induced plant volatiles: beyond the ‘cry for help’. Trends Plant Sci 15(3):167–175
EPPO (2012) European and Mediterranean Plant Protection Organization; PP 1/152(4), efficacy evaluation of plant protection products. Design and analysis of efficacy evaluation trials. EPPO Bull 42(3):367–381
Fox J (2003) Effect displays in R for generalised linear models. J Stat Softw 8(15):1–27
Hanson HI, Smith HG, Hedlund K (2015) Agricultural management reduces emergence of pollen beetle parasitoids. Agric Ecosyst Environ 205:9–14
Heil M (2014) Herbivore-induced plant volatiles: targets, perception and unanswered questions. N Phytol 204:297–306
Hokkanen HMT (2006) Phradis morionellus on Meligethes aeneus: long-term pattern of parasitism and impact on pollen beetle populations in Finland. IOBC–WPRS Bull 29(7):187–191
Hothorn T, Bretz F, Westfall P (2008) Simultaneous inference in general parametric models. Biom J 50(3):346–363
Jackowski J, Klukowksi Z, Irzykowicz M (2008) The effect of τ-fluvalinate and λ-cyhalothrin on two parasitic species of Phradis spp. (Hymenoptera, Ichneumonidae, Tersilochinae). Pesticides 3–4:79–92
Jansen JP, San Martin y Gomez G (2014) A large field trial to assess the short-term and long-term effects of 5 insecticides used to control the pollen beetle on parasitic Hymenoptera in oilseed rape. IOBC–WPRS Bull 103:9–16
Johnen A, Ulber B (2004) Perspektiven der Nützlingschonung im Winterraps durch die Entwicklung von phänologischen Modellen. Mitt Biol Bundesanst Land- Forstwirtsch 396:278
Johnen A, Williams IH, Nilsson C, Klukowski Z, Luik A, Ulber B (2010) The proPlant decision support system: phenological models for the major pests of oilseed rape and their key parasitoids in Europe. In: Williams IH (ed) Biocontrol-based integrated management of oilseed rape pests. Springer, Heidelberg, pp 381–403
Jönsson M, Lindkvist A, Anderson P (2005) Behavioural responses in three ichneumonid pollen beetle parasitoids to volatiles emitted from different phenological stages of oilseed rape. Entomol Exp Appl 115:363–369
Kaasik R, Kovacs G, Kaart T, Metspalu L, Williams IH, Veromann E (2014a) Meligethes aeneus oviposition preferences, larval parasitism rate and species composition of parasitoids on Brassica nigra, Raphanus sativus and Eruca sativa compared with on Brassica napus. Biol Control 69:65–71
Kaasik R, Kovacs G, Toome M, Metspalu L, Veromann E (2014b) The relative attractiveness of Brassica napus, B. rapa, B. juncea and Sinapis alba to pollen beetles. Biocontrol 59:19–28
Lancashire PD, Bleiholder H, van den Boom T, Langelüddeke P, Strauss R, Weber E, Witzenberger A (1991) A uniform decimal code for growth stages of crops and weeds. Ann Appl Biol 119:561–601
Lenth R (2015) lsmeans: least-squares means. R package version 2.20-23. http://CRAN.R-project.org/package=lsmeans
Longley M, Jepson PC (1996) Effects of honeydew and insecticide residues on the distribution of foraging aphid parasitoids under glasshouse and field conditions. Entomol Exp Appl 81:189–198
Mattiacci L, Dicke M, Posthumus MA (1994) Induction of parasitoid attracting synomone in Brussels sprouts plants by feeding of Pieris brassicae larvae: role of mechanical damage and herbivore elicitor. J Chem Ecol 20:2229–2247
Neumann N (2010) Lethal and sublethal effects of insecticides on mortality, migration and host searching behaviour of tersilochine parasitoids on winter oilseed rape. PhD Thesis, University of Göttingen
Nilsson C (1985) Impact of ploughing on emergence of pollen beetle parasitoids after hibernation. Z Angew Entomol 100:302–308
Nilsson C (1988) The pollen beetle (Meligethes aeneus F.) in winter and spring rape at Alnarp 1976–1978. III. Mortality factors. Växtskyddsnotiser 52(6):145–150
Nilsson C (2003) Parasitoids of pollen beetles. In: Alford DV (ed) Biocontrol of oilseed rape pests. Blackwell, Oxford, pp 73–85
Nilsson C, Andreasson B (1987) Parasitoids and predators attacking pollen beetles (Meligethes aeneus F.) in spring and winter rape in southern Sweden. IOBC–WPRS Bull 10(4):64–73
Nitzsche O (1998) Auftreten und Effizienz von Parasitoiden als natürliche Gegenspieler von Schadinsekten im Winterraps unter besonderer Berücksichtigung unterschiedlicher Bodenbearbeitungsmaßnahmen nach Winterraps. PhD Thesis, University of Göttingen
Osborne P (1960) Observations on the natural enemies of Meligethes aeneus (F.) and M. viridescens (F.) [Coleoptera: Nitidulidae]. Parasitology 50:91–110
Osborne P (1964) Morphology of the immature stages of Meligethes aeneus (F.) and M. viridescens (F.) (Coleoptera, Nitidulidae). Bull Entomol Res 55:747–759
R Core Team (2014) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. http://www.R-project.org/
Thies C, Steffan-Dewenter I, Tscharntke T (2008) Interannual landscape changes influence plant–herbivore–parasitoid interactions. Agric Ecosyst Environ 125:266–268
Ulber B, Nitzsche O (2006) Phenology of parasitoids (Hym., Ichneumonidae–Tersilochinae) of oilseed rape pests in northern Germany from 1995–1997. IOBC–WPRS Bull 29(7):173–179
Ulber B, Williams IH, Klukowski Z, Luik A, Nilsson C (2010a) Parasitoids of oilseed rape pests in Europe: key species for conservation biocontrol. In: Williams IH (ed) Biocontrol-based integrated management of oilseed rape pests. Springer, Heidelberg, pp 45–76
Ulber B, Klukowski Z, Williams IH (2010b) Impact of insecticides on parasitoids of oilseed rape pests. In: Williams IH (ed) Biocontrol-based integrated management of oilseed rape pests. Springer, Heidelberg, pp 337–355
Venables WN, Ripley BD (2002) In: Venables WN, Ripley BD (eds) Modern applied statistics with S. Springer, New York
Veromann E, Luik A, Metspalu L, Williams I (2006a) Key pests and their parasitoids on spring and winter oilseed rape in Estonia. Entomol Fenn 17:400–404
Veromann E, Luik A, Kevväi R (2006b) Oilseed rape pests and their parasitoids in Estonia. IOBC–WPRS Bull 29(7):165–172
Williams IH (2006) Integrating parasitoids into management of pollen beetle on oilseed rape. Agron Res 4:465–470
Williams IH, Cook SM (2010) Crop location by oilseed rape pests and host location by their parasitoids. In: Williams IH (ed) Biocontrol-based integrated management of oilseed rape pests. Springer, Heidelberg, pp 215–244
Williams IH, Büchi R, Ulber B (2003) Sampling, trapping and rearing oilseed rape pests and their parasitoids. In: Alford DV (ed) Biocontrol of oilseed rape pests. Blackwell, Oxford, pp 145–160
Wright DJ, Verkerk RHJ (1995) Integration of chemical and biological control systems for arthropods: evaluation in a multitrophic context. Pestic Sci 44:207–218
Zaller JG, Moser D, Drapela T, Schmöger C, Frank T (2009) Parasitism of stem weevils and pollen beetles in winter oilseed rape is differentially affected by crop management and landscape characteristics. Biocontrol 54:505–514
Acknowledgements
This study was supported by the Union zur Förderung von Oel- und Proteinpflanzen e.V. Special thanks to the staff of the plant protection services of the federal states Mecklenburg-Western Pomerania, Lower Saxony and Bavaria for conducting the external field trials. Furthermore thanks to Dr. Doreen Gabriel und Dr. Anke Dietzsch for statistical support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Miriama Malcicka.
Rights and permissions
About this article
Cite this article
Brandes, M., Heimbach, U. & Ulber, B. Effects of insecticide application on parasitism rates of pollen beetle larvae (Brassicogethes aeneus (Fabricius)) by tersilochine parasitoids. Arthropod-Plant Interactions 12, 799–809 (2018). https://doi.org/10.1007/s11829-017-9580-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11829-017-9580-y