Abstract
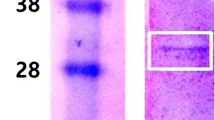
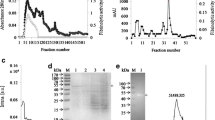
A serine protease was purified from Marphysa sanguinea through ammonium sulfate followed by ion exchange chromatography, and its N-terminal amino sequence was identified to be IVGGSEATPYQFPFQ. Fibrinolytic activity was depended on both direct fibrinolysis and indirect plasminogen-mediated cascade and had a consistent activity irrespective of pH. The serine protease could be confirmed to degrade α-, β-, and γ-chains of human fibrinogen through fibrinogenolytic assay and did not express significant cytotoxicity to endothelial cells. These imply the enzyme has anticoagulant as well as thrombolytic activity, not significantly impairing endothelial cells comprising brain blood brain barrier (BBB) tissue. Conclusively, the new serine protease is worthy of being a candidate to substitute tissue-Plasminogen Activator (t-PA) for acute ischemic reperfusion injury of brain.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
P. Libby, Am. J. Clin. Nutr., 83, 4565 (2006).
T. Ingall, J. Insur. Med., 36, 143 (2004).
P. Thorvaldsen, K. Asplund, K. Kuulasmaa, A. M. Rajakangas and M. Schroll, Stroke, 26, 1 (1995).
M. L. Simoons, P. Jaegere, R. Domburg, E. Boersma, A. P. Maggioni, M. G. Franzosi, J. D. Leimberger, R. Califf, R. Schröderm, G. Knatterud and E. Braunwald, The Lancet, 342, 1523 (1993).
R. E. Fromm, E. Hoskins, L. Cronin, C. M. Pratt, W. H. Spencer and R. Roberts, Ann. Emerg. Med., 20, 892 (1991).
B. Niego, R. Freeman, T. B. Puschmann, A. M. Turnley and R. L. Medcalf, Blood, 119, 4752 (2012).
P. R. Eisenberg, L. A. Sherman, A. J. Tiefenbrunn, P. A. Ludbrook, B. E. Sobel and A. S. Jaffe, Thromb. Haemost., 57, 35 (1987).
T. Nilsson, P. Wallen and G. Mellbring, Eur. J. Haematol., 33, 49 (1984).
J. P. Tsikouris and A. P. Tsikouris, Pharmacotherapy, 21, 207 (2001).
O. Matsuo, D. C. Rijken and D. Collen, Thromb. Haemost., 45, 225 (1981).
W. Hacke, G. Albers, Y. Ai-Rawi, J. Bogousslavsky, A. Davalos, M. Eliasziw, M. Fischer, A. Furlan, M. Kaste, K. R. Lees, M. Soehgen and S. Warach, Stroke, 36, 1 (2005).
G. W. Albers, R. von Kummor, T. Truelsen, J. S. Jensen, G. M. Ravn, B. A. Grønning, H. Chabriat, K. Chang, A. E. Davalos, G. A. Ford, J. Grotta, M. Kaste, L. H. Schwamm and A. Shuaib, Lancet. Neurol., 14, 575 (2015).
J. Loscalzo and E. Braunwald, N. Engl. J. Med., 319, 925 (1988).
B. Wiman and D. Collen, Nature, 272, 549 (1978).
T. Matys and S. Strickland, Nat. Med., 9, 371 (2003).
G. Micieli, S. Marcheselli and P. A. Tosi, Vasc. Health Risk Manag., 5, 397 (2009).
E. J. Su, L. Fredriksson, M. Geyer, E. Folestad, J. Cale, J. Andrae, Y. Gao, K. Pietras, K. Mann, M. Yepes, D. K. Strickland, C. Betsholtz, U. Eriksson and D. A. Lawrence, Nat. Med., 14, 731 (2008).
D. Liu, T. Cheng, H. Guo, J. A. Fernández, J. H. Griffin, X. Song and B. V. Zlokovic, Nat. Med., 10, 1379 (2004).
K. Benchenane, J. P. López-Atalaya, M. Fernández-Monreal, O. Touzani and D. Vivien, Trends Neurosci., 27, 155 (2004).
J. Kaur, Z. Zhao, G. M. Klein, E. H. Lo and A. M. Buchan, J. Cereb. Blood. Flow. Metab., 24, 945 (2004).
Z. Deng, S. Wang, Q. Li, X. Ji, L. Zhang and M. Hong, Bioresour. Technol., 101, 1954 (2010).
Y. Zhang, J. Cui, R. Zhang, Y. Wang and M. Hong, Biochimie, 89, 93 (2007).
K. Matsubara, K. Hori, Y. Matsuura and K. Miyazawa, Comp. Biochem. Physiol. B, Biochem. Mol. Biol., 125, 137 (2000).
S. Wang, Z. Deng, Q. Li, X. Ge, Q. Bo, J. Liu, J. Cui, X. Jiang, J. Liu, L. Zhang and M. Hong, Comp. Biochem. Physiol. B, Biochem. Mol. Biol., 159, 18 (2011).
D. Collen, Thromb. Haemost., 82, 259 (1999).
O. Matsuo, D. C. Rijken and D. Collen, Nature, 291, 590 (1981).
S. M. Camiolo, S. Thorsen and T. Astrup, Exp. Biol. Med., 138, 227 (1971).
E. Kotb, Appl. Biochem. Microbiol., 51, 34 (2015).
G. T. James, Anal. Biochem., 86, 574 (1978).
J. H. Choi, K. Sapkota, S. Kim and S. J. Kim, Biochimie, 105, 45 (2014).
S. M. Camiolo, S. Thorsen and T. Astrup, Exp. Biol. Med., 138, 277 (1971).
N. K. Isaev, E. V. Stelmashook, E. Y. Plotnikov, T. G. Khryapenkova, E. R. Lozier, Y. V. Doludin, D. N. Silachev and D. B. Zorov, Biochem. (Mosc. ), 73, 1171 (2008).
A. V. Alexandrov and J. C. Grotta, Neurology, 59, 862 (2002).
S. V. Pizzo, M. L. Schwartz, R. L. Hill and P. A. Mckee, J. Clin. Invest., 51, 2841 (1972).
D. H. Farrell, P. Thiagarajan, D. W. Chung and E. W. Davie, Proc. Natl. Acad. Sci. U. S. A., 89, 10729 (1992).
C. Ouyang and T. F. Huang, Biochim. Biophys. Acta-Enzymology, 571, 270 (1979).
J. W. Lee, J. H. Seu, I. K. Rhee, I. Jin, Y. Kawamura and W. Park, Biochem. Biophys. Res. Commun., 260, 665 (1999).
J. W. Park, J. E. Park, H. K. Choi, T. W. Jung, S. M. Yoon and J. S. Lee, Process Biochem., 48, 979 (2013).
Author information
Authors and Affiliations
Corresponding author
Additional information
These authors contributed equally to this work.
Rights and permissions
About this article
Cite this article
Yeon, S.J., Shim, K.H., Hong, J.S. et al. Identification of a new serine protease from polychaeta, Marphysa sanguinea, for its thrombolytic and anticoagulant activity. Korean J. Chem. Eng. 34, 781–786 (2017). https://doi.org/10.1007/s11814-016-0331-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-016-0331-z