Abstract
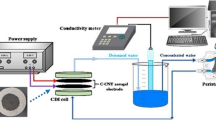
Carbon aerogel was chemically activated with KOH at various activation temperatures with the aim of improving the electrochemical performance of carbon aerogel for EDLC electrode. Electrochemical performance of activated carbon aerogel electrode was determined by cyclic voltammetry and galvanostatic charge/discharge methods using coin-type EDLC cell in organic electrolyte. Activation temperature played an important role in determining the electrochemical performance of activated carbon aerogel for EDLC electrode. Specific capacitance of activated carbon aerogel at a high current density (5 A/g) showed a volcano-shaped curve with respect to activation temperature. Excessively high activation temperature could have an adverse effect on the electrochemical properties of activated carbon aerogel due to the low electrical conductivity caused by a collapse of characteristic structure of carbon aerogel. Among the carbon samples, carbon aerogel activated at 800 °C with a high surface area and a well-developed porous structure exhibited the highest specific capacitance. In addition, carbon aerogel activated at 800 °C retained a considerable specific capacitance at a high current density even after 1000 cycles of charge/discharge. Therefore, it is concluded that carbon aerogel activated with KOH at 800 °C can serve as an efficient electrode material for commercial EDLC with a high power density.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
P. Sharma and T.S. Bhatti, Energy Convers. Manage., 51, 2901 (2010).
E. Frackowiak, Q. Abbas and F. Béguin, J. Energy Chem., 22, 226 (2013).
S. L. Candelariaa, Y. Shao, W. Zhouc, X. Li, J. Xiao, J.-G. Zhang, Y. Wang, J. Liu, J. Li and G. Cao, Nano Energy, 1, 195 (2012).
M.-G. Jeong, K. Zhuo, S. Cherevko and C.-H. Chung, Korean J. Chem. Eng., 29, 1802 (2012).
A. Thambidurai, J.K. Lourdusamy, J.V. John and S. Ganesan, Korean J. Chem. Eng., 31, 268 (2014).
E. Frackowiak and F. Béguin, Carbon, 39, 937 (2001).
L. Wei and G. Yushin, Nano Energy, 1, 552 (2012).
E. Jeong, M.-J. Jung, S.H. Cho, S. I. Lee and Y.-S. Lee, Colloids Surf., A, 377, 243 (2011).
A. Elmouwahidi, Z. Zapata-Benabithe, F. Carrasco-Marin and C. Moreno-Castilla, Bioresour. Technol., 111, 185 (2012).
Y. Zhang, H. Feng, X. Wu, L. Wang, A. Zhang, T. Xia, H. Dong, X. Li and L. Zhang, Int. J. Hydrog. Energy, 34, 4889 (2009).
Y. Guo, Z.-q. Shi, M.-m. Chen and C.-y. Wang, J. Power Sources, 252, 235 (2014).
Y. Huang, J. Liang and Y. Chen, Small, 8, 1805 (2012).
S.R. C. Vivekchand, C. S. Rout, K. S. Subrahmanyam, A. Govindaraj and C.N. R. Rao, J. Chem. Sci., 120, 9 (2008).
S.D. Perera, A.D. Liyanage, N. Nijem, J. P. Ferraris, Y. J. Chabal and K. J. Balkus Jr., J. Power Sources, 230, 130 (2013).
Z.-S. Wu, G. Zhou, L.-C. Yin, W. Ren, F. Li and H.-M. Cheng, Nano Energy, 1, 107 (2012).
S.-H. Yoon, S. Lim, Y. Song, Y. Ota, W. Qiao, A. Tanaka and I. Mochida, Carbon, 42, 1723 (2004).
J. Zhong, Z. Yang, R. Mukherjee, A.V. Thomas, K. Zhu, P. Sun, J. Lian, H. Zhu and N. Koratkar, Nano Energy, 2, 1025 (2013).
Z. Niu, P. Luan, Q. Shao, H. Dong, J. Li, J. Chen, D. Zhao, L. Cai, W. Zhou, X. Chen and S. Xie, Energy Environ. Sci., 5, 8726 (2012).
R.W. Pekala, J. C. Farmer, C.T. Alviso, T.D. Tran, S.T. Mayer, J.M. Miller and B. Dunn, J. Non-Cryst. Solids, 225, 74 (1998).
X. Wu and W. Jia, Chem. Eng. J., 245, 210 (2014).
C. Robertson, Micropor. Mesopor. Mater., 179, 151 (2013).
S.-W. Hwang and S.-H. Hyun, J. Non-Cryst. Solids, 347, 238 (2004).
X. Wang, X. Wang, L. Liu, L. Bai, H. An, L. Zheng and L. Yi, J. Non- Cryst. Solids, 357, 793 (2011).
Y. J. Lee, G.-P. Kim, Y. Bang, J. Yi, J. G. Seo and I. K. Song, Mater. Res. Bull., 50, 240 (2014).
K. Xia, Q. Gao, J. Jiang and J. Hu, Carbon, 46, 1718 (2008).
J.M.V. Nabaisa, P. Nunes, P. J.M. Carrott, M.M.L.R. Carrott, A.M. García and M. A. Díaz-Díez, Fuel Process. Technol., 89, 262 (2008).
S.-Y. Lee and S.-J. Park, J. Solid State Chem., 207, 158 (2013).
Ö. Sahin and C. Saka, Bioresour. Technol., 136, 163 (2013).
S.-Y. Lee and S.-J. Park, J. Colloid Interface Sci., 389, 230 (2013).
L.K.C. de Souza, N.P. Wickramaratne, A.S. Ello, M. J.F. Costa, C. E. F. da Costa and M. Jaroniec, Carbon, 65, 334 (2013).
X.-Y. Zhao, S.-S. Huang, J.-P. Cao, S.-C. Xi, X.-Y. Wei, J. Kamamoto and T. Takarada, J. Anal. Appl. Pyrol., 105, 116 (2014).
B. Xu, F. Wu, Y. Sub, G. Cao, S. Chen, Z. Zhou and Y. Yang, Electrochim. Acta, 53, 7730 (2008).
C. Lei, F. Markoulidis, Z. Ashitaka and C. Lekakou, Electrochim. Acta, 92, 183 (2013).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Kwon, S.H., Lee, E., Kim, BS. et al. Preparation of activated carbon aerogel and its application to electrode material for electric double layer capacitor in organic electrolyte: Effect of activation temperature. Korean J. Chem. Eng. 32, 248–254 (2015). https://doi.org/10.1007/s11814-014-0215-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-014-0215-z