Abstract
The growth performance, nutrient removal, lipid accumulation and morphological changes of Cyanobacterium aponinum OUC1 and Scenedesmus obliquus which were cultured in secondary effluents from two wastewater treatment plants: Tuandao Wastewater Treatment Plant (ETD) and Licun River Wastewater Treatment Plant (ELR) were investigated. The results showed that both C. aponinum OUC1 and S. obliquus have superior growth performances in both undiluted effluents, while the better of them was that in ETD effluent, with cell densities of C. aponinum OUC1 and S. obliquus increased by 159% and 66% over that of BG11 (control), respectively. Regarding nutrient removal, S. obliquus could completely remove inorganic phosphorus, and decrease ammonia nitrogen in ETD effluent by 81%. In addition, both C. aponinum OUC1 and S. obliquus cultivated in ETD exhibited extraordinary potential for biofuel production, increasing lipid productivities by 133% and 89% of that cultivated in ELR, respectively. As to ultrastructural changes, the differences in the lipoidal globules and glycogen granules of S. obliquus and C. aponinum OUC1 among the ETD and ELR treatments were mostly related to phosphorus limitations. The findings from this research reveal the probability using the secondary effluents as cultivation media to enhance algal biomass, nutrient removal and lipid productivity.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
AlMomani, F. A., and Örmeci, B., 2016. Performance of Chlorella vulgaris, neochloris oleoabundans, and mixed indigenous microalgae for treatment of primary effluent, secondary effluent and centrate. Ecological Engineering, 95: 280–289, DOI: https://doi.org/10.1016/j.ecoleng.2016.06.038.
Arbib, Z., Ruiz, J., Álvarez-Díaz, P., Garrido-Pérez, C., and Perales, J. A., 2014. Capability of different microalgae species for phytoremediation processes: Wastewater tertiary treatment, CO2 bio-fixation and low cost biofuels production. Water Research, 49: 465–474, DOI: https://doi.org/10.1016/j.watres.2013.10.036.
Arias, D. M., Rueda, E., Garcia-Galan, M. J., Uggetti, E., and Garcia, J., 2019. Selection of cyanobacteria over green algae in a photo-sequencing batch bioreactor fed with wastewater. Science of the Total Environment, 653: 485–495, DOI: https://doi.org/10.1016/j.scitotenv.2018.10.342.
Bligh, E. G., and Dyer, W. J., 1959. A rapid method of total lipid extraction and purification. Canadian Journal of Biochemistry and Physiology, 37(8): 911–917, DOI: https://doi.org/10.1139/o59-099.
Cai, T., Park, S. Y., and Li, Y., 2013. Nutrient recovery from wastewater streams by microalgae: Status and prospects. Renewable and Sustainable Energy Reviews, 19: 360–369, DOI: https://doi.org/10.1016/j.rser.2012.11.030.
Chen, G. Y., Zhao, L., and Qi, Y., 2015. Enhancing the productivity of microalgae cultivated in wastewater toward biofuel production: A critical review. Applied Energy, 137: 282–291, DOI: https://doi.org/10.1016/j.apenergy.2014.10.032.
Cheng, Q. L., Xu, L. G, Cheng, F. M., Pan, G., and Zhou, Q. F., 2018. Bicarbonate-rich wastewater as a carbon fertilizer for culture of Dictyosphaerium sp. of a giant pyrenoid. Journal of Cleaner Production, 202: 439–443, DOI: https://doi.org/10.1016/j.jclepro.2018.08.066.
Courchesne, N. M. D., Parisien, A., Wang, B., and Lan, C. Q., 2009. Enhancement of lipid production using biochemical, genetic and transcription factor engineering approaches. Journal of Biotechnology, 141(1): 31–41, DOI: https://doi.org/10.1016/j.jbiotec.2009.02.018.
Deegan, L. A., Johnson, D. S., Warren, R. S., Peterson, B. J., Fleeger, J. W., Fagherazzi, S., and Wollheim, W. M., 2012. Coastal eutrophication as a driver of salt marsh loss. Nature, 490(7420): 388–392, DOI: https://doi.org/10.1038/nature11533.
Diniz, G. S., Silva, A. F., Araujo, O. Q. F., and Chaloub, R. M., 2017. The potential of microalgal biomass production for biotechnological purposes using wastewater resources. Journal of Applied Phycology, 29: 821–832, DOI: https://doi.org/10.1007/s10811-016-0976-3.
Fujimoto, N., Sudo, R., Sugiura, N., and Inamori, Y., 1997. Nutrient-limited growth of microcystis aeruginosa and phormidium tenue and competition under various N:P supply ratios and temperatures. Limnology and Oceanography, 42(2): 250–256, DOI: https://doi.org/10.4319/lo.1997.42.2.0250.
Ge, S. J., and Champagne, P., 2016. Nutrient removal, microalgal biomass growth, harvesting and lipid yield in response to centrate wastewater loadings. Water Research, 88: 604–612, DOI: https://doi.org/10.1016/j.watres.2015.10.054.
Gojkovic, Z., Lindberg, R. H., Tysklind, M., and Funk, C., 2019. Northern green algae have the capacity to remove active pharmaceutical ingredients. Ecotoxicology and Environmental Safety, 170: 644–656, DOI: https://doi.org/10.1016/j.ecoenv.2018.12.032.
Gupta, S. K., Ansari, F. A., Shriwastav, A., Sahoo, N. K., Rawat, I., and Bux, F., 2016. Dual role of Chlorella sorokiniana and Scenedesmus obliquus for comprehensive wastewater treatment and biomass production for bio-fuels. Journal of Cleaner Production, 115: 255–264, DOI: https://doi.org/10.1016/j.jclepro.2017.01.144.
Ho, S. H., Chen, C. Y., and Chang, J. S., 2012. Effect of light intensity and nitrogen starvation on CO2 fixation and lipid/carbohydrate production of an indigenous microalga Scenedesmus obliquus CNW-N. Bioresource Technology, 113: 244–252, DOI: https://doi.org/10.1016/j.biortech.2011.11.133.
Kulaev, I. S., and Vagabov, V. M., 1983. Polyphosphate metabolism in micro-organisms. Advances in Microbial Physiology, 24: 83–171, DOI: https://doi.org/10.1016/S0065-2911(08)60385-9.
Li, X., Hu, H., and Yang, J., 2010. Lipid accumulation and nutrient removal properties of a newly isolated freshwater microalga, Scenedesmus sp. LX1, growing in secondary effluent. New Biotechnology, 27(1): 59–63, DOI: https://doi.org/10.1016/j.nbt.2009.11.006.
Li, Y., Han, D., Sommerfeld, M., and Hu, Q., 2011. Photosynthetic carbon partitioning and lipid production in the oleaginous microalga Pseudochlorococcum sp. (Chlorophyceae) under nitrogen-limited conditions. Bioresource Technology, 102(1): 123–129, DOI: https://doi.org/10.1016/j.biortech.2010.06.036.
Lomas, M. W., and Gilbert, P. M., 2000. Comparisons of nitrate uptake, storage, and reduction in marine diatoms and flagellates. Journal of Phycology, 36(5): 903–913, DOI: https://doi.org/10.1046/j.1529-8817.2000.99029.x.
Lorenzen, J., Larsen, L. H., Kjær, T., and Revsbech, N. P., 1998. Biosensor determination of the microscale distribution of nitrate, nitrate assimilation, nitrification, and denitrification in a diatom-inhabited freshwater sediment. Applied and Environmental Microbiology, 64: 3264–3269, DOI: https://doi.org/10.1002/(SICI)1097-0290(19980905)59:5<651::AID-BIT17>3.0.CO;2-C.
Marella, T. K., Datta, A., Patil, M. D., Dixit, S., and Tiwari, A., 2019. Biodiesel production through algal cultivation in urban wastewater using algal floway. Bioresource Technology, 280: 222–228, DOI: https://doi.org/10.1016/j.biortech.2019.02.031.
Markou, G., and Georgakakis, D., 2011. Cultivation of filamentous cyanobacteria (blue-green algae) in agro-industrial wastes and wastewaters: A review. Applied Energy, 88(10): 3389–3401, DOI: https://doi.org/10.1016/j.apenergy.2010.12.042.
Markou, G., Chatzipavlidis, I., and Georgakakis, D., 2012. Carbohydrates production and bio-flocculation characteristics in cultures of Arthrospira (Spirulina) platensis: Improvements through phosphorus limitation process. BioEnergy Research, 5(4): 915–925, DOI: https://doi.org/10.1007/s12155-012-9205-3.
Martin, C., Noüe, J. D. L., and Picard, G., 1985. Intensive cultivation of freshwater microalgae on aerated pig manure. Biomass, 7(4): 245–259, DOI: https://doi.org/10.1016/0144-4565(85)90064-2.
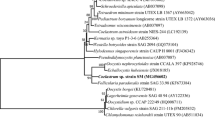
Meng, F. P., Cui, H. W., Wang, Y. J., and Li, X. L., 2018. Responses of a new isolated Cyanobacterium aponinum strain to temperature, pH, CO2 and light quality. Journal of Applied Phycology, 30(3): 1525–1532, DOI: https://doi.org/10.1007/s10811-018-1411-1418.
Ministry of Environmental Protection of China (MEP), 1987. Water quality-determination of nitrogen (nitrite)-spectro-photometric method (GB 7493-87), China Environmental Science Press, Beijing, China.
Ministry of Environmental Protection of China (MEP), 1989. Water quality-determination of total phosphorus-ammonium molybdate spectrophotometric method (GB 11893-89), China Environmental Science Press, Beijing, China.
Ministry of Environmental Protection of China (MEP), 2007. Water quality-determination of nitrate-nitrogen-ultraviolet spectrophotometry (HJ/T 346-2007), China Environmental Science Press, Beijing, China.
Ministry of Environmental Protection of China (MEP), 2009. Water quality-determination of ammonia nitrogen-nessler’s reagent spectrophotometry (HJ 535-2009), China Environmental Science Press, Beijing, China.
Mirzaei, R., Mesdaghinia, A., Hoseini, S. S., and Yunesian, M., 2019. Antibiotics in urban wastewater and rivers of Tehran, Iran: Consumption, mass load, occurrence, and ecological risk. Chemosphere, 221: 55–66, DOI: https://doi.org/10.1016/j.chemosphere.2018.12.187.
Quintana, N., Kooy, F. V. D., Rhee, M. D., Voshol, G. P., and Verpoorte, R., 2011. Renewable energy from Cyanobacteria: Energy production optimization by metabolic pathway engineering. Applied Microbiology and Biotechnology, 91(3): 471–490, DOI: https://doi.org/10.1007/s00253-011-3394-0.
Reynolds, E. S., 1963. The use of lead citrate at high pH as an electron-opaque stain in electron microscopy. The Journal of Cell Biology, 17(1): 208, DOI: https://doi.org/10.1083/jcb.17.1.208.
Ricci, J. N., Morton, R., Kulkarni, G., Summers, M. L., and Newman, D. K., 2017. Hopanoids play a role in stress tolerance and nutrient storage in the cyanobacterium Nostoc punctiforme. Geobiology, 15: 173–183, DOI: https://doi.org/10.1111/gbi.12204.
Sakthivel, R., Ramesh, K., Purnachandran, R., and Mohamed, S. P., 2018. A review on the properties, performance and emission aspects of the third generation biodiesels. Renewable and Sustainable Energy Reviews, 82: 2970–2992, DOI: https://doi.org/10.1016/j.rser.2017.10.037.
Stanier, R. Y., Kuniawa, R., Mandel, M., and Cohen-Bazire, G., 1971. Purification and properties of unicellular blue-green algae (order Chroococcales). Bacteriological Reviews, 35(2): 171–205.
Tang, D. Y. Y., Khoo, K. S., Chew, K. W., Tao, Y., Ho, S. H., and Show, P. L., 2020. Potential utilization of bioproducts from microalgae for the quality enhancement of natural products. Bioresource Technology, 304: 122997, DOI: https://doi.org/10.1016/j.biortech.2020.122997.
Tillberg, J. E., and Rowley, J. R., 1989. Physiological and structural effects of phosphorus starvation on the unicellular green alga Scenedesmus. Physiologia Plantarum, 75(3): 315–324, DOI: https://doi.org/10.1111/j.1399-3054.1989.tb04633.x.
Tiwari, B., Verma, E., Chakraborty, S., Srivastava, A. K., and Mishra, A. K., 2018. Tolerance strategies in cyanobacterium Fischerella sp. under pesticide stress and possible role of a carbohydrate-binding protein in the metabolism of methyl parathion (MP). International Biodeterioration & Biodegradation, 127: 217–226, DOI: https://doi.org/10.1016/j.ibiod.2017.11.025.
Tripathi, R., Gupta, A., and Thakur, I. S., 2019. An integrated approach for phycoremediation of wastewater and sustainable biodiesel production by green microalgae, Scenedesmus sp. Istga1. Renewable Energy, 135: 617–625, DOI: https://doi.org/10.1016/j.renene.2018.12.056.
Tuantet, K., Temmink, H., Zeeman, G., Janssen, M., Wijffels, R. H., and Buisman, C. J. N., 2014. Nutrient removal and microalgal biomass production on urine in a short light-path photobioreactor. Water Research, 55: 162–174, DOI: https://doi.org/10.1016/j.watres.2014.02.027.
Wang, L., Min, M., Li, Y., Chen, P., Chen, Y., Liu, Y., Wang, Y., and Ruan, R., 2010. Cultivation of green algae Chlorella sp. in different wastewaters from municipal wastewater treatment plant. Applied Biochemistry and Biotechnology, 162(4): 1174–1186, DOI: https://doi.org/10.1007/s12010-009-8866-7.
Yao, L. L., Shi, J. Y., and Miao, X. L., 2015. Mixed wastewater coupled with CO2 for microalgae culturing and nutrient removal. PLoS One, 10(9): 16, DOI: https://doi.org/10.1371/journal.pone.0139117.
Yu, C., Huang, X., Chen, H., Godfray, H. C. J., Wright, J. S., Hall, J. W., Gong, P., Ni, S., Qiao, S., Huang, G., Xiao, Y., Zhang, J., Feng, Z., Ju, X., Ciais, P., Stenseth, N. C., Hessen, D. O., Sun, Z., Yu, L., Cai, W., Fu, H., Huang, X., Zhang, C., Liu, H., and Taylor, J., 2019. Managing nitrogen to restore water quality in China. Nature, 567: 516–520, DOI: https://doi.org/10.1038/s41586-019-1001-1.
Zhu, L., Hiltunen, E., Shu, Q., Zhou, W., Li, Z., and Wang, Z., 2014. Biodiesel production from algae cultivated in winter with artificial wastewater through pH regulation by acetic acid. Applied Energy, 128: 103–110, DOI: https://doi.org/10.1016/j.apenergy.2014.04.039.
Acknowledgement
This work was supported by the National Marine Hazard Mitigation Service, Ministry of Natural Resources of the People’s Republic of China through its Commissioned Research Scheme (No. 2019005AC).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, B., Meng, F., Cui, H. et al. Microalgal Cultivation in Secondary Effluents: Enhancement of Algal Biomass, Nutrient Removal, and Lipid Productivity. J. Ocean Univ. China 19, 1461–1470 (2020). https://doi.org/10.1007/s11802-020-4480-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11802-020-4480-7