Abstract
Earthworms play an important role in soil processes and ecosystem services. Soil fauna responds quickly to any alteration in the soil environment induced by land-use changes. Earthworm distribution is usually diverse, and their populations fluctuate in relation to different soil variables and land use patterns of the soil. However, little is known about the impact of soil organic carbon pools (abiotic factors) and land-use changes on earthworm communities in north western Himalaya. This study examined distribution and diversity indices of earthworms and their relation to physico-chemical properties of soil. Earthworms were sampled from three different land-use systems (mixed forest, agricultural land and tea garden) in every season (summer, rainy and winter) from two different depths (0–15 and 15–30 cm) from April 2019 to March 2021. We further identified earthworms and investigated physico-chemical properties of soil along with different carbon pools. A total of 13 earthworm species belonging to three families (Megascolecidae, Lumbricidae, Octochaetidae) were identified, out of which Amynthas corticis (Kinberg, 1867) was the most abundant species and found in all land-use systems. Mixed forest showed the highest H index, Margalef index and also have good nutrient content. Principal Component Analysis (PCA) revealed a significant positive impact of abiotic factors on the distribution of earthworms. Our results indicate that earthworm diversity indices are strongly and positively correlated with the organic carbon pools of soil, which might become the potential factor in influencing the earthworm community pattern in north western Himalaya.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Climate change and land-use change threatened the ecological communities (Tylianakis et al. 2008; Newbold et al. 2015), thus affecting the population structure of the organisms (Bardgett et al. 2013). Soil biodiversity supports a wide range of ecosystem services (Wall et al. 2013) thereafter soil organisms are considered as indicators of the status and ‘health’ of the soil (Doube and Schmidt 1997; Capelle et al. 2012; Briones 2014). Among soil organisms, earthworms act as a key agent in maintaining soil fertility and sustainability (Jones et al. 1994).
Earthworms are important ‘ecosystem engineers’ (Doran and Zeiss 2000) as their bioturbation activity has a crucial impact on physical, chemical and biological characteristics of the soil (Bartlett et al. 2010; Tiwari et al. 2017). The potential role of earthworms in soil formation and their contribution in the functioning of soil ecosystems with varying species diversity was well explained (Lavelle and Spain 2001; Jouquet et al. 2006; Ngo et al. 2012). Earthworms represent a great portion of soil biomass which can increase to 80% (Yasmin and D’Souza 2010) in subtropical, tropical and temperate regions (Nainawat and Nagendra 2001). Many authors reported that earthworm diversity was severely affected due to large-scale destruction of the natural ecosystem (Lavelle et al. 1994; Bhaduria and Ramakrishnan 1991).
According to Darwin (1881), earthworms are nature’s plough and always show a positive impact on soil characteristics (Jones et al. 2006). Furthermore, earthworms also increase crop yield upto 25–35% (Van Groenigen et al. 2014). The fact that earthworms are essential for the entire ecosystem because they perform numerous functions like nutrient cycling, humus and aggregate formation. They have spurred keen interest in scientists in gaining an insight of the vast resource of soil worm diversity.
Overall, on the basis of physiology, feeding and burrowing behavior, earthworms are categorized into three ecological groups: endogeic, epigeic and anecic (Bouche 1972). Their density and distribution pattern are heterogeneous and totally depend on geographical and climatic factors of the area (Debbarma and Chaudhuri 2019; Bhadauria et al. 2000). Furthermore, Bhadauria and Ramakrishnan (1989) concluded that a variety of biotic and abiotic factors fluctuate the dispersal pattern of regional earthworm species.
Many authors found that different land-use systems have diverse vegetation cover, litter inputs and thereby affect the physiochemical properties and microbial diversity of the soil (Birkhofer et al. 2012; Xie et al. 2018; Marshall and Lynch 2020; Singh et al. 2020a). The earthworm community is affected predominantly by land-use system, soil organic carbon and soil moisture, thus being very sensitive to these changes (Bini et al. 2013; Dempsey et al. 2013). Ultimately, these changes directly influence the composition, diversity and distribution of earthworm communities in different agro-climatic regions (Lalthanzara et al. 2011). Since information related to earthworm diversity in Uttarakhand is limited, hence the present study has been undertaken with a view to ascertain species diversity of earthworms under different land-use systems.
There are approximately more than 7000 species of earthworm widely dispersed all over the world (Grdisa 2013; Wetzel and Reynolds 2021) out of which 3000–3500 are considered as valid (Csuzdi 2012). India is one of the major mega biodiversity country, accounting for 11.1% of total earthworm diversity in the world. (Chaudhuri and Nath 2011; Suthar 2011). Additionally, 505 earthworm species of 10 different families belonging to 69 genera have been discovered from India (Bora et al. 2021). At present, few studies have focused on taxonomic richness of earthworms in the western Himalayan region due to inaccessibility of some tough terrains. Hence, these regions have not yet been explored totally. Moreover, proper scientific investigations are required to explore the unique soil earthworm habitats of different land-use systems of western Himalaya. On the other hand, Kaushal and Bisht (1994) investigated the earthworm population density in the pasture land of Kumaun Himalayas. Later, Bhadauria et al. (2000) examined the effect of natural and regenerating ecosystems on the diversity and distribution of earthworm populations of central Himalaya and reported a significant population decline of endemic earthworm species. Rajwar et al. (2018) focused on earthworm populations of Kumaun Himalaya, however, all these studies were limited to a single land-use system. The gaps present in the information on comparative studies made in the past on different land-use systems and their effects on earthworm population density in Kumaun Himalayas has led to the present investigation. In order to gain a better understanding on the effect of soil organic carbon pools and abiotic factors on the earthworm populations, we carried out a study in north western Himalaya to understand (a) the population dynamics of earthworms in three different land-use systems (mixed forest, agricultural land and tea garden) along with their soil depth and seasonal variation and (b) how soil organic carbon pools and abiotic factors affect earthworm diversity.
Material and methods
Study area
Geographically, Champawat lies between 29°20’09.97” N and 80°05’27.70” E at an altitude of about 1615 m above mean sea level in the Himalayan region. The climate of the study area is sub–temperate with distinct warm (May–June), cold (December– January), and general dryness seasons in a year. The geographical coverage of Champawat is about 1613 km2 with 1085.62 mm mean annual rainfall. Deep, well-drained, coarse, and fine loamy soil with a loamy surface is found. Three different land-use systems (mixed forest, agricultural land, and tea garden) were selected for the study (Fig. 1). The complete description of the study sites is given in Table 1. Different land-use systems have different vegetation and thereby each vegetation type provides a different soil environment for the survival of earthworms. Soil and earthworm sampling was done during two consecutive years in pre-monsoon, monsoon, and post-monsoon seasons of the year 2019 and 2020 in the district Champawat (Uttarakhand, India). Monsoon and pre-monsoon periods have a maximum and high maturity of earthworms.
Earthworms sampling
Earthworms were sampled by using the tropical soil biology methodology from three different land-use systems. On each site, random sampling (25 × 25 cm) of the earthworms for three consecutive seasons was done. Earthworms were extracted by the hand-sorting method, then washed and properly stretched and fixed in 4% formalin (Anderson and Ingram 1993).
The collected earthworm samples were placed in polythene bags that were properly labelled with the place name, date of collection, and other requisite details. Further taxonomic identification upto species level was carried out at Zoological Survey of India (ZSI), Dehradun.
Soil sampling
Soil samples were collected from two incremental depths of 0–15 cm and 15–30 cm. The samples taken from the corresponding depths were thoroughly mixed and bulked to form one composite sample. Then, it was brought to the laboratory and was air-dried, crushed, and sieved through a 2 mm sieve. Thereafter, it was kept in polythene bags for subsequent analysis. Soil pH (H2O) and electrical conductivity were measured using a digital pH meter and conductivity meter. Organic carbon was determined by the wet digestion method (Walkey and Black 1934). Nitrogen was analyzed by acid digestion Kjeldahl procedure as given by Anderson and Ingram (1993). Soil moisture was determined by a gravimetric wet weight method. The content of K in soil was analyzed by a systronics flame photometer. Phosphorus was determined by using a spectrophotometer. The analysis of bulk density was done as per the protocols of Anderson and Ingram (1993). Particulate organic carbon was determined by using chemical dispersion technique of Cambardella (1992). An oxidation method by Blair et al. (1995) was used to analyze the value of labile organic carbon in the soil.
Statistical analysis
The data for physico-chemical, POC and LOC properties of the soil at successive depths of three consecutive seasons was represented as mean ± S.E. The results obtained were analyzed using Past, SPSS 20, and the significance level was set at α = 0.05. Pearson’s correlation analysis was performed to determine a correlation between the biological attributes of earthworms and soil parameters. The diversity indices such as Shannon-Weiner diversity index, species evenness, and Margalef’s index were used to compare the richness and diversity of earthworms among different land-use systems. All these indices were calculated by using the standard calculations by Shannon and Wiener (1949), Margalef (1958), and Pielou (1966), respectively. Principal Component Analysis (PCA) was used to characterize the major component of the soil with one or more variables that affects the diversity and distribution of earthworms at a particular site by using Varimax rotation with Kaiser Normalization.
Results
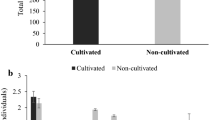
A total of 13 species belonging to three families viz. Megascolecidae, Lumbricidae, and Octochaetidae were identified from all three different land-use systems by following standard protocols by Julka and Senapati (1987). All the species were abundantly found in the rainy season and minimum in the winter season across all the sites due to extreme cold and dry weather conditions. Most of the species were present at a depth of 0–30 cm and thus possess different ecological categories (Table 2). Out of these, eight species belong to the family Megascolecidae (Amynthas corticis, Metaphire posthuma, Metaphire houlleti, Metaphire birmanica, Perionyx excavatus, Perionyx bainii, Perionyx nainianus and Lampito mauritii) and three species belong to family Lumbricidae (Eisenia fetida, Bimastos parvus and Aporrectodea trapezoides). The rest of the two species belong to the family Octochaetidae (Eutyphoeus waltoni, Eutyphoeus nainianus). Mixed forest recorded the highest earthworm diversity with 13 species, followed by agricultural land with ten species. On the other hand, tea garden had only eight species. Amynthas corticis was dominant earthworm species and was present in all land-use systems. The Shannon-Wiener diversity (H) index ranged from 0.85 to 2.40, Margalef species richness (DMg) index ranged from 0.89 to 2.19, and species evenness ranged from 0.91 to 1.99 across different sites (Table 3). Due to the presence of numerous earthworm species, mixed forest registered high Shannon-Weiner diversity index and species richness values (Fig. 2).
The pedological characteristics and carbon pools of different land-use varied depth wise and seasonally (Table 4). Moisture content at all sites was in the range of 45 − 15% with the highest value (45%) during the rainy season and the lowest (15%) in winter. The pH was found acidic in mixed forest irrespective to the seasons and depth whereas agricultural land and tea garden had basic to moderately acidic pH. Electrical conductivity and organic carbon were recorded highest at the surface layer of mixed forests. Also, their value increased during the rainy season due to the accumulation of a thick humus layer. The NPK content also varied significantly, with the highest value in a mixed forest in its surface layer. Moreover, the values of all variables significantly declined along with the vertical soil profile. But, bulk density gradually increased depth-wise and was recorded maximum in mixed forest. The labile and particulate organic carbon of all three land-use systems provides a unique insight (Table 5). All the land-use systems showed higher values of particulate organic carbon during the rainy season at a depth of 0–15 cm. Indeed, its higher value reflects the superiority to microbial activity and nutrient cycling. Labile organic carbon was maximum in the mixed forest because of the constant supply of decomposable leaf litter throughout the year. The values of soil characteristics were found significantly higher in the mixed forest in comparison with agricultural land and tea garden. It was evident that physico chemical attributes regulate the distribution of earthworm species, therefore the preferred range of physico-chemical soil characteristics of earthworms under different land-use systems are given in Table 6.
Pearson correlation analysis showed an association of earthworm diversity and indices with soil physico-chemical properties across the three land-use systems (Table 7). A highly positive significant correlation was observed between density and diversity with organic carbon, nitrogen, potassium, bulk density, moisture content, particulate and labile organic carbon. However, pH showed a negative correlation with the earthworm density.
The Principal component analysis (PCA) was applied to 13 different biotic and abiotic parameters for three different land-use systems to identify the most important soil factors affecting the distribution and diversity indices of earthworm species. PCA resulted into two major principal components, viz. PC1 and PC2 contributed a total variance of 58.47% and 41.52%, respectively. The different factors, their loading values, respective eigenvalues, and total variance (%) for each component are given in Table 8. The variance in PC1 was due to EC, N, BD, MC, and LOC while in PC2, it was due to P and K. However, PC2 showed strong negative loading with pH. Earthworm diversity indices and soil properties have a significant direct relation. The biplot of PCA of 13 different variables using varimax rotation with the Kaiser Normalization method is given in Fig. 3.
Discussion
A total of 13 earthworm species belonging to three families were identified from different land-use systems of north-western Himalaya. Kandpal (2018) also reported Amynthas corticis, Metaphire houlleti, Metaphire posthuma, Perionyx excavatus, Aporrectodea trapezoides, Eisenia fetida from Kathgodam, Terai region of north-western Himalaya. Earthworm populations are very sensitive to land use practices. It was observed during the study that earthworm community composition was largely dominated by endogeic species in all land-use systems, particularly in mixed forest. According to Decaens et al. (2003) forest provides abundant trophic resources such as litter, and organic matter to support earthworm population. The findings of our study go in line with this statement. Vegetation type, abiotic factors, physical, chemical and organic carbon pools of soil directly influence the distribution and diversity of earthworms (Ramanujam et al. 2000).
In agricultural land, the diversity of earthworm species was low due to the physical disturbance of the soil. Various agricultural management practices like ploughing, and heavy soil cultivation damage the burrows of endogeic species. Our results corroborated with the findings of Amador et al. (2013) and Lagerlof et al. (2002) who documented that ploughing decreased the earthworm population. In the tea garden, eight endogeic species (out of 13) predominated, with no anecic and epi-anecic in sight. Our results are consistent with the conclusions of Harbowy et al. (1997), Senapati et al. (1999) and Jamatia and Chaudhuri (2017) who concluded that tea leaf litter consists of high content of polyphenols, making it less palatable for earthworms.
Temporal variations (temperature, rainfall) alter functional categories of earthworms, leading to spatial and temporal heterogeneity in earthworm population (Chaudhuri and Paliwal 2008; Suthar 2012). Altitude shapes environmental factors (Jacquier et al. 2020) and the environmental conditions of Kumaun Himalayas are altitude-dependent. Furthermore, our results showed that the earthworm diversity of all land-use systems tends to decrease seasonally (rainy > summer > winter). Indeed, the rainy season has high precipitation, humidity, and appropriate temperature that successfully sustain the lives of earthworms and support their reproductive cycle. Similar observations are also made by Joshi and Aga (2009) and Walsh and Johnson-Maynard (2016).
During the study, many earthworm species were absent in the winter season. This could be related to the presence of frost in the study area, as even moderate frost in the soil was considered lethal for earthworms by Hopp and Linder (1947).
The value of the Shannon–Wiener index usually ranges from 0 to 4. In the present study, the earthworm diversity indices, i.e. H index, Margalef, and species evenness were recorded as highest in the mixed forest due to good canopy cover with maximum litter accumulation and less involvement of anthropic factors. Our results were in agreement with Haokip and Singh (2012). Similar to our findings, Sharma and Bharadwaj (2014) observed a higher H index value in agricultural fields due to the use of organic manure. Furthermore, the lower H index value of tea garden in our present study supports the findings of Jamatia and Chaudhuri (2017) who reported the same because of an absence of a canopy with unpalatable leaf litter and extreme anthropogenic interferences.
The physico-chemical characteristics of soil, different organic carbon pools, and organic matter at a particular site helped in determining the earthworm distribution and diversity of that area. During the present investigation, a variety of environmental factors such as moisture content, bulk density, conductivity, pH, organic carbon, nitrogen, potassium, particulate, and labile organic carbon directly affected the earthworm abundance and hence diversity indices.
We have observed that moisture content plays an important role due to the cutaneous mode of respiration, thus acting as a critical factor for earthworm distribution. Earthworm biomass contains 70–90% water. Soil temperature and moisture are inversely proportional thereafter affecting earthworm population through modes of metabolism, respiration, growth, reproduction, and osmoregulation. Similar to this, Debbarma and Chaudhuri (2019) documented a significant and positive correlation between the moisture of pasture soil and density and biomass of earthworms.
pH regulates acidity and alkalinity in the soil, therefore, did not directly influence earthworm dynamics in a particular site, but indirectly alters other chemical processes in earthworm ultimately leading to affect nutrient availability. In the present study, pH varied from 4.9 to 7.8 across sites and seasons. Earthworms can tolerate a pH range of 5.0 to 8.0. Bisht et al. (2003) support our study by recording a pH ranging from 5.6 to 8.0 in the terai region of central Himalaya.
Both organic carbon pools of soil (labile and particulate) are significantly and positively correlated with the biological attributes of earthworms. Hence, these carbon pools are also regarded as a critical edaphic factor influencing the earthworm population in any land-use system of north-western Himalaya. Also, according to Bayranvand et al. (2017) total organic carbon has a stronger impact on soil biological activity. While on the other hand, organic carbon determines the type and nature of food for an earthworm. Li et al. (2018) and Rajkhowa et al. (2014) observed high earthworm abundance in the sites having high organic carbon content and vice versa, which is clearly observed in our findings.
Two principal components were identified PC1 and PC2. Liu et al. (2003) classified factor loadings as weak, moderate, and strong when absolute loading values were < 0.50, 0.50–0.75, and > 0.75, respectively. Although PC1 was due to EC, N, BD, MC, and LOC. EC maintains the salt concentration and earthworms can survive only at a moderate salt level. Singh et al. (2020b) described that the growth and development of earthworms are directly affected by soil bulk density, which is clearly represented in our study. PC2 represents chemical factors and had a strong positive loading of K and P. This validates the outcomes of Singh et al. (2016a), who confirms that Metaphire posthuma increases nitrogen and phosphorus content in the soil, thereafter making it easily available for the plants. Thus, PCA proved that earthworm communities along with their diversity indices are directly and positively correlated with the physico-chemical properties of soil. The present observations are in agreement with the findings of other previous studies of Sankar and Patnaik (2018) and Singh et al. (2020c). Also, according to Singh et al. (2016b) earthworm distribution varied according to soil habitat, vegetation type, soil tillage, and land-use pattern. Overall, the findings of the study clearly indicate that changes in the land-use system along with the altitude have a plethora of effects on earthworm community structure. These statements are corroborated by the findings of Rajwar et al. (2022), who documented higher earthworm population density in forest systems as compared to agro-forest and cultivated systems in the Kumaun Himalayas.
Conclusion
Our study observed a pronounced effect of abiotic factors on the composition and diversity of the earthworm communities under three different land-use systems. In this study, 13 earthworm species have been documented. In particular, mixed forest supports the abundant and maximum earthworm population density as compared to the agricultural land and tea garden. To our knowledge, this is the first report on the distribution pattern of earthworms from Champawat district of north-western Himalaya. Amynthas corticis was the most dominant species that has found in all land-use systems. It was also noticed that the rainy season provides sufficient moisture to earthworms to sustain their life irrespective of land use. PCA showed that soil variables have a significant positive effect on the distribution and diversity indices of earthworm across three land-use patterns. Indeed, our study also revealed that LOC and OC have a strong positive impact on the earthworm community. Further, this study provides baseline information in establishing a relationship between earthworm population and different land-use systems in north-western Himalayas.
Data Availability
All the data generated or analysed during this study are included in this published article.
References
Amador JA, Winiarski K, Sotomayor-Ramirez D (2013) Earthworm communities along a forest–coffee agroecosystem gradient: preliminary evidences supporting the habitat-dependent feeding hypothesis. Trop Ecol 54:365–374
Anderson JM, Ingram JSI (1993) Tropical soil biology and fertility: a handbook of methods. CAB International. The Cambrian News, Aberstwyth, United Kingdom
Bardgett RD, Manning P, Morrien E, De Vries FT (2013) Hierarchical responses of plant-soil interactions to climate change: consequences for the global carbon cycle. J Ecol 101(2):334–343. https://doi.org/10.1111/1365-2745.12043
Bartlett MD, Briones MJI, Neilson R, Schmidt O, Spurgeon D, Creamer RE (2010) A critical review of current methods in earthworm ecology: from individuals to populations. Eur J Soil Biol 46(2):67–73. https://doi.org/10.1016/j.ejsobi.2009.11.006
Bayranvand M, Kooch Y, Rey A (2017) Earthworm population and microbial activity temporal dynamics in a Caspian Hyrcanian mixed forest. Eur J For Res 136:447–456. https://doi.org/10.1007/s10342-017-1044-5
Bhadauria T, Ramakrishnan PS (1989) Earthworm population dynamics and contribution to nutrient cycling during cropping and fallow phases of shifting agriculture (jhum) in north east India. J Appl Ecol 26(2):505–520
Bhadauria T, Ramakrishnan PS, Srivastava KN (2000) Diversity and distribution of endemic and exotic earthworms in natural and regenerating ecosystems in the central Himalayas, India. Soil Biol Biochem 32(14):2045–2054
Bhaduria T, Ramakrishnan PS (1991) Population dynamics of earthworms and their activities in forest ecosystems of north-east India. J Trop Ecol 7(3):305–318. https://doi.org/10.1017/S0266467400005575
Bini D, Cristiane ADS, Kellen BDC, Nagomi K, Galdino A, Waldemar Z, Marco AN (2013) Effects of land use on soil organic carbon and microbial processes associated with soil health in southern Brazil. Eur J Soil Biol 55(3):117–123. https://doi.org/10.1016/j.ejsobi.2012.12.010
Birkhofer K, Schoning I, Alt F, Herold N, Klarner B, Maraun M, Begerow D (2012) General relationships between abiotic soil properties and soil biota across spatial scales and different landuse types. PLoS One 7(8):e43292. https://doi.org/10.1371/journal.pone.0043292
Bisht R, Pandey H, Bharti D, Kaushal BR (2003) Population dynamics of earthworms (Oligochaeta) in cultivated soils of central Himalayan Tarai region. Trop Ecol 44(2):229–234
Blair GJ, Lefroy RDB, Lisle L (1995) Soil carbon fractions based on their degree of oxidation, and the development of a carbon management index for agricultural systems. Aust J Agric Res 46(7):1459–1466. https://doi.org/10.1071/AR9951459
Bouche MB (1972) Lombriciens de France. Ecologie et systemtique. INRA, Paris
Bora S, Bisht SS, Reynolds JW (2021) Global diversity of earthworms in various countries and continents: a short review. Megadrilogica 26(9):127–154
Briones MJI (2014) Soil fauna and soil functions: a jigsaw puzzle. Front Environ Sci 2(7):1–19. https://doi.org/10.3389/fenvs.2014.00007
Cambardella CA, Elliott ET (1992) Particulate soil organic matter changes across a grassland cultivation sequence. Soil Sci Soc Am J 56(3):777–783. https://doi.org/10.2136/sssaj1992.03615995005600030017x
Chaudhuri PS, Nath S (2011) Community structure of earthworms under rubber plantations and mixed forests in Tripura India. J Environ Biol 32(5):537–541
Chaudhuri PS, Paliwal SNR (2008) Earthworm population of rubber plantations (Hevea brasiliensis) in Tripura, India. Trop Ecol 49(2):225–234
Csuzdi CS (2012) Earthworm species, a searchable database. Opusc Zool Budapest 43(1):97–99. https://doi.org/10.5281/zenodo.1045525
Darwin CR (1881) The formation of vegetable mould, through the action of worms. John Murray, London
Debbarma B, Chaudhuri P (2019) Earthworm communities under pasture ecosystems in Tripura, India. Int J Ecol Environ Sci 45(2):179–190
Decaens T, Mariani L, Betancourt NS, Jimenez JJ (2003) Seed dispersion by surface casting activities of earthworms in colombian grasslands. Acta Oecol 24:175–185. https://doi.org/10.1016/S1146-609X(03)00083-3
Dempsey MA, Fisk MC, Yavitt JB, Fahey TJ, Balser TC (2013) Exotic earthworms alter soil microbial community composition and function. Soil Biol Biochem 67:263–270. https://doi.org/10.1016/j.soilbio.2013.09.009
Doran JW, Zeiss MR (2000) Soil health and sustainability: managing the biotic component of soil quality. Appl Soil Ecol 15(1):3–11. https://doi.org/10.1016/S0929-1393(00)00067-6
Doube BM, Schmidt O (1997) Can the abundance or activity of soil macro fauna be used to indicate the biological health of soils? In: Pankhurst CE, Doube BM, Gupta VVSR (eds) Biological indicators of soil health, CAB International, Wallingford, pp 265–295
Grdisa MK, Grsic, Grdisa MD (2013) Earthworms -role in soil fertility to the use in medicine and as a food. Invert Surviv J 10(1):38–45
Haokip SL, Singh TB (2012) Diversity and distribution of earthworms in a natural reserved and disturbed subtropical forest ecosystem of Imphal-West, Manipur, India. Int Multidiscip Res J 2(2):28–34
Harbowy ME, Balentine DA, Davies AP, Cai Y (1997) Tea chemistry. Crit Rev Plant Sci 16(5):415–480. https://doi.org/10.1080/07352689709701956
Hopp H, Linder PJ (1947) A principal for maintaining earthworms in farm soils. Science 105(2739):663–664. https://doi.org/10.1126/science.105.2739.663
Jacquier M, Calenge C, Say L, Devillard S, Ruette S (2020) Altitude shapes the environmental drivers of large-scale variation in abundance of a widespread mammal species. Ecol Evol 10(1):119–130. https://doi.org/10.1002/ece3.5851
Jamatia SKS, Chaudhuri PS (2017) Earthworm community structure under tea plantations (Camellia sinensis) of Tripura (India). Trop Ecol 58(1):105–113
Jones CG, Gutierrez JL, Groffman PM, Shachak M (2006) Linking ecosystem engineers to soil processes: a framework using the Jenny state factor equation. Eur J Soil Biol 42(1):39–53. https://doi.org/10.1016/j.ejsobi.2006.07.017
Jones CG, Lawton JH, Shachak M (1994) Organisms as ecosystem engineers. Oikos 69(3):373–386. https://doi.org/10.2307/3545850
Joshi N, Aga S (2009) Diversity and distribution of earthworms in a subtropical forest ecosystem in Uttarakhand, India. NHJCU 9(1):21–25
Jouquet P, Dauber J, Lagerlof J, Lavelle P, Lepage M (2006) Soil invertebrates as ecosystem engineers: intended and accidental effects on soil and feedback loops. Appl Soil Ecol 32(2):153–164. https://doi.org/10.1016/j.apsoil.2005.07.004
Julka JM, Senapati BK (1987) Records of the Zoological Survey of India, Miscellaneous Publication. Occ. pap. 92, Grafic Printall, Calcutta, pp 1–105
Kandpal J (2018) Population dynamic and vertical distribution of earthworm in managed soils of Kumaun, Uttarakhand. J Mountain Res 13:81–87
Kaushal BR, Bisht SPS (1994) Population dynamics of earthworm Amynthas alexandri (Annelida: Megascolecidae) in a Kuman Himalayan pasture soil. Biol Fertil Soils 17:9–13
Lagerlof J, Goffre B, Vincent C (2002) The importance of field boundaries for earthworms (Lumbricidae) in the Swedish agricultural landscape. Agric Ecosyst Environ 89(1):91–103. https://doi.org/10.1016/S0167-8809(01)00321-8
Lalthanzara H, Ramanujam SN, Jha LK (2011) Population dynamics of earthworm in relation to soil physico-chemical parameters in agroforestry systems of Mizoram, India. J Environ Biol 32(5):599–605
Lavelle P, Gilot C, Frogoso C, Pashanasi B (1994) Soil fauna and sustainable land use in the humid tropics. In: Greenland D, Szabolcs I (eds) Soil resilience and sustainable land use. CAB International, Wallingford, pp 291–308
Lavelle P, Spain AV (2001) Soil ecology. Kluwer Academic Publishers, Dordrecht
Li J, Zhang Z, Wang H, Wang S, Che Q (2018) Urban land-use impacts on composition and spatiotemporal variations in abundance and biomass of earthworm community. J For Res 31:325–331. https://doi.org/10.1007/s11676-018-0807-2
Liu CW, Lin KH, Kuo YM (2003) Application of factor analysis in the assessment of groundwater quality in a black foot disease area in Taiwan. Sci Tot Environ 313(1–3):77–89. https://doi.org/10.1016/S0048-9697(02)00683-6
Margalef R (1958) Temporal succession and spatial heterogeneity in phytoplankton. In: Buzzati-Traverso AA (ed) Perspectives in marine biology. Univ Calif Press, Berkeley, pp 323–347
Marshall CB, Lynch DH (2020) Soil microbial and macro fauna dynamics under different green manure termination methods. Appl Soil Ecol 148:103–505. https://doi.org/10.1016/j.apsoil.2020.103505
Nainawat R, Nagendra B (2001) Density and distribution of earthworms in different localities of Jaipur. J Eco-Physiol 4:9–13
Newbold T, Hudson LN, Hill SLL, Laginha M, Pinto Correia D, Martin CD, Meiri S, Novosolov M, Pan Y, Phillips HRP, Purves DW, Robinson A, Simpson J, Tuck SL, Weiher E, White HJ, Ewers RM, Mace GM, Scharlemann JPW, Purvis A (2015) Global effects of land use on local terrestrial biodiversity. Nature 520(7545):45–50. https://doi.org/10.1038/nature14324
Ngo PT, Rumpel C, Doan T, Jouquet P (2012) The effect of earthworms on carbon storage and soil organic matter composition in tropical soil amended with compost and vermicompost. Soil Biol Biochem 50(2012):214–220. https://doi.org/10.1016/j.soilbio.2012.02.037
Pielou EC (1966) The measurement of diversity in different types of biological collections. J Theor Biol 13:131–144. https://doi.org/10.1016/0022-5193(66)90013-0
Rajkhowa DJ, Bhattacharyya PN, Sarma AK, Mahanta K (2014) Diversity and distribution of earthworms in different soil habitats of Assam, North-East India, an Indo-Burma biodiversity hotspot. Proc Natl Acad Sci India Sect B Biol Sci 85(2):1–10. https://doi.org/10.1007/s40011-014-0380-1
Rajwar N, Bisht SS, Bhatt S, Miglani R, Singh V (2018) Investigations on seasonal abundance of the earthworm Octolasion cyaneum (Savigny, 1826) (Lumbricidae: Annelida) in high altitude forest system of the Kumaun Himalayas, India. Int J Life Sci Pharma Res 8(3):1–8
Rajwar N, Singh V, Bhatt S, Bisht SS (2022) Earthworm population dynamics in three different land use systems along an altitudinal gradient (208–2609 m asl) in Kumaun Himalayas India. Trop Ecol 63(1):134–140. https://doi.org/10.1007/s42965-021-00178-x
Ramanujam SN, Roy B, Jha LK (2000) Inventory studies on the earthworm population in agroforestry systems of Mizoram. In: Proceedings of the International Workshop on Agroforestry and Forest Products, Aizawl, pp 191–194
Sankar AS, Patnaik A (2018) Impact of soil physico-chemical properties on distribution of earthworm populations across different land use patterns in southern India. JOBAZ 79(1):1–18. https://doi.org/10.1186/s41936-018-0066-y
Senapati BK, Lavelle P, Giri S, Pashanasi B, Alegre, Decaens TJ, Jimenez J, Albrecht A, Blanchart E, Mahieux M, Rousseaux L, Thomas R, Panigrahi PK, Venkatachalan M (1999) Soil technologies for tropical ecosystems. In: Lavelle P, Brussard L, Hendrix P (eds) Earthworm Management in Tropical Agroecosystems. CAB International, Wallingford, pp 199–237
Shannon CE, Wiener W (1949) The mathematical theory of communication. University of Illinois Press, Urbana
Sharma RK, Bharadwaj P (2014) Earthworm diversity in transgangetic habitats of Haryana, India. Res J Agric For Sci 2(2):1–7
Singh J, Cameron E, Reitz T, Schadler M, Eisenhauer N (2020a) Grassland management effects on earthworm communities under ambient and future climatic conditions. Eur J Soil Sci 72(1):343–355. https://doi.org/10.1111/ejss.12942
Singh S, Sharma A, Khajuria K, Singh J, Vig AP (2020b) Soil properties changes earthworm diversity indices in different agro–ecosystem. BMC Ecol 20(27):1–14. https://doi.org/10.1186/s12898-020-00296-5
Singh S, Singh J, Vig AP (2020c) Diversity and abundance of earthworms in different landuse patterns: relation with soil properties. Asian J Biol Life Sci 9(2):111–118. https://doi.org/10.5530/ajbls.2020.9.18
Singh S, Singh J, Vig AP (2016a) Earthworm as ecological engineers to change the physico-chemical properties of soil: soil vs vermicast. Ecol Eng 90:1–5. https://doi.org/10.1016/j.ecoleng.2016.01.072
Singh S, Singh J, Vig AP (2016b) Effect of abiotic factors on the distribution of earthworms in different land use patterns. JOBAZ 74:41–50. https://doi.org/10.1016/j.jobaz.2016.06.001
Suthar S (2011) Earthworm biodiversity in western and semiarid land of India. Environmentalist 31(1):74–86. https://doi.org/10.1007/s10669-011-9308-y
Suthar S (2012) Seasonal dynamics in earthworm density, casting activity and soil nutrient cycling under Bermuda grass (Cynodon dactylon) in semiarid tropics, India. Environmentalist 32:503–511. https://doi.org/10.1007/s10669-012-9419-0
Tiwari N, Negi N, Raj R, Joshi N (2017) A study on physico-chemical and mycofloral proportions of compost and vermicompost. J Env Bio-Sci 31(2):375–377
Tylianakis JM, Rand TA, Kahmen A, Klein AM, Buchmann N, Perner J, Tscharntke T (2008) Resource heterogeneity moderates the biodiversity-function relationship in real world ecosystems. PLoS Biol 6(5):e122. https://doi.org/10.1371/journal.pbio.0060122
Capelle VC, Schrader S, Brunotte J (2012) Tillage-induced changes in the functional diversity of soil biota—a review with a focus on german data. Eur J Soil Biol 50:165–181. https://doi.org/10.1016/j.ejsobi.2012.02.005
Van Groenigen JW, Lubbers IM, Vos HMJ, Brown GG, De Deyn GB, Van Groenigen KJ (2014) Earthworms increase plant production: a meta-analysis. Sci Rep 4(6365):1–9. https://doi.org/10.1038/srep06365
Walkey A, Black IA (1934) Chromic acid titration for determination of soil organic matter. Soil Sci 63:251
Wall DH, Bardgett RD, Behan-Pelletier V, Herrick JE, Jones TH (2013) Soil ecology and ecosystem services. Oxford University Press, Oxford
Walsh CL, Johnson-Maynard JL (2016) Earthworm distribution and density across a climatic gradient within the Inland Pacific Northwest cereal production region. Appl Soil Ecol 104:104–110. https://doi.org/10.1016/j.apsoil.2015.12.010
Wetzel MJ, Reynolds JW (2021) A preliminary inventory of earthworms (Annelida, Clitellata) of the Nachusa Prairie Grassland Area, Lee and Ogle Counties, Illinois, USA. Megadrilogica 26(8):91–126
Xie T, Wang M, Chen W, Uwizeyimana H (2018) Impacts of urbanization and landscape patterns on the earthworm communities in residential areas in Beijing. Sci Tot Environ 626:1261–1269. https://doi.org/10.1016/j.scitotenv.2018.01.187
Yasmin S, D’Souza D (2010) Effects of pesticides on the growth and reproduction of earthworm: a review. Appl Environ Soil Sci 2010:1–9. https://doi.org/10.1155/2010/678360
Acknowledgements
The authors are thankful to the Department of Environmental Science, Kanya Gurukul campus, Gurukul Kangri (Deemed to be University), Haridwar, India for providing necessary lab facilities.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors have no competing interests to declare that are relevant to the content of this article.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tiwari, N., Joshi, N. The influence of abiotic factors and organic carbon pools of soil on the distribution and diversity indices of earthworms under different land-use systems in north western Himalaya, India. Biologia 78, 2489–2501 (2023). https://doi.org/10.1007/s11756-023-01387-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11756-023-01387-6