Abstract
Passive restoration offer better preservation for the diverse legacy of forest ecosystems, but many interactions on the restoration process remain poorly understood. In this study, the seasonality of accumulated litter layer, nutrient content, potential return, and soil quality were evaluated under the initial (subjected to 11-year) and advanced (subjected to 46-year) passive restoration conditions in Cerrado, Brazil. Measurements were carried out for a period of one year. Accumulated litter layer, nutrient content, potential return, and nutrient use efficiency were 50%, 43%, 13%, and 42% higher in the advanced passive restoration site compared to the initial restoration site. For both sites, the annual litter content followed the order: N > Ca > K > Mg > P > Fe > Mn > B > Zn > Cu. Significant increases in soil organic matter and cation exchange capacity were found in the advanced passive restoration. The annual average had shown a higher macronutrient concentration in the soil for the initial restoration, while a higher micronutrient concentration was found for the advanced restoration. The seasonality affected the accumulated litter layer, litter nutrients, potential return and soil quality. Some litter and soil nutrients were significantly correlated, evidencing the nutrient associations between litter and soil. Hence, both the passive restoration stages and rain were factors that regulated the temporal patterns of accumulated litter layer as well as the nutrient cycling in Cerrado passive restoration models.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Biodiversity conservation is one of the greatest challenges to be faced nowadays, given the high level of anthropic disturbances observed in natural ecosystems (Leverkus et al. 2018). In view of expanding human land use, increasing climate change and unmet conservation targets, area-based conservation requires efficiency and effectiveness more than ever (Hoffmann 2022). There are several techniques and models focused on recovering degraded areas and the selection of such models is, in general, based on the degradation level, specific features, and future use of the area to be recovered (Aide et al. 2000; Lima et al. 2018). One of the biggest problems related to forest restoration is the advance of the alien species: they are fast and extensive vegetative growth—covering large areas and displacing native species—also leading to an acidification of invaded soils (Lazzaro et al. 2020). The consequence is a loss in native species diversity, alteration of the chemical and physical soil conditions, as well the ecosystem functionality.
Active and passive restoration models have been proposed to reestablish the functionality of some ecological processes (Restrepo et al. 2013) in degraded lands. Active restoration includes a range of human interventions that aim to accelerate and influence the successional trajectory of recovery (Holl and Aide 2011) through planting trees at high density and their respective management (Celentano et al. 2011; Restrepo et al. 2013). Conversely, passive restoration models are based on ending the prior anthropogenic disturbance, allowing a natural successional process, or unassisted forest recovery (Holl and Aide 2011). Although passive restoration models are simple, inexpensive, and based on natural regeneration (Holl 2002; Schrautzer et al. 2007), they are relatively slow processes when compared to active restoration models (Pereira et al. 2021, 2022). As a natural process, passive restoration is unpredictable. That is, during natural regeneration processes in which the forest structure in nearby locations can change significantly. Sometimes, these processes are not always successful due to land use history or the establishment of aggressive species (Lazzaro et al. 2020). However, they can better preserve the diverse legacy of these forested systems (Bechara et al. 2016; Zhang et al. 2020). Furthermore, these processes also revealed in the tropics, the inherent capability of these systems to naturally recover and highlights the importance of considering passive restoration in management plans (Holl and Aide 2011; Bechara et al. 2016).
Protected area management often lacks the continuous availability of data on current states and trends of nature and threats (Hoffmann 2022). Despite the need for the scientific community to understand and characterize passive restoration processes as models for recovery of degraded lands, the models may fail to provide information on both the temporal dynamics of the litterfall (Pereira et al. 2021) as well as the nutrient return under similar conditions. Moreover, the comparative interpretation of passive restoration effects at different sites is often difficult due to possible pre-existing differences between the sites (such as environmental conditions, land use, vegetation type) that could mask the analysis.
Therefore, the knowledge of nutrients cycling is crucial for understanding the structure and functioning of forest ecosystems. Nutrients taken up by deep roots are transported into the above-ground parts and re-deposited on the soil surface through litterfall, stemflow or throughfall (Rengel 2007; Bessi et al. 2018; Tonello et al. 2021). These processes represent the main transfer of organic matter and nutrients from the vegetation to the soil surface (Celentano et al. 2011; Tonello et al. 2021), allowing the determination of positive trajectories in rehabilitating degraded land and restoring the ecosystem resilience (León and Osorio 2014). In many tropical soils (as those found in the Brazilian Cerrado), nutrients released from the litter are the most relevant sources of plant nutrients (Parzych and Trojanowski 2006) and humus formation (Souza 2022). The successional process influences nutrient cycling, but there are divergences to the pattern of this dynamic (Camara et al. 2018).
The above discussion and the data collected in this study were used to answer the following questions: (1) What are the patterns of accumulated litter layer and nutrient return to the forest in sites undergoing passive restoration in Cerrado for 11 and 46 years, under similar edaphic and climatic conditions? (2) Are there differences in the litter and soil nutrient content between these passive restoration sites? (3) Is the rain a factor that regulates the temporal patterns of accumulated litter layer and nutrients? (4) How are the litter and soil nutrients correlated? In order to answer above questions, it was hypothesized that: (a) accumulated litter layer, litter and soil nutrients increase due to passive restoration age (b) litter layer, litter and soil nutrients are influenced by the dry and rainy seasons. The answers for the above questions may help us to understand the dynamics and changes in ecosystems in a passive restoration scenario. The main goal of this work is to investigate the role of litter layer as a key-strategy in biogeochemical nutrient cycles and, thus, the soil quality improvement within the passive restoration sites in the Cerrado savanna.
Material and methods
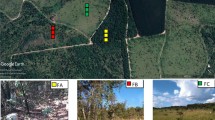
The study was carried out in Aguas Perenes Forest, which is a Private Reserve of Natural Patrimony (PRNP) located in Lagoa Seca microbasin, Brotas County – São Paulo State (22°11.754’S and 48°6.523’W). This forest is the water recharge area of Guarani Aquifer. Back in 2011, it was acknowledged by the Forest Stewardship Council as High Conservation Value Forest due to providing basic environmental services such as watershed protection. The PRNP covers more than 809.78 ha of Cerrado area, its phytophysiognomy is featured as secondary vegetation of Cerrado stricto sensu (trees cover more than 30% of the ground, but a fair amount of grass keeps on forming an open savanna) and Cerradão (closed woodland savanna without grass coverage) (Ratter et al. 1997; Oliveira-Filho and Ratter 2002; Durigan et al. 2012). After the removal of Eucalyptus sp. in 2006, silvicultural interventions and the establishment of Cerrado passive restoration have been carried out. The area has been exclusively dedicated to nature conservation and watershed protection. The Köppen climate-type of the region is Cwa (Dubreuil et al. 2019), corresponding to a subtropical climate (C), characterized by warm summers and dry, cool winters (w), such that the average temperature in the hottest month (January) is greater than 22 °C (a). Based on the meteorological data recorded from 2018, the annual average rainfall was 1337 mm, and the annual average temperature was 20 °C. The predominant soil type is quartzarenic neosol (Santos et al. 2018).
The present study was carried out in two forest sites within an experimental catchment with the same edaphic, climate, and disturbance history, on poor acid soil in Cerrado undergoing passive restoration stages: (1) site F11 concerned the initial passive restoration, and was subjected to 11-year restoration and (2) site F46 referred to the advanced passive restoration and was subjected to 46-year restoration (Fig. 1). Our study was performed in three triplicate plots of 20 × 20 m at each site. Accumulated litter layer, macro, and micronutrients from litter, soil, and rainfall were measured from May/2018 to April/2019. The structural vegetation attributes in the stand are shown in Table 1.
The rainfall was measured in an open area without any obstructions using three rainfall gauges made of polyethylene installed near the stand, with a maximum distance of 30 m. The pluviometers were installed at 1.20 m of height.
The litter layer stored on the soil surface was collected monthly with a litter traps of a 50 × 50 cm2 sampler. The collection was random, and only materials in one quadrant were collected. This procedure resulted in 10 collections at each plot, with a total of 3 composed samples per site/month. The soil was sieved and removed from the litter samples. Then, the fresh mass was measured in the field using a suitable scale and stored in plastic bags. Subsequently, the litter was dried in forced-air circulation oven at 70 °C, until it reached a constant dry mass, which was determined on a 0.01 g-precision scale. Monthly and annual accumulated litter layer were estimated by summing the fractions.
The litter nutrients analyses were performed in four composite samples collected every two months at each plot, allowing characterization of both the dry (April to September) and rainy season (October to March). Thus, each composite sample was formed by 9 subsamples per site. To quantify the nutrient content, the litter was crushed, packed in airtight plastic bags and labeled for further analysis. Litter contents were analyzed by different methods: nitrogen (N) by the Kjeldahl method (Kjeldahl 1883); phosphorus (P) by the molybdate-blue method; calcium (Ca), magnesium (Mg), potassium (K), boron (B), copper (Cu), iron (Fe), manganese (Mn), and zinc (Zn) by atomic absorption spectroscopy.
A random sampling design was used to collect soil samples from the restoration sites, as exactly as the one performed for the litter nutrients analysis. The soil samples used for soil nutrient analyses were first cleared of roots and litter by hand, then air-dried, crushed, and passed through a 2 mm mesh sieve. Coarse materials, such as gravel and roots, were removed, and samples from the < 2 mm fraction were weighted and used for analyses. The determination of soil nutrients content followed the same procedure reported for litter, except for nitrogen (N) that could not be analyzed. The pH was measured in a 0.01 mol L-1 CaCl2 solution (Embrapa 2017) at a 1: 5 soil:solution weight ratio and soil organic matter (SOM), according to Yeomans and Bremner (1988). Cation Exchange Capacity (CEC) and base saturation (V) were also calculated. The samples were obtained at a depth of 0 – 10 cm.
The nutrient transfer refers to the total amount of each element that returns to the soil within one hectare of forest. Thus, the potential return of some nutrient by the litterfall was calculated as the product of the nutrient concentration (g kg−1) and the litter dry mass (kg ha-1 ano−1) (Vitousek 1982). The annual nutrient use efficiency at each passive restoration site was estimated by using total biomass/nutrient ratios (Vitousek 1982).
Statistical analysis
Analysis of variance was applied to normal data through Tukey at a 5% probability level to analyze the means of the annual accumulated litter layer, litter, and soil nutrients between the passive restoration stages and between the dry and rainy season. Data that did not meet ANOVA assumptions were subjected to the non-parametric Kruskal-Wallis test. All analyses were performed using Minitab statistical software 14.0.
Results
Accumulated precipitation recorded throughout this research work was 887.3 mm, with 75% (668.4 mm) in the rainy and 25% (219.2 mm) in the dry seasons (Fig. 2a). The comparison between precipitation in the studied area and the data recorded over the period of 1981 to 2010 (climatological normal for the latest global standard normal period) showed an atypical year. Precipitation was approximately 44% below the climatological normal, whereas the mean temperature was 21.6 °C, 5% higher than that recorded at the climatological normal (20.6 °C). The highest mean temperature was recorded in December (24.7%), and the lowest one, in July (18.3%).
Accumulated litter layer was significantly higher in F46 than in F11 (Fig. 2b). In the first case, the annual litter deposited over one-year was 5.70 t ha−1, in which monthly litterfall peaks were recorded in May for F46 (770 kg ha−1). Accumulated litter layer in F46 differed between the dry (3.8 t ha−1 y−1) and the rainy seasons (1.90 t ha−1 y−1). Considering 3.70 t ha−1 y−1 of accumulated litter layer, the initial passive restoration showed a peak in March (552 kg ha−1), 1.95 t ha−1 year−1 in the rainy season, and 1.76 t ha−1 y−1 in the dry season. On the other hand, F11 did not present seasonal characteristics in the deposition, behaving more homogeneously throughout the year (Fig. 2b). Nevertheless, in F11 the litter deposition in the dry season was 51% lower than that of the rainy season.
Litter nutrients and seasonality
Mean annual litter nutrients were 43% higher in F46 than in F11 (Fig. 3). On average, litter from F46 showed 7.8 g kg−1 and 128.9 mg kg−1 of macro and micronutrients, respectively, whereas 7.0 g kg−1 and 84.7 mg kg−1 F11, respectively. It is important to stress that all macronutrient inputs were higher in advanced than initial restoration. Despite this, on the annual average, only magnesium, iron, and manganese differed between the sites. Considering the macronutrients, the nitrogen is exported in the greatest quantity, followed by calcium in both sites. In contrast, among the micronutrients, the higher values exported were for iron and manganese, and the lower values for copper. The concentration of macronutrients exported via the litterfall followed the order N > Ca > K > Mg > P for both sites, while for micronutrients the concentration was Mn > Fe > B > Zn > Cu for F11, and Fe > Mn > B > Zn > Cu for F46.
Regardless of passive restoration sites, nitrogen and potassium were higher in the rainy season (Table 2). These concentrations were 30% and 96% higher for nitrogen and potassium in F11. However, only potassium differed between the seasons. Considering the F46, phosphorus and magnesium were significantly higher in the drought period (78% and 22%, respectively), whereas in the rainy season, nitrogen was significantly higher (27%) and, despite it was not significant, even potassium was 41%. Comparing both passive restoration sites, they had shown differences in magnesium content in the dry season.
For both passive restoration sites, boron concentration was higher in the dry season, while copper, manganese and zinc were higher in the rainy season (Table 3). On the other hand, iron was higher in the dry season only for F11. The total micronutrient input was 6% and 33% higher in the rainy season for F11 and F46, respectively. When the two seasons were compared, statistical differences were observed only between boron and zinc in F11, while the same occurred for boron and manganese in F46. Again, comparing both passive restoration sites, manganese concentration was 104% higher in F46 in the dry season, while in the rainy season, the same occurred for iron and manganese (64% and 93%, respectively).
Litter nutrients potential return and nutrient use efficiency
The total annual estimated litter nutrients were 13% higher in F46 (231.15 kg ha−1 y−1) than F11 (204.44 kg ha−1 y−1) (Table 4). On the other hand, the transfer of macronutrients was 13% higher in F46 (226.74 kg ha−1 y−1) than in F11 (201.52 kg ha−1 y−1). Of this total, the percentage that occurred in the dry season was 55% in F46 and 52% in F11. Nitrogen and potassium showed a higher transfer value in the rainy season. Overall, nitrogen showed the greatest annual transfer (102.5 kg ha−1 for F11 and 106.4 kg ha−1 for F46), followed by calcium (52.2 kg ha−1 for F11 and 64.6 kg ha−1 for F46). Regardless of the season, the return of these macronutrients through the litter, for both restoration sites, followed the order: N > Ca > K > Mg > P.
The micronutrient concentration was 51% higher in F46 (4.41 kg ha−1 y−1) than in F11 (2.92 kg ha−1 y−1). In the rainy season, the potential returns in F46 and F11 were 52% and 53%, respectively. Considering the micronutrients, the return via litter was similar for both sites and seasons and followed the order: Fe > Mn > B > Zn > Cu.
The initial forest restoration (F11) was more efficient only in the use of manganese (Table 5). Among the macronutrients, the nutrient use efficiency in F46 was 50%, 42%, 45%, 27%, and 12% higher in nitrogen, phosphorus, potassium, calcium, and magnesium, respectively. Similarly, F46 also was 37%, 41%, 22%, and 55% higher in boron, copper, iron, and zinc, respectively.
Soil quality and seasonality
Annual significant increases in soil organic matter (SOM) and cation exchange capacity (CEC) were detected in F46, and the soil pH was found to be more acidic (Table 6). For both sites, macro and micronutrient concentrations were given according to the order Ca > P > Mg > K and Fe > Mn > Zn > Cu > B. The initial passive restoration (F11) had shown the highest macronutrient concentrations. Notably both sites showed significant differences in phosphorus and potassium. An inverse trend was observed for micronutrients, where the highest concentrations were associated with advanced passive restoration (F46), and only iron concentrations differed between the sites.
The SOM was significantly higher in dry season in F46 (Table 7). Considering F11 only, pH, SOM, V, and CEC did not differ between the seasons. Yet on this site, most nutrients concentration increased in the rainy season, except potassium, boron, and copper, while different concentrations were observed for iron and manganese between the seasons.
In F46 however, potassium, calcium, boron, and copper were higher in the drought period, but significant differences were observed only for boron and iron. Comparing the passive restoration sites, significant differences were noted in phosphorus and potassium concentrations in the dry season, while, in the rainy season, same trend has occurred for iron only.
Litter and soil nutrients correlation
The correlations between litter and soil nutrients varied between passive restoration sites (Table 8). The initial passive restoration (F11) was the only one to show that annual copper and iron concentrations in the litter content were significantly correlated. Considering the seasonality in F11, in the drought period, phosphorus concentration in the litter was strong and inversely correlated to the soil; in the rainy season, the strong correlation occurred for manganese. For F46, in contrast, potassium, boron (negative), and copper were significantly correlated in the drought period and boron in the rainy season.
Discussion
Effect of passive restoration age on accumulated litter layer and litter and soil nutrients content
The litter production in forest ecosystems depends on several ecological factors, such as the climate, species composition, stand age, and site quality (König et al. 2002; Yang et al. 2005; Dodonov et al. 2016; Pereira et al. 2022). In the present study, two sites possessing different passive restoration ages were compared, but with an identical macroclimate as well as soil type. Significant differences were observed in the annual litter production between passive restoration sites, which could be partly due to the physiological features of tree species as well as their different responses to the environmental cues. Although the sites studied are contiguous, having the same soil type and climate, as stated above, in each site the tree species had different nutritional demands, which implied differences in the deposition to the soil (Tables 2 and 3).
The initial passive restoration site (F11) has lower diversity, tree density, tree stratification, and, consequently, lower tree coverage (Table 1), which directly influenced material deposition rates. Considering the accumulated litter layer, the variability in its nutrient content depends on species, climate, and soil features. On the other hand, F46 present the opposite situation when compared with F11.
Nutrient cycling in forests has been studied for more than 100 years, however, there is limited information on micronutrient cycling, with most attentions traditionally being paid to nitrogen and phosphorus (Rengel 2007). Although the litter of restoration sites presented significant differences only for magnesium, iron, and manganese, the advanced passive restoration site showed the highest litter concentration for all macro and micronutrients studied. These pattern of higher production and concentration of nutrients in the litter from the youngest to the mature forest corroborates with records in tropical forests such as Atlantic Forest (Caldeira et al. 2008; Scheer et al. 2011), Semideciduous Seasonal Forest (Pinto et al. 2009), subtropical secondary rain forest (Dickow et al. 2012), Union Biological Reserve (Camara et al. 2018).
Macronutrient contents found in the litter were higher for nitrogen, especially in F46. The high content of nitrogen is related to the high nutritional demand for this element without reuse in other parts of the plant. This element is highly mobile and can easily be relocated from the older tissues to the younger ones, concentrating mainly on the leaves (Ribeiro et al. 2017). In the stocked litter, calcium and magnesium were the second and fourth contents, respectively, and both showed low mobility in the plant tissues. The low mobility is due to the structure of the pectic chains present in the cell wall (Hawkesford et al. 2012), which increases its content in the branches and leaves (Schumacher et al. 2004). Also, there is a direct relation with the leaf longevity, not being translocated to younger tissues (König et al. 2002). Calcium and magnesium are not very mobile chemical elements in the plant tissues, being, therefore, more strongly immobilized in the plant biomass (Camara et al. 2018). It was also verified that, although F46 presented a higher concentration of these elements in the litter, the availability in the soil was lower than that observed for F11. Due to the relative increase of its stock in forest biomass, the concentration of some nutrients in the soil may decrease as a result of gradual increase in biomass that takes place during ecosystem ripening (Vitoussek and Sanford 1986; Camara et al. 2018).
Since the tropics have both highly and slightly weathered soils, phosphorus is particularly limited in these regions (Cleveland et al. 2011; Rozendaal et al. 2019). Phosphorus in the litter presented the lowest concentration for both sites. This pattern has also been reported in several tropical forests (Vital et al. 2004; Caldeira et al. 2008; Pinto et al. 2009; Pimenta et al. 2011; Giácomo et al. 2017; Ribeiro et al. 2017). Phosphorus is one of the elements that have a high internal translocation rate in plants and, therefore, a high-efficient use. Results in this study have shown that phosphorus was the most efficient element in both passive restoration sites, but with greater efficiency in the advanced restoration site. Phosphorus can be translocated to other plant structures before the leaf senescence contributing to the creation of new plant structures or implementation of physiological processes (Palma et al. 2000; González-Rodríguez et al. 2011; López et al. 2013). On the other hand, the availability of phosphorus and potassium in the soil was significantly higher for F11. The variation in the results is probably due to the dynamics of phosphorus availability in the soil, which includes its temporary immobilization in microbial biomass and, crucially, the adsorption in colloidal particles in highly weathered soils developed under a tropical climate that make this element unavailable to plants (Santos et al. 2008). The cycling of potassium in the soil–plant relation is faster than the other nutrients, as it is a monovalent cation (Jordan 1985). The low potassium concentration levels in the accumulated litter can be related to the small rates of this nutrient in biogeochemical cycling. This nutrient has levels in the accumulated litter that are many times higher than those found in the above-ground biomass components. In general, biogeochemical cycling is the route in which the low mobility nutrients are cycled since for these nutrients, the biochemical cycling becomes not very significant, contrary to what occurs for high mobility nutrients in the plant (Caldeira et al. 2008; Pimenta et al. 2011).
Contrary to the macronutrients, the micronutrients are consumed in smaller quantities, but they are fundamental for plant development. The knowledge on the micronutrient dynamics in the native ecosystems is still incipient, which implies the absence of understanding of how the inorganic and organic inputs influence the micronutrient cycling. (Rengel 2007). The micronutrient order in the litter (Fe > Mn > B > Zn > Cu) was similar for passive restoration sites, and have been corroborated by other tropical forests (Lopez Hernandez et al. 2014; Klippel et al. 2016; Bianchin et al. 2017). After aluminum, iron is the second most abundant metal in the Earth's crust, possessing extremely low mobility (Broadley et al. 2012). Besides, high levels of iron and manganese in the litter can also be due to the high concentrations of these elements in the soil (Luciano et al. 2012) , as observed in this study (Table 6). Iron and manganese concentrations were significantly higher in the litter of F46, which in turn and in contrary to what was observed for macronutrients, presented the highest concentrations of microelements in the soil.
The economy in the use of nutrients possibly indicates a limitation in primary production in the environment, while low efficiency may indicate that the nutrients supply is more adequate (Vitousek 1982). The analysis of the nutrient use efficiency by passive restoration sites showed greater conservation in F11 (Table 5). Some authors have pointed out that high values in the nutrient use index indicate a more efficient nutrient cycling (Gama-Rodrigues and Barros 2002). Therefore, the higher levels of nutrients presented by litter from F46, in addition to the high annual transfer rates of these nutrients, reflect the best edaphic conditions in this environment. This shows that primary production in a mature forest is not limited by the availability of the elements studied (Vitousek 1982; Pinto et al. 2009).
Studying soil nutrient pool size change over time is challenging, since the difficulties are linked to the quantification of many unknown fluxes, especially within the ecosystem (Van der Heijden et al. 2013, 2014). The nutrients and the organic matter returned to the forest floor are important factors in forest restoration projects since the organic soil results in higher nutrients availability in this compartment. Through the litter, the vegetation can contribute to the soil quality improvement due to its capability to induce ecological and physicochemical changes in the soil (León and Osorio 2014). Hence, although the soil nutrients were similar across both sites, for other properties (pH, SOM and CEC), differences were found between passive restorations. Due to the cationic nutrients leaching, absorption of calcium and magnesium by vegetation, and probably the production of a more acidic litter throughout the forest development, soil acidification was observed in both initial to advanced passive restorations (Table 6). Hence, in this study, improvements were observed in soil quality from F11 to F46, with increased organic matter and cation exchange capacity, which resulted in greater nutrient use efficiency. However, the correlation between the annual litter and soil nutrient concentrations were site dependent. It means that nutrient proportion in the litter that was released into the soil is site dependent.
Thus, the results of this study partially corroborate the first hypothesis: differences in accumulated litter layer and quality increase with passive restoration age. It was confirmed that the accumulated litter and its nutrient content increase from initial to advanced passive restoration in Cerrado. Nevertheless, accumulated litter layer and litter content cannot be directly associated with the soil nutrients increase, but rather with the improvement on the soil quality.
Effect of dry and rainy seasons on accumulated litter layer, litter and soil nutrients
For tropical forests, the litter peaks mainly reflect drought stress (Okeke and Omaliko 1994; Vital et al. 2004; Barlow et al. 2007; Pimenta et al. 2011; Pereira et al. 2022). In this study, the drought season is represented by autumn and winter. Thus, the pattern of the litter production was found to be increased in the dry season in the advanced passive restoration in Cerrado, indicating that the physiological response to drought plays a major role in this process and in this restoration stage. The seasonal pattern of litterfall may be attributed to temperature and rainfall as a strategy by plants to control water loss by transpiration in the warmer periods with leaf abscission, branches, and other plant components (Pereira et al. 2022). Leaf aging, caused by photoinhibition, stomatal closure, and subsequent leaf overheating, causes leaf thinning at the end of the dry season (Röderstein et al. 2005). Besides, the lower night temperatures that prevail during the dry season, stimulate the abscisic acid synthesis in the foliage, which in turn stimulates the leaf senescence (Yang et al. 2005). Similar results were obtained in Cerrado (Valenti et al. 2008), Cerradão (Cianciaruso et al. 2006), Caatinga (Costa et al. 2010; Queiroz et al. 2019), forest-savanna transitions (Paiva et al. 2015), semideciduous seasonal forest (Pinto et al. 2009), and in the Amazon (Martins et al. 2018). On the other hand, the litter deposition in the initial restoration was shown not to be as drought-affected. The similarity of the litter deposition between the rain and dry season for the initial passive restoration site fits with similar studies in Cerrado sensu stricto (Ribeiro et al. 2017).
Independent of the amounts of nutrients in the incident rainfall, significant amounts of nutrients are added and transferred from above-ground plant parts to the forest floor as the rainwater passes through the canopy (Chuyong et al. 2004). Although rainfall could be a source of some nutrients, reduction of most nutrients in the litter and soil in the rainy season was observed. In the litter, only potassium and nitrogen contents increased in the rainy season in both sites, although significant differences were observed for potassium in F11 and nitrogen, in F46 (Table 2). Potassium is one of the ions most easily leached from tree crops by rain, as it is not a structural component of any organic compound, occurring in the soluble or adsorbed form in cells (Espig et al. 2009). This fact justifies the high values of potassium content in the rainy season, particularly the higher values in F11. In addition to litterfall and decomposition, rainfall represents the main nitrogen source for the soil–plant system (Luizao 1989), which may explain the increase in nitrogen content observed in the rainy season. A similar pattern was observed in pure forest plantations of Pterogyne nitens and Eucalyptus urophylla (Barbosa et al. 2017).
It was observed, although in different magnitudes, that both passive restorations presented the highest litter concentrations of calcium, phosphorus, magnesium and boron in the dry season (Table 2 and 3), as well as iron in the initial restoration. Due to its low mobility, the largest cycling of calcium in nature occurs by the fall and decomposition of senescent plant tissues (Marschner 1997) in the dry season, as reported in this study. The presence of magnesium is associated with phosphorus since magnesium is linked to its translocation by the plant (Schumacher et al. 2004). As magnesium participates in the constitution of chlorophyll a and b, its concentration will be higher in the leaves (Lima et al. 2010). This justifies the higher content of magnesium in F46, which presented a higher litter production in the dry season. However, in the soil, even though no differences were observed between the seasons, for both sites, magnesium concentration was higher in the rainy season. For phosphorus, the F11 showed an increase, but the F46 remained relatively stable throughout the seasons.
As for boron, the difference between its concentration in the litter in the dry and rainy seasons in the two studied areas was observed only in F46. Boron has low mobility in plant tissues, and this element is a constituent of the Ramnogalacturonanos II (O’Neill et al. 2004), molecules present in pectin which is the major component of the primary plant wall (Matoh et al. 1996). Accumulated litter layer in the dry season reflected in higher concentration of this element in the litter and in the soil of advanced restoration. On the other hand, although there was a significant difference in boron concentration between the dry and rainy seasons in F11, this was not enough to promote soil increments.
Iron was an element that had an inverse pattern between passive restoration sites, presenting low mobility, which may justify the high levels found in the litter deposited in F46 in the rainy season, which differed significantly from F11. An explanation for that may be due to the physiological behavior and nutritional needs of the species on this site, higher levels in the old leaves of some species, as well as higher average levels in the wood, bark, and branches (Caldeira et al. 2008).
It is important to note that the correlations between soil and litter nutrients became clearer when analyzed from the seasons, which is possibly related to the pattern of litter production, but due consideration should be given to the fact that the relations were site-specific. Also, precipitation in forest ecosystems can increase nutrient leaching from soils instead of increasing nutrient retention (Zhang et al. 2017; Tonello et al. 2021). This leaching may deplete some nutrients, leading to an availability decrease of these elements in the surface soils, potentially accounting for the poor correlations between the litter and soil nutrients.
All facts mentioned above reinforces the role of passive restoration in Cerrado, the importance of the forest structure, and the species in the restoration process. In this way, from a functional perspective, the standing litter on the soil surface is important in the regulation of several processes involving forest ecosystem maintenance and conservation, but ecosystem models need to consider litterfall seasonal patterns (Zhang et al. 2014). Hence, this study corroborates the second hypothesis that accumulated litter layer, litter and soil nutrients are influenced by the dry and rainy seasons in sites under passive restoration in Cerrado.
A comparison of annual nutrient returns in tropical forests
In tropical environments, litterfall represents the main process that determines the potential return of organic matter and nutrients to the soil (Scheer 2009; León and Osorio 2014), which supports plant development, soil recovery, and soil biota. The nutrients return verified in this study indicates the litter influence on the nutritional dynamics of systems undergoing passive restoration. The potential return of nutrients via litter production in forest ecosystems has been widely reported in many studies in tropical forests (Table 9). On the other hand, this kind of report in passive restoration sites is still scarce. Our results had shown that all nutrients return increased from initial to advanced passive restoration age in Cerrado.
The macronutrient return through total litterfall in both passive restoration sites was higher than those recorded in Advanced Atlantic forest (Scheer et al. 2011), Pine forest mixed with deciduous trees (González-Rodríguez et al. 2011), Ombrophilous Dense Forest (initial and advanced stage) (Caldeira et al. 2008), Caatinga (Queiroz et al. 2019), Mata mesofítica and Cerradão (Giácomo et al. 2012). Nitrogen return was higher than most of the studies in the tropical forest. Calcium inputs in F46, in turn, were higher than Ombrophilous Dense Forest in the intermediary stage (Caldeira et al. 2008). The potential return of potassium is consistently lower in most tropical forests. In these soils, the low level of potassium in the litter exerts a severe restriction for microbial activity and plant growth. Therefore, in tropical environments, one may find low litter concentrations of potassium, as observed in this study, representing a major limiting factor in nutrient cycling and plant nutrition. Despite this, the phosphorus return observed here was higher than for most of the other elements presented in Table 9.
There are few studies on micronutrient potential return. However, this study verifies that, the potential return of iron and manganese were lower than Dense Mountain Ombrophilous Forest (Freitas et al. 2015), as well as Ombrophilous Dense Forest (Caldeira et al. 2008) and Submontane Atlantic Rain Forest (advanced) (Bianchin et al. 2017). Boron had shown to be lower than the values observed in Lowland (Sayer et al. 2020) and Dense Mountain Ombrophilous Forest (Freitas et al. 2015) but higher than Ombrophilous Dense Forest (Caldeira et al. 2008). In the two passive restoration sites analyzed here, the potential return of copper and zinc was lower than most studies performed on tropical forests reported, (Table 9).
Conclusions
The litter deposition and nutrient transfer to the soil are key factors to understand the ecosystem recovery, although these studies are still incipient in passive restoration models. The variation in annual litter production, litter nutrient concentration and nutrient potential return increased from initial to advanced passive restoration in the study areas (11 and 46-year-old) in Cerrado, Brazil. However, the soil nutrients did not follow the same pattern. Based on this study, the greater annual nutrients supply from litter in the advanced forest has shown less need for conservation mechanisms of these elements, whereas it has shown greater conservation in the initial restoration site. Accumulated litter layer, litter and soil nutrients varied with the dry and rainy seasons, but the variations were site-specific. Some litter and soil nutrients were significantly correlated, exhibiting the nutrient associations between litter and soil, but they are seasonality and restoration stage dependent. It was also indicated that litterfall seasonal patterns were important for understanding the nutrient cycling and passive forest restoration in Cerrado. The evaluation of restoration models from a functional perspective, as proposed in this work, is relevant to guiding future interventions by restorers and in providing information to help in achievement of objectives in ongoing forest restoration process at different stages.. Furthermore, this study is expected to encourage future studies to make contributions to knowledge about soil-litter–nutrient dynamic in forest restoration.
Data availability statement
Data are contained within the article.
References
Aide TM, Zimmerman JK, Pascarella JB, Rivera L, Marcano-Vega H (2000) Forest regeneration in a chronosequence of tropical abandoned pastures: implications for restoration ecology. Restor Ecol 8(4):328–338. https://doi.org/10.1046/j.1526-100X.2000.80048.x
Barbosa V, Barreto-Garcia P, Gama-Rodrigues E, Paula A (2017) Biomassa, carbono e nitrogênio na serapilheira acumulada de florestas plantadas e nativa. Floresta Ambiente 24:1–9. https://doi.org/10.1590/2179-8087.024315
Barlow J, Gardner TA, Ferreira LV, Peres CA (2007) Litter fall and decomposition in primary, secondary and plantation forests in the Brazilian Amazon. For Ecol Manage 247(1–3):91–97. https://doi.org/10.1016/j.foreco.2007.04.017
Bechara FC, Dickens SJ, Farrer EC, Larios L, Spotswood EN, Mariotte P, Suding KN (2016) Neotropical rainforest restoration: comparing passive, plantation and nucleation approaches. Biodivers Conserv 25(11):2021–2034. https://doi.org/10.1007/s10531-016-1186-7
Bessi D, Tanaka MO, Costa LA, Correa CJP, Tonello KC (2018) Forest restoration and hydrological parameters effects on soil water conditions: a structural equation modelling approach. Rev Bras Recur Hidr 23. https://doi.org/10.1590/2318-0331.231820180043
Bianchin JE, Marques R, Blum H, Oliva EV, Donha CG, Silveira FM, Vargas GR (2017) Micronutrientes na serapilheira depositada em florestas secundárias no litoral do Paraná. Nativa 5(6):446–455. https://doi.org/10.5935/2318-7670.v05n06a11
Broadley M, Brown P, Cakmak I, Rengel Z, Zhao F (2012) Function of Nutrients: micronutrients. In: Marschner’s Mineral Nutrition of Higher Plants, 3rd edn, pp. 191–248. https://doi.org/10.1016/B978-0-12-384905-2.00007-8
Caldeira MVW, Vitorino MD, Schaadt SS, Moraes E, Balbinot R (2008) Quantification of litter and nutrients on an Atlantic Rain Forest. Semina Cienc Agrar 29(1):53–68. https://doi.org/10.5433/1679-0359.2008v29n1p53
Camara R, Silva VD, Delaqua GCG, Lisbôa CP, Villela DM (2018) Relação entre sucessão secundária, solo e serapilheira em uma reserva biológica no estado do Rio de Janeiro, Brasil. Ciência Florestal 28(2):674. https://doi.org/10.5902/1980509832066
Celentano D, Zahawi RA, Finegan B, Ostertag R, Cole RJ, Holl KD (2011) Litterfall dynamics under different tropical forest restoration strategies in Costa Rica. Biotropica 43(3):279–287. https://doi.org/10.1111/j.1744-7429.2010.00688.x
Chuyong GB, Newbery DM, Songwe NC (2004) Rainfall input, throughfall and stemflow of nutrients in a central African rain forest dominated by ectomycorrhizal trees. Biogeochemistry 67:73–91. https://doi.org/10.1023/b:biog.0000015316.90198.cf
Cianciaruso MV, Pires JSR, Delitti WBC, Silva ÉFLP (2006) Produção de serapilheira e decomposição do material foliar em um cerradão na Estação Ecológica de Jataí, município de Luiz Antônio, SP, Brasil. Acta Bot Bras 20(1):49–59. https://doi.org/10.1590/s0102-33062006000100006
Cleveland CC, Townsend AR, Taylor P, Alvarez-Clare S, Bustamante MMC, Chuyong G, Wieder WR (2011) Relationships among net primary productivity, nutrients and climate in tropical rain forest: a pan-tropical analysis. Ecol Lett 14(9):939–947. https://doi.org/10.1111/j.1461-0248.2011.01658.x
Costa CCA, Camacho RGV, Macedo ID, Silva PCM (2010) Análise comparativa da produção de serapilheira em fragmentos arbóreos e arbustivos em área de caatinga na Flona de açu-rn. Rev Arvore 34(2):259–265. https://doi.org/10.1590/s0100-67622010000200008
Dickow KMC, Marques R, Pinto CB, Höfer H (2012) Litter production in different successional stages of a subtropical secondary rain forest, in Antonina, PR. Cerne 18(1):75–86. https://doi.org/10.1590/S0104-77602012000100010
Dodonov P, Braga AL, Harper KA, Silva Matos DM (2016) Edge influence on plant litter biomass in forest and savanna in the Brazilian cerrado. Austral Ecol 42(2):187–197. https://doi.org/10.1111/aec.12420
Dubreuil V, Fante KP, Planchon O, Sant’AnnaNeto JL, (2019) Climate change evidence in Brazil from Köppen’s climate annual types frequency. Int J Climatol 39(3):1446–1456. https://doi.org/10.1002/joc.5893
Durigan G, Melo ACG, Brewer JS (2012) The root to shoot ratio of trees from open- and closed-canopy cerrado in south-eastern Brazil. Plant Ecol Divers 5(3):333–343. https://doi.org/10.1080/17550874.2012.691564
Embrapa (2017) Manual de Métodos de Análise de Solo, 3rd edn., Teixeira PC, Donagemma GK, Fontana A, Teixeira WG (eds) Centro Nacional de Pesquisa de Solos, Brasília-DF. Retrieved from https://www.agencia.cnptia.embrapa.br/Repositorio/Manual+de+Metodos_000fzvhotqk02wx5ok0q43a0ram31wtr.pdf. Acessed 1 Oct 2022
Espig SA, Freire FJ, Marangon LC, Caraciolo RL (2009) Sazonalidade, composição e aporte de nutrientes da serapilheira em fragmento de mata atlântica. Rev Arvore 33(5):949–956
Freitas CAA, Caldeira MVW, Horn SK, Castro KC, Viera M (2015) Serapilheira Acumulada Em Complexo Rupestre De Granito, Mimoso Do Sul, Es. Revista Árvore 39(4):671–681. https://doi.org/10.1590/0100-67622015000400009
Gama-Rodrigues AC, Barros NF (2002) Ciclagem De Nutrientes Em Floresta Natural E Em Plantios De Eucalipto E De Dandá No Sudeste Da Bahia, Brasil. Rev Árvore 26(2):193–207
Giácomo RG, Pereira MG, Machado DL (2012) Aporte e decomposição de serapilheira em áreas de cerradão e mata mesofítica na estação ecológica de Pirapitinga – MG. Ciência Florestal 22(4):669–680. Retrieved from http://www.scielo.br/pdf/cflo/v22n4/1980-5098-cflo-22-04-00669.pdf. Accessed 1 Oct 2022
Giácomo RG, Alves MC, Camara R, Pereira MG, Arruda OG, Souto SN, Moraes MLT (2017) Litterfall and nutrient input in a degraded area. Floresta Ambiente 24. https://doi.org/10.1590/2179-8087.002816
González-Rodríguez H, Domínguez-Gómez TG, Cantú-Silva I, Gómez-Meza MV, Ramírez-Lozano RG, Pando-Moreno M, Fernández CJ (2011) Litterfall deposition and leaf litter nutrient return in different locations at Northeastern Mexico. Plant Ecol 212(10):1747–1757. https://doi.org/10.1007/s11258-011-9952-9
Hawkesford M, Horst W, Kichey T, Lambers H, Schjoerring J, Møller IS, White P (2012) Functions of Macronutrients. In Marschner’s Mineral Nutrition of Higher Plants, 3rd edn. Elsevier Ltd, pp. 135–189. https://doi.org/10.1016/B978-0-12-384905-2.00006-6
Hoffmann S (2022) Challenges and opportunities of area-based conservation in reaching biodiversity and sustainability goals. Biodivers Conserv 31:325–352. https://doi.org/10.1007/s10531-021-02340-2
Holanda AC, Feliciano ALP, Freire FJ, Sousa FQ, Freire SRO, Alves AR (2017) Aporte de serapilheira e nutrienes em uma área de caatinga. Ciência Florestal 27(2):621–633. https://doi.org/10.5902/1980509827747
Holl KD, Aide TM (2011) Forest Ecology and Management When and where to actively restore ecosystems ? For Ecol Manage 261(10):1558–1563. https://doi.org/10.1016/j.foreco.2010.07.004
Holl KD (2002) Tropical Moist Forest Restoration. In Perrow M, Davy A (eds) Handbook of Restoration. Cambridge University Press, vol. II, pp. 539–558. https://doi.org/10.2307/2402276
Jordan C (1985) Nutrient cycling in tropical forest ecosystems. John Wiler, New York
Kjeldahl J (1883) A new method for the determination of nitrogen in organic matter. Z Anal Chem 22:366–382. https://doi.org/10.1007/BF01338151
Klippel VH, Pezzopane JEM, Caldeira MVWC, Silva GF, Castro KC (2016) Acúmulo de serapilheira e nutrientes em área com diferentes metodologias de Restauração Florestal. Comunicata Scientiae 7(2):241–250. https://doi.org/10.14295/CS.v7i2.521
König FG, Schumacher MV, Brun EJ, Seling I (2002) Avaliação da sazonalidade da produção de serapilheira numa Floresta Estacional Decidual no município de Santa Maria-RS. Rev Árvore 26(4):429–435. https://doi.org/10.1590/s0100-67622002000400005
Lanuza O, Casanoves F, Zahawi RA, Celentano D, Delgado D, Holl KD (2018) Litterfall and Nutrient Dynamics Shift in Tropical Forest Restoration Sites after a Decade of Recovery. Biotropica 50(3):491–498. https://doi.org/10.1111/btp.12533
Lazzaro L, Tondini E, Lombardi L, Giunti M (2020) The eradication of Carpobrotus spp. in the sand-dune ecosystem at Sterpaia (Italy, Tuscany): indications from a successful experience. Biologia 75:199–208. https://doi.org/10.2478/s11756-019-00391-z
León JD, Osorio NW (2014) Role of litter turnover in soil quality in tropical degraded lands of Colombia. Sci World J 2014(February). https://doi.org/10.1155/2014/693981
Leverkus AB, Lindenmayer DB, Thorn S, Gustafsson L (2018) Salvage logging in the world’s forests: interactions between natural disturbance and logging need recognition. Glob Ecol Biogeogr 27(10):1140–1154. https://doi.org/10.1111/geb.12772
Lima SS, Leite LFC, Aquino AM, Oliveira FC, Castro AAJF (2010) Serapilheira e teores de nutrientes em argissolo sob diferentes manejos no norte do piauí. Rev Arvore 34(1):75–84. https://doi.org/10.1590/s0100-67622010000100009
Lima MT, Ribeiro AÍ, Dias HCT, Rosa AG, Pires GT, Tonello KC (2018) The dynamics of the substrate recovery of waste dumps in calcary mining under natural regeneration. Cerne 24(1). https://doi.org/10.1590/01047760201824012476
López JH, González-Rodríguez H, Ramírez-Lozano RG, Cantú-Silva I, Gómez-Meza M, Pando-Moreno M, Estrada-Castillón AE (2013) Producción de hojarasca y retorno potencial de nutrientes en tres sitios del estado de Nuevo León, México. Polibotánica 35:41–64
Lopez Hernandez JM, Maiti R, Meza MVG, Rodriguez HG, Silva IC, Lozano RGR, Castillon AEE (2014) Litterfall production and nutrient deposition through leaf fallen in three Tamaulipan Thornscrub Communities, North-eastern Mexico. Int J Bio-Resource Stress Manag 5(2):168. https://doi.org/10.5958/0976-4038.2014.00551.x
Loumeto JJ (2003) Litterfall and nutrient return in tropical rainforest in the Chaillu area (southwest Congo). Adv Ecol Sci 19:1237–1249. https://doi.org/10.2495/ECO030462
Luciano RV, Albuquerque JA, Costa A, Batistella B, Warmling MT (2012) Atributos físicos relacionados à compactação de solos sob vegetação nativa em região de altitude no sul do Brasil. Rev Bras Cienc Solo 36(6):1733–1744. https://doi.org/10.1590/S0100-06832012000600007
Luizao FJ (1989) Litter production and mineral element input to the forest floor in a Central Amazonian forest. GeoJournal 19(4):407–417. https://doi.org/10.1007/BF00176910
Marschner H (1997) Mineral nutrition of higher pants, 2nd edn. Academic, San Diego
Martins WBR, Vale RL, Ferreira GC, Andrade VMS, Dionísio LFS, Rodrigues RP, Souza GMP (2018) Litterfall, litter stock and water holding capacity in post-mining forest restoration ecosystems, Eastern Amazon. Rev Bras Cienc Agrarias 13(3):1–9. https://doi.org/10.5039/agraria.v13i3a5546
Matoh T, Kawaguchi S, Kobayashi M (1996) Ubiquity of a borate-rhamnogalacturonan II complex in the cell walls of higher plants. Plant Cell Physiol 37(5):636–640. https://doi.org/10.1093/oxfordjournals.pcp.a028992
O’Neill MA, Ishii T, Albersheim P, Darvill AG (2004) RHAMNOGALACTURONAN II: structure and function of a borate cross-linked cell wall pectic polysaccharide. Annu Rev Plant Biol 55(1):109–139. https://doi.org/10.1146/annurev.arplant.55.031903.141750
Okeke AI, Omaliko CPE (1994) Litterfall and seasonal patterns of nutrient accumulation in Dactyladenia barteria (Hook f ex. Oliv.) Engl. bush fallow at Ozala, Nigeria. For Ecol Manag 67(1–3):345–351. https://doi.org/10.1016/0378-1127(94)90029-9
Oliveira-Filho A, Ratter J (2002) Vegetation physiognomies and woody flora of the Cerrado biome. In Marquis RJ, Oliveira PS (eds) The cerrados of Brazil: ecology and natural his- tory of a neotropical savanna, 1st edn. Columbia University, New York, pp. 91–120
Paiva AO, Silva LCR, Haridasan M (2015) Productivity-efficiency tradeoffs in tropical gallery forest-savanna transitions: linking plant and soil processes through litter input and composition. Plant Ecol 216(6):775–787. https://doi.org/10.1007/s11258-015-0466-8
Palma RM, Defrieri RL, Tortarolo MF, Prause J, Gallardo JF (2000) Seasonal changes of bioelements in the litter and their potential return to green leaves in four species of the Argentine subtropical forest. Ann Bot 85(2):181–186. https://doi.org/10.1006/anbo.1999.1005
Parzych A, Trojanowski J (2006) Precipitation and duff fall as natural sources of nitrogen and phosphorus forforest soils in the Słowiński National Park. Baltic Coastal Zone 10:47–59
Pereira LC, Balbinot L, Matus GN, Dias HCT, Tonello KC (2021) Aspects of forest restoration and hydrology: linking passive restoration and soil–water recovery in Brazilian Cerrado. J For Res. https://doi.org/10.1007/s11676-021-01301-3
Pereira LC, Balbinot L, Lima MT, Bramorski J, Tonello KC (2022) Aspects of forest restoration and hydrology: the hydrological function of litter. J For Res 33:543–552. https://doi.org/10.1007/s11676-021-01365-1
Pimenta JA, Rossi LB, Torezan JMD, Cavalheiro AL, Bianchini E (2011) Produção de serapilheira e ciclagem de nutrientes de um ref orestamento e de uma f oresta estacional semidecidual no Sul Do Brasil. Acta Bot Bras 25(1):53–57. https://doi.org/10.1590/s0102-33062011000100008
Pinto SIC, Martins SV, Barros NF, Dias HCT (2009) Ciclagem de nutrientes em dois trechos de floresta estacional semidecidual na reserva florestal mata do paraíso em Viçosa, MG, Brasil. Rev Árvore 33(4):653–663. https://doi.org/10.1590/s0100-67622009000400008
Queiroz MG, Silva TGF, Zolnier S, Souza CAA, Souza LSB, Neto S, Ferreira WPM (2019) Seasonal patterns of deposition litterfall in a seasonal dry tropical forest. Agric For Meteorol 279(August):107712. https://doi.org/10.1016/j.agrformet.2019.107712
Ratter JA, Ribeiro JF, Bridgewater S (1997) The Brazilian cerrado vegetation and threats to its biodiversity. Ann Bot 80(3):223–230. https://doi.org/10.1006/anbo.1997.0469
Rengel Z (2007) Cycling of Micronutrients in Terrestrial Ecosystems. In: Marschner ZRP (ed) Nutrient Cycling in Terrestrial Ecosystems. Springer-Verlag Berlin, vol. 10, pp. 93–121. https://doi.org/10.1007/978-3-540-68027-7_4
Restrepo MF, Florez CP, Osorio NW, León JD (2013) Passive and active restoration strategies to activate soil biogeochemical nutrient cycles in a degraded tropical dry land. ISRN Soil Sci 2013:1–6. https://doi.org/10.1155/2013/461984
Ribeiro FP, Bussinguer AP, Hodecker BER, Gatto A (2017) Conteúdo de nutrientes na serapilheira em três fisionomias do Cerrado do Distrito Federal. Pesquisa Florestal Bras 37(92):465–473. https://doi.org/10.4336/2017.pfb.37.92.1312
Röderstein M, Hertel D, Leuschner C (2005) Above- and below-ground litter production in three tropical montane forests in southern Ecuador. J Trop Ecol 21(5):483–492. https://doi.org/10.1017/S026646740500249X
Rozendaal DMA, Bongers F, Aide TM, Alvarez-Dávila E, Ascarrunz N, Balvanera P, Poorter L (2019) Biodiversity recovery of neotropical secondary forests. Sci Adv 5(3). https://doi.org/10.1126/sciadv.aau3114
Santos EMR, Franklin E, Luizão FJ (2008) Litter manipulation and associated invertebrate fauna in secondary forest, central Amazonia, Brazil. Acta Oecol 34(3):274–284. https://doi.org/10.1016/j.actao.2008.05.011
Santos HG, Jacomine PKT, Anjos LHC, Oliveira VA, Lumbreras JF, Coelho MR, Almeida JA, Cunha TJF (2018) Sistema Brasileiro de Classificação de Solos. Embrapa Solos
Sayer EJ, Rodtassana C, Sheldrake M, Bréchet LM, Ashford OS, Lopez-Sangil L, Tanner EVJ (2020) Revisiting nutrient cycling by litterfall—Insights from 15 years of litter manipulation in old-growth lowland tropical forest. Adv Ecol Res: 1–51. https://doi.org/10.1016/bs.aecr.2020.01.002
Scheer MB (2009) Fluxo de nutrientes pela precipitação pluviométrica em dois trechos de floresta ombrófila densa em Guaraqueçaba, Paraná. Floresta 39(1):117–130
Scheer MB, Gatti G, Wisniewski C (2011) Nutrient fluxes in litterfall of a secondary successional alluvial rain forest in Southern Brazil. Rev Biol Trop 59(4):1869–1882. https://doi.org/10.15517/rbt.v59i4.33192
Schrautzer J, Rinker A, Jensen K, Mϋller F, Schwartze P, Dierßen K (2007) Succession and Restoration of Drained Fens: perspectives from Northwestern Europe. In: Walker LR, Walker J, Hobbs RJ (eds) Linking Restoration and Ecological Succession. Springer, New York, pp. 90–120. https://doi.org/10.1007/978-0-387-35303-6_5
Schumacher MV, Brun EJ, Hernandes JI, König FG (2004) Produção de serapilheira em uma floresta de Araucaria angustifolia (Bertol.) Kuntze no município de Pinhal Grande-RS. Rev Árvore 28(1):29–37. https://doi.org/10.1590/s0100-67622004000100005
Souza T (2022) The living soil. in: soil biology in tropical ecosystems. Springer, Cham. https://doi.org/10.1007/978-3-031-00949-5_2
Terror VL, Sousa HC, Kozovits AR (2011) Produção, decomposição e qualidade nutricional da serapilheira foliar em uma floresta paludosa de altitude. Acta Bot Bras 25(1):113–121. https://doi.org/10.1590/s0102-33062011000100014
Tonello KC, Rosa AG, Pereira LC, Matus GN, Guandique MEG, Navarrete AA (2021) Rainfall partitioning in the Cerrado and its influence on net rainfall nutrient fluxes. Agric For Meteorol 303:108372. https://doi.org/10.1016/j.agrformet.2021.108372
Valenti M, Cianciaruso M, Batalha M (2008) Seasonality of litterfall and leaf decomposition in a cerrado site. Braz J Biol 68(3):459–465. https://doi.org/10.1590/S1519-69842008000300002
Van der Heijden G, Legout A, Pollier B, Mareschal L, Turpault MP, Ranger J, Dambrine E (2013) Assessing Mg and Ca depletion from broadleaf forest soils and potential causes - A case study in the Morvan Mountains. For Ecol Manag 293:65–78. https://doi.org/10.1016/j.foreco.2012.12.045
Van der Heijden G, Legout A, Pollier B, Ranger J, Dambrine E (2014) The dynamics of calcium and magnesium inputs by throughfall in a forest ecosystem on base poor soil are very slow and conservative: evidence from an isotopic tracing experiment (26Mg and 44Ca). Biogeochemistry 118(1–3):413–442. https://doi.org/10.1007/s10533-013-9941-2
Vital ART, Guerrini IA, Franken WK, Fonseca RCB (2004) Produção de serapilheira e ciclagem de nutrientes de uma floresta estacional semidecidual em zona ripária. Rev Árvore 28(6):793–800. https://doi.org/10.1590/s0100-67622004000600004
Vitousek P (1982) Nutrient cycling and nutrient use efficiency. Am Nat 119(4):553–572. https://doi.org/10.1086/283931
Vitoussek PM, Sanford RL (1986) Nutrient cycling in moist tropical forest. Annu Rev Ecol Syst 17(November 2003):137–167. https://doi.org/10.1146/annurev.es.17.110186.001033
Yang YS, Guo JF, Chen GS, Xie JS, Ren G, Zhen L, Zhao J (2005) Litter production, seasonal pattern and nutrient return in seven natural forests compared with a plantation in southern China. Forestry 78(4):403–415. https://doi.org/10.1093/forestry/cpi044
Yeomans JC, Bremner JM (1988) A rapid and precise method for routine determination of organic carbon in soil1. Commun Soil Sci Plant Anal 19(13):1467–1476. https://doi.org/10.1080/00103628809368027
Zhang H, Yuan W, Dong W, Liu S (2014) Seasonal patterns of litterfall in forest ecosystem worldwide. Ecol Complex 20:240–247. https://doi.org/10.1016/j.ecocom.2014.01.003
Zhang G, Zhang P, Peng S, Chen Y, Cao Y (2017) The coupling of leaf, litter, and soil nutrients in warm temperate forests in northwestern China. Sci Rep 7(1):1–11. https://doi.org/10.1038/s41598-017-12199-5
Zhang X, Zhang G, Hu C, Ping J, Shengqi J (2020) Response of soil moisture to landscape restoration in the hilly and gully region of the Loess Plateau, China. Biologia 75:827–839. https://doi.org/10.2478/s11756-020-00520-z
Funding
This research was funded by Coordination for the Improvement of Higher Education Personnel–CAPES through the Postgraduate Program in Planning and Using of Renewable Resources, Environmental Science Department, Federal University of São Carlos, São Paulo, Brazil, Finance Code 001; the Brazilian National Council for Scientific and Technological Development (CNPq) and Sylvamo Ltda from Brazil.
Author information
Authors and Affiliations
Contributions
Conceptualization, methodology, validation, LCP and KCT; Formal analysis, LCP, KCT and JB; Investigation, LCP, LB and KCT; Data curation, LCP, LB and KCT; Writing—original draft preparation, KCT and JB; Writing—review and editing, KCT, EON and MHM All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tonello, K.C., Pereira, L.C., Balbinot, L. et al. Patterns of litter and nutrient return to the soil during passive restoration in Cerrado, Brazil. Biologia 78, 399–414 (2023). https://doi.org/10.1007/s11756-022-01224-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11756-022-01224-2