Abstract
Renovascular hypertension is defined as the reduction in renal perfusion resulting in sustained hypertension. This study aims to investigate the possible effects of myrtle leaf extract (Myrtus communis L.) on, heart, kidney and aorta tissues in the experimental renovascular hypertension (RVH). 32 male Wistar Albino rats were divided into four groups as control, hypertension, hypertension+ramipril, and hypertension+myrtle leaf extract treatment groups. RVH model was induced by Goldblatt’s 2-kidney 1-clip method. 12 weeks later than the treatment blood pressures were recorded and oxidant and antioxidant parameters, tissue factor activity, and histological analysis were determined in the kidney, heart, and aortic tissues. The blood pressure levels of the hypertension group significantly increased compared to the control group. Administration of myrtle leaf extract and ramipril significantly decreased the increased blood pressure. In the hypertension group, oxidative damage increased in the kidney, heart, and aorta tissues. In the histological evaluation of tissues in RVH, heart muscle fibres degenerated. Bowman capsule and glomeruli dilated and tubules damaged in the kidney. Myrtle leaf extract administration regenerated the damages and degenerations. The administration of myrtle leaf extract restored the impaired oxidant-antioxidant balance in the heart, kidney and aorta tissues of hypertensive rats. Myrtle leaf extract can be considered as an alternative antihypertensive treatment target in the prevention of oxidative stress-induced damage in renovascular hypertension.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hypertension is one of the major risk factors in the development of cardiovascular diseases. It is classified as primary and secondary hypertension according to its formation mechanism (Mancia et al. 1997). The type of hypertension investigated in this study is related to secondary hypertension. Secondary hypertension is an increase in blood pressure caused by an underlying disease and often with a treatable cause (Textor 2017). The common cause of secondary hypertension is renovascular hypertension (RVH).
RVH is high blood pressure due to occlusive disease of one or both main renal arteries or their branches. Approximately 1% of all hypertension cases are of renovascular origin (Boutari et al. 2020). The cause of RVH is renal artery stenosis, and it may also cause renal diseases (Lerman et al. 2001). RVH is related to the renin-angiotensin-aldosterone system.
Experimental renovascular hypertension is generated in rats by clamping one of the renal arteries with silver clips. This model is also called as Goldblatt two-kidney, one-clip (2K1C) model (Goldblatt et al. 1934). This model has three phases. In the first phase (first six weeks) blood pressure rises rapidly as the renin-angiotensin system (RAS) is overactivated and plasma renin activity and circulating angiotensin II (A- II) levels increased. In the second phase (following two weeks) salt and water retention occur. This triggers additional pressor effects besides the stable high blood pressure. In the third phase (the ninth week after renal ischemia) RAS independent high blood pressure occurs (Martinez-Maldonado 1991; de Oliveira et al. 2012).
The cascade of the renin-angiotensin-aldosterone system begins with angiotensinogen, α-2-globulin secreted from the liver. Renin secreted from kidneys converts this substrate to angiotensin I (A-I). A-I is then converted to A-II via the angiotensin-converting enzyme (ACE) and binds to A-II receptors. Consequently, blood pressure increases through vasoconstriction, renal tubular and glomerular function changes, hypertrophy of the heart, and fibrosis (Basso and Terragno 2001). Recent studies reported some additional mechanisms of A-II that increase vascular resistance including vasoconstriction and endothelial dysfunction related to oxidative stress responsible for sustained increased blood pressure (Garovic and Textor 2005).
Angiotensin formation is prevented by ACE inhibitors. These inhibitors control the bradykinin breakdown and may be effective on other members of the renin-angiotensin-aldosterone system. These agents have many effects that can also protect coronary and peripheral vascularity. Ramipril is one of these agents that acts on the heart and blood vessels, and it is used in the treatment of hypertension and heart failure (Lonn et al. 2003).
Reactive oxygen species (ROS) cause oxidative damage to kidneys and vessels in renovascular hypertension (Briones and Touyz 2010). ROS mediate vasoconstriction by increasing intracellular calcium concentration. Although the role of antioxidant use in the treatment of hypertension remains a question, it is thought that hypertension-related tissue damage can be prevented by treating oxidative damage (Griendling et al. 2021). Targeted treatments against free radicals, either reducing the formation of ROS or increasing antioxidants, may reduce vascular damage and renal dysfunction, thereby preventing or regressing hypertensive end-organ damage (Rodrigo et al. 2011; Griendling et al. 2021).
Nowadays, the interest in natural antioxidant products to prevent and treat various complications increases. Although findings regarding the long-term use of antioxidants in the development of cardiovascular complications of hypertension are not yet clear, there are encouraging findings regarding the use of antioxidants in restoring endothelial function and blood pressure (Fang et al. 2002; Schulz et al. 2004).
Myrtle (M. communis L.) is a plant of the Myrtaceae family that is common in Turkey and many other Mediterranean countries. It has been used in folk medicine to treat hyperglycemia, hypertension, anxiety, lung infections like bronchitis, tuberculosis, heartburn, yeast infections and skin warts (Aydın and Özcan 2007; Sisay and Gashaw 2017). In the chemical content of the leaves and fruits of the myrtle plant, there are volatile compounds or essential oils like α-pinene and 1,8-cineole with terpenoid structure, flavonoids like quercetin, and catechin, fatty acids like oleic and linoleic acid, coumarins, galloyl-glucosides, ellagitannins, gallic and ellagic acids (Hennia et al. 2018). The antioxidant/anti-inflammatory activity and total phenolic content of myrtle that was used in this study were previously presented (Ozbeyli et al. 2020). It is no surprise that myrtle, which has such a rich chemical content, has antimicrobial, hypoglycemic, antihypertensive, hemostatic, antioxidant, anti-inflammatory, and anticarcinogenic features (Sumbul et al. 2011).
This study aims to examine the possible antihypertensive and antioxidant effects of the myrtle leaf extract on the kidney, heart, and aortic tissues in the experimental hypertension model and to compare them with the effects of ramipril, an antihypertensive agent.
Materials and methods
Plant material and extraction
The myrtle (M. communis) leaves were collected from Manisa, Turkey, and identified by Dr. Gizem Emre, a botanist in the School of Pharmacy, University of Marmara Voucher specimens were deposited in the Herbarium in the School of Pharmacy, Marmara University (MARE no: 13006). The 100 g dried plant leaves were extracted with 96% ethanol via the Soxhlet apparatus. The crude extract was filtered, and the filtrate was dried under a vacuum. The dried ethanol extract was stored at 4 °C.
Animals
Wistar albino rats (male, 200–300 g, n = 32) were kept in a humidity (65–70%) and a constant temperature (22 ± 2 °C) with regulated light and dark (12/12 h) cycles. Standard pellets and tap water was given to all animals ad libitum.
All the experimental protocols were approved by the Marmara University Animal Care and Use Committee (Approval date: 03.12.2018 Certification Number:112.2018.mar).
Experimental design
The rats were divided into four experimental groups as control, RVH, RVH + myrtle leaf extract (RVH + M), and RVH + ramipril treatment (RVH + R). The renovascular hypertension (RVH) model was applied intraperitoneally to rats under ketamine (100 mg/kg, intraperitoneally) and chlorpromazine (0.75 mg/kg, intraperitoneally) injection anaesthesia. Two kidneys, one clip (Goldblatt, 2K1C) procedure have been used to induce the RVH model (Cevikelli-Yakut et al. 2020). The silver clip (diameter 0.25 mm) was inserted over the left renal artery to form RVH. The rats in the control group underwent similar surgical procedures but no silver clips were inserted. The tail-cuff method was used to measure the blood pressure of the rats. The animals have received either myrtle leaf extract (100 mg/kg, intraperitoneally) or ramipril (10 mg/kg, intraperitoneally) for 12 weeks, starting with the surgery day. Animals in the control and RVH groups were received saline with 1% alcohol intraperitoneally (1 mL/kg/24 s) for 12 weeks. The study of Yakut-Çevikelli et al. was used to determine the antihypertensive dose of ramipril and M. communis extract (Yakut et al. 2020).
Systolic blood pressures were obtained in all groups before the surgical transactions, and measurement was repeated after three weeks and also at the end of the experiment. At the end of the twelfth week, all blood pressure measurements were done in all animals before they were decapitated. To evaluate injuries in the targeted organs, renal, cardiac, and aortic tissues were excised carefully and stored at -80 °C to determining the malondialdehyde (MDA), glutathione (GSH), nitric oxide (NO) levels, superoxide dismutase (SOD), catalase (CAT), myeloperoxidase (MPO), and tissue factor (TF) activities.
Measurement of blood pressure
Blood pressures were measured indirectly (Biopac MP35 Systems, Inc. COMMAT Ltd., Ankara, Turkey) by the tail-cuff method at the end of the third and twelfth weeks following the operations. Animals were placed in the pre-heated (35 °C) chamber. The animals were then placed in individual plastic holders and a cuff with a pulse sensor was wrapped around the tail. Blood pressure values were measured three consecutive times and then averaged.
Determination of malondialdehyde and glutathione
Malondialdehyde (MDA) activities in the kidney, heart, and aorta samples were measured according to the Ledwozyw method (Ledwozyw et al. 1986). Lipid peroxidation results were expressed in MDA nmol/g tissue. Glutathione (GSH) levels were measured using the Ellman method (Beutler 1984). The results are expressed as μmol GSH/g tissue.
Determination of superoxide dismutase and catalase activities
Superoxide dismutase (SOD) activity in the tissues of the heart, kidney, and aorta was measured following the Mylorie method (Mylroie et al. 1986). Bovine SOD (Sigma) was used in the preparation of the standard curve.
CAT activity was measured according to the Aebi method, which is based on the decomposition reaction of H2O2 to give H2O and O2 (Aebi 1974).
Determination of nitric oxide and myeloperoxidase activity
Nitric oxide (NO) levels in the heart, kidney and aortic tissues were determined by the Miranda method (Miranda et al. 2001).
Myeloperoxidase (MPO) activity has been assessed by measuring the oxidation of H2O2-dependent o-dianisidine.2HCl. The change in absorbance in 1.0 min−1 at 460 nm is defined as the one unit of MPO enzyme activity (Hillegass et al. 1990).
Determination of tissue factor activity
Tissue factor activity was determined by using blood plasma from healthy subjects and homogenates of heart, kidney, and aorta tissues according to the Ingram method (Ingram 1976). Briefly, 0.1 mL tissue homogenate and blood plasma were mixed and incubated at 37 °C for 2 min. The fibrin clot formation time was recorded after the addition of CaCl2 (0.02 M). Tissue factor activity decreases as the time taken for clot formation increases.
Histological evaluation
Kidney, heart, and aorta tissue samples were fixed in formaldehyde for histologic examination. After fixation, they were dehydrated in graded concentrations of alcohol series and embedded in paraffin. Three μm sections were taken from the paraffin blocks and used for Hematoxylin-eosin staining.
Histological evaluation was performed on stained sections by two histologists independently and blindly as described previously (Şener et al. 2005). In the evaluation of kidneys, degeneration status was scored in three different areas: glomerular, tubular, and vascular areas. For heart and kidney tissues, a whole evaluation was applied and along with degeneration status, inflammation was also scored. Scores were assigned from 0 to 3 (0: absent, 1: mild, 2: moderate, and 3: severe), and overall scores were determined by summing these individual scores (a maximum of 9 for kidney tissue, 6 for heart, and aorta tissue) and statistically evaluated.
LC-ESI-MS/MS analysis
LC-MS/MS analysis was carried out using an Absciex 3200 Q trap MS/MS detector. Experiments were performed with a Shimadzu 20A HPLC system coupled to an Applied Biosystems 3200 Q-Trap LC-MS/MS instrument equipped with an ESI source operating in negative ion mode. For the chromatographic separation, a GL Science Inertsil ODS - 3250 × 4.6 mm, 5 μm particle size, analytical column operating at 40 °C has been used. The solvent flow rate was maintained at 0.5 mL/min. Detection was carried out with a PDA detector. The elution gradient consisted of mobile phases (A) acetonitrile:water:formic acid (10:89:1, v/v/v) and (B) acetonitrile:water:formic acid (89:10:1, v/v/v). The composition of B was increased from 10% to 100% in 40 min. LC-ESI-MS/MS data were collected and processed by Analyst 1.6 software.
Statistical analysis
Statistical analysis of all parameters was performed using Graph Pad statistical package (San Diego; CA, USA). The findings were expressed as mean and standard deviation. One-way analysis of variance (ANOVA) followed by Tukey’s post hoc test was carried out for the evaluation of biochemical data.
Results
Body and tissue weights
At the end of the experiment, no significant difference was observed between the groups in terms of body weight measurements. Considering the weight of the tissues, a significant reduction was observed in the heart and left kidney tissues in the ramipril treatment group compared to all groups. The right kidney tissues did not change among all groups (Table 1).
Systolic blood pressure levels
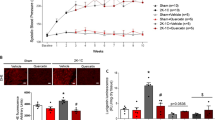
The basal systolic blood pressures were recorded before the surgery. There were no significant differences between the groups. In the RVH group, systolic blood pressure significantly increased at the third- and twelfth-week readings after the surgery compared to the basal levels. Moreover, myrtle leaf extract treatment to the RVH group caused slight increases in systolic blood pressure at the third- and twelfth-week compared to the RVH group. RVH + R group has also shown a slight increase in systolic blood pressure in both third- and twelfth-week compared to the RVH group after surgery. There were no significant differences in systolic blood pressure levels between the myrtle leaf extract and ramipril treated groups at both third and twelfth weeks (Table 2).
Heart tissue results
Renovascular hypertension increased MDA level, NO level, and MPO activity, and significantly decreased GSH level, SOD activity, TF activity compared to the control group. Myrtle leaf extract treatment significantly decreased MDA level, and significantly increased GSH level, SOD activity, CAT activity compared to the RVH group. In the RVH + R group, MDA and NO levels, MPO, and CAT activities significantly increased compared to the RVH group. When the myrtle leaf extract and ramipril treated RVH groups were compared MDA and NO levels, TF, MPO, and CAT activities were significantly higher than the myrtle leaf extract treated RVH group (Fig. 1). Histological evaluation of heart tissues showed that muscle fibres were severely damaged in the RVH group. Moreover, inflammatory cell infiltrations were detected in this group. RVH + M and RVH + R groups demonstrated less damage in muscle fibres compared to the RVH group (Fig. 4).
MDA, GSH, NO levels and MPO, SOD, CAT and TF activities of heart tissue. C: Control group, RVH: Renovascular hypertension group, RVH + M: Myrtle treatment group, RVH + R: Ramipril treatment group, MDA: Malondialdehyde, MPO: Myeloperoxidase activity, GSH: Glutathione, SOD: Superoxide dismutase, CAT: Catalase NO: Nitric oxide, (*): p < 0.05 compared to control group, (+): p < 0.05compared to RVH group, (x): p < 0.05 compared to RVH + M group, n = 8
Kidney tissue results
Right kidney
Renovascular hypertension significantly increased MDA and NO levels and significantly decreased CAT activity. Myrtle leaf extract treatment to RVH group significantly decreased MDA level, significantly increased GSH level, SOD, and CAT activities compared to RVH group. When the myrtle leaf extract treated and ramipril treated RVH groups were compared; MDA, GSH, and NO levels were significantly higher in the RVH + R group compared to the RVH + M group and, NO level and SOD activity were significantly lower in the RVH + R group. MPO and TF activities did not significantly change among all groups (Fig. 2).
MDA, GSH, NO levels and MPO, SOD, CAT and TF activities of right and left kidney tissues. C: Control group, RVH: Renovascular hypertension group, RVH + M: Myrtle treatment group, RVH + R: Ramipril treatment group, MDA: Malondialdehyde, MPO: Myeloperoxidase activity, GSH: Glutathione, SOD: Superoxide dismutase, CAT: Catalase NO: Nitric oxide, (*): p < 0.05 compared to control group, (+): p < 0.05compared to RVH group, (x): p < 0.05 compared to RVH + M group, n = 8
Left kidney
Renovascular hypertension significantly increased MDA and NO levels and TF and MPO activities and significantly decreased SOD activity compared to the control group. Myrtle leaf extract treatment to RVH group significantly decreased MDA level and TF activity and significantly increased NO compared to RVH and the control group. Ramipril treatment to the RVH group significantly increased MDA level and MPO activity and decreased GSH level, SOD activity compared to other groups. TF activity was significantly lower in the RVH+ R group compared to the RVH + M group. NO level was found to be significantly higher than the RVH + M group in the RVH + R group. CAT activity did not significantly change among all groups (Fig. 2).
The histological evaluation of the kidney revealed degenerated Bowman capsule and glomeruli, dilated tubules and epithelial damage of tubules, and interstitial oedema in the RVH group. These degenerations alleviated in RVH + M and RVH + R groups (Fig. 4).
Aortic tissue results
MDA and NO levels, and MPO activity significantly increased, GSH level and SOD activity significantly decreased in the RVH group compared to the control group. Myrtle leaf extract treatment significantly decreased MDA, MPO, and NO levels and significantly increased SOD and CAT activity compared to RVH and the control group. Ramipril treatment to the RVH group did not change the increased MDA and continued to increase MPO activity compared to the RVH group. In the RVH + R group; while SOD, CAT, and TF activities were significantly lower than the RVH + M group, NO level and MPO level significantly higher than the myrtle leaf extract treated group (Fig. 3).
MDA, GSH, NO levels and MPO, SOD, CAT and TF activities of aorta tisuue. C: Control group, RVH: Renovascular hypertension group, RVH + M: Myrtle treatment group, RVH + R: Ramipril treatment group, MDA: Malondialdehyde, MPO: Myeloperoxidase activity, GSH: Glutathione, SOD: Superoxide dismutase, CAT: Catalase NO: Nitric oxide, (*): p < 0.05 compared to control group, (+): p < 0.05compared to RVH group, (x): p < 0.05 compared to RVH + M group, n = 8
In histologic evaluation, aorta wall assessment disclosed that in the RVH group, many degenerated smooth muscle cells in tunica media and damaged and lost endothelial cells in tunica intima. In RVH + M group, smooth muscle cells were less damaged and endothelial cells were only partially lost. In RVH + R group both (Fig. 4).
Histological evaluation of heart, kidney, and aorta wall. (A) The normal morphology of cardiac muscles in the control group (1) severely damaged muscle fibers (arrow) and inflammatory cell infiltration (arrowhead) in the RVH group (2) and less damaged muscle fibers (arrow) in RVH + M and RVH + R groups (3, 4) Hematoxylene & Eosine stainings. Scale bar: 20 μm. (B) The normal morphology of kidney in the control group (1) degenerated Bowman capsule and glomeruli (*), dilated tubules (arrowhead), epithelial damage of tubules (arrow), interstitial edema (>) in RVH group (2) less damaged Bowman capsule, and glomeruli (*), epithelium of tubules (arrow), moderate interstitial edema (>) in RVH + M and RVH + R groups (3, 4). Hematoxylin & Eosin stainings. Scale bar: 50 μm. (C) The normal morphology of aorta wall in the control group (1) degenerated smooth muscle cells in tunica media (arrowhead), damage and loss of endothelial cells in tunica intima (arrow) in RVH group (2) less damaged smooth muscle cells in tunica media (arrowhead) and partially lost endothelial cells in tunica intima in RVH + M (3) less damaged smooth muscle cells in tunica media (arrowhead) and endothelial cells (arrow) in tunica intima in RVH + R group (4) Hematoxylene & Eosine stainings. Scale bar: A) 50 μm, B-D) 20 μm. (D) Damage scores of heart left kidney, and aorta wall. C: Control group, RVH: Renovascular hypertension group, RVH + M: Myrtle treatment group, RVH + R: Ramipril treatment group. (*): p < 0.05 compared to control group, (+): p < 0.05 compared to RVH group, n = 8
When the damage scores of the heart, left kidney, and aorta were compared, it was found that while the damage score increased significantly in the RVH group in all tissues, both myrtle leaf extract treatment, and ramipril were effective in decreasing the damage score (Fig. 4).
Phytochemical characterization of phenolic compounds Myrtle leaf extract by LC-MS/MS
Seven phenolic compounds including myricetin hexoside, myricetin rhamnoside, ellagic acid, quercetin rhamnoside, myricetin, trihydroxy cinnamic acid derivative, sinapinic acid derivative, were identified by LC/MS-MS (Fig. 5) (Table 3).
Discussion
This study investigated the possible effects of myrtle leaf extract, a medicinal plant with antioxidant properties on kidney, heart, and aortic tissues in experimental renovascular hypertension. The obtained results of the myrtle leaf extract administration were compared with those obtained with ramipril, a second-generation angiotensin-converting enzyme (ACE) inhibitor. The blood pressure levels of rats in which renovascular hypertension was induced by attaching a clip to the left kidney significantly increased compared to the control group. When the effects of increased blood pressure on tissue damage were evaluated, it was found that MDA and NO levels in the heart, right and left kidney and aorta were higher than in the control group. These findings indicate that oxidative damage occurs in the heart, kidney, and aorta and an increased NO was used for the adaptation of these tissues to high blood pressure. Besides, SOD activity increased and GSH level decreased in heart tissue. Heart MPO also increased in the renovascular hypertensive rats. When the kidney results were evaluated in the present study, it was observed that SOD activity decreased in the left kidney tissue and CAT activity decreased in the right kidney tissue. GSH level decreased in the aortic tissue. In line with these results, it has been determined that while hypertension increases oxidative damage in the heart, kidney, and aortic tissues and due to the use of antioxidant system elements against oxidative damage, the antioxidant defence capacity decreased. The results obtained from the hypertensive rat tissues were compatible with the literature. Agunloye et al. (Agunloye et al. 2021) determined the increased cardiac MDA and decreased heart and kidney CAT activity and GSH level in cyclosporine A-induced hypertensive rats. Similarly, Aekthammarat et al. (Aekthammarat et al. 2019) reported the increased thoracic aorta MDA level and NO activity, decreased SOD and CAT activities in the Nε-nitro-L-arginine methyl ester (L-NAME) induced in rats.
In the present study, TF activity decreased in the heart tissue of rats with hypertension. It also increased in the left kidney tissue and did not change in the right kidney and aorta. Histological examination results also confirmed the serious damage to heart muscle fibres. The increase in TF activity in the left kidney is related to the attached clip, indicating an increased tendency for clot formation in this tissue.
Today, the most commonly used herbs for hypertension treatment are lemon and garlic (Biçen et al. 2012), however, studies are showing that various plant extracts can also treat hypertension through different mechanisms. Wang et al. reported that naringenin improves kidney damage in renovascular hypertension by ensuring the balance of renin-angiotensin system components (Wang et al. 2019b). Toklu et al. stated that resveratrol, with its antioxidant effect, maintains the oxidant / antioxidant balance and reduces the oxidant damage caused by hypertension in the heart, kidney, and brain tissue in hypertension (Toklu et al. 2010). Talha et al. presented the list of plants containing anti-hypertensive activity (Talha et al. 2011). Çevikelli et al. stated in their study that myrtle protects the brain tissue against oxidative damage in experimental hypertension (Cevikelli-Yakut et al. 2020).
In the present study, the possible effects of myrtle leaf extracton heart, kidney, and aortic tissues in experimental renovascular hypertension were investigated. Myrtle leaf extract reduced MDA levels in heart, kidney, and aorta tissues in renovascular hypertension. It was determined that it did not change MPO activity and NO level in heart tissue, and increased heart GSH level, SOD, and CAT activity. Histological analysis also demonstrated less damage in muscle fibres of the heart by the myrtle leaf extract administration to hypertensive rats. It is obvious from these findings that myrtle leaf extract as a strong antioxidant activity on the heart in hypertension. While myrtle leaf extract decreases MDA and NO levels in the right kidney tissue, it also increases the right kidney GSH level and SOD activity. The protective effect of the myrtle leaf extract plant against oxidant damage was also observed in the right kidney in hypertension. Myrtle leaf extract also decreased TF activity and increased NO level in left kidney tissue. From these results, it was determined that myrtle leaf extract decreased the lipid peroxidation level in the left kidney by a mechanism other than GSH level, SOD, and CAT parameters. Myrtle leaf extract also alleviated the degenerations seen in both kidney tissue. Myrtle leaf extract decreased NO levels in aortic tissue in hypertension and increased SOD and CAT activity. Myrtle leaf extract showed its antioxidant properties by increasing aortic SOD and CAT activity, thus reducing lipid peroxidation. In the histologic examination, smooth muscle cells aorta were less damaged and endothelial cells were only partially lost in the myrtle leaf extract administered hypertensive rats compared to hypertensive rats.
The antioxidant potential of myrtle is attributed to the presence of polyphenolic compounds which may have many benefits in treating oxidative stress-related diseases (Bouaziz et al. 2015). The myrtle leaves in the current study were found to have phenolic compounds such as myricetin hexoside, myricetin rhamnoside, ellagic acid, quercetin rhamnoside, myricetin, trihydroxy cinnamic acid derivative, caffeic acid derivative and sinapinic acid derivative. Similarly, the HPLC analysis of myrtle leaves showed that they contained phenolic compounds rich in myricetin derivatives. In particular, myricetin 3-O-rhamnoside has been identified as one of major compounds in myrtle leaves (Romani et al. 1999). In a previous study, myrtle leaves were reported to have high phenolic content (472.7 ± 2.36 mg/g extract as gallic acid equivalent) with significant in vitro antioxidant activity (IC50 DPPH: 12.66 and IC50 ABTS: 17.29 μg/mL) (Ozbeyli et al. 2020). Phenolic compounds are generally known for their antioxidant activity (Haminiuk et al. 2012). Also, myricetin 3-O-rhamnoside have been reported to have antioxidant activity Hayder et al. 2008). Furthermore, phenolic compounds have been reported to act as modulators of NADPH oxidases in hypertension (Yousefian et al. 2019). Evidences indicated that myricetin derivative compounds had antihypertensive activity (Calassara et al. 2020; Wang et al. 2019a).
In this study, the effects of myrtle leaf extract on heart, kidney, and aortic tissues in hypertension were compared with the effects of ramipril, an ACE inhibitor, on hypertension. Administration of ramipril to the hypertensive rat significantly increased the MDA level in the heart, left kidney, right kidney, and aorta compared to the myrtle leaf extract given hypertensive group. The MPO activity except for the right kidney also increased heart, left kidney, and aorta compared to the myrtle leaf extract given hypertensive group. The increased MDA level and MPO activity mostly caused low antioxidant responses in the heart, kidney, and aorta. Based on these results, it is thought that ramipril does not have a significant antioxidant effect, but it contributes to the antioxidant capacity in the examined tissues by preventing the increase in blood pressure due to its blood pressure-lowering effect mechanism.
Conclusions
It was determined that both myrtle leaf extract and ramipril had antihypertensive effects. According to the results obtained, it was determined that myrtle leaf extract is effective on blood pressure due to its antioxidant effect. Although ramipril decreases blood pressure, it was ineffective at reducing the g LPO increase in the heart, kidney, and aorta tissues. These results demonstrate that myrtle (M. communis) leaf extract, significantly improves the oxidative damage in the heart, kidney, and aorta. Therefore, the ethanol extract of myrtle leaf can be an alternative therapeutic target against renovascular hypertension-induced oxidative stress-based heart, kidney and aorta damages.
Abbreviations
- RVH:
-
Renovascular Hypertension
- RAS:
-
Renin-Angiotensin System
- 2K1C:
-
Two-Kidney, One-Clip
- A- II:
-
Angiotensin II
- A- I:
-
Angiotensin I
- ACE:
-
Angiotensin-Converting Enzyme
- ROS:
-
Reactive oxygen species
- M:
-
Myrtle leaf extract leave extract
- R:
-
Ramipril
- MDA:
-
Malondialdehyde
- GSH:
-
Glutathione
- NO:
-
Nitric Oxide
- SOD:
-
Superoxide Dismutase
- CAT:
-
Catalase
- MPO:
-
Myeloperoxidase
- TF:
-
Tissue Factor
- HPLC:
-
High Performance Liquid Chromatography
- LC-ESI-MS:
-
Liquid Chromatography-Electrospray Ionization-Mass Spectrometry
- ANOVA:
-
One Way Analysis of variance
- L-NAME:
-
Nε-nitro-L-arginine methyl ester
References
Aebi H (1974) Catalase. In: HU B (ed.) Methods of enzymatic analysis. Verlag Chemie/Academic Press Inc, Weinheim/Newyork, pp. 673–684
Aekthammarat D, Pannangpetch P, Tangsucharit P (2019) Moringa oleifera leaf extractlowers high blood pressure by alleviating vascular dysfunction and decreasing oxidative stress in L-NAME hypertensive rats. Phytomedicine 54:9–16. https://doi.org/10.1016/j.phymed.2018.10.023
Agunloye OM, Oboh G, Bello GT, Oyagbemi AA (2021) Caffeic and chlorogenic acids modulate altered activity of key enzymes linked to hypertension in cyclosporine-induced hypertensive rats. J Basic Clin Physiol Pharmacol 32(3):169–177. https://doi.org/10.1515/jbcpp-2019-0360
Aydın C, Özcan MM (2007) Determination of nutritional and physical properties of myrtle (Myrtus communis L.) fruits growing wild in Turkey. J Food Eng 79:453–458. https://doi.org/10.1016/j.jfoodeng.2006.02.008
Basso N, Terragno NA (2001) History about the discovery of the renin-angiotensin system. Hypertension 38:1246–1249. https://doi.org/10.1161/hy1201.101214
Beutler E (1984) Red cell metabolism: a manual of biochemical methods
Biçen C, Elver Ö, Erdem E, Kaya C, Karataş A, Dilek M, Akpolat T (2012) Herbal product use in hypertension patients. J Exp Clin Med 29:109–112. https://doi.org/10.5835/jecm.omu.29.02.005
Bouaziz A, Abdalla S, Baghiani A, Charef N (2015) Phytochemical analysis, hypotensive effect and antioxidant properties of Myrtus communis L. growing in Algeria. Asian Pac J Trop Biomed 5:19–28. https://doi.org/10.1016/S2221-1691(15)30165-9
Boutari C, Georgianou E, Sachinidis A, Katsimardou A, Christou K, Piperidou A, Karagiannis A (2020) Renovascular hypertension: novel insights. Curr Hypertens Rev 16:24–29. https://doi.org/10.2174/1573402115666190416153321
Briones AM, Touyz RM (2010) Oxidative stress and hypertension: current concepts. Curr Hypertens Rep 12:135–142. https://doi.org/10.1007/s11906-010-0100-z
Calassara LL, Pinto SC, Condack CP, Leite BF, Nery LCE, Tinoco LW, Aguiar FA, Leal IC, Martins SM, Silva LL (2020) Isolation and characterization of flavonoids from Tapirira guianensis leaves with vasodilatory and myeloperoxidase-inhibitory activities. Nat Prod Res:1–4. https://doi.org/10.1080/14786419.2020.1784170
Cevikelli-Yakut ZA, Ertas B, Sen A, Koyuncuoglu T, Yegen BC, Sener G (2020) Myrtus communis improves cognitive impairment in renovascular hypertensive rats. J Physiol Pharmacol 71. https://doi.org/10.26402/jpp.2020.5.07
D’Urso G, Montoro P, Lai C, Piacente S, Sarais G (2019) LC-ESI/LTQOrbitrap/MS based metabolomics in analysis of Myrtus communis leaves from Sardinia (Italy). Ind Crop Prod 128(23):354–362. https://doi.org/10.1016/j.indcrop.2018.11.022
de Oliveira JC, Antonietto CRK, Scalabrini AC, Marinho TS, Pernomian L, Corrêa JWN, Restini CBA (2012) Antioxidant protective effects of the resveratrol on the cardiac and vascular tissues from renal hypertensive rats. Open J Med Chem 2(3):61–71. https://doi.org/10.4236/ojmc.2012.23008
Fang JC, Kinlay S, Beltrame J, Hikiti H, Wainstein M, Behrendt D, Suh J, Frei B, Mudge GH, Selwyn AP (2002) Effect of vitamins C and E on progression of transplant-associated arteriosclerosis: a randomised trial. Lancet 359:1108–1113. https://doi.org/10.1016/S0140-6736(02)08154-0
Francisco M, Moreno DA, Cartea ME, Ferreres F, García-Viguera C, Velasco P (2009) Simultaneous identification of glucosinolates and phenolic compounds in a representative collection of vegetable Brassica rapa. J Chromatogr A 1216(38):6611–6619. https://doi.org/10.1016/j.chroma.2009.07.055
Garovic V, Textor SC (2005) Renovascular hypertension: current concepts. Semin Nephrol 25:261–271. https://doi.org/10.1016/j.semnephrol.2005.02.010
Goldblatt H, Lynch J, Rf H, Summerville WW (1934) Studies on Experımental hypertension : I. the Productıon of Persıstent elevation of systolic blood pressure by means of renal Ischemıa. J Exp Med 59:347–379. https://doi.org/10.1084/jem.59.3.347
Griendling KK, Camargo LL, Rios FJ, Alves-Lopes R, Montezano AC, Touyz RM (2021) Oxidative stress and hypertension. Circ Res 128:993–1020
Haminiuk CW, Maciel GM, Plata-Oviedo MS, Peralta RM (2012) Phenolic compounds in fruits–an overview. Int J Food Sci Technol 47:2023–2044. https://doi.org/10.1161/CIRCRESAHA.121.318063
Hayder N, Bouhlel I, Skandrani I, Kadri M, Steiman R, Guiraud P, Mariotte A-M, Ghedira K, Dijoux-Franca M-G, Chekir-Ghedira L (2008) In vitro antioxidant and antigenotoxic potentials of myricetin-3-o-galactoside and myricetin-3-o-rhamnoside from Myrtus communis: modulation of expression of genes involved in cell defence system using cDNA microarray. Toxicol in Vitro 22:567–581. https://doi.org/10.1016/j.tiv.2007.11.015
Hennia A, Miguel MG, Nemmiche S (2018) Antioxidant activity of Myrtus communis L. and Myrtus nivellei batt. & Trab. Extracts: a brief review. Medicines 5:89. https://doi.org/10.3390/medicines5030089
Hillegass L, Griswold D, Brickson B, Albrightson-Winslow C (1990) Assessment of myeloperoxidase activity in whole rat kidney. J Pharm Methods 24:285–295. https://doi.org/10.1016/0160-5402(90)90013-B
Ingram G (1976) Reference method for the one stage prothrombin time test on human blood. Thromb Haemostas 36:237–238. https://doi.org/10.1055/s-0038-1648029
Ledwozyw A, Michalak J, Stepień A, Kadziołka A (1986) The relationship between plasma triglycerides, cholesterol, total lipids and lipid peroxidation products during human atherosclerosis. Clin Chim Acta; Int J Clin Chem 155:275–283. https://doi.org/10.1016/0009-8981(86)90247-0
Lerman LO, Nath KA, Rodriguez-Porcel M, Krier JD, Schwartz RS, Napoli C, Romero JC (2001) Increased oxidative stress in experimental renovascular hypertension. Hypertension 37:541–546. https://doi.org/10.1161/01.HYP.37.2.541
Lonn E, Gerstein H, Smieja M, Mann J, Yusuf S (2003) Mechanisms of cardiovascular risk reduction with ramipril:insights from HOPE and HOPE substudies. Eur Heart J Suppl 5:A43–A48. https://doi.org/10.1016/S1520-765X(03)90063-0
Mancia G, Sega R, Milesi C, Cessna G, Zanchetti A (1997) Blood-pressure control in the hypertensive population. Lancet 349:454–457. https://doi.org/10.1016/S0140-6736(96)07099-7
Martinez-Maldonado M (1991) Pathophysiology of renovascular hypertension. Hypertension 17:707–719. https://doi.org/10.1161/01.HYP.17.5.707
Miranda KM, Espey MG, Wink DA (2001) A rapid, simple spectrophotometric method for simultaneous detection of nitrate and nitrite. Nitric Oxide 5:62–71. https://doi.org/10.1006/niox.2000.0319
Mylroie AA, Collins H, Umbles C, Kyle J (1986) Erythrocyte superoxide dismutase activity and other parameters of copper status in rats ingesting lead acetate. Toxicol Appl Pharmacol 82:512–520. https://doi.org/10.1016/0041-008X(86)90286-3
Ozbeyli D, Sen A, Cilingir Kaya OT, Ertas B, Aydemir S, Ozkan N, Yuksel M, Sener G (2020) Myrtus communis leaf extract protects against cerulein-induced acute pancreatitis in rats. J Food Biochem 44:e13130. https://doi.org/10.1111/jfbc.13130
Park S, Arasu MV, Jiang N, Choi S-H, Lim YP, Park J-T, Al-Dhabi NA, Kim S-J (2014) Metabolite profiling of phenolics, anthocyanins and flavonols in cabbage (Brassica oleracea var. capitata). Ind Crop Prod 60:8–14. https://doi.org/10.1016/j.indcrop.2014.05.037
Rodrigo R, González J, Paoletto F (2011) The role of oxidative stress in the pathophysiology of hypertension. Hypertension Res 34:431–440
Romani A, Pinelli P, Mulinacci N, Vincieri F, Tattini M (1999) Identification and quantitation of polyphenols in leaves of Myrtus communis L. Chromatographia 49:17–20
Schulz E, Anter E, Keaney JF Jr (2004) Oxidative stress, antioxidants, and endothelial function. Curr Med Chem 11:1093–1104. https://doi.org/10.2174/0929867043365369
Şener G, Toklu H, Ercan F, Erkanlı G (2005) Protective effect of β-glucan against oxidative organ injury in a rat model of sepsis. Int Immunopharmacol 5:1387–1396. https://doi.org/10.1016/j.intimp.2005.03.007
Sisay M, Gashaw T (2017) Ethnobotanical, Ethnopharmacological, and phytochemical studies of Myrtus communis Linn: a popular herb in Unani system of medicine. J Evid Based Complement Altern Med 2:1035–1043. https://doi.org/10.1177/2156587217718958
Sumbul S, Ahmad MA, Asif M, Akhtar M (2011) Myrtus communis Linn.-a review. IJNPR 2(4):395–402
Taamalli A, Iswaldi I, Arráez-Román D, Segura-Carretero A, Fernández-Gutiérrez A, Zarrouk M (2014) UPLC–QTOF/MS for a rapid characterisation of phenolic compounds from leaves of Myrtus communis L. Phytochem Anal 25(1):89–96. https://doi.org/10.1002/pca.2475
Talha J, Priyanka M, Akanksha A (2011) Hypertension and herbal plants. Int Res J Pharm 2:26–30
Textor SC (2017) Renal arterial disease and hypertension. Med Clin North Am 101:65–79. https://doi.org/10.1016/j.mcna.2016.08.010
Toklu HZ, Sehirli O, Erşahin M, Süleymanoğlu S, Yiğiner O, Emekli-Alturfan E, Yarat A, Yeğen B, Sener G (2010) Resveratrol improves cardiovascular function and reduces oxidative organ damage in the renal, cardiovascular and cerebral tissues of two-kidney, one-clip hypertensive rats. J Pharm Pharmacol 62:1784–1793. https://doi.org/10.1111/j.2042-7158.2010.01197.x
Torras-Claveria L, Jauregui O, Bastida J, Codina C, Viladomat F (2007) Antioxidant activity and phenolic composition of lavandin (Lavandula x intermedia Emeric ex Loiseleur) waste. J Agric Food Chem 55(21):8436–8443. https://doi.org/10.1021/jf070236n
Wang L, Wu H, Yang F, Dong W (2019a) The protective effects of myricetin against cardiovascular disease. J Nutr Sci Vitaminol 65:470–476. https://doi.org/10.3177/jnsv.65.470
Wang Z, Wang S, Zhao J, Yu C, Hu Y, Tu Y, Yang Z, Zheng J, Wang Y, Gao Y (2019b) Naringenin ameliorates Renovascular hypertensive renal damage by normalizing the balance of renin-angiotensin system components in rats. Int J Med Sci 16:644–653. https://doi.org/10.7150/ijms.31075
Yakut ZAC, Ertas B, Sen A, Sener G (2020) Effect of Myrtus communis extract on serum cytokines in angiotensin dependent hypertensive rats. Medicine 9:404–407. https://doi.org/10.5455/medscience.2019.08.9204
Yousefian M, Shakour N, Hosseinzadeh H, Hayes AW, Hadizadeh F, Karimi G (2019) The natural phenolic compounds as modulators of NADPH oxidases in hypertension. Phytomedicine 55:200–213. https://doi.org/10.1016/j.phymed.2018.08.002
Živković J, Barreira JCM, Stojković D, Ćebović T, Santos-Buelga C, Maksimović Z, Ferreira ICFR (2014) Phenolic profile, antibacterial, antimutagenic and antitumour evaluation of Veronica urticifolia Jacq. J Funct Foods 9:192–201. https://doi.org/10.1016/j.jff.2014.04.024
Acknowledgments
The authors would like to thank Assoc.Prof. Gizem Emre for her help in identification of the plant material.
Funding
This study was funded by a grant from Marmara University Scientific Research Project Department (Project ID: SAG-C-YLP-170419-0142).
Author information
Authors and Affiliations
Contributions
Sule Arslan, Begüm Gürel Gökmen, Ozan Özcan: Biochemical analysis of heart, kidney and aorta tissues.
Zatiye Ayca Cevikelli Yakut, Goksel Sener: Conceptualization, experimental model and animal care.
Ali Şen, Fatih Göger: Myrtle leaf extract praparation and characterization.
Halil İbrahim Saygı, Gözde Erkanlı-Senturk: Histological Analysis of heart, kidney and aorta tissues.
Tuğba Tunali-Akbay: Conceptualization, methodology, investigation, manuscript writing and editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts interests that are relevant to the content of this article.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Arslan, S., Ozcan, O., Gurel-Gokmen, B. et al. Myrtle improves renovascular hypertension-induced oxidative damage in heart, kidney, and aortic tissue. Biologia 77, 1877–1888 (2022). https://doi.org/10.1007/s11756-022-01039-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11756-022-01039-1