Abstract
Drying treatments are an effective method for preserving original beneficial ingredients of postharvest plants. This work supplied the suitable drying conditions of three drying methods by investigating the changes of lotus leaves’ quality and antioxidant activity. The results indicated that: (1) Oven drying (55–65 °C) was the most appropriate way to keep the higher contents of alkaloids and amino acids, and the higher activities in DPPH radical scavenging and hydroxyl radical scavenging in lotus leaf treated with three drying methods; (2) Steam kill-enzyme torrefaction (3–5 min) was the most appropriate method for keeping the higher contents of flavone and the higher activities of superoxide radical scavenging in lotus leaf treated with three drying methods; (3) Microwave drying (680–850 W) was the most appropriate means to keep the higher contents of vitamin C, soluble sugar and protein in lotus leaf treated with three drying methods; (4) The color of lotus leaf treated with microwave treatments was more bright green than that treated with the other two methods; Therefore, the actual conditions of each drying means have to be taken into consideration when use the drying means.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Lotus (Nelumbo nucifera Gaertn.) is usually served as an economic ornamental, food, and pharmacy purposes, and generally grown in Eastern Asia, China, and India (Hu et al. 2012; Kato et al. 2015). The cultivation and medicinal use of lotus were discovered in China 2000 years ago (Zhao et al. 2013). Lotus leaves have been widely used as tonic medicine in Qin and Han dynasties. In November 1991, lotus leaves were included in the second batch of “Food and Drugs” list (Document No. 45 of the Health Supervision Bureau of the Ministry of Health of the People’s Republic of China).
Tea is brewed with a portion of the organs of medicinal plants, which has been popular in the world for thousands of years (Mukhtar and Ahmad 2000). Lotus leaves are rich in many active and nutritional ingredients, such as flavone, amino acids, vitamin C and so on (Huang and Zhang 2008). Additionally, many pharmacological and physiological activities have been confirmed in lotus leaf tea, such as anti-obesity, anti-oxidant, antiviral, anti-hypertensive, and so on (Ono et al. 2006; Liao and Lin 2013; Kato et al. 2015). The lotus leaf resources are abundant, and the price is low (Jin et al. 2016). Therefore, the lotus leaf tea has a broad development prospect. Currently, there are not many beverages related to lotus leaf on the market, and the processing methods reported and the product qualities obtained are different (Hao et al. 2012). Therefore, this article systematically studied the different processing techniques of lotus leaf tea and optimized the technical parameters of each process, which were able to enhance active and nutritional ingredients of lotus leaf tea.
Drying can be used to quickly protect the medicinal qualities of the medicinal plants in a simple manner, which is considered the most common and basic step needed for postharvest plants (Zhang et al. 2015; Yuan et al. 2015). It can be seen from former work, most researches on drying methods have been mainly conducted on medicinal plants, fruits, and vegetables. The aims of this study were to probe the changing rules of qualities and antioxidant activities of lotus leaf with three drying means and determine the most suitable drying method that simultaneously conserve the higher qualities and antioxidant activities in lotus leaves. Based on these, the results of this study will be helpful to develop lotus leaf tea as a healthy beverage.
Materials and methods
Equipment
Ultralow temperature refrigerated centrifuge (Thermo, Germany), Electronic balance (Sartorius, China), UV spectrophotometer (Unico, China), Electric blast drying oven (Teste, China), Microwave oven (Galanz, China), water-bath (Shengweili, China) and Ultrasonic cleaner (Kangjie, China).
Plant material and experimental design
Plant material
Lotus leaves were collected from Bai-yang Lake, Hebei province, China (38° 43′–39° 02′ N; 115° 38′-116° 07′ E), during September 2018. The lotus leaves were treated with three drying means.
Drying methods
(1) Oven drying was conducted in an electric blast drying oven at three different temperatures (55 °C, 65 °C, and 75 °C). Dry time was about 24 h; (2) Steam kill-enzyme torrefaction was carried out in a pot filled with boiling water in three different kill-enzyme times of 1 (Z1), 3 (Z2) and 5 min (Z3). The samples after kill-enzyme torrefaction were dried in electric blast drying oven at 65 °C. Drying time was about 24 h; and (3) Microwave drying was conducted in a microwave oven in three different power levels of 50% (450 W1), 80% (650 W2) and 100% (850 W3). Drying time was about 24 min, 6 min, and 3 min at W1, W2 and W3 treatments, separately. The eventual water contents were approximately 15 ± 0.73% dry basis in each of drying methods (Jafari et al. 2016).
After the above processes, the samples were ground into a powder and conserved in polyethylene bags.
Measurement methods
Flavone and alkaloids content
Flavone content was determined according to the method described by He and Liu (2007). Sample (0.2 g) was extracted with ethanol solution (20 mL, 50%) in an ultrasonic bath. The reaction mixture involved extract solution (5 mL), AlCl3 (8 mL, 1.5%), acetic acid-sodium acetate (4 mL). The absorption values were measured at 420 nm.
Alkaloids content was determined according to the method described by Wang et al. (2005). Sample (0.2 g) was extracted with trichloromethane solution (5 mL) and ammonium hydroxide (1 mL). The reaction mixture contained extract solution (2 mL), trichloromethane solution (8 mL), citric acid-sodium citrate (10 mL) and bromothymol blue (1 mL). The absorption values were detected at 416 nm.
Soluble sugar, free amino acids, vitamin C and protein content
Soluble sugar was measured according to the method described by Machado et al. (2013). Sample (0.2 g) was extracted with distilled water (5 mL). The reaction mixture included supernatant (1 mL), distilled water (1 mL), and anthrone-H2SO4 (5 mL). The absorption values were detected at 625 nm.
Free amino acids content was measured with minor modification according to the method described by Li et al. (2004). Sample (0.2 g) was extracted with acetic acid solution (8 mL, 10%). The reaction mixture was mixed with supernatant (2 mL), and ninhydrin (3 mL), ascorbic acid (0.1 mL). The absorption values were detected at 570 nm.
Vitamin C was determined according to State Standard of the People’s Republic of China (1990) (GB 12392-90). Sample (0.2 g) was extracted with oxalic acid solution (10 mL, 1%) and filtered with active carbon (0.2 g). The reaction mixture was mixed with supernatant (5 mL), thiourea solution (5 mL, 2%), 2, 4-dinitrophenylhydrazine solution (1 mL, 2%) and were cultivated in the water bath (37 °C) for 3 h. The absorption values were detected at 500 nm.
Protein content was measured according to the Coomassie brilliant blue staining created by Bradford (1976). Sample (0.2 g) was extracted with 5 mL phosphate buffer (50 mmol L−1) involving EDTA (1 mmol L−1), 2% (W/V) polyvinyl pyrrolidone. The reaction mixture involved extract solution (0.1 mL) and Coomassie brilliant blue (5 mL). The absorption values were detected at 595 nm.
Determination of antioxidant capacity
The DPPH free radical scavenging activity was measured according to the previously described method (Mohsen and Ammar 2009). Sample extract solution (2 mL) was mixed with DPPH solution (2 mL, 0.04 mg mL−1, prepared in 100% ethanol) for 0.5 h. The absorption values were detected at 517 nm, and the calculation formula as follows:
Scavenging activity (%) = (A0 − A1 + A2)/A0 × 100%
A0 is the absorption value of the control (the ethanol replaced the sample solution); A1 is the absorption value of the sample; A2 is the absorption value of the sample under similar conditions as A1 with the ethanol replaced the DPPH solution.
The superoxide radical scavenging activity was assayed using the modified pyrogallol autoxidation method (Chen et al. 2012). Sample extract solution (1 mL) was mixed with Tris–HCl (4 mL, 0.05 mol L−1, pH 8.2) and pyrogallol (1 mL, 0.2 mmol L−1) in a water bath (25 °C) for 5 min. The reaction was stopped immediately with two drops of concentrated HCl. The absorption values were detected at 325 nm, and the calculation formula was as follows:
Scavenging activity (%) = (A0 − A1)/A0 × 100%
A0 is the absorption value of the control; A1 is the absorption value of the sample.
The hydroxyl radical scavenging activity was measured according to the improved phenanthroline method (You et al. 2011). The mixtures were mixed with 1, 10-phenanthroline (1 mL, 0.75 mmol), phosphate buffer (2 mL, 0.2 mol L−1, pH 7.4), sample extract solution (1 mL), FeSO4 (1 mL, 0.75 mM) and H2O2 (1 mL, 0.01%) in a water bath (37 °C) for 1 h. The absorption values were detected at 536 nm, and the calculation formula was as follows:
Scavenging activity (%) = (A0 − A1)/(A2 − A1) × 100%
A0 is the absorption value of the sample; A1 is the absorption value of the control (the water replaced the sample); A2 is the absorption value of the solvent.
Statistical analysis
Data were the mean ± standard error. Differences among treatments were analyzed using one-way analysis of variance with Duncan’s test (p < 005). All analyses were performed by SPSS (version 13.0).
Results and discussion
The effects of drying methods on flavone and alkaloids in lotus leaf
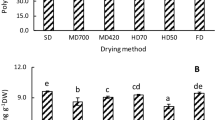
Flavone has various biological and medicinal properties, for example, anti-inflammatory, anti-cancer (Manthey et al. 2001) and anti-obesity agent (Kim et al. 2010). The experimental study showed that flavone content in lotus leaf was obviously raised as the enhance of stem kill-enzyme time (Fig. 1). The results were similar to Shi et al. (2017). In oven drying, flavone content rose first and then reduced as the enhance of the oven temperature. In the three drying methods, the lotus leaf processed with 5 min steam kill-enzyme exhibited the highest flavonoid content (2.537 mg g−1 DW), followed by the lotus leaf treated with the higher microwave power (2.337 mg g−1 DW) and lotus leaf dried by oven (65 °C) (1.836 mg g−1 DW).
The flavone content with three drying methods in lotus leaves. Oven drying (H1, 55 °C; H2, 65 °C; H3, 75 °C), steam kill-enzyme torrefaction (Z1, 1 min; Z2, 3 min; Z3, 5 min) and microwave drying (W1, 450 W; W2, 650 W; W3, 850 W). The bars with different letters are obviously different from each other at the same drying method (p < 0.05). Values are means of four replicates ± SE
Many studies have shown that the lotus leaf has a significant lipid-regulating effect, and its active site is mainly alkaloids (Du et al. 2000). In the current research, alkaloids content significantly increased as the enhance of microwave power (Fig. 2). Similar results have appeared in previous research (Zhang et al. 2011). In oven drying, alkaloids content in lotus leaf dropped first and rose subsequently as the enhance of oven temperature, and achieved a maximum content when oven temperature was 75 °C. However, the alkaloids content in lotus leaf significantly reduced with the enhance of the steam kill-enzyme time. Oven drying was beneficial to preserving the content of alkaloids in lotus leaves among the three drying methods.
The alkaloids content with three drying methods in lotus leaves. Oven drying (H1, 55 °C; H2, 65 °C; H3, 75 °C), steam kill-enzyme torrefaction (Z1, 1 min; Z2, 3 min; Z3, 5 min) and microwave drying (W1, 450 W; W2, 650 W; W3, 850 W). The bars with different letters are obviously different from each other at the same drying method (p < 0.05). Values are means of four replicates ± SE
The effects of drying methods on the contents of soluble sugar, amino acids, vitamin C and protein in lotus leaf
In recent years, bio-polysaccharides have been proven to be anti-tumor, anti-viral, anti-aging, enhancing immunity, and hematopoietic properties (Khaskheli et al. 2015; Hammi et al. 2016). At the same time, water-soluble sugar cannot only taste sweet alcohol in the tea field but also can produce some aroma substances and colored substances through Maillard reaction and caramelization in the process, which affected the tea quality. Dehnad et al. (2016) certified that the drying method has a significant effect on the polysaccharide content. Figure 3 indicated that the soluble sugar content in lotus leaf was affected by the actual conditions of each drying method. In the three drying means, higher microwave power (650–850 W) was beneficial for preserving soluble sugar content (1.550 and 1.630 mg g−1 DW) in lotus leaves, followed by lotus leaf treated by steam kill-enzyme (1.450 mg g−1 DW) and lotus leaf dried by oven (65–75 °C) (1.253 and 1.258 mg g−1 DW).
The soluble sugar content with three drying methods in lotus leaves. Oven drying (H1, 55 °C; H2, 65 °C; H3, 75 °C), steam kill-enzyme torrefaction (Z1, 1 min; Z2, 3 min; Z3, 5 min) and microwave drying (W1, 450 W; W2, 650 W; W3, 850 W). The bars with different letters are obviously different from each other at the same drying method (p < 0.05). Values are means of four replicates ± SE
Free amino acids are a crucial biologically active compound that has been shown specific functions in maintaining metabolism, growth, reproduction, and immune function (Cylwik et al. 2005; Morris et al. 2005; Mateo et al. 2007). Both the drying methods and the actual drying conditions will affect the content of free amino acids in the lotus leaf (Fig. 4). The present study showed amino acids in lotus leaf was obviously dropped while the enhance of the oven temperature (55–75 °C) and achieved a maximum content (1019 μg g−1 DW) while oven temperature was 55 °C. However, amino acids in lotus leaf increased firstly and then declined while the enhance of steam kill-enzyme time and microwave power, and achieved the maximum content (596 and 144 μg g−1 DW) as the steam kill-enzyme torrefaction and microwave drying was 3 min and 650 W, separately. The research showed that the decomposition of amino acids might be caused by the higher microwave power, the higher oven temperature, and the longer kill-enzyme time. In the three drying methods, oven drying was beneficial to preserving the amino acid content in lotus leaves. Therefore, the actual conditions of each processing methods should be considered.
The amino acids content with three drying methods in lotus leaves. Oven drying (H1, 55 °C; H2, 65 °C; H3, 75 °C), steam kill-enzyme torrefaction (Z1, 1 min; Z2, 3 min; Z3, 5 min) and microwave drying (W1, 450 W; W2, 650 W; W3, 850 W). The bars with different letters are obviously different from each other at the same drying method (p < 0.05). Values are means of four replicates ± SE
In humans, there are many important biosynthetic processes need vitamin C to take part, for example, to reduce the oxidative stress of ascorbate peroxidase substrates. Yang and He (2009) put forward that microwave-dried red dates had the highest vitamin C content, which was higher than that of natural drying and electrothermic drying. Lv et al. (2011) also reported that microwave drying could not only effectively shorten the drying time and increase the drying rate, but also could greatly improve the preservation rate of vitamin C in dry hawthorn slices compared with the traditional natural drying and hot air drying. In the current research, vitamin C in lotus leaf increased first and then reduced while the enhance of microwave power, and achieved the maximum content (22.344 mg g−1 DW) when the microwave power was 650 W (Fig. 5). Vitamin C content in lotus leaf significantly rose while the enhance of the steam kill-enzyme time. But, vitamin C content dropped first and rose subsequently with the increased of the oven temperature. Microwave drying was beneficial to preserving the content of vitamin C in lotus leaves among the three drying methods.
The vitamin C content with three drying methods in lotus leaves. Oven drying (H1, 55 °C; H2, 65 °C; H3, 75 °C), steam kill-enzyme torrefaction (Z1, 1 min; Z2, 3 min; Z3, 5 min) and microwave drying (W1, 450 W; W2, 650 W; W3, 850 W). The bars with different letters are obviously different from each other at the same drying method (p < 0.05). Values are means of four replicates ± SE
Protein is the material basis of life and the main undertaker of life activity. Wang et al. (2008) reported that the protein content of alfalfa hay could be reserved better with the high-temperature drying Wang et al. (2010). also reported that microwave-treated lotus leaves are more conducive to preserve protein content than traditional drying method. The study further showed that protein content in lotus leaf was also affected by microwave power. In the experimental condition, lotus leaf treated by 850 W microwave had the highest protein content (24.461 mg g−1DW), followed by lotus leaf treated by the oven (55 °C) (22.645 mg g−1 DW) and lotus leaf processed with 3 min steam kill-enzyme (21.720 mg g−1 DW) (Fig. 6).
The protein content with three drying methods in lotus leaves. Oven drying (H1, 55 °C; H2, 65 °C; H3, 75 °C), steam kill-enzyme torrefaction (Z1, 1 min; Z2, 3 min; Z3, 5 min) and microwave drying (W1, 450 W; W2, 650 W; W3, 850 W). The bars with different letters are obviously different from each other at the same drying method (p < 0.05). Values are means of four replicates ± SE
The effects of drying methods on the antioxidant activity in lotus leaf
Scientific researches have shown that cancer, aging, or other diseases were mostly related to the excess production of free radicals. So, antioxidant activity is listed as one of the main research directions by food, health care products, and cosmetics companies. DPPH was mainly used in the detection of vitro antioxidant activity of antioxidant substances (Kurd and Samavatib 2015). Superoxide radical could disrupt human functions (Seedevi et al. 2017). Hydroxyl radical was one of the reactive oxygen species that could kill red blood cells, degrade DNA, and cell membranes (Bagchi and Kumar 2016). Zhang et al. (2017) reported that the lotus plumule could be served as natural antioxidants in functional foods because it was an extremely high antioxidant activity. Jin et al. (2016) also reported that lotus leaf tea has been popular in China, which is a wholesome drink, especially for antioxidant function.
The DPPH radical scavenging activity in lotus leaf dropped first and rose subsequently with the increase of the oven temperature and microwave power, and reached a peak value at H1 (55 °C) and W1 (450 W), respectively. However, the DPPH radical scavenging activity was obviously declined as the enhance of steam kill-enzyme time (Fig. 7a). Among the three drying methods, the lotus leaf processed with oven drying (55 ℃) showed the higher DPPH radical scavenging activity, followed by the lotus leaf processed with the lower microwave power (450 W), and the lotus leaf processed with 1 min steam kill-enzyme.
The radical scavenging activity with three drying methods in lotus leaves (a DPPH radical scavenging activity; b superoxide radical scavenging activity; c hydroxyl radical scavenging activity). Oven drying (H1, 55 °C; H2, 65 °C; H3, 75 °C), steam kill-enzyme torrefaction (Z1, 1 min; Z2, 3 min; Z3, 5 min) and microwave drying (W1, 450 W; W2, 650 W; W3, 850 W). The bars with different letters are obviously different from each other at the same drying method (p < 0.05). Values are means of four replicates ± SE
In Fig. 7b demonstrated that the superoxide radical scavenging activity in lotus leaf rose first and then declined as the enhance of the steam kill-enzyme time and microwave power. The superoxide radical scavenging activity in lotus leaf treated by the oven temperature (55 ℃) reached the peak value. In the three drying methods, lotus leaf dried by the 3 min steam kill-enzyme exhibited the higher superoxide radical scavenging activity, while the lotus leaf treated by oven drying exhibited the lowest superoxide radical scavenging activity.
Figure. 7c indicated that the hydroxyl radical scavenging activity in lotus leaf significantly increased as the enhance of the steam kill-enzyme time and microwave power. However, the hydroxyl radical scavenging activity in lotus leaf rose first and then declined as the enhance of the oven temperature. Among the three drying methods, the lotus leaf processed with oven temperature (65 ℃) and 5 min steam kill-enzyme drying both had the higher hydroxyl radical scavenging activity than that treated by the microwave drying.
The comprehensive analysis showed that the DPPH radical and the superoxide radical scavenging activity were the higher in lotus leaf treated with oven drying and steam kill-enzyme torrefaction, respectively. In addition, the lotus leaves treated by oven drying and steam kill-enzyme torrefaction both had the higher hydroxyl radical scavenging activity.
The color of lotus leaves in drying methods.
Color parameters, which was one of the important sensory properties of tea (Wu and Sun 2013). Jin et al. (2016) reported that there were many diverse colors in herbal teas. As is shown in Plate 1, the color of lotus leaf treated with microwave treatments were more bright green than that treated with the other two methods. The reason might be that microwave treatments could be better to keep the higher chlorophyll contents in the lotus leaf. Next, experiments will be performed to prove the hypothesis and find the real reason.
Conclusion
Both the drying methods and the specific conditions of each processing methods will significantly affect the qualities and antioxidant activities of lotus leaves. The experimental study showed that: (1) Oven drying (55–65 ℃) was beneficial to keeping the higher contents of alkaloids and amino acids, and the higher activities in DPPH radical scavenging and hydroxyl radical scavenging; (2) Steam kill-enzyme torrefaction (3–5 min) could gain the higher contents of flavone and the higher activities of superoxide radical scavenging; (3) The appropriate microwave power and microwave time were about 680–850 W and 3–6 min, separately, which could gain the higher contents of vitamin C, soluble sugar and protein. (4) The color of lotus leaf treated with microwave treatments was more bright green than that treated with the other two methods. Therefore, the actual conditions of each processing methods should be considered when the drying method was selected. These results provided a theoretical basis for further research and improved the qualities of lotus leaf tea.
Author contribution statement
Contribution of the authors of the paper was as follows: (1) Idea of the project: XY, JC, CL. (2) Experimentation: CG (3). Data interpretation: CG, NZ, JX. (4) Manuscript writing: CG.
References
Bagchi S, Kumar KJ (2016) Studies on water-soluble polysaccharides from Pithecellobium dulce (Roxb.) Benth. Seeds Carbohyd Polym 138:215–221. https://doi.org/10.1016/j.carbpol.2015.11.018
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72(1–2):248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Chen R, Li Y, Dong H, Liu Z, Li S, Yang S, Li X (2012) Optimization of ultrasonic extraction process of polysaccharides from Ornithogalum caudatum ait and evaluation of its biological activities. Ultrason Sonochem 19(6):1160–1168. https://doi.org/10.1016/j.ultsonch.2012.03.008
Cylwik D, Mogielnicki A, Buczko W (2005) l-Arginine and cardiovascular system. Pharmacol Rep 57:14–22
Dehnad D, Jafari SM, Afrasiabi M (2016) Influence of drying on functional properties of food biopolymers: from traditional to novel dehydration techniques. Trends Food Sci Tech 57:116–131. https://doi.org/10.1016/j.tifs.2016.09.002
Du LJ, Sun H, Li M, Jin W, Xu LZ (2000) Studies on the active components in the leaf of Nelumbo nucifera and Black Soybean and their mixture with blood lipid-regulating effect. Chin Tradit Herbal Drugs 31(7):526–527
Hammi KM, Hammamib M, Rihouey C, Cerf DL, Ksouri R, Majdoub H (2016) Optimization extraction of polysaccharide from Tunisian Zizyphus lotus fruit by response surface methodology: composition and antioxidant activity. Food Chem 212:476–484. https://doi.org/10.1016/j.foodchem.2016.06.004
Hao LL, Wang ZP, Xu Y, Li HC, Ma SF (2012) Study on the preparation of lotus leaf tea beverage. J Anhui Agri Sci 40:13613–13615. https://doi.org/10.13989/j.cnki.0517-6611.2012.27.032
He SM, Liu JL (2007) Study on the determination method of flavone content in tea. Chin J Anal Chem 35:1365–1368
Hu JH, Pan L, Liu HG, Wang SZ, Wu ZH, Ke WD, Ding Y (2012) Comparative analysis of genetic diversity in sacred lotus (Nelumbo nucifera Gaertn) using AFLP and SSR markers. Mol Biol Rep 39(4):3637–3647. https://doi.org/10.1007/s11033-011-1138-y
Huang KY, Zhang ZG (2008) The researchsituation of Lotus leaves. Chin Pharm 17:77–78
Jafari SM, Azizi D, Mirzaei H, Dehnad D (2016) Comparing quality characteristics of oven-dried and refractance window-dried kiwifruits. J Food Process Pres 40(3):362–372. https://doi.org/10.1111/jfpp.12613
Jin L, Li XB, Tian DQ, Fang XP, Yu YM, Zhu HQ, Ge YY, Ma GY, Wang WY, Xiao WF, Li M (2016) Antioxidant properties and color parameters of herbal teas in China. Ind Crops Prod 87:198–209. https://doi.org/10.1016/j.indcrop.2016.04.044
Kato E, Inagaki Y, Kawabata J (2015) Higenamine 4′-O-β-d-glucoside in the lotus plumule induces glucose uptake of L6 cells through theβ2-adrenergic receptor. Bioorgan Med Chem 23(13):3317–3321. https://doi.org/10.1016/j.bmc.2015.04.054
Khaskheli SG, Zheng W, Sheikh SA, Khaskheli AA, Liu Y, Soomro AH, Feng X, Sauer MB, Wang YF, Huang W (2015) Characterization of Auricularia auricular polysaccharides and its antioxidant properties in the fresh and pickled product. Int J Biol Macromol 81:387–395. https://doi.org/10.1016/j.ijbiomac.2015.08.020
Kim J, Lee I, Seo J, Jung M, Kim Y, Yim N, Bae K (2010) Vitexin orientin and other flavonoids from Spirodela Polyrhiza inhibit adipogenesis in 3T3-L1 cells. Phytother Res 24(10):1543–1548. https://doi.org/10.1002/ptr.3186
Kurd F, Samavati V (2015) Water-soluble polysaccharides from Spirulina platensis: extraction and in vitro anti-cancer activity. Int J Biol Macromol 74:498–506. https://doi.org/10.1016/j.ijbiomac.2015.01.005
Li HS, Sun Q, Zhao SJ, Zhang WH (2004) Plant physiological and biochemical principle and experimental technique. Higher Education Press, Beijing
Liao CH, Lin JY (2013) Lotus (Nelumbo nucifera Gaertn.) plumule polysaccharide ameliorates pancreatic islets loss and serum lipid profiles in non-obese diabetic mice. Food Chem Toxicol 58:416–422. https://doi.org/10.1016/j.fct.2013.05.018
Lv YZ, Liang ZH, Liu G, Shi MJ (2011) Effects of different drying methods on the stability of vitamin C in the dry process of Cornus Officinalis. Process Agric Prod 6:70–71
Machado RAR, Ferrieri AP, Robert CAM, Glauser G, Kallenbach M, Baldwin IT, Erb M (2013) Leaf-herbivore attack reduces carbon reserves and regrowth from the roots via jasmonate and auxin signaling. New Phytol 200(4):1234–1246. https://doi.org/10.1111/nph.12438
Manthey JA, Guthrie N, Grohmann K (2001) Biological properties of citrus flavonoids pertaining to cancer and inflammation. Curr Med Chem 8(2):135–153. https://doi.org/10.2174/0929867013373723
Mateo RD, Wu G, Bazer FW, Park JC, Shinzato I, Kim SW (2007) Dietary l-arginine supplementation enhances the reproductive performance of gilts. J Nutr 137(3):652–656. https://doi.org/10.1093/jn/137.3.652
Mohsen SM, Ammar ASM (2009) Total phenolic contents and antioxidant activity of corn tassel extracts. Food Chem 112(3):595–598. https://doi.org/10.1016/j.foodchem.2008.06.014
Morris CR, Kato GJ, Poljakovic M, Wang X, Blackwelder WC, Sachdev V, Hazen SL, Vichinsky EP, Morris JSM, Gladwin MT (2005) Dysregulated arginine metabolism, hemolysis-associated pulmonary hypertension and mortality in sickle cell disease. J Am Med Assoc 294(1):81–90. https://doi.org/10.1001/jama.294.1.81
Mukhtar H, Ahmad N (2000) Tea polyphenols: prevention of cancer and optimizing health. Am J Clin Nutr 71(6):1698–1702. https://doi.org/10.1093/ajcn/71.6.1698s
Ono Y, Hattori E, Fukaya Y, Imai S, Ohizumi Y (2006) Anti-obesity effect of Nelumbo nucifera leaves extract in mice and rats. J Ethnopharmacol 106(2):238–244. https://doi.org/10.1016/j.jep.2005.12.036
Seedevi P, Moovendhan M, Viramani S, Shanmugam A (2017) Bioactive potential and structural characterization of sulfated polysaccharide from seaweed (Gracilaria cortical). Carbohyd Polym 155:516–524. https://doi.org/10.1016/j.carbpol.2016.09.011
Shi XF, Chu JJ, Zhang YF, Liu CQ, Yao XQ (2017) Nutritional and active ingredients of medicinal chrysanthemum flower heads affected by different drying methods. Ind Crops Prod 104:45–51. https://doi.org/10.1016/j.indcrop.2017.04.021
State Standard of the People’s Republic of China (1990) Method for determination of total ascorbic acid in fruits, vegetables and derived products (GB 12392–90)
Wang L, Zhao YJ, Zhang YY, Wu JF, Zhang XN, Ji ZQ, Yue W (2005) Determination of alkaloids in pinellia tuber and its antiemetic study. Chin Pharmacol bull 21:864–867
Wang XL, Yan XD, Xu YP, Zhao ZX (2008) Preliminary study on the factors affecting the protein contents of alfalfa hay. Pratacult Sci 3:47–50
Wang DL, Xu WX, Li C, Huang Y (2010) Microwave drying of tea beverage quality. In: Transformation of economic development mode and independent innovation-The 12th China Science and Technology Association Annual Meeting (Volume II)
Wu D, Sun DW (2013) Colour measurements by computer vision for food qualitycontrol—a review. Trends Food Sci Tech 29:5–20
Yang YJ, He HS (2009) Determination of vitamin C content in red dates of different drying methods. Mod Prevent Med 36:42–43
You L, Zhao M, Regenstein JM, Ren J (2011) In vitro antioxidant activity and in vivo anti-fatigue effect of loach (Misgurnus anguillicaudatus) peptides prepared by papain digestion. Food Chem 124(1):188–194. https://doi.org/10.1016/j.foodchem.2010.06.007
Yuan J, Hao LJ, Wu G, Wang S, Duan JA, Xie GY, Qin MJ (2015) Effects of drying methods on the phytochemicals contents and antioxidant properties of chrysanthemum flower heads harvested at two developmental stages. J Funct Foods 19:786–795. https://doi.org/10.1016/j.jff.2015.10.008
Zhang L, Wu HY, Deng DW, Huang GH (2011) Study on new technology of lotus leaf tea by microwave-assisted drying. Sci Tech Food Indus 32(04):295–298. https://doi.org/10.13386/j.issn1002-0306.2011.04.070
Zhang L, Liu TF, Xue YJ, Liu C, Ru H, Dong MH, Yu ZF (2015) Effects of drying methods on the aroma components and quality of Capsella Bursa-Pastoris L. J Food Process Eng 39(2):107–120. https://doi.org/10.1111/jfpe.12204
Zhang LJ, Cheng Z, Zhao Q, Wang MS (2017) Green and efficient PEG-based ultrasound-assisted extraction of polysaccharides from superfine ground lotus plumule to investigate their antioxidant activities. Ind Crops Prod 109:320–326. https://doi.org/10.1016/j.indcrop.2017.08.018
Zhao J, Deng JW, Chen YW, Li SP (2013) Advanced phytochemical analysis of herbal tea in China. J Chromatogr A 1313:2–23. https://doi.org/10.1016/j.chroma.2013.07.039
Funding
This work has been supported by the National Natural Science Foundation of China (No. 31300321), Natural Science Foundation of Hebei Province (No. C2012201080), and Open Foundation of Hebei Key Laboratory of Wetland Ecology and Conservation (No. hklk201904).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
Chun-Yan Guo declares that she has no conflict of interest. Nan Zhang declares that she has no conflict of interest. Cun-Qi Liu declares that he has no conflict of interest. Jing-Jing Xue declares that she has no conflict of interest. Jian-Zhou Chu declares that he has no conflict of interest. Xiao-Qin Yao declares that she has no conflict of interest.
Ethical approval
This article does not contain any studies involving human participants or animals performed by any of the authors.
Informed consent
Not applicable as this study does not include any human participants.
Additional information
Communicated by P. Wojtaszek.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Guo, C., Zhang, N., Liu, C. et al. Qualities and antioxidant activities of lotus leaf affected by different drying methods. Acta Physiol Plant 42, 14 (2020). https://doi.org/10.1007/s11738-019-2992-9
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11738-019-2992-9