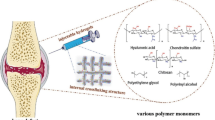
Abstract
Minimally invasive injectable self-setting materials are useful for bone repairs and for bone tissue regeneration in situ. Due to the potential advantages of these materials, such as causing minimal tissue injury, nearly no influence on blood supply, easy operation and negligible postoperative pain, they have shown great promises and successes in clinical applications. It has been proposed that an ideal injectable bone repair material should have features similar to that of natural bones, in terms of both the microstructure and the composition, so that it not only provides adequate stimulus to facilitate cell adhesion, proliferation and differentiation but also offers a satisfactory biological environment for new bone to grow at the implantation site. This article reviews the properties and applications of injectable bone repair materials, including those that are based on natural and synthetic polymers, calcium phosphate, calcium phosphate/polymer composites and calcium sulfate, to orthopedics and bone tissue repairs, as well as the progress made in biomimetic fabrication of injectable bone repair materials.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Hench L L, Polak J M. Third-generation biomedical materials. Science, 2002, 295(5557): 1014–1017
Dreifke M B, Ebraheim N A, Jayasuriya A C. Investigation of potential injectable polymeric biomaterials for bone regeneration. Journal of Biomedical Materials Research Part A, 2013, 101 (8): 2436–2447
He Y, Gao J, Li X, et al. Fabrication of injectable calcium sulfate bone graft material. Journal of Biomaterials Science: Polymer Edition, 2010, 21(10): 1313–1330
Low K L, Tan S H, Zein S H S, et al. Calcium phosphate-based composites as injectable bone substitute materials. Journal of Biomedical Materials Research Part B: Applied Biomaterials, 2010, 94(1): 273–286
Hile D D, Kowaleski M P, Doherty S A, et al. An injectable porous poly(propylene glycol-co-fumaric acid) bone repair material as an adjunct for intramedullary fixation. Bio-Medical Materials and Engineering, 2005, 15(3): 219–227
Yang X, Gan Y, Gao X, et al. Preparation and characterization of trace elements-multidoped injectable biomimetic materials for minimally invasive treatment of osteoporotic bone trauma. Journal of Biomedical Materials Research Part A, 2010, 95(4): 1170–1181
Zhu X S, Zhang Z M, Mao H Q, et al. A novel sheep vertebral bone defect model for injectable bioactive vertebral augmentation materials. Journal of Materials Science: Materials in Medicine, 2011, 22(1): 159–164
Cui F Z, Li Y, Ge J. Self-assembly of mineralized collagen composites. Materials Science and Engineering R: Reports, 2007, 57(1–6): 1–27
Wang X M, Cui F Z, Ge J, et al. Hierarchical structural comparisons of bones from wild-type and liliput(dtc232) genemutated Zebrafish. Journal of Structural Biology, 2004, 145(3): 236–245
Weiner S, Wagner H D. The material bone: Structure mechanical function relations. Annual Review of Materials Science, 1998, 28 (1): 271–298
Cui F Z, Wen H B, Su X W, et al. Microstructures of external periosteal callus of repaired femoral fracture in children. Journal of Structural Biology, 1996, 117(3): 204–208
Landis WJ, Song M J, Leith A, et al. Mineral and organic matrix interaction in normally calcifying tendon visualized in three dimensions by high-voltage electron microscopic tomography and graphic image reconstruction. Journal of Structural Biology, 1993, 110(1): 39–54
Weiner S, Traub W. Organization of hydroxyapatite crystals within collagen fibrils. FEBS Letters, 1986, 206(2): 262–266
Sharifi S, Imani M, Mirzadeh H, et al. Synthesis, characterization, and biocompatibility of novel injectable, biodegradable, and in situ crosslinkable polycarbonate-based macromers. Journal of Biomedical Materials Research Part A, 2009, 90(3): 830–843
Cruz D M, Ivirico J L, Gomes M M, et al. Chitosan microparticles as injectable scaffolds for tissue engineering. Journal of Tissue Engineering and Regenerative Medicine, 2008, 2(6): 378–380
Ma G, Yang D, Li Q, et al. Injectable hydrogels based on chitosan derivative/polyethylene glycol dimethacrylate/N,N-dimethylacrylamide as bone tissue engineering matrix. Carbohydrate Polymers, 2010, 79(3): 620–627
Abbah S A, Lu W W, Chan D, et al. In vitro evaluation of alginate encapsulated adipose-tissue stromal cells for use as injectable bone graft substitute. Biochemical and Biophysical Research Communications, 2006, 347(1): 185–191
Lee J Y, Choo J E, Park H J, et al. Injectable gel with synthetic collagen-binding peptide for enhanced osteogenesis in vitro and in vivo. Biochemical and Biophysical Research Communications, 2007, 357(1): 68–74
Bergman K, Engstrand T, Hilborn J, et al. Injectable cell-free template for bone-tissue formation. Journal of Biomedical Materials Research Part A, 2009, 91(4): 1111–1118
Boger A, Bohner M, Heini P, et al. Properties of an injectable low modulus PMMA bone cement for osteoporotic bone. Journal of Biomedical Materials Research Part B: Applied Biomaterials, 2008, 86(2): 474–482
Lewandrowski K U, Gresser J D, Wise D L, et al. Osteoconductivity of an injectable and bioresorbable poly(propylene glycol-co-fumaric acid) bone cement. Biomaterials, 2000, 21(3): 293–298
Kim C W, Talac R, Lu L, et al. Characterization of porous injectable poly-(propylene fumarate)-based bone graft substitute. Journal of Biomedical Materials Research Part A, 2008, 85(4): 1114–1119
Young A M, Ho S M. Drug release from injectable biodegradable polymeric adhesives for bone repair. Journal of Controlled Release, 2008, 127(2): 162–172
Vertenten G, Vlaminck L, Gorski T, et al. Evaluation of an injectable, photopolymerizable three-dimensional scaffold based on D,L-lactide and e-caprolactone in a tibial goat model. Journal of Materials Science: Materials in Medicine, 2008, 19(7): 2761–2769
Page J M, Harmata A J, Guelcher S A. Design and development of reactive injectable and settable polymeric biomaterials. Journal of Biomedical Materials Research Part A, 2013, 101 (12): 3630–3645
Shin H, Quinten Ruhé P, Mikos A G, et al. In vivo bone and soft tissue response to injectable, biodegradable oligo(poly(ethylene glycol) fumarate) hydrogels. Biomaterials, 2003, 24(19): 3201–3211
Guo X, Park H, Liu G, et al. In vitro generation of an osteochondral construct using injectable hydrogel composites encapsulating rabbit marrow mesenchymal stem cells. Biomaterials, 2009, 30(14): 2741–2752
Kim S Y, Lee S C. Thermo-responsive injectable hydrogel system based on poly(N-isopropylacrylamide-co-vinylphosphonic acid). I. Biomineralization and protein delivery. Journal of Applied Polymer Science, 2009, 113(6): 3460–3469
Lee K Y, Alsberg E, Mooney D J. Degradable and injectable poly (aldehyde guluronate) hydrogels for bone tissue engineering. Journal of Biomedical Materials Research, 2001, 56(2): 228–233
Rimondini L, Nicoli-Aldini N, Fini M, et al. In vivo experimental study on bone regeneration in critical bone defects using an injectable biodegradable PLA/PGA copolymer. Oral Surgery, Oral Medicine, Oral Pathology, Oral Radiology, and Endodontics, 2005, 99(2): 148–154
Chen F, Mao T, Tao K, et al. Injectable bone. The British Journal of Oral & Maxillofacial Surgery, 2003, 41(4): 240–243
Burdick J A, Anseth K S. Photoencapsulation of osteoblasts in injectable RGD-modified PEG hydrogels for bone tissue engineering. Biomaterials, 2002, 23(22): 4315–4323
Amouriq Y, Bourges X, Weiss P, et al. Skin sensitization study of two hydroxypropyl methylcellulose components (Benecel and E4M) of an injectable bone substitute in guinea pigs. Journal of Materials Science: Materials in Medicine, 2002, 13(2): 149–154
Lewis G, Koole L H, van Hooy-Corstjens C S J. Influence of powder-to-liquid monomer ratio on properties of an injectable iodine-containing acrylic bone cement for vertebroplasty and balloon kyphoplasty. Journal of Biomedical Materials Research Part B: Applied Biomaterials, 2009, 91(2): 537–544
Hernández L, Parra J, Vázquez B, et al. Injectable acrylic bone cements for vertebroplasty based on a radiopaque hydroxyapatite. Bioactivity and biocompatibility. Journal of Biomedical Materials Research Part B: Applied Biomaterials, 2009, 88(1): 103–114
Carrodeguas R G, Lasa B V, Del Barrio J S R. Injectable acrylic bone cements for vertebroplasty with improved properties. Journal of Biomedical Materials Research Part B: Applied Biomaterials, 2004, 68(1): 94–104
Hernandez L, Muñoz M E, Goñi I, et al. New injectable and radiopaque antibiotic loaded acrylic bone cements. Journal of Biomedical Materials Research Part B: Applied Biomaterials, 2008, 87(2): 312–320
Webb J C J, Spencer R F. The role of polymethylmethacrylate bone cement in modern orthopaedic surgery. Journal of Bone and Joint Surgery (British Volume), 2007, 89(7): 851–857
Robinson Y, Tschöke S, Stahel P F, et al. Complications and safety aspects of kyphoplasty for osteoporotic vertebral fractures: a prospective follow-up study in 102 consecutive patients. Patient Safety in Surgery, 2008, 2(1): 2 (10 pages)
Kalteis T, Lüring C, Gugler G, et al. Acute tissue toxicity of PMMA bone cements. Zeitschrift für Orthopädie und ihre Grenzgebiete, 2004, 142(6): 666–672
Brown W E, Chow L C. A new calcium phosphate setting cement. Journal of Dental Research, 1983, 62(1): 672–679
Gruninger S E S C, Chow L C, O'young A, et al. Evaluation of the biocompatibility of a new calcium phosphate setting cement. Journal of Dental Research, 1984, 63: 200
Horstmann W G, Verheyen C C, Leemans R. An injectable calcium phosphate cement as a bone-graft substitute in the treatment of displaced lateral tibial plateau fractures. Injury, 2003, 34(2): 141–144
Stankewich C J, Swiontkowski M F, Tencer A F, et al. Augmentation of femoral neck fracture fixation with an injectable calcium–phosphate bone mineral cement. Journal of Orthopaedic Research, 1996, 14(5): 786–793
Zimmermann R, Gabl M, Lutz M, et al. Injectable calcium phosphate bone cement Norian SRS for the treatment of intraarticular compression fractures of the distal radius in osteoporotic women. Archives of Orthopaedic and Trauma Surgery, 2003, 123 (1): 22–27
Aral A, Yalçin S, Karabuda Z C, et al. Injectable calcium phosphate cement as a graft material for maxillary sinus augmentation: an experimental pilot study. Clinical Oral Implants Research, 2008, 19(6): 612–617
Sato I, Akizuki T, Oda S, et al. Histological evaluation of alveolar ridge augmentation using injectable calcium phosphate bone cement in dogs. Journal of Oral Rehabilitation, 2009, 36 (10): 762–769
Hesaraki S, Nemati R. Cephalexin-loaded injectable macroporous calcium phosphate bone cement. Journal of Biomedical Materials Research Part B: Applied Biomaterials, 2009, 89(2): 342–352
Liu C, Shao H, Chen F, et al. Rheological properties of concentrated aqueous injectable calcium phosphate cement slurry. Biomaterials, 2006, 27(29): 5003–5013
Hesaraki S, Zamanian A, Moztarzadeh F. The influence of the acidic component of the gas-foaming porogen used in preparing an injectable porous calcium phosphate cement on its properties: acetic acid versus citric acid. Journal of Biomedical Materials Research Part B: Applied Biomaterials, 2008, 86(1): 208–216
Ryf C, Goldhahn S, Radziejowski M, et al. A new injectable brushite cement: first results in distal radius and proximal tibia fractures. European Journal of Trauma and Emergency Surgery, 2009, 35(4): 389–396
Lerouxel E, Weiss P, Giumelli B, et al. Injectable calcium phosphate scaffold and bone marrow graft for bone reconstruction in irradiated areas: an experimental study in rats. Biomaterials, 2006, 27(26): 4566–4572
Laschke M W, Witt K, Pohlemann T, et al. Injectable nanocrystalline hydroxyapatite paste for bone substitution: in vivo analysis of biocompatibility and vascularization. Journal of Biomedical Materials Research Part B: Applied Biomaterials, 2007, 82(2): 494–505
Wolff K D, Swaid S, Nolte D, et al. Degradable injectable bone cement in maxillofacial surgery: indications and clinical experience in 27 patients. Journal of Cranio-Maxillo-Facial Surgery, 2004, 32(2): 71–79
Li J, Qiu Z Y, Zhou L, et al. Novel calcium silicate/calcium phosphate composites for potential applications as injectable bone cements. Biomedical Materials, 2008, 3(4): 044102
Otsuka M, Ohshita Y, Marunaka S, et al. Effect of controlled zinc release on bone mineral density from injectable Zn-containing ß-tricalcium phosphate suspension in zinc-deficient diseased rats. Journal of Biomedical Materials Research Part A, 2004, 69(3): 552–560
Otsuka M, Oshinbe A, Legeros R Z, et al. Efficacy of the injectable calcium phosphate ceramics suspensions containing magnesium, zinc and fluoride on the bone mineral deficiency in ovariectomized rats. Journal of Pharmaceutical Sciences, 2008, 97(1): 421–432
Wu F, Su J, Wei J, et al. Injectable bioactive calcium–magnesium phosphate cement for bone regeneration. Biomedical Materials, 2008, 3(4): 1873–1884
Wang X, Ye J, Li X, et al. Production of in-situ macropores in an injectable calcium phosphate cement by introduction of cetyltrimethyl ammonium bromide. Journal of Materials Science: Materials in Medicine, 2008, 19(10): 3221–3225
del Valle S, Miño N, Muñoz F, et al. In vivo evaluation of an injectable macroporous calcium phosphate cement. Journal of Materials Science: Materials in Medicine, 2007, 18(2): 353–361
Wang X, Ye J, Wang Y. Influence of a novel radiopacifier on the properties of an injectable calcium phosphate cement. Acta Biomaterialia, 2007, 3(5): 757–763
Zhao F, Lu W W, Luk K D K, et al. Surface treatment of injectable strontium-containing bioactive bone cement for vertebroplasty. Journal of Biomedical Materials Research Part B: Applied Biomaterials, 2004, 69(1): 79–86
Hu G, Xiao L, Fu H, et al. Study on injectable and degradable cement of calcium sulphate and calcium phosphate for bone repair. Journal of Materials Science: Materials in Medicine, 2010, 21(2): 627–634
Iooss P, Le Ray A M, Grimandi G, et al. A new injectable bone substitute combining poly(e-caprolactone) microparticles with biphasic calcium phosphate granules. Biomaterials, 2001, 22 (20): 2785–2794
Rodríguez-Lorenzo L M, Fernández M, Parra J, et al. Acrylic injectable and self-curing formulations for the local release of bisphosphonates in bone tissue. Journal of Biomedical Materials Research Part B: Applied Biomaterials, 2007, 83(2): 596–608
Blouin S, Moreau MF, Weißs P, et al. Evaluation of an injectable bone substitute (βTCP/hydroxyapatite/hydroxy-propyl-methylcellulose) in severely osteopenic and aged rats. Journal of Biomedical Materials Research Part A, 2006, 78(3): 570–580
Weiss P, Layrolle P, Clergeau L P, et al. The safety and efficacy of an injectable bone substitute in dental sockets demonstrated in a human clinical trial. Biomaterials, 2007, 28(22): 3295–3305
Gauthier O, Goyenvalle E, Bouler J M, et al. Macroporous biphasic calcium phosphate ceramics versus injectable bone substitute: a comparative study 3 and 8 weeks after implantation in rabbit bone. Journal of Materials Science: Materials in Medicine, 2001, 12(5): 385–390
Chang C H, Liao T C, Hsu Y M, et al. A poly(propylene fumarate)–calcium phosphate based angiogenic injectable bone cement for femoral head osteonecrosis. Biomaterials, 2010, 31 (14): 4048–4055
Peter S J, Kim P, Yasko A W, et al. Croßslinking characteristics of an injectable poly(propylene fumarate)/β-tricalcium phosphate paste and mechanical properties of the crosslinked composite for use as a biodegradable bone cement. Journal of Biomedical Materials Research, 1999, 44(3): 314–321
Habraken W J E M, de Jonge L T, Wolke J G C, et al. Introduction of gelatin microspheres into an injectable calcium phosphate cement. Journal of Biomedical Materials Research Part A, 2008, 87(3): 643–655
Link D P, van den Dolder J, van den Beucken J J, et al. Bone response and mechanical strength of rabbit femoral defects filled with injectable CaP cements containing TGF-ß1 loaded gelatin microparticles. Biomaterials, 2008, 29(6): 675–682
Kai D, Li D, Zhu X, et al. Addition of sodium hyaluronate and the effect on performance of the injectable calcium phosphate cement. Journal of Materials Science: Materials in Medicine, 2009, 20(8): 1595–1602
Chazono M, Tanaka T, Komaki H, et al. Bone formation and bioresorption after implantation of injectable ß-tricalcium phosphate granules-hyaluronate complex in rabbit bone defects. Journal of Biomedical Materials Research Part A, 2004, 70(4): 542–549
Pek Y S, Kurisawa M, Gao S, et al. The development of a nanocrystalline apatite reinforced crosslinked hyaluronic acidtyramine composite as an injectable bone cement. Biomaterials, 2009, 30(5): 822–828
Plachokova A, Link D, van den Dolder J, et al. Bone regenerative properties of injectable PGLA–CaP composite with TGF-ß1 in a rat augmentation model. Journal of Tissue Engineering and Regenerative Medicine, 2007, 1(6): 457–464
Moreau J L, Xu H H K. Mesenchymal stem cell proliferation and differentiation on an injectable calcium phosphate–chitosan composite scaffold. Biomaterials, 2009, 30(14): 2675–2682
Liu H, Li H, Cheng W, et al. Novel injectable calcium phosphate/chitosan composites for bone substitute materials. Acta Biomaterialia, 2006, 2(5): 557–565
Montufar E B, Traykova T, Gil C, et al. Foamed surfactant solution as a template for self-setting injectable hydroxyapatite scaffolds for bone regeneration. Acta Biomaterialia, 2010, 6(3): 876–885
Jayabalan M, Shalumon K T, Mitha M K. Injectable biomaterials for minimally invasive orthopedic treatments. Journal of Materials Science: Materials in Medicine, 2009, 20(6): 1379–1387
Yang G J, Lin M, Zhang L, et al. Progress of calcium sulfate and inorganic composites for bone defect repair. Journal of Inorganic Materials, 2013, 28(8): 795–803
Peltier L F, Bickel E Y, Lillo R, et al. The use of plaster of paris to fill defects in bone. Annals of Surgery, 1957, 146(1): 61–69
Yu X W, Xie X H, Yu Z F, et al. Augmentation of screw fixation with injectable calcium sulfate bone cement in ovariectomized rats. Journal of Biomedical Materials Research Part B: Applied Biomaterials, 2009, 89(1): 36–44
Clayer M. Injectable form of calcium sulphate as treatment of aneurysmal bone cysts. ANZ Journal of Surgery, 2008, 78(5): 366–370
Yu B, Han K, Ma H, et al. Treatment of tibial plateau fractures with high strength injectable calcium sulphate. International Orthopaedics, 2009, 33(4): 1127–1133
Vlad M D, del Valle L J, Poeata I, et al. Injectable iron-modified apatitic bone cement intended for kyphoplasty: cytocompatibility study. Journal of Materials Science: Materials in Medicine, 2008, 19(12): 3575–3583
Herberg S, Siedler M, Pippig S, et al. Development of an injectable composite as a carrier for growth factor-enhanced periodontal regeneration. Journal of Clinical Periodontology, 2008, 35(11): 976–984
Song H Y, Esfakur Rahman A H, Lee B T. Fabrication of calcium phosphate–calcium sulfate injectable bone substitute using chitosan and citric acid. Journal of Materials Science: Materials in Medicine, 2009, 20(4): 935–941
Zhang W, Liao S S, Cui F Z. Hierarchical self-assembly of nanofibrils in mineralized collagen. Chemistry of Materials, 2003, 15 (16): 3221–3226
Liao S S, Cui F Z, Zhang W, et al. Hierarchically biomimetic bone scaffold materials: nano-HA/collagen/PLA composite. Journal of Biomedical Materials Research Part B: Applied Biomaterials, 2004, 69(2): 158–165
Chen Z, Liu H, Liu X, et al. Injectable calcium sulfate/mineralized collagen-based bone repair materials with regulable self-setting properties. Journal of Biomedical Materials Research Part A, 2011, 99(4): 554–563
Chen Z, Liu H, Liu X, et al. Injectable mineralized collagenbased bone repair materials. Journal of Controlled Release, 2013, 172(1): e148–e149
Hu N M, Chen Z, Liu X, et al. Mechanical properties and in vitro bioactivity of injectable and self-setting calcium sulfate/nano-HA/collagen bone graft substitute. Journal of the Mechanical Behavior of Biomedical Materials, 2012, 12: 119–128
Chen Z, Liu H, Liu X, et al. Improved workability of injectable calcium sulfate bone cement by regulation of self-setting properties. Materials Science & Engineering C: Materials for Biological Applications, 2013, 33(3): 1048–1053
Lian X J, Liu H Y, Wang X M, et al. Antibacterial and biocompatible properties of vancomycin-loaded nano-hydroxyapatite/collagen/poly(lactic acid) bone substitute. Progress in Natural Science: Materials International, 2013, 23(6): 549–556
Zalavras C G, Patzakis MJ, Holtom P. Local antibiotic therapy in the treatment of open fractures and osteomyelitis. Clinical Orthopaedics and Related Research, 2004, 427: 86–93
Jiang J L, Li Y F, Fang T L, et al. Vancomycin-loaded nanohydroxyapatite pellets to treat MRSA-induced chronic osteomyelitis with bone defect in rabbits. Inflammation Research, 2012, 61(3): 207–215
Joosten U, Joist A, Frebel T, et al. Evaluation of an in situ setting injectable calcium phosphate as a new carrier material for gentamicin in the treatment of chronic osteomyelitis: studies in vitro and in vivo. Biomaterials, 2004, 25(18): 4287–4295
Cui X, Zhao C, Gu Y, et al. A novel injectable borate bioactive glass cement for local delivery of vancomycin to cure osteomyelitis and regenerate bone. Journal of Materials Science: Materials in Medicine, 2014, 25(3): 733–745
Tsai Y F, Wu C C, Fan F Y, et al. Effects of the addition of vancomycin on the physical and handling properties of calcium sulfate bone cement. Process Biochemistry, 2014, 49(12): 2285–2291
Author information
Authors and Affiliations
Corresponding authors
Additional information
Z.C., X.Z. and L.K. contributed equally to this work.
Rights and permissions
About this article
Cite this article
Chen, Z., Zhang, X., Kang, L. et al. Recent progress in injectable bone repair materials research. Front. Mater. Sci. 9, 332–345 (2015). https://doi.org/10.1007/s11706-015-0310-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11706-015-0310-z