Abstract
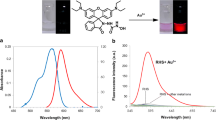
A novel probe RB for discriminative detection of Hg2+ and Cu2+ constructed by rhodamine and BODIPY was designed and synthesized. Probe RB consists of three parts: BODIPY as the energy donor, rhodamine as the energy receptor, and thiosemicarbazide as the reacting site. Probe RB selectively binds with Hg2+ and Cu2+ ions in in EtOH/H2O (V/V = 1:1) among tested metal ions. Probe RB exhibited a fluorescence red shift from 513 to 593 nm for Hg2+ and a fluorescence quenching for Cu2+. The mechanism study showed that the fluorescence changes of probe RB after the addition of Hg2+ was due to oxadiazole formation when the thiosemicarbazide moiety was liberated by Hg2+-facilitated ring opening of the spirocyclic group, while Cu2+ not only opened the spirolactam ring, but also bonded to the opening product through 1:2 mode. It is worth noting that all these types of probes could only detect Hg2+ or Cu2+ through metal ions-facilitated ring opening of the spirocyclic group mechanism. To the best of our knowledge, probe RB was firstly reported to detect Hg2+ and Cu2+ discriminatively. Moreover, probe RB demonstrated excellent sensitivity toward Hg2+ (LOD 8.36 nM) and Cu2+ (LOD 0.16 μM), fast responsive time (within 30 s) and wide pH range (6.0–10.0 for Hg2+, 6.0–11 for Cu2+). This probe was further successfully applied to Hela cells and could easily discriminate Hg2+ and Cu2+ through double-channel imaging.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Heavy metal pollutants have attracted much attention because of their harm to environments and human health. Therefore, selective detection of heavy metal pollutants, especially using fluorescent probes, is currently under in-depth research (Lian et al.2020; Nagarajan et al. 2021; Duong and Kim 2010). Among them, Cu2+ and Hg2+ ions as two common metal pollutants have been fully researched due to their wide application and high toxicity. Cu2+ is closely related to the hematopoietic function of human body. However, when a large number of Cu2+ ions are retained in the human body, it is easy to cause a burden to various organs in the body, especially the liver and gallbladder. When problems occur in these two organs, the maintenance of the metabolism in the human body will appear disorder (Hosseini et al. 2014; Zhao et al.2018). While its wide application in infrastructure constructions, power generation, as well as in the production of electronic products and equipment, industrial machinery and transport vehicles, the level of Cu2+ pollution in the environment is constantly increasing. Hence, it is of great significance to develop fluorescent probe for Cu2+ detection.
Unlike Cu2+, Hg2+ is considered to be one of the most toxic heavy metal ions to human health because it can coordinate with many negatively charged groups in enzymes or proteins, such as sulfhydryl groups, which affect many metabolic pathways in cells, such as energy generation, protein and nucleic acid synthesis, thus further affecting cell function and growth of cells (Tetsuro et al. 2005). Moreover, Hg2+ ions could easily pass through membranes and induce massive damage to central nervous system and organs, such as brain and kidney. Therefore, it is important to develop probes which could detect trace amounts of Hg2+ ions in living cells.
Currently, most reported fluorescent probes could only detect Cu2+ or Hg2+ (Zhu et al. 2021; Slassi et al. 2021; Du et al. 2021; Zhang et al. 2021; Yang et al. 2013, 2021; Culzoni et al. 2012; Chen et al. 2020, 2019; Li et al. 2020; Petdum et al. 2020; Liu et al. 2020a, b; Hu et al. 2020; Lin et al.2020), the fluorescent probes which could discriminately detect two metal ions through different spectra changes were rarely developed, especially in living cells (Huang et al.2013, 2019; Divya and Thennarasu 2020; Shi et al. 2019). Some of reported dual-signal fluorescence sensors have shortcomings in practical applications, such as the indistinguishability of Hg2+ and Cu2+ ions, long response time, low sensitivity and interference from other metal ions. Therefore, it is still needed to develop dual-signal fluorescence sensors which could discriminately detect Hg2+ and Cu2+ ions with large spectral differences.
Rhodamine and BODIPY dyes are widely used in the construction of fluorescent probes due to their excellent spectral properties, such as long absorption and emission wavelength, high fluorescence quantum yield, high extinction coefficient and excellent photostability (Zhang et al. 2020, 2016; Zhang and Wong 2020; Beija et al. 2009; Nguyen et al. 2021; Kaur and Singh 2019; Kowada et al. 2015). Most reported rhodamine probes for Hg2+ detection undergo a transformation from the spirocyclic type to the open-loop amide type, whose fluorescence properties are completely different. The spirocyclic (closed loop) form is basically non-fluorescent, while the open-loop form produces strong fluorescence emission. Based on this mechanism, some chemical sensors for Hg2+ and Cu2+ detection have been developed (Zhang and Zhang 2014; Saleem and Lee 2014; Chen et al. 2020). Among them, a series of ratiometric fluorescent probes by fluorescence resonance energy transfer (FRET) mechanism have been reported. Previously, our group has synthesized a FRET probe based on BODIPY- rhodamine system (Compound 1, Scheme 1), which shows high selectivity toward Hg2+ [Wen et al. 2021]. While all these types of probes could only detect Hg2+ or Cu2+ (Table S1). Recently, our group found a novel FRET probe system with thiosemicarbazide moiety (probe RB, Scheme 1) could discriminatively detect Hg2+ and Cu2+ with excellent sensitivity. Most importantly, probe RB was successfully applied to Hela cells and could easily discriminate Hg2+ and Cu2+ through double-channel imaging. To the best of our knowledge, this type of probe was firstly reported to discriminatively detect Hg2+ and Cu2+ through different mechanisms.
Experimental section
Materials and apparatus
All the materials used for the synthesis of probe RB and analytical experiments were purchased from Sigma-Aldrich without further purification. EtOH in HPLC grade purity and redistilled water were used in all analytical experiments. PerkinElmer Lambda 25 and HITACHI F-4600 Fluorescence spectrophotometer were used for absorption and fluorescence spectra measuring. Bruker Advance 400 MHz spectrometer was used for 1H-NMR and 13C-NMR recording. HRMS data were obtained from an SCIEX TripleTOF 5600 + high resolution spectrometer (American). Leica TCS SP8 Confocal Laser Scanning Microscope was used to obtain the fluorescence images of living cells.
Synthesis of probe RB
Compound 1 (0.3 g, 0.4 mmol) and phenyl isothiocyanate (0.2 mL, 1.3 mmol) were dissolved in dry DMF (5 mL), the mixture was stirred overnight at room temperature. After completion of the reaction, the solvent was removed by a rotary evaporator under reduced pressure to give a red residue, which was purified directly by gel column chromatography (CH2Cl2/MeOH = 50/1) to give a red solid (0.33 g, 0.36 mmol) in 90% yield. 1H NMR (400 MHz, CDCl3) δ 8.04 (d, J = 7.9 Hz, 1H), 7.69–7.60 (m, 2H), 7.52 (s, 1H), 7.18 (t, J = 7.5 Hz, 2H), 7.08 (dd, J = 19.0, 7.4 Hz, 3H), 6.90 (s, 1H), 6.68 (s, 1H), 6.53–6.46 (m, 4H), 6.33–6.31 (m, 1H), 6.06 (s, 2H), 3.79–3.75 (m, 2H), 3.62–3.58 (m, 2H), 3.44–3.26 (m, 4H), 3.21–3.20 (m, 4H), 3.13–3.00 (m, 2H), 2.51 (s, 6H), 2.46 (s, 6H), 2.15–1.91 (m, 2H), 1.66 (s, 3H), 1.16 (t, J = 7.0 Hz, 6H). 13C NMR (100 MHz, CDCl3) δ 170.28, 167.02, 154.10, 153.99, 125.12, 145.52, 140.46, 137.58, 134.46, 131.61, 129.36, 129.08, 128.34, 127.73, 126.12, 124.88, 124.70, 124.05, 121.81, 112.30, 103.23, 66.83, 48.27, 48.11, 45.19, 44.46, 41.25, 32.68, 27.57, 26.99, 16.51, 14.49, 12.51. HRMS (m/z): calculated for C52H56BF2N8O3S [M + H]+, 921.4257; found, 921.4333.
Absorption and fluorescence spectra measurement procedure
The stock solution of probe RB (1 mM) was prepared with EtOH in HPLC grade purity and stored in refrigerator at 4 °C. All the tested metal ions (Ag+, Ba2+, Ca2+, Cd2+, Co2+, Cu2+, Fe3+, K+, Hg2+, Mg2+, Mn2+, Na+, Ni+ and Zn2+, 10 mM) were prepared in ultrapure water. The UV–vis and fluorescence spectra were recorded in the mixture solution EtOH/H2O (V/V = 1:1) at room temperature.
Cell imaging
Hela cells were cultured in Dulbecco's modified Eagle medium (DMEM), which contains 10% Fetal Bovine Serum (FBS) in two days. Then Hela cells were firstly treated with probe RB (5 μM, PBS containing 1% DMSO), which were incubated over 30 min at 37 °C. Then the Hela cells were washed with phosphate buffer saline (PBS) (1 mL × 3). The treated cells were further incubated with Cu2+ or Hg2+ (5 μM, PBS containing 1% DMSO) over 30 min. After removing the culture solvent, the cells were washed with PBS for three times. The fluorescence images of living cells were captured in PBS with Leica TCS SP8 Confocal Laser Scanning Microscope, upon excitation at 488 nm. Emissions were collected with green channel (513 ± 15 nm) and red channel (593 ± 15 nm).
Results and discussion
Probe RB synthesis
The procedures of probe RB synthesis are depicted in Scheme 1. Probe RB was obtained by the reaction of phenyl isothiocyanate with compound 1 in 90% yield. Probe RB was fully characterized by 1H NMR, 13C NMR and HRMS data, which were offered in in the Experimental Section. The spectra of 1H NMR, 13C NMR and HRMS were provided in the supporting information.
Studies of spectral response of probe RB to different metal ions
With probe RB in hand, we firstly investigated its sensing ability toward several metal ions (Ag+, Ba2+, Ca2+, Cd2+, Co2+, Cu2+, Fe3+, K+, Hg2+, Mg2+, Mn2+, Na+, Ni+ and Zn2+) in the mixture solution EtOH/H2O (V/V = 1:1) by absorption and fluorescence spectra. As shown in Fig. 1a, probe RB displays an absorption band around 499 nm, which is the characteristic absorption band of BODIPY. Addition of Cu2+ or Ag+ led to partial decrease in absorption band around 499 nm, accompanied by the appearance of a new band around 564 nm, which was attributed to the typical absorption spectrum of rhodamine group. While with the addition of Hg2+, the absorption spectrum of open-loop rhodamine appeared immediately. The above phenomenon indicated that probe RB interacted with Cu2+/Hg2+/Ag+, further enhancing the conjugation degree of RB-Cu2+/Hg2+/Ag+ system, resulting in the red shift of RB absorption spectrum. In addition, probe RB is slight pink in the mixed solution of EtOH/H2O (V/V = 1:1). When Hg2+ was added, its color changed from pink to purple, while the addition of Cu2+ or Ag+ induced color changed from pink to lavender, indicating that probe RB can detect Hg2+ by naked eye (Fig. 2a). Moreover, the fluorescence spectra of probe RB in the presence of different metal ions were also measured. As shown in Fig. 1b, probe RB exhibited an emission band around 513 nm upon excitation at 480 nm. With addition of Cu2+, its fluorescence was almost quenched completely; while addition of Hg2+ induced the disappearance of emission band at 513 nm, along with the appearance of a new emission band centered at 593 nm. As shown in Fig. 2b, emission color changed from green to blank or red were easily observed by addition of Cu2+ or Hg2+ through naked eye under the illumination with a 365 nm UV-lamp. In addition, Ag+ ions had a slight effect, because it could also induce the fluorescence quenching of BODIPY, with the appearance of a new weak emission band centered at 593 nm. However, it has much longer equilibrium time (10 min, Fig. S1) than that of Hg2+ ions (30 s).
Fluorescence titrations of probe RB with different amounts of Cu2+ or Hg2+ were carried out in the mixture solution EtOH/H2O (V/V = 1:1). As depicted in Fig. 3a, probe RB showed a fluorescence emission band centered at 513 nm, which was decreased gradually and remained unchanged after addition of 8 Mμ Cu2+. Plot of fluorescence intensity changes at 513 nm as a function of concentrations of Cu2+ (0–2.5 Mμ) exhibited an excellent linearity (R2 = 0.99277). While upon progressive addition of Hg2+ (0–20 Mμ), fluorescence intensity at 513 nm decreased, along with a new fluorescence emission band around 593 nm appeared and increased gradually (Fig. 3b). In addition, the fluorescence titration curve showed that the ratio of fluorescence intensity of different emission band (I593 nm/I513 nm) increased dramatically with the increasing concentrations of Hg2+ and reached a plateau with addition of 20 μM Hg2+. Plot of I593 nm/I513 nm changes of probe RB as a function of concentrations of Hg2+ (5–15 μM) exhibited an excellent linearity (R2 = 0.99549). The detection limits of probe RB to Cu2+ and Hg2+ were calculated to be 0.16 μM (0.01 ppm) and 8.36 nM (1.68 ppb) (SI), respectively, which were lower than the maximum permissible amount of Cu2+ (1.3 ppm) and Hg2+ (2.0 ppb) in drinking water that proposed by USA Environmental Protection Agency (EPA).
a The fluorescence spectra changes of probe RB (10 μM) with increasing concentration of Cu2+ (0–8 μM) in EtOH/H2O (V/V = 1:1). Insert: plot of I513 nm changes of probe RB (10 μM) as a function of Cu2+ concentration (0–2.5 μM). b The fluorescence spectra changes of probe RB (10 μM) with increasing concentration of Hg2+ (0–20 μM) in EtOH/H2O (V/V = 1:1). Insert: plot of I593 nm/I513 nm changes of probe RB (10 μM) as a function of Hg2+ concentration (5–15 μM)
Moreover, in order to further confirm the sensing ability of probe RB to Cu2+ and Hg2+ in complicated environments, competition experiments of probe RB for Cu2+ and Hg2+ detection were measured through adding the above-mentioned tested metal ions, respectively. As shown in Fig. 4a, the fluorescence intensity of probe RB with tested metal ions (Ba2+, Ca2+, Cd2+, Co2+, Fe3+, K+, Mg2+, Mn2+, Na+, Ni+ and Zn2+) remained basically unchanged, while Cu2+ addition resulted a ca. 96% quenching of fluorescence at 513 nm. In addition, Ag+ could lead to a circa (ca.) 91% quenching of fluorescence at 513 nm, while the fluorescence intensity was further decreased to ca. 96%, following addition of Cu2+. Moreover, I593 nm/I513 nm was significantly increased with addition of Hg2+ in presence of other tested ions (Ag+, Ba2+, Ca2+, Cd2+, Co2+, Fe3+, K+, Mg2+, Mn2+, Na+, Ni+ and Zn2+), indicating excellent anti-interference ability of probe RB for the detection of Cu2+ and Hg2+. In addition, we also investigated whether Cu2+ and Hg2+ interfered with each other (Figs. S2, S3). The results showed that when Cu2+ was firstly added into the solution of probe RB in EtOH/H2O (V/V = 1:1), it induced a ca. 90% quenching of fluorescence at 513 nm, and the fluorescence intensity at 513 nm was further decreased, along with the appearance of a new emission band centered at 593 nm after another addition of Hg2+. While when Hg2+ was firstly added into the solution of probe RB in EtOH/H2O (V/V = 1:1), it led to the appearance of a new emission band centered at 593 nm, and then another addition of Cu2+had no effect on the new emission band. The above results indicated that the probe RB for detecting Hg2+ was not disturbed by other measured ions, while for detecting Cu2+ was interfered by Hg2+.
Sensing mechanism study
According to the literature (Yang et al. 2005; Ko et al. 2006; Wang et al. 2015; Li et al. 2019; Zhu et al. 2020; Ji et al. 2017), rhodamine fluorophore containing thiosemicarbazide group usually shows no fluorescence, and it could be easily transformed into 1,3,4-oxadiazole by Hg2+-promoted cyclization reaction. Therefore, we assume that probe RB with thiosemicarbazide group shows green fluorescence of BODIPY upon excitation at 480 nm, which is due to no FRET process. When Hg2+ facilitated the cyclization reaction of thiosemicarbazide to form oxadiazole, the FRET was switched on, accompanied by fluorescence emission of rhodamine appearance upon excitation at 480 nm. To verify this hypothesis, we carried out 1H NMR titration and HRMS experiments. The 1H NMR titration experiment was carried out in (CD3)2SO, due to its high solubility for probe RB. Moreover, the response of probe RB to Hg2+ in (CD3)2SO was the same as that in EtOH/H2O (V/V = 1:1). As shown in Fig. 5, the peaks at 8.86 (s, 1H) and 9.40 (s, 1H) are the protons of amine 1 and amine 2, addition of Hg2+ resulted the disappearance of proton 1 and downfield shift to 10.63 of proton 2. Moreover, the protons of rhodamine group showed a downfield shift and the protons of the pyrrole group on BODIPY kept the same at 6.23 (s, 2H) upon addition of Hg2+. These results clearly showed that thiosemicarbazide group could be transformed into oxadiazole by Hg2+ addition. In addition, the HRMS spectrum of probe RB with Hg2+ was recorded. The peak at m/z 887.4457 corresponded to the calculated m/z at 887.4375 for rhodamine containing oxadiazole group (Fig. S4). 1H NMR and HRMS data supported the mechanism which is depicted in Scheme 2. Moreover, The FRET energy transfer efficiency (E) was calculated as 97.8% (E = 1 − IDA/ID) through testing the fluorescence intensity of BODIPY and probe RB (Fig. S5).
Most reported rhodamine containing thiosemicarbazide group showed no response toward Cu2+, in order to understand the sensing mechanism of probe RB to Cu2+, we investigated reversibility, job plot, HRMS experiments. As shown in Fig. 6a, Cu2+ addition induced a ca. 96% quenching of fluorescence at 513 nm, and the fluorescence intensity was almost recovered after addition of EDTA, indicating that the coordination of probe RB with Cu2+ is chemically reversible. While the fluorescence changes induced by Hg2+ could not recover through addition of EDTA, due to the formation of oxadiazole (Fig. 6b). Job plot based on fluorescence change was applied to study the binding stoichiometry of probe RB and Cu2+. The fitting result is depicted in Fig S6 and 2:1 stoichiometry was calculated for probe RB bonding with Cu2+. In addition, the HRMS spectrum of probe RB with Cu2+ showed peaks at 1926.46, which was corresponding to the calculated m/z for 2(RB) + Cu2+ + Na+ (Fig. S7). All above results indicated that the response of probe RB to Cu2+ was due to the coordination of probe RB with Cu2+ through 2:1 binding mode (Scheme 2).
Kinetics study
Time dependent fluorescence change of probe RB in the presence of different concentrations of Cu2+ or Hg2+ were studied. As shown in Fig. 7a, the fluorescence intensity at 513 nm decreased sharply with the increasing concentrations of Cu2+. It was noted that I513 nm significantly decreased after 30 s and reached equilibrium within 1 min, which was much faster than most reported probes (5–30 min, Table S1). Moreover, Hg2+ addition also induced the value of I593 nm/I513 nm augmented with the increasing concentration of Hg2+, and reached equilibrium within 1 min (Fig. 7b). Because of its fast Kinetics, probe RB has a broad application prospect in real-time detection of Cu2+ and Hg2+.
pH influence
To evaluate the practical applicability of probe RB, the suitable operating pH ranges for Cu2+ and Hg2+ detection were measured through fluorescence spectra, respectively. As shown in Fig. 8, the fluorescence intensity at 513 nm or I593 nm/I513 nm of probe RB was basically stable in the pH range of 4–12. In the presence of Cu2+, the fluorescence intensity at 513 nm was decreased 45–75% of its initial level in the range of pH = 4–5. Interestingly, the fluorescence intensity decreased significantly (~ 95% of the initial level) at pH 6 to 12, especially at pH = 6–11 (Fig. 8a). The result showed that probe RB was adequate for Cu2+ detection in biological surroundings. On the other hand, upon addition of Hg2+, I593 nm/I513 nm of probe RB showed no obvious changes at pH below 5 or above 11, which indicated that Hg2+ could not promote the ring-closed reaction of thiosemicarbazides under strong acidic condition or basic condition. In the range of pH 6–10, the presence of Hg2+ significantly improved I593 nm/I513 nm of probe RB (Fig. 8b), indicating that probe RB is suitable for Hg2+ detection under physiological conditions.
Living cellular imaging
In order to further study the practical applicability of the probe RB in biological systems, fluorescence microscopy was used to conduct cell imaging experiments using Leica TCS SP8 Confocal Laser Scanning Microscope. Green channel (513 nm ± 15 nm) and red channel (593 nm ± 15 nm) were used to capture fluorescence emission images. As shown in Fig. 9, obvious green fluorescence in the cells was observed, after incubating living Hela cells with probe RB (5 μM) for 30 min at room temperature, indicating the cell permeability of probe RB. Moreover, in red channel no fluorescence can be observed, which demonstrated that probe RB is very steady and there is no interference for Hg2+ detection in Hela cells. Incubated the Hela cells stained with the probe RB with Cu2+ and Hg2+ (5 μM) in PBS for 30 min, respectively, and then washed with PBS three times. As shown in Fig. 9d, e, the green fluorescence intensity in the green channel was partially quenched (Fig. 9d), and the red color increased significantly, which indicated Hg2+ could promote the cyclization reaction of thiosemicarbazide group to form oxadiazole in living cells. Compared to Hg2+, Cu2+ only induced the green fluorescence intensity partially quenched, which was in accordance with the fluorescence spectrum changes in EtOH/H2O (V/V = 1:1) solution. All above results showed that the probe RB could be used as a fluorescent probe to discriminate Hg2+ and Cu2+ through double-channel imaging in living cells.
The confocal fluorescence images of Hela cells incubated with probe BR (5 μM) over 30 min a green channel, b red channel, c overlay image of a, b; the fluorescence images of Hela cells stained with the probe RB incubated with Hg2+ (5 μM) over 30 min d green channel, e red channel, f overlay image of d, e; the fluorescence images of Hela cells stained with the probe RB incubated with Cu2+ (5 μM) over 30 min g green channel, h red channel, i overlay image of g, h
Conclusion
In summary, we have synthesized a dual-function probe RB containing thiosemicarbazide group, which showed high sensitivity and selectivity toward Cu2+ and Hg2+. This probe could detect Cu2+ and Hg2+ through significant fluorescence on–off and ratiometric fluorescence changes in EtOH/H2O (V/V = 1:1) solution and living cells. The detection limits of probe RB to Cu2+ and Hg2+ were calculated to be 0.01 ppm and 1.68 ppb, respectively, which was lower than the maximum permissible amount of Cu2+ (1.3 ppm) and Hg2+ (2.0 ppb) in drinking water proposed by USA Environmental Protection Agency (EPA). Probe RB containing thiosemicarbazide group was firstly reported to detect Cu2+ through 2:1 binding mechanism. Most importantly, probe RB has been successfully applied for discriminating Hg2+ and Cu2+ through double-channel imaging in living cells.
References
Beija M, Afonso CAM, Martinho JMG (2009) Synthesis and applications of Rhodamine derivatives as fluorescent probes. Chem Soc Rev 38:2410–2433. https://doi.org/10.1039/B901612K
Chen CG, Vijay N, Thirumalaivasan N, Velmathi S, Wu SP (2019) Coumarin-based Hg2+ fluorescent probe: Fluorescence turn-on detection for Hg2+ bioimaging in living cells and zebrafish. Spectrochim Acta A Mol Biomol Spectrosc 219:135–140. https://doi.org/10.1016/j.saa.2019.04.048
Chen SX, Zhang SS, Ruhan A, Han YF (2020) A new rhodamine probe with large stokes shift for Hg2+ detection and its application in real sample analysis. Tetrahedron Lett 61(27):152077. https://doi.org/10.1016/j.tetlet.2020.152077
Culzoni MJ, Pea AMDL, Machuca A, Goicoechea HC, Babiano R (2012) Rhodamine and bodipy chemodosimeters and chemosensors for the detection of Hg2+, based on fluorescence enhancement effects. Anal Methods 5:30–49. https://doi.org/10.1039/C2AY25769F
Divya D, Thennarasu S (2020) A novel isatin-based probe for ratiometric and selective detection of Hg2+ and Cu2+ ions present in aqueous and environmental samples. Spectrochim Acta Part A Mol Biomol Spectrosc 243:118796. https://doi.org/10.1016/j.saa.2020.118796
Du JH, Zhao B, Kan W, Yin HC, Song TS, Wang LY, Sun L, Wang XW, Yin GM, Wang JX (2021) A phenanthrene[9,10-d]imidazole-phenol-based fluorescent probe combining ESIPT and AIE for the ‘“turn-on”’ detection of Cu2+ with green-emission and improved Stokes’ shift, and its application. New J Chem 45:15415–15422. https://doi.org/10.1039/d1nj02177j
Duong TQ, Kim JS (2010) Fluoro- and chromogenic chemodosimeters for heavy metal ion detection in solution and biospecimens. Chem Rev 110(10):6280–6301. https://doi.org/10.1021/cr100154p
Hosseini MJ, Shaki F, Ghazi-Khansari M, Pourahmad J (2014) Toxicity of copper on isolated liver mitochondria: impairment at complexes i, ii, and iv leads to increased ros production. Cell Biochem Biophys 70(1):367–381. https://doi.org/10.1007/s12013-014-9922-7
Hu JW, Yu XL, Zhang X, Jing CC, Liu TT, Hu XC, Lu SL, Uvdal K, Gao HW, Hu ZJ (2020) Rapid detection of mercury (II) ions and water content by a new rhodamine B-based fluorescent chemosensor. Spectrochim Acta Part A Mol Biomol Spectrosc 241:118657. https://doi.org/10.1016/j.saa.2020.118657
Huang JL, Ma XW, Liu B, Cai LB, Li Q, Zhang YQ, Jiang KZ, Yin SC (2013) A colorimetric and ratiometric turn-on bodipy-based fluorescent probe for double-channel detection of Cu2+ and Hg2+. J Lumin 141:130–136. https://doi.org/10.1016/j.jlumin.2013.03.038
Huang Y, Li CF, Shi WJ, Tan HY, He ZZ, Zheng LY, Liu FG, Yan JW (2019) A near-infrared BODIPY-based fluorescent probe for ratiometric and discriminative detection of Hg2+ and Cu2+ ions in living cells. Talanta 198:390–397. https://doi.org/10.1016/j.talanta.2019.02.012
Ji RX, Liu AK, Shen SL, Cao XQ, Li F, Ge YQ (2017) An indolizine–rhodamine based FRET fluorescence sensor for highly sensitive and selective detection of Hg2+ in living cells. RSC Adv 7(65):40829–40833. https://doi.org/10.1039/C7RA07938A
Kaur P, Singh K (2019) Recent advances in the application of BODIPY in bioimaging and chemosensing. J Mater Chem C 7:11361–11405. https://doi.org/10.1039/C9TC03719E
Ko KS, Yang YK, Tae JS, Shin I (2006) In vivo monitoring of mercury ions using a rhodamine-based molecular probe. J Am Chem Soc 128(43):14150–14155. https://doi.org/10.1021/ja065114a
Kowada T, Maeda H, Kikuchin K (2015) BODIPY-based probes for the fluorescence imaging of biomolecules in living cell. Chem Soc Rev 44:4953–4972. https://doi.org/10.1039/C5CS00030K
Li YZ, Qi SJ, Xia CC, Xu YH, Duan GY, Ge YQ (2019) A FRET ratiometric fluorescent probe for detection of Hg2+ based on an imidazo[1,2-a]pyridine-rhodamine system. Anal Chim Acta 1077:243–248. https://doi.org/10.1016/j.aca.2019.05.043
Li CP, Niu QF, Wang JG, Wei T, Li TD, Chen JB, Qin XY, Yang QX (2020) Bithiophene-based fluorescent sensor for highly sensitive and ultrarapid detection of Hg2+ in water, seafood, urine and live cells. Spectrochim Acta Part A Mol Biomol Spectrosc 233:118208. https://doi.org/10.1016/j.saa.2020.118208
Lian J, Xu Q, Wang Y, Meng F (2020) Recent developments in fluorescent materials for heavy metal ions analysis from the perspective of forensic chemistry. Front Chem 8:593291. https://doi.org/10.3389/fchem.2020.593291
Lin CX, Zhang M, Yan XQ, Zhang RQ, He XJ, Yuan YF (2020) A coumarin-boronic based fluorescent “on-off” probe for Hg2+ in aqueous solution. Z Anorg Allg Chem 646:1892–1899. https://doi.org/10.1002/zaac.202000135
Liu B, Liu J, He J, Zhang J, Gao C (2020a) A novel red-emitting fluorescent probe for the highly selective detection of Hg2+ ion with AIE mechanism. Chem Phys 539:110944. https://doi.org/10.1016/j.chemphys.2020.110944
Liu YL, Yang L, Li L, Liang XM, Li SJ, Fu Y (2020b) A dual thiourea-appended perylenebisimide “turn-on” fluorescent chemosensor with high selectivity and sensitivity for Hg2+ in living cells. Spectrochim Acta Part A Mol Biomol Spectrosc 241:118678. https://doi.org/10.1016/j.saa.2020.118678
Nagarajan R, Varadaraju C, Lee KH (2021) Recent advancements in the role of n-heterocyclic receptors on heavy metal ion sensing. Dyes Pigm 191:109331. https://doi.org/10.1016/j.dyepig.2021.109331
Nguyen VN, Ha J, Cho M, Li HD, Swamy KMK, Yoon J (2021) Recent developments of BODIPY-based colorimetric and fluorescent probes for the detection of reactive oxygen/nitrogen species and cancer diagnosis. Coord Chem Rev 439:213936. https://doi.org/10.1016/j.ccr.2021.213936
Petdum A, Faichu N, Sirirak J, Khammultri P, Wanichacheva N (2020) [5]helicene-rhodamine 6 g hybrid-based sensor for ultrasensitive Hg2+ detection and its biological applications. J Photochem Photobiol, A 394:112473. https://doi.org/10.1016/j.jphotochem.2020.112473
Saleem M, Lee KH (2014) Selective fluorescence detection of Cu2+ in aqueous solution and living cells. J Lumin 145:843–848. https://doi.org/10.1016/j.jlumin.2013.08.044
Shi WJ, Li CF, Huang Y, Tan HY, Wei YF, Liu FG, Feng LX, Zheng LY, Chen GS, Yan JW (2019) A remarkable colorimetric probe for fluorescent ratiometric and ON-OFF discriminative detection of Hg2+ and Cu2+ by double-channel imaging in living cells. Dyes Pigments 171:107782. https://doi.org/10.1016/j.dyepig.2019.107782
Slassi S, Aarjane M, Amine A (2021) A novel imidazole-derived schiff base as selective and sensitive colorimetric chemosensor for fluorescent detection of Cu2+ in methanol with mixed aqueous medium. Appl Organomet Chem 35:e6408. https://doi.org/10.1002/aoc.6408
Tetsuro A, Takashi K, Hisato I,In Monirith, Touch Seang T, Annamalai S, Shinsuke T (2005) Mercury contamination in human hair and fish from cambodia: levels, specific accumulation and risk assessment. Environ Pollut 134(1):79–86. https://doi.org/10.1016/j.envpol.2004.07.015
Wang M, Wen J, Qin ZH, Wang HM (2015) A new coumarine-rhodamine FRET system as an efficient ratiometric fluorescent probe for Hg2+ in aqueous solution and in living cells. Dyes Pigments 120:208–212. https://doi.org/10.1016/j.dyepig.2015.04.013
Wen D, Deng XK, Xu GV, Wu HR, Yu YH (2021) A novel FRET fluorescent probe based on BODIPY- rhodamine system for Hg2+ imaging in living cells. J Mol Struct 1236:130323. https://doi.org/10.1016/j.molstruc.2021.130323
Yang YK, Yook KJ, Tae JS (2005) A rhodamine-based fluorescent and colorimetric chemodosimeter for the rapid detection of Hg2+ ions in aqueous media. J Am Chem Soc 127(48):16760–16761. https://doi.org/10.1021/ja054855t
Yang Y, Zhao Q, Feng W, Li F (2013) Luminescent chemodosimeters for bioimaging. Chem Rev 113(1):192–270. https://doi.org/10.1021/cr2004103
Yang CC, Li YY, Wu N, Zhang YC, Feng W, Yu MM, Li ZX (2021) Ratiometric upconversion luminescence nanoprobes for quick sensing of Hg2+ and cells imaging. Sens Actuators, B Chem 326:128841. https://doi.org/10.1016/j.snb.2020.128841
Zhang QY, Wong KMC (2020) Photophysical, ion-sensing and biological properties of rhodamine-containing transition metal complexes. Coord Chem Rev 416:213336. https://doi.org/10.1016/j.ccr.2020.213336
Zhang L, Zhang XH (2014) A selectively fluorescein based colorimetric probe for detecting copper(II) ion. Spectrochim Acta Part A Mol Biomol Spectrosc 133:54–59. https://doi.org/10.1016/j.saa.2014.04.130
Zhang RQ, Yan FY, Huang YC, Kong DP, Ye QH, Xu JX, Chen L (2016) Rhodamine-based ratiometric fluorescent probes based on excitation energy transfer mechanisms: construction and applications in ratiometric sensing. RSC Adv 6:50732–50760. https://doi.org/10.1039/C6RA06956H
Zhang J, Wang NN, Ji X, Tao YF, Wang JM, Zhao WL (2020) BODIPY-Based fluorescent probes for biothiols. Chem: A Eur J 26(19):4172–4192. https://doi.org/10.1002/chem.201904470
Zhang CL, Zhang Y, Li MX, Gong S, Gao Y, Yang YQ, Wang ZL, Wang SF (2021) A novel AIE fluorescent probe based on myrtenal for Cu2+ detection in a near-perfect aqueous medium and bioimaging in vegetables and zebrafish. New J Chem 45(34):15247–15254. https://doi.org/10.1039/D1NJ02662C
Zhao D, Zhang XN, Li XF, Ru SG, Wang YW, Yin JB, Liu DS (2018) Oxidative damage induced by copper in testis of the red swamp crayfish procambarus clarkii and its underlying mechanisms. Aquat Toxicol 207:120–131. https://doi.org/10.1016/j.aquatox.2018.12.006
Zhu ZF, Ding HC, Wang YS, Fan CB, Tu YY, Liu G, Pu SZ (2020) A ratiometric and colorimetric fluorescent probe for the detection of mercury ion based on rhodamine and quinoline–benzothiazole conjugated dyad. J Photochem Photobiol, A 400:112657. https://doi.org/10.1016/j.jphotochem.2020.112657
Zhu DJ, Jiang SL, Zhao W, Yan XW, Xie W, Xiong YH, Wang SJ, Cai W, Gao YJ, Ren AS (2021) A novel ratiometric fluorescent probe for sensitive and selective detection of Cu2+ based on Boranil derivatives. Inorg Chim Acta 542:120438. https://doi.org/10.1016/j.ica.2021.120438
Acknowledgements
Thanks for the support of Jianghan University Foundation (2021KJZX001).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ma, L., Xu, G., Deng, X. et al. A dual-channel chemosensor based on rhodamine and BODIPY conjugated dyad for ratiometric detection of Hg2+ and fluorescence on–off recognition of Cu2+ in aqueous solution and living cells. Chem. Pap. 77, 583–593 (2023). https://doi.org/10.1007/s11696-022-02504-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-022-02504-6