Abstract
Bariatric surgery has been widely performed to treat morbid obesity. Our meta-analysis aims to provide an updated comparison between laparoscopic sleeve gastrectomy (LSG) and laparoscopic greater curvature plication (LGCP). Medline, EMBASE, Scopus, and Cochrane Central were searched. Ongoing clinical trials were identified from the clinicaltrials.gov website. References of the chosen literatures were manually reviewed for additional relevant studies. As a result, a total of 18 studies involving 1329 patients were selected. We demonstrated a significant higher excess weight loss (%EWL) after LSG at the 1-, 3-, 6-, 12-, and 18-month follow-up time points. However, no significant difference was found at 36 months. Body Mass Index Loss (BMIL) was better after LSG than LGCP at 12 and 24 months. The difference in the improvement of comorbidities (i.e., T2-DM, hypertension, and sleep apnea) did not reach statistical significance. The complications (i.e., bleeding, stenosis, leak, and abdominal pain), operative time, and length of hospital stay were comparable. More patients undergoing LGCP experienced nausea and vomiting. We obtained some different and new results compared to the previously published meta-analysis. Our meta-analysis showed significantly higher %EWL at 24 months (Z=2.08, p=0.04), significantly higher BMIL at 36 months (Z=9.11, p <0.00001), and significantly higher costs (Z=2.87, p=0.004) in the LSG group. In addition, for the first time, complications (i.e., GERD, wound infection, port-site hernia, and mortality) and improvement of dyslipidemia were compared between the two techniques. According to our pooled data, no significant differences were found in any of the above aspects. In conclusion, LSG is superior to LGCP with regard to providing effective weight loss in the short- and mid-term. LSG has a lower rate of minor complications, but was less effective when considering cost. The two procedures are similar in terms of improvement of comorbidities, major complications, operative time, and length of stay.
Graphical abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Since 1997, obesity has been officially recognized by the World Health Organization as a worldwide epidemic. Morbid obesity and its associated comorbidities have now placed a medical and economic burden on the world [1,2,3]. Bariatric surgery could ensure effective weight loss and maintain weight at a relatively satisfactory level [4]. In addition, it is effective for comorbidities of obesity, such as T2DM and hypertension [4].
Surgical treatment for obesity is either malabsorptive, restrictive, or a combination of the two. LSG and LGCP are the most commonly used restrictive procedures for morbid obesity [5]. In recent decades, LSG has been accepted as an effective and safe bariatric procedure and has been widely performed [6]. It involves resection of most of the stomach, including the entire fundus, along the long axis of the greater curvature, leaving the remaining stomach from the gastric antrum 2–6 cm above the pylorus to the gastroesophageal junction in a “banana shape.” The procedure reduces the volume of the stomach to approximately 180–200 ml, leading to a reduction in food intake [7]. LGCP, on the other hand, is another bariatric procedure that decreases the volume of the stomach by plication of the greater curvature [8, 9]. It could achieve the same weight loss effect as LSG to a certain extent through stomach restriction. Compared to LSG, LGCP has the advantages of technical simplicity and reversibility, preservation of the integrity of the stomach, lower leakage rate, and lower operative costs [10, 11].
Both procedures have advantages and disadvantages and thus need to be carefully examined and compared. However, there are limited comparative trials of the two procedures. The previously published meta-analysis [12] included only 12 comparative studies, and the sample sizes of some of the included studies were quite low. Suarez et al. [13] have just published an in-depth and extensive systematic review of the literature on LSG vs LGCP. However, only 9 of the included 28 studies compared the two procedures. Furthermore, due to the descriptive nature, their study was not subjected to formal statistical analysis. Thus, we conducted this updated meta-analysis to compare the efficacy, safety, operation duration, length of stay, and cost between LSG and LGCP.
Materials and Methods
Study Protocol
This meta-analysis was conducted in accordance with the principles described in the Cochrane Handbook and the PRISMA recommendations [14]. The present study was not registered in any database.
Eligibility Criteria
The inclusion criteria were as follows: (1) study design: any type of comparative study, (2) study population: patients undergoing LGCP or LSG, (3) intervention: comparison between LGCP and LSG, (4) outcomes: inclusion of information on efficacy or safety, (5) availability of the full text, and (6) publication in English. When a study reporting the same patient cohort was included in several publications, only the most recent or complete study was selected. The exclusion criteria included overlapping data and publications in the form of case series, case reports, editorials, letters, conference abstracts, and expert opinions.
Search Strategy
Electronic literature search was performed on Medline, EMBASE, Scopus, and the Cochrane Central inception to December 2020. The language was limited into English. The search terms were as follows: (plication OR imbrication OR gastroplication OR plicature OR verticalplication) AND (sleeve OR vertical) AND (laparoscopy OR laparoscopic). Ongoing clinical trials were identified from the clinicaltrials.gov website. References of the chosen literatures and published systematic reviews were reviewed manually for additional relevant studies.
Study Selection and Data Extraction
Two reviewers (Haoran Li and Weiqiang Wang) independently and blindly evaluated all titles and abstracts for studies that met the inclusion criteria, and excluded any articles that clearly did not meet the selection criteria. The potential inclusions were checked by one author (Xu Wang). Full reports (where available) of potentially relevant studies were retrieved and checked for eligibility. Then data extraction was performed independently and blindly by two investigators (Haoran Li and Weiqiang Wang) according to a standardized form. Efforts were made to get exact numerical data from authors via e-mail if not available in articles. Disagreements were resolved by two reviewers or even the third one if these two reviewers’ decisions could not reach a consensus.
The following data were extracted from included studies: %EWL, BMIL, improvement of comorbidities, major and minor complications, operative time, hospital stay, and cost. Complications were categorized as “minor” or “major” based on the original authors’ discretion, or the Clavien-Dindo scale if not explicitly categorized (grades I and II categorized as mild, grades III and IV categorized as major) [15]. In addition, characteristics of articles including study design, publication year, study location, baseline demographics, baseline BMI, and follow-up were routinely extracted. It is worth emphasizing that the exact numbers of cases at different follow-up time points were retrieved and used for the data analyses of %EWL and BMIL if the article clearly provided, so as to improve the reliability of the meta-analysis results.
Quality Assessment
Two authors (Haoran Li and Weiqiang Wang) assessed the risk of bias independently. We used the revised Jadad rating scale [16] to evaluate the methodological quality of randomized control trials (RCTs), with high-quality studies scoring 4–7 points. The Newcastle–Ottawa scale was used to judge study quality, as recommended by the Cochrane Collaboration [17]. This scale allocates a maximum of nine points with a score of ≥6 being of high quality. Any dispute was solved unanimously through discussion.
Statistical Analysis
Comprehensive meta-analysis was performed using Review Manager Version 5.3. Continuous variables were pooled using weighted mean difference (WMD) with 95% confidence interval (CI), while odds ratio (OR) with 95% CI was applied to perform the statistical analysis for dichotomous variables. A p value of <0.05 was regarded as statistically significant. The χ2 and I2 statistics were adopted to analyze the statistical heterogeneity across the included studies. An I2 value of 25 and 75% indicated low and high levels of heterogeneity, respectively [18]. We used fixed-effect models or random-effect models for comparisons. We considered statistically significant heterogeneity to be present at a p value of <0.10 with the use of a random-effect model.
Risk of Bias Across Studies
The funnel plot of the primary endpoint was visually inspected, in order to determine the presence of publication bias.
Additional Analyses
No sensitivity analysis was generated.
Results
Search Yields
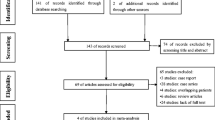
We got 132 search results from Medline, 446 results from EMBase, 60 results from the Cochrane Central Register of Controlled Trials, and 137 results from Scopus. Additional, one study was identified in the gray literature search. All titles and abstracts were screened and 753 studies were excluded. The remaining 23 trials were submitted to a full-text review in order to assess consistency with the predefined eligibility criteria. The full-text screening resulted in the identification and removal of 5 articles (3 repeated publications from the same medical center and 3 irrelevant records). Eighteen studies [19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36] were finally selected for meta-analysis. The flow chart for study selection is shown in Fig. 1.
Study Characteristics
The available study designs consisted of 6 retrospective studies, 7 prospective studies, and 5 RCTs. The analysis was performed on a total of 1329 patients, of whom 705 underwent LSG and 624 underwent LGCP. Studies were conducted in Spain, Romania, Czech Republic, Iran, Korea, Turkey, France, Ukraine, Italy, India, Egypt, and China. In total, 11 trials were conducted in a single institution, and 6 studies incorporated multiple surgical centers, except for one study that did not specify the institution. The study completion year spanned from 2011 to 2020. Postoperative follow-up extended from 1 month up to 3 years. All studies provided age, BMI, and gender data with a comparison of complete baseline profiles between two treatment groups. The baseline characteristics of included articles are represented in Table 1. Moreover, some of the included studies provided the exact number of cases at different follow-up time points, as summarized in Table 2.
Quality of Included Studies
The quality assessment of the included studies is illustrated in Tables 3 and 4. In general, all the 13 cohort studies were considered to be of high quality by gaining a score of ≥ 6 points according to the Newcastle–Ottawa scale, and the quality of 4 included RCTs was also satisfactory by gaining a score of ≥ 4 points in accordance with the revised Jadad rating scale. The quality level of the remaining 1 RCT was estimated to be low, with a score of 2 points.
Meta-Analysis Results
% EWL
This outcome was evaluated after 1, 3, 6, 12, 18, 24, and 36 months. Our meta-analysis demonstrated a significantly higher %EWL after LSG than LGCP at the follow-up time points of 1 month (MD 4.48, 95% CI [0.43, 8.53], I2 0%), 3 months (MD 5.37 ,95% CI [1.59, 9.16], I2 56%), 6 months (MD 8.47, 95% CI [5.81, 11.13], I2 24%), 12 months (MD 13.23, 95% CI [9.93, 16.54], I2 31%), 18 months (MD 10.63, 95% CI [3.72, 17.54], I2 69%), and 24 months (MD 19.62, 95% CI [1.15,38.08], I2 94%). However, no significant difference was found between these two procedures at 36 months (MD 24.63, 95% CI [−1.94, 51.21], I2 93%). High heterogeneity was noted in the 3-, 18-, 24- and 36-month analyses. Random effect was used, because it was not possible to exclude outliers. The forest plots are shown in Figs. 2, 3, 4, 5, 6, 7 and 8.
BMIL
This outcome was evaluated at 6, 12, 24, and 36 months. There was no difference between interventions at 6 months (MD 1.85, 95% CI [− 0.06, 3.76], I2 89%). The results were favorable for LSG at 12 months (MD 3.04, 95% CI [1.35, 4.74], I2 72%), 24 months (MD 5.25, 95% CI [3.65, 6.85], I2 64%), and 36 months (MD 6.03, 95% CI [4.73, 7.32, I2 13%). Random effect was used due to high heterogeneity in the 6-, 12-, and 24-month analyses. The forest plots are shown in Figs. 9, 10, 11 and 12.
Comorbidity Improvement
Diabetes, hypertension, dyslipidemia, and sleep apnea were evaluated in this meta-analysis. More specifically, no statistically significant differences between the two groups regarding improvement of diabetes (OR 1.55, 95% CI [0.55, 4.36], I2 0%), hypertension (OR 1.52, 95% CI [0.67, 3.03], I2 0%), dyslipidemia (OR 1.29, 95% CI [0.41, 4.00], I2 0%), and sleep apnea (OR 1.14, 95% CI [0.35, 3.70], I2 0%), were identified. We resorted to a fixed-effect model because there was no remarkable heterogeneity. The forest plots are shown in Fig. 13, 14, 15 and 16.
Major Complications
In Figs. 17, 18, 19 and 20, the pooled results of the eligible studies concerning the major complications are depicted. There are no differences between LSG and LGCP with regard to bleeding (OR 1.37, 95% CI [0.61, 3.09], I2 0%), stenosis (OR 0.57, 95% CI [0.23, 1.38], I2 0%), leak (OR 1.58, 95% CI [0.61, 4.15], I2 0%), and mortality (OR 1.39, 95% CI [0.09, 22.55], I2 35%). Heterogeneity was noted in the analysis of mortality and random effect was applied.
Minor Complications
Figures 21, 22, 23, 24 and 25 summarize the data regarding the comparisons between the two groups, in terms of minor complications. No differences were noted with regard to GERD (OR 1.38, 95% CI [0.67, 2.82], I2 0%), wound infection (OR 1.70, 95% CI [0.22, 13.27], I2 0%), port-site hernia (OR 1.26, 95% CI [0.28, 5.77], I2 0%), and abdominal pain (OR 0.48, 95% CI [0.21, 1.07], I2 0%). Statistically significant lower rate of nausea and vomiting (OR 0.36, 95% CI [0.23, 0.54], I2 0%) in the LSG group was recorded.
Operative Time
Twelve trials included this outcome for meta-analysis, but two were excluded because of lack of standard deviation [21, 33]. Eight hundred twenty-seven patients (435 in LSG group, 392 in LGCP group) from two studies were finally analyzed. A high heterogeneity was noted, and it was not possible to exclude outliers. Consequently, there is no difference for both surgical therapies with random effect (MD1.42, 95% CI [−9.49, 12.34], I2 94%). The forest plot is shown in Fig. 26.
Hospital Stay
Thirteen studies reported this information, but two could not be included because of lack of standard deviation [20, 32]. Eight hundred fourteen patients (419 in LSG group, 395 in LGCP group) were finally analyzed. A high heterogeneity was noted in the initial analysis. After identifying and excluding the outlier [36], the heterogeneity remained. A random effect was applied for analysis, and no significant difference was found (MD 0.60, 95% CI [− 1.18, 1.37], I2 97%). The forest plot is shown in Fig. 27.
Cost
Figure 28 summarizes the data regarding the comparison between the two groups, in terms of cost. A total of three articles reported this result. One hundred fifty-nine patients (80 in LSG group, 79 in LGCP group) were finally analyzed. A high heterogeneity was noted in the initial analysis. After identifying and excluding the outlier [23], the heterogeneity remained. A random effect was used for analysis, and LSG showed inferior result compared with LGCP considering cost (SMD 11.69, 95% CI [3.71, 19.67], I2 97%).
Publication Bias
Funnel plot of the primary outcome (i.e., %EWL at 6 months) was created to access the publication bias of the literature. Through visual inspection, the shape of the funnel plot did not reveal any evidence of obvious asymmetry (Fig. 29).
Discussion
Over the past few decades, LSG has been regarded as an effective and safe treatment for morbid obesity, not only to significantly reduce weight, but also for improving the associated comorbidities [37,38,39]. However, its irreversibility involving gastrectomy, operative complications, and partial mortality were the concern [40]. More specifically, the overall incidence of serious complications after LSG was estimated at 0.89%, which was principally attributed to the leak [41].
As an alternative to LSG, LGCP is performed by the plication of greater curvature with sutures. It could achieve the same weight loss effect as LSG to a certain extent through stomach restriction. As compared to LSG, LGCP could offer advantages including reversibility, preservation of the integrity of the stomach, and alleviating the risk of gastric content leak of the anastomosis [27]. However, its reliability is controversial. Some studies showed a comparable %EWL in the short or medium term for both procedures [42,43,44,45]. Nevertheless, some other trials have shown that LGCP is inferior to LSG regarding %EWL [46, 47]. Through our meta-analysis, although LGCP resulted in significant weight loss, LSG outperformed it in terms of %EWL and BMIL in the first 2 years. At 3 years postoperatively, both groups had achieved a similar %EWL; however, the results of BMIL were favorable for LSG. In an overall view, compared to the previously published meta-analysis, we provided further evidence of the advantage of LSG in terms of mid-term weight loss and preliminarily indicated that long-term weight loss (3 years) seems to be superior in the LSG patients. However, due to the low number of studies included and great heterogeneity of %EWL analysis, long-term results from our data have to be considered with caution.
The difference of weight loss may be due to the following major reasons. First of all, ghrelin plays a crucial role in regulating appetite and body weight [48,49,50,51]. This hormone is mainly produced by P/D1 cells in the fundus of the stomach [52]. Ghrelin secretion can cause hunger by increasing appetite, emptying the stomach, and moving the intestines. It decreased significantly after LSG but increased after LGCP [53]. This is due to the resection of the fundus of the stomach, where most ghrelin is produced, in LSG [54].
Another gastrointestinal hormone, peptide tyrosine tyrosine (PYY), must also be considered. It is produced in L cells located in the colon and distal ileum. This hormone has the effect of reducing appetite and sense of hunger. In Xanthakos’s trial [52], 12 obese and 12 lean human subjects received intravenous injection of PYY. The result showed a 30% reduction in single meal intake in both groups. In addition, PYY reduced ghrelin levels and enhanced its effect on reducing hunger. Karamanakos [55] reported that PYY did not increase after a trial meal after LGCP; however, PYY increased after LSG; this leads to better weight loss after LSG.
Another possible explanation for the different efficiencies of weight loss may be the technical difference between the two procedures. We must consider that the reduction in gastric volume in LGCP is mainly caused by the plication of the greater curvature of the stomach with sutures and the “space-occupying effect” of the mucosal in-folding. Abdelbaki et al. [56] performed gastroscopy on some patients who regained weight more than 1 year after LGCP. They found moderate atrophy of the infolded gastric mucosa. This mechanism may lead to loosening of the running suture and a diminished filling effect from the gastric mucosal in-folding, leading to a relative gastric enlargement and subsequent weight regain in patients undergoing gastric plication.
All of the above factors could explain why LSG is superior to LGCP considering weight loss. In addition, the improvement of obesity-related comorbidities also plays a non-negligible role in the evaluation of efficacy. In our meta-analysis, both procedures were effective treatments for obese patients with diabetes, hypertension, dyslipidemia, and sleep apnea, with no significant difference between the two groups. This may be due to multiple factors such as hormonal changes [57], BMI loss, and decrease in carbohydrate absorption [58] after bariatric surgery.
LGCP, considered as a less invasive technique, was expected to be better tolerated than LSG. We summarized the major complications as follows: bleeding, stenosis, leak, thromboembolism, and mortality. Other complications were secondary and included nausea and vomiting, GERD, wound infection, port-site hernia, sialorrhea, and abdominal pain. Contrary to expectation, this meta-analysis failed to show any advantage of LGCP in reducing complications. More specifically, at the time of classification, LGCP displayed a statistically significant higher rate of nausea and vomiting. This adverse effect may be attributed to the double row stitching, the large stomach folds imbricated in the gastric lumen [59], and mucosal edema due to venous stasis after LGCP [60]. In terms of major complications, bleeding, stenosis, and mortality outcomes were comparable between the two procedures. It should be noted that out of all 18 articles selected, only 12 cases of leakage were reported, with no significant differences between the two procedures. In addition to this, two of the selected articles reported leaks only after LGCP [31, 33]. As leakage is a serious complication of LSG, it is important to demonstrate that complications are not reduced with the use of LGCP.
Besides these, it is important to note that the choice between LSG and LGCP does not depend solely on their effectiveness and safety, but also on the comparison of operative time, length of stay, and economic costs. The expected outcome of LSG is a shorter operative time [11]. However, according to our pooled data, LSG was not associated with statistically shorter operative time. For this outcome interpretation, we must take into account the surgical technique. In the performance of LSG, some surgeons used reinforcement sutures in the stapling line, which may have prolonged the operation and reduce the difference between the two groups. Major complications are considered to be a crucial factor affecting the length of hospital stay. This meta-analysis demonstrated no significant difference between the two procedures regarding hospital stay, which was the same as the analysis of major complications. The cost of LSG was significantly higher as demonstrated in our meta-analysis. However, due to the high heterogeneity level, the low number of studies included, and the fact that one paper included [23] shows massive cost difference ($17100 vs $2620), the significance of the analysis was confined and we could not make a strong conclusion. It is worth emphasizing that the analysis of cost must take into account a variety of factors. Suarez et al. [13] have done important work in terms of comparing the costs of the two procedures. They examined the price of operative consumables (i.e., endoscopic stapler and sutures), length of stay, duration of surgery, and reoperation rates to evaluate the impact of these factors on the total cost. They draw a conclusion that LGCP is a little bit cheaper than LSG; however, the financial burden of treating the complications may lead to an underestimation of the overall cost of LGCP.
This meta-analysis has several limitations. Firstly, most of the included studies reported short- or mid-term results. Long-term result on follow-up of 4 years or more was lacking. Secondly, the small sample sizes of some of the included trials may have contributed to bias. Furthermore, of all 18 included studies, only 5 were RCTs, which may have biased the selection and detection. Last but not the least, only three of the included studies compared the costs of the two procedures. Therefore, the reliability of the cost advantage of LGCP was limited, which may be important for its popularity in economically underdeveloped areas.
Conclusion
The advantages of LSG in terms of short- and mid-term weight loss and minor complications were documented. However, both procedures were similar in terms of improvement of comorbidities, major complications, operation duration, and hospital stay. The cost-effectiveness of LGCP was better than that of LSG. However, given the limitations mentioned above, we should be cautious in drawing conclusions. More prospective studies with large samples and long-term follow-up are needed in the future, as well as more clinical trials on the charges for the procedures.
References
Schachter J, Martel J, Lin CS, et al. Effects of obesity on depression: a role for inflammation and the gut microbiota. Brain Behav Immun. 2018;69(1):1–8.
Wu YK, Berry DC. Impact of weight stigma on physiological and psychological health outcomes for overweight and obese adults: a systematic review. J Adv Nurs. 2017;74(5):1030–42.
Smith KB, Smith MS. Obesity statistics. Prim Care. 2016;43:121–35.
Azagury D, Papasavas P, Hamdallah I, et al. ASMBS Position Statement on medium- and long-term durability of weight loss and diabetic outcomes after conventional stapled bariatric procedures. Surg Obes Relat Dis. 2018;14:1425–41.
Carrano FM, Peev MP, Saunders JK, et al. The role of minimally invasive and endoscopic technologies in morbid obesity treatment: review and critical appraisal of the current clinical practice. Obes Surg. 2020;30:736–52.
Angrisani L, Santonicola A, Iovino P, et al. IFSO Worldwide Survey 2016: primary, endoluminal, and revisional procedures. Obes Surg. 2018;28:3783–94.
Kueper MA, Kramer KM, Kirschniak A, et al. Laparoscopic sleeve gastrectomy: standardized technique of a potential stand-alone bariatric procedure in morbidly obese patients. World J Surg. 2008;32(7):1462–5.
Khidir N, Al DM, El AW, et al. Outcomes of laparoscopic gastric greater curvature plication in morbidly obese patients. J Obes. 2017:1–6.
Doležalova-Kormanova K, Buchwald JN, Skochova D, et al. Five-year outcomes: laparoscopic greater curvature plication for treatment of morbid obesity. Obes Surg. 2017;27(11):2818–28.
Picot J, Jones J, Colquitt JL, et al. The clinical effectiveness and cost-effectiveness of bariatric (weight loss) surgery for obesity: a systematic review and economic evaluation. Health Technol Assess. 2009;13:1-190–215-357. iii-iv
Zerrweck C, Rodriguez JG, Aramburo E, et al. Revisional surgery following laparoscopic gastric plication. Obes Surg. 2017;27(1):38–43.
Perivoliotis K, Sioka E, Katsogridaki G, et al. Laparoscopic gastric plication versus laparoscopic sleeve gastrectomy: an up-to-date systematic review and meta-analysis. J Obes. 2018;3617458
Suarez DF, Gangemi A. How bad is “bad”? A cost consideration and review of laparoscopic gastric plication versus laparoscopic sleeve gastrectomy. Obes Surg. 2021;31(1):307–16.
Moher D, Liberati A, Tetzlaff J, et al. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoSMedicine. 2009;6(7):e1000097.
Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg. 2004;240(2):205–13.
Jadad AR, Moore RA, Carroll D, et al. Assessing the quality of reports of randomized clinical trials: is blinding necessary? Control Clin Trials. 1996;17:1–12.
Wells G (2014) The Newcastle-Ottawa Scale (NOS) for assessing the quality of non-randomised studies in meta-analyses. Symposium on Systematic Reviews: Beyond the Basics
Higgins JP, Thompson SG, Deeks JJ, et al. Measuring inconsistency in meta-analyses. BMJ. 2003;327(7414):557–60.
Lopeznava G, Asokkumar R, Bautista I et al. Endoscopic Sleeve Gastroplasty (ESG), Laparoscopic Sleeve Gastrectomy (LSG), and Laparoscopic Greater Curve Plication (LGCP): Do They Differ at 2-years?. Endoscopy 2020. https://doi.org/10.1055/a-1224-7231
Neagoe RM, Timofte D, Mureşan M, et al. Laparoscopic gastric plication vs laparoscopic sleeve gastrectomy - a single center, prospective and case-control study. Chirurgia (Bucur). 2019;114:769–78.
Li YH, Wang BY, Huang YC, et al. Clinical outcomes of laparoscopic greater curvature plication and laparoscopic sleeve gastrectomy: a case-matched control study. Obes Surg. 2019;29:387–93.
Talebpour M, Sadid D, Talebpour A, et al. Comparison of short-term effectiveness and postoperative complications: laparoscopic gastric plication vs laparoscopic sleeve gastrectomy. Obes Surg. 2018;28:996–1001.
Nabil TM, Moustafa KG, Balamoun HA (2018) Comparison of laparoscopic sleeve gastrectomy and laparoscopic gastric plication: two-year follow-up results. Bariatric Surgical Patient Care
Casajoana A, Pujol J, Garcia A, et al. Predictive value of gut peptides in T2D remission: randomized controlled trial comparing metabolic gastric bypass, sleeve gastrectomy and greater curvature plication. Obes Surg. 2017;27:2235–45.
Bužga M, Švagera Z, Tomášková H, et al. Metabolic effects of sleeve gastrectomy and laparoscopic greater curvature plication: an 18-month prospective, observational, open-label study. Obes Surg. 2017;27:3258–66.
Park YH, Kim SM. Short-term outcomes of laparoscopic greater curvature plication and laparoscopic sleeve gastrectomy in patients with a body mass index of 30 to 35 kg/m2. Yonsei Med J. 2017;58:1025–30.
Grubnik VV, Ospanov OB, Namaeva KA, et al. Randomized controlled trial comparing laparoscopic greater curvature plication versus laparoscopic sleeve gastrectomy. Surg Endosc. 2016;30:2186–91.
Toprak SS, Gültekin Y, Okuş A. Comparison of laparoscopic sleeve gastrectomy and laparoscopic gastric plication: one year follow-up results. Ulus Cerrahi Derg. 2016;32:18–22.
Chouillard E, Schoucair N, Alsabah S, et al. Laparoscopic gastric plication (LGP) as an alternative to laparoscopic sleeve gastrectomy (LSG) in patients with morbid obesity: a preliminary, short-term, case-control study. Obes Surg. 2016;26:1167–72.
Abdelnazer NA, Daoud EA, Sharouda SM. Laparoscopic gastric plication versus laparoscopic sleeve gastrectomy as a surgical treatment of morbid obesity. Life Science Journal. 2016;13(1)
Verdi D, Prevedello L, Albanese A, et al. Laparoscopic gastric plication (LGCP) vs sleeve gastrectomy (LSG): a single institution experience. Obes Surg. 2015;25:1653–7.
Abouzeid M, Taha O. Laparoscopic sleeve gastrectomy versus laparoscopic gastric greater curvature plication: a prospective randomized comparative study. Egyptian Journal of Surgery. 2015;34(1)
Sharma S, Narwaria M, Cottam DR, et al. Randomized double-blinded trial of laparoscopic gastric imbrication vs laparoscopic sleeve gastrectomy at a single Indian institution. Obes Surg. 2014;25(5):1–5.
Abdelbaki TN, Sharaan M, Abdel-Baki NA, et al. Laparoscopic gastric greater curvature plication versus laparoscopic sleeve gastrectomy: early outcome in 140 patients. Surg Obes Relat Dis. 2014;10:1141–6.
Shen D, Ye H, Wang Y, et al. Comparison of short-term outcomes between laparoscopic greater curvature plication and laparoscopic sleeve gastrectomy. Surg Endosc. 2013;27:2768–74.
Morshed G, Abdalla H. Laparoscopic gastric plication versus laparoscopic sleeve gastrectomy. Med J Cairo Univ. 2011;79(4)
Alexandrou A, Athanasiou A, Michalinos A, et al. Laparoscopic sleeve gastrectomy for morbid obesity: 5-year results. Am J Surg. 2015;209(2):230–4.
Golomb I, Ben DM, Glass A, et al. Long-term metabolic effects of laparoscopic sleeve gastrectomy. JAMA Surg. 2015;150(11):1051–7.
Varela JE, Nguyen NT. Laparoscopic sleeve gastrectomy leads the U.S. utilization of bariatric surgery at academic medical centers. Surg Obes Relat Dis. 2015;11(5):987–90.
Coblijn UK, Karres J, Raaff CALD, et al. Predicting postoperative complications after bariatric surgery: the Bariatric Surgery Index for Complications, BASIC. Surgical Endoscopy. 2017;31(11):4438–45.
Silecchia G, Iossa A. Complications of staple line and anastomoses following laparoscopic bariatric surgery. Ann Gastroenterol. 2017;31(1):56–64.
Lapatsanis D, Sidirokastritis G, Kontaxis V, et al. Laparoscopic total vertical gastric plication (LTVGP) and laparoscopic sleeve gastrectomy (LSG) as restrictive bariatric procedures. Results, comparison, cost effectiveness differences and similarities. Obes Surg. 2011;21:1010.
Ospanov OB, Sultanov EE. Laparoscopic sleeve gastroplication versus laparoscopic sleeve gastrectomy: preliminary results. Surg Endosc Interven Tech. 2013;27:S132.
Fardoun A. Gastric plication versus sleeve: 700 cases none randomized study. Obes Surg. 2014;24:1016–7.
Cesana G, Uccelli M, Ciccarese F, et al. Laparoscopic gastric plication versus sleeve gastrectomy, a randomized study. Obes Surg. 2013;23:1024–5.
Abou AHS, Zeineldin AA, Al KAF. Comparative study between laparoscopic sleeve gastrectomy and laparoscopic gastric plication: early results. Obes Surg. 2014;24:1139–40.
Ciemy M, Ciemy M, Kriz M, et al. Gastric plication versus gastric sleeve-a single center short-term comparison. Obes Surg. 2013;23:1024.
Cummings DE. Ghrelin and the short- and long-term regulation of appetite and body weight. Physiol Behav. 2006;89:71–84.
Kirchner H, Heppner KM, Tschop MH. The role of ghrelin in the control of energy balance. Handb Exp Pharmacol. 2012;209:161–84.
Guimarães M, Nora M, Ferreira T, et al. Sleeve gastrectomy and gastric plication in the rat result in weight loss with different endocrine profiles. Obes Surg. 2013;23(5):710–7.
Tschop M, Weyer C, Tataranni PA, et al. Circulating ghrelin levels are decreased in human obesity. Diabetes. 2001;50(4):707–9.
Raghay K, Akki R, Bensaid D, et al. Ghrelin as an anti-inflammatory and protective agent in ischemia/reperfusion injury. Peptides. 2020;124:170226.
WangY, Liu J. Plasma ghrelin modulation in gastric band operation and sleeve gastrectomy. Obes Surg. 2009;19:357–62.
Eleni S, George T, Konstantinos P, et al. Impact of laparoscopic sleeve gastrectomy on gastrointestinal motility. Gastroenterol Res Pract. 2018;4(5):1–17.
Karamanakos SN, Vagenas K, Kalfarentzos F, et al. Weight loss, appetite suppression, and changes in fasting and postprandial ghrelin and peptide-YY levels after Roux-en-Y gastric bypass and sleeve gastrectomy: a prospective, double blind study. Ann Surg. 2008;247(3):401–7.
Abdelbaki TN. An insight on the superior outcome of sleeve gastrectomy over gastric plication. Surg Obes Relat Dis. 2015;11:733–4.
Buchwald H, Avidar I, Brannwald E. Bariatric surgery: a systematic review and meta-analysis. JAMA. 292:1724–37.
Harvey EJ, Arroyo K, Korner J, et al. Hormone changes affecting energy homeostasis after metabolic surgery. Mt Sinai J Med. 2010;77(5):446–65.
Rogula T. Feasibility and early outcomes of laparoscopic plicated sleeve gastrectomy: a case-control study. Medical Studies. 2015;4:235–40.
Skrekas G, Antiochos K, Stafyla VK. Laparoscopic gastric greater curvature plication: results and complications in a series of 135patients. Obes Surg. 2011;21(11):1657–63.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical Approval
For this type of study, formal consent is not required.
Conflict of Interest
The authors declare no competing interests.
Informed Consent
Informed consent does not apply.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Key Points
• The advantages of LSG in terms of short- and mid-term weight loss were documented
• LSG has a lower rate of minor complications, but was less cost effective
• LSG and LGCP were similar regarding comorbidities improvement and major complications
• LSG and LGCP were similar in terms of operation duration and hospital stay
Rights and permissions
About this article
Cite this article
Li, H., Wang, J., Wang, W. et al. Comparison Between Laparoscopic Sleeve Gastrectomy and Laparoscopic Greater Curvature Plication Treatments for Obesity: an Updated Systematic Review and Meta-Analysis. OBES SURG 31, 4142–4158 (2021). https://doi.org/10.1007/s11695-021-05538-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-021-05538-z