Abstract
Background
Single-incision laparoscopic sleeve gastrectomy (SILSG) has been proposed as an alternative to conventional laparoscopic sleeve gastrectomy (CLSG) in obese patients. This study aims to compare the surgical outcomes of these two techniques.
Methods
A meta-analysis of existing literature obtained through a systematic literature search in the PubMed, EMBASE, and Cochrane Library CENTRAL databases from 2009 to 2019 was conducted.
Results
Eleven articles including 1168 patients were analyzed. Patients in the SILSG group reported greater satisfaction with cosmetic scar outcomes than those in the CLSG group (SMD = 2.47, 95% CI = 1.10 to 3.83, P = 0.00). There was no significant difference between the SILSG group and the CLSG group regarding operative time, intraoperative estimated blood loss, conversion rate, intraoperative complications, length of hospital stay, postoperative analgesia, postoperative complications, excess weight loss (EWL), and improvements in comorbidities (P > 0.05).
Conclusions
Compared to CLSG, SILSG resulted in improved cosmetic satisfaction and showed no disadvantages in terms of surgical outcomes; thus, SILSG can serve as an alternative to CLSG for obese patients. Nonetheless, high-quality randomized controlled trials (RCTs) with large study populations and long follow-up periods are needed.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In recent decades, the number of obese patients has increased rapidly, and the prevalence of obesity and related metabolic diseases has also increased substantially worldwide [1]. Bariatric surgery is currently recognized as the most effective method to treat obesity and related metabolic diseases; it can not only promote sustained and stable weight loss effects (WLEs) but also significantly alleviate obesity-related metabolic diseases [2]. As an independent bariatric surgery, laparoscopic sleeve gastrectomy was the most frequently performed bariatric procedure globally in 2014 [3]. Since the first case of single-incision laparoscopic sleeve gastrectomy (SILSG) was reported in 2008 [4], an increasing number of such cases have been reported. By 2018, 2028 patients had been reported to have undergone SILSG [5]. Compared with conventional laparoscopic sleeve gastrectomy (CLSG), SILSG has certain potential advantages, such as improved cosmesis and reduced postoperative pain [4,5,6]. In contrast, CLSG requires four or five skin incisions to place laparoscopic trocars for surgery [7, 8]. However, when a new technology emerges, the greatest concerns pertain to the safety of the procedure, and the single-incision surgical approach has been reported to be associated with higher incisional hernia rates [9]. To the best of our knowledge, no direct evidence supports EWL and improvements in comorbidities with the two types of approaches.
Based on the above findings, our study aimed to compare the surgical outcomes of CLSG and SILSG for the treatment of morbid obesity through a meta-analysis of existing literature obtained from the PubMed, EMBASE, and Cochrane Library CENTRAL databases from 2009 to 2019.
Methods
Literature Search
The meta-analysis was performed in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement [10]. A systematic literature search was conducted in the PubMed, Cochrane Library, and EMBASE databases to identify articles that compared CLSG with SILSG using the following terms in every possible combination: “single-incision,” “single-port,” “single-access,” “transumbilical,” “conventional,” “multiport,” “laparoscopic,” and “sleeve gastrectomy.” The latest search was conducted in September 2019.
Inclusion Criteria
The inclusion criteria of this study were as follows: (1) studies that compared CLSG and SILSG in obese patients, (2) studies published between 2009 and 2019, and (3) studies reporting original research in English. Conference abstracts, review articles, and clinical practice guidelines were excluded from this analysis. Two independent investigators (S.D.S. and P.P.W.) extracted data from the included studies. Any discrepancies between the two reviewers about the inclusion or exclusion of studies were discussed with the senior author until consensus was reached.
Data Extraction
Preoperative, intraoperative, and postoperative parameters were extracted from eleven included articles. The preoperative data included patient demographics, body mass index (BMI), and comorbidities. The intraoperative data included the single-incision devices, the mean operative time, estimated blood loss, the conversion rate, and intraoperative complications. Conversion to CLSG in the SILSG group and conversion to open surgery in either or both groups were considered conversion. The immediate postoperative data included the length of hospital stay, postoperative analgesia, and postoperative complications. The late postoperative data included the cosmetic outcome, EWL, and improvements in comorbidities. Two investigators performed data extraction and compared the validity of the data until consensus was reached.
Statistical Analysis
Statistical analysis was performed using Stata (version 15.1, StataCorp LP, TX, USA), and significance tests were two-sided at the 5% level. Dichotomous variables were analyzed using the random effects models (Mantel-Haenszel heterogeneity) and odds ratios (ORs) with 95% confidence intervals (CIs). ORs < 1 denoted outcomes that were more frequent in the SILSG group than in the CLSG group. If the values of the obtained continuous variables were mean- and median-cross-checked with maximum and minimum values, Hozo’s method was applied to estimate the mean and standard deviation (SD) [11]. Continuous variables were assessed using random effects models (inverse variance heterogeneity) and standard mean differences (SMDs) with 95% CIs. In cases where SMDs < 0, values in the SILSG group were increased. Heterogeneity among the included studies was examined using the I2 statistic. Statistical heterogeneity was evaluated using the I2 statistic. Statistical heterogeneity was evaluated using methods described by Higgins et al. [12]. I2 values of 0% to 25%, 25% to 75%, and > 75% were considered to indicate low, moderate, and high heterogeneity, respectively.
Results
Overview of Studies
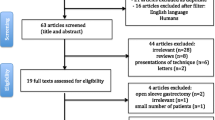
The flow diagram of the systematic literature search is shown in Fig. 1. In total, eleven publications including 1168 patients were analyzed [13,14,15,16,17,18,19,20,21,22,23]; one was a randomized controlled trial [16], three were prospective cohort studies [13, 15, 20], and seven were retrospective cohort studies [14, 17,18,19, 21,22,23] (Tables 1, 2, and 3). The included studies were from Austria, Italy, India, Spain, and the USA. In total, 1168 patients were included, of which 581 underwent CLSG and 587 underwent SILSG.
Intraoperative Data
Single-Incision Devices
The single-incision devices used in the included studies are listed in Table 1. The Covidien SILS™ Port (Covidien Surgical, Mansfield, MA) was the most frequently used device, which was used in seven studies [13, 15, 16, 19,20,21,22], while the LESS Triport/Quadport (Olympus Medical, Nagano, Japan) was used in only two studies [13, 14].
Operative Time
The mean operative time was reported in all eleven articles. No significant difference was observed (SMD = 0.27, 95% CI = − 0.01 to 0.55, P = 0.06) (Fig. 2a). The heterogeneity was moderate (I2 73.7%). A sensitivity analysis was performed and indicated that the study conducted by Delgado et al. [13] was responsible for the inconsistency. After removing this publication from the analysis, heterogeneity decreased to 60.4% (moderate heterogeneity), and the results remained unchanged (SMD = 0.15, 95% CI = − 0.09 to 0.38, P = 0.21).
Estimated Blood Loss
Estimated blood loss was reported in five of the eleven articles [16,17,18, 21, 22]. No significant difference was observed (SMD = − 0.59, 95% CI = − 1.36 to 0.17, P = .13) (Fig. 2b). The heterogeneity was high (I2 84.9%). A sensitivity analysis was performed and indicated that the study conducted by Saber et al. [22] was responsible for the inconsistency. After removing this publication from the analysis, the heterogeneity decreased to 62.3% (moderate heterogeneity), and the results remained unchanged (SMD = − 0.18, 95% CI = − 0.68 to 0.31, P = 0.47).
Conversion Rate
Three [13, 18, 19] of the eleven articles reported conversion to CLSG in the SILSG group. No conversion to open surgery was observed in the two groups. However, there was no significant difference (OR = 0.46, 95% CI = 0.07 to 3.02, P = 0.42). No heterogeneity was observed (I2 0%).
Intraoperative Complications
Intraoperative complications were reported in four [18,19,20,21] of the eleven articles, and they all occurred in the SILSG groups. Stapler line failure was reported in three studies [18,19,20]. Nguyen et al. [18] reported liver laceration, and Rogula et al. [21] reported air leakage. No significant difference was observed (OR = 0.32, 95% CI = 0.06 to 1.59, P = 0.16), and no heterogeneity was observed (I2 0%). The mortality was 0 in four [18,19,20,21] of the eleven articles.
Immediate Postoperative Data
Length of Hospital Stay
Length of hospital stay was reported in seven [14, 18,19,20,21,22,23] of the eleven articles, and no significant difference was observed (SMD = − 0.25, 95% CI = − 0.74 to 0.23, P = 0.31). The heterogeneity was high (I2 80.3%). A sensitivity analysis was performed and indicated that the study conducted by Nguyen et al. [18] was responsible for the inconsistency. After removing this publication from the analysis, the heterogeneity decreased to 70.3% (moderate heterogeneity), and the results remained unchanged (SMD = − 0.43, 95% CI = − 0.86 to 0.01, P = 0.05).
Postoperative Analgesia
Postoperative analgesia was reported in seven [15,16,17, 19,20,21,22] of the eleven articles. Pain visual analog scale (VAS) ratings at postoperative day (POD) 1, pain VAS ratings at POD 2, and doses of analgesics were used to measure postoperative analgesia more frequently than other indicators. Therefore, we decided to include these three indicators in the analysis (Fig. 3). The pain VAS rating at POD 1 was approximately the same in the CLSG and SILSG groups (SMD = − 0.39, 95% CI = − 1.15 to 0.37, P = 0.31), with high heterogeneity (I2 83.5%) (Fig. 3a). A sensitivity analysis was performed and indicated that the study conducted by Morales et al. [16] was responsible for the inconsistency. After removing this publication from the analysis, the heterogeneity decreased to 72.2% (moderate heterogeneity), and the results remained unchanged (SMD = − 0.08, 95% CI = − 0.71 to 0.56, P = 0.81). The pain VAS rating at POD 2 also appeared to exhibit a nonsignificant difference between the CLSG and SILSG groups (SMD = − 0.63, 95% CI = − 1.38 to 0.12, P = 0.10), with high heterogeneity (I2 79.4%) (Fig. 3b). A sensitivity analysis was performed and indicated that the study conducted by Morales et al. [16] was responsible for the inconsistency. After removing this publication from the analysis, the heterogeneity decreased to 0% (no heterogeneity), and the results remained unchanged (SMD = − 0.20, 95% CI = − 0.50 to 0.11, P = 0.21). There was no difference in the dose of analgesics between the two groups (SMD = 0.02, 95% CI = − 0.29 to 0.34, P = 0.88), with no heterogeneity (I2 0%) (Fig. 3c).
Postoperative Complications
Postoperative complications were reported in nine [13,14,15, 17,18,19,20,21, 23] of the eleven articles. Bleeding, wound infection, leaks, and incisional hernias were the major complications (Fig. 4). Bleeding was reported in five articles [13, 14, 16, 19, 23], and no significant difference was observed (OR = 1.18, 95% CI = 0.32 to 4.31, P = 0.81) (Fig. 4a). The risk of wound infection was also comparable between the two groups in four articles (OR = 0.51, 95% CI = 0.10 to 2.50, P = 0.41) [15, 17, 18, 20] (Fig. 4b). The incidence of leaks was comparable between the two groups (OR = 0.43, 95% CI = 0.06 to 2.98, P = 0.40) according to Lakdawala et al. [15] and Sucher et al. [23] (Fig. 4c). Only one study [15] reported a delayed incisional hernia at the trocar site in 3 patients (1%) in the SILSG group, and no incisional hernias were reported for the CLSG group. Moreover, reoperations were reported in five studies [13, 14, 17, 20, 23], and no significant difference was observed (OR = 0.81, 95% CI = 0.23 to 2.88, P = 0.74) (Fig. 4d). No heterogeneity was observed (I2 0%) for these four outcomes. One patient in the SILSG group developed a pulmonary embolus and recovered [19]. The mortality was 0 in all the articles.
Late Postoperative Data
Cosmetic Outcome
Cosmetic outcome was reported in four [15, 20, 21, 23] of the eleven articles. A cosmetic satisfaction scale was used in Porta et al. [20], Rogula et al. [21], and Sucher et al. [23], and outcome satisfaction was significantly higher in SILSG patients than in CLSG patients (SMD = 2.47, 95% CI = 1.10 to 3.83, P = 0.00), with very high heterogeneity (I2 92.8%) (Fig. 5a). According to a questionnaire by Lakdawala et al. [15], patients in the SILSG group reported greater satisfaction with their cosmetic outcome than those in the CLSG group. Rogula et al. reported that cosmetic satisfaction was high in the SILSG group (all patients scored 3/3) [21].
Excess Weight Loss
EWL was reported in all eleven articles. Only three articles [14,15,16] reported the mean and SD of the percentage of EWL (EWL%) at 6 months postoperatively, and the rest reported EWL or EWL% at different follow-up intervals. Therefore, we decided to include only these three articles in the analysis. No significant difference was observed (SMD = 0.06, 95% CI = − 0.09 to 0.20, P = 0.46) (Fig. 5b), and there was no heterogeneity (I2 0%) (Fig. 5b).
Improvements in Comorbidities
Improvements in comorbidities were reported in three [15, 20, 22] of the eleven articles. Diabetes mellitus, hypertension, dyslipidemia, and sleep apnea were the major comorbidities that were improved. However, there were no significant differences between the two groups. Sucher et al. [23] found no improvements in comorbidities.
Discussion
This meta-analysis identified eleven articles that compared CLSG with SILSG, measured patient outcomes, and was published from 2010 to 2017. The preoperative data were not different between these two groups. However, there were trends indicative of younger ages and lower BMIs in the SILSG groups than in the CLSG groups, which is potentially problematic in interpreting these results. To the best of our knowledge, this was the first meta-analysis to compare the estimated blood loss, EWL, and improvements in comorbidities associated with SILSG and CLSG in obese patients.
The main principle of SILSG is to perform the entire surgery through a single incision using a multiport device, while the greatest limitation is triangulation of the instruments and difficulties related to the parallel vision of the camera [24, 25]. Either a single-incision instrument or multiple trocars operated through a single incision can be used [26]. The most frequently used device in the included studies was the Covidien SILS™ Port. However, Muir et al. used the SPIDER™ device (TransEnterix Surgical, Inc., Durham, NC) without the loss of triangulation in the restricted space of the abdominal cavity, which offers true instrument triangulation and simulation of CLSG [17]. Therefore, SILSG can be a feasible and safe alternative to CLSG for obese patients.
With the restricted operating space due to a single incision, which increases the difficulty for the operator, the operative time may potentially be longer in the SILSG group. However, operative time was not significantly different between two groups. A sensitivity analysis indicating moderate heterogeneity was conducted by Delgado et al., who reported that the operative time was significantly increased in the first 10 cases due to the learning curve, probably due to the critical points of SILSG [13]. Another factor contributing to the high heterogeneity was the vast range of operative times among these studies, which included operations involving different single-port devices and surgical procedures.
Our study is the first meta-analysis to compare CLSG with SILSG to evaluate the outcome of estimated blood loss. In our analysis, the estimated blood loss was similar between the two groups, suggesting that SILSG can be safely performed in obese patients. The study by Saber et al. [22] was responsible for the high heterogeneity as they reported an obvious reduction in the estimated blood loss in the SILSG group compared with that in the CLSG group, although they did not mention the significance. It was believed that the surgeon was able to avoid dangerous zones of epigastric vessels by staying in the safe umbilical zone, further reducing the risk of trauma to abdominal wall blood vessels [27].
In previous studies, the conversion rate in the CLSG group was 1.96% [28] and that in the SILSG group was very low as well [29,29,30,32]. There was no conversion to open surgery in the two groups and three conversions to CLSG in the SILSG group in our study. Intraoperative complications and the length of hospitalization were comparable between the CLSG and SILSG groups, suggesting that SILSG can be a safe technique for obese patients.
Interestingly, SILSG was considered to cause less postoperative pain than CLSG in some included studies [15, 17, 19], although the result was not significantly different between the two groups in our study. A similar observation was reported between single-incision and conventional laparoscopic cholecystectomy [33]. Although previous studies recommended that postoperative analgesics can be expressed in morphine equivalents and paracetamol usage, these values were reported in only one article that found similar postoperative analgesic use in the two groups [20, 34, 35]. Notably, the provided data were very heterogeneous regarding the methods assessing pain and administered analgesics, and a certain publication bias exists.
Regarding postoperative complications, there was no significant difference between these two groups. Notably, incisional hernias are a substantial concern with the single-incision surgical approach as one study reported incisional hernia rates as high as 5.8% for single-incision laparoscopic cholecystectomy [9], but the rate was only 1% in our study. We believe that incisional hernia rates for single-incision surgery can be reduced by allocating appropriate attention to prevention measures during surgery and ensuring proper closure of the sheath. However, more RCTs with larger populations are needed to assess incisional hernias and other events. Unlike a previous study that found a mortality rate of 0.1% (n = 1 patient) in the SILSG group due to pulmonary embolism on POD 10 [36], the mortality was 0 in all eleven articles included in our meta-analysis.
Improvement in the cosmetic outcome was a prominent advantage of SILSG [37]. We found that cosmetic scar satisfaction was significantly higher in the SILSG group than that in the CLSG group, with very high heterogeneity. This very high heterogeneity was likely due to different definitions of the cosmetic satisfaction scale. The scale ranged from 1 to 4 (1 = poor; 2 = acceptable; 3 = good; 4 = excellent) in Porta et al. [20], while it ranged from 1 to 5 (1 = excellent; 2 = good; 3 = fair; 4 = acceptable; 5 = poor) in Sucher et al. [23]. When the single incision was a 2.5-cm umbilical incision, the specimen was extracted from the solitary port without extending the umbilical incision in the SILSG group, while the trocar incision located in the right upper quadrant required extension in the CLSG group. Furthermore, the site of the incision buried in the umbilicus yields an almost scarless surgery in the SILSG group. When given a choice, most patients select the scarless approach over the conventional laparoscopic approach as observed among many female patients in our study. Moreover, according to Saber et al. [22] and Porta et al. [20], a 2.5-cm incision is needed to extract the gastric specimen, making the single-incision approach ideal for this particular procedure, negating the claims that SILSG is a step back in minimally invasive surgery toward minilaparotomy. Therefore, due to greater patient satisfaction with cosmesis, SILSG can be an alternative to CLSG for obese patients.
To the best of our knowledge, our meta-analysis is also the first to compare EWL associated with CLSG and SILSG. We found no significant difference between the two groups regarding EWL. However, 6 months postoperatively may not be enough time to judge the effectiveness of obesity surgery [38]. Improvements in comorbidities were similar between the SILSG and CLSG group as well. A plethora of factors contribute to improvements in comorbidities after sleeve gastrectomy, including gastric restriction and related neurohumoral changes [39]. The nonsignificant difference between SILSG and CLSG suggests that these factors are likely to help improve comorbidities more or less equally across the two groups.
Our meta-analysis had two limitations. First, our study included only one randomized controlled trial (RCT), and the remaining ten articles were likely subject to bias. Second, we restricted our analysis to those studies that only compared CLSG with SILSG, which limited the sample size. In the future, if more studies are available, particularly studies with RCT designs, large sample sizes, and long follow-up periods, a new meta-analysis without these limitations can be conducted.
Conclusion
This meta-analysis identified eleven studies that compared CLSG with SILSG for the treatment of obese patients. Compared to CLSG, SILSG resulted in improved cosmetic satisfaction and showed no disadvantages in terms of other surgical outcomes; thus, SILSG can be an alternative to CLSG for obese patients. Nonetheless, high-quality RCTs with large study populations and long follow-up periods are needed.
References
Collaboration NCDRF. Trends in adult body-mass index in 200 countries from 1975 to 2014: a pooled analysis of 1698 population-based measurement studies with 19.2 million participants. Lancet. 2016;387(10026):1377–96.
Mingrone G, Panunzi S, De Gaetano A, et al. Bariatric-metabolic surgery versus conventional medical treatment in obese patients with type 2 diabetes: 5 year follow-up of an open-label, single-centre, randomised controlled trial. Lancet. 2015;386(9997):964–73.
Angrisani L. 2014: the year of the sleeve supremacy. Obes Surg. 2017;27(6):1626–7.
Saber AA, Elgamal MH, Itawi EA, et al. Single incision laparoscopic sleeve gastrectomy (SILSG): a novel technique. Obes Surg. 2008;18(10):1338–42.
Magouliotis DE, Tasiopoulou VS, Svokos AA, et al. Single incision versus conventional laparoscopic sleeve gastrectomy for morbid obesity: a meta-analysis. J Laparoendosc Adv Surg Tech A. 2018;28(6):690–9.
Dimitrokallis N, Alexandrou A, Schizas D, et al. Single-incision laparoscopic sleeve gastrectomy: review and a critical appraisal. J Laparoendosc Adv Surg Tech A. 2017;27(3):217–26.
Mognol P, Chosidow D, Marmuse JP. Laparoscopic sleeve gastrectomy as an initial bariatric operation for high-risk patients: initial results in 10 patients. Obes Surg. 2005;15(7):1030–3.
Roa PE, Kaidar-Person O, Pinto D, et al. Laparoscopic sleeve gastrectomy as treatment for morbid obesity: technique and short-term outcome. Obes Surg. 2006;16(10):1323–6.
Alptekin H, Yilmaz H, Acar F, et al. Incisional hernia rate may increase after single-port cholecystectomy. J Laparoendosc Adv Surg Tech A. 2012;22(8):731–7.
Moher D, Liberati A, Tetzlaff J, et al. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. J Clin Epidemiol. 2009;62(10):1006–12.
Hozo SP, Djulbegovic B, Hozo I. Estimating the mean and variance from the median, range, and the size of a sample. BMC Med Res Methodol. 2005;20(5):13.
Higgins JP, Thompson SG, Deeks JJ, et al. Measuring inconsistency in meta-analyses. BMJ. 2003;327(7414):557–60.
Delgado S, Ibarzabal A, Adelsdorfer C, et al. Transumbilical single-port sleeve gastrectomy: initial experience and comparative study. Surg Endosc. 2012;26(5):1247–53.
Gomberawalla A, Salamat A, Lutfi R. Outcome analysis of single incision vs traditional multiport sleeve gastrectomy: a matched cohort study. Obes Surg. 2014;24(11):1870–4.
Lakdawala M, Agarwal A, Dhar S, et al. Single-incision sleeve gastrectomy versus laparoscopic sleeve gastrectomy. a 2-year comparative analysis of 600 patients. Obes Surg. 2015;25(4):607–14.
Morales-Conde S, Del Agua IA, Moreno AB, et al. Postoperative pain after conventional laparoscopic versus single-port sleeve gastrectomy: a prospective, randomized, controlled pilot study. Surg Obes Relat Dis. 2017;13(4):608–13.
Muir KB, Rice WV. Weight-loss outcomes of SPIDER((R)) sleeve gastrectomy at 6 months compared to traditional laparoscopic technique. Surg Endosc. 2016;30(5):2043–8.
Nguyen NT, Smith BR, Reavis KM, et al. Strategic laparoscopic surgery for improved cosmesis in general and bariatric surgery: analysis of initial 127 cases. J Laparoendosc Adv Surg Tech A. 2012;22(4):355–61.
Park K, Afthinos JN, Lee D, et al. Single port sleeve gastrectomy: strategic use of technology to re-establish fundamental tenets of multiport laparoscopy. Surg Obes Relat Dis. 2012;8(4):450–7.
Porta A, Aiolfi A, Musolino C, et al. Prospective comparison and quality of life for single-incision and conventional laparoscopic sleeve gastrectomy in a series of morbidly obese patients. Obes Surg. 2017;27(3):681–7.
Rogula T, Daigle C, Dua M, et al. Laparoscopic bariatric surgery can be performed through a single incision: a comparative study. Obes Surg. 2014;24(7):1102–8.
Saber AA, El-Ghazaly TH, Dewoolkar AV, et al. Single-incision laparoscopic sleeve gastrectomy versus conventional multiport laparoscopic sleeve gastrectomy: technical considerations and strategic modifications. Surg Obes Relat Dis. 2010;6(6):658–64.
Sucher R, Resch T, Mohr E, et al. Single-incision laparoscopic sleeve gastrectomy versus multiport laparoscopic sleeve gastrectomy: analysis of 80 cases in a single center. J Laparoendosc Adv Surg Tech A. 2014;24(2):83–8.
Zhu JF. Which term is better: SILS, SPA, LESS, E-NOTES, or TUES? Surg Endosc. 2009;23(5):1164–5.
Ponsky JL, Rosen MJ, Poulose BK. NOTES: of caution. Surg Endosc. 2008;22(7):1561–2.
Moreno-Sanz C, Morandeira-Rivas A, Sedano-Vizcaino C, et al. Single-incision laparoscopic bariatric surgery: a systematic review. Surg Obes Relat Dis. 2015;11:248–58.
Saber AA, Meslemani AM, Davis R, et al. Safety zones for anterior abdominal wall entry during laparoscopy: a CT scan mapping of epigastric vessels. Ann Surg. 2004;239:182.
Zacharoulis D, Sioka E, Papamargaritis D, et al. Influence of the learning curve on safety and efficiency of laparoscopic sleeve gastrectomy. Obes Surg. 2012;22(3):411–5.
Pourcher G, Di Giuro G, Lafosse T, et al. Routine single-port sleeve gastrectomy: a study of 60 consecutive patients. Surg Obes Relat Dis. 2013;9(3):385–9.
Pourcher G, De Filippo G, Ferretti S, et al. Short-term results of single-port sleeve gastrectomy in adolescents with severe obesity. Surg Obes Relat Dis. 2015;11(1):65–9.
Farias C, Fernandez JI, Ovalle C, et al. Transumbilical sleeve gastrectomy with an accessory lateral port: surgical results in 237 patients and 1-year follow-up. Obes Surg. 2013;23(3):325–31.
Maluenda F, Leon J, Csendes A, et al. Single-incision laparoscopic sleeve gastrectomy: initial experience in 20 patients and 2-year follow-up. Eur Surg. 2014;46:32–7.
Lai EC, Yang GP, Tang CN, et al. Prospective randomized comparative study of single incision laparoscopic cholecystectomy versus conventional four-port laparoscopic cholecystectomy. Am J Surg. 2011;202(3):254–8.
O'Hara DA, Fragen RJ, Kinzer M, et al. Ketorolac tromethamine as compared with morphine sulfate for treatment of postoperative pain. Clin Pharmacol Ther. 1987;41(5):556–61.
Gordon DB, Stevenson KK, Griffie J, et al. Opioid equianalgesic calculations. J Palliat Med. 1999;2(2):209–18.
Gaillard M, Tranchart H, Lainas P, et al. Single-port laparoscopic sleeve gastrectomy as a routine procedure in 1000 patients. Surg Obes Relat Dis. 2016;12(7):1270–7.
Fernandez JI, Farias CO, Ovalle CL, et al. Transumbilical single-incision laparoscopic sleeve gastrectomy. Obes Surg. 2015;25(3):430–5.
O'Kane M, Parretti HM, Hughes CA, et al. Guidelines for the follow-up of patients undergoing bariatric surgery. Clin Obes. 2016;6(3):210–24.
Clinical Issues Committee of the American Society for M, Bariatric S. Updated position statement on sleeve gastrectomy as a bariatric procedure. Surg Obes Relat Dis. 2010;6(1):1–5.
Funding
This work was supported by research grants from the Startup Fund for Scientific Research of Fujian Medical University (2018QH1068).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflicts of interest.
Ethical Approval
This article does not contain any studies involving human participants or animals performed by any of the authors.
Statement Informed Consent
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sun, SD., Wu, PP., Zhou, JF. et al. Comparison of Single-Incision and Conventional Laparoscopic Sleeve Gastrectomy for Morbid Obesity: a Meta-Analysis. OBES SURG 30, 2186–2198 (2020). https://doi.org/10.1007/s11695-020-04437-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-020-04437-z