Abstract
Purpose
Obesity is associated with increased morbidity and mortality. Weight loss due to gastric bypass (GBP) surgery improves clinical outcomes and may be a cost-effective intervention. To estimate the cost-effectiveness of GBP compared to clinical treatment in severely obese individuals with and without diabetes in the perspective of the Brazilian public health system.
Materials and Methods
A Markov model was developed to compare costs and outcomes of gastric bypass in an open approach to clinical treatment. Health states were living with diabetes, remission of diabetes, non-fatal and fatal myocardial infarction, and death. We also included the occurrence of complications related to surgery and plastic surgery after the gastric bypass surgery. The direct costs were obtained from primary data collection performed in three public reference centers for obesity treatment. Utility values also derived from this cohort, while transition probabilities came from the international literature. A sensitivity analysis was performed to evaluate uncertainties. The model considered a 10-year time horizon and a 5% discount rate.
Results
Over 10 years, GBP increased quality-adjusted life years (QALY) and costs compared to clinical treatment, resulting in an incremental cost-effectiveness ratio (ICER) of Int$1820.17/QALY and Int$1937.73/QALY in individuals with and without diabetes, respectively. Sensitivity analysis showed that utility values and direct costs of treatments were the parameters that affected the most the ICERs.
Conclusion
The study demonstrated that GBP is a cost-effective intervention for severely obese individuals in the Brazilian public health system perspective, with a better result in individuals with diabetes.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The prevalence of obesity reaches global epidemic levels. In 2014, more than 640 million individuals were obese worldwide and almost 180 millions of these were severely obese. If these trends continue, by 2025 the global prevalence will reach 18% in men and more than 21% in women [1]. In Brazil, obesity prevalence increased by 7.5% between 2006 and 2016, reaching 53.8% and 18.9% of overweight and obesity, respectively [2].
Obesity and its comorbidities represent a very important public health problem worldwide regarding clinical and economic consequences. The impact in the global economy was estimated at US$2 trillion or 2.8% of the global gross domestic product (GDP) in 2014 [3]. In the perspective of the Brazilian public health system, the annual costs of obesity and associated diseases were estimated to be US$2.1 billion in 2010 [4].
In the Brazilian public health system (SUS), the number of Bariatric surgeries (BS) increased by 45% between 2010 and 2013, and BS health expenditures by 56%, totaling Int$20.5 million in 2013 [5]. At this time, only laparotomy surgeries were performed in the SUS [6]. In 2013, an overview of global estimates for BS placed Brazil in the second position in the number of surgeries performed per year (n = 86,840) [7]. Although there is a huge discrepancy between the number of individuals operated in the public and in the private systems, this large number of surgeries allows improvements in the learning curve and, consequently, better results.
Evidence indicates that bariatric surgery is the most effective intervention for the treatment of obesity. In several studies, the weight loss difference between the surgical and clinical groups was approximately 20%, besides allowing the control and/or remission of diseases associated with obesity, such as hypertension, type 2 diabetes mellitus, and cardiovascular diseases [8,9,10,11,12,13,14,15,16]. Furthermore, bariatric surgery has also been proven as a cost-effective treatment for obesity and its complications [17,18,19]. It has been suggested that patients with diabetes will present even better health outcomes compared with patients without diabetes [20, 21]. Given the current discussion about obesity management, surgical options, and its inclusion in diabetes treatment algorithms [22, 23], possible differences in patients’ baseline status are worth noting.
Since data on costs of severe obesity and bariatric surgery are still limited, the objective of this study was to develop a cost-utility model comparing the GBP surgical treatment of severely obese individuals with and without diabetes to clinical treatment.
Material and Methods
We carried out a cost-utility analysis of gastric bypass (GBP) surgery compared to clinical treatment for severely obese patients in the perspective of the public health system, taking into consideration a 10-year time horizon. We designed a Markov model for two target populations: (i) severely obese with diabetes and (ii) severely obese without diabetes. On this basis, the main questions explored were whether GBP surgery would be more cost-effective compared to clinical treatment, and if so, which target population would beneficiate the most. Our study complied with national [24] and international [25, 26] guidelines for economic evaluation. We also used the CHEERS checklist for reporting our cost-utility analysis [27].
Cost and Utility Data
We conducted a multicentric, cross-sectional study assessing health-related quality of life and costs of treatment of severely obese patients. Severe obesity was defined as BMI greater than 40 kg/m2 or greater than 35 kg/m2 associated with one or more obesity-related comorbidities [28]. The population was recruited in three reference hospitals, authorized by the Ministry of Health for the treatment of obesity. The patients who underwent gastric bypass surgery were selected in Hospital São Lucas (HSL) of the Pontifícia Universidade Católica do Rio Grande do Sul (PUC/RS). Patients undergoing clinical treatment were selected in Policlínica Piquet Carneiro of the Universidade do Estado do Rio de Janeiro (UERJ) and at the Instituto Estadual de Diabetes e Endocrinologia Luiz Capriglione (IEDE) in Rio de Janeiro.
We developed a standard questionnaire for collecting information on costs, including direct medical costs such as health professionals’ visits, laboratory tests, medications, hospital admissions, treatment of surgical complications, and the bariatric surgery. Non-medical direct costs (patient transportation, caregiver payment) and indirect costs (absenteeism) were included. The complete description of the micro-costing approach was previously published elsewhere [29].
For health-related quality of life, we used the Portuguese version of the EuroQol questionnaire (EQ-5D-3L), which was validated in Brazil in 2016 [30]. The EQ-5D-3L instrument was applied in 140 individuals with severe obesity submitted to gastric bypass (GBP) in open approach and 134 individuals undergoing clinical treatment. Utility values were generated based on the Brazilian value set for EQ-5D-3L results [30]. Subsequently, utility values were used to estimate the change in quality-adjusted life years (QALYs).
Comparators
Bariatric surgery strategy considered only patients who underwent the technique of Roux-en-Y gastric bypass by laparotomy, because the laparoscopic approach was not performed at public centers at the moment the study was carried out. Patients were grouped according to the time since surgery (up to 1 year, 1–2 years, 2–3 years, and > 3 years).
Clinical treatment is defined for the most updated guideline from the Brazilian Ministry of Health, which includes effective weight reduction (changes in lifestyle through diet and exercise education), public health strategies for prevention and health promotion, multidisciplinary assistance in primary health care services, nutritional follow-up, and referral for specialized assistance while waiting for surgical treatment when medically recommended [31].
Time Horizon, Discount Rate, Currency, Price Date, and Conversion
We adopted a 10-year-period horizon, considering the duration of the main randomized clinical trials for the treatment of severe obesity with the outcomes of interest. We used a 5% discount rate, which is recommended by the methodological guidelines published by the Brazilian Ministry of Health [24]. Costs were obtained in local currency (R$) related to 2015 prices, and converted to international dollars (Int$) using the purchasing power parity (PPP) of 1 Int$ = R$1.859 (World Bank, 2015) [32].
The Analytical Model
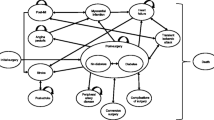
We developed a hybrid Markov decision tree model to compare the gastric bypass in open approach with the clinical treatment for severely obese individuals with and without diabetes. Figure 1 shows the rationale for each model. The health states considered into the models were living with diabetes, remission of diabetes, non-fatal and fatal myocardial infarction, and death for all causes. For the GBP arm, we included also the occurrence of complications related to surgery in the first year and the possibility to perform plastic surgery after the GBP. We included 10 Markov cycles of 1 year each, totaling 10-year time horizon.
The model was constructed using TreeAge software 2017 (Williamstown, MA, USA) and Microsoft Excel 2013 (Microsoft Corp., Redmond, WA, USA).
Clinical Effectiveness and Safety Data
The outcomes considered in the model were diabetes mellitus, fatal and non-fatal myocardial infarction, and death. We obtained transition probabilities from international literature [8, 9, 11,12,13,14] with the exception of mortality data that came from national registries [15, 16].
The most common postoperative surgical complications (cholecystectomy and hernioplasties), occurring in the first year post surgery, were included in the model using data from medical records of patients coming from a major center for surgical treatment of obesity in the public health system [6, 33].
Based on the total number of plastic surgeries performed in 1 year in the public health system after GBP surgeries (n = 1065) and the total number of GBP surgeries in 2014 (n = 7003), we assumed a proportion of individuals of 15.2% who would undergo at least one plastic surgery in 10 years (mammoplasty and abdominal, crural, and brachial dermolipectomy) [34].
Resource Utilization and Cost Data
Annual health resource utilization was obtained from the previous study with primary data collection of obese individuals undergoing clinical or surgical treatment [29].
The cost of GBP surgery was defined as Int$3409.08 (R$6337.48) and was obtained from the official reimbursement table of procedures of the Brazilian Public Health System for 2015 [35]. The costs of the four most common types of plastic surgery performed after bariatric surgery were also obtained from the official reimbursement table, and the weighted average value was considered for the model Int$498.46 (R$926.65) [35]. The average hospitalization cost of myocardial infarction was estimated at Int$2155.99 (R$4008) [36]. Table 1 presents the key parameters used in the models.
Analysis
The analysis was performed from the public payer perspective over a 10-year time horizon, and a 5.0% discount rate was applied to all variables considering Brazilian recommendations [24]. The incremental cost-effectiveness ratio (ICER) was calculated by dividing the difference in total costs (incremental cost) by the difference in quality-adjusted life years (QALY) (incremental effect) among group comparisons [24]. The willingness-to-pay threshold related to per capita GDP/QALY (GDP Brazil 2015 = Int$15,565.26) was assumed to identify the intervention as cost-effective [37].
Sensitivity Analysis
In order to evaluate the impact of the uncertainties of the variables included in the model, we performed both deterministic and probabilistic sensitivity analyses. For deterministic analysis, variability arbitrated was around the central measure of 25% for cost, utility, and probabilities in accordance with the recommendations of the Brazilian Economic Analysis Guidelines of the Ministry of Health [24]. For the probabilistic analysis, the Tornado diagram selected 10,000 Monte Carlo simulations with the most important variables. Gamma distribution was applied to costs, and beta distribution was applied to probabilities and utilities.
Results
Gastric bypass surgery was associated with higher costs (Int$10,481.25 (R$19,484.66)) and an additional 1.26 QALY, which resulted in an ICER of Int$1937.99 per QALY (R$3602.73) when compared to clinical treatment in the non-diabetic cohort.
In the diabetic cohort, an incremental cost of Int$3432.43 (R$6381.21) and an incremental effectiveness of 1.88 QALY were obtained, resulting in an ICER of Int$1820.17 per QALY (R$3383.70) for surgical treatment compared to clinical treatment at 10 years. Table 2 represents the results of the cost-effectiveness analysis.
Considering the willingness-to-pay (WTP) threshold related to per capita GDP, GBP surgery was a cost-effective intervention with ICER below the World Health Organization (WHO) threshold value of one GDP [38]. ICER in the diabetic group and non-diabetic group were 12.45% and 11.69% of per capita GDP, respectively.
Sensitivity Analysis
A base case scenario for deterministic one-way sensitivity analysis showed that utility values and direct costs of treatments were the parameters that affected the most the results. When taking into consideration the lower and upper limits of QALY, the ICER ranges from In$1938 to In$19,848.67 (Fig. 2).
Probabilistic sensitivity analysis indicated that GBP surgery was a cost-effective intervention in 99.5% of cases in patients with diabetes and in 93.4% of the patients without diabetes. (Figs. 3 and 4).
Conclusion
To the best of our knowledge, our study is the first study to investigate the cost-effectiveness of gastric bypass surgery in patients with severe obesity, with and without diabetes mellitus in the perspective of the Brazilian public health system. The results clearly showed that GBP surgery is a good value for money compared to clinical treatment, improving clinical outcomes at an affordable cost.
In our analysis, we developed a hybrid Markov model to evaluate the economic impact of GBP surgery in the context of the Brazilian Health Care System. Based on primary data collection at reference centers for obesity, this study especially estimated direct and indirect costs and utility data. Although based on only three reference centers, we performed a micro-cost analysis, considering the local setting. All utility and cost inputs used were obtained from the Brazilian population’s data. Bariatric surgery clearly reduces the risk of the obesity-related adverse events significantly at 10 years, especially in a young and severe obese population with diabetes, as demonstrated in the present analysis [8, 9, 11,12,13,14,15,16].
Several studies have already shown that BS is a cost-effective intervention compared to clinical treatment, with significant gains in quality of life, in addition to future economic benefits despite a higher initial expenditure with the surgery [17,18,19, 39,40,41,42]. The results of our study are similar to those previously published studies in European countries [40,41,42] and in the USA [43]. Another interesting issue is that only two studies explicitly considered the difference between individuals with and without diabetes [17, 18]. In this sense, our study brings new contributions to the international literature, since our results indicate that GBP surgery is more cost-effective in patients who already had diabetes at the time of surgical procedure.
It is worth noting the inclusion of plastic surgery costs in our model. It is the only study to show that surgery is cost-effective even including four types of plastic surgery (mammoplasty and abdominal, crural, and brachial dermolipectomy). Just one study assumed that only abdominoplasty was performed during the third year after bariatric surgery [41].
Considering distinctive features like health systems’ structure and financing, outcomes assessment, and the type of surgery, comparisons should be made cautiously. Gastric bypass in open approach is reimbursed by SUS since 1999 [44], but only in 2017, the laparoscopic approach was incorporated by the public health system [45], an intervention that is mainly performed in high-income countries [43]. Another point is the cost-effectiveness threshold adopted by different countries. Although the Brazilian government does not have an established cost-effectiveness threshold to consider a cost-effective health intervention, the estimated incremental cost-effectiveness ratios of this analysis are well below those suggested by the World Health Organization for interventions for low- and middle-income countries [38].
Obesity is a growing public health problem in Latin America, and around 58% of the inhabitants of the region are overweight [46]. During 2013, Latin America accounted for 30.5% of total procedures performed in four different regions in the world (USA/Canada, Europe, Latin/South America, and Asia/Pacific) [7]. Brazil is the country with the highest number of procedures in the region. Cost-effectiveness models are tools to decision-making and can further help to tackle with problems like obesity. Nevertheless, thus far, only developed countries have presented cost-effectiveness models of bariatric surgery for severe obesity [43].
It is worth mentioning the association between surgical skills and better or adverse outcomes after bariatric surgery. Hereupon, we have to consider the direct influence of the surgeon learning curve on operative outcomes [47]. In Brazil, high-complexity services are offered in a standardized manner, only in registered hospitals that develop specialized diagnostic and therapeutic support [48]. The results presented might be related to the years of experience and the growing number of bariatric surgeries performed by Brazilian specialized centers [5, 49].
Our study has some limitations. Every decision analytic model is a simplification of a health care system. The current surgical results may differ from the international study that provided the major outcomes inputs [8, 9, 11,12,13]. Compared to the private health care system, our conservative approach to estimating health care resources, such as lower medication prices and surgical costs according to the amounts reimbursed by the government, may underestimate the cost-effectiveness ratio. The analysis did not account for other obesity-related comorbidities such as stroke, heart failure, peripheral arterial disease, sleep apnea, cancer, and musculoskeletal disorders, and then it could undervalue the clinical benefits of surgery. Only two surgery complications (cholecystectomy and incisional hernia) were included in the model and did not consider for the possibility of other long-term surgery complications and clinical complications due to vitamin and iron deficiencies.
Brazil has no explicit value for a cost-effectiveness threshold in the public health system [50], which drives our choice for the WHO recommendations [38]. In addition, the current discussion about the use of GDP-based thresholds in decision-making processes [51] implies more conservative recommendations.
The development of this comprehensive decision analytic model for GBP surgery is a remarkable advance and can be further used for appropriate resources and decision-making processes. As many health care systems are operating under significant budget constraints, it is important to ensure that health interventions are cost-effective, i.e., reduce the cost of care or provide relevant clinical benefits for the money spent. Like all other health interventions, bariatric surgery must be evaluated from an economic perspective to support decision-making about the appropriateness of fund allocation for this service.
This economic evaluation demonstrated that GBP surgery is a cost-effective intervention for the treatment of severe obesity compared to clinical treatment in the perspective of public health system, showing a potential to reduce the risk of obesity-related conditions at 10 years at a reasonable cost, especially in the population with diabetes.
References
Zhou B, Lu Y, Hajifathalian K, Bentham J, Di Cesare M et al. Worldwide trends in diabetes since 1980: a pooled analysis of 751 population-based studies with 4.4 million participants. Lancet. NCD Risk Factor Collaboration.; 2016;387:1513–30. Available from: https://doi.org/10.1016/S0140-6736(16)00618-8
Brasil. Ministério da Saúde. Secretaria de Vigilância em Saúde. Vigitel Brasil 2016: vigilância de fatores de risco e proteção para doenças crônicas por inquérito telefônico. Ministério da Saúde, Secr. Vigilância em Saúde, Dep. Vigilância Doenças e Agravos não Transm. e Promoção da Saúde. 2017. Available from: www.saude.gov.br/svs
Tremmel M, Gerdtham U-G, Nilsson P, Saha S. Economic burden of obesity: a systematic literature review. Int J Environ Res Public Health. 2017 [cited 2018 Dec 15];14:435. Available from: http://www.ncbi.nlm.nih.gov/pubmed/28422077
Bahia L, Coutinho ES, Barufaldi LA, Abreu Gde A, Malhao TA, de Souza CP, et al. The costs of overweight and obesity-related diseases in the Brazilian public health system: cross-sectional study. BMC Public Health. 2012/06/21. Rio de Janeiro; 2012;12:440. Available from: https://doi.org/10.1186/1471-2458-12-440
Governo do Brasil. Aumenta número de cirurgias bariátricas realizadas pelo SUS. Governo do Brasil; 2017. Available from: http://www.brasil.gov.br/saude/2014/03/aumenta-numero-de-cirurgias-bariatricas-realizadas-pelo-sus
Ferrari MA. Colelitíase em pacientes bariátricos: correlação da perda de peso com a incidência de colelitíase em pacientes após a realização do by-pass gastrintestinal. Clínica Cirúrgica. [Porto Alegre]: Pontifícia Universidade Católica do Rio Grande do Sul; 2014. Available from: http://hdl.handle.net/10923/6812
Angrisani L, Santonicola A, Iovino P, Formisano G, Buchwald H, Scopinaro N. Bariatric surgery worldwide 2013. Obes Surg. 2015/04/04. General and Endoscopic Surgery Unit, S. Giovanni Bosco Hospital, Via Filippo Maria Briganti, 255, Naples, Italy, luigiangrisani@chirurgiaobesita.it.; 2015;25:1822–32. Available from: https://doi.org/10.1007/s11695-015-1657-z
Sjostrom L, Lindroos AK, Peltonen M, Torgerson J, Bouchard C, Carlsson B, et al. Lifestyle, diabetes, and cardiovascular risk factors 10 years after bariatric surgery. N Engl J Med. 2004/12/24. Department of Body Composition and Metabolism, Sahlgrenska University Hospital, Goteborg, Sweden.; 2004;351:2683–93. Available from: https://doi.org/10.1056/NEJMoa035622
Sjostrom L, Narbro K, Sjostrom CD, Karason K, Larsson B, Wedel H, et al. Effects of bariatric surgery on mortality in Swedish obese subjects. N Engl J Med. 2007/08/24. Gothenburg, Swedend; 2007;357:741–52. Available from: https://doi.org/10.1056/NEJMoa066254
Picot J, Jones J, Colquitt JL, Gospodarevskaya E, Loveman E, Baxter L, et al. The clinical effectiveness and cost-effectiveness of bariatric (weight loss) surgery for obesity: a systematic review and economic evaluation. Heal Technol Assess 2009/09/04. Southampton Health Technology Assessments Centre, University of Southampton, UK.; 2009;13:1–190, 215–357, iii–iv. Available from: https://doi.org/10.3310/hta13410, 2009
Sjostrom L, Peltonen M, Jacobson P, Sjostrom CD, Karason K, Wedel H, et al. Bariatric surgery and long-term cardiovascular events. Jama. 2012/01/05. Gothenburg, Swedend; 2012;307:56–65. Available from: https://doi.org/10.1001/jama.2011.1914
Romeo S, Maglio C, Burza MA, Pirazzi C, Sjoholm K, Jacobson P, et al. Cardiovascular events after bariatric surgery in obese subjects with type 2 diabetes. Diabetes Care. 2012/08/03. Gothenburg, Swedend; 2012;35:2613–7. Available from: https://doi.org/10.2337/dc12-0193
Sjostrom L, Peltonen M, Jacobson P, Ahlin S, Andersson-Assarsson J, Anveden A, et al. Association of bariatric surgery with long-term remission of type 2 diabetes and with microvascular and macrovascular complications. Jama. 2014/06/11. Gothenburg, Swedend; 2014;311:2297–304. Available from: https://doi.org/10.1001/jama.2014.5988
Eliasson B, Liakopoulos V, Franzen S, Naslund I, Svensson AM, Ottosson J, et al. Cardiovascular disease and mortality in patients with type 2 diabetes after bariatric surgery in Sweden: a nationwide, matched, observational cohort study. Lancet Diabetes Endocrinol. 2015/10/03. Gothenburg, Swedend; 2015;3:847–54. Available from: https://doi.org/10.1016/s2213-8587(15)00334-4
Kelles SMB, Diniz MF, Machado CJ, Barreto SM. Mortality rate after open Roux-in-Y gastric bypass: a 10-year follow-up. Braz J Med Biol Res. 2014/06/12. Centro de Pos Graduacao, Faculdade de Medicina, Universidade Federal de Minas Gerais, Belo Horizonte, MG, Brasil.; 2014;47:617–25.
Kelles SMB, Machado CJ, Barreto SM. Ten-years of bariatric surgery in Brazil: in-hospital mortality rates for patients assisted by universal health system or a health maintenance organization. Arq Bras Cir Dig. 2014;27:261–7. Available from: https://doi.org/10.1590/s0102-67202014000400008
Faria GR, Preto JR, Costa-Maia J. Gastric bypass is a cost-saving procedure: results from a comprehensive Markov model. Obes Surg. 2013 [cited 2019 Mar 4];23:460–6. Available from: http://www.ncbi.nlm.nih.gov/pubmed/23341033
Borisenko O, Lukyanov V, Johnsen SP, Funch-Jensen P. Cost analysis of bariatric surgery in Denmark made with a decision-analytic model. Dan Med J. 2017/09/05. funchjensen@gmail.com.; 2017;64.
Lucchese M, Borisenko O, Mantovani LG, Cortesi PA, Cesana G, Adam D, et al. Cost-utility analysis of bariatric surgery in Italy: results of decision-analytic modelling. Obes Facts. 2017/06/12. Bariatric, General Surgery and Metabolic Department, Santa Maria Nuova Hospital, Florence, Italy.; 2017;10:261–72. Available from: https://doi.org/10.1159/000475842
Hoerger TJ, Zhang P, Segel JE, et al. Cost-effectiveness of bariatric surgery for severely obese adults with diabetes. Diabetes care. North Carolina. 2010;33:1933–9. Available from: https://doi.org/10.2337/dc10-0554
Keating C, Neovius M, Sjöholm K, Peltonen M, Narbro K, Eriksson JK, et al. Healthcare costs during 15 years after bariatric surgery for patients with different baseline glucose status. Lancet Diabetes Endocrinol. Melbourne, Australia; 2015;3:855–65. Available from: https://doi.org/10.1016/s2213-8587(15)00290-9
Rubino F, Nathan DM, Eckel RH, Schauer PR, Alberti KG, Zimmet PZ, et al. Metabolic surgery in the treatment algorithm for type 2 diabetes: a joint statement by international diabetes organizations. Diabetes Care. 2016/05/26. London, U.K.; 2016;39:861–77. Available from: https://doi.org/10.2337/dc16-0236
Association AD. Obesity management for the treatment of type 2 diabetes: standards of medical care in diabetes-2018. Diabetes Care. 2017/12/10. 2018;41:S65-s72. Available from: https://doi.org/10.2337/dc18-S007
Ministério da Saúde. Secretaria de Ciência Tecnologia e Insumos Estratégicos. Departamento de Ciência e, Tecnologia. Diretrizes metodológicas - diretriz de avaliação econômica. Bras. Secr. Ciência, Tecnol. e Insumos Estratégicos. 2015.
Caro JJ, Briggs AH, Siebert U, Kuntz KM. Modeling good research practices--overview: a report of the ISPOR-SMDM Modeling Good Research Practices Task Force-1. Med Decis Mak. 2012/09/20. Montreal, Canada; 2012;32:667–77. Available from: https://doi.org/10.1177/0272989x12454577
Siebert U, Alagoz O, Bayoumi AM, Jahn B, Owens DK, Cohen DJ, et al. State-transition modeling: a report of the ISPOR-SMDM Modeling Good Research Practices Task Force--3. Value Heal. 2012/09/25. Tirol, Austria.; 2012;15:812–20. Available from: https://doi.org/10.1016/j.jval.2012.06.014
Husereau D, Drummond M, Petrou S, Carswell C, Moher D, Greenberg D, et al. Consolidated Health Economic Evaluation Reporting Standards (CHEERS)--explanation and elaboration: a report of the ISPOR Health Economic Evaluation Publication Guidelines Good Reporting Practices Task Force. Value Heal. 2013/03/30. Edmonton, Canada; 2013;16:231–50. Available from: https://doi.org/10.1016/j.jval.2013.02.002
Associação Brasileira para o Estudo da Obesidade e da Síndrome Metabólica. Diretrizes brasileiras de obesidade 2016. 2016.
Zubiaurre PR, Bahia LR, da Rosa MQM, Assumpção RP, Padoin AV, Sussembach SP, et al. Estimated costs of clinical and surgical treatment of severe obesity in the Brazilian Public Health System. Obes Surg. 2017 [cited 2019 Feb 18];27:3273–80. Available from: http://www.ncbi.nlm.nih.gov/pubmed/28717859
Santos M, Cintra MACT, Monteiro AL, Santos B, Gusmão-filho F, Andrade MV, Noronha K, Cruz LN, Camey S, Tura B, Kind P Brazilian valuation of EQ-5D-3L health states. Med Decis Mak. 2016 [cited 2018 Nov 15];36:253–263. Available from: http://www.ncbi.nlm.nih.gov/pubmed/26492896
Ministério da Saúde. Gabinete do Ministro. Portaria no 424, de 19 de março de 2013. Redefine as diretrizes para a organização da prevenção e do tratamento do sobrepeso e obesidade como linha de cuidado prioritária da Rede de Atenção à Saúde das Pessoas com Doenças Crônicas. Brasília; 2013.
World Bank. PPP conversion factor, LCU per international $. 2017. Available from: https://data.worldbank.org/indicator/PA.NUS.PPP
Grossi J. Post-operative complications of open gastric by-pass Roux-Y: consecutive incisional hernia repair - the sublay technique. 21st World Congr. Int. Fed. Surg. Obes. Metab. Disord. Rio de Janeiro/Brazil: Springer US; p. 459–60.
Portal da Saúde. Informações de Saúde (TABNET). Assistência à Saúde. Produção Hospitalar (SIH/SUS). Available from: http://www2.datasus.gov.br/DATASUS/index.php?area=0202
Ministério da Saúde. System of management of the table of procedures, medications and OPM from SUS. Available from: http://sigtap.datasus.gov.br/tabela-unificada/app/sec/inicio.jsp
Bahia LR, Rosa RS, Santos RD, Araujo D V. Estimated costs of hospitalization due to coronary artery disease attributable to familial hypercholesterolemia in the Brazilian public health system. Arch Endocrinol Metab. 2018/05/24. Rio de Janeiro; 2018; Available from: https://doi.org/10.20945/2359-3997000000030
Schwarzer R, Rochau U, Saverno K, Jahn B, Bornschein B, Muehlberger N, et al. Systematic overview of cost-effectiveness thresholds in ten countries across four continents. J Comp Eff Res. 2015/10/23. 2015;4:485–504. Available from: https://doi.org/10.2217/cer.15.38
World Health Organization (WHO). Macroeconomics and health: investing in health for economic development. Report of the Commission on Macroeconomics and Health: Executive Summary. Geneva: World Health Organization; 2001. Available from: http://apps.who.int/iris/handle/10665/42709
Craig BM, Tseng DS. Cost-effectiveness of gastric bypass for severe obesity. Am J Med. 2002/11/13. Wisconsin, USA.; 2002;113:491–8.
Ackroyd R, Mouiel J, Chevallier JM, Daoud F. Cost-effectiveness and budget impact of obesity surgery in patients with type-2 diabetes in three European countries. Obes Surg. 2006/11/30. South Yorkshire, UK.; 2006;16:1488–503. Available from: https://doi.org/10.1381/096089206778870067
Maklin S, Malmivaara A, Linna M, Victorzon M, Koivukangas V, Sintonen H. Cost-utility of bariatric surgery for morbid obesity in Finland. Br J Surg. 2011/09/03. Helsinki, Finland.; 2011;98:1422–9. Available from: https://doi.org/10.1002/bjs.7640
Borisenko O, Mann O, Duprée A. Cost-utility analysis of bariatric surgery compared with conventional medical management in Germany: a decision analytic modeling. BMC Surg. Hamburg, Germany; 2017;17. Available from: https://doi.org/10.1186/s12893-017-0284-0
Alsumali A, Al-Hawag A, Samnaliev M, Eguale T. Systematic assessment of decision analytic models for the cost-effectiveness of bariatric surgery for morbid obesity. Surg Obes Relat Dis. 2018/05/08. Boston, Massachusetts; 2018; Available from: https://doi.org/10.1016/j.soard.2018.03.005
Ministério da Saúde. Portaria GM/MS no 252, de 30 de março de 1999. Inclui o procedimento de gastroplastia na Tabela do Sistema de Informações Hospitalares do Sistema Único de Saúde – SIH/SUS e credencia hospitais para realizá-lo. Brasília; 1999. Available from: http://portal2.saude.gov.br/saudelegis/leg_norma_espelho_consulta.cfm?id=3558576&highlight=&tipoBusca=post&slcOrigem=0&slcFonte=0&sqlcTipoNorma=27&hdTipoNorma=27&buscaForm=post&bkp=pesqnorma&fonte=0&origem=0&sit=0&assunto=&qtd=10&tipo_norma=27&numero=252&
Ministério da Saúde. Gabinete do Ministro. Portaria no 482, de 06 de março de 2017. Inclui o procedimento Cirurgia Bariátrica por Videolaparoscopia na Tabela de Procedimentos, Medicamentos, Órteses, Próteses e Materiais Especiais do Sistema Único de Saúde - SUS. Secretaria de Atenção a Saúde, editor. Brasília; 2017. Available from: http://bvsms.saude.gov.br/bvs/saudelegis/sas/2017/prt0482_07_03_2017.html
PAHO FAO and. Panorama of Food and Nutrition Security in Latin America and the Caribbean. Santiago de Chile; 2017. Available from: http://www.fao.org/3/a-i7914e.pdf
Doumouras AG, Saleh F, Anvari S, Gmora S, Anvari M, Hong D. Mastery in bariatric surgery: the long-term surgeon learning curve of Roux-en-Y gastric bypass. Ann Surg. 2017/02/24. Department of Surgery, McMaster University, Hamilton, Ontario, Canada.Division of General Surgery, St Joseph’s Healthcare, Hamilton, Ontario, Canada.; 2018;267:489–94. Available from: https://doi.org/10.1097/sla.0000000000002180
Ministério da Saúde. Gabinete do Ministro. Portaria no 425, de 19 de Março de 2013. Estabelece regulamento técnico, normas e critérios para a Assistência de Alta Complexidade ao Indivíduo com Obesidade. Gabinete do Ministro, editor. Brasília; 2013. Available from: http://bvsms.saude.gov.br/bvs/saudelegis/gm/2013/prt0425_19_03_2013.html
El-Kadre L, Tinoco AC, Tinoco RC, Aguiar L, Santos T. Overcoming the learning curve of laparoscopic Roux-en-Y gastric bypass: a 12-year experience. Surg Obes Relat Dis. 2013/03/19. Rio de Janeiro, Brazil; 2013;9:867–72. Available from: https://doi.org/10.1016/j.soard.2013.01.020
Soarez PC, Novaes HMD. Cost-effectiveness thresholds and the Brazilian Unified National Health System. Cad Saúde Pública, vol. 33; 2017. Available from: http://www.scielo.br/scielo.php?script=sci_abstract&pid=S0102-311X2017000400301&lng=en&nrm=iso&tlng=en
Bertram MY, Lauer JA, De Joncheere K, Edejer T, Hutubessy R, Kieny MP, et al. Cost–effectiveness thresholds: pros and cons. Bull World Heal Organ. Geneva; 2016;94:925–930. Available from: https://doi.org/10.2471/blt.15.164418
Acknowledgments
We thank the support of the Brazilian Institute of Health Technology (IATS)/National Council for Scientific and Technological Development (CNPq). We thank Prof. Luiz Guilherme Kraemer de Aguiar (PhD) and Prof. Walmir Ferreira Coutinho (PhD) for the permission to collect data with patients from their obesity treatment centers.
Funding
The National Council for Scientific and Technological Development (CNPq), process no. 457440/2013-9, funded this study.
Author information
Authors and Affiliations
Contributions
LRB, DVA, and ENS conceived and designed the study; PRZ, MQR, and RPA performed the data collection and preliminary analysis; MGC developed the model and performed the statistical analysis; and LRB, RPA, and MQR wrote the draft manuscript. All authors revised and approved the manuscript.
Corresponding author
Ethics declarations
Conflicts of Interest
RPA received government grants to carry out the study. Authors RPA, LRB, DAV, MQR, RPA, ENS, PRZ, and CCM declare that they do not have any conflicts of interest. The study received funds for data monitoring and statistical analysis. The funder had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The Research Ethics Committees of each institution approved this study and approved on the Brazil Platform for clinical trials with their related numbers: HSL/PUCRS (1.488.130), IEDE (855.012), and UERJ (731.209).
Consent Statement
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Assumpção, R.P., Bahia, L.R., da Rosa, M.Q.M. et al. Cost-Utility of Gastric Bypass Surgery Compared to Clinical Treatment for Severely Obese With and Without Diabetes in the Perspective of the Brazilian Public Health System. OBES SURG 29, 3202–3211 (2019). https://doi.org/10.1007/s11695-019-03957-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-019-03957-7