Abstract
Introduction
Laparoscopic sleeve gastrectomy is one of the most common techniques in bariatric surgery, but there is no consensus on the optimal distance from the pylorus to start the gastric transection. The aim of this study is to determine the differences in gastric emptying, gastric distension and metabolic response between two starting distances.
Material and Methods
This is a prospective randomised study of 60 patients (30 patients with the section at 3 cm and 30 patients at 8 cm from the pylorus). We calculate at 6 and 12 months from surgery gastric emptying by scintigraphy (T1/2 min), gastric volume by CT scan (cc) and metabolic response by blood sample analysis (glucose, HbA1c, insulin, HOMA-IR, GLP-1, GIP and C-peptide).
Results
Gastric emptying increases the speed significantly in both groups but is greater in the 3-cm group (p < 0.05). Dividing groups into type 2 diabetic patients and non-diabetic patients, the speed in non-diabetic patients is significantly higher for the 3-cm group. Residual volume increases significantly in both groups, and there are no differences between them. One year after surgery, there are significant improvements in the hyperinsulinaemia in the patients of the 3-cm group with respect to the 8-cm group, but only in diabetic patients. No differences between groups are found regarding changes in GLP-1 or GIP.
Conclusions
Gastric emptying is faster in patients with antrum resection. The distance does not influence the gastric emptying of diabetic patients. Other mechanisms may explain metabolic response besides GLP-1 and its association with improvements in diabetes via gastric emptying.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Obesity is one of the main health problems in developed societies worldwide, and Spain is one of the European Union countries with the greatest prevalence of obesity [1]. In the case of morbid obesity (body mass index ≥40 kg/m2), excess weight is accompanied by multiple metabolic, hormonal and inflammatory disorders that are difficult to resolve and by important medical, psychological and social comorbidities that lead to important financial expense [2]. Bariatric surgery is the only efficient treatment for maintaining weight loss in the medium and long term and for resolving the associated comorbidities. Though it started out as a first-stage surgical procedure in biliopancreatic diversion with duodenal switch, laparoscopic sleeve gastrectomy (LSG) has become a good treatment for morbid obesity, thanks to the quality of its results in the short and medium term [3]. This first-stage procedure requires the surgeon to leave a residual antrum that is wide enough from the anatomical point of view to enable the application of a second surgical procedure. However, the tubulisation of the stomach must have enough of a restrictive component to be considered a separate or “alone” procedure whether it is performed on patients with a high BMI or on patients who are not clearly identified as candidates for malabsorptive procedures (e.g. patients with high-grade hepatic steatosis, hepatic cirrhosis, multiple prior abdominal interventions, adolescent morbid obesity) [4]. There is no consensus on the optimal distance from the pylorus to the beginning of the LSG section. The preservation or otherwise of the pyloric antrum may affect subsequent weight loss, the speed of gastric emptying and the residual volume. Although the initial dilatation of the gastric remnant appears to be a frequent occurrence, it has still not been possible to ascertain whether this is a physiological process or whether it is the cause of low weight loss since it is not observed in all patients who recover their initial weight [5]. The speed of gastric emptying is influenced by numerous factors, including the type of food ingested and the condition of the obese patient. A change in gastrointestinal motility—whether accelerated or delayed gastric emptying—has been observed in morbidly obese patients who have not undergone surgery compared with non-obese patients [6]. Whether the antrum is preserved or not may condition various responses in this regard [3, 7]. Hormone response after LSG may also be seen to be influenced through variations in gastric emptying [8]. In this case, surgery may be the means to justify the direct relationship between emptying and glucagon-like peptide 1 (GLP-1) levels, although from the clinical point of view, this has neither been demonstrated nor disproved [9,10,11,12]. Moreover, response to surgery may differ between diabetic and non-diabetic patients. However, although this hypothesis is supported by results from other experimental studies, it should be viewed with caution when deciding whether a certain technique is suitable for a patient with a certain metabolic profile [13].
The aim of our study is to determine the optimal distance between the pylorus and the beginning of the LSG section based on its effects on gastric distension (changes in gastric volume), the speed of gastric emptying and the metabolic response.
Material and Methods
This is a prospective and randomised clinical study of 60 morbidly obese patients with a BMI ≥50 kg/m2 divided into two surgical intervention groups. We perform this technique in super-obese patients in order to complete a second surgical time if it is required in the future (duodenal switch). General informed consent, approved by the ethics committee of our hospital, was obtained from all individual participants included in the study. All subjects signed a specific informed consent form. All patients received comprehensive written information about the project. The 60 patients were randomised into two groups and underwent LSG (30 patients with the section starting 3 cm from the pylorus and 30 patients with the section starting 8 cm from the pylorus) using a Web-based random number generator (www.random.org). The first participant joined the study in May 2012 and the last patient in November 2014. All operations were performed by the same bariatric surgeon.
Surgical Intervention
LSG was performed under general anaesthesia with the patient in the Lloyd-Davies position. A five-port technique was used in all patients: the first 10-mm midline trocar was inserted under direct optical control (Visiport, Covidien®, USA). A 15-mm trocar inserted into the right hypochondrium served to introduce the endostapler (4.8-mm staples) required for the first transections of the gastric antrum and was useful for extracting the resected stomach. Dissection of the greater gastric curvature was then conducted, separating it from the gastroepiploic arcade of the greater omentum. For this procedure, instruments for sealing and sectioning the tissues (LigaSure, Covidien®) were used. Dissection was continued to the angle of His; freeing the posterior fundal region is extremely important to avoid leaving a residual pocket. In all cases, the gastric transection was performed under the guidance of a 38-Fr Faucher bougie. The distance from the pylorus to the first section point (3 cm for group 1 and 8 cm for group 2) was measured intraoperatively with a ribbon. The suture line was reinforced using polycarbonate derivatives of polyglycolic acid (SEAMGUARD, W.L. Gore & Associates®, USA). Finally, a subhepatic Jackson-Pratt-type drain was placed along the suture. A methylene blue leak test was performed in all patients before the end of the surgery.
Weight Loss Evaluation
To evaluate weight loss, we used the percentage of excess BMI loss (PEBMIL) and the percentage of total weight loss (TWL) proposed by the American Society for Metabolic and Bariatric Surgery (ASBMS) [14].
Gastric Emptying by Scintigraphy
After 6 h of fasting, the patient was administered a food bolus comprising two 160-ml glasses of whole milk with seven ground cookies per glass and 1 mCi of technetium-99m sulfur colloid containing 400 kcal and a balanced composition of macronutrients (55% carbohydrates, 30% fats and 15% proteins). Images were collected while the patient ingested the food bolus for a short period of 1–2 min. Data and images were collected by scintigraphy (Infinia II, General Electric®, and LEHR (low-energy high-resolution)-type collimator). The patient sat in front of the gamma camera while ingesting the bolus. Dynamic anteroposterior images of the abdominal field were taken for 60 min with the sensors positioned vertically and in parallel. Activity/time curves were generated using regions of interest (ROI) at the level of the stomach in anterior and posterior projection calculated with the geometric mean. The result is expressed in T1/2 (minutes), or the average time it took for the stomach to empty 50% of the volume ingested (Fig. 1). Gastric emptying was measured before the operation and 6 and 12 months after the operation at our hospital’s Nuclear Medicine Service. After surgery, the volumes of the bolus and hydrosoluble contrast ingested depended on the patient’s capacity to ingest.
Gastric Volume by CT Scan
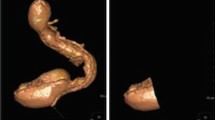
Gastric volume was determined by a low-resolution/dosage CT scan and a subsequent three-dimensional reconstruction by segmentation of the gastric chamber. This enabled the gastric volume to be calculated using IMAGE 3.0 for Windows software. Units of gastric volume are expressed in cubic centimetres (cc) (Fig. 2). Gastric volume was measured before the operation and 6 and 12 months after the operation at our hospital’s Radiology Service. Imaging was therefore done immediately after the patient had ingested the hydrosoluble contrast (Gastrografin®). The patient drank the contrast until he or she felt satiated, but ingestion of the mixture was not forced. Peristalsis was not stopped when these measurements were taken.
Analytical Data
Blood samples were taken before the operation and 6 and 12 months after the operation. We determined serum levels of glucose (mg/dl), glycosylated haemoglobin (% HbA1c), insulin (μUI/ml), HOMA-IR (homeostatic model assessment-insulin resistance) and C-peptide (pg/ml). Plasmatic glucagon-like peptide 1 (GLP-1) and gastric inhibitor polypeptide (GIP) (pg/ml) were measured by ELISA (MILLIPLEX®).
Data Analysis
We used the formulas for mean comparison and analysis of variance for each of the main variables according to Sample Power 3.0 software to calculate the number of patients in the study. With a minimum power of 80%, an α value of 0.05 and an expected difference of the means of 10 kg and 20 min of T1/2, we need 30 patients in each group to show significant differences. Results are expressed as means and standard deviations (SD). For the comparison of means before and after surgery, we applied Student’s t test and Fisher’s exact test on the assumption that the variable difference followed a normal distribution. We used the ANOVA test to compare differences between groups. In all cases, p values of less than 0.05 were considered statistically significant. The data were analysed using SPSS® software (22.0, SPSS Inc., Chicago, USA).
Results
A total of 60 patients with an average BMI of 51.1 kg/m2 and an average age of 50.9 were recruited prior to the operations. The baseline characteristics of the sample by group are shown in Table 1. No significant differences were observed between the two groups before surgery. A total of 98.3% of patients completed the established 1-year follow-up period by keeping all scheduled visits. The only loss during the follow-up period was caused by the death of one patient in the 8-cm group for reasons unrelated to surgery (car crash). One patient in the 8-cm group presented a leakage near the angle of His, which was self-limited with conservative measures within the first 10 days of the postoperative period.
We observed a significant decrease in weight in the two intervention groups after surgery. Despite absolute values being similar, as we published recently [15], the 3-cm group was in a higher percentile than the 8-cm group and obtained a lower percentage of poor results than the 8-cm group regarding PEBMIL (Tables 2 and 3). At the moment, no patient has undergone revision surgery due to an insufficient weight loss.
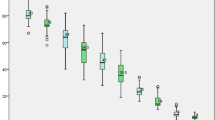
Table 4 shows the evolution of gastric volume and gastric emptying. Volumetric imaging tests indicate that 6 months after surgery, the volume was significantly lower in both groups but higher in the group whose section began 8 cm from the pylorus. Twelve months after surgery, the volume increased significantly in both groups and, importantly, the differences between the groups disappeared. On the other hand, the speed of gastric emptying increased significantly in both groups 6 and 12 months after surgery, but the speed was much greater in the 3-cm group even though at 12 months, the gastric volume began to increase progressively. Dividing the groups into type 2 diabetic patients and non-diabetic patients, in the group of non-diabetic patients, the speed of gastric emptying was significantly higher for the 3-cm group (Fig. 3). We found no differences between diabetic patients and non-diabetic patients regardless of the distance from the beginning of the section to the pylorus related to changes in volumetric data. The results of our metabolic response analysis are shown in Table 5. Overall, there were no significant differences between the glycaemic profiles of the patients in the 3-cm group and those of the patients in the 8-cm group whether the measurements were taken preoperatively, 6 months after surgery or 12 months after surgery. Dividing the groups into type 2 diabetic patients and non-diabetic patients, we found that preoperatively, the blood concentration of insulin was similar in all patients and in all groups. We also found that the 3-cm distance from the pylorus improved hyperinsulinaemia and the HOMA-IR index significantly compared with the 8-cm distance (Figs. 4 and 5). This result, only found in diabetic patients, was observed 6 months after surgery and was maintained 12 months after surgery. The other significant differences (in glycaemia and glycosylated haemoglobin) were observed between diabetic and non-diabetic patients rather than between patients in the two groups of resection distance. No differences were found between the 3-cm group and the 8-cm group in the concentration of incretins (GLP-1 and GIP) or in the concentration of C-peptide.
Discussion
Many technical aspects of LSG are currently under discussion. There is no consensus on the correct diameter for the guide used, though it is recommended that the gastric volume should be 100–120 ml [7, 16, 17]. There is also no consensus on the suitability of antrum resection or preservation. LSG can affect gastric emptying through multiple mechanisms, including altered contractility and gastric distensibility, as well as resection of the gastric fundus and its effects on receipt and propulsion of the food bolus and the disappearance of the body’s area for the gastric pacemaker [3]. Authors who advocate antral preservation, with a section beginning at 6 cm, believe that this preserves contractile function and promotes gastric emptying and therefore reduces intraluminal pressure and subsequent leakage [18], though the results of numerous studies in this area have been mixed. Antral preservation has several implications, including modulation of weight, intake, gastric volume and speed of gastric emptying. In morbidly obese patients who have not undergone surgery, a change in gastrointestinal motility (accelerated or delayed gastric emptying) has been observed in comparison with non-obese patients [6]. Since patients’ emptying profiles also depend on the characteristics of their intake (volume, caloric content, macronutrient composition and density), each laboratory or hospital should establish its own values of normality as well as its own protocol on the food to be ingested for gastric scintigraphy, which depends on whether catering facilities and ventilation are available on-site. As such catering facilities are unavailable at our location, we had to administer uncooked food to our patients. It contained approximately 400 kcal and satisfied all the conditions required for a balanced diet as far as macronutrients are concerned. This may partly account for the heterogeneity of the results found in the bibliography [19].
The studies by Braghetto et al. [5] show that the speed of gastric emptying for both solids and liquids is greater in obese patients with a starting distance of 3 cm from the pylorus. In our study, the speed of gastric emptying increased significantly in both groups 6 and 12 months after surgery, though the speed was much higher in the 3-cm group. However, other studies found no relationship between antrum preservation and speed of emptying in morbidly obese patients who underwent LSG with a starting distance of 8 cm from the pylorus [20]. Sista et al. [21] defend in their study the need to separate the speed of gastric emptying according to the density of the ingested food because the mechanisms of gastric emptying differ in solids and liquids, although they conclude that the rate of gastric emptying is increased after LSG for both solids and liquids. In our study, the patients ingested a semi-solid milled mixture of whole milk with biscuits, corroborating an increased gastric emptying rate. We should also mention our patients’ rather slow speed of gastric emptying (T1/2 = roughly 100 min) prior to surgery.
When we divided the groups into diabetic and non-diabetic patients, we also found that in the group of non-diabetic patients, the speed of gastric emptying was significantly higher for the 3-cm group. This indicates that gastric emptying is influenced not only by the distance from the section to the pylorus but also by the patient’s metabolic condition. Some studies exclude patients with poorly controlled type 2 DM or more than 10 years of evolution since this condition may influence the functionality of the vagus nerve and skew the study of gastric emptying [21]. This fact could explain the absence of differences in the diabetic group at 12 months of the intervention.
As we expected, patients in the 8-cm group had a larger gastric volume than those in the 3-cm group. Twelve months after surgery, these differences disappeared and the volume was the same in both groups regardless of the starting point of the tubular gastrectomy section. Although the gastric volume of both groups increased, the speed of gastric emptying also continued to increase for both groups. Moreover, the speed was greater (the stomach emptied faster) in the 3-cm group. The antrum therefore performs the role of a regulatory pump, which, in the case of the 3-cm group, disappears to produce a more direct or “less regulated” emptying. Although the volume increases, the lack of an antrum does not curtail the emptying. This dilatation of the remnant after surgery in our series of interventions coincides with that observed in the study by Vidal et al. [22], who applied a radiological volumetric model based on an oesophageal-gastric-duodenal transit to measure gastric volume after LVG with a starting point 5 cm from the pylorus. There appear to be no absolute differences between groups as far as weight is concerned, although, as we reported recently [15], the patients in the 3-cm group obtained better results, with a TWL located at a higher percentile than that in the 8-cm group; when we analysed the PEBMIL, the 8-cm group also presented a greater failure in terms of weight loss. The relationship between regaining gastric volume and losing weight does not appear to exist, at least in the short term. Numerous studies have reported conflicting results in the medium term with regard to this relationship [23, 24]. Further studies are therefore needed to analyse the relationship between the increase in gastric volume and its effect on long-term weight loss.
The classification of the techniques of bariatric surgery as restrictive, malabsorptive or mixed is becoming increasingly less common, mainly because of the role of the hormones that regulate appetite, satiety and weight control. Knowledge of these regulatory pathways and their various interactions is still at a very early stage. However, this is beginning to become the main reason why LSG does not operate as a purely restrictive technique. In non-obese individuals, incretins in the small intestine (such as GLP-1) work to reduce postprandial hyperglycaemia by decreasing or regulating the speed of gastric emptying. Bariatric surgery may be the means by which the direct relationship between emptying and GLP-1 levels is justified, although from the clinical point of view, this has neither been fully demonstrated nor fully disproved in the case of morbid obesity [3, 9,10,11, 22]. This is based on the hindgut hypothesis and it is supported by results from other experimental studies, although it should be viewed with caution when deciding whether a certain technique is suitable for a patient with a certain metabolic profile [13]. In this sense, Gagner recalls that GLP-1 decreases gastric emptying velocity and that this emptying is regulated after LSG by the length of the antrum left. A too short distance (<1 cm) will also produce the opposite effect, that is, a semi-obstruction pattern [12].
In this study, preoperatively, we found that the blood concentration of insulin was high in all patients. After surgery, and only in diabetic patients, we found significant improvements in hyperinsulinaemia, HbA1c (%) and HOMA-IR index in the 3-cm group compared with the 8-cm group. This result is observed both 6 and 12 months after surgery. No differences were found between the 3-cm group and the 8-cm group in the baseline concentration of incretins (GLP-1 and GIP) or in the concentration of C-peptide. The studies of Sista et al. concluded a fast gastric emptying after LSG that could be related to increased plasmatic GLP-1 levels, according to the hindgut theory [21]. Our study shows no significant difference between plasmatic GLP-1 levels before and after LSG and between the 3-cm group and the 8-cm group, although there is a tendency to decrease their plasmatic levels and it could be related to the improvement in the hyperinsulinaemia. Preoperative fasting plasmatic C-peptide levels were associated with increased type 2 diabetes remission after bariatric surgery [25], although there were other parameters that may influence such as age, BMI and duration of type 2 DM [26]. In our study, no differences have been found between plasmatic C-peptide levels before and after surgery and between the 3-cm and 8-cm groups. Neither have we found a relation between improvements in HbA1c (%) and plasmatic levels of C-peptide.
Conclusion
Non-antral preservation results in a higher speed of gastric emptying compared with antral preservation. Diabetic patients with antral preservation have a higher speed of gastric emptying than non-diabetic patients but a worse metabolic control, with no differences in baseline incretin levels compared with the other groups. This fact enhances the importance of the metabolic status of bariatric patients prior to surgery and the role of other hormones regulating the metabolic response beyond GLP-1. Further studies are needed to confirm these findings.
Limitations
This study did not analyse the postprandial response of incretins, which could have explained some differences between the groups. The follow-up is in the short-medium term, still without results in the long term, which will be necessary without doubt to corroborate our results. Changes in gastric volume after surgery may partially be subjected to the intake capacity of the oral contrast of every patient.
References
Rodríguez-Rodríguez E, López-Plaza B, López-Sobaler AM, et al. Overweight and obesity among Spanish adults. Nutr Hosp. 2011;26(2):355–63.
Sánchez-Santos R, Sabench Pereferrer F, Estévez Fernandez S, et al. Is the morbid obesity surgery profitable in times of crisis? A cost-benefit analysis of bariatric surgery. Cir Esp. 2013;91(8):476–84.
Milone L, Strong V, Gagner M. Laparoscopic sleeve gastrectomy is superior to endoscopic intragastric balloon as a first stage procedure for super-obese patients (BMI > or =50). Obes Surg. 2005;15(5):612–7.
Gagner M, Deitel M, Erickson AL, et al. Survey on laparoscopic sleeve gastrectomy (LSG) at the Fourth International Consensus Summit on Sleeve Gastrectomy. Obes Surg. 2013;23(12):2013–7.
Braghetto I, Davanzo C, Korn O, et al. Scintigraphic evaluation of gastric emptying in obese patients submitted to sleeve gastrectomy compared to normal subjects. Obes Surg. 2009;19:1515–21.
Segura Molina E, Chover González AJ, Partida Palma F, et al. Analysis of gastric emptying dynamics in relationship to qualitative composition of intake. Comparison between normal-weight and obese subjects. Rev Esp Med Nucl. 2006;25(3):172–9.
Weiner RA, Weiner S, Pomhoff I, et al. Laparoscopic sleeve gastrectomy—influence of sleeve size and resected gastric volume. Obes Surg. 2007;17:1297–305.
Melissas J, Daskalakis M, Koukouraki S, et al. Sleeve gastrectomy-a “food limiting” operation. Obes Surg. 2008;18(10):1251–6.
Deane AM, Nguyen NQ, Stevens JE, et al. Endogenous glucagon-like peptide-1 slows gastric emptying in healthy subjects, attenuating postprandial glycemia. J Clin Endocrinol Metab. 2009;95:215–21.
Peterli R, Wölnerhanssen B, Peters T, et al. Improvement in glucose metabolism after bariatric surgery: comparison of laparoscopic Roux-en-Y gastric bypass and laparoscopic sleeve gastrectomy: a prospective randomized trial. Ann Surg. 2009;250(2):234–41.
Shah S, Shah P, Todkar J, et al. Prospective controlled study of effect of laparoscopic sleeve gastrectomy on small bowel transit time and gastric emptying half-time in morbidly obese patients with type 2 diabetes mellitus. Surg Obes Relat Dis. 2010;6(2):152–7.
Gagner M. Faster gastric emptying after laparoscopic sleeve gastrectomy. Obes Surg. 2010;20(7):964–5.
Chambers AP, Smith EP, Begg DP, et al. Regulation of gastric emptying rate and its role in nutrient-induced GLP-1 secretion in rats after vertical sleeve gastrectomy. Am J Physiol Endocrinol Metab. 2014;306(4):E424–32.
Brethauer SA, Kim J, El Chaar M, et al., ASMBS Clinical Issues Committee. Standardized outcomes reporting in metabolic and bariatric surgery. Obes Surg. 2015;25(4):587–606. doi:10.1007/s11695-015-1645-3.
Sabench Pereferrer F, Molina López A, Vives Espelta M, et al. Weight loss analysis according to different formulas after sleeve gastrectomy with or without antral preservation: a randomised study. Obes Surg. 2016;27(5):1254–60. doi:10.1007/s11695-016-2454-z.
Kueper MA, Kramer KM, Kirschniak A, et al. Laparoscopic sleeve gastrectomy: standardized technique of a potential stand-alone bariatric procedure in morbidly obese patients. World J Surg. 2008;32(7):1462–5.
Parikh M, Gagner M, Heacock L, et al. Laparoscopic sleeve gastrectomy: does bougie size affect mean %EWL? Short-term outcomes. Surg Obes Relat Dis. 2008;4(4):528–33.
Michalsky D, Dvorak P, Belacek J, et al. Radical resection of the pyloric antrum and its effect on gastric emptying after sleeve gastrectomy. Obes Surg. 2013;23(4):567–73.
Farrell MB, Costello M, McKee JD, et al. Compliance with gastric-emptying scintigraphy guidelines: a report from the Intersocietal Accreditation Commission database. J Nucl Med Technol. 2017;45(1):6–13.
Bernstine H, Tzioni-Yehoshua R, Groshar D, et al. Gastric emptying is not affected by sleeve gastrectomy—scintigraphic evaluation of gastric emptying after sleeve gastrectomy without removal of the gastric antrum. Obes Surg. 2009;19(3):293–8.
Sista F, Abruzzese V, Clementi M, et al. The effect of sleeve gastrectomy on GLP-1 secretion and gastric emptying: a prospective study. Surg Obes Relat Dis. 2017;13(1):7–14. doi:10.1016/j.soard.2016.08.004.
Vidal P, Ramón JM, Busto M, et al. Residual gastric volume estimated with a new radiological volumetric model: relationship with weight loss after laparoscopic sleeve gastrectomy. Obes Surg. 2014;24(3):359–63.
Braghetto I, Cortes C, Herquiñigo D, et al. Evaluation of the radiological gastric capacity and evolution of the BMI 2-3 years after sleeve gastrectomy. Obes Surg. 2009;19(9):1262–9.
Obeidat F, Shanti H, Mismar A, et al. The magnitude of antral resection in laparoscopic sleeve gastrectomy and its relationship to excess weight loss. Obes Surg. 2015;25(10):1928–32.
Yan W, Bai R, Yan M, et al. Preoperative fasting plasma C-peptide levels as predictors of remission of type 2 diabetes mellitus after bariatric surgery: a systematic review and meta-analysis. J Investig Surg. 2017; doi:10.1080/08941939.2016.1259375.
Lee WJ, Chong K, Chen SC, et al. Preoperative prediction of type 2 diabetes remission after gastric bypass surgery: a comparison of DiaRem scores and ABCD scores. Obes Surg. 2016;26(10):2418–24.
Acknowledgements
This study was funded by the “Carlos III Research Institute”/Ministry of Economy and Competitiveness (registration number PI 11/02332), Spain. We are very grateful for the tasks of the nursing teams of the operating rooms and hospitalisation area, whose help has been indispensable for the correct functioning of the project, as well as all the staff of the Anesthesiology and Rehabilitation Service.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Informed consent was obtained from all individual participants included in the study. All procedures performed were in accordance with the ethical standards of the institutional research committee (registration number CEIC11–04-28/4PROJ3) and with the 1964 Helsinki declaration and its later amendments.
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Vives, M., Molina, A., Danús, M. et al. Analysis of Gastric Physiology After Laparoscopic Sleeve Gastrectomy (LSG) With or Without Antral Preservation in Relation to Metabolic Response: a Randomised Study. OBES SURG 27, 2836–2844 (2017). https://doi.org/10.1007/s11695-017-2700-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-017-2700-z