Abstract
Purpose
A new species of the genus Ceratocolax Vervoort, 1965 is described based on specimens collected from the Tomtate grunt Haemulon aurolineatum Cuvier, caught in the coast of Angra dos Reis, off the State of Rio de Janeiro, Brazil.
Methods
One hundred specimens of H. aurolineatum were purchased from the local fish market and examined for parasitic copepods. Parasites were fixed and preserved in 80% ethanol. Morphological features of the copepods were examined and drawn using an Olympus BX51 equipped with a drawing tube.
Results
Ceratocolax tavaresi n. sp. can be distinguished from all congeners by the following combination of characters in the adult female: (1) second endopodal segment of leg 3 with one seta, (2) lack of stout spinules along outer margins on rami of legs 2–4, (3) genital somite without flaplike structures, (4) terminal exopodal segment of leg 4 with seven elements; and in the adult male: (1) legs 1 to 4 with 3-segmented rami (except endopod of leg 4), (2) presence of a pair of blunt processes on dorsal surface of the third pedigerous somite, (3) second endopodal segment of leg 3 with one seta.
Conclusion
The number of species of Ceratocolax reported in the Atlantic Ocean was increased to three, including the new species. This is the forty-second species of copepod found parasitizing haemulid fish in marine waters from the Americas; however, the diversity of parasitic copepods off this continent is still underestimated.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cyclopoid copepods of the family Bomolochidae Claus, 1875 represent one of the most diverse groups of parasitic copepods, comprising about 162 species currently classified in 24 genera [1, 2]. Bomolochids are common parasites found on the head of marine fish, mainly in the branchial cavities but occasionally in the nostril, nasal sinuses, or orbits of its hosts [1]. The genus Ceratocolax Vervoort, 1965 includes only two valid species and are found in the nasal cavities of marine fish of the families Haemulidae Gill and Scombridae Linnaeus in the Atlantic waters [2, 3].
Among the 23 valid genera within the fish family Haemulidae, the genus Haemulon Cuvier currently comprises a total of 21 valid species [4]. The Tomtate grunt, Haemulon aurolineatum Cuvier occurs in costal zones from Massachusetts, USA to Brazil (including Gulf of Mexico and Bermuda), commonly inhabiting seagrass beds and patch reefs and feeding on a variety of invertebrates, mainly mollusks and small crustaceans [4, 5]. To date, four species of copepods have been reported parasitizing H. aurolineatum in the Atlantic Ocean, namely, Caligus robustus Bassett-Smith, 1898 (body surface), Colobomatus belizensis Cressey & Schotte, 1983 (mandibular canals), Hatschekia linearis Wilson, 1913 (gill filaments) and Mixtio inversa (Wilson, 1913) (gills) [6].
In the littoral of Brazil, nineteen species of bomolochids are recorded parasitizing bony fish, where seven belonging to the genus Bomolochus von Nordmann, 1832, followed by the genera Acantholochus Cressey, 1984 (three species), Ceratocolax Vervoort, 1965 (two species), Hamaticolax Ho & Lin, 2006 (two species), Orbitocolax Shen, 1957 (two species), Neobomolochus Cressey, 1981 (one species), Nothobomolochus Vervoort, 1962 (one species) and Unicolax Cressey & Cressey, 1980 (one species) [7, 8].
During parasitological studies on specimens of H. aurolineatum from the Brazilian coastal zone, some parasitic copepods were recovered from their nasal sinuses. Detailed morphological analysis of these parasites revealed that they represented a new species of Ceratocolax, which is described herein.
Materials and Methods
One hundred specimens of H. aurolineatum (total length 14.5–23.1; mean ± standard deviation 19.2 ± 1.8 cm) from the coast of Angra dos Reis (23°01′21ʺS, 44°19′13ʺW), State of Rio de Janeiro, Southeastern Brazil, were purchased from local fishermen between May 2022 and February 2023. Hosts were mostly fresh, but some specimens were kept frozen at -20 °C, until examination. Copepods were collected from the host, fixed and preserved in 80% ethanol until morphological analysis. For microscopical observation, specimens were cleared in 85% lactic acid and the appendages were dissected and examined using the wooden slide procedure described by Humes and Gooding [9]. Drawings were made using an Olympus BX51 microscope (Olympus Corporation, Tokyo, Japan) equipped with drawing tube. Measurements were based on eight adult females and four adult males, given as the ranges followed by mean and one standard deviation inside parentheses, all in micrometers or unless otherwise stated. Morphological terminology follows Huys and Boxshall [10]. Ecological terminology adopted for parasites is according to Bush et al. [11]. Host identification was based on Menezes and Figueiredo [12], and nomenclature and classification were updated according to Froese and Pauly [4]. Type specimens were deposited in the collection of the Museu de Zoologia da Universidade de São Paulo (acronym MZUSP), Brazil.
Systematics
-
Order Cyclopoida Burmeister, 1834
-
Family Bomolochidae Claus, 1875
-
Genus Ceratocolax Vervoort, 1965
-
Ceratocolax tavaresi n. sp.
Taxonomic Summary
Type host: The Tomtate grunt Haemulon aurolineatum Cuvier, 1830 (Actinopterygii: Haemulidae).
Site in host: nasal sinuses.
Type Locality: Angra dos Reis (23°01′21ʺS, 44°19′13ʺW), State of Rio de Janeiro, Brazil.
Prevalence and Intensity: 9% (09 infected fish out of 100 examined); mean of 1.4 copepods per infected fish (range 1–2).
Type material: Holotype: female (MZUSP-44978); allotype: male (MZUSP-44979); paratypes: five females and two males (MZUSP-44980). Three specimens (two females and one male) were dissected and kept in the personal collection of the second author.
ZooBank registration: urn:lsid:zoobank.org:pub:6D54EBBC-8A83-4F59-939B-4CF703B4897C.
Etymology: The new species is named after Prof. Luiz Eduardo Roland Tavares from Universidade Federal de Mato Grosso do Sul, Brazil, for his contributions to our knowledge of neotropical parasites.
Description (Figs. 1, 2, 3, 4 and 5).
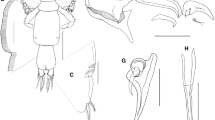
Ceratocolax tavaresi n. sp. (adult female). A Habitus, dorsal view, bp = blunt processes; B rostrum, ventral view; C cephalothoracic shield, lateral view, sp = spine, a1 = antennule, a2 = antenna, p1 = leg 1; D urosome, ventral view; E anal somite and caudal rami, ventral view; F antennule, ventral view, arrows showing the aesthetasc = ae
Ceratocolax tavaresi n. sp. (adult female). A detail of dorsally process on the antennule, lateral view; B antenna, ventral view; C labrum, ventral view; D mandible, ventral view; E paragnath, ventral view; F maxillule, ventral view; G maxilla, ventral view; H maxilliped, ventral view, arrow showing the accessory process = ap; I leg 5, ventral view
Adult Female
Body cyclopiform (Fig. 1A), 1,616–2,084 (1,838 ± 178) long; prosome length 1,015–1,147 (1,200 ± 173), maximum width 926–1,232 (1,021 ± 108). Prosome comprising broad cephalothorax incorporating first pedigerous somite, and free second to fourth pedigerous somites; third somite not overlapping fourth in dorsal view (Fig. 1A). Pedigerous somites on prosome distinctly separated from each other and becoming narrower posteriorly. Third pedigerous somite ornate with pair of blunt processes on dorsal part of anterior margin (Fig. 1A). Cephalothorax bearing pair of robust acutely-pointed tines in rostral area (Fig. 1B). Cephalothoracic shield with a pair of dorsal spines at anterior part (Fig. 1C). Urosome (Fig. 1D) 553–685 (645 ± 48) long, comprising fifth pedigerous somite, genital double-somite and 3 free abdominal somites. All urosomites wider than long; genital double-somite shorter than wide 264–416 (298 ± 58) × 388–645 (460 ± 92), with strongly convex lateral margins. Surfaces of anterior urosomites smooth, lacking ornamentation. Anal somite weakly incised posteromedially, with spinules present on ventral surface (Fig. 1E). Caudal rami (Fig. 1E) about 1.6 times longer than wide, with usual 2 long and 4 short setae.
Antennule (Fig. 1F) apparently 6-segmented, comprising broader proximal part and slender distal part; proximal part 3-segmented but second segment divided by partial suture; distal part slender, comprising 3 segments. First segment bearing 5 pilose setae, none modified; second segment bearing 7 pilose setae, plus 1 naked seta dorsally, and 2 short plumose setae, plus 3 naked setae on ventral surface. Arising dorsally between the junction of first and second segments, owns a robust process, with broad basis and rounded distal margin (Fig. 2A), tip of process curves medial towards to antennule. Third segment bearing 2 pilose setae, distalmost long, extending beyond the next segment, plus 1 naked seta on dorsal surface; segments 4 to 6 with setal formula 3, 2, 5 + 2ae.
Antenna (Fig. 2B) uniramous, 3-segmented; comprising long proximal segment (coxobasis) bearing single long seta, short middle (= first endopodal) segment armed with small naked seta and highly ornamented apical segment. Apical segment comprising partly fused second and third endopodal segments: proximal part (representing second endopodal segment) bearing blunt distal process, ornamented with multiple rows of tiny spines over ventral surface of segment, and with pectinate process dorsally; distal part (third endopodal segment) bearing 4 curved claws and 2 unequal naked setae.
Labrum (Fig. 2C) wider than long, ornamented with denticles on ventral surface and a pair of blunt elements on median part. Mandible (Fig. 2D) tipped with 2 unequal blades, each with single spinulate margin. Paragnath (Fig. 2E) bipartite; basal part with 1 naked seta, distal process blunt with smooth margins. Maxillule (Fig. 2F) lobate, armed with 1 naked seta and 3 unequal pilose setae. Maxilla (Fig. 2G) 2-segmented; proximal segment (syncoxa) larger, unarmed; second segment (basis) narrowing distally, bearing 2 spinulate apical elements plus naked seta. Maxilliped (Fig. 2H) 3-segmented; comprising syncoxa, armed with seta in proximal half; basis armed with 2 unequal pilose setae; terminal (endopodal) segment forming sigmoid claw provided with short accessory process, and armed with pilose seta proximally.
Legs 1 to 4 biramous, armature of legs (spines, Roman numerals; setae, Arabic numerals) as indicated in Table 1.
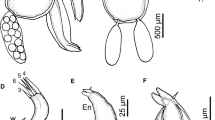
Leg 1 (Fig. 3A) biramous, modified with flattened rami as typical for bomolochids. Protopod ornamented with fine setules at outer margin; inner (coxal) seta of protopod transformed into pilose swollen element; basis with outer pilose seta; interpodal sclerite small, slightly longer than wide, ornamented with paired patches of spinules. Exopod 3-segmented: first segment with denticulate spine at outer distal corner; distal segment bearing total of 3 outer spines of unequal sizes. Endopod 3-segmented: all endopodal segments ornamented with outer margin setules; first and second segments with pointed element on the median margin.
Leg 2 (Fig. 3B) with 3-segmented rami; coxa with hirsute inner seta and setules at outer margin; basis with outer plumose seta. Ornamentation of long setules present along outer margin of the first exopodal segment. All outer spines on exopodal segments denticulate and provided with subterminal flagellum. Endopodal segments broad and flattened; outer margins of all segments ornamented with setules. Interpodal plate broad and flattened and ornamented with terminal row of spinules.
Leg 3 (Fig. 3C) with 3-segmented rami; coxa and basis armed with basal inner and outer plumose setae, respectively. All outer spines on exopodal segments unilaterally denticulate and provided with subterminal flagellum; outer margin of the first exopodal segment ornamented with row of spinules. Endopodal segments flattened; outer margins of all segments ornamented with setules. Interpodal plate broad and ornamented with terminal row of spinules.
Leg 4 (Fig. 3D) with 3-segmented rami; coxa lacking inner seta and spinules at outer margin; basis with outer plumose seta. All outer spines on exopodal segments denticulate and provided with subterminal flagellum; outer margin of the first exopodal segment ornamented with row of spinules. Outer margins of all endopodal segments ornamented with setules. Inner seta on first endopodal segment short, extending nearly to distal part of second segment. Inner seta on second endopodal segment extending to about mid-length of third segment. Third segment with spinule present adjacent to bases of outer and inner apical spines; apical seta about as long as segment. Interpodal plate broad and ornamented with terminal row of spinules as in leg 3.
Leg 5 (Fig. 2I) 2-segmented; protopodal segment small, ornamented with patch of spinules and armed with outer seta; free exopodal segment armed with spine at mid-length, plus outer and inner naked subterminal spines and middle terminal naked seta; exopodal segment ornamented with spinules extending along margins, plus distal patch of spinules.
Leg 6 (Fig. 1D) represented by 3 setae located in egg sac attachment area on genital double-somite.
Adult Male
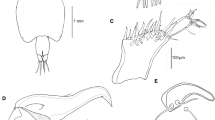
Body cyclopiform (Fig. 4A) 905–997 (960 ± 49) long; prosome length 489–559 (530 ± 36), maximum width 365–459 (408 ± 47); comprising cephalothorax incorporating first pedigerous somite, and free second to fourth pedigerous somites. Third pedigerous somite ornate with pair of blunt processes on dorsal part of anterior margin (Fig. 4A). Rostral area without tines. Urosome (Fig. 4B) length 399–445 (422 ± 23); comprising fifth pedigerous somite, elongate, pear-shaped genital somite, and two free abdominal somites. Genital somite longer than wide 205–223 (213 ± 9) × 170–198 (188 ± 15). Ventral surface of first free abdominal somite naked (Fig. 4B). Anal somite weakly incised, ornamented with transverse rows of spinules on the anterior and posterior margins (Fig. 4C). Caudal rami (Fig. 4C) about 2 times longer than wide; setation as in female.
Antennule (Fig. 4D) apparently 7-segmented; proximal 4 segments only slightly more robust than distal 3 cylindrical segments. First segment with 5 robust pilose setae, none modified; compound second segment with 10 setae: 5 pilose setae, plus 1 naked seta dorsally and 2 plumose, plus 2 naked setae on ventral surface. Third segment with 1 naked seta and 2 robust pilose setae; fourth segment bearing 2 pilose setae, distalmost long, extending beyond apex of antennule, plus 1 naked seta on anterior surface. Cylindrical distal segments with setal formula 3, 2 + 1ae and 7 + 1ae, respectively.
Antenna, mandible, maxillule and maxilla as in female.
Maxilliped (Fig. 4E) with elongate syncoxa armed with naked seta; basis robust, tapering distally, ornamented proximo-medially with multiple rows of short spinules and armed medially with 2 setae of unequal sizes; distal subchela incorporating unarmed endopodal segment, and bearing curved claw armed with seta proximally; concave margin of claw ornamented with row of denticles, plus a small seta at basis.
Legs 1 to 4 biramous with 3-segmented rami except 2-segmented endopod of leg 4; armature of legs (spines, Roman numerals; setae, Arabic numerals) as indicated in Table 2.
Leg 1 (Fig. 5A) with protopod and rami less flattened and less modified than in female. Coxa and basis distinct; coxa ornamented with row of spinules at outer margin and armed with inner plumose seta (not swollen as in female); basis armed with swollen outer seta and ornamented with inner patch of spinules, plus slender plumose seta near endopodal basis. Interpodal sclerite robust, ornamented with row of spinules. Outer spines on exopodal segments unilaterally denticulate and provided with subterminal flagellum. All endopodal segments ornamented with patches of setules on anterior surface near outer margins; second segment with tapered spine medially.
Leg 2 (Fig. 5B) with 3-segmented rami; coxa with long plumose inner seta and spinules at outer margin; basis with outer plumose seta. First exopodal segment with distal patch of spinules near outer margin. All outer spines on exopodal segments denticulate and provided with subterminal flagellum. Outer margins of all endopodal segments ornamented with setules. Interpodal plate broad and ornamented with terminal patches of thick spinules.
Leg 3 (Fig. 5C) with 3-segmented rami; coxa and basis armed with basal inner and outer plumose setae, respectively. All outer spines on exopodal segments denticulate and provided with subterminal flagellum. Endopodal segments flattened; outer margins of all segments ornamented with setules. Interpodal plate broad and ornamented with terminal patches of slender spinules.
Leg 4 (Fig. 5D) with 3-segmented exopod and 2-segmented endopod; coxa lacking inner seta and basis with outer plumose seta. All outer spines on exopodal segments denticulate and provided with subterminal flagellum. Inner plumose seta on proximal endopodal segment almost twice as long as ramus; distal endopodal segment with inner apical spine almost twice as long as outer spine; apical seta plumose, about twice as long as ramus. Long setules present on outer margins of both endopodal segments. Interpodal plate flattened and ornamented with terminal patches of slender spinules.
Leg 5 (Fig. 5E) 2-segmented; protopodal segment small, armed with outer seta and ornamented distally with of patch of spinules; free exopodal segment ornamented distally with patch of spinules, and bearing 2 unequal terminal setae.
Remarks
The adult females examined in the present study are identified as belonging to Ceratocolax by their possession of the following combination of characters: (1) all 5 setae at the proximal segment of antennule plumose, (2) presence of sclerotized dorsal process situated at the junction of the first and second segments of antennule, (3) pair of hook-like chitinous process on anterior margin of cephalothorax, (4) rostral hooks present, (5) maxilliped claw with accessory process, (6) legs 2–4 with 3-segmented rami, and (7) caudal rami with 2 major setae [1, 13].
The genus Ceratocolax was established by Vervoort [14] to accommodate their new species Ceratocolax euthynni Vervoort, 1965 from the scombrid fish Euthynnus alletteratus (Rafinesque) off Abidjan, Cote d’Ivoire. The type species was subsequently reported from two other scombrid host Thunnus albacares (Bonnaterre) [as Neothunnus albacora] and Sarda sarda (Bloch) in the Atlantic waters [12, 15]. After, Ceratocolax mykternastes Cressey, 1981 was described from the haemulid fish Haemulon sciurus (Shaw) off Carrie Bow Cay, Belize [3]. This second species was recently recorded from Haemulon plumierii (Lacepède) off Rio de Janeiro, Brazil [6].
The female of the new species can be readily distinguished from its congeners by the second endopodal segment of leg 3 with one seta and the lack of stout spinules along outer margins on rami of legs 2–4. In contrast, the two congeneric species, C. euthynni and C. mykternastes, have the second endopodal segment of leg 3 with two setae and the outer margins on rami of legs 2–4 possesses patches of stout spinules [3, 13].
In addition, the new species differs from C. euthynni by the genital double-somite without flaplike structures (vs. three flaplike structures in the latter) [13], as well as from C. mykternastes by the terminal exopodal segment of leg 4 with seven elements (vs. eight elements in the latter) [3].
Among the males, the new species is most similar to C. euthynni, since these two copepods share the legs 1 to 4 with 3-segmented rami (except 2-segmented endopod of leg 4), while in C. mykternastes the legs 1 to 4 have 2-segmented rami [3, 13]. However, C. tavaresi n. sp. easily differs from C. euthynni by the presence of a pair of blunt processes on dorsal surface of the third pedigerous somite (vs. absence in the latter) and by the second endopodal segment of leg 3 with one seta (vs. two in the latter) [3, 13].
Discussion
Determining boundaries between some bomolochid genera has proven problematic, especially due to the generic instability [16,17,18]. Nevertheless, the females in the genus Ceratocolax own a process situated at the junction of the first and second segments on antennule, making this feature the most assertive diagnostic for this bomolochid genus [1, 13]. In the present study, it was observed that this unusual structure differs in the morphology of the known species on Ceratocolax: long, curved, heavily sclerotized hook-like on C. euthynni; long, curved, sclerotized spine bearing a small naked seta at tip on C. mykternastes; and robust process, with broad basis and rounded distal margin on C. tavaresi n. sp. These observations imply that the morphology of this structure on the antennule in these species may be used for species identification as a supplementary character.
Recently, Izawa [19] proposed the genus Anomalocolax to accommodate Anomalocolax nemipteri Izawa, 2023 from the nemipterid fish Nemipterus japonicus (Bloch, 1791) off Japan. According to Izawa [19], the erected bomolochid genus is unique in having the first segment of the antennule with one process plus five plumose setae in the adult female. However, this process is very similar to the found in species of the genus Ceratocolax, which prompted us to re-consider the validity of the genus Anomalocolax was a possible synonym of Ceratocolax. Comparing the morphological features of these copepods, we found that the maxillipeds can be distinguish these genera, since the maxilliped claw in species of Ceratocolax have an accessory process, while Anomalocolax does not present. The presence or absence of an accessory process on the maxilliped claw among the bomolochids demonstrated to be useful for generic distinction; the genera Orbitacolax and Pseudorbitacolax Pillai, 1971, for example, can be also separated by this feature, since the first genus has such element, while the latter does not have [see 1]. Based on the previous information we consider that Anomalocolax is a distinct genus from Ceratocolax.
The parasite fauna of Haemulidae may be well considered high diverse [6, 20, 21]. Along the littoral of the American Continent, forty-two (including C. tavaresi n. sp.) copepods are known to parasites these fish, where 21 (50%) of these species are recorded off Brazil, followed by United States of America and Belize with 8 and 7 species recorded, respectively [6, 22, 23]. Moreover, the family Bomolochidae is the second group in number of species parasitizing grunts off Americas with 7 species, just behind to the family Caligidae Burmeister, 1835 with 14 species [22, 23]. This uneven biogeographical distribution of species richness of copepods is likely to be related to sampling effort of the researchers, especially by the diligent works of Cressey and Schotte [24], Cressey [16, 25] and Suárez-Morales et al. [26] in the North and Central America and by our research group in the South America [6, 27,28,29]. However, the present data emphasize that more effort should be directed to the study of parasitic copepods on haemulids, since its biodiversity most likely higher than that currently known in marine waters of the new world.
References
Boxshall GA, Halsey SH (2004) An introduction to copepod diversity. The Ray Society, London
Walter TC, Boxshall G (2023) World of Copepods Database. Bomolochidae Claus, 1875. WoRMS. https://www.marinespecies.org/aphia.php?p=taxdetails&id=128564. Accessed 27 Jul 2023
Cressey HB (1981) Ceratocolax mykternastes, new species (Copepoda, Bomolochidae) parasitic in the nasal sinus of Haemulon sciurus (Pisces, Pomadasydae) from Belize. Proc Biol Soc Wash 94(2):514–524
Froese R, Pauly D (2023) FishBase. World Wide Web electronic publication. http://www.fishbase.org. Accessed 27 Jul 2023
Manooch CS, Barans CA (1982) Distribution, abundance, and age and growth of tomtate, Haemulon aurolineatum, along the southeastern United States. US Fish Bull 80:1–19
Paschoal F, Cezar AD, Luque JL (2015) Checklist of metazoan associated with grunts (Perciformes, Haemulidae) from the Nearctic and neotropical regions. Check List 11(1):1501. https://doi.org/10.15560/11.1.1501
Luque JL, Pavanelli G, Vieira F, Takemoto R, Eiras J (2013) Checklist of Crustacea parasitizing fishes from Brazil. Check List 9(6):1449–1470. https://doi.org/10.15560/9.6.1449
Chaves L, Paschoal F (2020) Community ecology of the metazoan parasites of the Atlantic thread herring, Opisthonema oglinum (Lesueur, 1818) (Actinopterygii: Clupeidae) from the Sepetiba Bay, Rio De Janeiro, Brazil. Braz J Biol 81(2):418–423. https://doi.org/10.1590/1519-6984.229814
Humes AG, Gooding RU (1964) A method for studying the external anatomy of copepods. Crustaceana 6(3):238–240. https://doi.org/10.1163/156854064x00650
Huys R, Boxshall GA (1991) Copepod evolution. The Ray Society, London
Bush JO, Lafferty KD, Lotz JM, Shostak AW (1997) Parasitology meets ecology on its own terms: Margolis. Revisit J Parasitol 83(4):575–583. https://doi.org/10.2307/3284227
Menezes NA, Figueiredo JL Manual de Peixes Marinhos do Sudeste do Brasil. IV., Teleostei (1980) (3). Museu de Zoologia da Universidade de São Paulo, São Paulo
Cressey RF, Cressey HB (1980) Parasitic copepods of mackerel- and tuna-like fishes (Scombridae) of the world. Smith Contrib to Zoo 311:1–86. https://doi.org/10.5479/si.00810282.311.i
Vervoort W (1965) Three new species of Bomolochidae (Copepoda, Cyclopoida) from tropical Atlantic tunnies. Zool Verh Lei 76(1):1–40
Vervoort W (1971) New hosts for Ceratocolax Euthunni Vervoort, 1965 (Copepoda, Cyclopoida). Crustaceana 21(2):223–223. https://doi.org/10.1163/156854071X00454
Cressey R (1984) A new genus of bomolochid copepod from eastern Pacific haemulid fishes. Bull Mar Sci 35(2):182–186
Morales-Serna FN, Gómez S (2010) A new bomolochid copepod parasitic on bullseye puffer Sphoeroides annulatus (Jenyns) from Mexico, with reassignment of some species of Acantholochus Cressey and Hamaticolax Ho & Lin Zootaxa 2336(1):36–50. https://doi.org/10.11646/zootaxa.2336.1.3.
Pérez-i-García D, Carrassón M, Boxshall GA (2017) A new species of Hamaticolax Ho & Lin, 2006 (Copepoda: Bomolochidae) from deep water macrourids in the Mediterranean. Syst Parasitol 94:243–254. https://doi.org/10.1007/s11230-016-9693-1
Izawa K (2023) Anomalocolax nemipteri gen. et sp. nov. (Copepoda, Cyclopoida, Bomolochidae) recovered from Nemipterus japonicus (Bloch, 1791) (Actinopterygii) in Japan. Crustaceana 96(6):595–600. https://doi.org/10.1163/15685403-bja10297
García-Teh JG, Vidal-Martínez VM, Mariño-Tapia I, Árcega-Cabrera F, Ordoñez-López U, Aguirre-Macedo ML (2022) Metazoan parasite infracommunities of the Tomtate Grunt (Haemulon aurolineatum) as potential bioindicators of environmental conditions in the Yucatan Continental Shelf, Mexico. Bull Environ Contam Toxicol 108(1):49–54. https://doi.org/10.1007/s00128-021-03305-5
Guillén-Hernández S, Canto A, Villegas-Hernández H, González-Salas C, Poot-López G, Espínola-Novelo JF, Pérez-España H (2023) Spatial variation in Helminth Parasite communities of the French grunt, Haemulon flavolineatum (Teleostei: Haemulidae), from the Yucatan Peninsula. J Parasitol 109(2):76–86. https://doi.org/10.1645/21-103
Paschoal F, Cezar AD, Pereira FB, Luque JL (2023) Structure of the metazoan parasite communities of haemulid fish (Actinopterygii: Perciformes) in the South Atlantic Ocean: a comparative approach. An Acad Bras Cienc 95:e20220205. https://doi.org/10.1590/0001-3765202320220205
Morales-Serna FN, Cana-Bozada V, Mera-Loor G, Loor-Andrade P, Fajer-Avila EJ, Ho JS (2015) New records of sea lice (Copepoda: Caligidae) from marine fishes in Jaramijó, an area with potential for sea-cage aquaculture in Ecuador. Zootaxa 3920(2):366–380. https://doi.org/10.11646/zootaxa.3920.2.8
Cressey RF, Schotte M (1983) Three new species of Colobomatus (Copepoda: Philichthyidae) parasitic in the mandibular canals of haemulid fishes. Proc Biol Soc 96(2):189–201
Cressey RF (1991) Parasitic copepods from the Gulf of Mexico and Caribbean Sea, III: Caligus. Smith Contrib to Zoo 497:1–53. https://doi.org/10.5479/si.00810282.497
Suárez-Morales E, Reyes-Lizama C, González-Solís D (2010) Parasitic copepods from reef grunts (Teleostei, Haemulidae) with description of a new species of Lernanthropus (Siphonostomatoida, Lernanthropidae) from the Mexican Caribbean. Acta Parasitol 55(2):167–176. https://doi.org/10.2478/s11686-010-0025-3
Luque JL, Takemoto RM (1996) Parasitic copepods on Orthopristis ruber and Haemulon steindachneri (Osteichthyes: Haemulidae) from the Brazilian littoral, with the description of a new species of Caligus (Siphonostomatoida: Caligidae). Braz J Biol 56:529–546
Paschoal F, Cezar AD, Luque JL (2013) A new species of Acantholochus (Cyclopoida, Bomolochidae) parasitic on the barred grunt Conodon nobilis (Linnaeus, 1758) (Osteichthyes, Haemulidae) from Rio De Janeiro, Brazil. Crustaceana 86(2):212–220. https://doi.org/10.1163/15685403-00003153
Paschoal F, Couto JV, Pereira FB, Luque JL (2022) A New species of Hatschekiid Copepod (Crustacea: Hatschekiidae) parasitic on the Porkfish Anisotremus virginicus (Linnaeus, 1758) (Actinopterygii: Haemulidae), with notes on previously known species of Hatschekia Poche, 1902 collected from actinopterygians off Brazil. Acta Parasitol 67(3):1126–1135. https://doi.org/10.1007/s11686-022-00551-3
Acknowledgements
Fabiano Paschoal was supported by Fundação de Amparo à Pesquisa e ao Desenvolvimento Científico e Tecnológico do Maranhão (FAPEMA, Process no. 84516/2022), Brazil. José L. Luque was supported by a research fellowship from the Conselho Nacional de Desenvolvimento Científico e Tecnologico do Brazil (CNPq).
Funding
No funding was received for conducting this study.
Author information
Authors and Affiliations
Contributions
FP and JLL performed field collections; FP and TC analyzed the copepods and prepared the illustrations; TC and FP prepared the main manuscript document; all authors reviewed the manuscript & approved the final version.
Corresponding author
Ethics declarations
Ethical Approval
All applicable institutional, national and international guidelines for the care and use of animals were followed.
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Casanova, T., Paschoal, F. & Luque, J.L. Ceratocolax tavaresi n. sp. (Copepoda: Bomolochidae) parasitic in the Tomtate Grunt Haemulon aurolineatum Cuvier, 1830 (Actinopterygii: Haemulidae) off Rio de Janeiro, southeastern Brazil. Acta Parasit. 68, 903–912 (2023). https://doi.org/10.1007/s11686-023-00733-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11686-023-00733-7