Abstract
Hamaticolax resupinus n. sp. is described from specimens collected from the gill cavities of Coelorinchus mediterraneus Iwamoto & Ungaro and Coryphaenoides mediterraneus (Giglioli) (Gadiformes: Macrouridae) caught in the Western Mediterranean Sea at depths between 1,236 and 1,626 m. Hamaticolax resupinus n. sp. closely resembles H. maleus Oldewage, 1994, but differs from the latter by its smaller body size and in having a genital double-somite in the female that is markedly wider than the free abdominal somites and has strongly convex lateral margins. The new species is only the second bomolochid found on a macrourid host and is the first from depths in excess of 1,200 m. Hamaticolax resupinus n. sp. also represents the first parasitic copepod recorded from Coe. mediterraneus and only the third one from Cor. mediterraneus worldwide.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The genus Hamaticolax Ho & Lin, 2006 (Bomolochidae Sumpf, 1871) is a small genus currently comprising ten species. Typically parasites of the gill cavity, these species have been reported from different families of fishes from both the Atlantic and Pacific Oceans: Hamaticolax attenuatus (Wilson, 1913) described from Scorpaena plumieri Bloch (Scorpaenidae) from off Jamaica; Hamaticolax embiotocae (Hanan, 1976) described from Cymatogaster aggregata Gibbons and other Embiotocidae from off California; Hamaticolax galeichthyos (Luque & Bruno, 1990) described from Galeichthys peruvianus Lütken (Ariidae) from off Perú; Hamaticolax maleus (Oldewage, 1994) described from Malacocephalus laevis (Lowe) (Macrouridae) from off South Africa; Hamaticolax occultus (Kabata, 1971) described from Hippoglossoides elassodon Jordan & Gilbert and Lyopsetta exilis (Jordan & Gilbert) (Pleuronectidae) from off British Columbia, Canada; Hamaticolax paralabracis (Luque & Bruno, 1990) described from Paralabrax humeralis (Valenciennes) (Serranidae) from off Perú; Hamaticolax prolixus (Cressey, 1969) described from Pleuronichthys coenosus Girard (Pleuronectidae) and subsequently found on other Pleuronectidae plus some Batrachoididae, Cottidae, Cynoglossidae, Embiotocidae, Hexagrammidae, Paralichthyidae and Sciaenidae from off California; Hamaticolax scutigerulus (Wilson, 1935) described from Pseudupeneus maculatus (Bloch) (Mullidae) from off Belize; Hamaticolax spinulus (Cressey, 1969) described from Scorpaena guttata Girard (Scorpaenidae) and some Sebastidae and Hexagrammidae from off California; and Hamaticolax unisagittatus (Tavares & Luque, 2003) from Centropomus undecimalis (Bloch) (Centropomidae) from off Brazil (Wilson, 1913; Wilson, 1935; Cressey, 1969; Kabata, 1971; Hanan, 1976; Cressey, 1983; Luque & Bruno, 1990; Oldewage, 1994; Tavares & Luque, 2003).
During a parasitological survey of macrourid species from the deep-sea off Catalonia and the Balearic Islands (Western Mediterranean), a new bomolochid of the genus Hamaticolax was collected from the gills and inner surface of the opercula of Coelorinchus mediterraneus Iwamoto & Ungaro and Coryphaenoides mediterraneus (Giglioli). This is only the second Hamaticolax species reported from macrourids and is the first member of the Bomolochidae to be found in the deep Mediterranean Sea. This paper describes both sexes of the new Hamaticolax species from Coe. mediterraneus and Cor. mediterraneus, and provides a key to the species of the genus.
Materials and methods
Three individuals of Coe. mediterraneus (preanal length: 7.0–8.0 cm) and one of Cor. mediterraneus (preanal length: 3.2 cm) were captured during the oceanographic project ANTROMARE (Spanish Ministry of Science and Innovation) on the research vessel García del Cid with a semi-balloon otter-trawl (OTSB) fished at depths between 1,236 and 1,626 m off Barcelona and the Balearic Islands. Fish were measured, weighed, assigned unique codes and stored at −20 °C on board until examination.
The copepods were removed from freshly thawed fish, washed in physiological saline and preserved in 70% ethanol. They were dissected and mounted in lactophenol as temporary slide preparations and examined on an Olympus microscope equipped with differential interference contrast optics. Drawings were made with the aid of a drawing tube. Measurements were made using an ocular micrometer and are presented as the range followed by the mean (in parentheses); all measurements are in millimetres. Morphological terminology follows Huys & Boxshall (1991). Host names were validated against FishBase (Froese & Pauly, 2016). Type-material is deposited at the Natural History Museum, London (NHMUK).
Family Bomolochidae Sumpf, 1871
Genus Hamaticolax Ho & Lin, 2006
Hamaticolax resupinus n. sp.
Type-host: Coelorinchus mediterraneus Iwamoto & Ungaro (Gadiformes, Macrouridae).
Other hosts: Coryphaenoides mediterraneus (Giglioli) (Gadiformes, Macrouridae).
Type-locality: Off Barcelona, Western Mediterranean (40°41.96′N, 01°37.46′E – 40°54.35′N, 02°06.06′E); depth: 1,236–1,269 m; 10.vii.2010, 16.x.2011).
Other localities: Off Ibiza, Western Mediterranean (39°56.20′N, 01°37.91′E; depth: 1,626 m; 19.xi.2011).
Type-material: Holotype female (NHMUK 2015.2974), allotype male (NHMUK 2015.2975), 9 female paratypes (2 damaged) and 2 male paratypes (1 damaged) (NHMUK 2015.2977–2986) ex Coelorinchus mediterraneus; 2 female paratypes and 2 male paratypes (1 damaged) (NHMUK 2015.2987–2990) ex Coryphaenoides mediterraneus.
Site on host: Gill filaments and inner surface of the opercula.
Etymology: The species name, resupinus, alludes to the reflexed form of the outer margin spines on the exopods of legs 3 and 4.
Description (Figs. 1–4)
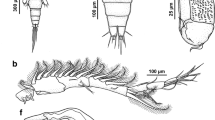
Adult female. Body cyclopiform (Fig. 1A), 1.22 to 1.31 (1.26) long (n = 10); prosome length 0.79–0.92 (0.86), maximum width 0.65–0.79 (0.74). Prosome comprising broad cephalothorax and free second to fourth pedigerous somites; third somite not overlapping fourth in dorsal view (Fig. 1A). Cephalothorax bearing pair of acutely-pointed, tapering tines in rostral area (Fig. 1C). Urosome (Fig. 1B) 0.40–0.44 (0.43) long (n = 9), comprising fifth pedigerous somite, genital double-somite and 3 free abdominal somites. All urosomites wider than long; genital double-somite with strongly convex lateral margins, about 1.6 times wider than first free abdominal somite; anal somite weakly incised posteromedially. Surfaces of anterior urosomites smooth, lacking ornamentation; paired transverse rows of spinules present on ventral surface of anal somite and longitudinal row present ventrally on each caudal ramus (Fig. 1B). Caudal rami (Fig. 1B) about 1.3 times longer than wide, bearing single principal seta, plus 5 smaller setae.
Antennule (Fig. 1C) comprising broader proximal part and slender distal part; proximal part 3-segmented but third segment divided by partial suture; distal part slender, comprising 3 segments. First segment bearing 5 pilose setae, none modified; second segment bearing 5 pilose setae, 3 naked setae dorsally, and 4 short plumose setae on ventral surface; third segment bearing 5 pilose setae, distalmost long, extending beyond apex of antennule, 4 naked setae on dorsal surface plus 1 small naked seta on ventral surface; segments 4 to 6 with setal formula: 4, 2+1ae, 7+1ae.
Antenna (Fig. 1D) uniramous, 3-segmented; comprising long proximal segment (coxobasis) bearing single long seta, short middle (=first endopodal) segment armed with small naked seta and highly ornamented apical segment. Apical segment comprising partly fused second and third endopodal segments: proximal part (representing second endopodal segment) produced into blunt distal process ornamented with rows of spinules ventrally, continuous with multiple rows over ventral surface of segment, and armed with stout curved claw and pectinate process distally; distal part (third endopodal segment) bearing 3 curved claws and 4 unequal naked setae.
Labrum (Fig. 2A) wider than long, ornamented with 3 sensilla on ventral surface. Mandible (Fig. 1E) tipped with 2 unequal blades, each with single spinulate margin. Paragnath (Fig. 2A) bipartite; basal part ornamented with long setules, distal process blunt, covered with shorter setules. Maxillule (Fig. 1F) lobate, armed with 1 minute naked seta and 3 unequal pilose setae. Maxilla (Fig. 2B) 2-segmented; proximal segment (syncoxa) larger, unarmed; second segment (basis) narrowing distally, bearing 2 spinulate apical elements plus naked seta. Maxilliped (Fig. 2C) 3-segmented; comprising syncoxa, armed with seta in proximal half; basis armed with 1 pilose seta and vestigial seta located on medial margin; terminal (endopodal) segment forming sigmoid claw provided with short accessory process, and armed with spinulate seta proximally.
Legs 1 to 4 biramous, with armature as indicated in Table 1.
Leg 1 (Fig. 2D) modified, with flattened rami. Protopod with swollen, hirsute outer basal seta; inner coxal seta transformed into flattened broad, hirsute element and inner basal element modified into short knob-like structure; basis ornamented with patches of fine surface spinules; interpodal sclerite with 2 rounded anterior lobes ornamented with short spinules around anterior margin. Exopod indistinctly 2-segmented, retaining almost complete suture between first and second segments but only partial suture between second and third; first segment with large spine at outer distal corner; compound distal segment bearing total of 4 outer spines and 6 plumose setae. Endopod 3-segmented: all endopodal segments ornamented with outer margin setules; second and third segments with inner margin setules; first segment with rows of fine surface spinules; third segment with minute spine located proximal to base of outermost seta.
Leg 2 (Fig. 2E) with 3-segmented rami; coxa with hirsute inner seta and basis with outer plumose seta and inner patch of spinules. All outer spines on exopodal segments unilaterally denticulate and provided with subterminal flagellum. Endopodal segments broad and flattened; outer margins of all segments ornamented with long setules, second and third segments with inner row of setules. Interpodal sclerite unornamented.
Leg 3 (Fig. 3A) with 3-segmented rami; coxa and basis armed with long inner and outer plumose setae, respectively; basis ornamented with inner patch of spinules. Exopodal segments with proximal outer spines reflexed over ramus. First exopodal segment with patch of spinules on outer distal margin. Endopodal segments broad, flattened and ornamented as in leg 2. Interpodal plate ornamented with paired patches of short spinules.
Leg 4 (Fig. 3B) with 3-segmented rami; coxa lacking inner seta; basis with outer plumose seta. All outer spines on exopodal segments finely denticulate, provided with subterminal flagellum and reflexed over ramus as in leg 3. Outer margins of all endopodal segments ornamented with long setules. Inner seta on first endopodal segment short, extending nearly to middle of second segment. Inner seta on second endopodal segment extending to about mid-length of third segment. Third segment with spinules present adjacent to bases of outer and inner apical spines; apical seta about as long as segment. Interpodal sclerite ornamented with paired patches of fine spinules as in leg 3.
Leg 5 (Fig. 2F) 2-segmented; protopodal segment small, ornamented with patch of spinules and armed with outer seta; free exopodal segment armed with spinulate spine at mid-length, outer naked subterminal spine, and inner spinulate and outer plumose terminal elements; inner distal spine longer than outer subterminal spine. Exopodal segment ornamented with spinules extending along outer margin, plus distal rows.
Leg 6 (Fig. 1B) represented by 3 short setae located in egg sac attachment area on genital double-somite.
Adult male. Body cyclopiform (Fig. 3C); 0.64–0.74 (0.69) long (n = 2). Prosome length 0.44–0.46 (0.45), maximum width 0.31–0.35 (0.33); comprising cephalothorax incorporating first pedigerous somite, and free second to fourth pedigerous somites. Rostral area without tines (Fig. 4A). Urosome (Fig. 3C) length 0.21–0.28 (0.24); comprising fifth pedigerous somite, elongate, pear-shaped genital somite, and two free abdominal somites. Ventral surface of first free abdominal somite naked (Fig. 3D). Anal somite weakly incised, ornamented with transverse row of spinules anteriorly and paired rows posteriorly (Fig. 3D). Caudal rami (Fig. 3D) ornamented with crescentic row of spinules ventrally; setation as in female.
Antennule (Fig. 4A) 5-segmented; proximal 2 segments only slightly more robust than distal 3 cylindrical segments. First segment with 5 robust pilose setae, none modified; compound second segment with 10 pilose setae, 5 naked setae dorsally, and 3 plumose and 2 naked setae ventrally; distalmost pilose element on second segment shorter than in female. Cylindrical distal segments with setal formula 4, 2+1ae and 7+1ae, respectively.
Antenna, mandible, maxillule and maxilla as in female.
Maxilliped (Fig. 3E) with broad syncoxa armed with plumose seta; basis robust, tapering distally, ornamented proximo-medially with multiple rows of short spinules and armed medially with 2 spinulate setae; distal subchela incorporating unarmed endopodal segment, and bearing curved claw armed with long seta proximally; concave margin of claw ornamented with row of denticles, plus cluster at tip.
Legs 1 to 4 biramous with 3-segmented rami except 2-segmented endopod of leg 4; setal armature as indicated in Table 2.
Leg 1 (Fig. 4B) protopod and rami less flattened and less modified than in female. Coxa and basis distinct; coxa ornamented with row of spinules and armed with inner plumose seta (not swollen as in female); basis armed with swollen hirsute outer seta and ornamented with inner patch of spinules. Interpodal sclerite linear, ornamented with paired patches of small spinules. Outer spines on exopodal segments each finely spinulate along outer margin and provided with subterminal flagellum. All endopodal segments ornamented with patches of spinules on anterior surface near outer margins; second and third segments each with row of setules along inner margin.
Legs 2 and 3 (Fig. 4C–D) with outer spines on exopodal segments finely unilaterally spinulate, and bearing subterminal flagellum. First exopodal segment with distal patch of spinules near outer margin and short row of setules on inner margin. Long setules present on outer margins of endopodal segments. Interpodal sclerites linear, ornamented with paired patches of small spinules.
Leg 4 (Fig. 4E) with 3-segmented exopod and 2-segmented endopod; outer margin spines on exopod segments finely unilaterally spinulate and bearing subterminal flagellum. Outer distal patch of spinules and inner row of setules present on first exopodal segment. Inner plumose seta on proximal endopodal segment about twice as long as ramus, extending almost to tip of long seta on distal segment; distal endopodal segment with inner apical spine almost twice as long as outer spine; apical seta plumose, about twice as long as ramus. Long setules present on outer margins of both endopodal segments.
Leg 5 (Fig. 4F) 2-segmented; protopodal segment small, armed with outer seta; free exopodal segment ornamented distally with patches of spinules, and bearing 2 unequal terminal setae.
Remarks
The new species is similar to Hamaticolax maleus, the only other member of the genus reported from a macrourid host. Detailed comparisons with H. maleus are difficult because of the number of observational errors and misinterpretations apparent in Oldewage’s (1994) original description of H. maleus. Apparent differences, such as the lack of the small outer spine on the third endopodal segment of leg 1 in H. maleus, are almost certainly observational errors. Others errors are more difficult to interpret: the figures of the legs are incorrectly labelled in Oldewage (1994) and the illustrations do not match the information in the text. Using the exopodal setation formula as a reference we identify Oldewage’s figure 2c as leg 2 (third exopodal segment with IV, 5) and figure 2d as leg 3 (third exopodal segment with III, 5). However the setation of the third endopodal segment is the same in both figures, whereas this segment of leg 3 typically has 2 setae and 2 spines in female bomolochids compared with 3 setae and 2 spines present on leg 2.
As a result of the inconsistencies in Oldewage (1994) we cannot rely on either the illustrations or the written descriptions of the limbs to provide reliable characters for species discrimination. The basic body shape is, however, more readily comparable. Oldewage (1994) shows that the genital double-somite is narrow and has more or less parallel lateral margins and he specifically states that “genital segment and three abdominal segments of equal diameter”. In contrast, the genital double-somite of the new species has strongly convex lateral margins (Fig. 1B) and is about 1.6 times wider than the first free abdominal somite. The body length of the female was not given by Oldewage (1994) but from the scale provided with the figure, the body length can be calculated as 1.6 mm, distinctly longer than the maximum length (1.31 mm) of the new species. These differences in size and gross body morphology might be supported by characters based on the limbs, such as the different proportional lengths of the 4 setae on the maxillules, but the uncertainty surrounding Oldewage’s figures is so high that we are unable to rely on them. Clearly, H. maleus is in urgent need of redescription.
Hamaticolax maleus was found on the macrourid Malacocephalus laevis caught off the west coast of South Africa (Oldewage, 1994). The depth of capture was unknown, but this host fish is typically found between depths of 300 and 750 m (Geistdoerfer, 1990), although its overall depth range is 200 to 1,000 m according to Cohen et al. (1990). The hosts of the new species were collected in the western basin of the Mediterranean at depths of 1,236 to 1,626 m.
Discussion
Determining boundaries between some bomolochid genera has proven problematic (Vervoort, 1962, 1969; Cressey, 1984) and the genera Acantholochus Cressey, 1984 and Hamaticolax have shown a particularly high level of instability (Ho & Lin, 2006; Morales-Serna & Gómez, 2010). Morales-Serna & Gómez (2010) addressed this problem and provided new diagnoses of both Acantholochus and Hamaticolax. They transferred three species from Acantholochus to Hamaticolax and another three species in the opposite direction and then created keys to species of both genera. The key to Acantholochus was updated by Pashoal et al. (2013) who described a new species, A. lamellatus Pashoal, Cezar & Luque, 2013 from Conodon nobilis (Linnaeus) caught off Brazil. The new bomolochid described here is placed in Hamaticolax on the basis of the presence of a pair of rostral hooks in the female, the form of the proximal segments of the female antennule (i.e. the anterior margin of the first two segments is straight), and the presence of an accessory process on the claw of the maxilliped. However, we consider that generic boundaries are still rather labile and that the validity of bomolochid genera should be tested by a comprehensive phylogenetic analysis.
Attempting to identify our material from Coelorinchus mediterraneus and Coryphaenoides mediterraneus using the key of Morales-Serna & Gómez (2010) was problematic since the key contains a number of errors. The second couplet distinguishing between the type-species Hamaticolax attenuatus (Wilson, 1913) and H. spinulus (Cressey, 1969), uses two characters, the number of outer spines on the mid-exopod segment of leg 3 and the total number of setal elements on the terminal exopod segment of leg 4. Morales-Serna & Gómez (2010) refer to two outer spines on the mid-exopod segment of leg 3, a character state not found in the entire order Cyclopoida. Indeed, in the whole of the Copepoda, two outer spines are present on the second exopodal segment of leg 3 only in the bizarre harpacticoid family Superornatiremidae (Huys, 1996). They also refer to the presence of only 5 elements on the terminal exopod segment of leg 4, a character state that would be extremely unusual for the Bomolochidae. Using Vervoort’s (1969) redescription of H. attenuatus, which was based on re-examination of Wilson’s (1913) type material, it is clear that the mid-exopod segment of leg 3 carries only one outer spine, as normal, and that the terminal exopod segment of leg 4 carries a total of 8 setal elements. So the characters provided for the key couplet do not serve to distinguish between the species. A new key is provided below, avoiding characters such as antennulary segmentation which are often difficult to interpret as segments are often incompletely separated in bomolochids.
Key to the species of Hamaticolax (based on females)
-
1a
Urosome as long as or longer than prosome ……… 2
-
1b
Urosome shorter than prosome ……… 3
-
2a
Hook-like spine on antenna extending beyond tip of pectinate process ……… H. attenuatus
-
2b
Hook-like spine on antenna not extending beyond tip of pectinate process ……… H. spinulus
-
3a
Second endopodal segment of legs 2 and 3 with 1 inner seta ……… H. unisagittatus
-
3b
Second endopodal segment of legs 2 and 3 with 2 inner setae ……… 4
-
4a
Apical segment of leg 4 endopod with 2 setal elements ……… H. galeichthyos
-
4b
Apical segment of leg 4 endopod with 3 setal elements ……… 5
-
5a
Third exopodal segment of legs 3 and 4 each with 7 elements ……… H. embiotocae
-
5b
Third exopodal segment of legs 3 and 4 each with 8 elements ……… 6
-
6a
Endopod of leg 4 elongate, almost twice as long as exopod; third endopodal segment of leg 4 more than 4 times longer than wide ……… H. paralabracis
-
6b
Endopod of leg 4 typically about as long as exopod, at most 1.5 times longer; third endopodal segment of leg 4 at most 1.5 times longer than wide …… 7
-
7a
Third exopodal segment of leg 2 with 8 elements (III, 5) ……… 8
-
7b
Third exopodal segment of leg 2 with 9 elements (IV, 5) ……… 9
-
8a
Leg 4 with long inner seta on first and second endopodal segments; seta on first segment reaching beyond distal margin of second and seta on second reaching beyond distal margin of third segment ……… H. occultus
-
8b
Leg 4 with short inner seta on first and second endopodal segments; seta on first segment not reaching beyond distal margin of second, and seta on second segment not reaching beyond distal margin of third ……… H. scutigerulus
-
9a
Leg 4 with long inner seta on first and second endopodal segments; seta on first segment reaching beyond distal margin of second and seta on second reaching beyond distal margin of third segment ……… H. prolixus
-
9b
Leg 4 with short inner seta on first and second endopodal segments; seta on first segment not reaching beyond distal margin of second, and seta on second segment not reaching beyond distal margin of third ……… 10
-
10a
Body length of adult female 1.6 mm; genital double-somite with linear lateral margins, about equal in width to free abdominal somites ……… H. maleus
-
10b
Body length of adult female 1.3 mm; genital double-somite with strongly convex lateral margins, markedly wider than free abdominal somites ……… H. resupinus n. sp.
Hamaticolax resupinus n. sp. is the first species of the genus found below 1,300 m. Considering the bathymetric distribution of Coe. mediterraneus and Cor. mediterraneus in the studied area, this parasite could potentially be found up to 2,250 m depth, although it is likely to occur around 1,200 to 1,800 m, where its hosts are more abundant (Stefanescu et al., 1992, 1993). Hamaticolax resupinus n. sp. is the first recorded parasitic copepod from Coe. mediterraneus and the third from Cor. mediterraneus worldwide, after Chondacanthodes deflexus Wilson, 1932 (see Kellermans et al., 2009) and Sarcotaces sp. (see Bullock et al., 1986), and its first Hamaticolax species from the Mediterranean Sea. Bomolochids are uncommon parasites of Macrouridae. The most common copepod families reported from macrourids are the Chondracanthidae Milne Edwards, 1840, Lernaeopodidae Milne Edwards, 1840 and Sphyriidae Wilson, 1919, especially the genera Clavella Oken, 1815 and Lophoura Kölliker, in Gegenbaur, Kölliker & Muller, 1853 (see Boxshall, 1998; Klimpel et al., 2009). However, the Macrouridae is a family with a worldwide distribution comprising over 400 species (Froese & Pauly, 2016), and only few have been targeted for extensive parasitological studies, such as Cor. mediterraneus (Kellermanns et al., 2009), Coryphaenoides rupestris Gunnerus (Campbell et al., 1980; Zubchenko, 1981) and Macrourus berglax Lacépède (Zubchenko, 1981; Palm & Klimpel, 2008). Therefore, as parasitological studies on members of the Macrouridae increase, the number of copepod parasites probably will do so, including the number of Bomolochidae.
References
Boxshall, G. A. (1998). Host specificity in copepod parasites of deep-sea fishes. Journal of Marine Systems, 15, 215–223.
Bullock, A. M., Phillips, S. E., Gordon, J. D. M., & Roberts, R. J. (1986). Sarcotaces sp., a parasitic copepod infection in two deep-sea fishes, Lepidion eques and Coelorhynchus occa. Journal of the Marine Biological Association U.K., 66, 835–843.
Campbell, R. C., Haedrich, R. L., & Munroe, T. A. (1980). Parasitism and ecological relationships among deep-sea benthic fishes. Marine Biology, 57, 301–313.
Cohen, D. M., Inada, T., Iwamoto, T., & Scialabba, N. (1990). FAO species catalogue. Vol. 10. Gadiform fishes of the world (Order Gadiformes). An annotated and illustrated catalogue of cods, hakes, grenadiers and other gadiform fishes known to date. FAO Fish Synopses, 125(10), FAO, Rome, 442 pp.
Cressey, R. (1969). Five new parasitic copepod from California inshore fish. Proceedings of the Biological Society of Washington, 82, 409–427.
Cressey, R. (1983). Parasitic copepods from the Gulf of Mexico and Caribbean Sea, II: Bomolochidae. Smithsonian Contributions to Zoology, 389, 1–35.
Cressey, R. (1984). A new genus of bomolochid copepod from eastern Pacific haemulid fishes. Bulletin of Marine Sciences, 35, 182–186.
Froese, R., & Pauly, D. (2016). Fishbase. World Wide Web electronic publication. www.fishbase.org, version (01/2016).
Geistdoerfer, P. (1990). Macrouridae. In: Quero, J.C., Hureau, J.C., Karrer, C., Post, A. & Saldanha, L. (Eds). Check-list of the fishes of the eastern tropical Atlantic (CLOFETA). JNICT, Lisbon; SEI, Paris; and UNESCO, Paris. Vol. 2, 541–563 pp.
Hanan, D. (1976). A new species of cyclopoid copepod, parasitic on shiner surfperch, Cymatogaster aggregata Gibbons, in Anaheim Bay and Huntington Harbor, California, with notes on Bomolochus cuneatus Fraser and Ergasilus lizae Krøyer. Bulletin of the Southern California Academy of Sciences, 75, 22–28.
Ho, J.-S., & Lin, C.-L. (2006). A new bomolochid copepod parasitic on marine fishes of Taiwan, with reassignment of species of Holobomolochus Vervoort, 1969. Crustaceana, 78, 1369–1381.
Huys, R. (1996). Superornatiremidae fam. nov. (Copepoda: Harpacticoida): An enigmatic family from North Atlantic anchihaline caves. Scientia Marina, 60, 497–542.
Huys, R., & Boxshall, G. A. (1991). Copepod Evolution (p. 468). London: The Ray Society.
Kabata, Z. (1971). Four Bomolochidae (Copepoda) from fishes of British Columbia. Journal Fisheries Research Board of Canada, 28, 1563–1572.
Kellermanns, E., Klimpel, S., & Palm, H. W. (2009). Parasite fauna of the mediterranean grenadier Coryphaenoides mediterraneus (Giglioli, 1893) from the Mid-Atlantic Ridge (MAR). Acta Parasitologica, 54, 158–164.
Klimpel, S., Busch, M. W., Kellermanns, E., Kleinertz, S., & Palm, H. W. (2009). Metazoan Deep-Sea Fish Parasites. Acta Biologica Benrodis, Supplementband II. Heinrich-Heine University Düsseldorf, 384 pp.
Luque, J. L., & Bruno, M. (1990). Two new species of Acantholochus Cressey, 1984 (Copepoda: Bomolochidae) parasitic on Peruvian marine fishes. Journal of Natural History, 24, 241–249.
Morales-Serna, F. N., & Gómez, S. (2010). A new bomolochid copepod parasitic on bullseye puffer Sphoeroides annulatus (Jenyns) from Mexico, with reassignment of some species of Acantholochus Cressey and Hamaticolax Ho & Lin. Zootaxa, 2336, 36–50.
Oldewage, W. H. (1994). A new species of Holobomolochus Vervoort, 1969 (Copepoda, Poecilostomatoida) from the west coast of South Africa. Crustaceana, 67, 324–328.
Palm, H. W., & Klimpel, S. (2008). Metazoan fish parasites of Macrourus berglax Lacépède, 1801 and other macrourids in the north Atlantic: Invasion of the deep sea from the continental shelf. Deep-Sea Research Part II, 55, 235–242.
Pashoal, F., Cezar, A. D., & Luque, J. L. (2013). A new species Acantholochus (Cyclopoida, Bomolochidae) parasitic on the barred grunt Conodon nobilis (Linnaeus, 1758) (Osteichthyes, Haemulidae) from Rio de Janeiro, Brazil. Crustaceana, 86, 212–220.
Stefanescu, C., Lloris, D., & Rucabado, J. (1992). Deep-living demersal fishes in the Catalan Sea (western Mediterranean) below a depth of 1000 m. Journal of Natural History, 26, 197–213.
Stefanescu, C., Lloris, D., & Rucabado, J. C. (1993). Deep-sea fish assemblages in western Mediterranean. Deep-Sea Research Part I, 40, 695–707.
Tavares, L., & Luque, J. L. (2003). A new species of Acantholochus (Copepoda: Bomolochidae) parasitic on Centropomus undecimalis (Osteichthyes: Centropomidae) from the coastal zone of the State of Rio de Janeiro, Brazil. Memórias do Instituto Oswaldo Cruz, 98, 241–245.
Vervoort, W. (1962). A review of the genera and species of the Bomolochidae (Crustacea, Copepoda), including the description of some old and new species. Zoologische Verhandelingen, Leiden, 56, 1–111.
Vervoort, W. (1969). Caribbean Bomolochidae (Copepoda: Cyclopoida). Studies on the Fauna of Curaçao and other Caribbean Islands, 28, 1–125.
Wilson, C. B. (1913). Crustacean parasites of West Indian fishes and land crabs, with descriptions of new genera and species. Proceedings of the United States National Museum, 44, 189–277.
Wilson, C. B. (1935). Parasitic copepods from the Dry Tortugas. Papers from Tortugas Laboratory, 29, 329–347.
Zubchenko, A. V. (1981). Parasitic fauna of some Macrouridae in the northwest Atlantic. Journal of the Northwest Atlantic Fishery Science, 2, 67–72.
Acknowledgements
We thank all participants in the ANTROMARE 1 and 3 cruises and the staff of the Department of Animal Biology, Vegetal Biology and Ecology of the Universitat Autònoma de Barcelona (Spain) for their assistance during the oceanographic campaign.
Funding
This study was supported by Spanish Science and Innovation Ministry project ANTROMARE (CTM2009-12214-C02-02). DPG benefited from a PIF PhD student grant of the Universitat Autònoma de Barcelona.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable institutional, national and international guidelines for the care and use of animals were followed.
Rights and permissions
About this article
Cite this article
Pérez-i-García, D., Carrassón, M. & Boxshall, G.A. A new species of Hamaticolax Ho & Lin, 2006 (Copepoda: Bomolochidae) from deep water macrourids in the Mediterranean. Syst Parasitol 94, 243–254 (2017). https://doi.org/10.1007/s11230-016-9693-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11230-016-9693-1