Abstract
Post-traumatic stress disorder (PTSD) manifests as emotional suffering and problem-solving impairments under extreme stress. This meta-analysis aimed to pool the findings from all the studies examining emotion and cognition in individuals with PTSD to develop a robust mechanistic understanding of the related brain dysfunction. We identified primary studies through a comprehensive literature search of the MEDLINE and PsychINFO databases. The GingerALE software (version 2.3.6) from the BrainMap Project was used to conduct activation likelihood estimation meta-analyses of the eligible studies for cognition, emotion and interface of both. Relative to the non-clinical (NC) group, the PTSD group showed greater activation during emotional tasks in the amygdala and parahippocampal gyrus. In contrast, the NC group showed significantly greater activation in the bilateral anterior cingulate cortex (ACC) than did the PTSD group in the emotional tasks. When both emotional and cognitive processing were evaluated, the PTSD group showed significantly greater activation in the striatum than did the NC group. No differences in activation between the PTSD and NC groups were noted when only the cognitive systems were examined. Individuals with PTSD exhibited overactivity in the subcortical regions, i.e., amygdala and striatum, when processing emotions. Underactivity in the emotional and cognitive processing intermediary cortex, i.e., the ACC, was especially prominent in individuals with PTSD relative to the NC population following exposure to emotional stimuli. These findings may explain the trauma-related fear, irritability, and negative effects as well as the concentration difficulties during cognitive distress associated with emotional arousal, that are commonly observed in individuals with PTSD.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Post-traumatic stress disorder (PTSD), which may develop following a traumatic experience, is a debilitating illness characterized by emotional distress associated with physiological arousal and poor concentration (Atwoli et al. 2015). The traumatic experience may also lead to stress-induced functional changes in multiple domains of the brain. In order to develop precise treatments for the PTSD symptoms, identifying the biological targets of treatment is imperative (Bremner 2007).
The neural correlates of the emotional dysregulation in PTSD likely involve the fronto-limbic system, given the corresponding symptoms of anger, nervousness, fear, intrusive, recurrent recollections, flashbacks, and nightmares. However, only a few publications have utilized cognitive tasks that probe the poor concentration and other related executive function difficulties that are commonly noted in PTSD (Moores et al. 2008; Sailer et al. 2008; Felmingham et al. 2009; Cisler et al. 2015; Shaw et al. 2009; Elman et al. 2009; Astur et al. 2006; Strigo et al. 2010; Geuze et al. 2008; Bryant et al. 2005; Falconer et al. 2008; Jovanovic et al. 2013). Given that emotions influence cognition, it is hypothesized that there would be whole-brain involvement, with combined emotional and cognitive domain dysfunction and corresponding network abnormalities in both the cognitive and emotional brain networks. Therefore, the present meta-analysis included published studies that examined two brain domains, namely cognition and emotion, based on different levels of the domain hierarchy from the brainmap coding scheme (Laird et al. 2009). We also added a third domain, i.e., the interface between emotion and cognition, since the underlying networks show close connectivity, as informed by our prior work (Pavuluri et al. 2010), and hence may be involved in the pathophysiology of PTSD (Hayes et al. 2012b).
The majority of individual functional neuroimaging studies on PTSD are proof-of-concept trials with small sample sizes. One way to amplify the power of these studies is to perform a meta-analysis, which integrates the data from all published studies on PTSD. One of the most common algorithms for performing coordinate-based meta-analyses that offer robust and reliable findings is the recently developed activation likelihood estimation (ALE) technique (Eickhoff et al. 2009). This method assumes that the peak coordinates reported by each study represent the activation maps from which they are derived and uses the reported coordinates in voxel-wise analyses to assess the consistency of activation in any given set of studies (Eickhoff et al. 2009; Kober and Wager 2010; Turkeltaub et al. 2012). By using this type of quantitative voxel-wise meta-analysis of already published results we can compare the findings from the PTSD population to those from the non-clinical control (NC) group and yield objective, unbiased, and statistically based quantified evidence of aberrant brain activation during cognitive, emotional and interface of both processing in PTSD. As far as we know, there has been no previous study about the meta-analysis of PTSD using the ALE method for those three domains – cognition, emotion, and interface of both.
Here, we conducted a separate meta-analysis for each individual domain of cognition, emotion processing, and the interface between cognition and emotion, as they relate to the PTSD diathesis. We hypothesized that emotional systems and circuits, in individuals with and without PTSD, would be closely linked to cognitive circuits. By separating which probe or domain dysfunction, cognition, emotion, and/or the interface of both, contributes to the activity in any given coordinate in this meta-analysis, we can quantitatively evaluate the results of neuroimaging studies performed in individuals with PTSD and understand that cognition and emotion are separate but interacting sub-systems of the brain in PTSD.
Methods
Search strategy
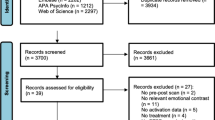
We performed a systematic literature search following the flow suggested by the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) statement (Moher et al. 2009). We identified primary studies through a comprehensive literature search of the MEDLINE (using both free-text and Medical Subject Headings searches) and PsychINFO databases using the following keywords: PTSD or traumatic or post-traumatic, and functional magnetic or fMRI. In addition, manual searches of the reference sections of review articles and individual studies were conducted to check for any missing studies not identified by the computerized searches. There were no language restrictions, as all included manuscripts were written in English. An initial list of studies was produced that included any report of functional magnetic resonance imaging (fMRI) studies of PTSD published in print or online by December 31, 2017. The selection process for the final list of primary studies for the planned meta-analyses in this study was very clear and specific. Accordingly, the inclusion of specific study could not be misinterpreted.
The first-level literature search with further manual search yielded 179 unique published articles with 73 studies meeting the following selection criteria. All reports included in the meta-analysis satisfied the following criteria: (1) included an NC comparison group; (2) conducted whole-brain analyses with or without region of interest (ROI) analyses; (3) provided standard Talairach or Montreal Neurological Institute (MNI) spatial coordinates for the key findings; (4) patient groups consisted of individuals diagnosed with PTSD according to the specific diagnostic criteria such as Diagnostic and statistical manual of mental disorders; (5) each subject group consisted of at least five subjects; (6) reported activation foci as three-dimensional coordinates in stereotactic space; (7) utilized active task constructs; and (8) presented the findings at the disorder vs. NC group level. Excluded manuscripts consisted of the following: (1) reviews or other meta-analyses; (2) studies with subject overlap; and (3) studies using other MRI modalities (e.g., structural imaging, spectroscopy, diffusion tensor imaging, and functional connectivity studies).
After a second-level review of these 73 studies, only 56 studies reported the coordinates that were essential for inclusion in this meta-analysis (Table 1). Any ambiguity in inclusion was resolved through discussion by the authors of this manuscript until consensus was reached. Study data (e.g., coordinates, subject numbers) were entered and cross-checked by the participating authors. The grouping of the studies based on the task type, i.e., emotion, cognition, and the interface of emotion and cognition, was also determined through independent classification decision-making by researchers, with any differences in classification being resolved through discussion until consensus (Fig. 1).
ALE methods and pairwise ALE meta-analysis
The GingerALE software (version 2.3.6) from the BrainMap Project was used to conduct ALE meta-analyses of eligible studies (Eickhoff et al. 2009; Turkeltaub et al. 2012; Eickhoff et al. 2012). We separately analyzed the data by the type of the task or brain domain probed, i.e., cognition, emotion, and the interface between cognition and emotion. Activation coordinates originally reported in MNI space were converted to Talairach coordinates using the Lancaster transform (icbm2tal) program the in GingerALE software. Our meta-analyses were conducted in Talairach space. For uniformity, Talairach coordinates expressed by the previous Brett transformation (Brett et al. 2002) were converted into MNI space and re-transformed into Talairach space.
We performed pairwise ALE meta-analyses that included the following comparisons in each domain (cognition, emotion, interface between cognition and emotion): greater activation in PTSD vs. NC and greater activation in NC vs. PTSD. The input coordinates were weighted to form estimates of activation likelihood for each intracerebral voxel. The activation likelihood of each voxel in standard space was then combined to form a statistic map of the ALE score at each voxel. Statistical significance of the ALE scores was determined by a permutation test controlling the false discovery rate at p < 0.05 (Laird et al. 2005). The statistic maps were thresholded by default at this critical value, and a recommended minimum cluster size was suggested from the cluster statistics. By using this minimum cluster size of supra-threshold voxels, we obtained the thresholded ALE image. The results of the pairwise ALE analyses are reported at p = 0.05 and are whole-brain corrected. The Talairach daemon was used to determine the anatomical locations of significant clusters.
Analyses were performed in two steps. In step one, only whole-brain analyses were included. In step two, both the whole-brain analyses and the ROI analyses were included.
Results
Our meta-analyses included 56 fMRI studies with PTSD and NC groups (emotion: 28 studies, cognition: 12, interface between cognition and emotion: 16 studies). No overlap in subjects that completed the same task across the selected studies was noted. The primary studies included in the meta-analyses are listed in Table 1. Findings are summarized in Table 2 and Fig. 2.
Results from pairwise activation likelihood estimation analysis. Each coordinate represents center coordinate of each cluster. A) Emotional task, PTSD>normal control, combined results of whole brain plus ROI analysis. Amygdala, x = 23.6, y = −6.7, z = −13.3, cluster size = 168 mm3, extreme value = 0.020 (B) Emotional task, normal control>PTSD, whole brain only analysis. Anterior cingulate cortex, x = −0.6, y = 34, z = −4.3, cluster size = 144 mm3, extreme value = 0.023 (C) Emotional task, normal control>PTSD, whole brain plus ROI analysis. Anterior cingulate cortex, x = −0.6, y = 34, z = −4.3, cluster size = 104 mm3, extreme value = 0.023 (D) interface between cognition and emotion, PTSD>normal control, whole brain only analysis. Lentiform nucleus, lateral globus pallidus, x = −18.6, y = −2.2, z = −7.2, cluster size = 240 mm3, extreme value = 0.024 (E) interface between cognition and emotion, PTSD>normal control, whole brain plus ROI analysis. Lentiform nucleus, lateral globus pallidus, x = −18.7, y = −2.2, z = −7.2, cluster size = 248 mm3, extreme value = 0.024
Cognition
No significant differences were found between the PTSD and NC groups for the cognitive task results either in the whole-brain only analyses or in the whole-brain plus ROI analyses.
Emotion
The PTSD group did not show any clusters with significantly greater activation than the NC group in the whole-brain only analyses. However, in the whole-brain plus ROI analyses, the PTSD group showed greater activation than the NC group in one cluster, i.e., the limbic lobe including the amygdala and parahippocampal gyrus within the right cerebrum (Fig. 2, Table 2).
In the whole-brain only analyses, the NC group showed significantly greater activation than the PTSD group in one cluster, namely the bilateral anterior cingulate cortex (ACC), at the interface of both the dorsal and the ventral ACC. In whole-brain plus ROI analyses, the NC group also showed significantly greater activation than the PTSD group in the same cluster (Fig. 2).
Interface between cognition and emotion
The PTSD group showed significantly greater activation than the NC group in both the whole-brain analyses and in the whole-brain plus ROI analyses in one cluster, i.e., the caudate, putamen, and globus pallidus (Fig. 2).
When comparing the NC group relative to the PTSD group, no clusters of significantly greater activation were found in either the whole-brain analyses or the whole-brain plus ROI analyses.
Discussion
The present meta-analysis is the first to collectively evaluate studies that used tasks probing emotion, cognition, and the interface of emotion and cognition. Given the small number of studies, we employed the broad approach of using whole-brain and ROI analyses to assess the affective circuitry’s operations in individuals with PTSD. Our findings illustrate that PTSD predominantly affects the emotional circuitry regions either directly or in association with the cognitive circuitry. Our results are particularly important as extreme affect dysregulation in the PTSD patients when faced with exterrnal triggers may often be confused with that of bipolar disorder. This study is one of the many steps to discern the underlying brain pathophysiology of emotional and cognitive systems in the PTSD.
Fear, negativity, avoidance, and hyperarousal are known clinical features of PTSD, which may explain the increased amygdala activation that we observed in individuals with PTSD compared to NC individuals (emotion domain, PTSD>NC, Right parahippocampal gyrus and amygdala in whole brain + region of interest analysis). Indeed, increased activation of the limbic system, especially the amygdala, has been observed in individual studies of patients with PTSD (Lanius et al. 2010). The amygdala is known to play a key role in fear conditioning (Rogan et al. 1997), including regulating learned fear during Pavlovian fear conditioning, and individuals with PTSD show fear associated with such conditioning (Mahan and Ressler 2012). The parahippocampal gyrus is also related to memory encoding (Alkire et al. 1998; Brewer et al. 1998). Interestingly, the amygdala reportedly modulates memory processes linked with the hippocampus-parahippocampus complex (Packard et al. 1994). In line with these studies, a hyperactive limbic lobe, including the amygdala in individuals with PTSD, modulates memory through its connectivity with the parahippocampal gyrus and the frontal regions during emotionally arousing learning situations that mimic traumatic stress (Kilpatrick and Cahill 2003). Our results support the hypothesis that individuals with PTSD may show an exaggerated response to fearful stimuli owing to hyperresponsiveness of the amygdala and related structures including the parahippocampal gyrus.
In contrast, individuals with PTSD showed less activation in the bilateral ACC, the key interface region involving the dorsal and ventral ACC (emotion domain, NC > PTSD, both ACC in whole brain analysis). Activation of the ACC is associated with decreased limbic activity, perhaps explaining the resolution of emotional conflict at the cortical level coupled with the top-down inhibition of limbic activity (Etkin et al. 2006). Indeed, the ACC relays projections from higher-order sensory areas to other regions of the prefrontal cortex and subcortical striatum (Johnson et al. 2003; Gunaydin and Kreitzer 2016), as well as the amygdala (Etkin et al. 2006).
It is quite important to understand the dynamic interplay between emotion and cognition. The lentiform nucleus (globus pallidus and putamen), parahippocampal gyrus and amygdala all showed greater activity in the PTSD than NC group when both the emotional and cognitive systems were jointly probed the interface domain (interface domain, PTSD>NC, lentiform nucleus, lateral & medial globus pallidus, parahippocampal gyrus, amygdala, putamen in whole brain analysis & whole brain + region of interest analysis). The PTSD group showed greater activity in amygdala than the normal control group. Other recent meta-analysis of imaging studies in the PTSD subjects showed that amygdala and mid-anterior cingulate cortex is hyperactive, whereas lateral and medial prefrontal cortex is hypoactive in that group for negative emotional stimuli vs. neutral or positive stimuli (Hayes et al. 2012a). A neurocircuitry model of PTSD shows that dysfunction of the ventromedial prefrontal cortex results in failure to inhibit an overactive amygdala, leading to an exaggerated fear response and impaired fear extinction learning (Hayes et al. 2012b).
The basal ganglia, especially the striatum, is involved in concentration and emotional processing along with the amygdala (Wise et al. 1996; Hollerman et al. 2000). The striatum is also a critical component of the motor and reward systems. Motor activity and planning, as moderated by the globus pallidus and striatum including putamen, where significant activity differences were shown in this meta-analysis, may explain why individuals with PTSD are unable to take action or avoidance measures in the context of emotional arousal (Stark et al. 2015). Unfortunately, our data could not afford to confirm this in the present study, as we could not address the correlation with specific PTSD clinical symptoms.
The small number of individual studies that were available for inclusion in this meta-analysis posed some limitations. First, the control group consisted of both healthy volunteers and trauma-exposed controls without clinical symptoms. Second, we presented the combined results of the ROI analyses and whole-brain analyses to assess the involvement of the hypothesized regions as simply mixing the ROI activation results with whole-brain scanning can bring the overrepresentation of specific ROI activations. However, we presented the combined results of whole brain and ROI analysis only as an option in addition to the whole brain analysis results. Finally, we could not provide a developmental perspective, as only two of the included studies specifically evaluated youths with PTSD.
In conclusion, the findings from this meta-analysis imply that individuals with PTSD process emotion and cognition in the subcortical regions, including the striatum, and in the limbic regions, including the amygdala, when the emotional system is probed with and without the cognitive effort, respectively. We also found that individuals with PTSD exhibited prominent underactivity in the ACC, especially when processing emotions, suggesting that PTSD may be an emotional disorder. These findings underscore the mechanistic dysfunction in key subcortical regions that potentially explain the emotional and cognitive distress experienced by clinically ill individuals diagnosed with PTSD.
Data availability
Data are available on request.
References
Alkire, M. T., Haier, R. J., Fallon, J. H., & Cahill, L. (1998). Hippocampal, but not amygdala, activity at encoding correlates with long-term, free recall of nonemotional information. Proceedings of the National Academy of Sciences of the United States of America, 95(24), 14506–14510.
Astur, R. S., St Germain, S. A., Tolin, D., Ford, J., Russell, D., & Stevens, M. (2006). Hippocampus function predicts severity of post-traumatic stress disorder. Cyberpsychology & Behavior, 9(2), 234–240. https://doi.org/10.1089/cpb.2006.9.234.
Atwoli, L., Stein, D. J., Koenen, K. C., & McLaughlin, K. A. (2015). Epidemiology of posttraumatic stress disorder: Prevalence, correlates and consequences. Current Opinion in Psychiatry, 28(4), 307–311. https://doi.org/10.1097/YCO.0000000000000167.
Aupperle, R. L., Allard, C. B., Grimes, E. M., Simmons, A. N., Flagan, T., Behrooznia, M., et al. (2012). Dorsolateral prefrontal cortex activation during emotional anticipation and neuropsychological performance in posttraumatic stress disorder. Archives of General Psychiatry, 69(4), 360–371. https://doi.org/10.1001/archgenpsychiatry.2011.1539.
Blair, K. S., Vythilingam, M., Crowe, S. L., McCaffrey, D. E., Ng, P., Wu, C. C., Scaramozza, M., Mondillo, K., Pine, D. S., Charney, D. S., & Blair, R. J. R. (2013). Cognitive control of attention is differentially affected in trauma-exposed individuals with and without post-traumatic stress disorder. Psychological Medicine, 43(1), 85–95. https://doi.org/10.1017/s0033291712000840.
Bluhm, R. L., Frewen, P. A., Coupland, N. C., Densmore, M., Schore, A. N., & Lanius, R. A. (2012). Neural correlates of self-reflection in post-traumatic stress disorder. Acta Psychiatrica Scandinavica, 125(3), 238–246. https://doi.org/10.1111/j.1600-0447.2011.01773.x.
Bremner, J. D. (2007). Neuroimaging in posttraumatic stress disorder and other stress-related disorders. Neuroimaging Clinics of North America, 17(4), 523–538, ix. https://doi.org/10.1016/j.nic.2007.07.003.
Brett, M., Johnsrude, I. S., & Owen, A. M. (2002). The problem of functional localization in the human brain. Nature Reviews. Neuroscience, 3(3), 243–249. https://doi.org/10.1038/nrn756.
Brewer, J. B., Zhao, Z., Desmond, J. E., Glover, G. H., & Gabrieli, J. D. (1998). Making memories: Brain activity that predicts how well visual experience will be remembered. Science, 281(5380), 1185–1187.
Brunetti, M., Sepede, G., Mingoia, G., Catani, C., Ferretti, A., Merla, A., del Gratta, C., Romani, G. L., & Babiloni, C. (2010). Elevated response of human amygdala to neutral stimuli in mild post traumatic stress disorder: Neural correlates of generalized emotional response. Neuroscience, 168(3), 670–679. https://doi.org/10.1016/j.neuroscience.2010.04.024.
Bryant, R. A., Felmingham, K. L., Kemp, A. H., Barton, M., Peduto, A. S., Rennie, C., Gordon, E., & Williams, L. M. (2005). Neural networks of information processing in posttraumatic stress disorder: A functional magnetic resonance imaging study. Biological Psychiatry, 58(2), 111–118. https://doi.org/10.1016/j.biopsych.2005.03.021.
Bryant, R. A., Kemp, A. H., Felmingham, K. L., Liddell, B., Olivieri, G., Peduto, A., Gordon, E., & Williams, L. M. (2008). Enhanced amygdala and medial prefrontal activation during nonconscious processing of fear in posttraumatic stress disorder: An fMRI study. Human Brain Mapping, 29(5), 517–523. https://doi.org/10.1002/hbm.20415.
Cisler, J. M., Bush, K., Scott Steele, J., Lenow, J. K., Smitherman, S., & Kilts, C. D. (2015). Brain and behavioral evidence for altered social learning mechanisms among women with assault-related posttraumatic stress disorder. Journal of Psychiatric Research, 63, 75–83. https://doi.org/10.1016/j.jpsychires.2015.02.014.
Eickhoff, S. B., Laird, A. R., Grefkes, C., Wang, L. E., Zilles, K., & Fox, P. T. (2009). Coordinate-based activation likelihood estimation meta-analysis of neuroimaging data: A random-effects approach based on empirical estimates of spatial uncertainty. Human Brain Mapping, 30(9), 2907–2926. https://doi.org/10.1002/hbm.20718.
Eickhoff, S. B., Bzdok, D., Laird, A. R., Kurth, F., & Fox, P. T. (2012). Activation likelihood estimation meta-analysis revisited. Neuroimage, 59(3), 2349–2361. https://doi.org/10.1016/j.neuroimage.2011.09.017.
El Khoury-Malhame, M., Reynaud, E., Soriano, A., Michael, K., Salgado-Pineda, P., Zendjidjian, X., et al. (2011). Amygdala activity correlates with attentional bias in PTSD. Neuropsychologia, 49(7), 1969–1973. https://doi.org/10.1016/j.neuropsychologia.2011.03.025.
Elman, I., Lowen, S., Frederick, B. B., Chi, W., Becerra, L., & Pitman, R. K. (2009). Functional neuroimaging of reward circuitry responsivity to monetary gains and losses in posttraumatic stress disorder. Biological Psychiatry, 66(12), 1083–1090. https://doi.org/10.1016/j.biopsych.2009.06.006.
Etkin, A., Egner, T., Peraza, D. M., Kandel, E. R., & Hirsch, J. (2006). Resolving emotional conflict: A role for the rostral anterior cingulate cortex in modulating activity in the amygdala. Neuron, 51(6), 871–882. https://doi.org/10.1016/j.neuron.2006.07.029.
Falconer, E., Bryant, R., Felmingham, K. L., Kemp, A. H., Gordon, E., Peduto, A., Olivieri, G., & Williams, L. M. (2008). The neural networks of inhibitory control in posttraumatic stress disorder. Journal of Psychiatry & Neuroscience, 33(5), 413–422.
Fani, N., Jovanovic, T., Ely, T. D., Bradley, B., Gutman, D., Tone, E. B., & Ressler, K. J. (2012). Neural correlates of attention bias to threat in post-traumatic stress disorder. Biological Psychology, 90(2), 134–142. https://doi.org/10.1016/j.biopsycho.2012.03.001.
Felmingham, K. L., Williams, L. M., Kemp, A. H., Rennie, C., Gordon, E., & Bryant, R. A. (2009). Anterior cingulate activity to salient stimuli is modulated by autonomic arousal in posttraumatic stress disorder. Psychiatry Research, 173(1), 59–62. https://doi.org/10.1016/j.pscychresns.2008.12.005.
Felmingham, K. L., Falconer, E. M., Williams, L., Kemp, A. H., Allen, A., Peduto, A., & Bryant, R. A. (2014). Reduced amygdala and ventral striatal activity to happy faces in PTSD is associated with emotional numbing. PLoS One, 9(9), e103653. https://doi.org/10.1371/journal.pone.0103653.
Fonzo, G. A., Simmons, A. N., Thorp, S. R., Norman, S. B., Paulus, M. P., & Stein, M. B. (2010). Exaggerated and disconnected insular-amygdalar blood oxygenation level-dependent response to threat-related emotional faces in women with intimate-partner violence posttraumatic stress disorder. Biological Psychiatry, 68(5), 433–441. https://doi.org/10.1016/j.biopsych.2010.04.028.
Frewen, P. A., Dozois, D. J., Neufeld, R. W., Lane, R. D., Densmore, M., Stevens, T. K., et al. (2012). Emotional numbing in posttraumatic stress disorder: A functional magnetic resonance imaging study. The Journal of Clinical Psychiatry, 73(4), 431–436. https://doi.org/10.4088/JCP.10m06477.
Garrett, A. S., Carrion, V., Kletter, H., Karchemskiy, A., Weems, C. F., & Reiss, A. (2012). Brain activation to facial expressions in youth with PTSD symptoms. Depression and Anxiety, 29(5), 449–459. https://doi.org/10.1002/da.21892.
Geuze, E., Vermetten, E., Ruf, M., de Kloet, C. S., & Westenberg, H. G. (2008). Neural correlates of associative learning and memory in veterans with posttraumatic stress disorder. Journal of Psychiatric Research, 42(8), 659–669. https://doi.org/10.1016/j.jpsychires.2007.06.007.
Gunaydin, L. A., & Kreitzer, A. C. (2016). Cortico-basal ganglia circuit function in psychiatric disease. Annual Review of Physiology, 78, 327–350. https://doi.org/10.1146/annurev-physiol-021115-105355.
Hayes, J. P., LaBar, K. S., McCarthy, G., Selgrade, E., Nasser, J., Dolcos, F., et al. (2011). Reduced hippocampal and amygdala activity predicts memory distortions for trauma reminders in combat-related PTSD. Journal of Psychiatric Research, 45(5), 660–669. https://doi.org/10.1016/j.jpsychires.2010.10.007.
Hayes, J. P., Hayes, S. M., & Mikedis, A. M. (2012a). Quantitative meta-analysis of neural activity in posttraumatic stress disorder. Biol Mood Anxiety Disord, 2, 9. https://doi.org/10.1186/2045-5380-2-9.
Hayes, J. P., Vanelzakker, M. B., & Shin, L. M. (2012b). Emotion and cognition interactions in PTSD: A review of neurocognitive and neuroimaging studies. Frontiers in Integrative Neuroscience, 6, 89. https://doi.org/10.3389/fnint.2012.00089.
Hollerman, J. R., Tremblay, L., & Schultz, W. (2000). Involvement of basal ganglia and orbitofrontal cortex in goal-directed behavior. Progress in Brain Research, 126, 193–215. https://doi.org/10.1016/s0079-6123(00)26015-9.
Hou, C., Liu, J., Wang, K., Li, L., Liang, M., He, Z., Liu, Y., Zhang, Y., Li, W., & Jiang, T. (2007). Brain responses to symptom provocation and trauma-related short-term memory recall in coal mining accident survivors with acute severe PTSD. Brain Research, 1144, 165–174. https://doi.org/10.1016/j.brainres.2007.01.089.
Jatzko, A., Schmitt, A., Demirakca, T., Weimer, E., & Braus, D. F. (2006). Disturbance in the neural circuitry underlying positive emotional processing in post-traumatic stress disorder (PTSD). An fMRI study. European Archives of Psychiatry and Clinical Neuroscience, 256(2), 112–114. https://doi.org/10.1007/s00406-005-0617-3.
Johnson, P. A., Hurley, R. A., Benkelfat, C., Herpertz, S. C., & Taber, K. H. (2003). Understanding emotion regulation in borderline personality disorder: Contributions of neuroimaging. Journal of Neuropsychiatry and Clinical Neurosciences, 15(4), 397–402. https://doi.org/10.1176/jnp.15.4.397.
Jovanovic, T., Ely, T., Fani, N., Glover, E. M., Gutman, D., Tone, E. B., Norrholm, S. D., Bradley, B., & Ressler, K. J. (2013). Reduced neural activation during an inhibition task is associated with impaired fear inhibition in a traumatized civilian sample. Cortex, 49(7), 1884–1891. https://doi.org/10.1016/j.cortex.2012.08.011.
Kemp, A. H., Felmingham, K. L., Falconer, E., Liddell, B. J., Bryant, R. A., & Williams, L. M. (2009). Heterogeneity of non-conscious fear perception in posttraumatic stress disorder as a function of physiological arousal: An fMRI study. Psychiatry Research, 174(2), 158–161. https://doi.org/10.1016/j.pscychresns.2009.04.012.
Kilpatrick, L., & Cahill, L. (2003). Amygdala modulation of parahippocampal and frontal regions during emotionally influenced memory storage. Neuroimage, 20(4), 2091–2099.
Kim, M. J., Chey, J., Chung, A., Bae, S., Khang, H., Ham, B., Yoon, S. J., Jeong, D. U., & Lyoo, I. K. (2008). Diminished rostral anterior cingulate activity in response to threat-related events in posttraumatic stress disorder. Journal of Psychiatric Research, 42(4), 268–277. https://doi.org/10.1016/j.jpsychires.2007.02.003.
Kober, H., & Wager, T. D. (2010). Meta-analysis of neuroimaging data. Wiley Interdisciplinary Reviews: Cognitive Science, 1(2), 293–300. https://doi.org/10.1002/wcs.41.
Laird, A. R., Fox, P. M., Price, C. J., Glahn, D. C., Uecker, A. M., Lancaster, J. L., Turkeltaub, P. E., Kochunov, P., & Fox, P. T. (2005). ALE meta-analysis: Controlling the false discovery rate and performing statistical contrasts. Human Brain Mapping, 25(1), 155–164. https://doi.org/10.1002/hbm.20136.
Laird, A. R., Eickhoff, S. B., Kurth, F., Fox, P. M., Uecker, A. M., Turner, J. A., et al. (2009). ALE meta-analysis workflows via the Brainmap database: Progress towards a probabilistic functional brain atlas. Frontiers in Neuroinformatics, 3, 23. https://doi.org/10.3389/neuro.11.023.2009.
Landre, L., Destrieux, C., Andersson, F., Barantin, L., Quide, Y., Tapia, G., et al. (2012). Working memory processing of traumatic material in women with posttraumatic stress disorder. Journal of Psychiatry & Neuroscience, 37(2), 87–94. https://doi.org/10.1503/jpn.100167.
Lanius, R. A., Williamson, P. C., Densmore, M., Boksman, K., Gupta, M. A., Neufeld, R. W., Gati, J. S., & Menon, R. S. (2001). Neural correlates of traumatic memories in posttraumatic stress disorder: A functional MRI investigation. The American Journal of Psychiatry, 158(11), 1920–1922. https://doi.org/10.1176/appi.ajp.158.11.1920.
Lanius, R. A., Williamson, P. C., Boksman, K., Densmore, M., Gupta, M., Neufeld, R. W., et al. (2002). Brain activation during script-driven imagery induced dissociative responses in PTSD: A functional magnetic resonance imaging investigation. Biological Psychiatry, 52(4), 305–311.
Lanius, R. A., Williamson, P. C., Hopper, J., Densmore, M., Boksman, K., Gupta, M. A., Neufeld, R. W. J., Gati, J. S., & Menon, R. S. (2003). Recall of emotional states in posttraumatic stress disorder: An fMRI investigation. Biological Psychiatry, 53(3), 204–210.
Lanius, R. A., Vermetten, E., Loewenstein, R. J., Brand, B., Schmahl, C., Bremner, J. D., & Spiegel, D. (2010). Emotion modulation in PTSD: Clinical and neurobiological evidence for a dissociative subtype. The American Journal of Psychiatry, 167(6), 640–647. https://doi.org/10.1176/appi.ajp.2009.09081168.
Mahan, A. L., & Ressler, K. J. (2012). Fear conditioning, synaptic plasticity and the amygdala: Implications for posttraumatic stress disorder. Trends in Neurosciences, 35(1), 24–35. https://doi.org/10.1016/j.tins.2011.06.007.
Mazza, M., Catalucci, A., Mariano, M., Pino, M. C., Tripaldi, S., Roncone, R., & Gallucci, M. (2012). Neural correlates of automatic perceptual sensitivity to facial affect in posttraumatic stress disorder subjects who survived L'Aquila eartquake of April 6, 2009. Brain Imaging and Behavior, 6(3), 374–386. https://doi.org/10.1007/s11682-012-9151-x.
Mazza, M., Tempesta, D., Pino, M. C., Nigri, A., Catalucci, A., Guadagni, V., Gallucci, M., Iaria, G., & Ferrara, M. (2015). Neural activity related to cognitive and emotional empathy in post-traumatic stress disorder. Behavioural Brain Research, 282, 37–45. https://doi.org/10.1016/j.bbr.2014.12.049.
Mickleborough, M. J., Daniels, J. K., Coupland, N. J., Kao, R., Williamson, P. C., Lanius, U. F., Hegadoren, K., Schore, A., Densmore, M., Stevens, T., & Lanius, R. A. (2011). Effects of trauma-related cues on pain processing in posttraumatic stress disorder: An fMRI investigation. Journal of Psychiatry & Neuroscience, 36(1), 6–14. https://doi.org/10.1503/jpn.080188.
Moher, D., Liberati, A., Tetzlaff, J., Altman, D. G., & PRISMA Group. (2009). Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. BMJ (Clinical Research Ed.), 339, b2535. https://doi.org/10.1136/bmj.b2535.
Moores, K. A., Clark, C. R., McFarlane, A. C., Brown, G. C., Puce, A., & Taylor, D. J. (2008). Abnormal recruitment of working memory updating networks during maintenance of trauma-neutral information in post-traumatic stress disorder. Psychiatry Research, 163(2), 156–170. https://doi.org/10.1016/j.pscychresns.2007.08.011.
Moser, D. A., Aue, T., Wang, Z., Rusconi Serpa, S., Favez, N., Peterson, B. S., & Schechter, D. S. (2013). Limbic brain responses in mothers with post-traumatic stress disorder and comorbid dissociation to video clips of their children. Stress, 16(5), 493–502. https://doi.org/10.3109/10253890.2013.816280.
Moser, D. A., Aue, T., Suardi, F., Kutlikova, H., Cordero, M. I., Rossignol, A. S., Favez, N., Rusconi Serpa, S., & Schechter, D. S. (2015). Violence-related PTSD and neural activation when seeing emotionally charged male-female interactions. Social Cognitive and Affective Neuroscience, 10(5), 645–653. https://doi.org/10.1093/scan/nsu099.
New, A. S., Fan, J., Murrough, J. W., Liu, X., Liebman, R. E., Guise, K. G., Tang, C. Y., & Charney, D. S. (2009). A functional magnetic resonance imaging study of deliberate emotion regulation in resilience and posttraumatic stress disorder. Biological Psychiatry, 66(7), 656–664. https://doi.org/10.1016/j.biopsych.2009.05.020.
Packard, M. G., Cahill, L., & McGaugh, J. L. (1994). Amygdala modulation of hippocampal-dependent and caudate nucleus-dependent memory processes. Proceedings of the National Academy of Sciences of the United States of America, 91(18), 8477–8481.
Pavuluri, M. N., Passarotti, A. M., Parnes, S. A., Fitzgerald, J. M., & Sweeney, J. A. (2010). A pharmacological functional magnetic resonance imaging study probing the interface of cognitive and emotional brain systems in pediatric bipolar disorder. Journal of Child and Adolescent Psychopharmacology, 20(5), 395–406. https://doi.org/10.1089/cap.2009.0105.
Protopopescu, X., Pan, H., Tuescher, O., Cloitre, M., Goldstein, M., Engelien, W., Epstein, J., Yang, Y., Gorman, J., LeDoux, J., Silbersweig, D., & Stern, E. (2005). Differential time courses and specificity of amygdala activity in posttraumatic stress disorder subjects and normal control subjects. Biological Psychiatry, 57(5), 464–473. https://doi.org/10.1016/j.biopsych.2004.12.026.
Rabinak, C. A., MacNamara, A., Kennedy, A. E., Angstadt, M., Stein, M. B., Liberzon, I., & Phan, K. L. (2014). Focal and aberrant prefrontal engagement during emotion regulation in veterans with posttraumatic stress disorder. Depression and Anxiety, 31(10), 851–861. https://doi.org/10.1002/da.22243.
Rauch, S. L., Whalen, P. J., Shin, L. M., McInerney, S. C., Macklin, M. L., Lasko, N. B., Orr, S. P., & Pitman, R. K. (2000). Exaggerated amygdala response to masked facial stimuli in posttraumatic stress disorder: A functional MRI study. Biological Psychiatry, 47(9), 769–776.
Rogan, M. T., Staubli, U. V., & LeDoux, J. E. (1997). Fear conditioning induces associative long-term potentiation in the amygdala. Nature, 390(6660), 604–607. https://doi.org/10.1038/37601.
Sailer, U., Robinson, S., Fischmeister, F. P., Konig, D., Oppenauer, C., Lueger-Schuster, B., et al. (2008). Altered reward processing in the nucleus accumbens and mesial prefrontal cortex of patients with posttraumatic stress disorder. Neuropsychologia, 46(11), 2836–2844. https://doi.org/10.1016/j.neuropsychologia.2008.05.022.
Schechter, D. S., Moser, D. A., Wang, Z., Marsh, R., Hao, X., Duan, Y., Yu, S., Gunter, B., Murphy, D., McCaw, J., Kangarlu, A., Willheim, E., Myers, M. M., Hofer, M. A., & Peterson, B. S. (2012). An fMRI study of the brain responses of traumatized mothers to viewing their toddlers during separation and play. Social Cognitive and Affective Neuroscience, 7(8), 969–979. https://doi.org/10.1093/scan/nsr069.
Shaw, M. E., Moores, K. A., Clark, R. C., McFarlane, A. C., Strother, S. C., Bryant, R. A., Brown, G. C., & Taylor, J. D. (2009). Functional connectivity reveals inefficient working memory systems in post-traumatic stress disorder. Psychiatry Research, 172(3), 235–241. https://doi.org/10.1016/j.pscychresns.2008.07.014.
Shin, L. M., Whalen, P. J., Pitman, R. K., Bush, G., Macklin, M. L., Lasko, N. B., Orr, S. P., McInerney, S. C., & Rauch, S. L. (2001). An fMRI study of anterior cingulate function in posttraumatic stress disorder. Biological Psychiatry, 50(12), 932–942.
Shin, L. M., Wright, C. I., Cannistraro, P. A., Wedig, M. M., McMullin, K., Martis, B., Macklin, M. L., Lasko, N. B., Cavanagh, S. R., Krangel, T. S., Orr, S. P., Pitman, R. K., Whalen, P. J., & Rauch, S. L. (2005). A functional magnetic resonance imaging study of amygdala and medial prefrontal cortex responses to overtly presented fearful faces in posttraumatic stress disorder. Archives of General Psychiatry, 62(3), 273–281. https://doi.org/10.1001/archpsyc.62.3.273.
Simmons, A. N., Paulus, M. P., Thorp, S. R., Matthews, S. C., Norman, S. B., & Stein, M. B. (2008). Functional activation and neural networks in women with posttraumatic stress disorder related to intimate partner violence. Biological Psychiatry, 64(8), 681–690. https://doi.org/10.1016/j.biopsych.2008.05.027.
Simmons, A. N., Flagan, T. M., Wittmann, M., Strigo, I. A., Matthews, S. C., Donovan, H., Lohr, J. B., & Paulus, M. P. (2013). The effects of temporal unpredictability in anticipation of negative events in combat veterans with PTSD. Journal of Affective Disorders, 146(3), 426–432. https://doi.org/10.1016/j.jad.2012.08.006.
St Jacques, P. L., Botzung, A., Miles, A., & Rubin, D. C. (2011). Functional neuroimaging of emotionally intense autobiographical memories in post-traumatic stress disorder. Journal of Psychiatric Research, 45(5), 630–637. https://doi.org/10.1016/j.jpsychires.2010.10.011.
Stark, E. A., Parsons, C. E., Van Hartevelt, T. J., Charquero-Ballester, M., McManners, H., Ehlers, A., et al. (2015). Post-traumatic stress influences the brain even in the absence of symptoms: A systematic, quantitative meta-analysis of neuroimaging studies. Neuroscience and Biobehavioral Reviews, 56, 207–221. https://doi.org/10.1016/j.neubiorev.2015.07.007.
Steuwe, C., Daniels, J. K., Frewen, P. A., Densmore, M., Pannasch, S., Beblo, T., Reiss, J., & Lanius, R. A. (2014). Effect of direct eye contact in PTSD related to interpersonal trauma: An fMRI study of activation of an innate alarm system. Social Cognitive and Affective Neuroscience, 9(1), 88–97. https://doi.org/10.1093/scan/nss105.
Stevens, J. S., Jovanovic, T., Fani, N., Ely, T. D., Glover, E. M., Bradley, B., & Ressler, K. J. (2013). Disrupted amygdala-prefrontal functional connectivity in civilian women with posttraumatic stress disorder. Journal of Psychiatric Research, 47(10), 1469–1478. https://doi.org/10.1016/j.jpsychires.2013.05.031.
Strigo, I. A., Simmons, A. N., Matthews, S. C., Grimes, E. M., Allard, C. B., Reinhardt, L. E., Paulus, M. P., & Stein, M. B. (2010). Neural correlates of altered pain response in women with posttraumatic stress disorder from intimate partner violence. Biological Psychiatry, 68(5), 442–450. https://doi.org/10.1016/j.biopsych.2010.03.034.
Thomaes, K., Dorrepaal, E., Draijer, N. P., de Ruiter, M. B., Elzinga, B. M., van Balkom, A. J., et al. (2009). Increased activation of the left hippocampus region in complex PTSD during encoding and recognition of emotional words: A pilot study. Psychiatry Research, 171(1), 44–53. https://doi.org/10.1016/j.pscychresns.2008.03.003.
Thomaes, K., Dorrepaal, E., Draijer, N., de Ruiter, M. B., Elzinga, B. M., Sjoerds, Z., van Balkom, A. J., Smit, J. H., & Veltman, D. J. (2013). Increased anterior cingulate cortex and hippocampus activation in complex PTSD during encoding of negative words. Social Cognitive and Affective Neuroscience, 8(2), 190–200. https://doi.org/10.1093/scan/nsr084.
Turkeltaub, P. E., Eickhoff, S. B., Laird, A. R., Fox, M., Wiener, M., & Fox, P. (2012). Minimizing within-experiment and within-group effects in activation likelihood estimation meta-analyses. Human Brain Mapping, 33(1), 1–13. https://doi.org/10.1002/hbm.21186.
van Rooij, S. J., Rademaker, A. R., Kennis, M., Vink, M., Kahn, R. S., & Geuze, E. (2015). Neural correlates of trauma-unrelated emotional processing in war veterans with PTSD. Psychological Medicine, 45(3), 575–587. https://doi.org/10.1017/s0033291714001706.
Whalley, M. G., Rugg, M. D., Smith, A. P., Dolan, R. J., & Brewin, C. R. (2009). Incidental retrieval of emotional contexts in post-traumatic stress disorder and depression: An fMRI study. Brain and Cognition, 69(1), 98–107. https://doi.org/10.1016/j.bandc.2008.05.008.
Wise, S. P., Murray, E. A., & Gerfen, C. R. (1996). The frontal cortex-basal ganglia system in primates. Critical Reviews in Neurobiology, 10(3–4), 317–356.
Xiong, K., Zhang, Y., Qiu, M., Zhang, J., Sang, L., Wang, L., Xie, B., Wang, J., & Li, M. (2013). Negative emotion regulation in patients with posttraumatic stress disorder. PLoS One, 8(12), e81957. https://doi.org/10.1371/journal.pone.0081957.
Yang, P., Wu, M. T., Hsu, C. C., & Ker, J. H. (2004). Evidence of early neurobiological alternations in adolescents with posttraumatic stress disorder: A functional MRI study. Neuroscience Letters, 370(1), 13–18. https://doi.org/10.1016/j.neulet.2004.07.033.
Zhang, J. N., Xiong, K. L., Qiu, M. G., Zhang, Y., Xie, B., Wang, J., Li, M., Chen, H., Zhang, Y., & Zhang, J. J. (2013). Negative emotional distraction on neural circuits for working memory in patients with posttraumatic stress disorder. Brain Research, 1531, 94–101. https://doi.org/10.1016/j.brainres.2013.07.042.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All the authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. This article does not contain any studies with animals performed by any of the authors.
Informed consent
This article does not contain any studies with human participants performed by any of the authors.
Role of the sponsor
None.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lee, MS., Anumagalla, P. & Pavuluri, M.N. Individuals with the post-traumatic stress disorder process emotions in subcortical regions irrespective of cognitive engagement: a meta-analysis of cognitive and emotional interface. Brain Imaging and Behavior 15, 941–957 (2021). https://doi.org/10.1007/s11682-020-00303-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11682-020-00303-9