Abstract
Differences in forest attributes and carbon sequestration of each organ and layer between broadleaved and conifer forests of central and outer urban areas are not well-defined, hindering the precise management of urban forests and improvement of function. To clarify the effect of two forest types with different urbanization intensities, we determined differences in vegetation composition and diversity, structural traits, and carbon stocks of 152 plots (20 m × 20 m) in urban park forests in Changchun, which had the largest green quantity and carbon density effectiveness. We found that 1.1-fold thicker and healthier trees, and 1.6- to 2.0-fold higher, healthier, denser, and more various shrubs but with sparser trees and herbs occurred in the central urban forests (p < 0.05) than in the outer forests. The conifer forests exhibited 30–70% obviously higher tree aboveground carbon sequestration (including stem and leaf) and 20% bigger trees, especially in the outer forests (p < 0.05). In contrast, 1.1- to 1.5-fold higher branch stocks, healthier and more diverse trees were found in broadleaved forests of both the inner and outer forests (p < 0.05). Plant size and dominant species had similarly important roles in carbon stock improvement, especially big-sized woody plants and Pinus tabuliformis. In addition, a higher number of deciduous or needle species positively affected the broadleaved forest of the central urban area and conifer forest of the outer urban area, respectively. These findings can be used to guide precise management and accelerate the improvement of urban carbon function in Northeast China in the future.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Since the industrial revolution, technological advances and rapid urban development have been prevalent, resulting in increased atmospheric greenhouse gases, such as CO2, and these global changes threaten anthropogenic development (Seto and Shepherd 2009; Kashiwagi 2016). Urban areas are essential locations where human populations are directly affected by the deteriorating environment (Mitchell et al. 2018). Urban forests have played a crucial role in reducing atmospheric CO2 and improving the urban environment through storing excess carbon as biomass, and increasing biomass and organic matter accumulation (Main-Knorn et al. 2011; Ni 2013).
Although many innovative methods have been applied to resolve urbanization problems, such as terrestrial laser scanning and ENVI-met software to estimate vegetation structural traits and microclimate for specific regions (Wu et al. 2022; Wang et al. 2023), field survey investigation has been found to be more reliable and irreplaceable in terms of providing detailed data on each layer. Forest vegetation composition and community structural characteristics are the foundation of terrestrial ecosystems, which support green infrastructure for cities (Wang et al. 2019a). Moreover, shrubs and herbs with a relatively small contribution to urban forests compared with trees, which also play an indispensable role in urban greenspace, should not be underestimated (Wang et al. 2021b). A comprehensive understanding of the composition and diversity of taxonomic species among the forest vegetation, as well as structural characteristics at different vertical layers, is conducive to providing a holistic view of the forest attributes and reveal the mechanisms underlying functional improvement of the forest ecosystem (Wang et al. 2020a, 2021a). In urban forests with extreme fragmentation and manual management interference, the forest assembly, community traits, and carbon function were found to be more complex between different species and categories (Escobedo et al. 2011; Gratani et al. 2016). Broadleaf and conifer forests are common species categories in plantations and natural forests (Marshall and Waring 1984), and comparison between the two forest types was found to be beneficial in revealing forest ecosystem function and successional development (Liu et al. 2013). Therefore, the effect on forest attributes and carbon sink function of a plant arrangement dominated by broadleaf and conifer trees is deemed worth exploring.
Proper forestry management effectively mitigates increasing atmospheric CO2 concentrations in addressing serious environmental challenges (Zhang et al. 2013; Carretero et al. 2017). Decoupling the complex relationship among urban forest communities (species composition and diversity, and plant size) and ecosystem function (carbon sequestrated) with different urbanization intensity supports the suitable management to reach standardized and accept conclusions for urban forests. Selecting an appropriate statistical analysis method is crucial to decoupling the associations among dominant species, diversity, and forest structure, as well as tree and shrub carbon stocks, to discover the underlying mechanism of the improvement of carbon sink function. Redundancy analysis (RDA) has been used to identify these complex associations (Zhang et al. 2018; Wang et al. 2018, 2019b). Forest attributes, including tree-size structures and leaf functional traits, in natural forests can play a crucial role in aboveground biomass or productivity (Ali et al. 2018; Dong et al. 2019). Among these, the “big-sized tree hypothesis” has recently been a topic of discussion (Ali et al. 2019). In previous studies, the top 1% of larger-size trees in individual plots was used to indicate the big‐sized trees, which could better explain the association among species composition, richness, and aboveground biomass in natural forests (Lutz et al. 2018; Ali et al. 2019), and the percentile scores of larger vegetation were attributed to statistical stability, such as high R2 and low root mean square error (RMSE). However, urban forests are usually heterogeneous, fragmented, scattered, and surrounded by many impervious surfaces compared with natural forests (Shen et al. 2020). The morphological traits, functions, and structure of vegetation provide a wide range of ecosystem services and benefits that can alleviate the adverse effects of urbanization (Lin et al. 2019).
Changchun, the capital city of Jilin Province in China, is important in the urban agglomeration of northeastern China and has many adventitious plants from Changbai Mountain. Changchun is considered a “Forest City,” the studies on its urban forest are necessary. In this study, broadleaved and coniferous forests located in urban parks of Changchun’s inner and outer urban areas were investigated in detail. We aimed to achieve the following objectives:
-
(1)
Clarify the effect of urbanization and forest-type effect on forest attributes trees, shrubs, and herbs and their carbon stocks;
-
(2)
Explore the association among carbon stocks, dominant vegetation composition, structural characteristics, and species diversity;
-
(3)
Assess possible carbon sequestration improvement from species and structural trait perspectives and future implications.
This study precisely evaluated carbon sink function and provided active measures for urban forest management (Technical route in Fig. 1). The results are expected to be instructive for similar greenspaces planned in the future.
Materials and methods
Study area
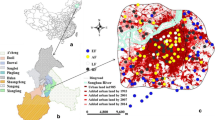
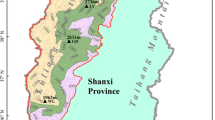
The study area was located within the ring expressway of Changchun, Jilin Province, China (125°15′–125°29′ E, 43°45′–43°59′ N; Fig. 2). By the end of 2020, the permanent urban resident population was approximately 15 million, and the urbanization rate of the population was 62.64% (China Statistical Yearbook 2020). Changchun City is located in the hinterland of the Great Plains in Northeast China, with an average annual temperature of 4.8 °C and an average annual precipitation of 567 mm (Chang et al. 2022). Park forests, with the most abundant species and the highest carbon density in the urban forest, were the main forest type involved in this study (Wang et al. 2021b).
Experimental design, field investigation, and data collection
The stratified random sampling method was used to determine and allocate the number of sample plots. All sampling plots were located in urban parks, with the largest green amount and carbon density. Ring Roads were adopted as the dividing lines for investigating the urbanized areas based on Changchun’s concentric circle structure (Ma et al. 2021). A total of 152 plots were divided into inner (70 plots located in the inner area) and outer areas (82 plots located in the outer area) based on the urban development and location. Sampling sites on the ring-road 1 and 2 in the light green area belong to the inner urban area; sampling sites on the ring-road 3, 4 and 5, and suburban park in the dark green area belong to the outer area (Fig. 2). The broadleaved and conifer forests were defined as the relative abundance of trees of more than 50% of these two types. The number of broadleaved and conifer forests was 76 in the 152 plots. Among these, 41 broadleaved and 29 conifer forests were surveyed in the inner urban area, and 35 broadleaved and 47 conifer forests were found in the outer area.
Field surveys were conducted from June to August 2022. Each plot measured 20 m × 20 m. During the survey, plant species names and structural traits of trees, shrubs and herbs were investigated in detail. For trees, tree height (Th), the diameter at breast height (DBH), tree under crown height (Uch), tree crown diameter (Tcd), tree density (Td), and tree health degree (Thd) were recorded. For shrubs, shrub height (Sh), shrub crown diameter (Scd), shrub density (Sd), and shrub health degree (Shd) were measured. Moreover, the herb surveyed items included the relative coverage of each species (Hc), which was measured as the ratio of the area of the surveyed species to the total surveyed area in percentage. Plant density was calculated as the total individual quantity in each plot divided by the plot area. The health degree ranged from 0 to 4, according to the comprehensive evaluation of leaves, branches, stems, and individual plants. These grades were as follows: (0) dead, (1) dying, 76–99% of the crown was composed of dead branches (2) critical, 51–75% dead branches, (3) poor, 26–50% dead branches, and (4) excellent and fair, no prominent dead branches inside the tree crown (< 25%). Plot information such as canopy density, altitude, latitude, and longitude of each sampling plot were also recorded.
Species diversity calculation
Using the field survey data, four species diversity indices were calculated (Eqs. 1–4) (Ma et al. 1997):
where, Pi is the proportion of the number of species i to the total number of the species, and S is the total of species i in the sampling plot.
Structural parameter calculation
In the plot, the mean value of most community structural parameters was calculated using the general average formula outlined in Eq. 5, including tree diameter at DBH, Th, Uch, Tcd, Thd, Sh, Scd and Shd. Td and Sd refer to Eq. 6, and Hc refers to Eq. 7:
where, Dij is the structural index of the No. j tree in the No. ith species; m is the total species number; n is the measured tree for each species; ci is the coverage of ith species; and A is the area of the plot.
Dominant species abundance calculation
Dominant species were recognized by ranking all the species of the pooled data for all plots according to the important value. All species names and their quantities (relative abundance, frequency, prominence, and important value) are listed in the supplementary material (Tables S1 − S3), and the top five tree and shrub species were defined as the dominant species. The calculations of relative abundance, frequency, prominence, and important value are shown in Eqs. 8–11.
Subsequently, the relative abundance of dominant species (genera and families) in each plot was calculated as the average value of the proportion of the species individual quantity and the total individual amount of all species in each plot:
where, C, F and P are the relative coverage, frequency, and abundance of each species, respectively; m is total species number; Ci is the coverage of the ith species; Fi is the frequency of the ith species; and Pi is the abundance of the ith species.
Aboveground carbon storage calculation
The aboveground biomass was evaluated by species-specific allometric equations, using tree DBH, height, and shrub crown diameter, respectively (Hu et al. 2015; Fan et al. 2011; Wang et al. 2005, 2022). The total aboveground biomass was the sum of individual aboveground biomass of all plants in each plot and the aboveground carbon amount was derived by total aboveground biomass multiplied by a factor of 0.5 (Guo et al. 2010). Carbon stocks were calculated by total carbon storage per unit area in each plot (Mg ha−1).
Data processing and association decoupling
To identify the broadleaved versus conifer effects averaged among the central and outer urban forests, two-way analysis of variance (Two way ANOVA) was conducted for checking the interaction effect of forest type and location (inner and outer). The paired difference (broadleaf/conifer ratio of marginal-mean from Two way ANOVA; XB/C = XB/XC) was used to describe the differences between broadleaved and conifer forests, and the least statistical significances between two types are shown as *; the significant differences among different urbanization areas are shown in the top right corner of the associated figures; All analyses were performed using SPSS 22.0 (IBM SPSS, USA).
The top 50% of the big‐sized trees and 25% of big‐sized shrubs were used to quantify the effect of big‐sized trees and shrubs within each plot. The top 50% of big‐sized trees was quantified using the 50th percentile scores for tree diameter, height, and crown diameter within each plot. The top 25% of big‐sized shrubs was quantified using the 75th percentile shrub height and crown diameter scores within each plot. In this study, we used the 50th percentile score (50%) of tree DBH, height, and crown diameter to represent the big‐sized tree attributes because of the stability of high R2 and low RMSE values among the 50% tree diameter, height, crown diameter, and aboveground carbon density data (although R2 increased and RMSE decreased from 10 to 75%, the raise rate decreased from 50 to 75% attributes, see Figs. S1a–c). In addition, the 75th percentile score (25%) of shrub height and crown diameter was chosen to represent the big‐sized shrubs’ attributes owing to the relatively high R2 and lowest RMSE (Figs. S1d and e). The variation partitioning analysis of “Var-part-3 groups-Conditional-effects-tested” and forward selection were used to determine the most important part and significant factors. Canoco 5 software was used for RDA and variation partitioning analysis (Biometrics Ltd., Plant Research International, Netherlands).
Results
Urban forest vegetation composition and dominant species
In the park’s urban forest, 69 tree species belonging to 33 genera and 20 families were surveyed in our study. The most important tree species was Pinus tabuliformis, comprising 12.4% of all species, followed by Larix gmelinii and Pinus sylvestris, accounting for 10.7% and 7.9%, respectively. Quercus mongolica, Pinus koraiensis, and Ulmus pumila accounted for 5–7% (Fig. 3a, in bold). Phellodendron amurense, Prunus padus and Picea koraiensis accounted for 3.2–4.1% (Fig. 3a). Twenty-two shrub species belonging to 19 genera and 11 families were surveyed in our study, and the most important three shrub species in the urban forest were Lonicera japonica, Sambucus williamsii and Syringa oblate, which accounted for 11.9–24.4%, followed by Sorbaria sorbifolia and Acer tataricum, at 6–6.5% (Fig. 3b, in bold). Prunus triloba, Philadelphus schrenkii, and Lespedeza bicolo accounted for 4–5% (Fig. 3b). The top five species (shown in bold in the figure) were considered the dominant tree and shrub species in the urban forest according to the important value (IV).
Two-way analysis of variance of location and forest type
The interaction effect of forest type and location on structural traits, plant species diversity, and biomass carbon stocks of each organ were non-significant except for tree density (Table 1). Moreover, the main effect of forest type showed that biomass carbon stocks of tree leaf, stem, and aboveground of broadleaved forests accounted for 40–70% of conifer forests, and tree under crown height, tree height, and DBH in broadleaved forests accounted for 60–90% of the conifer forests. However, tree health degree and species diversity in broadleaved forests accounted for 1.2- to 1.7-fold of conifer forests (p < 0.05, Table 2 left). Larger and healthier tree and shrubs, and two- to three fold denser and more diverse shrubs but with only 70% of herb coverage and richness, were found in the inner area (p < 0.05, Table 2 right).
Tree height (Th), the diameter at breast height (DBH), tree crown diameter (Tcd), shrub height (Sh), shrub crown diameter (Scd), tree under crown height (Uch), tree health degree (Thd), tree density (Td), shrub health degree (Shd), shrub density (Sd), herb coverage (Hc), and herb Richness index (HS); TH, TD, TS, TJ and SH, SD, SS, SJ are the Shannon–Wiener, Simpson, richness and evenness indices of trees and shrubs. TCs, TCb, TCl, TCr are the carbon storage of tree stem, branch, leaf and root, respectively. Tcag is tree aboveground carbon storage.
Urbanization and different forest-type effects on carbon stocks
The mean carbon density of trees and shrubs in the central urban area was 1.0- to 1.3-fold of those in the outer area, except for root carbon density, and the differences were non-significant (Table 2). For tree aboveground and leaf carbon stocks, the significant differences between forest types, mainly attributed to the outer area, manifested as the carbon stocks of leaf and aboveground in broadleaved forests and only accounted for 30–50% of the conifer forests (Fig. 4a, b). Moreover, the differences of stem and branch carbon stocks were obvious in both the inner and outer areas (Fig. 4c, d). However, the root and shrub carbon stocks had no significant differences between forest types and locations (Fig. 4e, f).
Carbon density difference of each organ of trees as well as carbon sequestration of aboveground trees and shrubs between the broadleaved forests (B) and coniferous forests (C) of inner and outer urban forests. The y-coordinate is the mean value of the parameters, numbers above the error bars: broadleaf to conifer ratio (XB/C = XB/XC), error bars: standard error. TCs: tree carbon density of stem; TCb: tree carbon density of branch; TCl: tree carbon density of leaves; TCr: tree carbon density of root; TCag: tree aboveground carbon density; SC: shrub carbon density
Differences in forest characteristics and species diversity of two types under different urbanization areas
For tree height and DBH, the average in the broadleaved forest was significantly decreased, by 20%, compared to conifer forests of the outer area (p < 0.05, Fig. 5a, b). The under crown height in the broadleaved forest was 60% of that of the conifer forest, but the tree health degree was 20–50% higher in broadleaved forests of all urban forests (p < 0.05, Fig. 5c, d). Although tree density was obviously higher in the outer area (Table 2), non-significant changes were found between forest types, neither associated with tree crown diameter (Fig. 5e, f).
Difference of structural characteristics of trees between the broadleaved forests (B) and coniferous forests (C) of inner and outer urban forests. The y-coordinate is the mean value of the parameters, numbers above the error bars: broadleaf to conifer ratio (XB/C = XB/XC), error bars: standard error. Th: tree height; DBH: tree diameter at breast height; Uch: tree under crown height; Tcd: tree crown diameter; Thd: tree health degree; Td: tree density
The shrub layer, shrub height, health degree, and density were found to be 1.6- to two fold higher in the central park than in the outer parks (Table 2). Shrub height, crown diameter and health degree in the broadleaved forest was 1.1- to 1.2-fold higher than those in the conifer forest of urban forests; shrubs were denser in conifer forests (40–50%) of urban forests (Fig. 6a − d). However, all differences between forest types were non-significant (p > 0.05).
Difference of structural characteristics of shrubs between the broadleaved forests (B) and coniferous forests (C) of different urbanization areas. The y-coordinate is the mean value of the parameters; numbers above the error bars: broadleaf to conifer ratio (XB/C = XB/XC); error bars: standard error. Sh: shrub height; Scd: shrub crown diameter; Shd: shrub health degree; Sd: shrub density
Herb coverage and richness of outer areas were 30% higher than those in inner area (p < 0.05, Table 2), and parameters in conifer forests were 20% higher than those in broadleaved forests of outer area (p > 0.05, Fig. 7a, b).
Difference of structural and diversity characteristics of herbs between the broadleaved forests (B) and coniferous forests (C) of different urbanization areas. The y-coordinate is the mean value of the parameters; numbers above the error bars: broadleaf to conifer ratio (XB/C = XB/XC); error bars: standard error. Hc: herb coverage; HS: herb richness
Tree species diversity in the broadleaved forest was 1.2- to 1.5-fold higher than that in the conifer forest (p < 0.05, Fig. 8a–d). Furthermore, the shrub diversity in the central area was two- to three fold higher of that in the outer area (Table 2, p < 0.05), and diversity in broadleaved forests accounted for 60–90% of those in conifer forests (Fig. 8e–h).
Difference of species diversity of tree and shrubs between the broadleaved forests (B) and coniferous forests (C) of different urbanization areas. The y-coordinate is the mean value of the parameters; numbers above the error bars: broadleaf to conifer ratio (XB/C = XB/XC); error bars: standard error. TH: tree Shannon–Wiener; TD: tree Simpson; TS: tree richness. TJ: tree evenness; SH: shrub Shannon–Wiener; SD: shrub Simpson; SS: shrub richness. SJ: shrub evenness
RDA and variation partitioning analysis
Structural traits and dominant species abundance had similar important roles in carbon stock improvement at 40%, and the impact of diversity accounted for only a quarter. The explanation of all three parts is 63% (Fig. 9). Communities with an increased top 50% tall trees, top 25% big crown shrubs, and Pinus tabuliformis were available for the large tree and shrub carbon sequestration, whose explanations were more than three fold of the other parameters (Fig. 10). Among these, Pinus tabuliformis had a strongly positive effect on tree leaf carbon. Increased big-sized trees (top 50% largest tree height, DBH, and crown diameter) directly increased tree branch, stem, and aboveground carbon density. Other dominant species, such as Pinus koraiensis, Larix gmelinii, Ulmus pumila and Acer tataricum, as well as denser trees, had a strong positive effect on tree roots, branches, and stem carbon density. Increased the amount of Pinus sylvestris and big-sized and healthy shrubs (top 25% largest shrub crown diameter and higher health degree) induced high shrub carbon stocks (Fig. 10).
Variation partitioning analysis results of “Var-part-3 groups-Conditional-effects-tested.” Three groups were selected as X1: 11 structural traits (the average of Uch, Thd, Td, Shd, Sd, and Hc, the top 50% Th, DBH and Tcd, the top 25% Sh and Scd); X2: 9 species diversity parameters (four tree and shrub diversity indices, respectively, and herb richness); X3: 10 dominant species abundance (top five tree and shrub dominant species, respectively)
RDA (a) and forward selection results (b) of the association among dominant species composition and diversity, structural traits and carbon sink function of trees and shrubs. The factors chosen are the significant elements under the forward selection effects as follows: 50%Th, 50% highest tree height; PinusT, Pinus tabuliformis; 25%Scd, 25% biggest shrub crown diameter; PinusK, Pinus koraiensis; LarixG, Larix gmelinii; UlmusP, Ulmus pumila; PinusS, Pinus sylvestris; Td, tree density; AcerT, Acer tataricum; 50%DBH, 50% biggest DBH; 50%Tcd, 50% biggest tree crown diameter; Shd, shrub health degree
Despite different urbanization areas (inner or outer) and forest types (broadleaved or conifer forest), the top three significant impact elements were similar: the top 50% tall trees, top 25% big crown shrubs, and Pinus tabuliformis, but their rank was different (Figs. 10, S2 and S3). In addition, an increased number of deciduous species, including Ulmus pumila and Acer tataricum, had a positive effect on tree roots, stems, and branches as well as shrub carbon density in the central urban area and broadleaved forests. The more extensive coverage of herbs was attributed to high tree biomass carbon sink, except for leaves in the inner urban area (Fig. S2a, b). However, increased needle trees (Pinus koraiensis, Larix gmelinii, and Pinus sylvestris) influenced the carbon stocks of the outer urban area and conifer forest. Pinus koraiensis and Larix gmelinii positively increased tree roots, branches, and stem carbon, and Pinus sylvestris directly promoted shrub carbon sink (Figs. S2c, d and S3). Moreover, in conifer forests, increased clutter distribution of trees and abundant herb trees conducive to aboveground carbon function included leaves, stems, and branches (Fig. S3).
Discussion
Urbanization effect-induced carbon sequestration, structural traits, and species diversity changes
The mean carbon density of aboveground trees (including stem, leaf and branch) and shrubs in the central urban forest was 1.0- to 1.3-fold of those in the outer forest, but the differences were not significant (p > 0.05, Table 2). According to the results of two large, crucial parks with similar established time inside and outside central urban, South Lake (SL) and Jingyuetan National Forest Park (JYT), the tree carbon sink in JYT was slightly higher (10–30%) than in the SL Park (p > 0.05, Table S4). Nevertheless, carbon sequestration was higher mainly in other “Old parks” than in “New parks” and attributed to the older big-sized trees in the “Old park” in the central urban forests (p < 0.05, Table S5). This finding demonstrates that favorable practices had been implemented to protect urban green infrastructure during recent years, especially in “Old parks”, such as urban forest planning, afforestation and remnant tree conservation (Wang et al. 2020b). Moreover, “New parks” dominated with the young and middle-aged forests had a large potential for C sequestration-raising in the future (Dai et al. 2018). With the rise of urban development, certain cities developed through increasing density of the current urban area, resulting in urban forest decline. In contrast, most cities have expanded by extending “sprawl” into the wider countryside, where additional “New park” was established, which also involved the investment of a large amount of money by local government in response to climate change problems (Dallimer et al. 2011; Ren et al. 2019). However, a higher number of larger, older trees attributed to extraordinary carbon sequestration in the “Old park” area, which should be paid more attention in the future owing to their poorer health and the high susceptibility of the land-type to various human influences (Lindenmayer and Laurance 2017; Nizamani et al. 2021). Furthermore, the healthier trees in the heavily urbanized area could be mainly attributed to SL Park, which is consistent with the findings of a previous study (Zhang et al. 2017). Thus, solely considering the differences between the urban ring road and location to reflect the urbanization pattern is challenging; the establishment time is a necessary factor that cannot be ignored in future studies.
Generally, the total aboveground (including stems and leaves) and root carbon sink in conifer forests were 20–70% higher than those in broadleaved forests (Fig. 4), which is consistent with previous studies in which conifer trees had higher carbon sequestration efficiency than other species (Weissert et al. 2016). However, the result was different from that in southern China (Fu et al. 2015). The 20% larger trees and 40% increased under crown height were found in the conifer forests, especially of outer area. Moreover, 1.6- to twofold higher, healthier and denser shrubs, but 30% fewer herbs were found in the central park than in the outer parks; and sparser but larger, healthier shrubs were found in broadleaved forests, mainly attributed to density-dependent effect; Tree species diversity in the broadleaved forest was 1.2- to 1.5-fold higher than that in the conifer forest, but the shrub diversity accounted for 60–90% of those in conifer forests. Consequently, suitable greenspace management according to different forest types is essential to protect vegetation health, and can be expected to determine the urban appearance and green quantity (Jin et al. 2022; Kendal et al. 2020).
Complex association among vegetation composition, structural traits, species diversity, and carbon sequestration
We evaluated the importance of plant composition and diversity, as well as the structural characteristics of each organ and aboveground biomass carbon stocks of trees and shrub carbon in urban forests. We provide observational evidence of the crucial role of the top 50% highest trees and 25% largest crown diameter shrubs on carbon sink function. Our results support the generic concept that the crucial contribution of big-sized plants on forests aboveground biomass rather than species richness, whose tall statures and big crowns can occupy most of the growing space at the top of the canopy than small and medium-sized plants (Lutz et al. 2018; Ali et al. 2019; Bastin et al. 2018; Pelt et al. 2016). Moreover, experts have revealed that the greater carbon sequestration ability of older forests is attributed to the regeneration of new trees, which offset respiration rate of older trees to slow global warming (Dimri et al. 2017; Yang et al. 2021; Gilhen-Baker et al. 2022).
In this study, we found that conifer trees such as Pinus tabuliformis strongly affected aboveground biomass carbon stocks, especially leaf carbon, and this result was also found in urban greenspace in Beijing (Wang et al. 2021b). The explanation of needle trees (26.8%) was six-fold that of the broadleaved trees (4.4%), indicating the high yield of needle trees. This conclusion disproves the inherent idea that conservative species with lower photosynthetic rates and stem hydraulic conductivity function induce lower biomass productivity. The result might be attributable to conservative species, which increase plant growth and performance as well as productivity because of reduced cell elasticity, and transpire less water response in the onset of the harsher environment and freeze–thaw cycles (Klein and Niu 2014; Poorter and Bongers 2006).
This study summarizes a series of forest management strategies for promoting urban carbon sequestration in specific urbanization areas and types. For example, increased broadleaved trees, such as Ulmus pumila and Acer tataricum, are attributed more to broadleaved forests in central urban areas. However, increased needle trees, including Pinus koraiensis, Larix gmelinii, and Pinus sylvestris, had an evident effect in conifer forests of outer urban areas. Furthermore, Pinus sylvestris directly promoted shrub carbon sink, and denser trees raised tree carbon stocks (Fig. 11). Our results confirmed that densely planted forests are much more effective in carbon sequestration, as reported in previous studies (De la Sota et al. 2019). These strategies will provide references for urban forest management to improve carbon sequestration, but must be tested and optimized in other construction practices.
In natural forests, stand structural complexity and diverse species promote different tree canopy heights and niche complementary, thereby increasing aboveground biomass and productivity (Yuan et al. 2020; Wang et al. 2020a). However, in urban forests, the dominant species and “big-size effect” overvalued diversity-determined carbon stocks. In addition, the strength and magnitude of the key species effect demonstrated that it was available to increase urban forest carbon sequestration via artificial management measures.
In our research, aboveground carbon stocks are a critical function for provisioning and regulating services, which is strongly linked to forest timber production (Trogisch et al. 2017). Comprehensive assessment from multiple organs (leaves, stems, branches, and roots) and layers (trees and shrubs), which played key roles in the urban carbon cycle, was conducive to the proposal of scientific and systematic practices and measures for urban forest management.
Implication
Communities dominated by dominant species and big-sized individuals are expected to have a high biomass carbon stocks. Large trees in urban forests should be strictly protected without transplanting and disturbance by anthropogenic effects because of their irreplaceable role in carbon stocks (aboveground biomass and soil) (Jin et al. 2022). Furthermore, trade-offs among affluent and clutter trees and herbs and aboveground carbon function (including leaves, stems, and branches, Fig. S3) highlight the potential for "win–win" forest strategies to achieve carbon sink function improving and diversity protection in conifer urban forests. The optimized strategies will help to promote urban carbon sequestration and contribute to sustainable and stable carbon sequestration.
Our findings clarified that central urban forests play a considerable role in carbon stock improvement, including trees and shrubs, and confirmed that precise and effective management measures were implemented in central urban areas. However, further actions should be implemented urgently in low urbanization areas, especially for the “New park” area.
Compared with previous studies on aboveground carbon sink and structural traits of Changchun in 2012 (Zhang et al. 2017), over 10 years, the carbon stocks increased by 50–60% in heavy and low urbanized areas, tree DBH increased by 10–40% from low to heavy urbanization, tree height in 2022 increased by 2- to 2.3-fold of historical measurements, and trees were denser with a 1.2- to 1.6-fold increase (Table 3). However, notably, the tree diversity in heavily urbanized areas decreased by 10–30% over the 10 years.
Conclusion
This is the first study on the effects of urbanization and forest-type on forest attributes and carbon stocks in the urban forests of Changchun parks. Intense urbanization areas accompanied by big-sized and additional healthy trees and shrubs induced higher aboveground tree and shrub carbon stocks. However, denser trees, larger coverage, and diverse herbs were found in lower urbanization areas. Conifer forests comprised big-sized trees, diverse herbs and shrubs, and higher tree carbon sequestration. Broadleaved forests showed higher shrub carbon density with increased big-sized and healthy yet sparse shrubs, as well as increased healthy and diverse trees. Among the various factors, big-sized woody plants and Pinus tabuliformis played important roles in controlling carbon sink. Future management measures should consider different urbanization intensities and forest types and strengthen the management of the “New park” area. Our findings provide a scientific basis for the further precise management of urban forests.
References
Ali A, Lin S, He J, Kong F, Yu J, Jiang H (2018) Climate and soils determine aboveground biomass indirectly via species diversity and stand structural complexity in tropical forests. For Ecol Manage 432:823–831. https://doi.org/10.1016/j.foreco.2018.10.024
Ali A, Lin S, He J, Kong F, Yu J, Jiang H (2019) Big-sized trees overrule remaining trees’ attributes and species richness as determinants of aboveground biomass in tropical forests. Glob Change Biol 25(8):2810–2824. https://doi.org/10.1111/gcb.14707
Baker M, Roviello V, Kroeger D, Roviello G (2022) Old growth forests and large old trees as critical organisms connecting ecosystems and human health. A Rev Environ Chem Lett 20(2):1529–1538. https://doi.org/10.1007/s10311-021-01372-y
Bastin J, Rutishauser E, Kellner J, Saatchi S, Pélissier R, Hérault B, Slik F et al (2018) Pan-tropical prediction of forest structure from the largest trees. Glob Ecol Biogeogr 27(11):1366–1383. https://doi.org/10.1111/geb.12803
Carretero E, Moreno G, Duplancic A, Abud A, Vento B, Jauregui J (2017) Urban forest of Mendoza (Argentina): the role of Morus alba (Moraceae) in carbon storage. Carbon Management 8(3):237–244. https://doi.org/10.1080/17583004.2017.1309206
Chang Y, Wang Z, Zhang D, Fu Y, Zhai C, Wang T, Yang Y, Wu J (2022) Analysis of urban woody plant diversity among different administrative districts and the enhancement strategy in Changchun City. China Sustainability 14(13):7624. https://doi.org/10.3390/su14137624
Dimri S, Baluni P, Sharma C (2017) Biomass production and carbon storage potential of selected old-growth temperate forests in Garhwal Himalaya, India. Proc Natl Acad Sci India Sect b: Biol Sci 87(4):1327–1333. https://doi.org/10.1007/s40011-016-0708-0
Dong S, Sha W, Su X, Zhang Y, Li S, Gao X, Liu S, Shi J, Liu Q, Hao Y (2019) The impacts of geographic, soil and climatic factors on plant diversity, biomass and their relationships of the alpine dry ecosystems: Cases from the Aerjin Mountain Nature Reserve, China. Ecol Eng 127:170–177. https://doi.org/10.1016/j.ecoleng.2018.10.027
Escobedo F, Kroeger T, Wagner J (2011) Urban forests and pollution mitigation: analyzing ecosystem services and disservices. Environ Pollut 159(8–9):2078–2087. https://doi.org/10.1016/j.envpol.2011.01.010
Fan W, Zhang H, Yu Y, Mao X, Yang J (2011) Comparison of three models of forest biomass estimation. Chin J Plant Ecol 35(4):402–410. https://doi.org/10.3724/sp.j.1258.2011.00402
Fu W, Fu Z, Ge H, Ji B, Jiang P, Li Y, Wu J, Zhao K (2015) Spatial variation of biomass carbon density in a subtropical region of Southeastern China. Forests 6(12):1966–1981. https://doi.org/10.3390/f6061966
Gratani L, Varone L, Bonito A (2016) Carbon sequestration of four urban parks in Rome. Urban Forestry & Urban Greening 19:184–193. https://doi.org/10.1016/j.ufug.2016.07.007
Guo Z, Fang J, Pan Y, Birdsey R (2010) Inventory-based estimates of forest biomass carbon stocks in China: A comparison of three methods. For Ecol Manage 259(7):1225–1231. https://doi.org/10.1016/j.foreco.2009.09.047
Hu Y, Su Z, Li P, Li W (2015) Comparative studies on biomass-carbon accounting models at forest stand scale. J Central South Univ For Technol 35(1):84–88
Jin T, Liu W, Wang Y, Zhao M, Fu Y, Dong Y, Luo T, Fu H, Wang Q (2022) Effects of urbanization intensity on glomalin-related soil protein in Nanchang, China: Influencing factors and implications for greenspace soil improvement. J Environ Manage 318:115611. https://doi.org/10.1016/j.jenvman.2022.115611
Kashiwagi H (2016) Atmospheric carbon dioxide and climate change since the Late Jurassic (150Ma) derived from a global carbon cycle model. Palaeogeogr Palaeoclimatol Palaeoecol 454:82–90. https://doi.org/10.1016/j.palaeo.2016.04.002
Kendal D, Egerer M, Byrne J, Jones P, Marsh P, Threlfall C, Allegretto G, Kaplan H, Nguyen HK, Pearson S, Wright A, Flies EJ (2020) City-size bias in knowledge on the effects of urban nature on people and biodiversity. Environ Res Lett 15(12):124035. https://doi.org/10.1088/1748-9326/abc5e4
Klein T, Niu S (2014) The variability of stomatal sensitivity to leaf water potential across tree species indicates a continuum between isohydric and anisohydric behaviours. Funct Ecol 28(6):1313–1320. https://doi.org/10.1111/1365-2435.12289
Knorn M, Moisen G, Healey S, Keeton W, Freeman E, Hostert P (2011) Evaluating the Remote Sensing and Inventory-Based Estimation of Biomass in the Western Carpathians. Remote Sens 3(7):1427–1446. https://doi.org/10.3390/rs3071427
Lin J, Kroll C, Nowak D, Greenfield E (2019) A review of urban forest modeling: Implications for management and future research. Urban For Urban Greening 43:126366. https://doi.org/10.1016/j.ufug.2019.126366
Lindenmayer D, Laurance W (2017) The ecology, distribution, conservation and management of large old trees. Biol Rev Camb Philos Soc 92(3):1434–1458. https://doi.org/10.1111/brv.12290
Liu C, Xiang W, Lei P, Deng X, Tian D, Fang X, Peng C (2013) Standing fine root mass and production in four Chinese subtropical forests along a succession and species diversity gradient. Plant Soil 376(1–2):445–459. https://doi.org/10.1007/s11104-013-1998-0
Lutz J, Furniss T, Johnson D, Davies S, Allen D, Alonso A, Teixeira K et al (2018) Global importance of large-diameter trees. Glob Ecol Biogeogr 27(7):849–864. https://doi.org/10.1111/geb.12747
Ma J, Li X, Jia B, Liu X, Li T, Zhang W, Liu W (2021) Spatial variation analysis of urban forest vegetation carbon storage and sequestration in built-up areas of Beijing based on i-Tree Eco and Kriging. Urban For Urban Greening. https://doi.org/10.1016/j.ufug.2021.127413
Ma K, Liu C, Yu S, Wang W (1997) Plant community diversity in Dongling Mountain, Beijing, China III. Species-abundance relations of several types of forest communities. Acta Ecologica Sinica 17(6):573–583. https://www.plant-ecology.com/EN/Y1999/V23/I6/490
Marshall J, Waring R (1984) Conifers and broadleaf species: stomatal sensitivity differs in western Oregon. Can J for Res 14(6):905–908. https://doi.org/10.1139/x84-161
Mitchell M, Johansen K, Maron M, McAlpine C, Wu D, Rhodes J (2018) Identification of fine scale and landscape scale drivers of urban aboveground carbon stocks using high-resolution modeling and mapping. Sci Total Environ 622–623:57–70. https://doi.org/10.1016/j.scitotenv.2017.11.255
Ni J (2013) Carbon storage in Chinese terrestrial ecosystems: approaching a more accurate estimate. Clim Change 119(3):905–917. https://doi.org/10.1007/s10584-013-0767-7
Nizamani M, Harris A, Cheng X, Zhu Z, Jim C, Wang H (2021) Positive relationships among aboveground biomass, tree species diversity, and urban greening management in tropical coastal city of Haikou. Ecol Evol 11(17):12204–12219. https://doi.org/10.1002/ece3.7985
Pelt R, Sillett S, Kruse W, Freund J, Kramer R (2016) Emergent crowns and light-use complementarity lead to global maximum biomass and leaf area in Sequoia sempervirens forests. For Ecol Manage 375:279–308. https://doi.org/10.1016/j.foreco.2016.05.018
Poorter L, Bongers F (2006) Leaf traits are good predictors of plant performance across 53 rain forest species. Ecology 87(7):1733–1743. https://doi.org/10.1890/0012-9658(2006)87[1733:ltagpo]2.0.co;2
Ren Z, Zheng H, He X, Zhang D, Shen G, Zhai C (2019) Changes in spatio-temporal patterns of urban forest and its above-ground carbon storage: Implication for urban CO2 emissions mitigation under China’s rapid urban expansion and greening. Environ Int 129:438–450. https://doi.org/10.1016/j.envint.2019.05.010
Seto K, Shepherd J (2009) Global urban land-use trends and climate impacts. Curr Opin Environ Sustain 1(1):89–95. https://doi.org/10.1016/j.cosust.2009.07.012
Shen G, Wang Z, Liu C, Han Y (2020) Mapping aboveground biomass and carbon in Shanghai’s urban forest using Landsat ETM+ and inventory data. Urban For Urban Greening 51:1226655. https://doi.org/10.1016/j.ufug.2020.126655
Sota C, Ferreira R, García R, Alvarez S (2019) Urban green infrastructure as a strategy of climate change mitigation. A case study in northern Spain. Urban Forestry & Urban Greening 40:145–151. https://doi.org/10.1016/j.ufug.2018.09.004
Trogisch S, Schuldt A, Bauhus J, Blum J, Both S, Buscot F, Izaguirre N et al (2017) Toward a methodical framework for comprehensively assessing forest multifunctionality. Ecol Evol 7(24):10652–10674. https://doi.org/10.1002/ece3.3488
Wang W, Zu Y, Wang H, Matsuura Y, Sasa K, Koike T (2005) Plant biomass and productivity of larix gmelinii forest ecosystems in northeast China: intra- and inter- species comparison. Eurasian J. Forest Res Hokkaido Univ (Japan) 8(1):21–41
Wang W, Zhang B, Xiao L, Zhou W, Wang H, He X (2018) Decoupling forest characteristics and background conditions to explain urban-rural variations of multiple microclimate regulation from urban trees. PeerJ 6:e5450. https://doi.org/10.7717/peerj.5450
Wang W, Zhang B, Zhou W, Lv H, Xiao L, Wang H, Du H, He X (2019a) The effect of urbanization gradients and forest types on microclimatic regulation by trees, in association with climate, tree sizes and species compositions in Harbin city, northeastern China. Urban Ecosyst 22(2):367–384. https://doi.org/10.1007/s11252-019-0823-9
Wang Q, Wang W, Zhong Z, Wang H, Fu Y (2019b) Variation in glomalin in soil profiles and its association with climatic conditions, shelterbelt characteristics, and soil properties in poplar shelterbelts of Northeast China. J For Res 31:279–290. https://doi.org/10.1007/s11676-019-00909-w
Wang Y, Yu J, Xiao L, Zhong Z, Wang Q, Wang W (2020a) Dominant species abundance, vertical structure and plant diversity response to nature forest protection in Northeastern China: conservation effects and implications. Forests 11(3):295. https://doi.org/10.3390/f11030295
Wang Q, Zhang D, Zhou W, He X, Wang W (2020b) Urbanization led to a decline in glomalin-soil-carbon sequestration and responsible factors examination in Changchun, Northeastern China. Urban For Urban Greening 48:126506. https://doi.org/10.1016/j.ufug.2019.126506
Wang Y, Wen H, Wang K, Sun J, Yu J, Wang Q, Wang W (2021a) Forest plant and macrofungal differences in the Greater and Lesser Khingan Mountains in Northeast China: A regional-historical comparison and its implications. J For Res 33:623–641. https://doi.org/10.1007/s11676-021-01313-z
Wang Y, Chang Q, Li X (2021b) Promoting sustainable carbon sequestration of plants in urban greenspace by planting design: A case study in parks of Beijing. Urban For Urban Greening 64:127291. https://doi.org/10.1016/j.ufug.2021.127291
Wang H, Cai Y, Deng W, Li C, Dong Y, Zhou L, Sun J et al (2023) The effects of tree canopy structure and tree coverage ratios on urban air temperature based on ENVI-Met. Forests 14(1):80. https://doi.org/10.3390/f14010080
Wang Y, Wang B, Wang K, Wang H, and Wang W (2022) Land Degradation Neutrality: Achieving SDG 15 by Forest Management, Nature Conservation Effects on Forest Carbon Sequestration, Biodiversity of Plants, and Macrofungi: A Case Study in Central Lesser Khingan Mountains, NE China.
Weissert L, Salmond J, Schwendenmann L (2016) Photosynthetic CO2 uptake and carbon sequestration potential of deciduous and evergreen tree species in an urban environment. Urban Ecosyst 20(3):663–674. https://doi.org/10.1007/s11252-016-0627-0
Wu L, Shi Y, Zhang F, Zhou Y, Ding Z, Lv S, Xu L (2022) Estimating carbon stocks and biomass expansion factors of urban greening trees using terrestrial laser scanning. Forests 13(9):1389. https://doi.org/10.3390/f13091389
Yang Z, Zheng Q, Zhuo M, Zeng H, Hogan J, Lin T (2021) A culture of conservation: How an ancient forest plantation turned into an old-growth forest reserve – The story of the Wamulin forest. People Nat 3(5):1014–1024. https://doi.org/10.1002/pan3.10248
Yuan Z, Ali A, Benito P, Jucker T, Mori A, Wang S, Zhang X et al (2020) Above- and below-ground biodiversity jointly regulate temperate forest multifunctionality along a local-scale environmental gradient. J Ecol 108(5):2012–2024. https://doi.org/10.1111/1365-2745.13378
Zhang H, Qi Z, Ye X, Cai Y, Ma W, Chen M (2013) Analysis of land use/land cover change, population shift, and their effects on spatiotemporal patterns of urban heat islands in metropolitan Shanghai, China. Appl Geogr 44:121–133. https://doi.org/10.1016/j.apgeog.2013.07.021
Zhang D, Wang W, Zheng H, Ren Z, Zhai C, Tang Z, Shen G, He X (2017) Effects of urbanization intensity on forest structural-taxonomic attributes, landscape patterns and their associations in Changchun, Northeast China: Implications for urban green infrastructure planning. Ecol Ind 80:286–296. https://doi.org/10.1016/j.ecolind.2017.05.042
Zhang J, Wang W, Du H, Zhong Z, Xiao L, Zhou W, Zhang B, Wang H (2018) Differences in community characteristics, species diversity, and their coupling associations among three forest types in the Huzhong area, Daxinganling Mountains. Acta Ecol Sin 13:4684–4693. https://doi.org/10.5846/stxb201706261149
Acknowledgements
We would like to thank Z.W., J.W., X.Z., Y.Y. for their dedication to our fieldwork campaigns.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Project funding: This work was supported by the Youth Growth Technology Project, Science and Technology Department of Jilin Province (20230508130RC), and project from Bureau of Forestry and Landscaping of Changchun.
Corresponding editor:Yu Lei
The online version is available at https://springerlink.bibliotecabuap.elogim.com/.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, Y., Dai, X., Chen, X. et al. Effects of urbanization and forest type on species composition and diversity, forest characteristics, biomass carbon sink, and their associations in Changchun, Northeast China: implications for urban carbon stock improvement. J. For. Res. 35, 36 (2024). https://doi.org/10.1007/s11676-023-01688-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11676-023-01688-1