Abstract
Forests are important ecosystems for economic and social development. However, the response of tree radial growth to climate has produced ‘divergent problems’ at high latitudes under global warming. In this study, the response stability and trend of Picea schrenkiana radial growth to variability in climate factors were analyzed in the mid-latitudes of the western Tien Shan Mountains. Radial growth of P. schrenkiana was mainly limited by minimum and mean temperatures. The divergent responses of radial growth occurred in response to the minimum and mean temperatures at the beginning of the growing season (April–May) of the current year, but responses to drought occurred in July–September of the previous year. And the mean and minimum temperatures in June–September of the current year were both stable. Radial growth first increased and then decreased according to the basal area increment, with a gradual increase in temperature. Therefore, forest ecosystems in mountainous arid areas will be increasingly affected by future climate warming.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Forests are important natural resources covering approximately 3.45 billion ha or 30% of the global land area. Forest play important roles in the formation and maintenance of biodiversity, carbon sequestration, the carbon balance, the hydrological cycles and paleoclimatic reconstructions (FAO 2006; Anderegg et al. 2020). Global warming has a significant effect on the sensitivity of tree species and the composition of forest ecosystems (Williams et al. 2010). The Intergovernmental Panel on Climate Change (IPCC) fifth report reported that the global mean temperature at the Earth’s surface increased by 0.85 °C ± 0.20 °C between 1880 and 2012, the hottest 30-year-period in the past 800 years, and the future climate was forecast to warm further (IPCC 2013). Therefore, the sustainable development of forest ecosystems has been integrated with the core global ecological problems as global warming continues to intensify (Jiao et al. 2017).
As a database of climate change, tree rings are widely selected to study regional historical climate reconstructions and growth-climate relationships due to accurate dating, strong continuity, high sensitivity and easy access (Wang et al. 2020). However, several scholars have found that climate warming led to a decrease in the sensitivity of trees to temperatures at high latitudes in the Northern Hemisphere (Wang et al. 2017; Zhang et al. 2019). The phenomenon of an unstable response between radial growth and climatic factors was termed the ‘divergence problem’, which challenged the accuracy and scientificity of tree-ring climate reconstruction work based on the ‘uniformitarian principle’ (D'Arrigo et al. 1992; Briffa et al. 1998; Wilmking et al. 2020). At present, the results of divergent responses differ between regions, tree species, elevation gradients and age groups. For example, Siberian Larch (Larix sibirica Ledeb.) did not show a distinct divergent response to temperature in the Altai Mountains, but divergent response of Larix sibirica and the relatively stable responses by Schrenk spruce (Picea schrenkiana Fisch. & C.A. Mey.) to climate factors were shown in the eastern Tien Shan (Shang et al. 2010; Jiao et al. 2019). Global warming is believed to have positive effects on forests at high elevations but negative effects at low elevations reflected by radial growth to climate along elevation gradients according to dendroclimatology, as observed in the western mountains of North America (Salzer et al. 2009). Radial growth response of Chinese white pine (Pinus armandii Franch.) of different age groups to climate factors were different in the Baiyunshan National Nature Reserve, central China (Peng et al. 2018). Moreover, the mechanisms of the ‘divergence problem’ are also controversial, mainly focusing on the heterogeneous age effects of biological species, the threshold changes of the growth responses to climate, inappropriate detrended methods, and low-quality climate data (Cook et al. 1997; D'Arrigo et al. 2008; Allen et al. 2018; Wilmking et al. 2020). Therefore, it is necessary to further assess the stability of the responses to climate changes by different tree species and in diverse areas, and to explore the causes of the divergent responses.
The Tien Shan Mountains or simply Tien Shan, as a wet island in an arid region, play an important role in maintaining the stability of oases and the sustainability of water resources in northwest China (Liang et al. 2016). Moreover, the Tien Shan adequately reflects the fluctuations in regional climate change as there is limited interference from areas of human activities. Thus, the Tien Shan has become a “hot spot” for dendrochronological research (Zhang et al. 2016a). Although numerous studies on paleoclimate reconstruction have been performed in the Tien Shan since the 1960s, few have focused on the ‘divergence problem’ (Zhang et al. 2016b). At the same time, climate transformations from warm-dry to warm-wet areas since the end of the 1980s also provide a possibility for further analysis of the impacts of climate change on forest ecosystems (Shi et al. 2007). However, conclusions of climatic impacts on trees and forests have been inconsistent. For example, the basal area increment (BAI) of P. schrenkiana has shown n an increase in the Tien Shan with the extension of the growing season caused by global warming (Qi et al. 2015). However, other studies found that P. schrenkiana growth declined trend with intensified water deficits in inland Asia (Liu et al. 2013). Therefore, the impacts of climate change on ecosystems in the Tien Shan need further analysis.
A virgin P. schrenkiana forest in the western Tien Shan was selected and the stability of the responses to climate changes were analyzed and changes to the growth patterns of dominant coniferous trees with climate transformation were described. The research questions were: (1) What are the principal climatic factors limiting the growth of P. schrenkiana in the western Tien Shan? (2) Is there a ‘divergence problem’ in the dynamic growth-climate relationship? and (3) What was the trend in growth of P. schrenkiana under climate change?
Materials and methods
Study site and tree ring acquisition
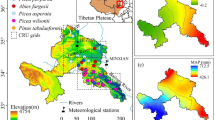
Tree rings were collected from species in the P. schrenkiana National Nature Reserve in western Tien Shan (XTS) (Fig. 1). The research region has a typical temperate continental climate with rainy and hot conditions in the same period. However, warm, humid air from the Atlantic enters the western Tien Shan under the influence of the westerly winds, resulting in relatively warmer and more humid climates than eastern Tien Shan. Annual total precipitation is approximately 508 mm, mainly concentrated in April−September, accounting for 84.0% of the annual rainfall. Annual mean temperature is 3.4 °C, with January the coldest month at − 11.2 °C, and July the hottest month at 15.1 °C (Figs. 2, 3). The annual mean temperature in the study region has been increasing since 1959 (mean maximum temperature: 0.19 °C per decade, mean temperature: 0.33 °C per decade, mean minimum temperature: 0.53 °C per decade, P < 0.001). The interannual variation in precipitation was not significant (3.82 mm per decade, P = 0.574). We further studied the rate of temperature increase (slope/10a) and found that it had a higher rate of increase in the 1980s and the decade before the twenty-first century (P < 0.01). The above meteorological data are from Zhaosu (ZS) Meteorological station. P. schrenkiana is the dominant conifer species in the region and mainly distributed at an altitude of 1500−2800 m a.s.l. with a canopy density of approximately 40%.
To minimize the effects of non-climate factors, cores from healthy trees in undisturbed open stands with similar slopes (10°−30°), aspects (north-facing) and soil conditions (grey cinnamon soil) were selected. Two cores from each tree were taken at the 120° position with 5-mm increment borers. Sixty cores from 30 live trees were obtained in June 2017. The characteristics of the forest communities are shown (Table 1).
Tree-ring chronology development
In the laboratory, the cores were mounted on woody supports, air- dried, sanded to produce clear boundaries for identification, and the widths measured using LINTAB (RINNTECH e.K. Heidelberg, Germany) with 0.001 mm resolution (Stokes and Smiley 1968; Schweingruber 1996). Two cores with ambiguous rings and large growth-curve variations were removed. The accuracy of all measurement series was verified by the COFFECHA program (Holmes 1983). Ring widths were detrended using negative exponential or straight line curves, and single chronologies were combined with the individual series by calculating the bi-weight robust means chronology using the ARSTAN program (Cook et al. 2013). The variance in chronologies was stabilized with the Briffa Rbar-weighted method (Cook and Kairiukstis 1990). Finally, a standard chronology (STD) was developed.
Statistical parameters of the chronology reflect the strength and quality of the climate information (Fritts 1976). Mean sensitivity (MS) and standard deviation (SD) were used to evaluate chronology sensitivity to climate factors. The interpretation of the variance in the first principal component (PC1) and signal-to-noise ratio (SNR) characterized the common climate information contained in the chronology. The average correlation coefficient (R1, R2, R3) indicated the consistency of the radial growth fluctuations. An expressed population signal (EPS) greater than 0.85 indicated that the signal contained in the tree-ring series represented the overall characteristics (Wigley et al. 1984). A high first-order autocorrelation coefficient (AC1) indicated a high growth response in the current year to the climate in the previous year (Fig. 4; Table 2).
Meteorological data
Meteorological data were acquired from the ZS meteorological station at 43.15°N, 83.13°E, and at 1854 m (Fig. 1). The climatic data included monthly maximum, mean, and minimum temperatures and total precipitation (1959−2016). These temperature data were adjusted for the altitude differences between the sampling sites and the meteorological station (the temperature decreases by 0.6 °C for every 100 m increase). Radial growth exhibited a delayed response to climate, showing that trees are affected not only by the current year's climate but also by the previous year’s climate. Therefore, the monthly climate data were selected from the previous July to the current September for the analysis of the growth-climate relationship (Fritts 1976).
The standardized precipitation and evapotranspiration index (SPEI) were also calculated using monthly climate data (Vicente-Serrano et al. 2010). The SPEI is an ideal tool for monitoring and studying the process of drought because it organically integrates the Palmer drought severity index (PDSI) with evaporation demand changes and standardized precipitation index (SPI) properties over multiple time scales (Diego Galván et al. 2015). The SPEI data on a 24-month scale were calculated using R (R Core Team R 2014) with the ‘SPEI’ R package (Beguería and Vincente-Serrano 2013).
Statistical methods
Climatic factors controlling radial growth were determined by calculating the Pearson correlation coefficient between monthly climate data and chronology with SPSS 22.0 (Kirkpatrick and Feeny 2014). Two significance levels (0.05 and 0.01) were set and a two-tailed test was performed.
where Y is the ecological security index, Xi the i landscape pattern index, C1(Y, Xi) the Pearson correlation coefficient between Y and Xi, C2(Y, Xi) the Pearson correlation coefficient between Y and lnXi, C(Y, Xi) the Pearson correlation coefficient between Y and Xi. The sign (positive or negative) of the coefficient is consistent with that of the original value.
The moving correlation analysis was used to test the dynamic responses of radial growth to climate using the Dendroclim 2002 program (Biondi and Waikul 2004). We employed a fixed 30-year period starting from 1960 that slid across time to compute the correlation coefficients between tree-ring width chronologies and seasonal climatic factors with a bootstrap procedure using 1000 replications.
Radial growth pattern
Basal area index (BAI) is a more realistic indicator than tree-ring chronology to quantify trends in tree radial growth. In fact, the BAI overcomes undetectable increasing growth trends caused by a decrease in ring width with age in mature trees (Weiner and Thomas 2001; Gomez-Guerrero et al. 2013). The BAI eliminates any data-transformation biases resulting from the use of the original tree-ring width data; in particular, downward trends of BAI indicate that growth was restricted by the environment (Schuster and Oberhuber 2013). Therefore, BAI is considered a better means for assessing radial growth (LeBlanc 1990; Pedersen 1998). The formula is:
where R is the tree radius in years t-1, and TRW is the raw width of the tree-ring in year t.
Results
Correlation between chronology and climatic factors
The standard chronology (STD) for P. schrenkiana contained extensive climate information of good quality and sensitive to climate change. Thus, it was suitable for the analysis of radial growth-climate relationships (Table 2). Correlation analysis indicated that temperature was the controlling factor for tree growth (Fig. 5). On a monthly scale, the STD chronology was positively correlated to total precipitation (previous July: r = 0.382; previous September: r = 0.290), with a high level of significance (P < 0.01, P < 0.05), negatively correlated to temperatures (maximum, mean and minimum temperature in the previous July to October and the current April to September), with a high level of significance (P < 0.05), and positively correlated to the SPEI (previous July: r = 0.399) with a high level of significance (P < 0.01). On the growing season scale, STD was correlated to the maximum temperature (r = − 0.669), mean temperature (r = − 0.813), minimum temperature (r = − 0.806), total precipitation (r = 0.413) and SPEI (r = 0.361) with a high level of significance (P < 0.01) late in the growing season of the previous year (July−September). The STD was correlated to the maximum, mean and minimum temperatures in the early growing season (April–May) and the middle-late growing season (June–September) of the current year with a high level of significance (P < 0.01, P < 0.05).
Dynamic growth-climate relationship
Based on the results of the growth-climate relationships, the moving correlations of radial growth and climatic factors in the growing season were tested during 1960−2016 (Fig. 6). The tree-ring width chronology to mean and minimum temperatures, precipitation and SPEI were stable during the middle-late growing season of the previous year (July–September), and the significant negative correlation between the STD and mean and minimum temperatures was also stable during the middle-late growth season of the current year (June–September). However, the divergent responses of the STD to mean and minimum temperatures in the early growing season of the current year showed gradually increasing trends (mean temperature: y = − 0.015x + 30.158, R2 = 0.693, P < 0.001; minimum temperature: y = − 0.013x + 26.553, R2 = 0.692, P < 0.001).
Radial growth trend
Interannual variation in the BAI was evaluated during the period 1816 to 2016 (Fig. 7). The radial growth trended to increase and then decrease, specifically an upward trend from 1816 to 1958 (1.05 cm2 per decade, P < 0.001), but downward trend from 1959 to 2016 (− 2.16 cm2 per decade, P < 0.001). To avoid influence of the age effect, the trend of radial growth of trees of different age classes (˃ 200 years old; ˂ 200 years old) was analyzed. The results show that the radial growth of the two age classes trended upwards (1.60 and 2.22 cm2 per decade, respectively) from 1816 to 1958, and downward from 1959 to 2016 (− 3.01 and − 1.60 cm2 per decade, respectively, P < 0.001).
Discussion
Ecological response of radial growth to the main limiting climatic factors
To verify that our tree-ring chronology contained sufficient climate information, we compared MS, SD, EPS and SNR with those near the research area and found that our chronology had a high quality and could meet the requirements of growth-climate research (Cook and Kairiukstis 1990). Tree rings not only record climate information but also represent the physiological response of vegetation to climate change (Liang et al. 2009). P. schrenkiana growth in western Tien Shan is mainly restricted by previous drought conditions and current temperatures in the growing season.
Due to the influence of elevation-dependent responses in plants, trees at high altitudes are restricted by low temperatures, a positive correlation with temperature. Trees at low altitudes are mainly restricted by drought (Shao et al. 2005). In our study, STD was negatively correlated with temperature and positively with precipitation and the SPEI from the previous July to September period, indicating that drought in the middle to late growing seasons of previous years had a profound impact on tree growth (Fig. 5). This is because the water storage capacity was poor for P. schrenkiana distributed on steep slopes with shallow roots at low elevations, so the limited water barely met the needs of trees during the growing season (Table 1). Moreover, high temperatures increased the transpiration of leaves and evaporation of surface moisture, increasing the scarcity of available water resources and reducing the accumulation of photosynthetic nutrients, easily resulting in the formation of narrow tree rings (King et al. 2013; Gou et al. 2015; Zhang et al. 2017).
There was a significant negative response reflected in tree-ring width to temperature in the growing season of the current year, especially the mean minimum temperature (Fig. 5). This indicated that radial growth of P. schrenkiana was mainly limited by the mean minimum temperature. The same result has been found in the Tien Shan and other regions (Wu et al. 2013; Liu et al. 2016). As global warming further intensifies, temperatures in the western Tien Shan will show a significant upward trend, with the mean minimum temperature rising at the highest rate (Fig. 3). In fact, the mean minimum temperature effects plant respiration, net productivity, nutrient absorption and other physiological processes (Palombo et al. 2014). Therefore, a significant increase in the mean minimum temperature would result in a decrease in the diurnal temperature differences during the growing season and an increase in respiration and photosynthetic consumption, thereby restricting the differentiation and growth of P. schrenkiana cambium cells (Yu et al. 2013).
Response stability of radial growth to climate change in time
The results of growth-climate dynamic relationships exhibit complex characteristics due to differences in habitat, tree species, age, and climate change patterns (D'Arrigo et al. 2008; Leonelli et al. 2009; Fonti et al. 2018; Jiao et al. 2019). In our study, P. schrenkiana was affected by the persistence of drought conditions during the mid to late growing season of the previous year (Fig. 5). Temperature increase resulted in increased evaporation which increased the adsorption of water by leaf mesophyll cells and slowed the movement of water, resulting in closing of the stomata and weakening photosynthesis (Berninger et al. 2004). In addition, decreased precipitation did not satisfy tree water demands, so the trees were under water shortage stress for considerable time (Jiang et al. 2017). Therefore, radial growth exhibited consistent responses to the temperature, precipitation and SPEI of the previous year. A similar study took place in the mid-western Tien Shan, showing that Schrenk spruce exhibited positive responses to the standardized precipitation and evapotranspiration index (SPEI) from July–October of the previous growing season (Chen et al. 2016).
The limiting effects of the mean and minimum temperatures on annual rings were also relatively stable during the mid to late growing season of the current year (Fig. 6). This was because the relatively high amount of precipitation would address the demands of tree growth in this period, so the radial growth exhibited no correlation with precipitation or drought. However, the continuously high temperatures during this period increased tree respiration and carbohydrates consumption, leading to the maintenance of radial growth sensitivity of radial growth to temperature (Schaberg et al. 2000). Therefore, the ring chronology showed a stable, negative response to temperature in the mid to late growing season of the current year.
The response by Schrenk spruce showed divergence to mean and minimum temperatures at the beginning of the growing season of the current year, mainly supporting the range from uncorrelated to significantly negatively correlated (Fig. 6). Carbohydrates produced by photosynthesis are a major source of energy and nutrients for tree growth. When increased temperatures were still within the physiological threshold range of Schrenk spruce, relatively high temperatures increase photosynthetic efficiency and promote radial growth (Jiang et al. 2014). However, Franceschini et al. (2013) found that the physiological adaptive threshold of Norway spruce (Picea abies Franch.) was gradually exceeded with an abrupt increase in mean temperature in the early growing season, and the species showed a divergent response to the effect of temperature as reflected by the annual rings changing from promotion to reduction. At the same time, the increase in precipitation from rain and melting snow increased soil moisture, largely satisfying moisture demands for radial growth in the early growing season (Dang et al. 2007). Therefore, species sensitivity to total precipitation did not change significantly in the early growing season. The same result was also found in a study on the divergent response of Larix sibirica Ledeb. and climatic factors at high altitudes of the eastern Tien Shan (Jiao et al. 2015).
Radial growth under climate change
The effects of global climate change on tree growth at different temporal and spatial scales remain highly uncertain (IPCC 2013). Our research results show BAI increased initially and then decrease (Fig. 7). In particular, BAI of trees > 200 years old and < 200 years old showed the same growth patterns, indicating that tree-ring width was mainly controlled by climate and is not related to tree age.
Species are often limited by primary ecological factors, and a change beyond the threshold of ecological adaptation will lead to growth reduction and often death (Wiens 1989). At the same time, studies of tree physiology have also concluded that the ‘threshold effect’ occurs, supporting the inverted U-shaped relationships of tree growth to climate (Kramer and Kozlowski 1979). This was also verified by simulating the effect of temperature rise on the size and number of stem cells of trees along the northern edge of a forest (Tuovinen 2005). Precipitation in the western parts of the Tien Shan is more abundant than in the eastern. The length of the growing season is prolonged and the rate of cambium cell division is accelerated with temperature increase. Therefore, radial growth was promoted in the western Tien Shan. However, with further increases in temperature, the tree-ring width chronology showed a significant negative response, and the BAI also showed a significant downward trend, indicating temperatures rose beyond the optimal threshold value in recent decades, and that the effects of temperature on growth have not been restricted with increased respiration, worsening drought and decreased photosynthetic nutrient accumulation. The same result was also found in the eastern Tien Shan (Jiao et al. 2019).
Drought due to climate warming has led to an increase in tree mortality, a decline in forest productivity and the instability of forest ecosystems worldwide (Sarris et al. 2010; Anderegg et al. 2012), especially in arid inland Asia (Poulter et al. 2013; Anderegg et al. 2015). Forest decline caused by climate warming represents a significant threat to forest resources and, to a certain extent, ecological problems such as substantial reductions in available water resources, adverse changes to forest carbon storage, and weakened forest climate regulation (Liang et al. 2006). In addition, increasing temperatures will not only cause a decline in forests in arid regions but also impact humid area and high-altitude forests in the future (Jiao et al. 2015).
Conclusion
In the mid-latitude western Tien Shan of the Northern Hemisphere, the minimum and mean temperatures in the growing season are the main factors limiting the radial growth of P. schrenkiana. Moreover, the dynamic responses of radial growth to the different climate factors differ, showing relatively stable response to drought in the mid to late growing season of the previous year and to the minimum and mean temperatures in same period of the middle-late growing seasons of the current year. However, responses to minimum and mean temperatures in the early growing season of the current year diverged. In addition, radial growth of Schrenk spruce increased and then decreased with climate warming. If temperatures continue to rise in the future, radial growth of Schrenk spruce will become more restricted. Therefore, the influence of climatic change on mountain forest ecosystems in arid regions is important. According to the regular responses by radial growth to climate factors, the monitoring of dominant tree species, targeted forest conservation management and scientific implementation of forest resource protection measures should be strengthened to mitigate the threat of future climate warming.
References
Allen KJ, Villalba R, Lavergne A, Palmer JG, Cook EC, Fenwick P, Drew DM, Turney CSM, Baker PJ (2018) A comparison of some simple methods used to detect unstable temperature responses in tree-ring chronologies. Dendrochronologia 48:52–73. https://doi.org/10.1016/j.dendro.2018.02.002
Anderegg WRL, Kane J, Anderegg LDL (2012) Consequences of widespread tree mortality triggered by drought and temperature stress. Nat Clim Change 3:30–36. https://doi.org/10.1038/nclimate1635
Anderegg WRL, Hicke JA, Fisher RA, Allen CD, Aukema J, Bentz B, Hood S, Lichstein JW, Macalady AK, Mcdowell N, Pan Y (2015) Tree mortality from drought insects and their interactions in a changing climate. New Phytol 208(3):674–683. https://doi.org/10.1111/nph.13477
Anderegg WRL, Trugman AT, Badgley G, Konings AG, Shaw J (2020) Divergent forest sensitivity to repeated extreme droughts. Nat Clim Change. https://doi.org/10.1038/s41558-020-00919-1
Beguería S, Vicente-Serrano SM (2013) SPEI: calculation of the standardised precipitation-evapotranspiration index. Retrieved from http://cran.r-project.org/package=SPEI
Berninger F, Hari P, Nikinmaa E, Lindholm M, Merilainen J (2004) Use of modeled photosynthesis and decomposition to describe tree growth at the northern tree line. Tree Physiol 24(2):193–204. https://doi.org/10.1093/treephys/24.2.193
Biondi F, Waikul K (2004) DENDROCLIM 2002: A C++ program for statistical calibration of climate signals in tree-ring chronologies. Comput Geosci 30(3):303–311. https://doi.org/10.1016/j.cageo.2003.11.004
Briffa KR, Schweingruber FH, Jones PD, Osborn TJ, Shiyatov SG, Vaganov EA (1998) Reduced sensitivity of recent tree-growth to temperature at high northern latitudes. Nature 391(6668):678–682. https://doi.org/10.1038/35596
Chen F, Shang H, Yuan Y (2016) Dry/wet variations in the eastern Tien Shan (China) since AD 1725 based on Schrenk spruce (Picea schrenkiana Fisch. et Mey) tree rings. Dendrochronologia 40:110–116. https://doi.org/10.1016/j.dendro.2016.07.003
Cook ER, Kairiukstis LA (1990) Methods of dendrochronology. Kluwer Academic, Dordrecht
Cook ER, Peters K (1997) Calculating unbiased tree-ring indices for the study of climatic and environmental change. The Holocene 7(3):361–370. https://doi.org/10.1177/095968369700700314
Cook ER, Krusic PJ, Melvin T (2013) Program RCSigFree: Version 43ptr05. Lamont–Doherty Earth Obs. Columbia Univ Palisades
Dang HS, Jiang MK, Zhang QF, Zhang YJ (2007) Growth responses of subalpine fir (Abies fargesii) to climate variability in the Qinling Mountains of China. For Ecol and Manag 240:143–150. https://doi.org/10.1016/j.foreco.2006.12.021
D’Arrigo RD, Jacoby GC, Free RM (1992) Tree-ring width and maximum latewood density at the North American tree line: parameters of climatic change. Can J For Res 22(9):1290–1296. https://doi.org/10.1139/x92-171
D’Arrigo R, Wilson R, Liepert B, Cherubini P (2008) On the ‘Divergence Problem’ in Northern Forests: a review of the tree-ring evidence and possible causes. Glob Planet Change 60(3):289–305. https://doi.org/10.1016/j.gloplacha.2007.03.004
Diego Galván J, Büntgen U, Ginzler C, Grudd H, Gutierrez E, Labuhn I, Camarero JJ (2015) Drought-induced weakening of growth-temperature associations in high-elevation Iberian pines. Global Planet Change 124:95–106. https://doi.org/10.1016/j.gloplacha.2014.11.011
FAO (2006) Global forest resources assessment 2005—progress towards sustainable forest management. FAO Forestry Paper No. 147. Rome.
Fonti MV, Vaganov EA, Wirth C, Shashkin AV, Astrakhantseva NV, Schulze ED (2018) Age-effect on intra-annual δ13c-variability within scots pine tree-rings from central Siberia. Forests 9(364):1–14. https://doi.org/10.3390/f9060364
Franceschini T, Bontemps JD, Perez V, Leban JM (2013) Divergence in latewood density response of Norway spruce to temperature is not resolved by enlarged sets of climatic predictors and their non-linearities. Agric For Meteorol 180:132–141. https://doi.org/10.1016/j.agrformet.2013.05.011
Fritts HC (1976) Tree rings and climate. Academic Press, New York, pp 1–567
Gomez-Guerrero A, Silva LCR, Barrera-Reyes M, Kishchuk B, Velazquez-Martinez A, Martinez-Trinidad T, Plascencia-Escalante FO, Horwath WR (2013) Growth decline and divergent tree ring isotopic composition (δ13C and δ18O) contradict redictions of CO2 stimulation in high altitudinal forests. Glob Chang Biol 19:1748–1758. https://doi.org/10.1111/gcb.12170
Gou XH, Deng Y, Gao LL, Chen FH, Cook E, Yang MX, Zhang F (2015) Millennium tree-ring reconstruction of drought variability in the eastern Qilian Mountains northwest China. Clim Dyn 45(7–8):1761–1770. https://doi.org/10.1007/s00382-014-2431-y
Holmes RL (1983) Computer-assisted quality control in tree-ring dating and measurement. Tree-Ring Bull 43(3):69–75
IPCC (2013) Climate Change 2013: The Physical science basis: contribution of working group I to the fifth assessment report of the intergovernmental panel on climate change. Cambridge University Press Cambridge
Jiang Y, Zhang WT, Wang MC, Kang MY, Dong MY (2014) Radial Growth of two dominant montane conifer tree species in response to climate change in North-Central China. PLoS ONE 9:e112537. https://doi.org/10.1371/journal.pone.0112537
Jiang P, Liu HY, Wu XC, Wang HY (2017) Tree-ring-ased SPEI reconstruction in central Tien Shan of China since A.D. 1820 and links to westerly circulation. Int J Climatol 37(6):2863–2872. https://doi.org/10.1002/joc.4884
Jiao L, Jiang Y, Zhang WT, Wang MC, Zhang LN, Zhao SD (2015) Divergent responses to climate factors in the radial growth of Larix sibirica in the eastern Tianshan Mountains northwest China. Trees 29(6):1673–1686. https://doi.org/10.1007/s00468-015-1248-6
Jiao L, Jiang Y, Wang MC, Zhang WT, Zhang YP (2017) Age-Effect radial growth responses of Picea schrenkiana to climate change in the Eastern Tianshan Mountains Northwest China. Forests 8:294. https://doi.org/10.3390/f8090294
Jiao L, Jiang Y, Zhang WT, Wang MC, Wang SJ, Liu XR (2019) Assessing the stability of radial growth responses to climate change by two dominant conifer trees species in the Tianshan Mountains northwest China. For Ecol Manag 433:667–677. https://doi.org/10.1016/j.foreco.2018.11.046
King G, Fonti P, Nievergelt D, Buntgen U, Frank D (2013) Climatic drivers of hourly to yearly tree radius variations along a 6 °C natural warming gradient. Agric For Meteorol 168:36–46. https://doi.org/10.1016/j.agrformet.2012.08.002
Kirkpatrick LA, Feeney BC (2014) A Simple Guide to IBM SPSS: for version 22.0
Kramer PJ, Kozlowski TT (1979) Physiology of woody plants. Academic Press, New York
LeBlanc DC (1990) Relationships between breast-height and whole-stem growth indexes for red spruce on Whiteface Mountain New York. Can J For Res 20:1399–1407. https://doi.org/10.1139/x90-185
Leonelli G, Pelfini M, Battipaglia G, Cherubini P (2009) Site-aspect influence on climate sensitivity over time of a high-altitude Pinus cembra tree-ring network. Clim Chang 96:185–201. https://doi.org/10.1007/s10584-009-9574-6
Liang EY, Liu XH, Yuan YJ, Qin NS, Fang XQ, Huang L, Zhu HF, Wang L, Shao XM (2006) The 1920s drought recorded by tree rings and historical documents in the semi-arid and arid areas of Northern China. Clim Chang 79(3–4):403–432. https://doi.org/10.1007/s10584-006-9082-x
Liang EY, Eckstein D, Liu H (2009) Assessing the recent grassland greening trend in a long-term context based on tree-ring analysis: a case study in North China. Ecol Indic 9(6):1283. https://doi.org/10.1016/j.ecolind.2009.02.007
Liang EY, Leuschner C, Dulamsuren C, Wagner B, Hauck M (2016) Global warming-related tree growth decline and mortality on the north-eastern Tibetan plateau. Clim Chang 134(1–2):163–176. https://doi.org/10.1007/s10584-015-1531-y
Liu HY, Willians AP, Allen CD, Gou DL, Wu XC, Anenkhonov OA, Liang EY, Sandanov DV, Yin Y, Qi ZH, Badmaeva NK (2013) Rapid warming accelerates tree growth decline in semi-arid forests of Inner Asia. Glob Chang Biol 19(8):2500–2510. https://doi.org/10.1111/gcb.12217
Liu WH, Gou XH, Li JB, Huo YX, Fang KY (2015) A method to separate temperature and precipitation signals encoded in tree-ring widths for the western Tien Shan Mountains, northwest China. Glob Planet Chang 133:141–148. https://doi.org/10.1016/j.gloplacha.2015.08.002
Liu N, Liu Y, Bao G, Bao M, Wang YC, Zhang LZ, Ge YX, Bao W, Tian H (2016) Drought reconstruction in eastern Hulun Buir steppe China and its linkages to the sea surface temperatures in the Pacific Ocea. J Asian Earth Sci 115:298–307. https://doi.org/10.1016/j.jseaes.2015.10.009
Nie CY, Zhang QB, Lyu LX (2017) Millennium-long tree-ring chronology reveals megadroughts on the southeastern Tibetan Plateau. Tree-Ring Res 73(1):1–10. https://doi.org/10.3959/1536-1098-73.1.1
Palombo C, Battipaglia G, Cherubini P, Chirici G, Garfi V, Lasserre B, Lombardi F, Marchetti M, Tognetti R (2014) Warming-related growth responses at the southern limit distribution of mountain pine (Pinus mugo Turra subsp. mugo). J Veg Sci 25:571–583. https://doi.org/10.1111/jvs.12101
Pedersen BS (1998) The role of stress in the mortality of midwestern oaks as indicated by growth prior to death. Ecology 79:79–93. https://doi.org/10.1890/0012-9658(1998)079[0079:TROSIT]2.0.CO;2
Peng JF, Peng KY, Li JB (2018) Climate-growth response of Chinese white pine (Pinus armandii) at different age groups in the Baiyunshan National Nature Reserve, central China. Dendrochronologia 49:102–109. https://doi.org/10.1016/j.dendro.2018.02.004
Poulter B, Pederson N, Liu H, Zhu Z, D’Arrigo R, Ciais P, Davi N, Frank D, Leland C, Myneni R, Piao S, Wang T (2013) Recent trends in Inner Asian forest dynamics to temperature and precipitation indicate high sensitivity to climate change. Agric For Meteorol 178–179:31–45. https://doi.org/10.1016/j.agrformet.2012.12.006
Qi ZH, Liu HY, Wu XC, Hao Q (2015) Climate-driven speedup of alpine treeline forest growth in the Tianshan mountains Northwestern China. Glob Chang Biol 21(2):816–826. https://doi.org/10.1111/gcb.12703
R Core Team (2014) R 2014: a language and environment for statistical computing. Retrieved from http://www.r-project.org/
Salzer MW, Hughes MK, Bunn AG, Kipfmueller KF (2009) Recent unprecedented tree-ring growth in bristlecone pine at the highest elevations and possible causes. PNAS 106:20348–20353. https://doi.org/10.1073/pnas.0903029106
Sarris D, Christodoulakis D, Körner C (2010) Impact of recent climatic change on growth of low elevation eastern Mediterranean forest trees. Clim Chang 106:203–223. https://doi.org/10.1007/s10584-010-9901-y
Schaberg PG (2000) Winter photosynthesis in red spruce (Picea rubens Sarg.): limitations, potential benefits and risks. Arct Antarct Alp Res 32:375–380. https://doi.org/10.2307/1552385
Schuster R, Oberhuber W (2013) Drought sensitivity of three co-occurring conifers within a dry inner Alpine environment. Trees 27(1):61–69. https://doi.org/10.1007/s00468-012-0768-6
Schweingruber FH (1996) Tree rings and environment: dendroecology. Vienna Paul Haupt Bern, Stuttgart, pp 1–609
Shang HM, Wei WS, Yuan YJ, Yu SL, Zhang TW, Aizemaiti W, Li JX (2010) Response of tree ring width to recent climate change south slope of Altai Mountains. Acta Ecol Sin 30(9):2246–2253 (In Chinese with English summary)
Shao XM, Huang L, Liu HB, Liang EY, Fang XQ, Wang LL (2005) Reconstruction of precipitation variation from tree rings in recent 1000 years in Delingha Qinghai. Sci China 48(7):939–949. https://doi.org/10.1360/03yd0146
Shi YF, Shen YP, Kang AS, Li DL, Ding YP, Zhang GW, Hu RJ (2007) Recent and Future Climate Change in Northwest China. Clim Chang 80(3–4):379–393. https://doi.org/10.1007/s10584-006-9121-7
Stokes MA, Smiley TL (1968) An introduction to tree-ring dating. University of Arizona Press, Tucson
Tuovinen M (2005) Response of tree-ring width and density of Pinus sylvestris to climate beyond the continuous northern forest line in Finland. Dendrochronologia 22(2):83–91. https://doi.org/10.1016/j.dendro.2005.02.001
Vicente-Serrano SM, Begueria S, Lopez-Moreno JI (2010) A multiscale drought index sensitive to global warming: the standardized precipitation evapotranspiration index. J Clim 23(7):1696–1718. https://doi.org/10.1175/2009JCLI2909.1
Wang XC, Zhang MH, Ji Y, Li ZS, Li M, Zhang YD (2017) Temperature signals in tree-ring width and divergent growth of Korean pine response to recent climate warming in northeast Asia. Trees 31(2):415–427. https://doi.org/10.1007/s00468-015-1341-x
Wang SJ, Jiao L, Jiang Y, Chen K, Liu XP, Qi CL, Xue RH (2020) Extreme climate historical variation based on tree-ring width records in the Tianshan mountains of northwestern china. Int J Biometeorol 64(12):2127–2139. https://doi.org/10.1007/s00484-020-02003-x
Weiner J, Thomas SC (2001) The nature of tree growth and the ‘age-related decline in forest productivity.’ Oikos 94:374–376. https://doi.org/10.2307/3547583
Wiens JA (1989) Spatial scaling in ecology. Funct Ecol 3(4):385–397. https://doi.org/10.2307/2389612
Wigley TML, Briffa KR, Jones PD (1984) On the average value of correlated time series with applications in dendroclimatology and hydrometeorology. J Clim Appl Meteorol 23(2):201–213. https://doi.org/10.1175/1520-0450(1984)023%3c0201:OTAVOC%3e2.0.CO;2
Williams AP, Allen CD, Millar CI, Swetnam TW, Michaelsen J, Still CJ, Leavitt SW (2010) Forest responses to increasing aridity and warmth in the southwestern United States. PNAS 107(50):21289–21294. https://doi.org/10.1073/pnas.0914211107
Wilmking M, van der Maaten-Theunissen M, van der Maaten E (2020) Global assessment of relationships between climate and tree growth. Global Change Biol 26(6):3212–3220. https://doi.org/10.1111/gcb.15057
Wu XC, Liu HY, Wang YF, Deng MH (2013) Prolonged limitation of tree growth due to warmer spring in semi-arid mountain forests of Tianshan, northwest China. Environ Res Lett 8(2):024016. https://doi.org/10.1088/1748-9326/8/2/024016
Yu SL, Yuan YJ, Wei WS, Chen F, Zhang TW, Shang HM, Zhang RB, Qing L (2013) A 352-year record of summer temperature reconstruction in the western Tianshan Mountains China as deduced from tree-ring density. Quat Res 80(2):158–166. https://doi.org/10.1016/j.yqres.2013.05.005
Zhang RB, Yuan YJ, Gou XH, He Q, Shang HM, Zhang TW, Chen F, Ermenbaev B, Yu SL, Qin L, Fan ZA (2016a) Tree-ring-based moisture variability in western Tianshan Mountains since A.D. 1882 and its possible driving mechanism. Agric For Meteorol Sci 218–219:267–276. https://doi.org/10.1016/j.agrformet.2015.12.067
Zhang RB, Yuan YJ, Wei WS, Gou XH, Yu SL, Shang HM, Zhang TW, Chen F, Qin L (2016b) Research advances of dendroclimatology in Tianshan Mountains. Desert Oasis Meteorol 10(4):1–9 (In Chinese with English summary)
Zhang RB, Yuan YJ, Yu SL, Chen F, Zhang TW (2017) Past changes of spring drought in the inner Tianshan Mountains China as recorded by tree rings. Boreas 46(4):688–696. https://doi.org/10.1111/bor.12238
Zhang L, Shi H, Yu PT, Wang YH, Pan SF, Wang B, Tian HQ (2019) Divergent growth responses to warming between stand-grown and open-grown trees in a Dryland Montane Forest in Northwestern China. Forests 10(12):1133. https://doi.org/10.3390/f10121133
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This research was supported by the National Natural Science Foundation of China (Projects Nos. 41861006 and 41630750), the Scientific Research Program of Higher Education Institutions of Gansu Province (2018C-02) and the Research Ability Promotion Program for Young Teachers of Northwest Normal University (NWNU-LKQN2019-4).
The online version is available at http://www.springerlink.com.
Corresponding editor: Yu Lei.
Rights and permissions
About this article
Cite this article
Jiao, L., Wang, S., Chen, K. et al. Dynamic response to climate change in the radial growth of Picea schrenkiana in western Tien Shan, China. J. For. Res. 33, 147–157 (2022). https://doi.org/10.1007/s11676-021-01336-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11676-021-01336-6