Abstract
Forest structure and function are subject to risks of growth declines from intensified drought and frequent extreme events related to climate warming. Knowledge of tree growth declines will help anticipate future responses of forests to climate change. In this study, we investigated tree growth declines over the last four centuries in a juniper forest on the eastern Tibetan Plateau. By analyzing the radial growth trajectories of individual trees, we identified two events of intense growth decline, one in 1817–1830 and the other in 1969–1999 over the past four centuries. The intensity of the recent decline was unprecedented in the period under study. Ring-width chronology showed a positive correlation with self-calibrating Palmer Drought Severity Indices and a negative correlation with mean monthly temperatures in May and June. The recent intensified growth decline may have been due to temperature-induced frequent droughts in the study area. Our findings suggest that trees in this juniper forest may face a higher risk of growth decline and even mortality under continued climate warming.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Tree growth declines, which affect the structure and function of forests, are expected to increase with global warming (Allen et al. 2010; Anderegg et al. 2016; Cailleret et al. 2017). While much attention has been paid to recent periods of growth reductions (Williams et al. 2010; Shestakova et al. 2016; Chen et al. 2017), little is known about the history of such events. It is a question whether recent declines have intensified relative to those in the past. Answers to this question are important for detecting the causes of tree growth declines and for predicting potentially dangerous thresholds of decline in scenarios of future climate extremes and disturbances (Trumbore et al. 2015).
Researchers study the history of tree growth declines through dendrochronological methods. Their findings have been inconsistent over the intensity of recent growth declines relative to those in the past. Some researchers considered that growth reductions intensified due to climate warming in recent years (Liu et al. 2013; Gómez-Guerrero et al. 2013; Wong and Daniels 2017). Others suggest that there were more intensive growth declines in the past than in recent decades, but that declines would intensify with continued climate warming (Williams et al. 2013). Further studies of historical growth declines in different forests are required to settle this argument.
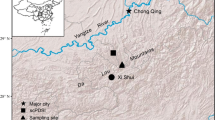
In this study, we address this issue by looking at tree-ring data in a juniper forest in Nangqian County of the eastern Tibetan Plateau (96.64° E, 32.19° N). The County is an important part of the headwaters of the Yangze, Yellow, and Mekong rivers (Fig. 1). Forests in this area are important for maintaining ecosystem services in and beyond the three rivers’ source (Xu et al. 2008; Shen et al. 2015). These services depend on healthy forests. As climate changes and disturbances become more frequent, forest health may be affected. Growth decline and mortality in recent decades have been observed in the northeastern margin of the Tibetan Plateau (Fang et al. 2015; Yu et al. 2015; Liang et al. 2016). Compared to the current symptoms of decline, little attention has been given to long-term dynamics of growth reduction in this region. Putting the current growth declines in a longer time scale will help better understand the characteristics of decline events in this region. The objective of this study was to identify the history of tree growth declines for the forest in Dongba, Nangqian County, and to examine whether the current forest exhibits intensified decline relative to the past.
Data and methods
Study area and tree-ring data
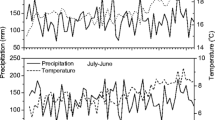
The sampling site is isolated from villages and the elevation is 4200 m a.s.l. where there is little human activity (Fig. 2). According to instrumental records at the Nangqian meteorological station (96.48° E, 32.20° N) since 1957, the annual mean temperature is 4.0 °C, and mean monthly temperatures range from − 6.5 °C in January to 13.2 °C in July. Annual total precipitation is 520 mm with approximately 79% occurring June to September. Annual mean temperatures show a rising trend whereas annual total precipitation does not (Fig. 3).
The forest consists of a single species, Tibetan juniper, Juniperus tibetica Kom. Twenty-nine trees were sampled in June 2002, and measured and crossdated. The site chronology was integrated into a tree-ring network for the reconstruction of past climates (Zhang et al. 2015). We examined the ring-width sequences of individual trees in detail. Age-related growth trends in each ring sequence was removed using software ARSTAN (Cook 1985), in which a negative exponential curve or horizontal line through the means was fitted to the data. Standardized tree-ring indices were obtained for each sample by dividing the measured ring widths by the fitted curves or lines. These tree-ring indices formed the database for comparison of growth trajectories among individual trees.
Detection of tree growth decline
We examined the radial growth trajectory of each tree and used the definition for tree growth decline of Li and Zhang (2017). This definition is based on the assumption that normal growth of trees fluctuates around a mean, and a sustained below average growth together with a severe growth reduction is outside its natural variation (Li and Zhang 2017). The periods of growth declines were identified when tree-ring indices met the following four criteria:
-
1.
Tree-ring indices are persistently lower than one for at least 9 years;
-
2.
The mean of the tree-ring indices is lower than 0.7 in each growth decline period;
-
3.
At least 2 years of tree-ring indices reach a minimum level of 0.5 during each growth decline period; and,
-
4.
There exists at least one tree in each growth decline period whose tree-ring indices do not match one of the above rules.
We tested a sequence of the threshold values and considered that these values were reasonable to illustrate a decline in tree growth. The percentage of trees with growth decline was calculated by years using the Expressed Population Signal (EPS) above 0.85. A historical decline event was defined when this percentage was greater than 25%.
The recovery times of individual trees in the declining periods were calculated. If the mean of the tree-ring indices over five consecutive years was > 1, it was considered that the tree had recovered to its normal growth state, and the years from the beginning of the declining period to the recovery year were calculated.
Climate-response analysis
To explore the climate-growth relationship, a site tree-ring chronology was developed by averaging ring-width indices. Pearson correlation analysis was then carried out between the standard chronology and climatic variables, including monthly mean temperatures and monthly total precipitation for the period 1957–2001. This correlation analysis was also calculated between standard chronology and the self-calibrating Palmer Drought Severity Index (scPDSI), a measure of soil moisture that incorporates the influence of both temperature and precipitation (Dai et al. 2004). The scPDSI values were obtained from the global data set (http://climexp.knmi.nl) at the grid (0.5° × 0.5° latitude and longitude) nearest the sample site during 1957–2001. Correlation analysis was performed for a 12-month period from October of the previous year to September of the current year.
Results
Tree-ring data
Descriptive statistics of the tree-ring site are in Table 1. The master series is 542 years from 1460 to 2001. This site includes 29 tree-ring series, 123,031 rings in total and 31 absent rings. The mean length of all series is 335 years. The average mean sensitivity is 0.274, indicating that the ring-width chronology shows relatively high inter-annual variability. The series intercorrelation and EPS are 0.667 and 0.864, respectively. The tree-ring chronologies meet the 0.85-EPS criterion after the year 1570. Therefore, we used the period 1570–2001 (432 years) for further analysis.
Historical growth declines
Tree growth declines are ubiquitous in individual trees throughout the past four centuries, and such declines synchronized to form decline events in 1817–1830 and in 1969–1999 (Fig. 4). Decline in tree growth intensified abruptly in 1969–1999, in which the percentage of trees with growth decline and the duration are both twice those in 1817–1830 (Fig. 4). The highest percentage of trees with growth decline reached 38% and 70% in 1817–1830 and 1969–1999, respectively. The first growth decline events lasted 14 years since 1817. The recent growth decline lasted 31 years since 1969. The recovery time of individual trees averaged about 21 years in the 1817–1830 period (Table 2).
Average ring indices were compared in trees with growth decline and those not showing a decline in growth during the two periods. During 1817–1830, seven trees showed a growth decline and 22 did not. There were 18 trees with growth decline and six with regular growth in the period 1969–1999. There are significant differences in growth trajectories between the two groups (Fig. 5). The trends in growth in the recent period of decline show that the number of trees with reduced growth decreased in 1969–1980 and slowly increased after 1980 (Fig. 5b). These growth trends also show in standard chronology of this study site (Fig. 4b).
Growth-climate relationship
The radial growth shows a positive correlation with the mean scPDSI in and before the growing season, which are displayed in October (r = 0.34, n = 46, p < 0.05) to November (r = 0.30, n = 46, p < 0.05) of the previous year and June (r = 0.29, n = 46, p < 0.05) of the current year. Zhang et al. (2015) also reported a positive correlation (r = 0.31, n = 45, p < 0.05) between tree-ring chronology in Dongba, southwest China, and moisture conditions in the growing season. A negative correlation is shown between growth and mean temperatures in May (r = − 0.43, n = 46, p < 0.05) and June (r = − 0.34, n = 46, p < 0.05) of current year, the growing period (Fig. 6).
Discussion
This study revealed that declines in tree growth were prevalent over the past four centuries in this juniper forest on the eastern Tibetan Plateau. However, intense declines occurred twice and the most recent was more intense than that of the earlier one.
Past growth declines can be identified by a comparison of growth trajectories of individual trees. Previous studies reveal that low radial growth could be an indicator of growth decline. By comparing the radial growth of trees showing a decline in growth with those showing regular growth, growth decline is accompanied with long-time low radial growth (Amoroso et al. 2012; Camarero et al. 2015). In our study, growth indices of trees with growth decline were significantly lower than trees showing regular growth during two intense growth decline periods (Fig. 5). Thus, by defining tree growth decline as long-term low radial growth, we successfully identified growth declines in the past decades.
Growth decline intensified dramatically in recent decades in the moisture-limited forests of our study area. A major decline was also observed in a nearby sample site in Dongzhong, Yushu County and in Qinghai Province, which is also located in the headwaters of the three rivers (Fig. 1). This growth decline occurred in 1968–1981, with 43% of trees with growth decline, which is much higher than the percentage of trees with growth decline over the past 200 years (Fig. 7). The recent intensified growth decline in the study area supports a marked vegetation degradation in the region (Yu et al. 2009; Zhao et al. 2012). Similar growth declines were also identified in areas of the northeastern Tibetan Plateau. An analysis of regional NDVI data and regional forest basal area increments calculated by tree-ring data at upper tree lines along the Qilian mountains, northeastern Tibetan Plateau, reveal a major growth decline in the late twentieth century (Yu et al. 2015). In the northeast, increasing frequency of missing tree rings and rising tree mortality in recent decades were observed in high-elevation, semi-arid Qinghai spruce forests (Liang et al. 2016). These growth declines were attributed to warming-induced drought. In contrast to the results in this study, previous research demonstrated that tree growth decline was not intensified in recent decades, such as stable tree growth in high-elevation cold-limited forests of the Spanish Pyrenees (Granda et al. 2017), and increasing tree growth in the southern area of the polar and alpine tree-line ecotone of Finnish Lapland (Franke et al. 2017). Tree growth in these areas is limited by low temperatures that could benefit from climate warming (Granda et al. 2017). However, moisture is the main limiting factor of growth in our study area, supported by the positive relationship between tree growth and scPDSI (Fig. 6). This growth-climate relationship likely is the cause of trees showing intense growth decline under recent climate warming. Previous studies also demonstrated that tree growth declined markedly in moisture-limited forests under climate warming on the margins of the Tibetan Plateau (Fang et al. 2015; Liang et al. 2016) and in semi-arid forests of Inner Asia (Liu et al. 2013). Combined with a significant negative correlation between tree growth and mean summer temperature (Fig. 6), recent climate warming most likely has a negative impact on tree growth in this moisture-limited forest.
The recent unprecedented tree growth decline was possibly caused by warming-induced frequent droughts. Increased temperatures in summer would intensify the magnitude and frequency of droughts through rising moisture deficits (Chen et al. 2017) especially as the amount of precipitation remained stable since 1957 (Fig. 3). Frequent droughts in recent decades have been recorded by data in the Third Pole Environment Database (http://www.tpedatabase.cn). Severe droughts occurred more than ten times since the 1970s in Nangqian County. Frequent droughts were also detected in the southeastern periphery of the Tibetan Plateau (Ma et al. 2017). These warming-induced droughts could cause an abrupt decline in tree growth (Cailleret et al. 2017). Trees under drought stress will close their stomata to prevent hydraulic failure. As a result, photosynthetic uptake of carbon dioxide would diminish, causing carbon starvation (Manion 1991; Bréda et al. 2006; Adams et al. 2013). In addition, elevated temperatures during a drought period could increase respiratory demands, which will exacerbate the carbon-starvation process (McDowell et al. 2008). The difference in carbohydrate balances among individual trees might cause the phenomenon that growth in some trees decline, but others do not (Sevanto et al. 2014). Trees that have adequate carbon reserves for metabolic demand for carbohydrates could maintain normal growth during a drought period. This causal link between tree growth and climate warming is supported by the growth-climate relationship (Fig. 6), and has been revealed by previous studies in forests worldwide. For example, a drastic reduction of radial growth over several years in the past century was caused by warming-induced droughts in the northern Yunnan-Guizhou Plateau (Su et al. 2017). In aspen-dominated forests in the boreal forest of western Canada, prolonged and large-scale droughts associated with climate warming have caused significant growth decline (Chen et al. 2017). Decreasing tree growth and increasing mortality in recent decades within the southwestern United States were caused by increasing aridity and higher temperatures (Williams et al. 2010). Based on these studies, the recent intensified growth decline in our study area was most likely related to climate warming.
Recent temperature increases might have a more significant negative impact on tree growth than did past volcanic eruptions in this juniper forest. Intense growth declines occurred twice in the past four centuries (Fig. 4). The first appeared in 1817–1830 on the Tibetan Plateau with an abnormal climate fluctuation induced by the 1815 Mount Tambora eruption in Indonesia (Liu et al. 2005; Bräuning 2006; Liang et al. 2008). This intense decline in growth resulted from this abnormal climate fluctuation. A possible explanation is that volcanic aerosols would intercept incoming solar radiation, lowering normal temperatures and reducing photosynthesis (Yadav 2007). The most recent intense growth decline event, caused by frequent temperature-induced droughts, was twice as severe as the one in 1817–1830 (Fig. 4), which indicates that recent climate warming has a stronger negative impact on tree growth in this juniper forest than a past volcanic eruption.
Conclusion
We propose a method that compares growth trajectories among individual trees to identify historical growth declines. The history of tree growth demonstrated an unprecedented decline over several years in the past four centuries in this juniper forest on the eastern Tibetan Plateau. In addition, a significant relationship between tree growth and climate factors indicates that climate warming has a strong negative impact in this moisture-limited forest. By putting the current growth decline in the context of the past four centuries, we found that the present forest shows an intensified growth decline because of climate warming. Trees in this juniper forest might be at a higher risk of growth reduction, and even mortality, with continued climate warming. Further examination of tree growth declines on the Tibetan Plateau is necessary to understand forest dynamics in future climate scenarios and to improve forest management.
References
Adams HD, Germino MJ, Breshears DD, Barron-Gafford GA, Guardiola-Claramonte M, Zou CB, Huxman TE (2013) Nonstructural leaf carbohydrate dynamics of Pinus edulis during drought-induced tree mortality reveal role for carbon metabolism in mortality mechanism. New Phytol 197:1142–1151
Allen CD, Macalady AK, Chenchouni H, Bachelet D, McDowell N, Vennetier M, Kitzberger T, Rigling A, Breshears DD, Hogg EH, Gonzalez P, Fensham R, Zhang Z, Castro J, Demidova N, Lim JH, Allard G, Running SW, Semerci A, Cobb N (2010) A global overview of drought and heat-induced tree mortality reveals emerging climate change risks for forests. For Ecol Manag 259(4):660–684
Amoroso MM, Daniels LD, Larson BC (2012) Temporal patterns of radial growth in declining Austrocedrus chilensis forests in Northern Patagonia: the use of tree-rings as an indicator of forest decline. For Ecol Manag 265:62–70
Anderegg WRL, Martinez-Vilalta J, Cailleret M, Camarero JJ, Ewers BE, Galbraith D, Gessler A, Grote R, Huang CY, Levick SR, Powell TL, Rowland L, Sánchez-Salguero R, Trotsiuk V (2016) When a tree dies in the forest: scaling climate-driven tree mortality to ecosystem water and carbon fluxes. Ecosystems 19(6):1133–1147
Bräuning A (2006) Tree-ring evidence of ‘Little Ice Age’ glacier advances in southern Tibet. Holocene 16(3):369–380
Bréda N, Huc R, Granier A, Dreyer E (2006) Temperate forest trees and stands under severe drought: a review of ecophysiological responses, adaptation processes and long-term consequences. Ann For Sci 63:625–644
Cailleret M, Jansen S, Robert EMR, Desoto L, Aakala T, Antos JA, Beikircher B, Bigler C, Bugmann H, Caccianiga M, Čada V, Camarero JJ, Cherubini P, Cochard H, Coyea MR, Čufar K, Das AJ, Davi H, Delzon S, Dorman M, Gea-Izquierdo G, Gillner S, Haavik LJ, Hartmann H, Hereş AM, Hultine KR, Janda P, Kane JM, Kharuk VI, Kitzberger T, Klein T, Kramer K, Lens F, Levanic T, Calderon JCL, Lloret F, Lobo-Do-Vale R, Lombardi F, Rodríguez RL, Mäkinen H, Mayr S, Mészáros I, Metsaranta JM, Minunno F, Oberhuber W, Papadopoulos A, Peltoniemi M, Petritan AM, Rohner B, Sangüesa-Barreda G, Sarris D, Smith JM, Stan AB, Sterck F, Stojanović DB, Suarez ML, Svoboda M, Tognetti R, Torres-Ruiz JM, Trotsiuk V, Villalba R, Vodde F, Westwood AR, Wyckoff PH, Zafirov N, Martínez-Vilalta J (2017) A synthesis of radial growth patterns preceding tree mortality. Glob Change Biol 23(4):1675–1690
Camarero JJ, Gazol A, Sangüesa-Barreda G, Oliva J, Vicente-Serrano SM (2015) To die or not to die: early warnings of tree dieback in response to a severe drought. J Ecol 103:44–57
Chen L, Huang JG, Alam SA, Zhai LH, Dawson A, Stadt KJ, Comeau PG (2017) Drought causes reduced growth of trembling aspen in western Canada. Glob Change Biol 23:2887–2902
Cook ER (1985) A time series analysis approach to tree ring standardization. University of Arizona, Tucson, pp 76–88
Dai AG, Trenberth KE, Qian TT (2004) A global data set of Palmer drought severity index for 1870–2002: relationship with soil moisture and effects of surface warming. J Hydrometeorol 5(6):1117–1130
Fang KY, Frank D, Zhao Y, Zhou FF, Seppä H (2015) Moisture stress of a hydrological year on tree growth in the Tibetan Plateau and surroundings. Environ Res Lett 10:034010
Franke AK, Bräuning A, Timonen M, Rautio P (2017) Growth response of Scots pines in polar-alpine tree-line to a warming climate. For Ecol Manag 399:94–107
Gómez-Guerrero A, Silva LCR, Barrera-Reyes M, Kishchuk B, Velázquez-Martínez A, Martínez-Trinidad T, Plascencia-Escalante FO, Horwath WR (2013) Growth decline and divergent tree ring isotopic composition (δ 13C and δ 18O) contradict predictions of CO2 stimulation in high altitudinal forests. Glob Change Biol 19:1748–1758
Granda E, Camarero JJ, Galván JD, Sangüesa-Barreda G, Alla AQ, Gutierrez E, Dorado-Liñán I, Andreu-Hayles L, Labuhn I, Grudd H, Voltas J (2017) Aged but withstanding: maintenance of growth rates in old pines is not related to enhanced water-use efficiency. Agric For Meteorol 243:43–54
Li Y, Zhang QB (2017) History of tree growth declines recorded in old trees at two sacred sites in northern China. Front Plant Sci 8:1779
Liang EY, Shao XM, Qin NS (2008) Tree-ring based summer temperature reconstruction for the source region of the Yangtze River on the Tibetan Plateau. Glob Planet Change 61(3–4):313–320
Liang EY, Leuschner C, Dulamsuren C, Wagner B, Hauck M (2016) Global warming-related tree growth decline and mortality on the north-eastern Tibetan plateau. Clim Change 134(1–2):163–176
Liu XH, Qin DH, Shao XM, Chen T, Ren JW (2005) Temperature variations recovered from tree-rings in the middle Qilian Mountain over the last millennium. Sci in China Ser D 48(4):521–529
Liu HY, Williams AP, Allen CD, Guo DL, Wu XC, Anenkhonov OA, Liang EY, Sandanov DV, Yin Y, Qi ZH, Badmaeva NK (2013) Rapid warming accelerates tree growth decline in semi-arid forests of Inner Asia. Glob Change Biol 19(8):2500–2510
Ma SM, Zhou TJ, Angélil O, Shiogama H (2017) Increased chances of drought in southeastern periphery of the Tibetan Plateau induced by anthropogenic warming. J Clim 30(16):6543–6560
Manion PD (1991) Tree disease concepts. Prentice Hall, Upper Saddle River
McDowell N, Pockman WT, Allen CD, Breshears DD, Cobb N, Kolb T, Plaut J, Sperry J, West A, Williams DG, Yepez EA (2008) Mechanisms of plant survival and mortality during drought: why do some plants survive while others succumb to drought? New Phytol 178:719–739
Sevanto S, McDowell NG, Dickman LT, Pangle R, Pockman WT (2014) How do trees die? A test of the hydraulic failure and carbon starvation hypotheses. Plant Cell Environ 37(1):153–161
Shen MG, Piao SL, Jeong SJ, Zhou LM, Zeng ZZ, Ciais P, Chen DL, Huang MT, Jin CS, Li LZX, Li Y, Myneni RB, Yang K, Zhang GX, Zhang YJ, Yao TD (2015) Evaporative cooling over the Tibetan Plateau induced by vegetation growth. PNAS 112(30):9299–9304
Shestakova TA, Gutiérrez E, Kirdyanov AV, Camarero JJ, Génova M, Knorre AA, Linares JC, Resco de Dios V, Sánchez-Salguero R, Voltas J (2016) Forests synchronize their growth in contrasting Eurasian regions in response to climate warming. PNAS 113(3):662–667
Su JJ, Gou XH, Deng Y, Zhang RB, Liu WH, Zhang F, Lu M, Chen Y, Zheng WJ (2017) Tree growth response of Fokienia hodginsii to recent climate warming and drought in southwest China. Int J Biometeorol 61(12):2085–2096
Trumbore S, Brando P, Hartmann H (2015) Forest health and global change. Science 349(6250):814–818
Williams AP, Allen CD, Millar CI, Swetnam TW, Michaelsen J, Still CJ, Leavitt SW (2010) Forest responses to increasing aridity and warmth in the southwestern United States. PNAS 107(50):21289–21294
Williams AP, Allen CD, Macalady AK, Griffin D, Woodhouse CA, Meko DM, Swetnam TW, Rausche SA, Seager R, Grissino-Mayer HD, Dean JS, Cook ER, Gangodagamage C, Cai M, McDowell NG (2013) Temperature as a potent driver of regional forest drought stress and tree mortality. Nat Clim Change 3:292–297
Wong CM, Daniels LD (2017) Novel forest decline triggered by multiple interactions among climate, an introduced pathogen and bark beetles. Glob Change Biol 23(5):1926–1941
Xu XD, Lu CG, Shi XH, Gao ST (2008) World water tower: an atmospheric perspective. Geophys Res Lett 35(20):L20815
Yadav RR (2007) Basin specificity of climate change in western Himalaya, India: tree-rings evidences. Curr Sci India 92(10):1424–1429
Yu BH, Lu CH, Lu TT, Yang AQ, Liu C (2009) Regional differentiation of vegetation change in the Qinghai-Tibet Plateau. Progr Geogr 28(3):391–397 (in Chinese with an English abstract)
Yu L, Huang L, Shao XM, Xiao FJ, Wilmking M, Zhang YX (2015) Warming-induced decline of Picea crassifolia growth in the Qilian Mountains in recent decades. PLoS ONE 10(6):e0129959
Zhang QB, Eavns MN, Lyu LX (2015) Moisture dipole over the Tibetan Plateau during the past five and a half centuries. Nat Commun 6:8062
Zhao X, Zhou DJ, Fang JY (2012) Satellite-based studies on large-scale vegetation changes in China. J Int Plant Biol 54(10):713–728
Acknowledgements
We thank professor Qi-Bin Zhang for his supervision on this thesis. We thank members in Qi-Bin Zhang’s team for assistance in tree-ring sampling, crossdating and help with preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Project funding: This study was financially supported by the National Natural Science Foundation of China project (No. 31330015).
The online version is available at http://www.springerlink.com
Corresponding editor: Hu Yanbo.
Rights and permissions
About this article
Cite this article
Mou, YM., Fang, O., Cheng, X. et al. Recent tree growth decline unprecedented over the last four centuries in a Tibetan juniper forest. J. For. Res. 30, 1429–1436 (2019). https://doi.org/10.1007/s11676-018-0856-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11676-018-0856-6