Abstract
Agarwood, the resinous product of Aquilaria spp. (Thymelaeaceae), is highly valued for medicinal and fragrant purposes. Unsustainable forest harvesting contributes to the declining population in the wild, threatening their existence. As a protection effort, cultivation occurred in range countries, mainly in Asia, effectively establishing mass plantations. The success of domesticating Aquilaria relies heavily on technological advancement in agarwood induction, without which the entire thriving industry will collapse. In this paper, we describe efforts since 1929 and current progress and variation in induction as practiced in Asia. The methods include traditional practices and artificial induction via inoculum and chemical inducer, experimented and patented or otherwise. Artificial induction methods are being developed to intensify agarwood production in terms of yield and quality. While traditional methods are commonly applied by rural planters, artificial methods with the use of specific agents/chemicals are seen as more appealing for inducing agarwood, and have a higher demand from entrepreneurs and plantation owners. Several issues related to induction technology faced by stakeholders in agarwood cultivation are presented, such as safety levels, cost, yields, and quality. We conclude by highlighting remaining challenges in induction methods and their associated technologies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The plant family Thymelaeaceae is known for its incense-producing tree species, especially from the genera Aquilaria and Gyrinops. Endemic to the Indomalayan region, there are a total of 21 recorded Aquilaria spp. and nine Gyrinops spp. (The Plant List 2013). Aquilaria is the main agarwood-producing genus. The natural range for Aquilaria includes Bangladesh, Bhutan, Cambodia, China, India, Indonesia, Laos, Malaysia, Papua New Guinea, Thailand and Vietnam (Lee and Mohamed 2016). In the wild, agarwood formation is often related to natural factors such as thunder strikes, broken branches due to strong winds, and pest and disease infestations. These factors can create wounds or openings for pathogenic microbes to enter and trigger the tree’s defense system (Mohamed et al. 2010). The infected area surrounding the wound over time turns dark brown or black into what is known as agarwood or ‘gaharu’ in the Malay language (Mohamed et al. 2014). Agarwood formation takes place under the bark, thus making it difficult to detect from the outside. A quick way to search for agarwood as practiced by unscrupulous harvesters is by indiscriminately felling the wild Aquilaria trees, causing their numbers to decline significantly (Wyn and Anak 2010). The extensive harvesting of wild mother trees disrupts the reproduction cycle and can limit the availability of juvenile trees for future supplies. Due to this concern, international conservation initiatives listed nine Aquilaria spp. under the International Union for Conservation of Nature (IUCN) Red List of Threatened Species in 1998 (IUCN 2017), and placed all Aquilaria and Gyrinops spp. under Appendix II of the Convention on International Trade in Endangered Species (CITES) in 2005 (CITES Secretariat 2004). Given the significance of its sustainability, any trading activity involving the species must adhere to relevant licenses (UNEP-WCMC 2015).
Agarwood is a valuable product, and its future prospects are bleak if current exploitation practices continue. In this review, we examine historical records and summarize the advancement of agarwood induction methods, essential information to allow for better understanding of current technologies and the development of improved ones. By reviewing its evolution over time, planters, traders, and researchers will be better informed about the effectiveness of these induction methods. In addition, stakeholders may become more inclined to the concept of sustainability in the production and management of agarwood resources, both from cultivated and natural forests. Generally, this review examines methods in artificial agarwood induction by taking a closer look at the examples of induction practices in Asia, outlining their progress over almost a decade, and concludes by emphasizing future directions.
Efforts to cultivate agarwood in Asia
The chance of coming upon a wild-sourced agarwood in a natural forest is extremely low, estimated at about 10% (Ng et al. 1997), which explains its high market demand and price. With the international law governing natural agarwood resources in place, and in line with the conservation efforts of CITES member countries, those who are in the agarwood trade have diverted their attention to cultivated Aquilaria. The range countries in Asia are now promoting Aquilaria cultivation to ensure a continuous supply of agarwood. Due to its protected status, Aquilaria planting is under regulation by CITES representatives in each member country. In Malaysia, efforts in Aquilaria planting cover a total of 1300 hectares and 1.2 million trees at present (Hashim et al. 2016). In Indonesia, the Forestry Research and Development Agency (FORDA) reported a rough estimation of the total planted agarwood-producing trees at over 3.4 million. The survey represented only 1/3 of the provinces that have planted the species (Turjaman and Hidayat 2017). In China, over 20 million Aquilaria trees have been cultivated, covering nearly 5300 hectares. The number is expected to double due to the recognition of cultivated agarwood for pharmaceutical purposes (Yin et al. 2016). In Bangladesh in 2007 alone, almost 800,000 Aquilaria seedlings were planted (Chowdhury et al. 2016). Bhutan and India have approximately 20,000 and 10 million trees, respectively, while Nepal has recorded less than 1000 trees in home gardens. Southeast Asian countries like Myanmar and Vietnam have 35,000 and > 1 million trees, respectively, in home gardens. Cambodia and Thailand are also active in planting Aquilaria spp., however, no estimation has been given on plantation size and number of trees (ITTO-CITES 2015).
At present, the cultivation of agarwood-producing species has gone from home gardens on small lands in the backyards to large-scale plantations under systematic management. Some of these plantations have been developed out of smallholder initiatives, and some were set up by enterprising individuals with trade interests who put hopes on high returns on their investment. Others are plantations run by private companies and government agencies. The species of choice for cultivation in the range countries is influenced by: (a) the species preferred by the targeted market; (b) land suitable for planting a particular species; and, (c) the establishment of an efficient induction technique for specific species. At present, the commonly planted commercial species are A. crassna Pierre ex Lecomte, A. malaccensis Lam. and A. sinensis (Lour.) Gilg. They are widely cultivated in Southeast Asia. Aquilaria malaccensis is the main species planted in India, while A. sinensis is primarily planted in southern China (Lee and Mohamed 2016).
Methods in agarwood induction
With the increased number of Aquilaria plantations, efforts have been made to develop artificial induction methods to stimulate agarwood formation. The aim is to increase the yield of cultivated agarwood to an acceptable quality for targeted downstream purposes. These methods must be practical for large-scale plantations, with the primary objective to yield maximum agarwood supplies in the shortest time possible. Coupled with good agriculture practices, the artificial method is anticipated to provide higher agarwood yields in comparison to the natural process, where ideally its quality must closely resemble wild agarwood, if not higher.
Agarwood is a resinous compound produced by Aquilaria spp. as a response to abiotic and biotic injury. The process of agarwood formation takes place in the parenchyma cells, including the phloem, and vessels (Nobuchi and Sahri 2008; Mohamed et al. 2013). The tree reacts to stress and foreign intrusions by activating its cell machinery to synthesize substances that act as a biochemical defense against physical or biological agents (Rasool and Mohamed 2016). These anatomical structures are responsible for producing, storing and distributing agarwood constituents to the wounded area, filling up the compartments, becoming solid and impregnating the wood. This process is the foundation for developing artificial methods to induce agarwood. At present, artificial agarwood induction methods can be classified into two groups: conventional and non-conventional.
Conventional methods
Conventional methods are traditional practices passed down from one generation to another and practiced sustainably on trees growing on individual and ancestral lands as well as in natural forests. China has a long history of agarwood usage and had developed various artificial agarwood-inducing methods since before the Song Dynasty (A.D. 960) (Liu et al. 2013). The basic concept is to physically wound the tree as it is the main trigger to agarwood formation (Pojanagaroon and Kaewrak 2005). This explains why farmers use severe methods like burning-chisel-drilling, partial-trunk-pruning, wounding using an axe or machete, bark removal, cauterizing, cutting, holing and nailing on their trees (Liu et al. 2013; Rasool and Mohamed 2016). These methods may be cost-effective but they are laborious. In addition, a longer time may be required to yield agarwood of uncertain quality. Using conventional techniques, agarwood is formed only at the injured area of the stem, implying that the yield correlates with the number and magnitude of physical injuries.
Non-conventional methods
Non-conventional induction methods are developed to mimic natural events leading to agarwood formation. Generally, these methods apply a catalyst or inducer into the tree to stimulate agarwood formation. A minor physical wounding is required on the trunk to make an entry point prior to applying the inducer. Depending on its application, the inducer may be liquid, semi-solid or solid. Currently available inducers in the market are classified into (1) biological inocula, and (2) chemical inducers. A summary of the relevant studies and the development of agarwood inducers using these non-conventional methods are shown in Table 1.
Biological Inducers
These commonly contain microbes such as yeast, fungi and other microbial flora found naturally. Another terminology often used by planters is ‘inoculum’. Some traders use the same term for chemical inducers, which is technically incorrect because inoculum refers to a biological agent. The technique involves applying fungal cultures into the tree to mimic the pathological condition of Aquilaria in the wild. The culture could be ‘pure’, as normally isolated in a controlled environment such as in a laboratory, or ‘mixed’ as those commonly produced by individual proprietors. The pure culture can be in solid form, grown on agar media in Petri dishes, or liquid, grown in broth media. Most of the mixed cultures are concocted by proprietors based on their own endeavors in formulating inocula and experimenting with an inoculum on their own trees. Such inocula are produced using locally available ingredients such as molasses, yeast, salt, and fruit juice. The inoculum may contain various types of microbes and fermentation products due to the uncontrolled environment in which they were prepared. Biological inducers may require a long incubation time before harvesting to produce darker wood and subsequently a better form of agarwood (Mohamed et al. 2014). The long incubation period allows the microorganism to multiply and colonize the tree. Triggered by the invasion, the tree’s defense system is activated to obstruct further foreign penetration by producing agarwood resin as a barrier (Rasool and Mohamed 2016).
Inoculum development started prior to 1929 when Tunstall first reported inoculating fungi into Aquilaria agallocha Roxb (Gibson 1977). Others have since isolated fungi from naturally occurring agarwood in wild trees to investigate the role of fungi in agarwood formation (Table 1). The selection of fungi was mostly endophytic, isolated from either healthy or diseased wood, and then inoculated into a healthy Aquilaria tree. One fungal genus that has attracted interest, especially researchers in Indonesia is Fusarium (e.g. Santoso et al. 2011; Iskandar and Suhendra 2012). Other fungi such as Paraconiothyrium variabile Riccioni, Damm, Verkley and Crous, Botryosphaeria dothidea (Moug. ex Fr.) Ces. & De Not., Aspergillus niger Tiegh, Fomitopsis spp., and the parasitic fungus Lasiodiplodia theobromae (Pat.) Griffon and Maubl. (Cui et al. 2013; Tian et al. 2013; Zhang et al. 2014) were also investigated, and several have been patented for commercial inocula production (Huang 2013; Wei et al. 2014). A comprehensive list of fungi is available with some supporting results from field trials (Table 1). The purpose of testing a wide variety of fungal species is to identify the most suitable species or isolate that can produce high quality agarwood, however the outcomes may vary when applied at different sites.
Chemical inducers
Chemical inducers appear promising as they act rapidly, are easy to apply, and are available in precise strengths. Using chemicals to induce the tree’s defense mechanism toward promoting resin production is not solely applied to Aquilaria spp., but is also widely used with other species such as Norway spruce [Picea abies (L.) H. Karst.] (Martin et al. 2002). Picea abies, similar to other conifers, produces terpenoid-based oleoresins which act as constitutive and inducible defenses against insects, herbivores and pathogens (Martin et al. 2002; Zeneli et al. 2006; Phillips et al. 2007). For Aquilaria, it is uncertain when people first experimented with chemicals as many have used salt (sodium chloride) and vinegar (acetic acid). However, specialized chemicals came to light when signaling molecules such as methyl jasmonate, ethylene, and salicylic acid were found to induce the production of agarwood fragrant constituents in Aquilaria callus and suspension cells (Okudera and Ito 2009) (Table 1). It is not surprising that these molecules are common ingredients in commercial inducers. The content of an inducer may include chemicals such as methyl jasmonate, hydrogen peroxide, sodium methyl bisulfite, sodium chloride, ferric chloride, ferrous chloride, formic acid, cellobiose, salicylic acid, and iron powder, and biological-derived substances like yeast extract and chitin (Blanchette and van Beek 2005; Okudera and Ito 2009; Wijitphan 2009; Wei et al. 2010; Lan and Li 2013; Thanh et al. 2015; Fu and Xu 2016; Tang and Liu 2016). However, the exact chemicals, combinations and concentrations are only known to the proprietor who developed the inducer. Chemical inducers need to be applied at an optimal strength because an excess could kill the tree. Therefore, many field trials must be conducted before the inducer may be commercialized.
Agarwood induction methods currently available in the Asian market
Countries in Asia are actively planting Aquilaria as the climate favors the growth of the numerous species, and this highlights the need for agarwood induction techniques. Innovative induction technology and effective inducers are needed to shorten the period before harvesting and to increase yields and quality of cultivated agarwood.
Agarwood induction technology
This technology is developing well in many Asian countries (Table 1). A significant improvement was achieved when field-tested Cultivated Agarwood Kits (CA-Kits) were introduced in Vietnam and became the first patented agarwood induction method (Blanchette and van Beek 2005). The technology produces agarwood by forming an artificial wound into the stem and inserting a tube into the wound to aid in aeration. A resin-inducing agent is then applied through the tube to reach the wounded tissues and stimulates resin production. Many of the recently modified and optimized agarwood induction techniques have been built on this technique.
Agarwood inducers based on chemical formulations are often developed together with a suitable delivery method. The various delivery methods were developed to ease the induction process, allowing for systematic application, which is suitable for large-scale plantations. Recently, a chemical inducer with an innovative delivery system has been invented which can induce agarwood formation in the whole tree. The patented technology is known as the “Whole-tree Agarwood-Inducing Technique” (Agar-WIT) (Wei et al. 2010; Zhang et al. 2012). Through this technology, less than 2 years is needed to produce agarwood of high quality (Liu et al. 2013). The method implements a device functioning as a drip bag to slowly release the liquid chemical into the tree through a drilled hole and then relies on the tree’s transportation system to distribute the chemical throughout the tree.
In the case of a successful fungal inoculum developed in Indonesia, preliminary selection of suitable fungi was carried out by isolating endophytic fungi from diseased wild Aquilaria trees. A fast-growing fungus is a prerequisite for a suitable inoculum, followed by the quality of agarwood produced upon infection (Santoso et al. 2011). The selection and testing process may take many years of evaluation. The inoculums were prepared in a broth culture and used fresh. Prior to inoculation, 0.3-mm diameter holes were drilled perpendicular to the tree bole, with pre-calculated depths and distances between each hole, from the bottom to the top of the tree stem, wherever accessible. Inoculation was carried out using a sterile syringe to deliver the broth-liquid into each hole. After 12 weeks of incubation, the tree produces a mixture of sesquiterpenes and aromatic compounds in considerable quantities. This suggests that the inoculum is a promising technique for agarwood formation (Faizal et al. 2017).
Inducers available in the market
Currently, there are many types of inducer formulations in the market; most are biological-based, although there are some chemicals (Table 2). Intellectual proprietors are in a race to develop convincing methods to influence planters to purchase their system as the potential profits are high. Some are backed by scientific evidence from field trials, others are based on personal experiences and observations. Generally, inducers are marketed through “word of mouth”, which are reviews from one client to other potential clients. Some inducer companies may begin by introducing themselves to planters and visiting plantations ready for induction. They would offer their products on a trial basis with certain options to choose from in regards to payment. Most sellers are planters themselves and often advertise their inducers by displaying evidences from their successful trials to build up the confidence of their clients. Some established companies offer a discounted price for their services by agreeing to share the profit for the agarwood sold using their system, while other companies would do the common ‘sell and buy’ practice. The confidence level for planters to use a certain induction technique escalates if the seller provides a yield promise, which arguably is not applicable at all time.
Maximizing yield
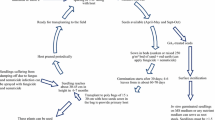
A proper delivery system ensures maximum agarwood yields which sometimes could be laborious and costly, depending on its handling protocol. To maximize yields, the tree has to be induced in a certain pattern depending on the nature of the inducer, biological or chemical. Generally, there are two types of formation. The first is when the agarwood is formed near the inoculation site as a result of fungal inoculum in solid or semi-solid form being directly injected into the pre-drilled hole in the stem. One example is the technique developed by Santoso et al. (2011), which applies F. solani culture into holes drilled in a certain pattern on the tree (Fig. 1a). The goal is to maximize yields by increasing the contact area between the inoculum and the host. In this example, agarwood formation is localized in the inoculation area. Another example also applies fungal culture onto the stem but in a spiral pattern (Chong et al. 2015; Fig. 1b). Whether the pattern is spiral, diamond or other, the aim is to create as many holes as possible, at the same time taking into account the structural strength of the tree as it has to withstand natural elements such as wind and rain during the incubation period which usually lasts one or more years. The resulting agarwood from the localized formation is usually in the form of wood chips.
The second type of formation has the agarwood formed from roots/base up to the top of the trunk in a systemic manner. It uses a suitable delivery method and a high volume of liquid solution delivered into the tree through holes along the stem at several levels from the base to the top (Fig. 1c). One end of a plastic or PVC tubing is inserted into the hole; while the other end is attached to a plastic bottle/bag; which functions as a reservoir, thus allowing the liquid to flow into the whole tree by transpiration uptake. This delivery concept is preferred by many planters because it is easy to apply and inexpensive. Due to the advancement in technology, delivery methods have evolved to suit various kinds of inocula and chemicals (Wei et al. 2010; Tian et al. 2013; Yang et al. 2014; Tang 2016; Yan and Lin 2016; Chen et al. 2017; Huang et al. 2017).
Quality of cultivated agarwood
Traditionally, agarwood is graded based on physical appearance, with its commercial value depending on resin content, colour and aroma (Mohamed and Lee 2016; Liu et al. 2017). A better practice to grade agarwood might be to determine its chemical content that is generally a complex mixture of aromatic compounds, sesquiterpenes, and chromones (Naef 2011). The latter are also responsible for the color intensity that also is a major contributor to grade level (Li et al. 2014). Arguably, agarwood of higher grades should contain specific sesquiterpene compounds such as agarofuran, agarol, jinkohol, agarospirol and others (Ismail et al. 2013, 2014). Although this could be subjective, these compounds are often detected in good quality agarwood in the market, regardless if natural or cultivated (Jamil et al. 2014; Jong et al. 2014). For comparison, the chemical compounds present in natural agarwood or agarwood induced by fungi or chemicals include major aromatic compounds such as agarospirol, aristolene, aromadendrene oxide-(1), benzylacetone, caryophyllene oxide, copaenol, eudesmol, guaiol, and hinesol (Appendix S1; Naef 2011). Others such as agarofuran, anethole, butyl isobutyl phthalate, cubebol, cubenol, dehydrofukinon and eremophilone are found in both fungal- and chemically-induced agarwood. This suggests that cultivated agarwood with a quality comparable to natural agarwood can be obtained using modern formulations regardless if they are biological- or chemical-based.
Advantages and disadvantages of non-conventional methods
Induction technology is the key to a successful sustainable agarwood production program. Aquilaria planters can now produce reliable yields of artificial agarwood, cultivated through the application of non-conventional methods. While there are a variety of inducers in the market from which to choose, the selection of a suitable inducer relies on the needs of the planter and is much affected by the advantages or disadvantages of the chosen inducing method.
Biological inducers and localized formation
This type of inducer is widely used among Aquilaria planters because it is easily available and dominates the market in Asia (Table 2). In addition, the biological aspect seems to be attractive and often relates to its being organic, environmentally friendly, and safe for handling and human consumption. Fungal-inoculated agarwood has a higher concentration of sesquiterpene compounds compared to mechanically wounded material (Chhipa et al. 2017; Faizal et al. 2017). One major drawback when using this method is that it requires extensive labor to make the holes to obtain maximum agarwood yields (Fig. 1a, b). This laborious and time-consuming approach is not practical for large-scale plantations or in a high-cost labor environment, but is applicable for home gardens. In addition, the agarwood formed concentrates near the inoculation site and rarely spreads. Different fungal strains or species may result in inconsistency of agarwood quality, and therefore are less promising. Optimization could be laborious as the environment could influence fungal efficacy (Tamuli et al. 2005; Santoso et al. 2011; Faizal et al. 2017).
Chemical inducers and systemic formation
Chemicals appear to be less popular than inocula because planters and agarwood users are skeptical on its impact on human health and the environment. The introduction of chemical inducers is to provide agarwood a consistent yield and quality, as well as for easy implementation in large-scale plantations. This delivery technique is thought to provide better distribution of agarwood formation compared to the localized application of inoculum. However, the application of chemical inducers requires good technical skills and precision, which many planters consider as complicated.
When properly formulated, chemical inducers could stimulate artificial agarwood, which closely resembles natural agarwood in terms of quality (Liu et al. 2013). This method may give a fast result and high yields, but more tests and demonstrations are needed to prove its effectiveness and efficiency. The most critical test is to prove that the agarwood produced using chemical inducers is safe to consume and for other applications. In general, chemical inducers could replace biological inducers in the market due to ease of application and rapid results. For quality control purposes, it is easier to prepare chemical inducers compared to biological ones as the latter contain living microorganisms. In industries where agarwood is used in pharmaceuticals and in traditional medicines, a standard quality is important to maintain the medicinal properties.
Recommendations and conclusions
Current stocks of natural agarwood are insufficient to meet demands in the international market. By cultivating agarwood, this pressure can be lifted. However, efficient techniques for inducing agarwood formation and practicalities of application need to be assessed. The techniques should be practical, inexpensive, and the results are reproducible. Further studies are needed to optimize and increase the efficiency of potential fungal strains for use as inocula suitable for various Aquilaria spp. in different geographical regions. Safety aspects regarding chemically-induced agarwood need to be clarified to convince consumers to use these types of inducer. Chemical inducers appear to be a new alternative that need more attention and wider dissemination. With the current progress in research and technologies, in the near future, it can be concluded that both types of inducers can be used optimally to produce agarwood of high quality comparable to wild agarwood.
References
Akhsan N, Mardji D, Sutisna M (2015) Response of Aquilaria microcarpa to two species of Fusarium under two different cultivation systems. J Trop For Sci 27:447–455
Bhattacharyya B, Datta A, Baruah HK (1952) On the formation and development of agaru in Aquilaria agallocha. Sci Cult 18:240–241
Biobenua Teknoloji Sdn Bhd (2013) Black Gold Bio Booster (BGBB). http://biobenuagaharu.com.my/?page_id=24. Accessed 12 Dec 2016
Bisnis Gaharu (2015) Inokulan Nanoteknologi Gaharuda. http://bisnisgaharu.com/tag/harga-serum-inokulasi/. Accessed 12 Dec 2016
Blanchette RA, van Beek HH (2005) Cultivated agarwood. U.S. Patent and Trademark Office, Washington, DC, pp 1–15. US Patent 6848211
Bose SR (1943) Agaru production by fungal inoculation in Aquilaria agallocha trees in Assam. In: Proceedings of the 30th Indian Scientific Congress. https://www.cabdirect.org/cabdirect/abstract/19430625104. Accessed 12 Dec 16
Budi S, Santoso E, Wahyudi A (2010) Identification of potential types of fungi on establishment agarwood stem of Aquilaria spp. Jurnal Silvikultur Tropika 1:1–5
Chen X, Sui C, Liu Y, Yang Y, Liu P, Zhang Z, Wei J (2017) Agarwood formation induced by fermentation liquid of Lasiodiplodia theobromae, the dominating fungus in wounded wood of Aquilaria sinensis. Curr Microbiol 74:460–468
Chhipa H, Chowdhary K, Kaushik N (2017) Artificial production of agarwood oil in Aquilaria sp. by fungi: a review. Phytochem Rev. https://doi.org/10.1007/s11101-017-9492-6
Chong SP, Osman MF, Bahari N, Nuri EA, Zakaria R, Abdul-Rahim K (2015) Agarwood inducement technology: a method for producing oil grade agarwood in cultivated Aquilaria malaccensis Lamk. J Agrobiotechnol 6:1–16
Chowdhury M, Hussain MD, Chung SO, Kabir E, Rahman A (2016) Agarwood manufacturing: a multidisciplinary opportunity for economy of Bangladesh—a review. CIGR J 18:171–178
CITES Secretariat (2004) Convention on international trade in endangered species of Wild Fauna and Flora. CoP13 Prop. 49. Consideration of proposals for amendment of Appendices I and II-Aquilaria spp. and Gyrinops spp. Thirteenth meeting of the conference of the parties, 2–14 Oct 2004, Bangkok. International Environment House, Geneva, pp 1–9
Cui J, Wang C, Guo S, Yang L, Xiao P, Wang M (2013) Evaluation of fungus-induced agilawood from Aquilaria sinensis in China. Symbiosis 60:37–44
Dinh XB (2015) Vietnamese farmers uses ants to create rare aloeswood. J Glob Issues Solut. http://www.bwwsociety.org/journal/archive/applied_biology.htm. Accessed 26 Dec 2016
Faizal A, Esyanti RR, Aulianisa EN, Santoso E, Turjaman M (2017) Formation of agarwood from Aquilaria malaccensis in response to inoculation of local strains of Fusarium solani. Trees 31:189–197
Fu J, Xu Y (2016) A natural method of bionic colorectal fragrant incense. CN104082058B. State Intellectual Property office of the P.R.C., Beijing, pp 1–8
Gaharu Anugerah Sarawak (2013) GAS de_Denai agarwood inducer. http://gaharu-anugerah-sarawak.blogspot.my/p/researh-and-development.html. Accessed 13 Jan 2017
Gaharu Sarawak (2015) Inokulan SGB. http://gaharusarawak.blogspot.my/. Accessed 30 Nov 2016
Gibson IAS (1977) The role of fungi in the origin of oleoresin deposits (agaru) in the wood of Aquilaria agallocha Roxb. Bano Biggyan Patrika 6:16–26
Gunawan H (2012) Gaharu on. http://hergunfish.blogspot.my/2012/07/inokulan-gaharu-on-hergun-pengembang.html. Accessed 13 Jan 2017
Hashim JRN, Mustapa MZ, Othman K (2016) Agarwood: policy and regulations in Malaysia. Paper presented at the 2nd international scientific symposium on agarwood, 10–12 Oct 2016, Putrajaya, Malaysia. Universiti Putra Malaysia, Serdang, p 12
Hawksworth DL, Gibson IAS (1976) Phialophora parasitica. C.M.I. Descriptions of pathogenic fungi and bacteria. Commonwealth Mycological Institute, Kew, p 504
Huang Z (2013) Method for producing high-quality Aquilaria sinensis material through Aspergillus niger conversion. CN103215311A. State Intellectual Property office of the P.R.C., Beijing, pp 1–12
Huang J, Liao Y, Chen H, Zhang Z (2017) Chemical solution is an efficient method to induce the formation of 2-(2-phenylethyl) chromone derivatives in Aquilaria sinensis. Phytochem Lett 19:64–70
Iskandar D, Suhendra A (2012) Uji inokulasi Fusarium sp. untuk produksi gaharu pada budidaya A. beccariana. Jurnal Sains dan Teknologi Indonesia 14:182–188
Ismail N, Nor Azah MA, Jamil M, Rahiman MH, Tajuddin SN, Taib MN (2013) Analysis of high quality agarwood oil chemical compounds by means of SPME/GC-MS and Z-score technique. Malays J Anal Sci 17:403–413
Ismail N, Ali NAM, Jamil M, Rahiman MH, Tajuddin SN, Taib MN (2014) A review study of agarwood oils and its quality analysis. Jurnal Teknologi (Sci Eng) 68:37–42
ITTO-CITES (2015) Report of the Asian regional workshop on management of wild and planted agarwood taxa, 19–23 Jan 2015. Guwahati, Assam, India. International Timber Trade Organization (ITTO), Yokohama, pp 1–17
IUCN (2017) The IUCN red list of threatened species. Version 2017-1. http://www.iucnredlist.org. Accessed 12 Dec 2016
Jailani AK (2015). Inokulan Gaharu Bio Inducer. http://gaharu84.blogspot.my/2015/11/harga-gubal-gaharu-budidaya-dan.html. Accessed 20 Dec 2016
Jalaluddin M (1977) A useful pathological condition of wood. Econ Bot 31:222–224
Jamil M, Ali NAM, Saidin SH, Lias S, Zainal MH, Aziz A, Patah MFZ (2014) Volatile components, extractive content and E-Nose study on two natural B grade agarwood samples. In: Jamal JA, Ali MS, Jalil J, Husain K, Azmi N, Jasamai M, Kumulosasi E (eds) Proceeding of the 30th annual seminar of the Malaysian Natural Products Society, 18–19 Mar 2014, Bangi, Selangor. Universiti Kebangsaan Malaysia, Bangi, pp 93–96
Jayaraman S, Mohamed R (2015) Crude extract of Trichoderma elicits agarwood substances in cell suspension culture of the tropical tree, Aquilaria malaccensis Lam. Turk J Agric For 39:163–173
Jong PL, Tsan P, Mohamed R (2014) Gas chromatography-mass spectrometry analysis of agarwood extracts from mature and juvenile Aquilaria malaccensis. Int J Agric Biol 16:644–648
Lan J, Li H (2013) Method for producing agilawood on Aquilaria plant by eccentric perfusion method. CN102668908A. State Intellectual Property office of the P.R.C., Beijing, pp 1–10
Lee SY, Mohamed R (2016) The origin and domestication of Aquilaria, an important agarwood-producing genus. In: Mohamed R (ed) Agarwood: science behind the fragrance. Springer, Singapore, pp 1–20
Li W, Cai C, Dong W, Guo Z, Wang H, Mei W, Dai H (2014) 2-(2-Phenylethyl) chromone derivatives from Chinese agarwood induced by artificial holing. Fitoterapia 98:117–123
Lin F, Mei W, Wu J, Dai H (2010) GC-MS analysis of volatile constituents from Chinese eaglewood produced by artificial methods. Zhong Yao Cai 33:222–225
Lisdayani L, Anna N, Siregar EBM (2015) Isolation and identifying of fungi from the stem of agarwood (Aquilaria malaccensis Lamk.) was had been inoculation. Peronema For Sci J 4:1–5
Liu Y, Chen H, Yang Y, Zhang Z, Wei J, Meng H, Gao Z (2013) Whole-tree agarwood-inducing technique: an efficient novel technique for producing high-quality agarwood in cultivated Aquilaria sinensis trees. Molecules 18:3086–3106
Liu Y, Wei J, Gao Z, Zhang Z, Lyu J (2017) A review of quality assessment and grading for agarwood. Chin Herb Med 9:22–30
Ma H, Liang K, Zhou Z, Huang G, Lin M (2012) Method for rapid formation of agilawood of agallochum through induction by using fungus fermentation liquid. CN102550311A. State Intellectual Property office of the P.R.C., Beijing, pp 1–6
Marapurna A (2016) Bio Serum Gaharu Lampung. http://bioserumgaharulampung.blogspot.my/. Accessed 26 Dec 2016
Martin D, Tholl D, Gershenzon J, Bohlmann J (2002) Methyl jasmonate induces traumatic resin ducts, terpenoid resin biosynthesis, and terpenoid accumulation in developing xylem of Norway spruce stems. Plant Physiol 129:1003–1018
Mei W, Dai H, Wang H, Cai Z (2014) Agilawood production method. CN103229677A. State Intellectual Property office of the P.R.C., Beijing, pp 1–8
Mohamed R, Lee SY (2016) Keeping up appearances: agarwood grades and quality. In: Mohamed R (ed) Agarwood: science behind the fragrance. Springer, Singapore, pp 149–167
Mohamed R, Jong PL, Zali MS (2010) Fungal diversity in wounded stems of Aquilaria malaccensis. Fungal Divers 43:67–74
Mohamed R, Wong MT, Halis R (2013) Microscopic observation of ‘gaharu’ wood from Aquilaria malaccensis. Pertanika J Sci Technol 36:43–50
Mohamed R, Jong PL, Kamziah AK (2014) Fungal inoculation induces agarwood in young Aquilaria malaccensis trees in the nursery. J For Res 25:201–204
Monggoot S, Popluechai S, Gentekaki E, Pripdeevech P (2017) Fungal endophytes: an alternative source for production of volatile compounds from agarwood oil of Aquilaria subintegra. Microb Ecol. https://doi.org/10.1007/s00248-016-0908-4
Naef R (2011) The volatile and semi-volatile constituents of agarwood, the infected heartwood of Aquilaria species: a review. Flavour Frag J 26:73–87
Ng LT, Chang YS, Azizol AK (1997) A review on agar (gaharu) producing Aquilaria species. J Trop For Prod 2:272–285
Nobuchi T, Sahri MH (2008) The formation of wood in tropical forest trees: a challenge from the perspective of functional wood anatomy. Penerbit Universiti Putra Malaysia, Serdang
Okudera Y, Ito M (2009) Production of agarwood fragrant constituents in Aquilaria calli and cell suspension cultures. Plant Biotechnol 26:307–315
Phillips MA, Walter MH, Ralph SG, Dabrowska P, Luck K, Uros EM (2007) Functional identification and differential expression of 1-deoxy-D-xylulose 5-phosphate synthase in induced terpenoid resin formation of Norway spruce (Picea abies). Plant Mol Biol 65:243–257
Pojanagaroon SC, Kaewrak C (2005) Mechanical methods to stimulate aloes wood formation in Aquilaria crassna Pierre ex H. Lec. (kritsana) trees. Acta Hortic 676:161–166
Rahman MA, Basak AC (1980) Agar production in agar trees by artificial inoculation and wounding. Bano Biggyan Patrika 9:87–93
Rahman MA, Khisa SK (1984) Agar production in agar tree by artificial inoculation and wounding, part-II, further evidences in favor of agar formation. Bono Biggyan Patrika 9:57–63
Rasool S, Mohamed R (2016) Understanding agarwood formation and its challenges. In: Mohamed R (ed) Agarwood: science behind the fragrance. Springer, Singapore, pp 39–56
Rusli J (2011) Inokulan Gaharu Biotech (IGB) and Nano Tech. http://gaharucandanku.blogspot.my/search/label/Inokulan%20Gaharu%20Biotech. Accessed 20 Dec 2016
Sadgopal VB (1960) Exploratory studies in the development of essential oils and their constituents in aromatic plants. Part I. Oil of agarwood. Soap Perfum Cosmet (Lond) 33:41–46
Santoso E (1996) Pembentukan gaharu dengan cara inokulasi. In: Turjaman M (ed) Makalah diskusi hasil penelitian dalam menunjang pemanfaatan hutan yang lestari, 11–12 Mar 1996, Bogor. Pusat Litbang Hutan dan Konservasi Alam, Bogor, pp 1–3
Santoso E, Irianto RSB, Turjaman M, Sitepu IR, Santoso S, Najmulah, Yani A, Aryanto (2011) Gaharu-producing tree induction technology. In: Turjaman M (ed) Proceedings of the development of gaharu production technology, 1 July 2011. Research and Development Centre for Forest Conservation and Rehabilitation, Bogor, pp 31–46
Sripetkla W (2011) Witsawa’s Agarwood Inducer. http://agarwoodinducement.blogspot.my/. Accessed 08 Jan 2017
Tabata Y, Widjaja E, Mulyaningsih T, Parman I, Wiriadinata H, Mandang YI, Itoh T (2003) Structural survey and artificial induction of aloeswood. Wood Res : Bull Wood Res Inst Kyoto Univ 90:11–12
Tamuli P, Boruah P, Nath SC, Samanta R (2000) Fungi from diseased agarwood tree (Aquilaria agallocha Roxb.): two new records. Adv For Res India 22:182–187
Tamuli P, Boruah P, Nath SC, Leclercq P (2005) Essential oil of eaglewood tree: a product of pathogenesis. J Essent Oil Res 17:601–604
Tanali Esteem (2013) Gaharu Resin Essence. http://www.tanali.my/Gaharu-Essence.html. Accessed 05 Jan 2017
Tang W (2016) Method for generating fragrance of agilawood through natural fungi. CN105830765A. State Intellectual Property office of the P.R.C., Beijing, pp 1–6
Tang J, Liu Y (2016) A natural incense inducer and methods of producing agarwood. CN103858689A. State Intellectual Property office of the P.R.C., Beijing, pp 1–5
Telupid Gaharu Enterprise (2009) Vaksin NVI, PLT (Gred A, B, and C), and Vaksin Kuning. http://telupidgaharu.blogspot.my/. Accessed 12 Dec 16
Thanh L, Do T, Son NH, Sato T, Kozan O (2015) Impacts of biological, chemical and mechanical treatments on sesquiterpene content in stems of planted Aquilaria crassna trees. Agrofor Syst 6:973–981
The Plant List (2013) Version 1.1. Published on the Internet. http://www.theplantlist.org/. Accessed 12 Dec 2016
Tian J, Gao X, Zhang W, Wang L, Qu L (2013) Molecular identification of endophytic fungi from Aquilaria sinensis and artificial agarwood induced by pinholes-infusion technique. Afr J Biotechnol 12:3115–3131
Triadiati T, Carolina DA, Miftahudin (2016) Induksi pembentukan gaharu menggunakan berbagai media tanam dan cendawan Acremonium sp. dan Fusarium sp. pada Aquilaria crassna. Jurnal Sumberdaya Hayati 2:1–6
Turjaman M, Hidayat A (2017) Agarwood-planted tree inventory in Indonesia. IOP Conf Ser Earth Environ Sci 54:012062
UNEP-WCMC (Comps) (2015) The checklist of CITES species website. CITES Secretariat, Geneva, Switzerland. Compiled by UNEP-WCMC, Cambridge, UK. http://checklist.cites.org. Accessed 17 Jan 2017
Verma VPS (1977) Trials of herbicides for inducing formation of agarwood in Aquilaria agollocha Roxb. Indian Perfum 21:151–153
Wei J, Yang Y, Zhang Z, Meng M, Feng J, Gan B (2010) Liquid infusion method for producing linaloe on Aquilaria sinensis tree. CN101755629. State Intellectual Property office of the P.R.C., Beijing, pp 1–5
Wei J, Chen X, Yang Y, Liu Y, Zhang Z, Su Z (2014) Fungus for promoting Aquilaria plants to generate agilawood and application of fungus. CN103651151A. State Intellectual Property office of the P.R.C., Beijing, pp 1–8
Wei J, Chen X, Yang Y, Liu Y, Meng H, Zhang Z, Gan B (2016) Microorganism liquid infusion method-based agilawood induction method and application. CN105340601A. State Intellectual Property office of the P.R.C., Beijing, pp 1–12
Wijitphan P (2009) Method to stimulate resin formation by wounding on the Aquilaria’s trunk. Patent and Trademark Office, Washington, DC, pp 1–11. US Patent 7485309
Wyn LT, Anak NA (2010) Wood for the trees: a review of the agarwood (gaharu) trade in. TRAFFIC Southeast Asia, Selangor
Yan H, Lin C (2016) Method for resin secretion process of agilawood. CN105409715A. State Intellectual Property office of the P.R.C., Beijing, pp 1–8
Yang M, Fu H, Liang Y, Huang H, Zhao B, Xie C, Chen N (2014) Modified transfusion devices, inducer, and procedure for agarwood-inducing by infusion technique. J Chem Pharm Res 6:2566–2571
Yazid A, Al-Fattah SS (2017) PIT2008. http://wafiqahfarmnursery.blogspot.my/. Accessed 12 Dec 2016
Yin Y, Jiao L, Dong M, Jiang X, Zhang S (2016) Wood resources, identification, and utilization of agarwood in China. In: Mohamed R (ed) agarwood. Springer, Singapore, pp 21–38
Zeneli G, Krokene P, Christiansen E, Krekling T, Gershenzon J (2006) Methyl jasmonate treatment of mature Norway spruce (Picea abies) trees increases the accumulation of terpenoid resin components and protects against infection by Ceratocystis polonica, a bark beetle-associated fungus. Tree Physiol 26:977–988
Zhang X, Liu Y, Wei J, Yang Y, Zhang Z, Huang J, Liu Y (2012) Production of high-quality agarwood in Aquilaria sinensis trees via whole-tree agarwood-induction technology. Chin Chem Lett 23:727–730
Zhang Z, Han X, Wei J, Xue J, Yang Y, Liang L, Gao Z (2014) Compositions and antifungal activities of essential oils from agarwood of Aquilaria sinensis (Lour.) Gilg induced by Lasiodiplodia theobromae (Pat.) Griffon. and Maubl. J Braz Chem Soc 25:20–26
Author information
Authors and Affiliations
Corresponding author
Additional information
Project funding: This study was supported by Universiti Putra Malaysia under the Putra Grant scheme (GP-IPS/2017/9554600).
The online version is available at http://www.springerlink.com
Corresponding editor: Zhu Hong.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Azren, P.D., Lee, S.Y., Emang, D. et al. History and perspectives of induction technology for agarwood production from cultivated Aquilaria in Asia: a review. J. For. Res. 30, 1–11 (2019). https://doi.org/10.1007/s11676-018-0627-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11676-018-0627-4