Abstract
Nutrient resorption before abscission is an important nutrient conservation mechanism regulated by climatic conditions and soil nutrients. However, our current understanding of leaf nutrient resorption is primarily derived from site-specific studies or from the use of green-leaf nutrient concentrations to represent those in soils. It remains unknown how nutrient resorption responds to natural soil-nutrient concentrations at a global scale. The effects of plant functional groups, climatic conditions, and soil nutrients and their interactions on leaf nutrient resorption are also unknown. In this study, we established a global database derived from 85 published papers, including 547 reports of nitrogen and phosphorus resorption efficiency (NRE and PRE), climatic factors (LAT, latitude; MAT, mean annual temperature; MAP, mean annual precipitation) and soil-nutrient data (STN, soil total nitrogen; STP, soil total phosphorus) across 111 research sites. The results demonstrated that mean NRE and PRE were 48.4 and 53.3%, respectively. NRE of trees was lower than those of shrubs. NRE and PRE of coniferous species were both higher than those of broad-leaved species. Evergreen species had higher PRE than did deciduous species. NRE was negatively related to STN, but PRE and STP were not related. Both NRE and PRE decreased with increasing MAT and MAP but increased with increasing LAT. Plant functional groups, climate and soil nutrients jointly explained 22 and 32% of the variations in NRE and PRE, respectively. It is important to note that climate (especially MAT) explained 12 and 29% of the variations in NRE and PRE, respectively, implying that continuing global warming will exert an increasingly profound influence on plant nutrient cycles.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Global warming and associated alteration of precipitation regimes will probably influence soil nutrient availability and plant nutrient economies of terrestrial ecosystems (Reich and Oleksyn 2004; Ordoñez et al. 2009; Schmidt et al. 2011). Soil nutrient availability is a primary determinant of the species composition of plant communities, and vice versa, as plants have species-specific effects on soil nutrient availability. These patterns of plant–soil interaction are based on trade-offs between nutrient use and plant growth (Wright et al. 2004; Ordoñez et al. 2009; Freschet et al. 2012). Climate and soils are the most important factors regulating leaf traits (Ordoñez et al. 2009; Chen et al. 2013). In a changing environment, the plant–soil interactions might be changed, leading to a shift in plant nutrient economies (Ordoñez et al. 2009; Zhang et al. 2012). Thus, an essential component of accurate impact forecasting and mitigation planning is an understanding of the adaptation mechanisms of plants to accelerating rates of environmental change.
Nutrient resorption is an efficient nutrient conservation mechanism for plants to adapt to environmental conditions, which refers to the process whereby nutrients are retranslocated from senescing organs to living or storage organs (Aerts 1996; Brant and Chen 2015). The nutrients resorbed during senescence are directly available for further plant growth, which reduces dependence on soil nutrients supply (Aerts 1996). Variations of nutrient resorption could also regulate stand-level biogeochemical cycling via its effect on litter-fall quality, which, in turn, influences litter decomposition and soil nutrient availability (Aerts 1997; Oleksyn et al. 2003).
Plants are frequently exposed to soil nutrients limitations (Grime et al. 1997; Han et al. 2012). Within a given habitat, the leaf traits may coexist to make the balance between growth and environmental conditions (Wright and Westoby 2003). Several leaf traits, e.g., leaf nitrogen (N) and phosphorus (P) concentrations, leaf N:P, and particularly N and P resorption, have been intensively studied (Oleksyn et al. 2003; Ordoñez et al. 2009; Maire et al. 2015). In these site-specific studies, positive (Hidaka and Kitayama 2011), negative (Tully et al. 2013; Hayes et al. 2014) or no (Wright and Westoby 2003; Huang and Boerner 2007) relationship was reported between nutrient resorption and soil nutrients. Few major reviews have attempted to explore the relationships between nutrient resorption and soil nutrients globally (Aerts 1996; Kobe et al. 2005; Vergutz et al. 2012). However, these studies have methodological limitations, namely, they all replaced soil nutrients though green leaf nutrient concentrations but did not use soil nutrients directly (Brant and Chen 2015). Therefore, Brant and Chen (2015) emphasized that it is imperative to explore the relationships between nutrient resorption and natural gradients of soil nutrients for woody plants at large scale. Climate factors have profound effects on nutrient resorption (Brant and Chen 2015). Yuan and Chen (2009) reported that NRE decreased but PRE increased with increasing mean annual precipitation (MAP) and temperature (MAT). Vergutz et al. (2012) reported that both NRE and PRE decreased with increasing MAP and MAT. Thus, no universal relationship was identified to date.
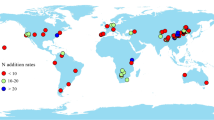
To the best of our knowledge, no study has considered the relationships between nutrient resorption and natural soil nutrients at a global scale. Additionally, the relative effects of plant functional groups, climatic conditions and soil nutrients, and their interactions on nutrient resorption are also unknown. In this study, we developed a global database derived from 85 published papers, which included nutrient resorption efficiency, climate and soil nutrients. Finally, 547 reports from 111 research sites were obtained (Fig. 1). Based on this database, the following research questions were addressed: (1) whether and how the variations in nutrient resorption of woody species respond to soil nutrients and (2) whether and how the relative effects of plant functional groups, climatic conditions, soil nutrients, and their interactions relate to nutrient resorption.
Materials and methods
Data collection
We collected leaf nutrient resorption efficiency, climate and soil nutrient data from the published literature (Table S1). Despite obtaining many relevant papers, only studies which included species-specific nutrient resorption efficiency were selected (NRE, N resorption efficiency; PRE, P resorption efficiency, directly obtained or by allowed calculation), climate (LAT, latitude; MAT, mean annual temperature; MAP, mean annual precipitation) and soil nutrients (STN, soil total N concentration; STP, soil total P concentration) at the same site synchronously. We conducted searches using Web of Science and Google Scholar search engines with the following keywords: resorption, retranslocation, reabsorption, translocation, nutrient resorption, nutrient retranslocation, nutrient reabsorption and nutrient translocation. For additional manipulations (i.e., water-, N- or P- addition) studies, we only selected the unmanipulated controls, and N-fixing species were also included in our dataset (Wright and Westoby 2003; Vergutz et al. 2012). For studies reporting nutrient concentrations throughout the year, we used the maximum value for nutrient concentration of mature leaves to calculate nutrient resorption efficiency. The majority of our data points (nutrient resorption efficiency) were based on leaf mass and leaf area. Overall, the database was derived from 85 studies, encompassing 547 reports of nutrient resorption, climate and soil nutrients for trees and shrubs across 111 research sites which were distributed across most of the world’s biomes (Fig. 1).
According to Aerts (1996) and van Heerwaarden et al. (2003), due to the resorption of soluble carbon compounds during leaf senescence, leaves lost mass in this process. Therefore, we used a mass loss correction factor (MLCF) to eliminate the underestimations of nutrient resorption efficiency, with the calculation as follows:
where NuRE was nutrient resorption efficiency, and Nm and Ns were nutrient concentrations in mature and senesced leaves, respectively. The MLCF values were different for plant growth forms, i.e., 0.745 for conifers, 0.780 for evergreen broadleaved species, and 0.784 for deciduous broad-leaved species (Vergutz et al. 2012; Yuan and Chen 2015).
We only assessed soil total nitrogen (STN) and soil total phosphorus (STP) concentrations in this study (Ordoñez et al. 2009), measured for the upper 0–10 cm of soil in the majority of cases. Leaf nutrient concentrations and soil nutrients were expressed on a mass basis. To ensure data comparability, the unit of percentages (%) for leaf nutrient concentrations was converted to unit of mg g−1, and the unit of percentages (%), mg kg−1 and μg g−1 for corresponding soil nutrients was converted to unit of mg g−1. When the plant functional groups (tree vs. shrub, broad-leaved vs. conifer, and deciduous vs. evergreen) were not provided in the original papers, it was referred and extracted from other papers or one of several online databases (http://en.wikipedia.org/wiki/Main_Page; http://www.theplantlist.org/1/; http://efloras.org/). Data was obtained from tables, texts, additional supporting information directly, or from figures by the data thief software GetData Graph Digitizer 2.24 (http://getdata-graph-digitizer.com). Resorption was quantified by nutrient resorption efficiency, which is defined as the proportional withdrawal of nutrients between green and senesced leaves (Killingbeck 1996). Climatic variable data, including LAT (°), MAT, (°C) and MAP (mm year−1), was derived from the original papers, but if the information was not available, we obtained it from the WorldClim (http://www.worldclim.org/). If several years of temperature and precipitation data were available, we preferred data in the experimental years. Among the studied sites, a wide range of climate and soil nutrient data was obtained. MAT and MAP ranged from − 12.4 to 31.6 °C, and 56 to 5500 mm year−1, respectively, and STN and STP ranged from 0.031 to 26.45 mg g−1, and 0.06 to 2.90 mg g−1, respectively. Geographic coordinates were obtained from Google Earth for those sites where longitude and latitude data had not been reported in the original papers (Zhang et al. 2012).
Data analysis
Before statistical analysis, all leaf, climate and soil data was tested for normality and homoscedasticity of variances using Kolmogorov–Smirnov’s and Levene’s tests, respectively. Soil variables were log10-transformed before statistical analysis to improve the data normality (Han et al. 2011). Of the climatic variables, only MAP was log10-transformed, while MAT was not transformed, as it showed approximately normal distribution (Ordoñez et al. 2009). We used one-way analysis of variance (ANOVA) when we compared the significant differences in nutrient resorption efficiency between tree versus shrub, broad-leaved versus conifer, and deciduous versus evergreen. Linear regression analysis of nutrient resorption efficiency (NRE and PRE) on LAT was performed to address the geographic pattern of nutrient resorption. Linear regression analyses were also performed to examine the response patterns of NRE and PRE to climatic conditions (MAT and MAP) and soil nutrients (STN and STP). Stepwise multiple regressions were used to identify the most influential variables (MAT, MAP, STN and STP) on NRE and PRE. General linear model (GLM) and partial GLM were conducted, with nutrient resorption efficiency as the dependent variable, and plant functional groups (tree and shrub, broadleaf and conifer and deciduous and evergreen), climate (MAP and MAT) and soil total nutrient (STN for NRE, and STP for PRE) as independent variables, to evaluate the relative effects and the interactions between independent variables on nutrient resorption (Heikkinen et al. 2005; Han et al. 2012). To examine how these three variables influence nutrient resorption, the variation partitioning method led to the identification of seven fractions: independent effects of plant functional group (a), climate (b) and soil total nutrient (c), joint effects of functional group and climate (ab), functional group and soil total nutrient (ac), climate and soil total nutrient (bc), and joint effects of all three explanatory variables (abc). The statistical analyses were performed with SPSS 13.0 for Windows (SPSS Inc., Chicago, IL, USA) and R 3.2.0 (R Development Core Team 2015).
Results
Variations in nutrient resorption among plant functional groups
Globally, the mean values of NRE and PRE (both corrected for leaf mass loss) in woody plants were 48.4 and 53.3%, respectively (Table 1). Significant differences in NRE and PRE were observed among plant functional groups. Trees had lower resorption efficiency than those of shrubs, but the difference was significant only in NRE (P = 0.012 and P = 0.329 for NRE and PRE, respectively). Both NRE and PRE of broad-leaved species were significantly lower than those of conifer species (P = 0.042 and P < 0.001, respectively). In addition, evergreen species had higher PRE than those of deciduous species (P = 0.014) (Table 1).
Relationships between nutrient resorption and soil nutrients
The NRE was significantly decreased with the increasing soil total N (P = 0.001). However, there was no significant relationship between PRE and soil total P (P = 0.681) (Fig. 2).
Relationships between NRE (a) and PRE (b) with log-soil total N (Log-STN) and log-soil total P (Log-STP), respectively. For each relationship, coefficient of determination (R 2), P value (P) and number of observations (n) are reported. The solid line is shown when statistically significant (P < 0.05), and the dashed line shown indicates no significant trend of nutrient resorption efficiency among soil nutrients (STN and STP)
Latitudinal pattern of nutrient resorption
Significant latitudinal trends were identified for NRE and PRE at a global scale (Fig. 3). Generally, NRE and PRE were both positively and significantly related to LAT (P < 0.001, R 2 = 0.074 and P = 0.003, R2 = 0.023, respectively) for all species pooled together (Fig. 3).
Relationships between nutrient resorption and climate
The NRE and PRE were both negatively and significantly related to log-MAP (P < 0.001 and P = 0.044, respectively). Meanwhile, NRE and PRE also displayed similar relationships with MAT (both P < 0.001) (Fig. 4). MAT had more influence on NRE than MAP or STN (Table 1S). The same pattern was also true for PRE (Table 1S). With the exception of the relationship between NRE and MAT for conifer, NRE and PRE were both negatively related to MAT, MAP, STN and/or STP, regardless of whether all data were pooled together or the plant functional groups were examined individually (Fig. 4, Table 1S).
Effects of plant functional group, climate and soil nutrients on NRE and PRE
GLM and partial GLM analyses showed that plant functional group, climate and total soil nutrients had significant effects on NRE and PRE. These three explanatory variables together explained 22 and 32% of the variations in NRE and PRE, respectively (Fig. 5). Both the independent (b) and total effects of climate (b + ab + bc + abc) accounted for the largest contribution to the variations in NRE and PRE (16 and 22%; 12 and 29%, respectively). The total effect of plant functional group (a + ab + ac + abc) in PRE (9%) was larger than that of NRE (5%), while the total effect of soil nutrients (c + ac + bc + abc) in PRE (1%) was lower than that of NRE (2%). The joint effects of plant functional group and climate (ab) were 1% and 6% for NRE and PRE, respectively (Fig. 5).
Variation partitioning (R 2, %) of plant functional groups (PFG), climate and soil total nutrient in accounting for NRE (a) and PRE (b). Variables a, b and c denote the independent effects of PFG, climate and soil nutrient, respectively. Variables ab, ac, bc and abc denote the joint effects of PFG and climate, PFG and soil total nutrient, climate and soil total nutrient and the combined three explanatory variables, respectively
Discussion
Variations of NRE and PRE among functional groups
Our results showed that the mean value of NRE and PRE (corrected with MLCF) were 48.4 and 53.3%, respectively. These results were comparable to 49.1 and 56.3%, and 51.0 and 56.9% for the NRE and PRE reported by Tang et al. (2013) and Han et al. (2013), respectively. The NRE and PRE showed significant differences among functional groups (Table 1). The NRE of trees was lower than that of shrubs, while there was no significant difference for PRE, which was in agreement with Yuan and Chen (2009). Both NRE and PRE of coniferous species were higher than those of broad-leaved species (Table 1). Similar observations were also found by Tang et al. (2013). Given that shrubs or coniferous species were always found in poor environmental conditions, therefore, the elevated nutrient resorption could help them survive and adapt to the stressed habitats (Aerts and Chapin 2000; Yuan et al. 2005; Lambers et al. 2008; Yan et al. 2016). Consistent with Yuan and Chen (2009), our observation showed that evergreen species had significantly higher PRE than deciduous species (Table 1). The possible explanation may be that, first, higher abundance of evergreen species grow in P-limited low latitudes, thus a higher PRE characteristic for evergreen species could support their nutrient demands and adapt to P-limitation. Second, evergreen species would prolong nutrient retention time in leaf biomass and leaf longevity to increase nutrient use efficiency and lead to low nutrient loss rates (Escudero et al. 1991; Aerts 1996). The above results reflect diverse nutrient use by different plant functional groups.
Patterns of NRE and PRE along soil nutrients
Our data demonstrated that NRE was negatively related to STN, but PRE and STP were not significantly related (Fig. 2). It has been widely reported that plants grown in nutrient-poor habitats would be characterized by high nutrient resorption efficiency (Vergutz et al. 2012; Yuan and Chen 2015). This expectation was also supported by our observations for the relationship between NRE and STN. Our results were partially supported by the previous findings, which demonstrated that nutrient resorption was strongly related to soil fertility (Kobe et al. 2005; Vergutz et al. 2012; Zhang et al. 2015). The finding contradicted the reports by Aerts (1996), which showed that the relationship between nutrient resorption and soil fertility was absent or very weak, or by Tang et al. (2013) which found that NRE increased with increasing STN, but PRE decreased with increasing STP. The decreased NRE with increasing STN may be explained by the theoretical model, which considered that plants in fertile habitats would require less energy to acquire nutrients from soil than by resorption from senescent leaves (Wright and Westoby 2003). Plants grown in high soil nutrient (e.g., STN) habitats would be characterized by low nutrient resorption efficiency, return higher-quality litter to soils, which in turn would produce a positive plant–soil feedback. By contrast, plants grown in low soil nutrient habitat would have high nutrient resorption efficiency, and the lower-quality litter reinforced the infertile soils (Aerts 1997; Ordoñez et al. 2009; Lü et al. 2012; Yan et al. 2016). Given that leaf nutrients, therefore, can either be recycled within the plant or returned to the soil via litter, plants may strive to make a trade-off between nutrient requirements and soil conditions.
Patterns of NRE and PRE along climatic gradients
In our present study, whether all data were pooled together or plant functional groups were examined individually, both NRE and PRE decreased with increasing MAT and MAP, but increased with increasing LAT (Figs. 3, 4, Table 1S). Our results were in agreement with Vergutz et al. (2012) and Sun et al. (2016), but in disagreement with some of the conclusions of Yuan and Chen (2009) and Tang et al. (2013), which found that NRE was negatively related to MAT and MAP, but positively related to LAT, whereas the opposite relationships were true for PRE. The inconsistency in PRE may be due to the methodological differences among different studies (Yuan and Chen 2009; Vergutz et al. 2012; Brant and Chen 2015). Alternatively, P-related leaf traits are usually more responsive to environmental conditions relative to N-related leaf traits (Han et al. 2011; Chen et al. 2013; Tang et al. 2013).
Our results suggested that plant species in higher-latitude (lower temperature and precipitation) regions were more dependent on internal nutrient cycling processes (Oleksyn et al. 2003). This finding is likely to reflect an adaptive trait with higher nutrient conservation for plants from colder habitats (Oleksyn et al. 2003). Two main reasons can be ascribed to this phenomenon. First, lower temperature would limit the movement of soil nutrient solution, as well as nutrients uptake via root (Chapin and Oechel 1983; Körner 1999; Reich and Oleksyn 2004). Thus plants may prefer nutrient resorption which costs relatively less energy than uptake from soil (Wright and Westoby 2003; Viers et al. 2013). Conversely, plants grown in higher temperature regions would have higher potential evapotranspiration, and thus plants could enhance the transpiration rate to cool leaves (Cramer et al. 2009; Pan et al. 2010; Maire et al. 2015). Accompanied by this adaptive feature, lots of mass-flow nutrients would be delivered from soil to plant tissues (Cramer et al. 2009; Pan et al. 2010), which may subsequently weaken the process of nutrient resorption. Second, decomposition and nutrient mineralization are slow at high latitudes, which in turn, would reduce soil N and P availability (Robinson 2002; Oleksyn et al. 2003; Reich and Oleksyn 2004). Consequently, plants would improve their capacities of nutrient resorption to meet nutrient requirements. Similar latitudinal patterns were observed by Oleksyn et al. (2003), which reported that the NRE and PRE of Scots pine populations from colder habitats were greater than those from warmer habitats throughout Europe.
Effects of plant functional group, climate and soil nutrients on NRE and PRE
GLM and partial GLM showed that NRE was primarily driven by plant functional group, climate, soil nutrients, and their interactions, while PRE was primarily driven by plant functional group, climate and the joint effects. Climate explained the largest part of the variations in both NRE and PRE (12 and 29%, respectively), which implied that continuing global warming will exert an increasingly profound influence on plant nutrient cycles, particularly for PRE. Evidence from Chen et al. (2013) also reported that leaf P was more flexible than leaf N in response to changing climate. The relatively small proportion explained by soil nutrients may be attributed to the fact that STN and STP are just potential soil nutrient supplies and were rarely utilized by plants (Aerts and Chapin 2000). These results implied that the applications of STN and STP were inadequate to characterize the response of nutrient resorption to soil nutrient conditions, and the response may be driven by collective soil variables, e.g., soil nutrient availability, soil moisture and soil texture (Ordoñez et al. 2009; Lü et al. 2012; Reed et al. 2012). Therefore, further studies should also take soil available N and P into consideration, because these available nutrients can be directly utilized by plants. Overall, the above biogeographic patterns of nutrient resorption in woody species are generally considered to be the acclimation or adaptation responses to their hydrothermal environmental and soil nutrient conditions.
Conclusions
Our present study is, to the best of our knowledge, the first attempt to quantify the relationships between nutrient resorption and natural soil nutrients directly, and to assess the integrated effects of plant functional groups, climatic factors, soil nutrients, and their interactions on nutrient resorption of woody species at a global scale. Thus, we provided the opportunity to explore the widely debated issue of whether and how soil nutrients affect nutrient resorption. Our results demonstrate that NRE decreased with increasing STN, while by contrast, PRE was not related to STP. There existed biogeographic patterns in nutrient resorption of woody species at a global scale, which likely is a result of the collective forcing of plant functional group, climate and soil nutrient gradients. In particular, climatic factors (especially MAT) had more contributions to the variations in both NRE and PRE (12 and 29%, respectively) than those of plants functional groups and soil nutrients. These results implied that the ongoing global warming will modulate the patterns of nutrient resorption, and subsequently affect the nutrient returning and cycling.
References
Aerts R (1996) Nutrient resorption from senescing leaves of perennials: are there general patterns? J Ecol 84:597–608
Aerts R (1997) Nitrogen partitioning between resorption and decomposition pathways: a trade-off between nitrogen use efficiency and litter decomposability? Oikos 80:603–606
Aerts R, Chapin FS (2000) The mineral nutrition of wild plants revisited: a re-evaluation of processes and patterns. Adv Ecol Res 30:1–67
Brant AN, Chen HYH (2015) Patterns and mechanisms of nutrient resorption in plants. Crit Rev Plant Sci 34:471–486
Chapin FS, Oechel WC (1983) Photosynthesis, respiration, and phosphate absorption by Carex aquatilis ecotypes along latitudinal and local environmental gradients. Ecology 64:743–751
Chen YH, Han WX, Tang LY, Tang ZL, Fang JY (2013) Leaf nitrogen and phosphorus concentrations of woody plants differ in responses to climate, soil and plant growth form. Ecography 36:178–184
Cramer MD, Hawkins HJ, Verboom GA (2009) The importance of nutritional regulation of plant water flux. Oecologia 161:15–24
Escudero A, del Arco JM, Sanz IC, Ayala J (1991) Effects of leaf longevity and retranslocation efficiency on the retention time of nutrients in the leaf biomass of different woody species. Oecologia 90:80–87
Freschet GT, Aerts R, Cornelissen JHC (2012) A plant economics spectrum of litter decomposability. Funct Ecol 26:56–65
Grime JP, Thompson K, Hunt R, Hodgson JG, Cornelissen JHC, Rorison IH, Hendry GAF, Ashenden TW, Askew AP, Band SR, Booth RE, Bossard CC, Campbell BD, Cooper JEL, Davison AW, Gupta PL, Hall W, Hand DW, Hannah MA, Hillier SH, Hodkinson DJ, Jalili A, Liu Z, Mackey JML, Matthews N, Mowforth MA, Neal AM, Reader RJ, Reiling K, Ross-Fraser W, Spencer RE, Sutton F, Tasker DE, Thorpe PC, Whitehouse J (1997) Integrated screening validates primary axes of specialisation in plants. Oikos 79:259–281
Han WX, Fang JY, Reich PB, Woodward FI, Wang ZH (2011) Biogeography and variability of eleven mineral elements in plant leaves across gradients of climate, soil and plant functional type in China. Ecol Lett 14:788–796
Han WX, Chen YH, Zhao FJ, Tang LY, Jiang RF, Zhang FS (2012) Floral, climatic and soil pH controls on leaf ash content in China’s terrestrial plants. Glob Ecol Biogeogr 21:376–382
Han WX, Tang LY, Chen YH, Fang JY (2013) Relationship between the relative limitation and resorption efficiency of nitrogen vs phosphorus in woody plants. PLoS ONE 8:e83366. https://doi.org/10.1371/journal.pone.0083366
Hayes P, Turner BL, Lambers H, Laliberté E (2014) Foliar nutrient concentrations and resorption efficiency in plants of contrasting nutrient-acquisition strategies along a 2-million-year dune chronosequence. J Ecol 102:396–410
Heikkinen R, Luoto M, Kuussaari M, Pöyry J (2005) New insights into butterfly-environment relationships using partitioning methods. Proc Biol Sci 272:2203–2210
Hidaka A, Kitayama K (2011) Allocation of foliar phosphorus fractions and leaf traits of tropical tree species in response to decreased soil phosphorus availability on Mount Kinabalu, Borneo. J Ecol 99:849–857
Huang JJ, Boerner REJ (2007) Effects of fire alone or combined with thinning on tissue nutrient concentrations and nutrient resorption in Desmodium nudiflorum. Oecologia 153:233–243
Killingbeck KT (1996) Nutrients in senesced leaves keys to the search for potential resorption and resorption proficiency. Ecology 77:1716–1727
Kobe RK, Lepczyk CA, Iyer M (2005) Resorption efficiency decreases with increasing green leaf nutrients in a global data set. Ecology 86:2780–2792
Körner C (1999) Alpine plant life: functional plant ecology of high mountain ecosystems. Springer, Berlin, pp 1–338
Lambers H, Chapin FS, Pons TL (2008) Plant physiological ecology, 2nd edn. Springer, New York, pp 212–546
Lü XT, Freschet GT, Flynn DFB, Han XG (2012) Plasticity in leaf and stem nutrient resorption proficiency potentially reinforces plant–soil feedbacks and microscale heterogeneity in a semi-arid grassland. J Ecol 100:144–150
Maire V, Wright IJ, Prentice C, Batjes NH, Bhaskar R, van Bodegom PM, Cornwell WK, Ellsworth D, Niinemets Ü, Ordonez A, Reich PB, Santiago LS (2015) Global effects of soil and climate on leaf photosynthetic traits and rates. Glob Ecol Biogeogr 24:706–717
Oleksyn J, Reich PB, Zytkowiak R, Karolewski P, Tjoelker MG (2003) Nutrient conservation increases with latitude of origin in European Pinus sylvestris populations. Oecologia 136:220–235
Ordoñez JC, van Bodegom PM, Witte JPM, Wright IJ, Reich PB, Aerts R (2009) A global study of relationships between leaf traits, climate and soil measures of nutrient fertility. Glob Ecol Biogeogr 18:137–149
Pan RC, Wang XJ, Li NH (2010) Plant physiology. Higher Education Press, Beijing, pp 14–24
R Development Core Team (2015) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. http://www.r-project.org/
Reed SC, Townsend AR, Davidson EA, Cleveland CC (2012) Stoichiometric patterns in foliar nutrient resorption across multiple scales. New Phytol 196:173–180
Reich PB, Oleksyn J (2004) Global patterns of plant leaf N and P in relation to temperature and latitude. Proc Natl Acad Sci USA 101:11001–11006
Robinson CH (2002) Controls on decomposition and soil nitrogen availability at high latitudes. Plant Soil 242:65–81
Schmidt MWI, Torn MS, Abiven S, Dittmar T, Guggenberger G, Janssens IA, Kleber M, Kögel-Knabner I, Lehmann J, Manning DAC, Nannipieri P, Rasse DP, Weiner S, Trumbore SE (2011) Persistence of soil organic matter as an ecosystem property. Nature 478:48–56
Sun X, Kang H, Chen HYH, Björn B, Samuel BF, Liu C (2016) Biogeographic patterns of nutrient resorption from Quercus variabilis Blume leaves across China. Plant Biol 18:505–513
Tang LY, Han WX, Chen YH, Fang JY (2013) Resorption proficiency and efficiency of leaf nutrients in woody plants in eastern China. J Plant Ecol 6:408–417
Tully KL, Wood TE, Schwantes AM, Lawrence D (2013) Soil nutrient availability and reproductive effort drive patterns in nutrient resorption in Pentaclethra macroloba. Ecology 94:930–940
van Heerwaarden LM, Toet S, Aerts R (2003) Current measures of nutrient resorption efficiency lead to a substantial underestimation of real resorption efficiency: facts and solutions. Oikos 101:664–669
Vergutz L, Manzoni S, Porporato A, Novais RF, Jackson RB (2012) Global resorption efficiencies and concentrations of carbon and nutrients in leaves of terrestrial plants. Ecol Monogr 82:205–220
Viers J, Prokushkin AS, Pokrovsky OS, Auda Y, Kirdyanov AV, Beaulieu E, Zouiten C, Oliva P, Dupré B (2013) Seasonal and spatial variability of elemental concentrations in boreal forest larch foliage of Central Siberia on continuous permafrost. Biogeochemistry 113:435–449
Wright IJ, Westoby M (2003) Nutrient concentration, resorption and lifespan: leaf traits of Australian sclerophyll species. Funct Ecol 17:10–19
Wright IJ, Reich PB, Westoby M, Ackerly DD, Baruch Z, Bongers F, Cavender-Bares J, Chapin T, Cornelissen JHC, Diemer M, Flexas J, Garnier E, Groom PK, Gulias J, Hikosaka K, Lamont BB, Lee T, Lee W, Lusk C, Midgley JJ, Navas ML, Niinemets Ü, Oleksyn J, Osada N, Poorter H, Poot P, Prior L, Pyankov VI, Roumet C, Thomas SC, Tjoelker MG, Veneklaas EJ, Villar R (2004) The worldwide leaf economics spectrum. Nature 428:821–827
Yan T, Lü XT, Yang K, Zhu JJ (2016) Leaf nutrient dynamics and nutrient resorption: a comparison between larch plantations and adjacent secondary forests in Northeast China. J Plant Ecol 9:165–173
Yuan ZY, Chen YH (2009) Global-scale patterns of nutrient resorption associated with latitude, temperature and precipitation. Global Ecol Biogeogr 18:11–18
Yuan ZY, Chen YH (2015) Negative effects of fertilization on plant nutrient resorption. Ecology 96:373–380
Yuan ZY, Li LH, Han XG, Huang JH, Wan SQ (2005) Foliar nitrogen dynamics and nitrogen resorption of a sandy shrub Salix gordejevii in northern China. Plant Soil 278:183–193
Zhang SB, Zhang JL, Slik JWF, Cao KF (2012) Leaf element concentrations of terrestrial plants across China are influenced by taxonomy and the environment. Glob Ecol Biogeogr 21:809–818
Zhang JH, Tang ZY, Luo YK, Chi XL, Chen YH, Fang JY, Shen HH (2015) Resorption efficiency of leaf nutrients in woody plants on Mt. Dongling of Beijing, North China. J Plant Ecol 8:530–538
Acknowledgements
We thank Mr. Xu Kuang and Dr. Zuoqiang Yuan for their help to conduct the variation partitioning in R 3.2.0, and we also thank Mr. Guiduo Shang for his help with the production of Fig. 1 by ArcGIS. We are very grateful to the editor and the anonymous reviewers for their helpful and constructive comments and suggestions that greatly improved this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Project funding: This study was funded by the National Basic Research Program of China (973 Program) (2012CB416906).
The online version is available at http://www.springerlink.com
Corresponding editor: Chai Ruihai.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yan, T., Zhu, J. & Yang, K. Leaf nitrogen and phosphorus resorption of woody species in response to climatic conditions and soil nutrients: a meta-analysis. J. For. Res. 29, 905–913 (2018). https://doi.org/10.1007/s11676-017-0519-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11676-017-0519-z