Abstract
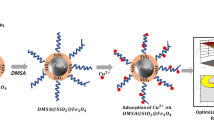
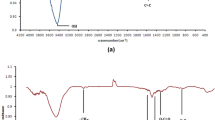
Fe3O4@SiO2@THPP (MSTHPP) nanocomposite was prepared as an adsorbent for the removal of lead ions. The structural characteristics of thisnanocomposite were determined using Fourier transform-infrared (FT-IR) spectroscopy, scanning electron microscopy (SEM), transmission electron microscopy, x-ray diffraction, vibrating sample magnetometry (VSM), and N2 adsorption-desorption analyses. SEM images showed that the magnetic nanoparticles have uniform morphologies with a mean size of 20 nm. The magnetic properties of the synthesized nanocomposite were measured on a VSM with maximum saturation magnetization values of 40 emug-1 and 10 emug-1 for Fe3O4@SiO2 and MSTHPP nanocomposites, respectively. MSTHPP has been efficiently used to remove lead ions from aqueous solutions. After the lead sorption process, the nanocomposite was magnetically separated from the mixture and showed good reusability. The effects of pH, contact time, adsorbent dosage and initial concentration of lead in the removal of lead were investigated. Optimization of the parameters was performed by using Taguchi design method to obtain the maximum removal efficiency. The optimized condition can be achieved when pH, contact time, adsorbent dosage and initial concentration of lead are 5.3 min, 30 min, 50 mg, 20 ppm, respectively. The M, MS, and MSTHPP lead removal efficiency was found to be 18%, 25%, and 97%, respectively.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
M. Golshekan and S. Shariati, Acta chem. Slov 60, 358 (2013).
M. Emadi, E. Shams, and M.K. Amini, J. Chem 2013, 1 (2012).
T. Poursaberi, H. Ghanbarnejad, and V. Akbar, J. Nano. Struct. 2, 417 (2013).
E. Bilgin, M. Yuce, K. Kose, K. Erol, and D.A. Kose, Hitite. J. Sci. Eng. 4, 1 (2017).
S. Bakhshayesh and H. Dehghani, Mater. Res. Bull. 48, 2614 (2013).
E. Blicharska, M. Tatarczak-Michalewska, A. Plazińska, W. Plaziński, A. Kowalska, A. Madejska, M. Szymańska-Chargot, A. Sroka-Bartnicka, and J. Flieger, J. Sep. Sci 41, 3129 (2018).
H.M. Baker, A.M. Massadeh, and H.A. Younes, Environ. Monit. Assess 157, 319 (2009).
M.J. Gonzalez-Munoz, M.A. Rodriguez, S. Luque, and J.R. Alvarez, Desalination 200, 742 (2006).
M.G. Khedr, Water Treat. 2, 342 (2009).
Z.-H. Huang, X. Zheng, W. Lv, M. Wang, Q.-H. Yang, and F. Kang, Langmuir 27, 7558 (2011).
L. Fan, C. Luo, M. Sun, X. Li, and H. Qiu, Colloid Surf. B. Biointerfaces 103, 523 (2013).
X. Liu, Q. Hu, Z. Fang, X. Zhang, and B. Zhang, Langmuir 25, 3 (2008).
M. Khazaei, S. Nasseri, M.R. Ganjali, M. Khoobi, R. Nabizadeh, A.H. Mahvi, S. Nazmara, and E. Gholibegloo, J. Environ. Health Sci. Eng. 14, 1 (2016).
Y. Tan, M. Chen, and Y. Hao, Chem. Eng. J. 191, 104 (2012).
Z. Khayat Sarkar and F. Khayat Sarkar, Int. J. Nanosci. Nanotechnol 9, 109 (2013).
S. Rajput, C.U. Pittman Jr, and D. Mohan, J. Colloid Interface Sci. 468, 334 (2016).
A.Z.M. Badruddoza, Z.B.Z. Shawon, W.J.D. Tay, K. Hidajat, and M.S. Uddin, Carbo. Poly 91, 322 (2013).
X. Xin, Q. Wei, J. Yang, L. Yan, R. Feng, G. Chen, B. Du, and H. Li, Chem. Eng. J. 184, 132 (2012).
X. Peng, F. Xu, W. Zhang, J. Wang, C. Zeng, M. Niu, and E. Chmielewská, Colloids Surf., A: Phys.Chem. Eng.Asp 443, 27 (2014).
S. Recillas, A. Garcia, E. Gonzalez, E. Casals, V. Puntes, A. Sanchez, and X. Font, Desalination 277, 213 (2011).
M. Kumari, C.U. Pittman Jr, and D. Mohan, J. Colloid Interface Sci. 442, 120 (2015).
L. Dong, Z. Zhu, Y. Qiu, and J. Zhao, Chem. Eng. J. 165, 827 (2010).
P. Panneerselvam, N. Morad, and K.A. Tan, J. Hazard. Mater. 186, 160 (2011).
L. Sun, Y. Li, M. Sun, H. Wang, S. Xu, C. Zhang, and Q. Yang, New J. Chem. 35, 2697 (2011).
Y. Deng, D. Qi, C. Deng, X. Zhang, and D. Zhao, J. ACS. 130, 28 (2008).
R. Hao, R. Xing, Z. Xu, Y. Hou, S. Gao, and S. Sun, Adv. Mater. 22, 2729 (2010).
B.J. Kim, J. Bang, C.J. Hawker, J.J. Chiu, D.J. Pine, S.G. Jang, S.-M. Yang, and E.J. Kramer, Langmuir 23, 12693 (2007).
S. Hou, X. Li, H. Wang, M. Wang, Y. Zhang, Y. Chi, and Z. Zhao, RSC Adv. 7, 51993 (2017).
T. Sen, A. Sebastianelli, and I.J. Bruce, J. ACS. 128, 7130 (2006).
M.O. Ojemaye, O.O. Okoh, and A.I. Okoh, J. Nanomater. 2017, 1 (2017).
T. Poursaberi, V. Akbar, and S.M.R. Shoja, Iran. J. Chem. Eng. 34, 41 (2015).
E. Aguilera-Ruiz, U.M. Garcĺa-Pérez, M. de la Garza-Galván, P. Zambrano-Robledo, B. Bermúdez-Reyes, and J. Peral, Appl. Surf. Sci. 328, 361 (2015).
J. Niu, B. Yao, Y. Chen, C. Peng, X. Yu, J. Zhang, and G. Bai, Appl. Surf. Sci. 271, 39 (2013).
W.M. Campbell, A.K. Burrell, D.L. Officer, and K.W. Jolley, Coord. Chem. Rev. 248, 1363 (2004).
E. Gholamrezapor and A. Eslami, J. Mater. Sci: Mater. Elec. 30, 4705 (2019).
V.N. Nair, B. Abraham, J. MacKay, G. Box, R.N. Kacker, T.J. Lorenzen, J.M. Lucas, R.H. Myers, G.G. Vining, and J.A. Nelder, Technometrics 34, 127 (1992).
P.T. Dhorabe, D.H. Lataye, A.R. Tenpe, and R.S. Ingole, SN Appl. Sci. 1, 250 (2019).
F. Googerdchian, A. Moheb, R. Emadi, and M. Asgari, J. Hazard. Mater. 349, 186 (2018).
V.C. Srivastava, I.D. Mall, and I.M. Mishra, Chem. Eng. J. 140, 136 (2008).
X. Zheng, L. Zhang, J. Li, S. Luo, and J.-P. Cheng, Chem. Commun. 47, 12325 (2011).
J.M. Perez, F.J. Simeone, Y. Saeki, L. Josephson, and R. Weissleder, J. ACS. 125, 10192 (2003).
M. Shokouhimehr, Y. Piao, J. Kim, Y. Jang, and T. Hyeon, Angew. Chem. 119, 7169 (2007).
A. Zielińska-Jurek, Z. Bielan, S. Dudziak, I. Wolak, Z. Sobczak, T. Klimczuk, and J. Hupka, Catalysts 7, 360 (2017).
V.D. Rumyantseva, A.S. Gorshkova, and A.F. Mironov, Macroheterocycles 6, 59 (2013).
N. Salamun, H.X. Ni, S. Triwahyono, A.A. Jalil, and A.H. Karim, Malays. J. Fundam. Appl. Sci. 7, 89 (2011).
A. Patterson, Phys. Rev. 56, 978 (1939).
Y. Liu, R. Fu, Y. Sun, X. Zhou, S.A. Baig, and X. Xu, Appl. Surf. Sci. 369, 267 (2016).
H. Ghanbarnejad, T. Poursaberi, and V. Akbar, J. Nano. Struc. 2, 417 (2012).
Acknowledgments
We gratefully acknowledged financial support from the Research Council of the University of Mazandaran.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gholamrezapor, E., Eslami, A. Removal of Lead by Tetra Hydroxyl Phenyl Porphyrin-Linked Magnetic Nanoparticles: Process Optimization by Using Taguchi Design Method. J. Electron. Mater. 49, 743–751 (2020). https://doi.org/10.1007/s11664-019-07767-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11664-019-07767-6