Abstract
Summary
Swimming has little effect on bone mass. Therefore, adolescent swimmers should complement their water training with a short and intense weight-bearing training, aiming to increase their bone acquisition. Forty swimmers performed a six-month whole-body vibration (WBV) training. WBV had no effect on adolescent swimmers’ bone mass or lean mass.
Purpose
The aims of the present study were to evaluate the effects of a whole-body vibration (WBV) intervention on bone mineral density (BMD), bone mineral content (BMC) and lean mass (LM) in adolescent swimmers.
Methods
Forty male and female adolescent swimmers (VIB; mean age 14.2 ± 1.9 years) completed the WBV protocol that consisted of 15 min of training 3 days per week during a 6-month period (ranging from 3.6 to 11.6 g), while 23 swimmers (SWI; mean age 15.0 ± 2.2 years) continued with their regular swimming training alone. VIB were divided into tertiles according to training compliance in order to evaluate if any dose-effect relation existed. BMD, BMC and LM were measured longitudinally by dual energy X-ray at the whole body, lumbar-spine and hip.
Results
No group by time interactions and no differences in change percentage were found for BMD, BMC or LM in any of the measured variables. The mean change percentage of the subtotal body (whole body minus the head) for VIB and SWI, respectively, was 2.3 vs. 2.4% for BMD, 5.7 vs 5.7% for BMC and 7.3 vs. 8.0% for lean mass. Moreover, no indication for dose-response was observed.
Conclusions
The proposed WBV protocol had no effect on BMD, BMC and LM in adolescent swimmers. Other types of training should be used in this population to improve both bone and lean mass.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Physical activity has been suggested as one of the best methods of combating osteoporosis. It can regulate bone maintenance, stimulate bone formation including the accumulation of mineral [1], in addition to strengthening muscles, improving balance [2] and thus reducing the overall risk of falls and consequent fractures [2]. Development of osteoporosis is variably attributed to either excessive rate of bone loss during aging or to inadequate accumulation of peak bone mass prior to attainment of skeletal maturity [3]. Regarding the latter, several studies have found that high-impact physical activity can improve bone mass acquisition during adolescence [4, 5]. Nevertheless, not all physical activities exert a positive effect on bone mass, as sports such as cycling [6] and swimming [7] seem to be ineffective in increasing bone mass.
Focusing on swimming, a systematic review [7] and a more recent meta-analysis [8] showed that swimming seemed to be neutral (having no effect) to bone mineral content (BMC) and density (BMD) during adolescence. This could possibly be interpreted as a positive conclusion, as swimming is not negatively affecting bone mass acquisition. Nevertheless, we should consider that firstly, adolescence is a key period for increasing bone mass and density values (at least 25% of the adult total BMC is attained in just a 2-year period of fast bone mineral accrual during growth [9]), and secondly, swimming participation will entail a significant amount of water training that may exclude other weight-bearing activities [10]; not improving bone acquisition might be a negative condition associated to swimming.
Although swimmers present similar bone mass values [8], they generally present higher lean mass values when compared to sedentary controls [11]. Lean mass has been positively associated with bone mass in several studies [12, 13]. Therefore, specific training aiming to improve lean mass might also entail some minor benefits to bone mass. In swimmers, even if lean mass is improved without an increase in bone mass, a performance benefit would be attained, as lean mass has been positively associated with swimming performance [14].
In order to improve both lean mass and bone mass acquisition, short interventions should be performed before or after swimming training. To this end, jumping interventions have been proved to be very effective [4] and thus could be a feasible option for adolescent swimmers. Although teenagers may find these types of intervention boring and might require incentives to adhere to them. Whole-body vibration (WBV) platforms appear to be devices that might appeal to teenagers and thus improve adherence to the program.
WBV training was originally proposed as a means of increasing BMD in astronauts in space who, like swimmers, operate in a hypogravitational medium. WBV, like other weight-bearing physical activities, requires muscles and bones to work against gravity, and several studies have shown that it is an effective tool regarding bone mass augmentation in both disabled [15] and healthy populations [16, 17]. Few studies have evaluated the effects of WBV training on body composition with inconclusive results as some researchers have found positive effects in untrained participants [18, 19] while others have found no effects [20, 21]. It thus seems as if WBV could be a good tool for adolescent swimmers to improve their lean mass and bone mass.
Therefore, the aim of the present study was to determine the effects of WBV on BMD, BMC and lean mass in a group of swimmers and compare these values to those of other swimmers who perform the same water training without the WBV training.
Material and methods
Experimental design
For this study, participants were requested to visit our laboratory on two occasions (pre- and post-intervention). The first evaluation took place between September and October 2012, while the second evaluation took place between May and June 2013. All the tests and questionnaires were performed by qualified trained researchers from the University of Zaragoza.
Participants
Swimmers were recruited from four different swimming clubs from Aragon (Spain). When the study began, inclusion criteria were as follows: participants had to be between the ages of 11 and 18 years, Caucasian, healthy, non-smokers and with no chronic disease or musculoskeletal disorders (fibromyalgia, gout, osteoarthritis, rheumatoid arthritis, tendinitis), bone fractures or medication. Swimmers had to have a history of swimming and competing in regional tournaments for more than 3 years and training for a minimum of 6 h per week.
Participant classification
All swimmers were randomly allocated vibration training in addition to swimming or simply the continuation of their normal swimming training. Some of the swimmers who continued with their normal swimming training performed an extra weight-bearing sport (running, free weights, etc.) on a weekly basis that could modify or mask intervention results, and these participants were thus excluded. Therefore, for the present study, swimmers who only swam (SWI; n = 23) were compared to swimmers who swam and performed WBV training (VIB; n = 40).
Ethics statement
Written informed consent from parents and verbal assent from the participants were obtained. The study was performed following the ethical guidelines of the Declaration of Helsinki 1961 (revision of Fortaleza 2013). The protocol study was approved by the Ethics Committee of Clinical Research from the Government of Aragón (ref. CP08/2012, CEICA, Spain). The present study is part of a larger randomized controlled trial which is registered in clinicaltrials.gov (identification number NCT02380664).
Evaluation of pubertal stage
Pubertal maturation was determined by self-assessment of secondary sexual characteristics according to the criteria devised by Tanner [22]. This method has been reported to be both valid and reliable in assessing sexual maturity among adolescent athletes [23].
Calcium intake
Calcium intake was estimated as milligrams per day through a validated food frequency questionnaire that is explained in detail elsewhere [24].
Bone mass and body composition
Dual energy X-ray scans were performed with the pediatric version of the QDR-Explorer software (Hologic Corp., Software version 12.4, Bedford, MA, USA) for the whole body (and its sub-regions), lumbar spine and hip (trochanter, femoral neck, total hip). For bone mass, outcomes of interest were BMD and BMC. For body composition, the outcome of interest was lean mass. All the DXA analyses were performed by the same operator. The coefficients of variation of the DXA in our laboratory for total body scans are published elsewhere and were 2.3% for BMC, 1.3% for BMD and 1.9% for lean mass [25].
Whole-body vibration program
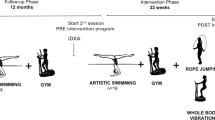
A synchronous vibration platform (Power Plate® Pro5; PowerPlate, Amsterdam, The Netherlands) was used to perform the vibration intervention. A synchronous vibration device was selected over a rotational one as the asynchronous nature of the rotational vibration may make it more difficult to perform certain exercises on the platform because both feet must remain on the unit at all times [26]. In addition, there is not enough data to support the effectiveness of one type of vibration unit over another [26, 27]. Participants allocated to the WBV group performed three WBV training sessions per week, supervised by a researcher who ensured safety and guided participants in the correct performance of all sets. The researcher that supervised the WBV trainings completed a diary to register participants’ assistance. From this diary, final WBV training compliance was calculated by dividing the number of session attendances by the total number of sessions and multiplying that number by 100 (i.e. a participant that attended 60 sessions out of the 81 possible sessions had performed 74% of the training). Each swimming center had a platform where the intervention was carried out. Swimmers performed the exercises in alternating pairs with one swimmer resting while the other used the vibration platform. A graphic description of the exercises performed is represented in Fig. 1. The intervention comprised five exercises which were performed in the following order: (1) half squat (knees bent at 120°) on the platform (Fig. 1a); (2) squat (knees bent at 90°) (Fig. 1b); (3) dynamic flexion and extension from 120 to 90° at a rate of 2 s up and 2 s down; (4) lunge with right foot in front (Fig. 1c); and (5) lunge with left foot in front (Fig. 1d). Swimmers performed the sequence twice, taking approximately 15 min to complete the whole protocol. Participants were asked to attend the sessions with sports shoes to standardize the damping of the vibration caused by footwear.
The intensity of the intervention protocol was gradually increased during the 6-month period, and specific amplitudes and frequencies are detailed in Fig. 1. The duration of 6 months was selected on account of the temporal limitations of the training season (September to June). As measuring the whole sample took 2 months, the first evaluation was carried out between September and October while the second evaluation was performed between May and June. Consequently, the WBV intervention began in November and finished in April (both months included). The present WBV protocol was chosen following positive results found with similar interventions performed in previous studies [28, 29] with an attempt to maintain minimal effective doses. High-vibration amplitudes were selected as they appear to produce better adaptations for hypertrophy than low-vibration amplitudes [30].
Statistical analyses
Data was examined with box-plots in order to check for outliers. One participant was detected as an outlier for both BMD and BMC at pre- and post-intervention in most measured sites and was therefore excluded from further analyses.
Analyses of variance for repeated measures: 2 (group) by 2 (time) by 2 (sex), were performed in order to ascertain whether there was an interaction between group, time and sex. As no significant interactions were found for any of the outcome variables, all analyses were performed with male and female participants together.
Both an intention-to-treat analysis (ITT) which included all participants who began the protocol at baseline and a per-protocol (PP) analysis designed to evaluate the effects of compliance to WBV training were performed. All data were analyzed using SPSS version 15.0 (SPSS, Chicago, IL, USA). The same analyses were performed for the ITT and the PP. The PP analysis was performed to identify if any dose-response relationship existed. In this post-hoc analysis, the experimental cohort was subdivided into tertiles that were determined from the sample size, in order to have the same number of participants in each group. Firstly, a comparison between high- and medium-level compliers (over 50% attendance) and swimmers was performed. Secondly, the three groups (high, medium and low compliers) were compared to each other in order to ascertain whether any dose-response relationship existed.
Independent t tests were used to examine baseline differences between SWI and VIB for all anthropometric characteristics and dietary calcium intake. In addition, pre- and post-training bone variables (BMD and BMC) and lean mass were compared with ANCOVAs adjusting by age, Tanner stage and height. Chi-square tests were performed to evaluate differences in Tanner stage before and after the intervention.
A percentage of change was calculated for BMD, BMC and lean mass variables from the pre- and post-intervention values. ANOVAS were then performed with these scores in order to ascertain the differences between groups.
Analysis of covariance (ANCOVA) for repeated measures × 2 was performed to check differences within groups between pre- and post-intervention and to determine the effects of the intervention on BMD, BMC and lean mass values adjusting by change in height, initial age and final Tanner stage.
The repeated measures were performed to determine whether BMD, BMC and lean mass improved in both SWI and VIB within time, and if a group by time interactions existed. Thus, for a small- to medium-effect size (f = 0.20) and a correlation of 0.7 between the pre- and post-evaluation measurements of the within factors, repeated measures calculation determined a total sample size of 32 (16 per group), for a power of 0.8 and α of 0.05. For the repeated measures, within-between interaction, the required sample size was also of 32 (16 per group).
For the repeated measures changes within group analysis, partial eta squared (n 2 p ) calculated by SPSS was presented. Values of 0.01 can be considered small, and those around 0.06 moderate and over 0.14 large.
Results
Participants
A summary of the enrollment, loss of participants and final number of adolescents included in the study is presented in Fig. 2.
The final sample consisted of 40 VIB (22 males/18 females) and 23 SWI (14 males/9 females).
The included participants trained an average of 10 weekly hours (10.1 ± 2.6 h SWI vs. 10.1 ± 1.9 h VIB) during the follow-up period.
Compliance to training and side effects
Of all the included participants following the WBV protocol, one reported to increase his episodes of migraines due to the WBV training. No other adverse effects were reported.
The average WBV training compliance was of 62 ± 17% ranging from 32 to 97%. Regarding tertiles, the low-complier group (mean compliance 42%) consisted of 13 VIB who did not attain 50% of compliance (range between 32 and 49%). Participants in the low-compliance group trained for between 204 and 315 min on the platform (408 to 630 min when taking into account rest periods off the platform during the WBV training). Thirteen VIB were classified as medium compliers (mean compliance 60%) with compliance ranging from 50 to 67.5%. These participants trained for between 351 and 420 min on the platform (700 to 840 min of training including rest periods). Fourteen participants formed the high-complier group (mean compliance 82%) and trained between 67.5 and 97%, from 426 to 609 min (850 to 1200 min including rest periods).
Anthropometric characteristics and calcium intake
Participants’ physical characteristics are shown in Table 1. No differences were found for any of the measured variables between groups (Hedges’ g from 0.051 to 0.410; all p > 0.05).
DXA measurements
Table 2 summarizes the average-adjusted values of BMD, BMC and lean mass at pre-evaluation and the mean change percentage with the 95% limits agreement.
No group by time interactions were found for any of the studied variables (all p > 0.05; Table 2), suggesting that both groups (SWI and VIB) increased BMD, BMC and lean mass similarly.
Differences in BMD, BMC and lean mass change percentage for both ITT and PP analyses are plotted in Fig. 3. No significant differences were found for BMD, BMC or lean mass change percentage between SWI and VIB, although VIB presented a tendency towards higher change percentage for the total hip BMC (p = 0.07; Fig. 3.2). Results were similar when grouping high- and medium-compliers and comparing them to swimmers.
Regarding individual group changes, the VIB group increased trochanter, total hip, lumbar spine, subtotal body, arms and legs BMD and BMC (all p < 0.05; Table 2). While the SWI group increased femoral neck, lumbar spine, subtotal body, arms and legs BMD and BMC and total hip BMD (all p < 0.05; Table 2). These improvements were all large for both groups, as partial eta-squared was always above 0.14 (Table 2).
When comparing the three compliance groups, no differences were found among them for BMD or BMC values (all comparisons p > 0.05; data not presented).
Discussion
The main finding of the present study was that the WBV protocol presented, in combination with swimming training, did not provide any major benefits to BMD, BMC or lean mass acquisition.
As expected, both groups of adolescents increased BMD, BMC and lean mass values due to growth. Nevertheless, previous longitudinal studies evaluating swimmers’ bone mass found that swimmers increased BMD and BMC similarly to normo-active controls [31, 32] but less than other athletes [33] suggesting that swimmers were not stimulating bone mass as much as other adolescent athletes and thus might not reach a high-peak bone mass.
In light of the apparent similarity in bone mass between swimmers and controls and due to cross-sectional findings [34, 35], it was deemed appropriate to perform an intervention attempting to increase bone mass in this population. As adolescent swimmers attend regular swimming training sessions occupying considerable amounts of time (an average of 10 weekly hours for our sample), an intervention involving large amounts of time was not feasible. Therefore, a synchronous WBV training appeared to constitute a timesaving intervention as only 15 min three times per week was required. A platform could be placed in each swimming club in order for swimmers to perform the training just before or just after their regular training, thus maximizing time effectiveness. The results found in the present study were unpromising as 45 min per week over 6 months of WBV with increasing amplitudes and frequencies had no effect on bone mass. The only tendency towards differences between VIB and SWI was found for the total hip BMD and BMC values, with the VIB group gaining more BMC than the SWI. Nevertheless, no group by time interactions or differences were found for the measured regions. It therefore seems likely that WBV may not be the best method to employ in subjects with normally developed bone mass. Although it has shown to be effective to a degree in disabled [15] and overweight children [16], postmenopausal women [36] and older adults [37], all of which are populations that present compromised bones, it was not effective in the present group of adolescent swimmers who presented normally developed bone mass.
Similar results were found for lean mass, as there were no interactions between SWI and VIB and no differences in change percentage. These results were surprising as the WBV training was performed in addition to regular swimming training and not in substitution of it, and therefore, the extra weight-bearing training performed while vibrating could very probably have improved lean mass. Nevertheless, this was not the case. It is possible that the swimmers’ lean mass was already adapted to the high-intensity training performed while swimming, and therefore, the WBV training protocol performed might have not been sufficiently intense to cause a stimulus in order to induce muscle improvements. These results are similar to previous research that also found no effects of WBV on body composition in elderly [38], adults [39] or young adults [21]. In fact, few authors have found improvements in lean mass with WBV. Roelants et al. [19] found minor improvements in fat-free mass in an untrained group of young adult females who performed WBV three times per week during a 24-week period, with varying accelerations ranging from 2.28 to 5.09 g which were lower than those presented by our protocol (3.6 to 11.6 g). Similarly Martinez-Pardo et al. [30] found that WBV during a 6-week period performed with high amplitudes (4 mm) and 50 Hz twice per week could improve total lean mass in adults. Although the frequencies were slightly higher than those in the present study, the participants did not perform a specific sport and did not train more than three times per week for approximately 20–30 min. It is thus possible that they had a higher margin of improvement regarding body composition than the adolescent swimmers included in the present study. From the present results and taking into account the above-named studies, it seems as if WBV could be effective in improving lean mass in untrained participants but might not be an effective method with trained adolescents, as no benefits were found in highly trained swimmers. This lack of effect could be due to the WBV doses used, as it is possible that in the present training protocol, the minimum threshold to produce adaptations in this population was not reached. From a practical point of view, it is of great importance to ascertain the real effects of different training protocols in order to provide trainers with effective means of obtaining maximum performance with the least expenditure of time and effort, particularly among those sports groups with time-demanding training hours.
The lack of effect found with the ITT was reinforced by the PP results as no differences were found between high-medium compliers and swimmers. In addition, the lack of differences between the three WBV groups (high compliers vs. medium compliers vs. low compliers) suggests that the employed protocol conferred no training effect. Nonetheless, this lack of differences among the three vibration groups could also be due to the low sample size within each group that might have caused a type II error or a false negative, as it is possible that if there was a larger number of participants in each group, differences might have emerged. Consequently, the results regarding the per-protocol analysis should be interpreted with caution.
Although the present results regarding the effects of WBV on bone and lean mass are discouraging, further studies should evaluate the effects of these interventions on performance-related variables, as Torvinen et al. [40] who did evaluate performance-related variables in addition to body composition, found improvements in vertical jump height without improvements in bone or lean mass. Improving jump height could be highly beneficial for swimming performance as several studies have found that it is associated with swimming performance [41, 43].
It is worth mentioning that the WBV platforms used were synchronous devices that transferred the vibration to both feet synchronously. A study performed by Ritzmann et al. [42] comparing synchronous devices to side-alternating devices (WBV platforms that vibrate in a side-alternating way so that the right foot is lower when the left foot is higher and vice versa) showed that the side-alternating platforms provoked higher neuromuscular activity in the lower limb muscles which may consequently mean a higher bone mass stimulus. Therefore, the present findings do not necessarily apply to side-alternating devices and further studies should evaluate the effects of them on adolescent swimmers’ bone and lean mass. Another important variable regarding WBV platforms is vibration transmission as showed by the study developed by Kiiski et al. [43]. These researchers evaluated the transmission of vertical whole-body vibration to the human body demonstrating that at high frequencies and amplitudes (similar to those presented in the current study), the transmitted vibration power declines. Therefore, a higher vibration transmission might occur at lower frequencies (10 Hz was found to be the optimal frequency for vibration transmission to the lumbar spine). Consequently, it is possible that results might have varied if a different WBV protocol with lower frequencies had been used.
It is worth remarking that just a handful of studies have used WBV interventions with the aim of improving bone mass in healthy adolescents; thus, the protocol designed for this study was based on previous results in our laboratory [28, 29] that found bone mass improvements in adolescents with disabilities. Nevertheless, other protocols might be more beneficial to bone mass improvements; thus, it would be interesting to perform further studies with larger sample sizes that divide participants into different WBV protocols that aim to determine which is the optimum frequency and amplitude for WBV training to improve bone mass. The lack of strong scientific evidence regarding optimal WBV protocols to improve bone mass leaves researchers to choose WBV protocols based on few studies. It is therefore possible that if a different protocol had been used, different results would have been found.
In addition to the previous limitation, training intensities were not specific to each participant; therefore, training stimuli might have not been sufficient to improve bone mass in some participants. Several studies have found substantial individual heterogeneity in physiological responses and benefits from standardized exercise [44, 45]. Moreover, specifically designed exercise interventions have been proven to be more effective than general ones [46].
It would appear that the WBV training protocol presented has no effects on bone or lean mass acquisition. Owing to the results of the present intervention and the cost of the devices employed in it, it would be interesting to perform future studies evaluating the effects of other interventions such as jumping training protocols on the bone mass of adolescent swimmers.
References
Frost HM (2003) Bone’s mechanostat: a 2003 update. Anat Rec A Discov Mol Cell Evol Biol 275:1081–1101
Sherrington C, Whitney JC, Lord SR, Herbert RD, Cumming RG, Close JC (2008) Effective exercise for the prevention of falls: a systematic review and meta-analysis. J Am Geriatr Soc 56:2234–2243
Specker B, Minett M (2013) Can physical activity improve peak bone mass? Curr Osteoporos Rep 11:229–236
Hind K, Burrows M (2007) Weight-bearing exercise and bone mineral accrual in children and adolescents: a review of controlled trials. Bone 40:14–27
Vicente-Rodriguez G (2006) How does exercise affect bone development during growth? Sports Med 36:561–569
Gonzalez-Aguero A, Casajus JA, Vicente-Rodriguez G (2012) Cycling and adolescent bone health. Apunts Med Esport 47:169
Gomez-Bruton A, Gonzalez-Aguero A, Gomez-Cabello A, Casajus JA, Vicente-Rodriguez G (2013) Is bone tissue really affected by swimming? A systematic review. PLoS One 8:e70119
Gomez-Bruton A, Montero-Marin J, Gonzalez-Aguero A, Garcia-Campayo J, Moreno LA, Casajus JA, Vicente-Rodriguez G (2015) The effect of swimming during childhood and adolescence on bone mineral density: a systematic review and meta-analysis. Sports Med 46:365–379
Bailey DA, Martin AD, McKay HA, Whiting S, Mirwald R (2000) Calcium accretion in girls and boys during puberty: a longitudinal analysis. J Bone Miner Res 15:2245–2250
Gomez-Bruton A, Gonzalez-Aguero, A, Gomez-Cabello, A, Matute-Llorente, A, Casajus, JA, Vicente-Rodriguez, G (2015) Swimming and bone: is low bone mass due to hypogravity alone or does other physical activity influence it? Osteoporos Int.
Gomez-Bruton A, González-Agüero A, Gómez-Cabello A, Matute-Llorente A, Casajús J, Vicente-Rodríguez G (2015) The effects of swimming training on bone tissue in adolescence. Scand J Med Sci Sports:25
Schoenau E (2005) The “functional muscle-bone unit”: a two-step diagnostic algorithm in pediatric bone disease. Pediatr Nephrol 20:356–359
Courteix D, Lespessailles E, Loiseau-Peres S, Obert P, Ferry B, Benhamou CL (1998) Lean tissue mass is a better predictor of bone mineral content and density than body weight in prepubertal girls. Rev Rhum (English Edition) 65:328–336
Avlonitou E, Georgiou E, Douskas C, Louizi A (1997) Estimation of body composition in competitive swimmers by means of three different techniques. Int J Sports Med 18:363–368
Matute-Llorente A, Gonzalez-Aguero A, Gomez-Cabello A, Vicente-Rodriguez G, Casajus Mallen JA (2014) Effect of whole-body vibration therapy on health-related physical fitness in children and adolescents with disabilities: a systematic review. J Adolesc Health 54:385–396
Erceg DN, Anderson LJ, Nickles CM, Lane CJ, Weigensberg MJ, Schroeder ET (2015) Changes in bone biomarkers, BMC, and insulin resistance following a 10-week whole body vibration exercise program in overweight Latino boys. Int J Med Sci 12:494–501
Slatkovska L, Alibhai SM, Beyene J, Cheung AM (2010) Effect of whole-body vibration on BMD: a systematic review and meta-analysis. Osteoporos Int 21:1969–1980
Gonzalez-Aguero A, Matute-Llorente A, Gomez-Cabello A, Casajus JA, Vicente-Rodriguez G (2013) Effects of whole body vibration training on body composition in adolescents with Down syndrome. Res Dev Disabil 34:1426–1433
Roelants M, Delecluse C, Goris M, Verschueren S (2004) Effects of 24 weeks of whole body vibration training on body composition and muscle strength in untrained females. Int J Sports Med 25:1–5
Song GE, Kim K, Lee DJ, Joo NS (2011) Whole body vibration effects on body composition in the postmenopausal Korean obese women: pilot study. Korean J Fam Med 32:399–405
Osawa Y, Oguma Y, Onishi S (2011) Effects of whole-body vibration training on bone-free lean body mass and muscle strength in young adults. J Sports Sci Med 10:97–104
Tanner JM, Whitehouse RH, Takaishi M (1966) Standards from birth to maturity for height, weight, height velocity, and weight velocity: British children, 1965. II. Arch Dis Child 41:613–635
Leone M, Comtois AS (2007) Validity and reliability of self-assessment of sexual maturity in elite adolescent athletes. J Sports Med Phys Fitness 47:361–365
Julián-Almárcegui C, Huybrechts I, Gómez-Bruton A, Matute-LLorente A, González-Agüero A, Gómez-Cabello A, Moreno L, Casajús J, Vicente-Rodríguez G (2015) Validity of a food-frequency questionnaire for estimating calcium intake in adolescent swimmers. Nutr Hosp:32
Gracia-Marco L, Ortega FB, Jimenez-Pavon D, Rodriguez G, Castillo MJ, Vicente-Rodriguez G, Moreno LA (2012) Adiposity and bone health in Spanish adolescents. The HELENA study. Osteoporos Int 23:937–947
Merriman H, Jackson K (2009) The effects of whole-body vibration training in aging adults: a systematic review. J Geriatr Phys Ther 32:134–145
Sitja-Rabert M, Rigau D, Fort Vanmeerghaeghe A, Romero-Rodriguez D, Bonastre Subirana M, Bonfill X (2012) Efficacy of whole body vibration exercise in older people: a systematic review. Disabil Rehabil 34:883–893
Matute-Llorente A, Gonzalez-Aguero A, Gomez-Cabello A, Olmedillas H, Vicente-Rodriguez G, Casajus JA (2015) Effect of whole body vibration training on bone mineral density and bone quality in adolescents with Down syndrome: a randomized controlled trial. Osteoporos Int 26:2449–2459
Matute-Llorente A, Gonzalez-Aguero A, Gomez-Cabello A, Tous-Fajardo J, Vicente-Rodriguez G, Casajus JA (2016) Effect of whole-body vibration training on bone mass in adolescents with and without Down syndrome: a randomized controlled trial. Osteoporos Int 27:181–191
Martinez-Pardo E, Romero-Arenas S, Alcaraz PE (2013) Effects of different amplitudes (high vs. low) of whole-body vibration training in active adults. J Strength Cond Res 27:1798–1806
Czeczelewski J, Dlugolecka B, Czeczelewska E, Raczynska B (2013) Intakes of selected nutrients, bone mineralisation and density of adolescent female swimmers over a three-year period. Biol Sport 30:17–20
Maimoun L, Coste O, Philibert P, Briot K, Mura T, Galtier F, Castes-de-Paulet B, Mariano-Goulart D, Sultan C, Paris F (2013) Testosterone secretion in elite adolescent swimmers does not modify bone mass acquisition: a 1-year follow-up study. Fertil Steril 99:270–278
Ferry B, Lespessailles E, Rochcongar P, Duclos M, Courteix D (2013) Bone health during late adolescence: effects of an 8-month training program on bone geometry in female athletes. Joint Bone Spine 80:57–63
Gomez-Bruton A, González-Agüero A, Casajus JA, Vicente-Rodríguez G (2014) Swimming training repercussion on metabolic and structural bone development. Benefits of the incorporation of whole body vibration or pliometric training. The RENACIMIENTO project. Nutr Hosp 30:399–409
Dlugolęcka B, Czeczelewski J, Raczyńska B (2011) Bone mineral content and bone mineral density in female swimmers during the time of peak bone mass attainment. Biol Sport 28:69–74
Oliveira LC, Oliveira RG, Pires-Oliveira DA (2016) Effects of whole body vibration on bone mineral density in postmenopausal women: a systematic review and meta-analysis. Osteoporos Int 27:2913–2933
Gusi N, Raimundo A, Leal A (2006) Low-frequency vibratory exercise reduces the risk of bone fracture more than walking: a randomized controlled trial. BMC Musculoskelet Disord 7:92
Gomez-Cabello A, Gonzalez-Aguero A, Ara I, Casajus JA, Vicente-Rodriguez G (2012) Effects of a short-term whole body vibration intervention on lean mass in elderly people. Nutr Hosp 28:1255–1258
Tapp LR, Signorile JF (2014) Efficacy of WBV as a modality for inducing changes in body composition, aerobic fitness, and muscular strength: a pilot study. Clin Interv Aging 9:63–72
Torvinen S, Kannus P, Sievanen H, Jarvinen TA, Pasanen M, Kontulainen S, Nenonen A, Jarvinen TL, Paakkala T, Jarvinen M, Vuori I (2003) Effect of 8-month vertical whole body vibration on bone, muscle performance, and body balance: a randomized controlled study. J Bone Miner Res 18:876–884
West DJ, Owen NJ, Cunningham DJ, Cook CJ, Kilduff LP (2011) Strength and power predictors of swimming starts in international sprint swimmers. J Strength Cond Res 25:950–955
Ritzmann R, Gollhofer A, Kramer A (2013) The influence of vibration type, frequency, body position and additional load on the neuromuscular activity during whole body vibration. Eur J Appl Physiol 113:1–11
Kiiski J, Heinonen A, Jarvinen TL, Kannus P, Sievanen H (2008) Transmission of vertical whole body vibration to the human body. J Bone Miner Res 23:1318–1325
Wilmore JH, Leon AS, Rao DC, Skinner JS, Gagnon J, Bouchard C (1997) Genetics, response to exercise, and risk factors: the HERITAGE family study. World Rev Nutr Diet 81:72–83
Bouchard C, Rankinen T (2001) Individual differences in response to regular physical activity. Med Sci Sports Exerc 33:S446–S451 discussion S452–443
Neto CF, Neto GR, Araujo AT, Sousa MS, Sousa JB, Batista GR, Reis VM (2014) Can programmed or self-selected physical activity affect physical fitness of adolescents? J Hum Kinet 43:125–130
Acknowledgements
We would like to thank participants and their families and coaches for the collaboration. Special thanks are given to Lindsey A. Bruton for her work in reviewing the English style and grammar. This work was supported by the Spanish “Ministerio de Economia y competitividad” “Plan Nacional I+D+i 2008-2011 (Project DEP2011-29093).” AGB received a Grant FPI 2012 (BES-2012-051888) from the “Ministerio Economía y Competitividad.” AML received a Grant AP2012/02854 from the “Ministerio de Educación Cultura y Deportes.” CJ received a Grant FPU 2013 (FPU13/00421) from the “Ministerio de Educación Cultura y Deportes.” GLB received a Grant FPU 2013 (FPU13/02111) from the “Ministerio de Educación, Cultura y Deporte.”
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None.
Rights and permissions
About this article
Cite this article
Gómez-Bruton, A., González-Agüero, A., Matute-Llorente, A. et al. Do 6 months of whole-body vibration training improve lean mass and bone mass acquisition of adolescent swimmers?. Arch Osteoporos 12, 69 (2017). https://doi.org/10.1007/s11657-017-0362-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11657-017-0362-z