Abstract
Objective
To investigate the effects of Shengmai Injection (生脉注射液, SMI) on the proliferation, apoptosis and N-myc downstream-regulated gene 2 (NDRG2, a tumour suppressor gene) expression in varying densities of human hepatic stellate cells LX-2.
Methods
LX-2 cells were cultured in vitro. Then, cells were plated in 96-well plates at an approximate density of 2.5×104 cells/mL and cultured for 48, 72, 96 or 120 h followed by the application of different concentrations of SMI (0.6, 1.2, 2.4, 4.8 or 6 μL/mL). Cell proliferation was measured after an additional 24 or 48 h using the 3(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. The effects of SMI on different cell growth states (cultured for 48, 72, 96, or 120 h) were observed by light microscopy at 24 h after treatment. When the cells reached 80% conflfluence, apoptosis was detected by flflow cytometry after 24 h. Lastly, LX-2 cells were treated with different concentrations of SMI and extracted with protein lysis buffer. The levels of NDRG2 were measured by Western blot.
Results
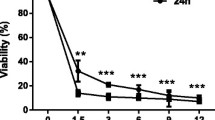
When the LX-2 cells grew for 48, 72, 96 and 120 h, 4.8 and 6 μL/mL of SMI significantly inhibited cell proliferation at 24 and 48 h after treatment (P<0.05). And 2.4 μL/mL of SMI also inhibited cell proliferation at 24 h after treatment when cell growth for 48 h (P<0.05) and at 48 h after treatment when cell growth for 72, 96 and 120 h (P<0.05). The NDRG2 expression level in the LX-2 cell was significantly increased when treated with SMI at concentrations of 1.2, 2.4, 4.8 or 6 μL/mL (P<0.05).
Conclusion
The inhibitory effects of SMI on the proliferation of LX-2 cells were related to not only concentration dependent but also cell density. In addition, SMI (2.4, 4.8 and 6 μL/mL) could accelerate apoptosis in LX-2 cells, and the mechanism might be associated with NDRG2 over-expression.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Li F, Ma N, Zhao R, Wu G, Zhang Y, Qiao Y, et al. Overexpression of miR–483–5p/3p cooperate to inhibit mouse liver fibrosis by suppressing the TGF–beta stimulated HSCs in transgenic mice. J Cell Mol Med 2014;18:966–974.
Gou X, Tao Q, Feng Q, Peng J, Zhao Y, Dai J, et al. Urine metabolic profile changes of CCl4–liver fibrosis in rats and intervention effects of Yi Guan Jian Decoction using metabonomic approach. BMC Complement Altern Med 2013;13:123.
D’Argenio G, Amoruso DC, Mazzone G, Vitaglione P, Romano A, Ribecco MT, et al. Garlic extract prevents CCl4–induced liver fibrosis in rats: the role of tissue transglutaminase. Dig Liver Dis 2010;42:571–577.
Fan WM, Shi BY. Advances in studies on apoptotic factors of hepatic stellate cell. Med Recapitul (Chin) 2012;18:1797–1799.
Henderson NC, Mackinnon AC, Farnworth SL, Poirier F, Russo FP, Iredale JP, et al. Galectin–3 regulates myofibroblast activation and hepatic fibrosis. Proc Natl Acad Sci USA 2006;103:5060–5065.
Huang CL, Zhao WX. Progress on treating hepatic fibrosis in Chinese medicine. Clin J Chin Med (Chin) 2014;2:146–147.
Yang Q, Feng Y, Jiang SL. Experiences of Prof. YAO Xixian in treating chronic hepatic fibrosis based on blood stasis theory. Chin J Chin Med Pharm (Chin) 2007;22:168–171.
Cheng Y, Mai JY, Wang MF, Chen GF, Ping J. Antifibrotic effect of total flavonoids of Astmgali Radix on dimethylnitrosamine–induced liver cirrhosis in rats. Chin J Integr Med 2017;23:48–54.
Liu P. Inhibition of pathological angiogenesis of Chinese medicine against liver fibrosis. Chin J Integr Med 2016;22:569–572.
Xuan J. Clinical study of Huagan Tongluo Recipe and the mechanism of Th17 cell differentiation involved in hepatic fibrosis. Nanjing: Nanjing University of Chinese Medicine;2018.
Deng ZJ, ed. Formulas of Chinese medicine. Beijing: China Press of Traditional Chinese Medicine;2011:152–153.
Rong XF, Yao WY. Influence of Shenmai Injection combined with Puerarin Injection on liver fibrosis indexes in patients with cirrhosis. World Chin J Digestol (Chin) 2006;14:3326–3329.
Wang L, Liu N, Yao L, Li F, Zhang J, Deng Y, et al. NDRG2 is a new HIF–1 target gene necessary for hypoxia–induced apoptosis in A549 cells. Cell Physiol Biochem 2008;21:239–250.
Shen L, Zhao ZY, Wang YZ, Ji SP, Liu XP, Liu XW, et al. Immunohistochemical detection of NDRG2 in the mouse nervous system. Neuroreport 2008;19:927–931.
Boulkroun S, Fay M, Zennaro MC, Escoubet B, Jaisser F, Blot–Chabaud M, et al. Characterization of rat NDRG2 (N–Myc downstream regulated gene 2), a novel early mineralocorticoidspecific induced gene. J Biol Chem 2002;277:31506–31515.
Zheng J, Li Y, Yang J, Liu Q, Shi M, Zhang R, et al. NDRG2 inhibits hepatocellular carcinoma adhesion, migration and invasion by regulating CD24 expression. BMC Cancer 2011;11:251.
Yang J, Zheng J, Wu L, Shi M, Zhang H, Wang X, et al. NDRG2 ameliorates hepatic fibrosis by inhibiting the TGFbeta1/Smad pathway and altering the MMP2/TIMP2 ratio in rats. PLoS One 2011;6:e27710.
Yang JD. Expression of NDRG2 in liver injury–repair and preliminary study on its function. Xi’an: The Fourth Military Medical University;2013.
Xu L, Hui AY, Albanis E, Arthur MJ, O’Byrne SM, Blaner WS, et al. Human hepatic stellate cell lines, LX–1 and LX–2: new tools for analysis of hepatic fibrosis. Gut 2005;54:142–151.
Zhou WC, Zhang QB, Qiao L. Pathogenesis of liver cirrhosis. World J Gastroenterol 2014;20:7312–7324.
Duval F, Moreno–Cuevas JE, González–Garza MT, Rodríguez–Montalvo C, Cruz–Vega DE. Protective mechanisms of medicinal plants targeting hepatic stellate cell activation and extracellular matrix deposition in liver fibrosis. Chin Med 2014;9:27.
Su TH, Kao JH, Liu CJ. Molecular mechanism and treatment of viral hepatitis–related liver fibrosis. Int J Mol Sci 2014;15:10578–10604.
Hu W, Fan C, Jiang P, Ma Z, Yan X, Di S, et al. Emerging role of N–myc downstream–regulated gene 2 (NDRG2) in cancer. Oncotarget 2016;7:209–223.
Yoon SH, Nam YM, Hong JT, Kim SJ, Ko SK. Modification of ginsenoside composition in red ginseng (Panax ginseng) by ultrasonication. J Ginseng Res 2016;40:300–303.
Yao C, Shi X, Lin X, Shen L, Xu D, Feng Y. Increased cardiac distribution of mono–PEGylated Radix Ophiopogonis polysaccharide in both myocardial infarction and ischemia/reperfusion rats. Int J Nanomed 2015;10:409–418.
Szopa A, Ekiert R, Ekiert H. Current knowledge of Schisandra chinensis (Turcz.) Baill. (Chinese magnolia vine) as a medicinal plant species: a review on the bioactive components, pharmacological properties, analytical and biotechnological studies. Phytochem Rev 2017;16:195–218.
Liu X, Tan W, Yang F, Wang Y, Yue S, Wang T, et al. Shengmai Injection reduces apoptosis and enhances angiogenesis after myocardial ischaemia and reperfusion injury in rats. Biomed Pharmacother 2018;104:629–636.
Yang H, Li L, Zhou K, Wang Y, Guan T, Chai C, et al. Shengmai Injection attenuates the cerebral ischemia/reperfusion induced autophagy via modulation of the AMPK, mTOR and JNK pathways. Pharm Biol 2016;10:2288–2297.
Duan B, Xie J, Rui Q, Zhang W, Xi Z. Effects of Shengmai Injection add–on therapy to chemotherapy in patients with non–small cell lung cancer: a meta–analysis. Support Care Cancer 2018;26:2103–2111.
Acknowledgement
We sincerely thank Prof. BIAN Hui-jie from the Fourth Military Medical University for donating LX-2 cell lines, and thank Dr. BAI Yang and Dr. ZHANG Ge from the Fourth Military Medical University for revising the manuscript. We also thank all of our colleagues for their generous support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the National Natural Sciences Foundation of China (No. 81072973)
Rights and permissions
About this article
Cite this article
Zhang, Y., Ma, Lt., Li, J. et al. Anti-fibrotic Effects and Mechanism of Shengmai Injection (生脉注射液) on Human Hepatic Stellate Cells LX-2. Chin. J. Integr. Med. 25, 197–202 (2019). https://doi.org/10.1007/s11655-018-2849-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11655-018-2849-x