Abstract
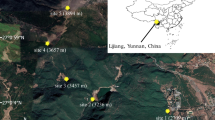
Maximum plant height (Hmax), stem tissue mass density (SD), leaf mass per area (LMA), and relative growth rate (RGR) have all been linked to flowering phenology. However, it is still unknown whether these functional traits varying with flowering phenology are related to other floral traits associated with pollinator preference and reproductive success. We investigated the relationship between vegetative and floral traits, as well as the rates of insect visitation and seed set of five Gentiana species in eastern Tibetan meadows. Our results showed that flowering onset and flowering offset time were all found to be positively correlated with the Hmax, SD, and LMA, but negatively correlated with the RGR. Flowering onset time was positively correlated with corolla diameter and pollen grain number, whereas was negatively correlated with flower number and flower longevity. The rates of insect visitation were positively related to flowering onset time, pollen grain number, corolla diameter, and seed set, but negatively related to flower number and longevity. Early flowering species have higher RGR but lower SD, LMA and Hmax, as well as smaller flowers, fewer pollen grains, longer flower longevity, and lower insect visitation rates than late-flowering plant species. Our findings indicate that floral traits are related to vegetative traits in Gentiana species. Also these plant trait relationships were associated with pollinator preference, and plant reproductive success of eastern Tibetan meadows. We speculate that these traits relationships are likely adaptive in unpredictable and often pollinator-limited environments in the Tibet alpine meadows.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ågren J (2019) Pollinators, herbivores, and the evolution of floral traits. Science 364(6436): 122–123. https://doi.org/10.1126/science.aax1656
Althoff DM, Segraves KA, Pellmyr O (2005) Community context of an obligate mutualism: Pollinator and florivore effects on Yucca filamentosa. Ecology 86(4): 905–913. https://doi.org/10.1890/04-1454
Arroyo MTK, Armesto JJ, Primack RB (1985) Community studies in pollination ecology in the high temperate Andes of central Chile. II. Effect of temperature on visitation rates and pollination possibilities. Plant Syst Evol 149(3): 187–203. https://doi.org/10.1007/BF00983305
Barberis M, Bogo G, Bortolotti L, et al. (2021) Gender-biased nectar targets different behavioral traits of flower visitors. Plant Ecol 222: 233–246. https://doi.org/10.1007/s11258-020-01101-5
Bolmgren K, Cowan PD (2008) Time-size tradeoffs: a phylogenetic comparative study of flowering time, plant height and seed mass in a north-temperate flora. Oikos 117(3): 424–429. https://doi.org/10.1111/j.2007.0030-1299.16142.x
Brown VK, Lawton JH (1991) Herbivory and the evolution of leaf size and shape. Philos T R Soc B 333(1267): 265–272. https://doi.org/10.1098/rstb.1991.0076
Broz AK, Randle AM, Sianta SA, et al. (2017) Mating system transitions in Solanum habrochaites impact interactions between populations and species. New Phytol 213(1): 440–454. https://doi.org/10.1111/nph.14130
Cruden RW (1977) Pollen-ovule ratios: a conservative indicator of breeding systems in plants. Evolution 31:32–46. https://doi.org/10.2307/2407542
Dai WF, Yang YL, Patch HM, et al. (2022). Soil moisture affects plant-pollinator interactions in an annual flowering plant. Philos T R Soc B 377(1853): 20210423. https://doi.org/10.1098/rstb.2021.0423
Donoghue M, Ree RH, Baum DA (1998) Phylogeny and the evolution of flower symmetry in the Asteridae. Trends Plant Sci 3(8):311–317. https://doi.org/10.1016/S1360-1385(98)01278-3
Du GZ, Qi W (2010) Trade-offs between flowering time, plant height, and seed size within and across 11 communities of a QingHai-Tibetan flora. Plant Ecol 209(2): 321–333. https://doi.org/10.1007/s11258-010-9763-4
Fenster CB, Cheely G, Dudash MR, et al. (2006) Nectar reward and advertisement in hummingbird-pollinated Silene virginica (Caryophyllaceae). Am J Bot 93(12): 1800–1807. https://doi.org/10.3732/ajb.93.12.1800
Firon N, Shaked R, Peet M, et al. (2006). Pollen grains of heat tolerant tomato cultivars retain higher carbohydrate concentration under heat stress conditions. Sci Hortic 109, 212–217. https://doi.org/10.1016/j.scienta.2006.03.007
Galen C (1989) Measuring pollinator-mediated selection on morphometric floral traits: bumblebees and the alpine sky pilot, Polemonium viscosum. Evolution 43(4): 882–890. https://doi.org/10.1111/j.1558-5646.1989.tb05185.x
Geber MA, Moeller DA, Harder LD, et al. (2006) Pollinator responses to plant communities and implications for reproductive character evolution, Ecology and Evolution of Flowers, Oxford University Press. pp 102–119.
Hansen J, Møller IB (1975) Percolation of starch and soluble carbohydrates from plant tissue for quantitative determination with anthrone. Anal Biochem 68(1): 87–94. https://doi.org/10.1016/0003-2697(75)90682-x
He YP, Duan YW, Liu JQ, et al. (2005) Floral closure in response to temperature and pollination in Gentiana straminea Maxim. (Gentianaceae), an alpine perennial in the Qinghai-Tibetan Plateau. Plant Syst Evol 256(1/4): 17–33. https://doi.org/10.1007/s00606-005-0345-1
Heady FH (1957) The measurement and value of plant height in the study of herbaceous vegetation. Ecology 38(2): 313–320. https://doi.org/10.2307/1931691
Hovenden MJ, Wills KE, Schoor JKV, et al. (2008) Flowering phenology in a species-rich temperate grassland is sensitive to warming but not elevated CO2. New Phytol 178(4): 815–822. https://doi.org/10.1111/j.1469-8137.2008.02419.x
Hu L, Wu XW (2019) The difference in pollen harvest between Apis mellifera and Apis cerana in a Tibetan alpine meadow. J Mt Sci-Engl 16: 1598–1605. https://doi.org/10.1007/s11629-018-5245-2
Hunt R (1982) Plant growth curves. The functional approach to plant growth analysis. London: Edward Arnold.
Hunt R, Cornelissen JHC (1997) Components of relative growth rate and their interrelations in 59 temperate plant species. New Phytol 135(3): 395–417. https://doi.org/10.1046/j.1469-8137.1997.00671.x
Junker RR, Larue-Kontić AAC (2018) Elevation predicts the functional composition of alpine plant communities based on vegetative traits, but not based on floral traits. Alpine Bot 128:13–22. https://doi.org/10.1007/s00035-017-0198-6
König P, Tautenhahn S, Cornelissen JHC, et al. (2018) Advances in flowering phenology across the Northern Hemisphere are explained by functional traits. Global Ecol Biogeogr 27(3): 310–321. https://doi.org/10.1111/geb.12696
Kudo G (1993) Relationship between flowering time and fruit set of the entomophilous alpine shrub, Rhododendron aureum (Ericaceae), inhabiting snow patches. Am J Bot 80(11): 1300–1304. https://doi.org/10.1002/j.1537-2197.1993.tb15368.x
Kühsel S, Blüthgen N (2015) High diversity stabilizes the thermal resilience of pollinator communities in intensively managed grasslands. Nat Commun 6(1): 7989. https://doi.org/10.1038/ncomms8989
Kulbaba MW, Worley NC (2012) Selection on floral design in Polemonium brandegeei (Polemoniaceae): female and male fitness under hawkmoth pollination. Evolution 66(5): 1344–1359. https://doi.org/10.1111/j.1558-5646.2011.01536.x
Levin DA (2012) Mating system shifts on the trailing edge. Ann Bot-London 109(3): 613–620. https://doi.org/10.1093/aob/mcr159
Li JK, Song YP, Xu H, et al. (2015) High ratio of illegitimate visitation by small bees severely weakens the potential function of heteranthery. J Plant Ecol 8(2): 213–223. https://doi.org/10.1093/jpe/rtv021
Liu YZ, Li GY, Wu XW, et al. (2021) Linkage between species traits and plant phenology in an alpine meadow. Oecologia 195(2):409–419. https://doi.org/10.1007/s00442-020-04846-y
Lloyd DG (1965) Evolution of self-compatibility and racial differentiation in Leavenworthia (Cruciferae). Contributions of the Gray Herbarium Harvard University 195: 3–134. https://doi.org/10.1093/aob/mcr159
Mitchell RJ (2004). Heritability of nectar traits: why do we know so little. Ecology 85(6): 1527–1533. https://doi.org/10.1890/03-0388
Mu JP, Li GY, Niklas KJ, et al. (2011) Difference in floral traits, pollination, and reproductive success between white and blue flowers of Gentiana leucomelaena (Gentianaceae) in an Alpine Meadow. Arct Antarct Alp Res 43(3): 410–416. https://doi.org/10.1657/1938-4246-43.3.410
Mu JP, Peng YH, Xi XQ, et al. (2014a) Domesticated honeybees evolutionarily reduce flower nectar volume in a Tibetan Asteraceae. Ecology 95(11): 3161–3172. https://doi.org/10.1890/13-2055.1
Muth F, Francis JS, Leonard AS (2016) Bees use the taste of pollen to determine which flowers to visit. Biol Letters 12(7): 20160356. https://doi.org/10.1098/rsbl.2016.0356
Nicolson SW, Wright GA (2017) Plant-pollinator interactions and threats to pollination: perspectives from the flower to the landscape. Funct Ecol 31(1): 22–25. https://doi.org/10.1111/1365-2435.12810
Nock CA, Vogt RJ, Beisner BE (2016) Functional Traits. In: eLS. John Wiley & Sons, Ltd: Chichester. https://doi.org/10.1002/9780470015902.a0026282
Norris K (1965) The bionomics of blow flies. Annu Rev Entomol 10(1): 47–68. https://doi.org/10.1146/annurev.en.10.010165.000403
Parachnowitsch AL, Manson J S, Sletvold N (2019) Evolutionary ecology of nectar. Ann Bot-London 123(2): 247–261. https://doi.org/10.1093/aob/mcy132
Pélabon C, Osler N, Diekmann M, et al. (2013) Decoupled phenotypic variation between floral and vegetative traits: distinguishing between developmental and environmental correlations. Ann Bot-London 111(5): 935–944. https://doi.org/10.1093/aob/mct050
Petanidou T (2001) Differential pollination success in the course of individual flower development and flowering time in Gentiana pneumonanthe L. (Gentianaceae). Bot J Linn Soc 135(1): 25–33. https://doi.org/10.1006/bojl.2000.0356
R Core Team (2020) R: A Language and Environment for Statistical Computing. Vienna, Austria: R Foundation for Statistical Computing. Retrieved from https://www.R-project.org/
Rathcke B, Lacey EP (1985) Phenological patterns of terrestrial plants. Annu Rev Ecol Syst 16(1): 179–214. http://www.jstor.org/journals/annrevs.html
Real LA, Rathcke BJ (1991). Individual Variation in Nectar Production and Its Effect on Fitness in Kalmia Latifolia. Ecology 72(1): 149–155. https://doi.org/10.2307/1938910
Sahli HF, Conner JK (2011) Testing for conflicting and nonadditive selection: floral adaptation to multiple pollinators through male and female fitness. Evolution 65(5): 1457–1473. https://doi.org/10.1111/j.1558-5646.2011.01229.x
Sargent RD, Goodwillie C, Kalisz S, et al. (2007) Phylogenetic evidence for a flower size and number trade-off. Am J Bot 94(12): 2059–2062. https://doi.org/10.3732/ajb.94.12.2059
Shaked R, Rosenfeld K, Pressman E (2004) The effect of low night temperatures on carbohydrates metabolism in developing pollen grains of pepper in relation to their number and functioning. Sci Hortic-Amsterdam 102(1): 29–36. https://doi.org/10.1016/j.scienta.2003.12.007
Sheoran IS, Saini HS (1996) Drought-induced male sterility in rice: Changes in carbohydrate levels and enzyme activities associated with the inhibition of starch accumulation in pollen. Sexual Plant Reprod 9(3): 161–167. https://doi.org/10.1007/BF02221396
Su RJ, Dai WF, Yang YL, et al. (2022) Introduced honey bees increase host plant abundance but decrease native bumble bee species richness and abundance. Ecosphere 13(6): e4085. https://doi.org/10.1002/ecs2.4085
Sun SC, Frelich LE (2011) Flowering phenology and height growth pattern are associated with maximum plant height, relative growth rate and stem tissue mass density in herbaceous grassland species. J Ecol 99(4): 991–1000. https://doi.org/10.1111/j.1365-2745.2011.01830.x
Vaudo AD, Patch HM, Mortensen DA, et al. (2014) Bumble bees exhibit daily behavioral patterns in pollen foraging. Arthropod-Plant Inte 8(4): 273–283. https://doi.org/10.1007/s11829-014-9312-5
Vaudo AD, Patch HM, Mortensen DA, et al. (2016) Macronutrient ratios in pollen shape bumble bee (Bombus impatiens) foraging strategies and floral preferences. P Natl Acad Sci US A 113(28): E4035–E4042. https://doi.org/10.1073/pnas.1606101113
Vaudo AD, Tooker JF, Patch HM, et al. (2020) Pollen Protein: Lipid Macronutrient Ratios May Guide Broad Patterns of Bee Species Floral Preferences. Insects 11(2): 132. https://doi.org/10.3390/insects11020132
Waites AR, Ågren J (2004) Pollinator visitation, stigmatic pollen loads and among population variation in seed set in Lythrum salicaria. J Ecol 92(3): 512–526. https://doi.org/10.1111/j.0022-0477.2004.00893.x
Wang ZH, Chlus A, Geygan R, et al. (2020) Foliar functional traits from imaging spectroscopy across biomes in eastern North America. New Phyto 228(2): 494–511. https://doi.org/10.1111/nph.16711
Young HJ, Stanton ML (1990) Influences of floral visitation on pollen removal and seed production in wild Radish. Ecology 71(2): 536–547. https://doi.org/10.2307/1940307
Yoshida S, Forno D, Cock J, et al. (1976) Laboratory manual for physiological studies of rice. Manila, Philippines: International Rice Research Institute.
Acknowledgements
We thank Hui Wang, Jie Xiong, Yuling Zeng, Yanling Luo, Rui Cao, Hanxiang Chen, Xue Wei, and Kai He for the field and lab assistance. This study was funded by National Natural Science Foundation of China (31870393, 31270513) and the open project from the Ecological Security and Protection Key Laboratory of Sichuan Province (ESP111503).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dai, Wf., Gao, R., He, My. et al. Linkages among the functional traits, insect visitation rate and seed set of Gentiana taxa on the Tibetan Plateau. J. Mt. Sci. 19, 2312–2321 (2022). https://doi.org/10.1007/s11629-021-6749-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11629-021-6749-8