Abstract
Background
Preoperative pulmonary function assessment is useful for selecting surgical candidates and operative methods and assessing the risk of postoperative pulmonary complications. However, few studies have investigated the relationship between preoperative pulmonary function and short- and long-term outcomes in patients who underwent gastrectomy for gastric cancer.
Methods
Of the 1040 patients with gastric cancer (stages I–III) who had undergone R0 gastrectomy between 2009 and 2020, 750 who underwent preoperative spirometry were included. Restrictive ventilatory impairment was defined as a vital capacity of the predicted value (%VC) < 80%, while obstructive ventilatory impairment was defined as forced expiratory volume in one second (FEV1%) < 70%. Postoperative complications were assessed using the Clavien–Dindo (CD) classification. The relationship between clinical factors, including %VC, FEV1%, severe postoperative complications (CD ≥ 3b), overall survival (OS), and relapse-free survival, were assessed.
Results
The mean age of the 750 patients was 68 ± 10.5 years. Severe postoperative complications were observed in 25 (3.3%) patients and were significantly associated with FEV1% < 70% in the univariate analysis. The 5-year OS was 72.5%. Multivariate analysis showed that the cancer stage, age > 75 years, preoperative comorbidities, %VC < 80%, total gastrectomy, severe postoperative complications, and postoperative adjuvant chemotherapy were the significant independent factors affecting OS. Pneumonia was significantly associated with %VC < 80%.
Conclusions
FEV1% < 70%was associated with the development of severe postoperative complications, while %VC < 80% was associated with poor OS independent of the cancer stage because of death from pneumonia. Spirometry helps surgeons and patients discuss the risks and benefits of surgery.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Gastric cancer (GC) is the fifth-most common cancer and the third-most common cause of cancer-related deaths worldwide1. Curative resection is the gold standard treatment for non-metastatic GC. It is widely performed for older patients and those with multiple comorbidities; however, major concerns regarding its safety and efficacy exist in the elderly, and it is associated with increased mortality and morbidity, or decreased long-term survival.2,3,4,5,6,7,8,9
Preoperative pulmonary function tests have been performed for the risk assessment for thoracic surgery.10,11 Spirometry, the most readily available pulmonary function test, measures the total exhaled volume, or the forced vital capacity (FVC), the volume exhaled in the first second (FEV1) being the most important variable.12 Spirometry is used to predict postoperative pulmonary complication risks in high-risk situations, including chronic obstructive pulmonary disease or asthma, current smoking, and indicated surgery.13 However, it can promote coughing and aerosol generation, leading to the spread of coronavirus disease (COVID-19). Screening for active COVID-19 is challenging, particularly in patients with underlying respiratory symptoms. Furthermore, asymptomatic patients can shed the virus. The American Thoracic Society recommends limiting spirometry to patients whose results are essential for immediate management decisions14 Therefore, the clinical significance of spirometry requires reevaluation.
Aging is becoming a global public health concern, and the incidence of GC in the elderly seems to be rising owing to recent increases in life expectancy.15,16 Elderly patients are vulnerable to and affected by various physiological dysfunctions, with an age-dependent decline in pulmonary function. Older age is associated with high complication rates and severity after gastrectomy.17 Radical resection is feasible in selected elderly patients with resectable GC, whereas less invasive palliative resection may be beneficial for vulnerable patients. Therefore, the preoperative risk assessment and the estimation of the benefits of gastrectomy are important.
Few studies have investigated the relationship between preoperative pulmonary function and short- and long-term outcomes in patients who underwent abdominal surgeries for GC and colorectal cancer.18,19,20,21,22,23 This study aimed to test the hypothesis that preoperative pulmonary function is related to short- and long-term outcomes in patients who underwent gastrectomy for GC.
Materials and Methods
Patients
Of the 1040 patients with GC (stages I–III) who underwent R0 gastrectomy at our hospital between 2009 and 2020, those with R1/R2 resection (n = 50), stage IV (n = 4), and an unknown cancer stage (n = 8) were excluded. Patients who underwent surgical procedures other than distal or total gastrectomy (n = 3) or did not undergo preoperative spirometry (n = 225) were excluded. Finally, 750 patients who underwent preoperative spirometry were included in the study (Fig. 1). At the final follow-up in December 2020, the median follow-up period for the surviving patients was 62 (interquartile range [IQR]: 40–91) months. We investigated the relationship between clinical factors, including preoperative lung function and postoperative complications, and overall survival (OS) and relapse-free survival (RFS). Age was stratified to < 75 and ≧ 75 years according to the Japan Geriatrics Society24. This retrospective study was approved by the institutional review board of the Japanese Red Cross Aichi Medical Center Nagoya Daiichi Hospital (approval number: 2022–004).
Study Criteria
Preoperative lung function was assessed by spirometry using CHEST AC-8900 (CHEST, Tokyo, Japan). Restrictive ventilatory impairment was defined as a vital capacity percentage (%VC) < 80%, and obstructive ventilatory impairment was defined as a percent forced expiratory volume in one second (FEV1%) < 70%. Pathological staging was determined according to the 15th edition of the Japanese Classification of Gastric Carcinoma.25 The extent of gastric resection and lymph node dissection was determined in accordance with the Japanese Gastric Cancer Treatment Guidelines 2018.26 The reconstruction methods were determined by the surgeons who had performed the gastrectomy. Preoperative comorbidities were classified into 15 categories using the Charlson comorbidity index as a reference: myocardial, hypertension, cerebrovascular, vascular, pulmonary, dementia, neurologic, diabetes, endocrine, renal, gastroenterological, immune, musculoskeletal, hematological disease, and anemia.27 Postoperative complications were evaluated using the Clavien–Dindo (CD) classification.28 Severe postoperative complications were defined as CD ≥ 3b, considering the complications that required treatment by surgery, endoscopy, or interventional radiology under general anesthesia to be serious. Postoperative surveillance was performed in accordance with the Japanese Gastric Cancer Treatment Guidelines 2018.26 Blood tests and computed tomography were performed every 6 months, and upper gastrointestinal endoscopy was performed annually. S-1 monotherapy was administered as postoperative adjuvant chemotherapy for stage II/III GC based on the patients’ consent and general condition.26
Statistical Analysis
Continuous variables were expressed as mean ± standard deviation or median (IQR) and were compared using Student’s t-test, Mann–Whitney U test, or Kruskal–Wallis test, as appropriate. Categorical variables were compared using Pearson’s chi-square or Fisher’s exact test. Correlations between continuous variables were analyzed using Pearson’s correlation coefficient. Correlations between continuous and ordinal variables were analyzed using Spearman’s rank correlation test. The OS and RFS were calculated from the date of surgery to that of the last follow-up. A survival curve was drawn using the Kaplan–Meier method, and comparisons between the groups were performed using the log-rank test. Considering the statistical significance and clinical implications of the univariate analysis, variables were entered into multivariate analyses via logistic regression analysis or a Cox proportional hazards model. Statistical significance was set at p < 0.05. Statistical analyses were performed using the JMP software program (version 13.2.0) for Windows (SAS Institute Inc., Cary, NC, USA).
Results
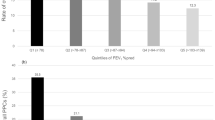
Table 1 shows the patients’ backgrounds. The average age of the patients was 68 ± 10.5 years. There were 494 males (65.9%) and 256 females (34.1%). Preoperative comorbidities were observed in 465 (62.0%) patients. Hypertension, diabetes, myocardial disease, anemia, and pulmonary disease were more frequently (Supplementary Table 1). The mean %VC and FEV1% were 106.1% ± 17.3% and 74.0% ± 13.0%, respectively. The number of patients who had %VC < 80% and FEV1% < 70% were 41 (5.5%) and 223 (29.7%), respectively. Diabetes, myocardial disease, anemia, and renal disease were significantly related to %VC < 80%, while vascular and neurologic diseases were significantly related to FEV1% < 70% (Supplementary Table 1).
The number of patients with postoperative complications classified as CD 1, 2, 3a, 3b, 4a, 4b, and 5 were 11, 77, 81, 12, 6, 1, and 6, respectively. Thirty-six severe postoperative complications (CD ≥ 3b) were observed in 25 patients (3.3%; median, 2; IQR, 1–4 in a patient; Table 2). Organ/space surgical site infection, pneumonia, intra-abdominal bleeding, and anastomotic passage disturbance were the most frequent complications. Univariate analysis of the clinical factors related to severe postoperative complications showed that age ≥ 75 years, FEV1% < 70%, serum albumin level < 3.5 g/dL, hemoglobin level < 10 g/dL, and platelet count < 15 × 104 μ/L were significant. Multivariate analysis revealed that FEV1% < 70% and serum albumin level < 3.5 g/dL approached, but were not statistically significant (p = 0.0833 and p = 0.0690, respectively; Table 3).
The correlation between FEV1% and %VC was weak and significant (γ = 0.171, p < 0.0001, Supplementary Fig. 1). Reduced FEV1% was associated with the frequent incidence of severe postoperative complications (< 70% vs. ≧ 70%, 12/223 (5.4%) vs. 13/527 (2.5%), p = 0.0421), whereas reduced %VC and severe postoperative complications showed no significant association (< 80% vs. ≧ 80%, 2/41 (4.9%) vs. 23/709 (3.2%), p = 0.6416).
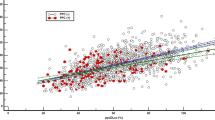
An investigation of the relationship between clinical factors and pulmonary function showed that %VC < 80% was significantly related to age ≧ 75 years, preoperative comorbidity, serum albumin < 3.5 g/dL, hemoglobin < 10 g/dL, neutrophil-to-lymphocyte ratio (NLR) ≧ 4, and less postoperative adjuvant chemotherapy, and FEV1% < 70% was significantly related to age ≧ 75, male, and preoperative comorbidity (Table 4). Figure 2a shows the correlation between age and %VC in terms of severe postoperative complications. The correlation was weak and significant (γ = 0.280, p < 0.0001). Severe postoperative complications were frequent in patients aged ≥ 75 years and with a %VC of 80–100%. Figure 2b shows the correlation between age and FEV1% in terms of severe postoperative complications. The correlation was weak and significant (γ = 0.206, p < 0.0001). Severe postoperative complications were frequent in patients aged ≥ 70 years and those with FEV1% < 85%. Pneumonia and anastomotic passage disturbance were more frequent in patients with FEV1% < 70% than in those with FEV1% ≥ 70% (3/223 [1.3%] vs. 2/527 [0.9%] and 3/223 [1.3%] vs. 1/527 [0.2%], respectively). The correlations between the number of comorbidities and %VC and FEV1% were weak and significant (Supplementary Fig. 2a and b).
a Correlation between age and %VC in terms of severe postoperative complication. The correlation was weak and significant. Severe postoperative complications were frequent in patients with age ≥ 75 years and %VC of 80–100%. b Correlation between age and FEV1% in terms of severe postoperative complications. The correlation was weak and significant. Severe postoperative complications were frequent in age ≥ 70 years and FEV1% < 85%
Postoperative adjuvant chemotherapy was administered to 220 (29.3%) patients. The 5-year OS was 74.9% during a median follow-up period of 61 months (IQR: 27–82 months). Table 5 shows the results of the univariate and multivariate analyses for OS. Univariate analysis showed that age ≥ 75 years, preoperative comorbidities, %VC < 80%, preoperative serum albumin level < 3.5 g/dL, hemoglobin level < 10 g/dL, and platelet count < 15 × 104 μ/L, neutrophil-to-lymphocyte ratio ≥ 4, laparotomy, total gastrectomy, the cancer stage, severe postoperative complications, and the presence of postoperative adjuvant chemotherapy were significantly associated with unfavorable OS. Multivariate analysis revealed that age ≥ 75 years, preoperative comorbidities, %VC < 80%, total gastrectomy, the cancer stage, severe postoperative complications, and the absence of postoperative adjuvant chemotherapy were significantly associated with unfavorable OS. The hazard ratio of OS in patients with %VC < 80% was 1.95 compared to that in patients with %VC ≥ 80%. Figure 3a–d show the OS curves of all patients and those with stages I–III disease according to the %VC. The OS of patients with %VC < 80% was consistently lower than those with %VC ≥ 80% in stages I–III. The number of comorbidities was the lowest in stage I (Supplementary Fig. 3).
During the follow-up period (median, 27 months, IQR: 15–45 months), 192 patients (25.6%) died. Table 6 shows the relationship between the cause of death and %VC. The incidence of death from non-malignant diseases and pneumonia was significantly higher in patients with %VC < 80% than in those with %VC ≥ 80%.
Relapse occurred in 160 (21.3%) patients during the follow-up period (median: 14 months, IQR: 7–26 months), and the 5-year RFS was 76.6%. Univariate and multivariate analyses of RFS showed that %VC < 80% or FEV1% < 70% were not associated with RFS (Supplementary Table 2).
Discussion
In this study, FEV1% < 70% was significantly correlated with severe postoperative complications. OS was consistently lower in patients with %VC < 80% than in those with %VC ≥ 80% in stages I–III due to frequent pneumonia-related deaths.
Univariate analysis revealed FEV1% < 70% as a significant factor associated with severe postoperative complications, whereas multivariate analysis revealed FEV1% < 70% as an independent factor that approached, but was not statistically significant. These results were partially supported by previous studies. Miki Y et al. showed that %VC < 80% or FEV1% < 70% was associated with postoperative pneumonia after gastrectomy.20 Jeong O et al. reported that FEV1/VC < 0.7 was associated with local and systemic complications after gastrectomy.18 Similar results were reported by a previous study investigating postoperative complications after colorectal resection.22 Reduced airway clearance may explain the frequent occurrence of pneumonia in patients with low FEV1%. Pneumonia and anastomotic passage disturbances were more frequent in patients with FEV1% < 70% than in those with FEV1% ≥ 70%, possibly because of hypoxia due to reduced airway clearance.29 Additionally, severe postoperative complications were more common in elderly patients and those with multiple comorbidities (Table 3 and Supplementary Fig. 2), suggesting an association between decreased FEV with older age and multiple comorbidities.
In this study, OS did not significantly differ between patients with FEV1% < 70% and ≥ 70%, consistent with a previous study. Sugawara K et al. reported that the OS of patients with stages I–III GC was similar to those with FEV1% < 70% and ≥ 70%.19 The relationship between low FEV1% and mortality is controversial in various populations.30,31,31 FEV1% is reportedly unrelated to muscle mass and muscularity,33,34 while Bellelli G et al. reported that decreasing FEV1% was a predictor of 3-year mortality in outpatients with bronchiectasis.35
In this study, %VC and FEV1% decreased with aging (Fig. 2a and b). In the elderly, the airspace expands, and the elastic contractile force of the lungs decreases without inflammatory changes or alveolar wall rupture. Moreover, the thorax stiffens, and the respiratory muscle strength decreases; therefore, FEV1%, %VC, and lung diffusivity decrease.36 Skeletal muscle loss correlates better with %VC < 80%.37 In our study, OS was lower in patients with %VC < 80% than in those with %VC ≥ 80% because of frequent deaths from non-malignant diseases (particularly pneumonia), whereas RFS was not different, consistent with previous studies.19,21 The incidence of %VC < 80% is reportedly 7–13% in various populations.32,38 %VC < 80% is associated with cardiovascular disease, diabetes, stroke, systemic inflammation, metabolic syndrome, deficits in the physical quality of life, and mortality.32,38,39,40,41 Accordingly, it can be used as a surrogate marker for decreased systemic function. Many patients with %VC < 80% are asymptomatic or have no interstitial lung disease. Therefore, %VC < 80% cannot be detected without a pulmonary function test. Spirometry is useful in predicting severe postoperative complications and long-term outcomes. These results can offer important information to surgeons and help patients make serious decisions before gastrectomy for GC.
In this study, RFS was slightly lower in patients aged ≥ 75 years than in those aged < 75 years (73.3% vs. 77.9%); however, OS was significantly lower in patients aged ≥ 75 years than in those aged < 75 years (62.9 vs. 79.3%, p < 0.0001), probably because of death from non-malignant disease. Several studies have assessed disease-specific survival rather than OS to compare the long-term outcomes of elderly patients who underwent gastrectomy for GC.2,5 We believe that OS is preferable for primary outcome of studies investigating surgical results to disease-specific survival or RFS, particularly in the elderly.
After selecting surgical candidates for gastrectomy, what would surgeons do for patients with impaired pulmonary function? Previous studies suggest several interventions including smoking cessation,42 control of respiratory infection, preoperative lung expansion interventions (incentive spirometry,43 deep breathing exercises,44 and continuous positive airway pressure45), preoperative total enteral or parenteral nutrition46 including immunonutrition,47 improvement of anemia,48 reducing the duration of surgery,49 epidural analgesia,50 use of nasogastric tubes for gastric decompression after surgery,51 and laparoscopic operations that can reduce postoperative pain and pulmonary compromise.52 In addition, preoperative rehabilitation (pre-rehabilitation) before upper abdominal surgery may help improve respiratory muscle strength.53,54,55 We identified a poor OS in patients who underwent total gastrectomy, which is consistent with previous studies.56,57,58 Total gastrectomy leads to a limited postoperative diet, dysphagia, dry mouth, and reflux symptoms, which affect the patient’s quality of life; therefore, total gastrectomy should be avoided if the proximal margin is negative.59 Considering that it is technically possible, “function-preserving” gastrectomy including proximal gastrectomy and subtotal gastrectomy may be considered.60 Less invasive palliative resection may be beneficial, especially for vulnerable patients.
This study has some limitations. First, the results might have been biased because of the retrospective and single-center design; therefore, the results could not be extrapolated to other institutions. The usefulness of spirometry should be determined in a well-designed randomized controlled trial. Second, we did not analyze the lung diffusing capacity, cardiopulmonary function tolerance, or blood gas, which might have helped precisely evaluate pulmonary function. However, %VC and FEV1% are widely used and are easily understandable respiratory parameters. Third, relatively few patients underwent laparoscopic gastrectomy (n = 153; 20.4%). The clinical significance of preoperative pulmonary function varies in the era of laparoscopic and robotic surgery. Further investigations are required in patients undergoing laparoscopic or robotic gastrectomy. Fourth, the multivariate analysis of clinical factors related to severe postoperative complications revealed no statistically significant independent factors because of the small number of patients with CD ≥ 3b. The outcome criteria for postoperative complications in previous studies include any complication, CD ≥ 2, CD ≥ 3a, infectious complications, and anastomotic leakage.61,62 We selected CD ≥ 3b (serious complications requiring interventions with general anesthesia) as an outcome criterion because it affects the preoperative treatment decisions. Additional analysis for factors related to postoperative complications (CD ≥ 3a) indicated significant associations with FEV1% < 70%, serum albumin < 3.5 g/dl, and total gastrectomy (Supplementary Table 3). Fifth, frailty (unintentional weight loss, exhaustion, low physical activity, slowness, and weakness), a widely recognized impaired physical response to stress and prevalence with increasing age, was not investigated as factors affecting short- and long-term patient outcomes.63,64 The definitions of frailty vary across studies, with no consensus in terms of preoperative physical function and short- and long-term postoperative outcomes.6,8,9,65 However, our results suggest that multiple comorbidities, %VC < 80% and FEV1% < 70%, are potential predictors of frailty. Further research based on a common definition of frailty in a larger cohort is required.
In conclusion, FEV1% < 70% is associated with severe postoperative complications (CD ≥ 3b), whereas %VC < 80% is associated with poor OS independent of the cancer stage because of death from non-malignant disease and pneumonia. Spirometry can provide important information and help surgeons discuss the risks and benefits of surgery with patients, perform perioperative management, and select a surgical procedure for patients undergoing gastrectomy for GC, particularly in patients aged ≥ 75 years and those with multiple comorbidities.
Data Availability
The data that support the findings of this study are available from the corresponding author (NY) upon reasonablerequest.
References
Bray F, Ferlay J, Soerjomataram I, et al (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68:394-424. https://doi.org/10.3322/caac.21492
Tran TB, Worhunsky DJ, Squires MH 3rd, et al (2015) Outcomes of gastric cancer resection in octogenarians: A multi-institutional study of the U.S. Gastric Cancer Collaborative. Ann Surg Oncol 22:4371-4379. https://doi.org/10.1245/s10434-015-4530-3
Kiyokawa T, Hiki N, Nunobe S, et al (2015) Feasibility of gastrectomy with standard lymphadenectomy for patients over 85 years old with gastric cancer. Ann Surg Oncol 22:3962-3969. https://doi.org/10.1245/s10434-015-4489-0
Endo S, Shimizu Y, Ikenaga M, Ohta K, Yamada T (2017) Survival benefit of gastrectomy for gastric cancer in patients ≥85 years old: A retrospective propensity score-matched analysis. Surgery 161:984-994. https://doi.org/10.1016/j.surg.2016.10.012
Yang JY, Lee HJ, Kim TH, et al (2017) Short- and long-term outcomes after gastrectomy in elderly gastric cancer patients. Ann Surg Oncol 24:469-477. https://doi.org/10.1245/s10434-016-5482-y
Lu J, Cao LL, Zheng CH, et al (2017) The preoperative frailty versus inflammation-based prognostic score: which is better as an objective predictor for gastric cancer patients 80 years and older? Ann Surg Oncol 24:754-762. https://doi.org/10.1245/s10434-016-5656-7
Mengardo V, Cormack OM, Weindelmayer J, et al (2018) Multicenter study of presentation, management, and postoperative and long-term outcomes of septegenerians and octogenerians undergoing gastrectomy for gastric cancer. Ann Surg Oncol 25:2374-2382. https://doi.org/10.1245/s10434-018-6543-1
Lu J, Zheng HL, Li P, et al (2018) High preoperative modified frailty index has a negative impact on short- and long-term outcomes of octogenarians with gastric cancer after laparoscopic gastrectomy. Surg Endosc 32:2193-2200. https://doi.org/10.1007/s00464-018-6085-4
Tanaka T, Suda K, Inaba K, et al (2019) Impact of frailty on postoperative outcomes for laparoscopic gastrectomy in patients older than 80 years. Ann Surg Oncol 26:4016-4026. https://doi.org/10.1245/s10434-019-07640-0
Osuka S, Hashimoto N, Sakamoto K, et al (2015) Risk stratification by the lower limit of normal of FEV1/FVC for postoperative outcomes in patients with COPD undergoing thoracic surgery. Respir Investig 53:117-123. https://doi.org/10.1016/j.resinv.2015.01.005
Smetana GW, Lawrence VA, Cornell JE, American College of Physicians (2006) Preoperative pulmonary risk stratification for noncardiothoracic surgery: systematic review for the American College of Physicians. Ann Intern Med 144:581-595. https://doi.org/10.7326/0003-4819-144-8-200604180-00009
Culver BH, Graham BL, Coates AL, et al (2017) Recommendations for a standardized pulmonary function report. An official American Thoracic Society technical statement. Am J Respir Crit Care Med 196:1463-1472. https://doi.org/10.1164/rccm.201710-1981ST
Zibrak JD, O’Donnell CR, Marton K (1990) Indications for pulmonary function testing. Ann Intern Med 112:763-771. https://doi.org/10.7326/0003-4819-112-10-763
Pulmonary Function Laboratories Advice regarding COVID-19. https://www.thoracic.org/professionals/clinical-resources/disease-related-resources/pulmonary-function-laboratories.php Accessed Jul 24, 2022
Chang AY, Skirbekk VF, Tyrovolas S, Kassebaum NJ, Dieleman JL (2019) Measuring population ageing: an analysis of the global burden of disease study 2017. Lancet Public Health 4:e159-e167. https://doi.org/10.1016/S2468-2667(19)30019-2
Katanoda K, Matsuda T, Matsuda A, et al (2013) An updated report of the trends in cancer incidence and mortality in Japan. Jpn J Clin Oncol 43:492-507. https://doi.org/10.1093/jjco/hyt038
Hamilton TD, Mahar AL, Haas B, et al (2018) The impact of advanced age on short-term outcomes following gastric cancer resection: an ACS-NSQIP analysis. Gastric Cancer 21:710-719. https://doi.org/10.1007/s10120-017-0786-6
Jeong O, Ryu SY, ParkYK (2013) The value of preoperative lung spirometry test for predicting the operative risk in patients undergoing gastric cancer surgery. J Korean Surg Soc. 84(1):18-26. https://doi.org/10.4174/jkss.2013.84.1.18. Epub 2012 Dec 26.
Sugawara K, Yamashita H, Yajima S, et al (2020) Preoperative restrictive pulmonary dysfunction influences the survival after gastrectomy for elderly patients with gastric carcinoma. Surg Today 50:1065-1073. https://doi.org/10.1007/s00595-020-01983-y
Miki Y, Makuuchi R, Tokunaga M, et al (2016) Risk factors for postoperative pneumonia after gastrectomy for gastric cancer. Surg Today 46:552-556. https://doi.org/10.1007/s00595-015-1201-8
Feng F, Tian Y, Zang Y, et al (2017) Low forced vital capacity predicts poor prognosis in gastric cancer patients. Oncotarget 8:28897–28905. https://doi.org/10.18632/oncotarget.15953
Wei J, Zhang Y, Yu P, et al. (2021) Maximal voluntary ventilation and forced vital capacity of pulmonary function are independent prognostic factors in colorectal cancer patients: A retrospective study of 2323 cases in a single-center of China, Medicine (Baltimore). 100(20):e25793. https://doi.org/10.1097/MD.0000000000025793.
Cheng WJ, Chiang CC, Peng MT, et al. (2021) Chronic Obstructive Pulmonary Disease Increases the Risk of Mortality among Patients with Colorectal Cancer: A Nationwide Population-Based Retrospective Cohort Study. Int J Environ Res Public Health. 2021 Aug 19;18(16):8742. https://doi.org/10.3390/ijerph18168742.
Ouchi Y, Rakugi H, Arai H, et al. (2017) Redefining the elderly as aged 75 years and older: Proposal from the Joint Committee of Japan Gerontological Society and the Japan Geriatrics Society. Geriatr Gerontol Int. 17(7):1045-1047. https://doi.org/10.1111/ggi.13118.
Japanese Gastric Cancer Association (2017). (in Japanese) Japanese classification of gastric carcinoma, 15th edn. Kanehara, Tokyo
Japanese Gastric Cancer Association (2021) Japanese gastric cancer treatment guidelines 2018 (5th edition). Gastric Cancer, 5th edn. 24:1–21. https://doi.org/10.1007/s10120-020-01042-y
Charlson ME, Pompei P, Ales KL, MacKenzie CR (1987) A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 40:373-383. https://doi.org/10.1016/0021-9681(87)90171-8
Dindo D, Demartines N, Clavien PA (2004) Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240:205-213. https://doi.org/10.1097/01.sla.0000133083.54934.ae
Jacobi CA, Zieren HU, Zieren J, Müller JM (1998) Is tissue oxygen tension during esophagectomy a predictor of esophagogastric anastomotic healing? J Surg Res 74:161-164. https://doi.org/10.1006/jsre.1997.5239
Beaty TH, Cohen BH, Newill CA, et al (1982) Impaired pulmonary function as a risk factor for mortality. Am J Epidemiol 116:102-113. https://doi.org/10.1093/oxfordjournals.aje.a113385
Peto R, Speizer FE, Cochrane AL, et al (1983) The relevance in adults of air-flow obstruction, but not of mucus hypersecretion, to mortality from chronic lung disease. Results from 20 years of prospective observation. Am Rev Respir Dis 128:491-500. https://doi.org/10.1164/arrd.1983.128.3.491
Burney PGJ, Hooper R (2011) Forced vital capacity, airway obstruction and survival in a general population sample from the USA. Thorax 66:49-54. https://doi.org/10.1136/thx.2010.147041
Shirai H, Kaido T, Hamaguchi Y, et al (2018) Preoperative low muscle mass and low muscle quality negatively impact on pulmonary function in patients undergoing hepatectomy for hepatocellular carcinoma. Liver Cancer 7:76-89. https://doi.org/10.1159/000484487
Jeon YK, Shin MJ, Kim MH, et al (2015) Low pulmonary function is related with a high risk of sarcopenia in community-dwelling older adults: the Korea National Health and Nutrition Examination Survey (KNHANES) 2008–2011. Osteoporos Int 26:2423-2429. https://doi.org/10.1007/s00198-015-3152-8
Bellelli G, Chalmers JD, Sotgiu G, et al (2016) Characterization of bronchiectasis in the elderly. Respir Med 119:13-19. https://doi.org/10.1016/j.rmed.2016.08.008
Yamaguchi Y (2014) Respiratory and pulmonary functions in the elderly. J Japan Broncho-esphageal Soc 65:395–402. (in Japanese with English abstract). https://doi.org/10.2468/jbes.65.395
Park CH, Yi Y, Do JG, Lee YT, Yoon KJ (2018) Relationship between skeletal muscle mass and lung function in Korean adults without clinically apparent lung disease. Med (Baltim) 97:e12281. https://doi.org/10.1097/MD.0000000000012281
Guerra S, Sherrill DL, Venker C, et al (2010) Morbidity and mortality associated with the restrictive spirometric pattern: A longitudinal study. Thorax 65:499-504. https://doi.org/10.1136/thx.2009.126052
Godfrey MS, Jankowich MD (2016) The vital capacity is vital: Epidemiology and clinical significance of the restrictive spirometry pattern. Chest 149:238-251. https://doi.org/10.1378/chest.15-1045
Guerra S, Carsin AE, Keidel D, et al (2017) Health-related quality of life and risk factors associated with spirometric restriction. Eur Respir J 49:1602096. https://doi.org/10.1183/13993003.02096-2016
Sharma G, Goodwin J (2006) Effect of aging on respiratory system physiology and immunology. Clin Interv Aging 1:253-260. https://doi.org/10.2147/ciia.2006.1.3.253
Møller AM, Villebro N, Pedersen T, et al. Effect of preoperative smoking intervention on postoperative complications: a randomised clinical trial. Lancet. 359:114–7. [PMID: 11809253]
Hall JC, Tarala R, Harris J, et al. (1991) Incentive spirometry versus routine chest physiotherapy for prevention of pulmonary complications after abdominal surgery. Lancet. 1991;337:953-6. [PMID: 1678039]
Fagevik Olse´n M, Hahn I, Nordgren S, et al. (1997) Randomized controlled trial of prophylactic chest physiotherapy in major abdominal surgery. Br J Surg. 84:1535-8. [PMID: 9393272]
Bo¨hner H, Kindgen-Milles D, Grust A, et a. (2002) Prophylactic nasal continuous positive airway pressure after major vascular surgery: results of a prospective randomized trial. Langenbecks Arch Surg. 387:21-6. [PMID: 11981680]
Moore FA, Feliciano DV, Andrassy RJ, et al. (1992) Early enteral feeding, compared with parenteral, reduces postoperative septic complications. The results of a meta-analysis. Ann Surg 216:172-83. [PMID: 1386982]
Gianotti L, Braga M, Nespoli L, et al. (2002) Randomized controlled trial of preoperative oral supplementation with a specialized diet in patients with gastrointestinal cancer. Gastroenterology. 122:1763-70. [PMID: 12055582]
Fowler AJ, Ahmad T, Phull MK, et al. (2015) Meta-analysis of the association between preoperative anaemia and mortality after surgery. Br J Surg. 102(11):1314-24. https://doi.org/10.1002/bjs.9861.
Gastmeier P, Sohr D, Breier A, et al. (2011) Prolonged duration of operation: an indicator of complicated surgery or of surgical (mis)management? Infection. 39(3):211-5. https://doi.org/10.1007/s15010-011-0112-x.
Park WY, Thompson JS, Lee KK. (2001) Effect of epidural anesthesia and analgesia on perioperative outcome: a randomized, controlled Veterans Affairs cooperative study. Ann Surg. 234:560–9; discussion 569–71. [PMID: 11573049]
Nelson R, Tse B, Edwards S. (2005) Systematic review of prophylactic nasogastric decompression after abdominal operations. Br J Surg 92:673-80. [PMID: 15912492]
Honda M, Kumamaru H, Etoh T, et al (2019) Surgical risk and benefits of laparoscopic surgery for elderly patients with gastric cancer: a multicenter prospective cohort study. Gastric Cancer 22:845-852. https://doi.org/10.1007/s10120-018-0898-7
Yamamoto K, Nagatsuma Y, Fukuda Y, et al (2017) Effectiveness of a preoperative exercise and nutritional support program for elderly sarcopenic patients with gastric cancer. Gastric Cancer 20:913-918. https://doi.org/10.1007/s10120-016-0683-4
Valkenet K, Trappenburg JCA, Ruurda JP, et al (2018) Multicentre randomized clinical trial of inspiratory muscle training versus usual care before surgery for oesophageal cancer. Br J Surg 105:502-511. https://doi.org/10.1002/bjs.10803
Jianjun W, Xing W, Guozhong Y, Chuming Z, Jiang Y (2019) Application of exercised-based pre-rehabilitation in perioperative period of patients with gastric cancer. Open Med (Wars) 14:875-882. https://doi.org/10.1515/med-2019-0103
Martin AN, Das D, Turrentine FE, et al (2016) Morbidity and mortality after gastrectomy: identification of modifiable risk factors. J Gastrointest Surg 20:1554-1564. https://doi.org/10.1007/s11605-016-3195-y
Sugoor P, Shah S, Dusane R, et al (2016) Proximal gastrectomy versus total gastrectomy for proximal third gastric cancer: total gastrectomy is not always necessary. Langenbecks Arch Surg 401:687-697. https://doi.org/10.1007/s00423-016-1422-3
Liu Z, Feng F, Guo M, et al (2017) Distal gastrectomy versus total gastrectomy for distal gastric cancer. Med (Baltim) 96:e6003. https://doi.org/10.1097/MD.0000000000006003
Li Z, Bai B, Xie F, Zhao Q (2018) Distal versus total gastrectomy for middle and lower-third gastric cancer: A systematic review and meta-analysis. Int J Surg 53:163-170. https://doi.org/10.1016/j.ijsu.2018.03.047
Kosuga T, Tsujiura M, Nakashima S, Masuyama M, Otsuji E (2021) Current status of function-preserving gastrectomy for gastric cancer. Ann Gastroenterol Surg 5:278-286. https://doi.org/10.1002/ags3.12430
Fujiya K, Tokunaga M, Mori K, et al (2016) Long-term survival in patients with postoperative intra-abdominal infectious complications after curative gastrectomy for gastric cancer: A propensity score matching analysis. Ann Surg Oncol 23:809-816. https://doi.org/10.1245/s10434-016-5577-5
Wang S, Xu L, Wang Q, et al (2019) Postoperative complications and prognosis after radical gastrectomy for gastric cancer: a systematic review and meta-analysis of observational studies. World J Surg Oncol 17:52. https://doi.org/10.1186/s12957-019-1593-9
Rockwood K, Stadnyk K, MacKnight C, et al (1999) A brief clinical instrument to classify frailty in elderly people. Lancet 353:205-206. https://doi.org/10.1016/S0140-6736(98)04402-X
Fried LP, Tangen CM, Walston J, et al (2001) Frailty in older adults: evidence for a phenotype. J Gerontol A Biol Sci Med Sci 56:M146-M156. https://doi.org/10.1093/gerona/56.3.M146
Meng Y, Zhao P, Yong R (2021) Modified frailty index independently predicts postoperative pulmonary infection in elderly patients undergoing radical gastrectomy for gastric cancer. Cancer Manag Res 13:9117-9126. https://doi.org/10.2147/CMAR.S336023
Funding
This work is supported by Japanese Red Cross Nagoya First Hospital Research Grant (NFRCH 22-0012). The funders had no role in study design, data collection or analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
AS wrote the manuscript, analyzed the data, and received funding. HM, HN, and YY prepared the materials and collected the data. NY contributed to the study conception and design and analyzed the data. The first draft of the manuscript was written by AS, and all authors commented on the previous versions of the manuscript. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval
All procedures followed were in accordance with the ethical standards of human experimentation and the Helsinki Declaration. This study was approved by our institutional ethics committee.
Informed Consent
Informed consent was waived owing to the retrospective nature of this study (2022–004).
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
11605_2023_5582_MOESM2_ESM.pptx
Supplementary file2 Fig. 1 Correlation between FEV1% and %VC in terms of severe postoperative complications (Clavien–Dindo classification ≥ 3b). The correlation between FEV1% and %VC was weak and significant. Reduced FEV1% was associated with a high frequency of severe postoperative complications, while reduced %VC and severe postoperative complications showed no correlation. Fig. 2 a Correlation between the number of comorbidities and %VC. A weak but significant correlation was observed. b Correlation between the number of comorbidities and FEV1%. A weak but significant correlation was observed. Fig. 3 The number of comorbidities according to the stage of gastric cancer. The number of comorbidities was lowest in stage I (p = 0.0192) (PPTX 179 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sekimoto, A., Miyake, H., Nagai, H. et al. Significance of Preoperative Pulmonary Function on Short- and Long-Term Outcomes Following Gastrectomy for Gastric Cancer. J Gastrointest Surg 27, 866–877 (2023). https://doi.org/10.1007/s11605-023-05582-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-023-05582-3